Abstract
Cortical brain damage was produced in rats by a focal pulse from a Nd-YAG laser, and evolution of the lesion was evaluated at 30 min, and 2, 8, and 24 h with respect to microvascular perfusion, blood–brain barrier (BBB) permeability, and expression of both the heat-shock/stress protein, hsp72, and the c-fos proto-oncogene transcription factor. A double-labeling fluorescence technique employing intravenously injected Evans blue albumin (EBA) and fluorescein-labeled dextran was used to map and measure BBB damage and microvascular perfusion in fresh frozen brain sections. Hsp72 and c-fos mRNAs were localized by in situ hybridization, and the respective proteins were identified by immunocytochemistry. Parallel sections were stained for glial fibrillary acidic protein and for routine histologic examination. Striking hsp72 mRNA expression was evident by 2 h in an ∼300 μm wide rim surrounding an area of expanding BBB damage. Increased hsp72 mRNA was observed only in regions of preserved microcirculation, where the hsp72 protein was subsequently localized exclusively in the vasculature at 24 h after the insult. Hsp72-positive endothelial cells spanned the narrow margin between the lesion and histologically normal, glial fibrillary acidic protein (GFAP)-positive cortical tissue. There was no hsp72 expression in the area of subcortically migrating edema fluid. Inductions of c-fos mRNA and Fos protein were not strikingly evident around the focal brain lesion, but were observed transiently throughout the injured hemisphere at 30 min and 2.5 h, respectively, indicating that spreading depression was triggered by the focal injury. These results are in striking contrast to those previously obtained from studies of models of focal ischemic or traumatic brain injury, which are characterized by a complex pattern of glial and neuronal hsp72 expression in the periphery of an infarct, and which suggest that the tightly demarcated lesion produced by the Nd-YAG laser lacks these components of graded injury that are evident following other types of focal brain damage.
Cerebral injury caused by thermal extremes, or by mechanical or ischemic mechanisms, results in a striking reprogramming of gene expression throughout the brain. Global ischemia gives rise to a predominantly neuronal induction of the 70 kDa heat shock/stress protei, hsp72 (Vass et al., 1988; Nowak, 1991; Simon et al., 1991), in keeping with neuronal vulnerability to injury in such models. There is also parallel neuronal expression of the proto-oncogene c-fos and related transcription factors after global ischemia (Jörgensen et al., 1989; Nowak et al., 1990b; Wessel et al., 1991, Kiessling et al., 1993). Hyperthermie challenges result in more pronounced glial and vascular expression of heat shock proteins and mRNAs, although neuronal responses have also been documented (Sprang and Brown, 1987; Blake et al., 1990; Marini et al., 1990; Pardue et al., 1992). There is a prominent induction of the stress response in the area of focal traumatic or ischemic lesions (Brown et al., 1989; Kawagoe et al., 1992; Li et al., 1992; Welsh et al., 1992; Kinouchi et al., 1993; Tanno et al., 1993), with potential involvement of multiple cell types. Hsp72 is expressed in neurons adjacent to a necrotic focus, but is only detected in the surviving vasculature within such lesions, while hsp72-positive glia are evident at an infarct rim (Gonzalez et al., 1989; Sharp et al., 1991; Li et al., 1992; Kinouchi et al., 1993). Focal insults also result in the expression of c-fos and related mRNAs and proteins, not only adjacent to the lesion but also in remote regions of ipsilateral cortex (Dragunow and Robertson, 1988; Uemura et al., 1991; Gass et al., 1992; Welsh et al., 1992; An et al., 1993), presumably as result of propagated depolarizations elicited by local tissue damage (Herrera and Robertson 1990).
In the present study, we examined such responses to focal cortical injury in a reproducible, recently-validated model of laser-induced progressive brain damage (Frerichs et al., 1990; Lindsberg et al., 1991). In view of growing evidence that hsp72 expression constitutes a valid marker for potentially injurious stress (Sloviter and Lowenstein, 1992), we used in situ hybridization and immunocytochemistry to map the distribution, time course, and cell type specificity of hsp72 expression as indices of injury in this model. We examined c-fos expression to further characterize both local and remote responses to the focal injury. Changes in gene expression were correlated with vascular parameters including blood–brain barrier damage, edema fluid migration, and microcirculatory perfusion in order to examine the possible relationships of these changes to cellular pathophysiology in the periphery of cortical lesions.
MATERIALS AND METHODS
Surgical protocol and dye infusion
Twenty-five male Sprague-Dawley rats (300–500g) were purchased from Taconic Farms (Germantown, NY, U.S.A.) and housed at 22°C with a 12/12 h light/dark cycle. Rats were anesthetized by inhalation of halothane and oxygen/nitrous oxide and placed in a stereotactic device (David Kopf Instruments, Tujunga, CA, U.S.A.). Using an operating microscope, a rectangular craniotomy (∼3 × 2 mm) was performed with a dental drill to expose the parietal lobe of the right hemisphere to enable laser lesioning. A similar craniotomy was performed over the left (control) hemisphere. For systemic infusion of fluorescent tracers, the left femoral vein was catheterized with PE-50 tubing, which was then tunneled s.c. through dorsal skin for chronic implantation. Rats were then subjected to the injury protocol (see below) and permitted to recover from anesthesia. The bone flap was repositioned and the scalp sutured. Rats were studied at different times after the injury as follows: 30 min (n = 8), and 2–2.5 (n = 10), 8 (n = 5), and 24 h (n = 2). A volume of 0.1 ml/100 g body wt of 0.9% saline solution containing 2% Evans blue (EB) (Sigma Chemicals St. Louis, MO, U.S.A.) and 1% albumin was infused i.v. over 1 min and allowed to circulate for 30 min before decapitation. Prior to killing, rats were reanesthetized and 15 mg/100 g body wt of fluorescein isothiocyanate (FITC)-dextran (Sigma) average molecular weight 71.2 kD, was injected in 0.5 ml of 0.9% saline over 5 s followed by another 0.5 ml of saline. These procedures have been shown to allow visualization of the network of perfused cerebral microvessels in the rat using fluorescence microscopy (Williams et al., 1993) and have been applied previously in the present injury model (Lindsberg et al., 1991b). At 30 s following FITC-dextran administration, animals were decapitated and the heads dropped into dry ice-cooled isopentane and stored at −70°C until analyzed.
Injury protocol
A focal cortical laser injury was produced following procedures described in detail elsewhere (Lindsberg et al., 1991a). Briefly, a neurosurgical neodymium:yttrium-aluminum-garnet (Nd-YAG) laser unit was used to generate a standardized energy pulse conveyed by a fixed fiberoptic cable onto the exposed brain surface. Laser was used at an incident power of 20 W and an exposure time of 1.0 s. The total energy delivered was, thus, 20 J. All other radiation parameters used were as previously described (Lindsberg et al., 1991a).
Scanning laser fluorescence microscopy
Frozen brains were removed at −20°C. Fresh-frozen 8 and 16 μm serial sections were cut through macroscopically determined epicenters of EB-stained laser-induced brain lesions and air dried on polylysine-coated slides. Regional assessment of fluorescence was performed in the 8 μm sections by delineating circumscribed areas of interest for fluorescence analysis. For this purpose, a computerized scanning argon laser-excited fluorescence microscopy system (ACAS 470, Meridian Instruments Inc., Okemos, MI, U.S.A.), capable of spectral splitting, spatial integration, quantitation, and recombination of the double-detected fluorescence emissions for two-dimensional imaging was used. Details of this equipment are described elsewhere (Schindler et al., 1987, 1989), and we have recently described its application to the investigation of plasma protein extravasation and density, and the filling of perfused microvessels with high spatial resolution (Lindsberg et al., 1991b). Briefly, fluorescence was excited by a focal 1 μm argon laser beam at 488 nm one spot at a time with the use of a computerized X-Y coordinated precision motorized microscope stage. Emissions were gathered with two separate photomultiplier tubes and the resulting photo-currents integrated and analyzed by a computer fitted with this system. The two-dimensional fluorescence map (image) was constructed by moving the tissue past the point of laser focus and relating the measured photo-currents to the coordinates of the motion. Splitting of emissions of the two fluorescent probes was accomplished by the use of a dichroic filter at 610 nm and two secondary isolation filters allowing passage at 500–560 nm for FITC and >630 nm for EB. In each section, fluorescence was excited, optically split, and analyzed simultaneously for both EB and FITC fluorescence using two different step sizes of 40–50 and 1.5 μm for the scanning laser beam, which enabled both low and high resolution imaging. The 40–50 μm step size was used for evaluation of lesion dimensions. High-resolution imaging was accomplished with step size 1.5 μm by scanning a grid of 5–25 adjacent standardized predetermined rectangular areas (180 × 165 μm) and used for quantitative image analysis of the microcirculation.
Image analysis
From the rectangular areas scanned perpendicular to the lateral cortical border of the lesion, those encompassing an abrupt five-to-10-fold increase in interstitial EB fluorescence above background levels (autofluorescence) were identified. From these and neighboring areas, fields located at 0–80, 80–240, 240–400, and 400–560 μm distances on both sides of the observed edge were analyzed on the image analyzer screen (Dasy Image Analysis System, Meridian Instruments Inc., Okemos, MI, U.S.A.). Values for fluorescence intensity were recorded for interstitial EBA. FITC-filled microvessels (<30 μm) were counted in each field and their peak fluorescence recorded individually and averaged for that field to indicate the filling status of the perfused micro vasculature. The homologous cortical region of the uninjured contralateral hemisphere was scanned for control measurements. Four 180 × 165 μm (0.0297 mm2) areas typically containing 8–13 perfused microvessels were analyzed for intensity of intravascular FITC-fluorescence and background EBA-fluorescence (arbitrary intensity units, see Fig. 1A) and density of perfused vessels (per mm2). Positions of analyzed areas with respect to an index mark carved on the slide edge were recorded by the computerized X-Y coordinated motorized microscope stage and stored in a data file. These location data were retrieved to reposition each slide after in situ hybridization and liquid emulsion autoradiography to correlate events in microcirculation with mRNA expression field by field. An inverted light microscope (IMT-2, Olympus Optical Co., Tokyo, Japan) attached to the image analyzer was used to examine the silver grain deposits localizing hsp72 mRNA (see below).
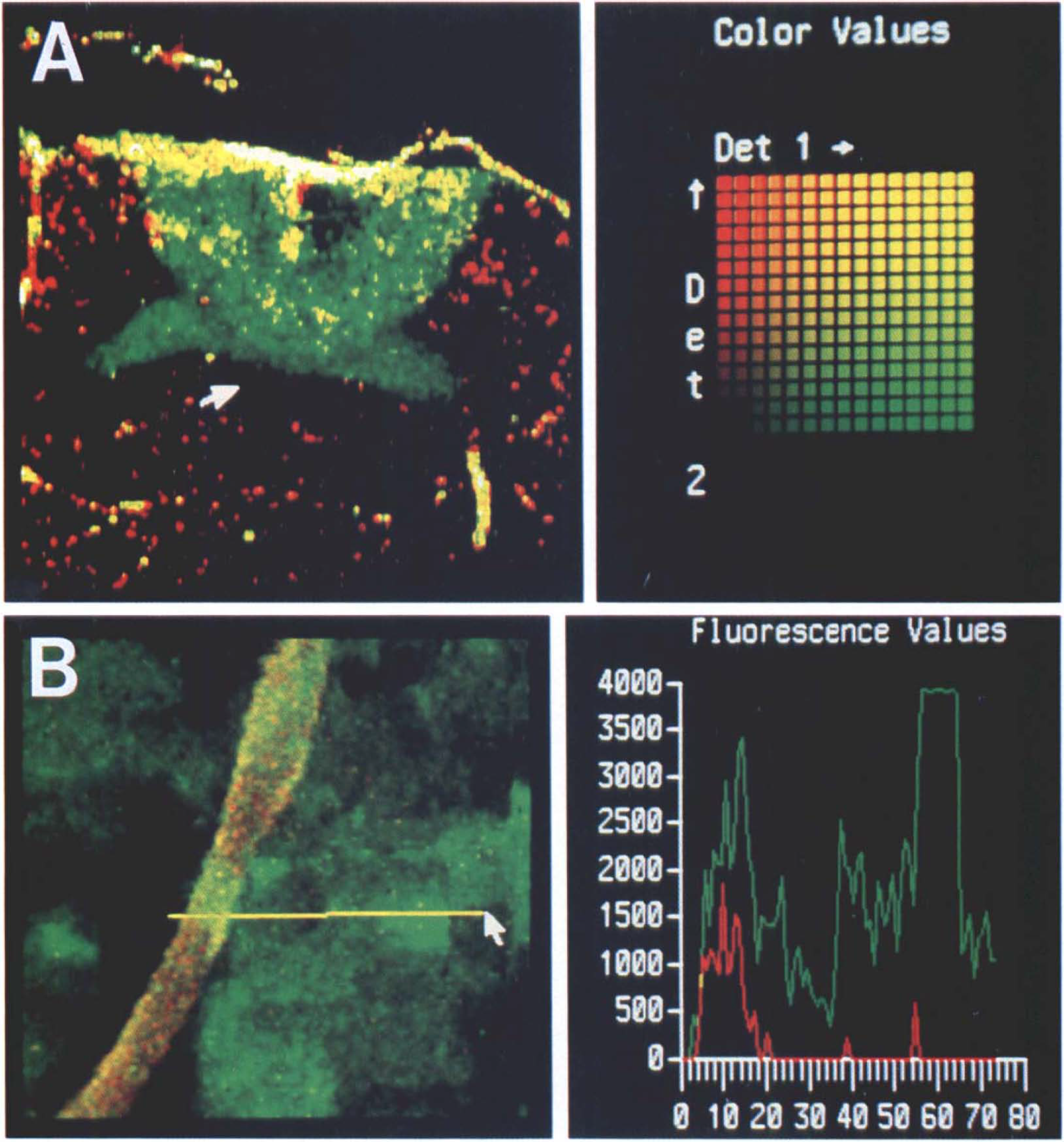
Measurement of microcirculation and BBB damage.
In situ hybridization of hsp72 and c-fos mRNA
Frozen 16 μm sections were stored dessicated at −70°C until use. Prior to hybridization, sections were fixed for 5 min in 4% paraformaldehyde in 0.1 M sodium phosphate buffer (pH 7.4) and rinsed twice in 70% ethanol followed by two rinses in 0.3 M NaCl, 0.03 M Na-citrate (2x SSC)-1x SSC-0.15M NaCl + 0.015M sodium citrate, pH 7.0). Sections were acetylated for 10 min with acetic anhydride (0.25% final concentration) added dropwise with stirring to slides immersed in 0.1 M triethanolamine (pH 8.0) followed by immersion for 30 min in 0.2 M Tris and 0.1 M glycine (pH 6.5). Slides were rinsed in 2x SSC, dehydrated through a graded ethanol series, extracted with chloroform for 5 min, and prewarmed in a humidified chamber. Hybridizations took place overnight at 37°C in a volume of 10 μl per section in a solution consisting of 2x SSC, 50% formaldehyde, 10% dextran sulfate, 1x Denhardt's solution (0.02% Ficoll, 0.02% polyvinylpyrrolidone, and 0.02% bovine serum albumin), 0.5 mg/ml salmon DNA, 0.25 mg/ml yeast tRNA, 100 mM dithiothreitol, and labeled probe (0.2–0.5 pmol/section). After hybridization, sections were washed with four changes of 2x SSC and 50% formamide at 42°C for 1 h each, rinsed in 2x SSC, and dehydrated through graded ethanols. Hybridized radioactivity was visualized by film autoradiography followed by liquid emulsion exposure (Kodak NTB2).
The probe used for hsp72 hybridization was an oligonucleotide (30 mer) with the sequence 5′-CGAT CTC CTT CAT CTT GGT CAG CAC CAT GG-3′ complementary to a conserved region of the coding sequence that can selectively hybridize to inducible RN As of the hsp72 family (Nowak et al., 1990a; Miller et al., 1991). The probe was 3′end-labeled with α[35S]dATP using terminal nucleotide transferase (Roychoudhury and Wu, 1980) to a specific activity of ∼5 × 106 dpm/pmol in a kit from Boehringer Mannheim, Indianapolis, IN, U.S.A. The probe used for c-fos hybridization was a commercially available cloned sequence (Miller et al., 1984) (Lofstrand Laboratories, Gaithersburg, MD, U.S.A.) labeled by a random primer method (Feinberg and Vogelstein, 1983) using kit reagents (Gibco BRL, Gaithersburg, MD, U.S.A.).
Immunocytochemistry
Additional animals were subjected to laser injury and perfused with 4% paraformaldehyde in 0.1 M Naphosphate buffer (pH 7.4) at intervals of 2.5 and 24 h after insult. Immunocytochemistry was performed as described previously (Vass et al., 1988). Briefly, sections were incubated for 1 h in 50 mM NH4Cl in phosphate-buffered saline (PBS), followed by 0.3% Triton X-100 in PBS. Blocking was accomplished with normal goat serum for several hours at room temperature; subsequent washes and antibody dilutions contained 10% normal goat serum in PBS. The Fos antibody used in these studies was an affinity purified rabbit polyclonal IgG preparation from Oncogene Science, Inc., Manhasset, NY, U.S.A. (Product PC05) raised against the N-terminal domain of Fos. The hsp72 antibody was a mouse monoclonal IgG (Amersham RPN. 1197), previously characterized (Vass et al., 1989). Glial fibrillary acidic protein (GFAP) was stained with a monoclonal antibody G 3893 (Sigma). Sections were incubated in 1/400 dilutions of anti-Fos or anti-GFAP, or 1/200 dilutions of anti-hsp72 overnight at 4°C. Mouse IgG against a nonrelevant rat antigen was used as a negative control (OX6, Sera-lab). After several washes, sections were incubated in 1/200 dilutions of biotinylated goat anti-rabbit or anti-mouse IgG (Kirkegaard and Perry Laboratories, Gaithersburg, MD, U.S.A.) for several hours at room temperature, followed by a series of washes. Endogenous peroxidase was blocked with 3% H2O2 and 10% methanol in PBS followed by extensive washes. Streptavidin-conjugated peroxidase (Kirkegaard and Perry Laboratories) was applied at a dilution of 1/200 for 1 h, after which sections were washed and bound peroxidase detected with 1 mg/ml diaminobenzidine and 0.015% H2O2 in 100 mM Tris-HCl (pH 7.6). Some sections were counterstained with hematoxylin-eosin (H & E) following immunohistochemistry.
Data presentation
Fluorescence intensity values for interstitial EBA and circulating FITC, and density of perfused vessels recorded from the scanned areas in the control hemisphere were averaged to obtain control values in each case. Data from the ipsilateral hemisphere were calculated as a percentage of the control. All data are presented as mean ± SEM.
RESULTS
The Nd-YAG laser pulse induced a highly localized intracortical hemispheroid lesion as described earlier (Frerichs et al., 1990; Lindsberg et al., 1991b), although the reduced power employed in the present study resulted in injury that was confined to cortex without involvement of the underlying hippocampus. A zone of bright EBA fluorescence indicated a sharp demarcation between the area of extravasation and surrounding tissue with an intact BBB (Fig. 1A). Intense extravasation was observed at all time points along the hemispheroidal centrifugally advancing lesion margins, but extensive edema fluid migration into the subcortical white matter tracts took place only at 2 h after injury. Simultaneous visualization of BBB damage and perfused microvessels with separate circulating fluoroprobes allowed high-resolution, quantitative measurement of ongoing BBB damage and perturbation of microvascular patency (Fig. 1B).
Striking hsp72 mRNA induction took place in a narrow zone surrounding the laser-induced brain lesions. While no hsp72 mRNA was observed at 30 min after injury, clear hsp72 induction occurred at 2 h and persisted at 8 h postinjury (Fig. 2). Hsp72 induction was not observed in distant cortical or subcortical areas or in the CA1 hippocampal sector below the site of injury. Furthermore, the hsp72 mRNA rim appeared to define the limits of the original BBB lesion and did not accompany the migration of extravasated EBA along the subcortical structures (Figs. 1A, 2B). Immunocytochemical studies confirmed that endothelial cells within the region of heavy hsp72 mRNA expression at 2–8 h were capable of message translation into proteins, since a corresponding circular rim of hsp72 positive vessels was observed in the lesion margin 24 h after injury (Fig. 3). No hsp72-positive neurons or glia were detected, and the zone of vascular hsp72 expression precisely spanned the transition between the lesion and histologically normal GFAP-positive tissue (not shown). At 30 min after injury (prior to pronounced hsp72 induction), c-fos mRNA expression was increased diffusely throughout the injured hemisphere, with no preferential induction either in the vicinity of BBB damage or in the lesion margin (not shown). The c-fos hybridization signal was not evident at later time points. Consistent with hybridization results, minimal Fos protein accumulation was observed in the lesion vicinity at either 2.5 or 24 h after insult. However, moderate Fos accumulation was detected throughout the ipsilateral, but not the contralateral, hemisphere at 2.5 h after injury, with the most prominent expression in the densely packed neurons in prepiriform cortex (Fig. 4).
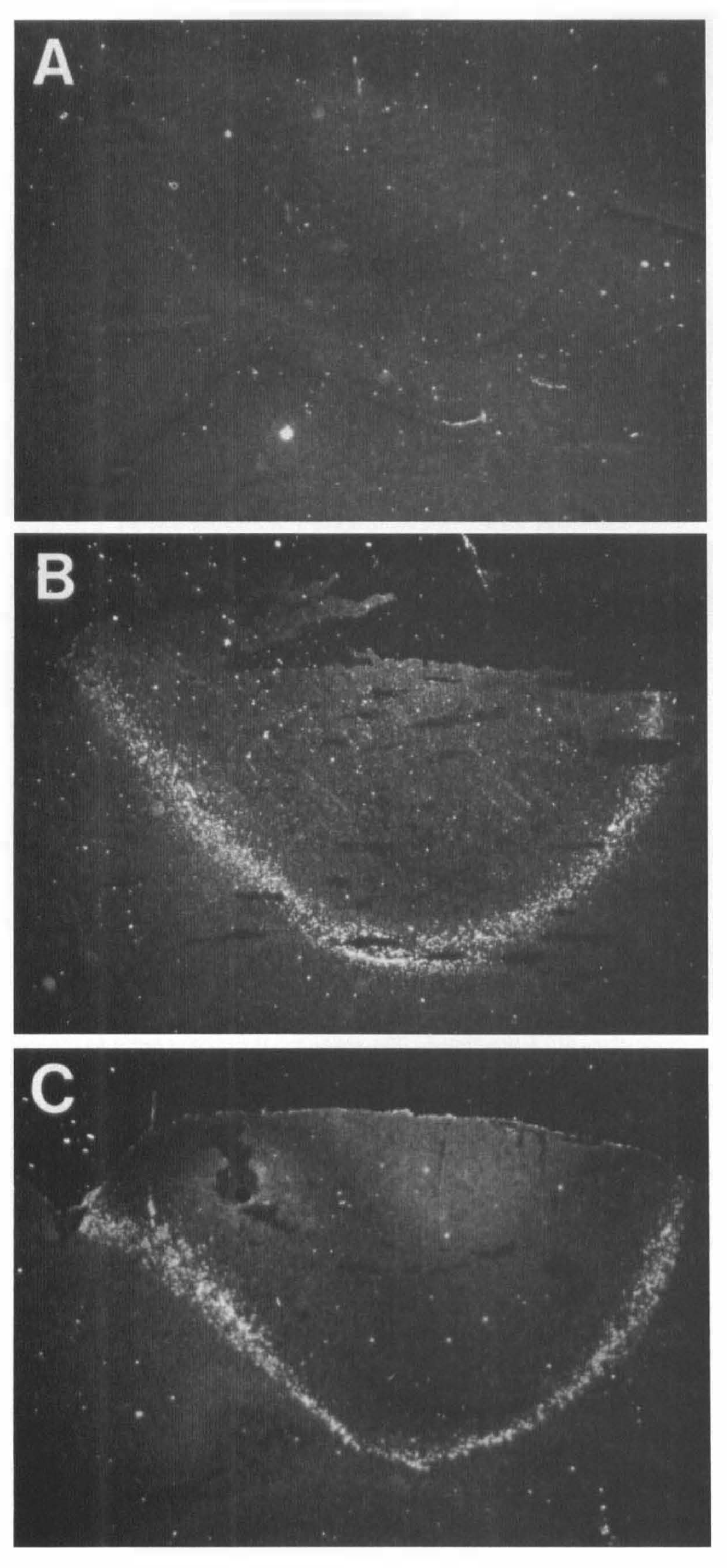
Hsp72 mRNA induction. Hsp72 hybridization Signal was visualized by liquid emulsion autoradiography and photographed under dark-field light microscopy. No significant hsp72 mRNA was evident at 30 min after injury

Immunocytochemistry of hsp72 at 24 h after injury, demonstrating that the stress response was exclusively localized in the microvasculature bordering the lesion.
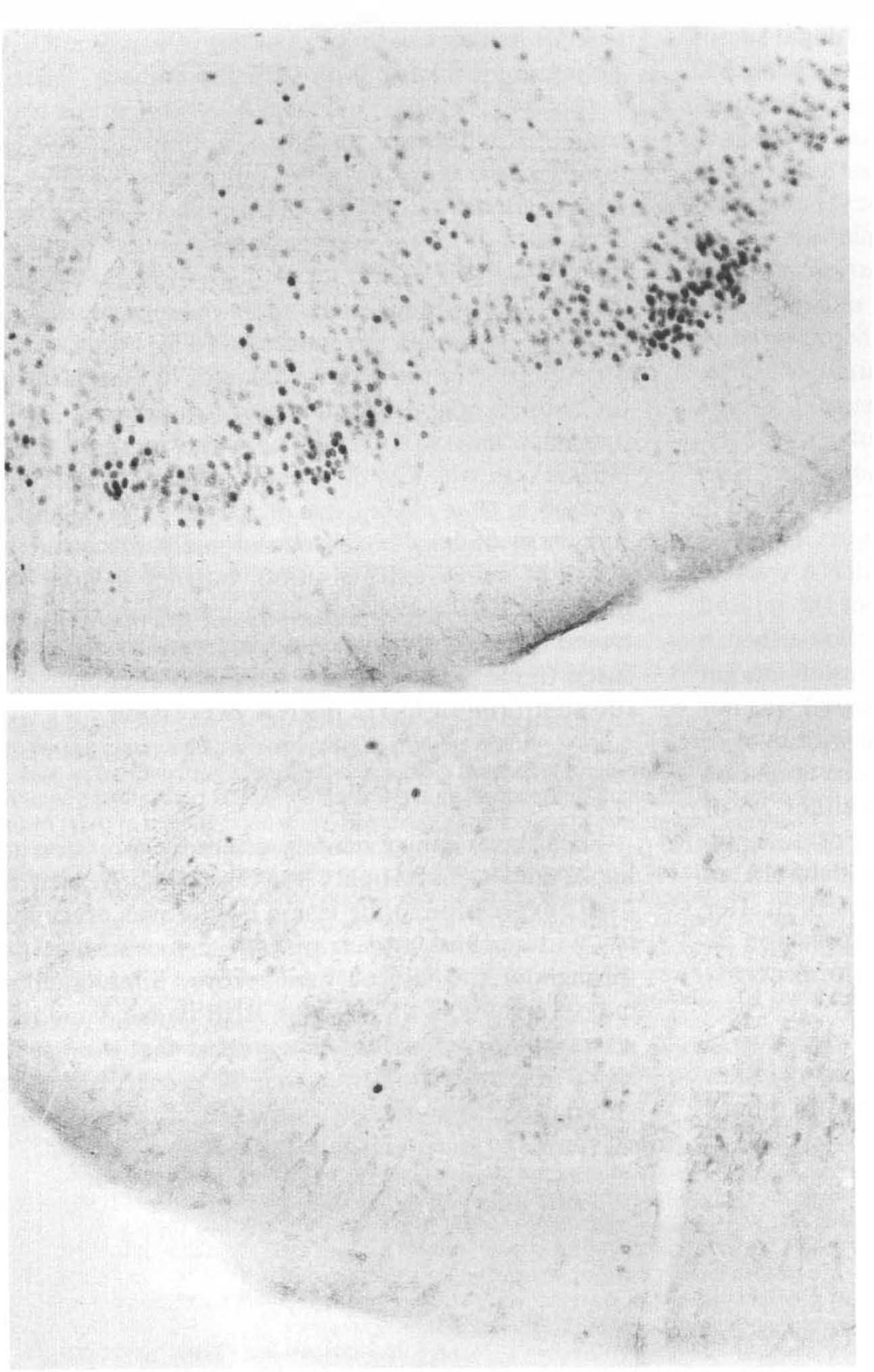
Immunocytochemistry of Fos protein in the prepiriform cortex 2.5 h after laser injury. Increased expression is evident in the ipsilateral hemisphere (
We determined the relationship between hsp72 induction and changes in microcirculatory parameters by comparing scanned images of the two fluorescent markers with localization of hsp72 mRNA. Topographic distribution of BBB-damage, microvascular occlusion, and hsp72 hybridization were examined with high spatial resolution 2 h after injury (Fig. 5). This time point represents the interval during which secondary permeability change and expansion of tissue damage were previously found to be underway at the lesion margin (Lindsberg et al., 1991a, b ). Indexes of microvascular perfusion (vessel density and intravascular FITC content) decreased toward the lesion epicenter in parallel with the number of grain clusters corresponding to hsp72 hybridization. Association with the margin of the BBB lesion was less clear cut, but hsp72 induction seemed to peak at the zone of arising extravasation. Induction of hsp72 was occasionally observed in zones of heavy extravasation, but not in areas of microcirculatory collapse. The transition zone between normally perfused and edematous underperfused tissue was ∼300 μm wide and encompassed the sharp rim of hsp72 mRNA expression.
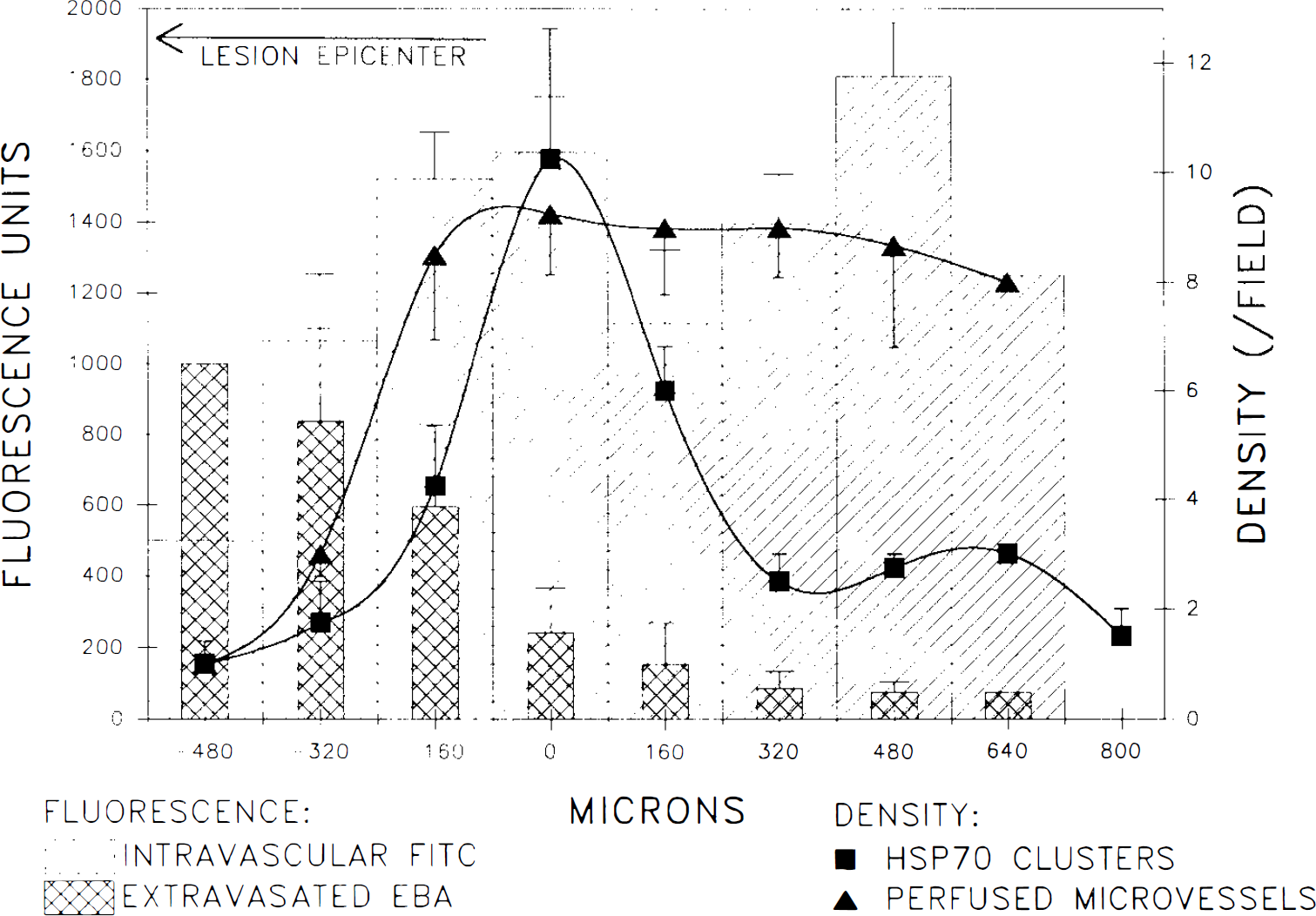
Quantitative analysis of hsp72 expression and microcirculation at the lesion border. Spatial distribution of hsp72 mRNA induction is compared with that of the measured EBA extravasation and indexes of microvascular patency and density of perfused microvessels (triangles) and their FITC content (hatched bars). Data are arranged according to the observed narrow peak in the distribution of silver grains corresponding to hsp72 hybridization, which is indicated as the 0 point on the x-axis. Each datum point represents averaged measurements in a single field from each of four animals at 2 h after injury.
DISCUSSION
Focal laser injury acutely altered expression of hsp72 and c-fos. Highly localized microvascular hsp72 expression at the lesion border was preceded by widespread and transient c-fos induction throughout the injured hemisphere. Simultaneous measurements of the ongoing BBB damage and microvascular perfusion demonstrated that the hsp72 signal was restricted to a zone of preserved blood circulation immediately adjacent to the lesion. Flooding of the extracellular milieu with extravasated plasma constituents was unrelated to the more restricted distribution of hsp72 expression. With the additional immunocytochemical observations, we conclude that hsp72 induction is a component of the injury response of viable microvasculature.
The most surprising observation in these studies was the absence of neuronal or glial hsp72 expression in the vicinity of the laser-induced lesion, since both are prominent components of focal vascular occlusion and trauma (Kinouchi et al., 1993; Tanno et al., 1993). This model of laser-induced focal brain injury has recently been validated by histopathological analysis (Lindsberg et al., 1991a) and entails a component of secondary brain damage, which involves some features of focal ischemia models. While the fixed laser energy pulse immediately photocoagulates the irradiated area, previous studies have demonstrated that elements of progressive brain damage in the surrounding region include extravasation of plasma constituents, edema, secondary hypoperfusion, and progressive neuronal death (Frerichs et al., 1990; Lindsberg et al., 1991a, Kiessling et al., 1990). Therapeutic intervention designed to preserve microcirculation was found to attenuate these components of secondary brain damage (Frerichs et al., 1990). In further studies with this model, progressive enlargement of the hemispheroid, cortical area of extravasated plasma protein was demonstrated (Lindsberg et al., 1991b). Thus, this model should allow observation of proposed self-propagatory processes underlying secondary microvascular brain damage surrounding focal, edematous brain lesions (Klatzo, 1985). Significant increases in lesion dimensions have been reported between 2 and 48 h after induction of such injuries (Kiessling et al., 1990), although, in our hands, most of the expansion has been noted by 2–8 h (Lindsberg et al., 1991 b). The present studies, however, suggest that this process is not associated with propagation of hsp72 expression to an area of histologically normal appearing neurons and glia. The rim of microvascular hsp72 expression observed in these studies appeared to be established within 2 h (Fig. 2). In a solely hyperthermic brain insult, hsp72 expression is predominantly glial and vascular, and peaks already by 1 h after injury at the mRNA level (Sprang and Brown, 1987; Marini et al., 1990, Nowak et al., 1990a). Hsp72 induction at the lesion border in the present studies appeared to occur somewhat more slowly after injury. This suggests that it may represent a later step in lesion maturation rather than a direct result of the thermal insult, although this delay may simply reflect a lag in the accumulation of detectable mRNA levels resulting from transient, local metabolic deficits following the intense heating produced by the laser injury.
The pattern of hsp72 response underscores the microvascular character of the damage produced in this model of focal injury and vasogenic edema as well as the relative resilience of the endothelium. While all cellular elements are rapidly lost within the injury focus, there appears to be little persistent stress to surviving neurons and glia in the lesion periphery outside the zone of vascular hsp72 expression, based on the absence of hsp72 staining in such cells (Sloviter and Lowenstein, 1992). This is consistent with a very steep gradient of thermal injury, with endothelial cells representing the only surviving elements at the margin of the lesion that retain the capacity for a successful stress response. The present results suggest that hsp72 expression in diverse cell types in other vascular occlusion models may be directly related to the more persistent graded ischemic insult to which tissue is necessarily subjected surrounding the core of such lesions (Jacewicz et al., 1992). Recent evidence suggests the potential importance of repeated intervals of metabolic stress in the region of repeated depolarization surrounding an ischemic core (Takeda et al., 1993). In contrast, quantitative autoradiographic studies have demonstrated an absence of persistent blood flow deficits outside the necrotic focus at intervals of ≥30 min after laser injury (Kiessling et al., 1990) and our own results demonstrate an abrupt step in microvascular filling at the border of the lesion (Fig. 5). Correlation of hsp72 expression with microcirculatory parameters has not been reported previously in other models of focal cerebral injury. Other investigations have employed either global ischemic stimuli or local trauma without simultaneous documentation of the vascular permeability and blood flow changes. Hsp72 induction has been observed in cortical regions suffering from only moderately ischemic blood flow reduction (<40 mg/ml/100 g) (Jacewicz et al., 1986). Conversely, an irreversibly damaged central infarct area in a model of combined middle cerebral artery (MCA) occlusion and bilateral carotid artery occlusion did not express significant hsp72 mRNA (Welsh et al., 1992). In the present investigation, quantitative analysis of microcirculation also demonstrated that hsp72 expression decreases sharply in parallel with the number of perfused microvessels toward the lesion epicenter. This result supports the suggestion that hsp72 is preferentially induced in viable yet injured cells (Nowak, 1991; Sloviter and Lowenstein, 1992). Further study is needed to clarify whether the hsp72 positive cells of the injured vasculature will ultimately survive.
The pattern of ipsilateral c-fos expression observed throughout cortex in the present study is similar to that seen in diverse models of focal injury, reflecting the consequences of propagated depolarization in response to local tissue damage (Herrera and Robertson, 1989, 1990; Dragunow and Robertson, 1988; Welsh et al., 1992). Several studies have described a component of the induction of c-fos and other immediate early genes in the region surrounding the lesion focus that is refractory to inhibition by the N-methyl-D-aspartate antagonist, MK-801 (Uemura et al., 1991; Gass et al., 1992), arguing that distinct mechanisms may account for local versus remote effects of the insult on expression of these genes. We have not examined the effect of MK-801 on expression of c-fos mRNA or Fos protein in this model and, therefore, cannot determine whether this component of expression is present after laser injury. In contrast to neuronal localization of diffuse hemispheric c-fos induction, a glial distribution of Fos protein expression has been observed surrounding a region of mechanical trauma (Dragunow and Robertson, 1988). The intensity of Fos expression in the present study was relatively modest, even in regions of high cell density such as prepiriform cortex (Fig. 4), and distinct Fos expression was evident in only a few cells in the lesion periphery. This may be consistent with the lack of neuronal and glial responses in the region noted above with respect to hsp72 expression.
Studies of focal ischemia produced by MCA occlusion have identified hsp72 or proto-oncogene induction in ipsilateral and, to some extent, contralateral hippocampus—regions that are not subject to overt damage in these models (Welsh et al., 1992; An et al., 1993; M. Jacewicz et al., unpublished data). Absence of evident changes in hippocampal gene expression in this study suggests that the transcriptional responses observed in this region in the MCA occlusion models may be secondary to effects of ischemia or trauma in ventrolateral cortical regions, including prepiriform/entorhinal cortex that project to hippocampus. However, under conditions similar to those of the present experiments, the laser pulse has been observed to elicit delayed damage in the hippocampal CA1 neurons underlying the lesion (Frerichs et al., 1990), suggesting that a more detailed evaluation of hippocampal responses in the present model may be warranted.
In summary, these studies demonstrate a striking microvascular stress response following focal cortical laser injury, consistent with the primary vascular damage well documented in this model and with the vascular localization of stress protein induction observed following other hyperthermic insults. Absence of detectable expression in neuronal or glial elements in the periphery of such lesions indicates that such cells outside the primary lesion remain relatively unaffected by the laser, while those in the transition zone of vascular hsp72 expression are so rapidly damaged that they are unable to mount an effective transcriptional/translational response. From these results, it is clear that the nature of cellular pathology in the border zone of Nd-YAG laser-induced lesions is distinct from that occurring in the penumbra of lesions produced in other vascular occlusion models. However, the present model of focal vasogenic edema surrounding a core of necrosis should provide a useful comparison for studies investigating the topography of gene expression following other types of focal brain injury.
Footnotes
Acknowledgment:
Dr. P. J. Lindsberg is supported by Paulo Foundation and Maire Taponen Foundation.
