Abstract
The functional consequences of increased capillary densities in the brain resulting from vascular endothelial growth factor (VEGF165) overexpression are unknown. Therefore, the authors measured local CBF using the iodo-[14C]antipyrine technique in transgenic mice expressing brain-specifically sixfold higher VEGF165 levels and in nontransgenic littermates. To reveal possible compensatory vasoconstriction, CBF was also measured during severe hypercapnia (PaCO2 > 130 mm Hg). Simultaneously, local capillary density, perfusion state, and blood–brain-barrier permeability were assessed. Using the 2-[14C]deoxyglucose method, metabolic effects of VEGF overexpression could be excluded. In transgenic mice all capillaries showed normal morphology and a tight blood–brain barrier. However, 3% nonperfused capillaries in some brain structures indicate ongoing angiogenesis. Capillary density was drastically increased in transgenic mice in white matter structures (70% to 185%), the dentate gyrus (143%), and caudate nucleus (86%). In all other brain structures investigated, capillary densities were moderately increased by approximately 20%. Normocapnic CBF did not differ between transgenic and nontransgenic mice. During maximal hypercapnic vasodilation, CBF was 20% to 30% higher in transgenic mice, although only in brain structures where capillary density was increased more than twofold. These findings suggest that attenuated CBF in transgenic mice during normocapnia is only partly due to a compensatory vasoconstriction, and that microvascular networks in transgenic brains might be ineffectively constructed.
Keywords
Because of oxygen depletion, anaerobic glycolysis in brain tissue surrounding cerebral infarcts (penumbra) is markedly increased (Cramer et al., 1997; Weiller et al., 1993). Hypoxia induces neoangiogenesis that can restore the oxygen supply to the penumbral tissue (Marti et al., 2000). Fast and efficient growth of new vessels may improve the survival of cells in the penumbra and therefore the functional outcome of patients after stroke. This idea is supported by postmortem analysis of brains from stroke patients indicating the correlation of survival with the density of newly formed blood vessels in the penumbra (Krupinski et al., 1994). The effectiveness of the physiologic response of new vessel formation in penumbral tissue could be enhanced by therapeutically induced angiogenesis, which might be especially beneficial in patients with a diminished hemodynamic reserve and consequential lower penumbral blood flow (Friberg and Olsen, 1991; Witt et al., 1994). However, newly formed vessels have to be integrated effectively into the preexisting network to enable an optimal tissue supply and to avoid areas of “luxury perfusion” next to areas of reduced perfusion (steal). This integration process, called remodeling, is driven by metabolic signals such as local hypoxia (Pfeifer et al., 1998) as well as mechanical forces of the flowing blood (Bongrazio et al., 2000; Pries et al., 2001). These stimuli are acting not only locally but are transferred upstream over the entire vascular network (Dora, 2001; Pries et al., 2001). Model calculations have shown that metabolic signals such as the local PO2 and mechanical forces such as shear stress are essential for the maintenance of the capillary network structure as well as for the adaptation to an altered metabolic situation or to occlusions of pathways within the network (Pries and Secomb, 2000). The process of remodeling appears to ensure an optimal tissue supply with a minimum of capillaries, each perfused over time to the same extent. From simultaneous measurements of local CBF, local glucose utilization and local capillary density in rat brain tissue it is known that in each brain structure blood flow as well as metabolic rate per single capillary is indeed the same, independent of the local capillary density (Klein et al., 1986; Kuschinsky, 1991). In addition, it has been shown that the density of glucose transporters per single capillary is similar in each brain structure (Duelli and Kuschinsky, 2001). These findings indicate a similar supply capacity of each capillary all over the brain whose establishment and maintenance might require precise regulation of vascular growth and regression. However the functional significance of the postulated parallel action of metabolic and mechanical signaling during remodeling (Pries and Secomb, 2000) is still unclear.
This question can be investigated with our newly developed transgenic mice that overexpress human vascular endothelial growth factor (VEGF165) in the brain. Assuming that additional capillaries in the brain of these mice do not increase the metabolic demand of the tissue, additional capillaries would not be necessary. The new capillaries might be formed correctly, but remodeling might be disturbed resulting in an ineffective perfusion. In the present study we measured local capillary densities and correlated them with local CBF measurements, allowing the estimation of perfusion in relation to single capillaries. To unmask possible compensations of a “luxury perfusion” by vasoconstriction, local CBF measurements were also performed during severe hypercapnia. In addition, local cerebral glucose utilization (CGU) was measured to elucidate metabolic effects of angiogenesis in our mice.
MATERIALS AND METHODS
Generation of transgenic animals
A full-length cDNA for human VEGF165 (provided by Dr. G. Breier, Bad Nauheim, Germany) was modified by replacing the 3′ untranslated region with a polyadenylation signal derived from the bovine growth hormone, and placed under the control of a 1.8-kb rat NSE-promotor fragment (Forss-Petter et al., 1990), resulting in brain-specific expression. The constructs were microinjected into fertilized oocytes of (C57BL/6 X C3H/He)F1 hybrid mice. Transgenic founders were identified by screening DNA samples from these litters. Founders were subsequently bred with C57BL/6 wild-type mice (Harlan, Borchen, Germany) to establish the transgenic line TgN(NSEVEGF)1651 (V1). Heterozygous V1 animals showed sixfold increased VEGF levels in the brain compared with nontransgenic littermates as determined by ELISA (Schoch et al., 2002). Homozygous animals were not viable. All experiments were performed in accordance with the German and Swiss animal protection laws and institutional guidelines.
In vivo experiments
Anesthesia was induced by a gas mixture containing 4% halothane, 70% N20, and remainder O2, and was maintained by reducing the halothane concentration to 1% to 1.5%. Body temperature was maintained at 37°C using a temperature-controlled heating pad. The animals were equipped with catheters inside the right femoral artery and vein to enable the infusion of iodo[14C]antipyrine (Biotrend, Cologne, Germany) for measurements of the local CBF, the injection of Evans blue (Sigma, Deisenhofen, Germany), measurement of the arterial input function of iodo[14C]antipyrine and 2-[14C]deoxyglucose, and measurement of blood acid-base status and glucose concentration. After surgery, the anesthesia was changed to intravenous etomidate (Etomidat Lipuro, B. Braun, Melsungen, Germany) in a dose of 0.025 mg/kg body wt/min to minimize the effects of anesthesia on CBF (Famewo and Odugbesan, 1978; Janssen et al., 1975). One hour later the animals were randomized to different experimental groups. In the first group CBF was measured during normocapnia, whereas in the second group hypercapnia was induced by allowing the mice to breathe a water vapor-saturated gas mixture containing 18% to 22% CO2, 22% O2, and 56% to 60% N2 for 15 minutes to obtain arterial PCO2 values > 135 mm Hg. Ten minutes before the decapitation in the CBF and CGU experiments, 0.2 mL of 4% Evans blue in saline was injected. CBF was measured according to the autoradiographic method of Sakurada et al. (1978) and the modifications of Jay et al. (1988). To this end 125 μCi/kg body weight iodo[14C]antipyrine dissolved in 200 μL saline was infused with an increasing infusion rate for 1 minute. Parallel to this, 12 to 16 timed arterial blood samples were taken for determination of the time course of the arterial iodo[14C]antipyrine concentration. At the end of the infusion period the animals were decapitated, and the brains were removed as quickly as possible and frozen in 2-methylbutane chilled to −60°C. In another experimental group local CGU was measured according to Sokoloff et al. (1977), and 125 μCi/kg body weight 2-[14C]deoxyglucose was infused during 20 seconds. Parallel and after this, arterial blood samples (20 μL) were collected 0.25, 0.5, 0.75, 1, 2, 4, 12, 20, 30, and 45 minutes after starting the infusion. After taking the last sample, the animals were decapitated, and the brain was dissected out and frozen in 2-methylbutane chilled to −60°C. The brains of all mice were embedded in M-1 embedding matrix (Lipshaw, Detroit, MI, U.S.A.) and cut into 20-μm coronal sections at −20°C in a cryomicrotome. After drying them on a heating plate at 60°C, the sections were exposed together with a [14C] standard set on a Kodak MinR1 x-ray film for 21 days. From the optical densities of the autoradiograms the local CBF or the local CGU respectively was calculated using an image analyzing system (MCID; Imaging Research Inc., St. Catherines, Ontario, Canada). For the calculation of CBF, a blood—brain partition coefficient of 0.7 was determined in our mice.
Assessment of capillary densities and perfusion state
Five-micrometer sections adjacent to those used for autoradiography were used for assessment of the Evans blue extravasation and the determination of the capillary density and perfusion state according to Gobel et al. (1990). To this end the sections were transferred to egg-white/glycerin-coated (1:1) −20°C precooled slides. The sections on the slides were immediately dehydrated in −20°C cold acetone for 2 minutes and then transferred to acetone at room temperature. Thereafter, they were observed and photographically documented using an incident fluorescent microscope (Zeiss, Deisenhofen, Germany). After rehydration in a humid chamber overnight, indirect immunofluorescence against fibronectin was performed as described previously (Gobel et al., 1990). Positions previously photographed were relocalized and again photographically documented using the incident fluorescent microscope. Capillary densities and perfusion state were determined using the image analyzer mentioned earlier.
Determination of brain water content and vascular permeability
To quantify brain water content in brains from transgenic and nontransgenic animals, brains were weighed immediately after removal as a wet weight. The samples were dried at 110°C for 24 hours and reweighed as a dry weight. The difference between wet and dry weight accounts for the in vivo water content. Vascular permeability of brain vessels was quantified as described (Schoch et al., 2002). Briefly, 200 μL of sodium-fluorescein (Sigma) at a concentration of 6 mg/mL in phosphate-buffered saline were injected through the tail vein. Sodium-fluorescein (molecular weight = 376) is a fluorescent tracer that does not cross an intact blood-brain barrier (Williams et al., 1984). Thirty minutes later, mice were anesthetized and then perfused with phosphate-buffered saline (20 mL) through the left heart ventricle to remove the fluorescent tracer from the vascular bed. Subsequently, both hemispheres were removed and frozen in liquid nitrogen. To assess fluorescence brain hemispheres were homogenized in 0.5-mol/L borate buffer (pH = 10), centrifuged (3,000g) for 15 minutes at 4°C, and the supernatant was added to 1.2 mL of ethanol to precipitate proteins. Probes were again centrifuged (13,000g) for 20 minutes at 4°C and fluorescence of the supernatant was measured at 485 nm at an excitation wavelength of 330 nm, using a Lambda Fluoro 320 Fluoroscope (MWG Biotech AG, Reinach, Switzerland) (Baba et al., 1988). Results are presented as relative fluorescence units (rfu) per mg of brain tissue.
Statistics
The data measured in the experimental groups were compared using a two-tailed t-test with Bonferroni correction. The level of statistical significance was set at P < 0.05.
RESULTS
Table 1 shows the physiologic variables measured in the different experimental groups. No significant differences were found between nontransgenic and transgenic mice within each experimental group. Statistically significant differences between the hypercapnic and the normocapnic and CGU groups are indicated in the table.
Physiologic variables of the different experimental groups

Data are presented as means±SD, n = 6 per group.
CGU, cerebral glucose utilization.
P < 0.01 vs. normocapnic CBF.
P < 0.01 vs. CGU.
P < 0.05 vs. normocapnic CBF.
P < 0.05 vs. CGU.
After Evans blue injection, all detectable dye spots, assigning perfused blood vessels, were sharply confined, showed no visible diffusion halo, and were comparable with the vessels found in nontransgenic mice (Fig. 1, right panels), indicative of an intact blood–brain barrier in transgenic mice. Furthermore, no changes in brain water content as a sign of edema formation in this organ were detected (transgenic: 78.15%±0.10%, n = 3; nontransgenic: 77.99%±0.31%, n = 11). Direct quantification of vascular permeability of brain vessels using sodium-fluorescein again revealed no significant differences between nontransgenic (78.2±15.01 rfu/mg brain tissue; n = 3) and transgenic (53.2±7.86 rfu/mg; n = 3) animals.
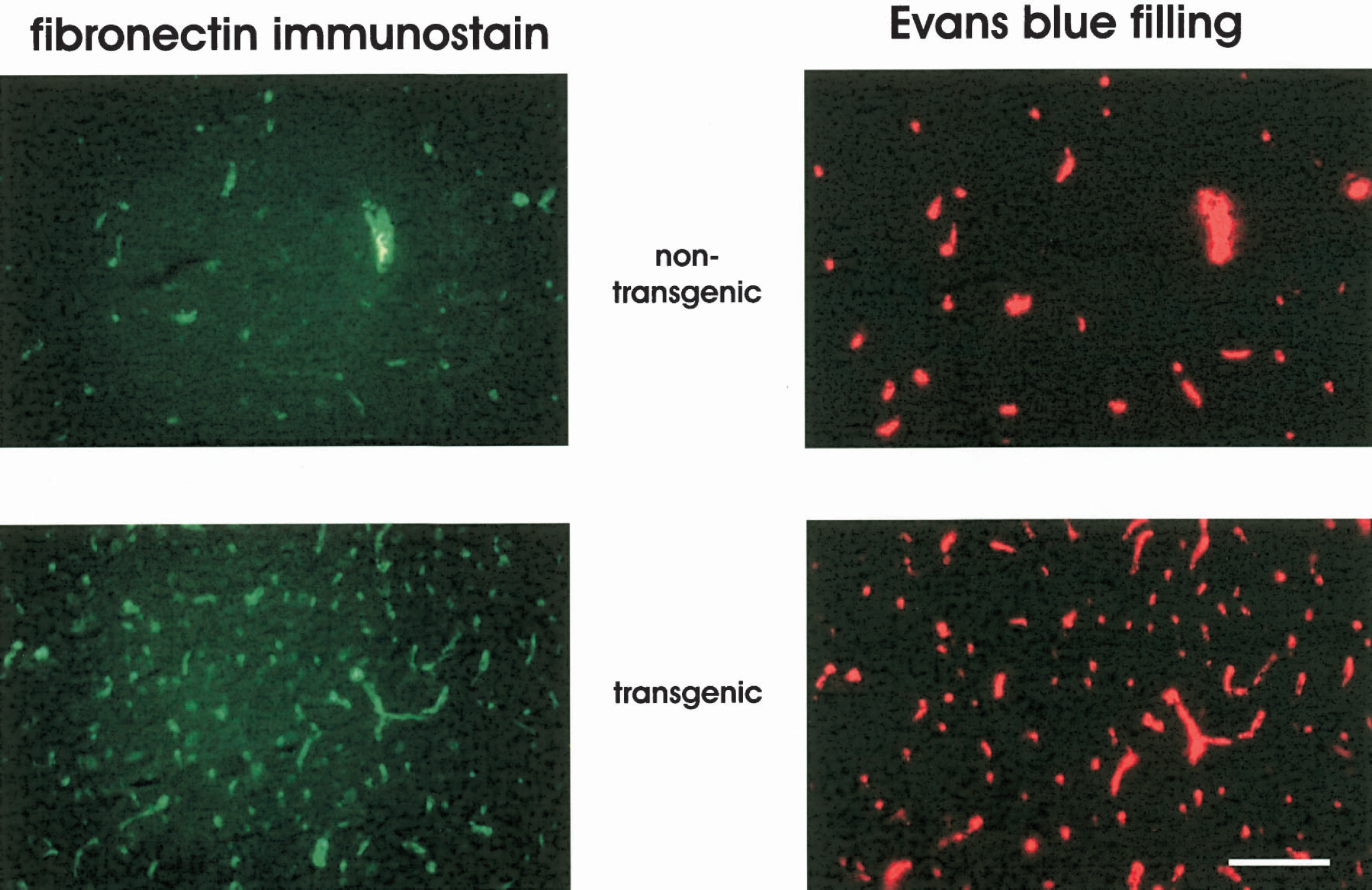
Fluorescent microscopic photographs of two representative brain sections showing indirect immunofluorescence of fibronectin (left) and Evans blue filling and extravasation (right) in the forceps minor of a nontransgenic (upper panels) and a transgenic mouse (lower panels). In both groups, all capillary spots are sharp on a dark background and no extravasation of the intravascular marker can be detected, indicating a tight blood–brain barrier. In addition, almost all Evans blue–filled capillary spots are associated with fibronectin immunoreactivity, indicating no major perfusion deficits of the capillaries. Note the higher density of capillary spots in the transgenic mouse. Scale bar = 50 μm.
In addition, in Fig. 1 each capillary spot can be detected in both images, indicating no major perfusion deficits of capillaries. Figure 2 shows the results of the capillary density and perfusion state measurements. The capillary density was significantly higher in transgenic mice in all brain structures investigated compared with nontransgenic littermates. The most prominent effects were found in white matter structures and the dentate gyrus. In these structures capillary density had more than doubled (Fig. 2a). As shown in Fig. 2b, measurement of the perfusion state showed slightly fewer perfused capillaries in some brain structures of transgenic mice. This might be due to a higher rate of capillary sprouts as angiogenesis might be ongoing in transgenic mice. However, because more than 97% of all capillaries are filled with Evans blue, the vast majority of the additional capillaries in the transgenic mice are functional. Capillary densities were increased preferentially in those brain structures that naturally have the lowest capillary densities, resulting in a more homogeneous distribution of the capillaries among the brains of transgenic mice. This is evident from the coefficient of variation of the capillary densities that was 0.308 in nontransgenic and 0.171 in transgenic mice.
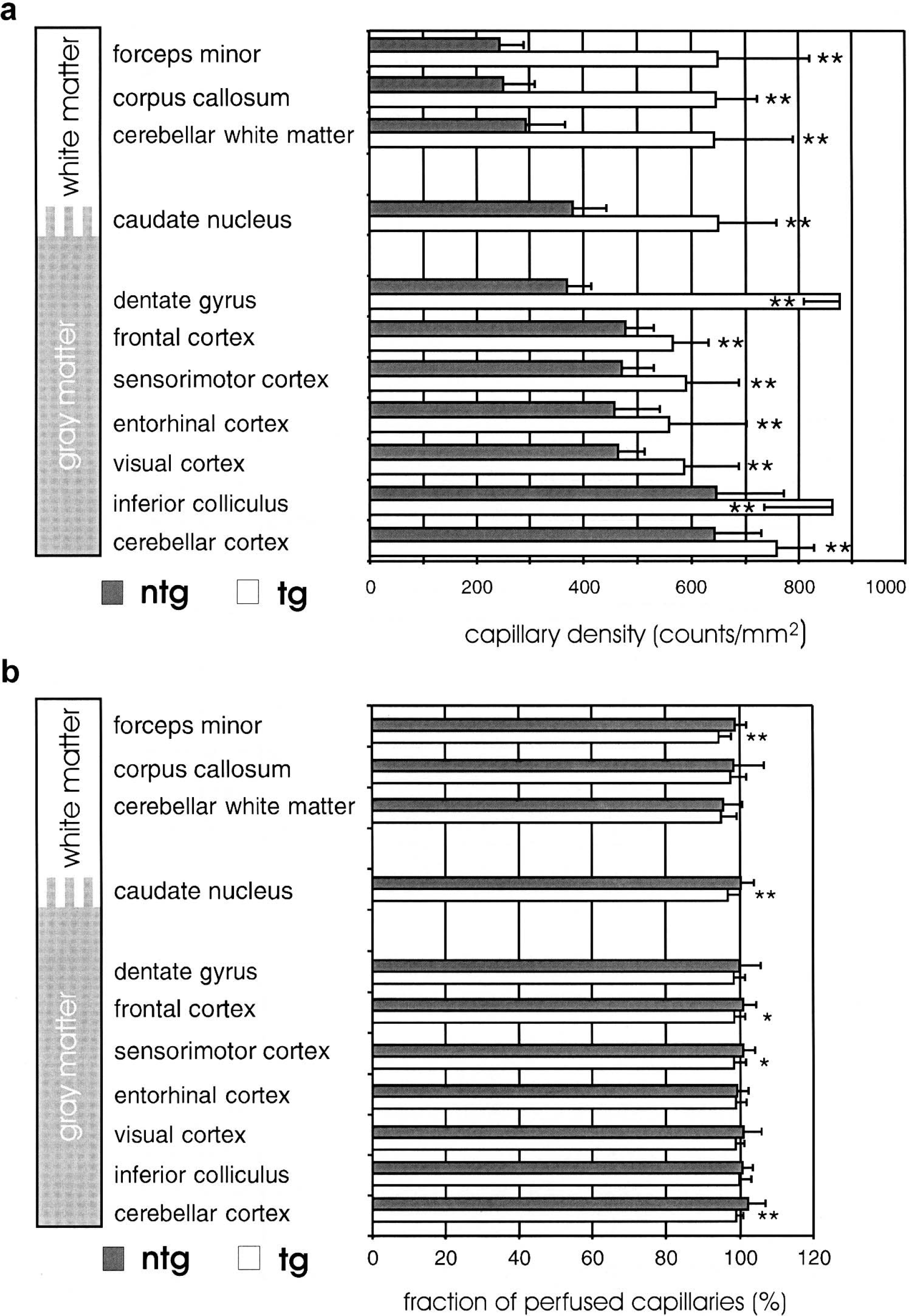
Capillary density and perfusion state of different brain structures of nontransgenic (ntg, gray bars) and transgenic (tg, white bars) mice. (
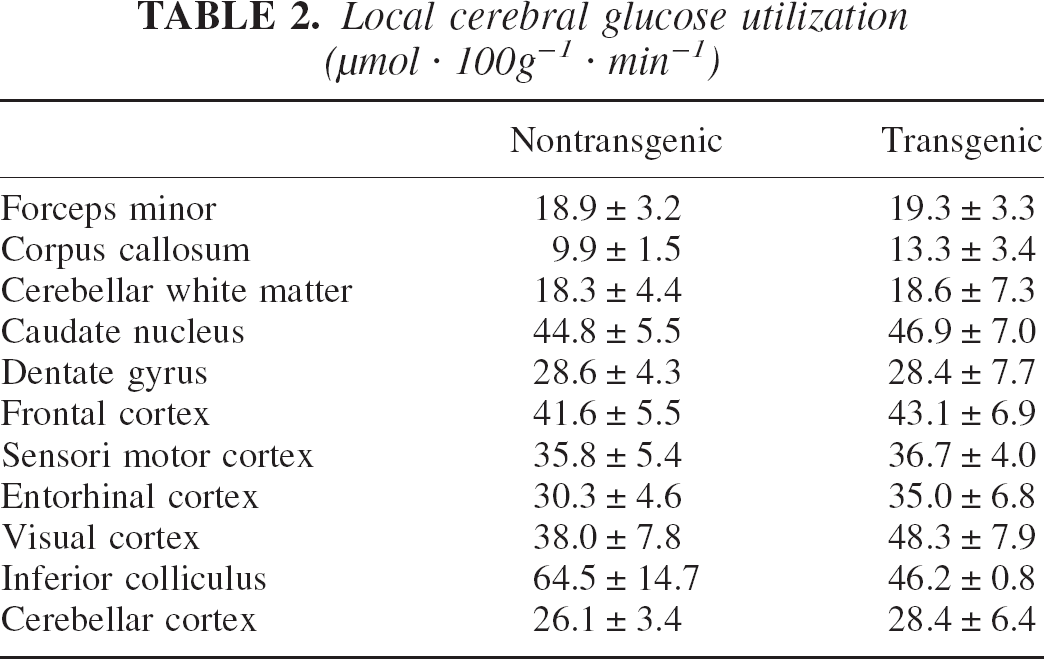
As shown in Table 2, local CGU did not differ between transgenic and nontransgenic animals in any brain structure investigated, indicating that new vessel growth in transgenic mice did not result in a changed metabolic demand of the brain tissue.
Local cerebral glucose utilization (μmol · 100g−1 · min−1)
Measurements of local CBF had been performed during normocapnia and hypercapnia. The results of these measurements are presented in Fig. 3. During normocapnia, local CBF of all brain structures investigated did not differ between groups. In contrast, during extreme hypercapnia accompanied by a maximal vasodilation, local CBF in transgenic mice increased to a significantly greater extent in the dentate gyrus and white matter structures (i.e., in all those brain structures where vessel density had more than doubled) (Fig. 3b). This finding indicates that a compensatory vasoconstriction during normocapnia might be one of the mechanisms responsible for suppressed luxury perfusion in transgenic mouse brains. However, compared with the up to 2.7-fold increase in capillary density in some transgenic brain structures, the elevation in local CBF observed during extreme hypercapnia in these areas was maximally 50%. Because during normocapnia capillaries contribute to approximately 30% of the total vascular resistance, at least a doubling of CBF is expected during extreme dilation of the upstream resistance vessels under hypercapnic conditions. Thus, the additional capillaries in the transgenic mice might be ineffectively perfused. We therefore assessed perfusion of single capillaries, and correlated to this end vessel density and local CBF in all brain structures. In nontransgenic mice a tight positive correlation between capillary density and CBF during normocapnia as well as hypercapnia was found (Fig. 4a), a finding similar to that found in rats (Klein et al., 1986; Kuschinsky, 1991). In contrast, this strong correlation was completely lost in transgenic mice (Fig. 4b). In line with this, the average perfusion per single capillary was reduced by approximately 30% in transgenic compared with nontransgenic mice, supporting the idea of some ineffective capillary perfusion even during maximal vasodilation.
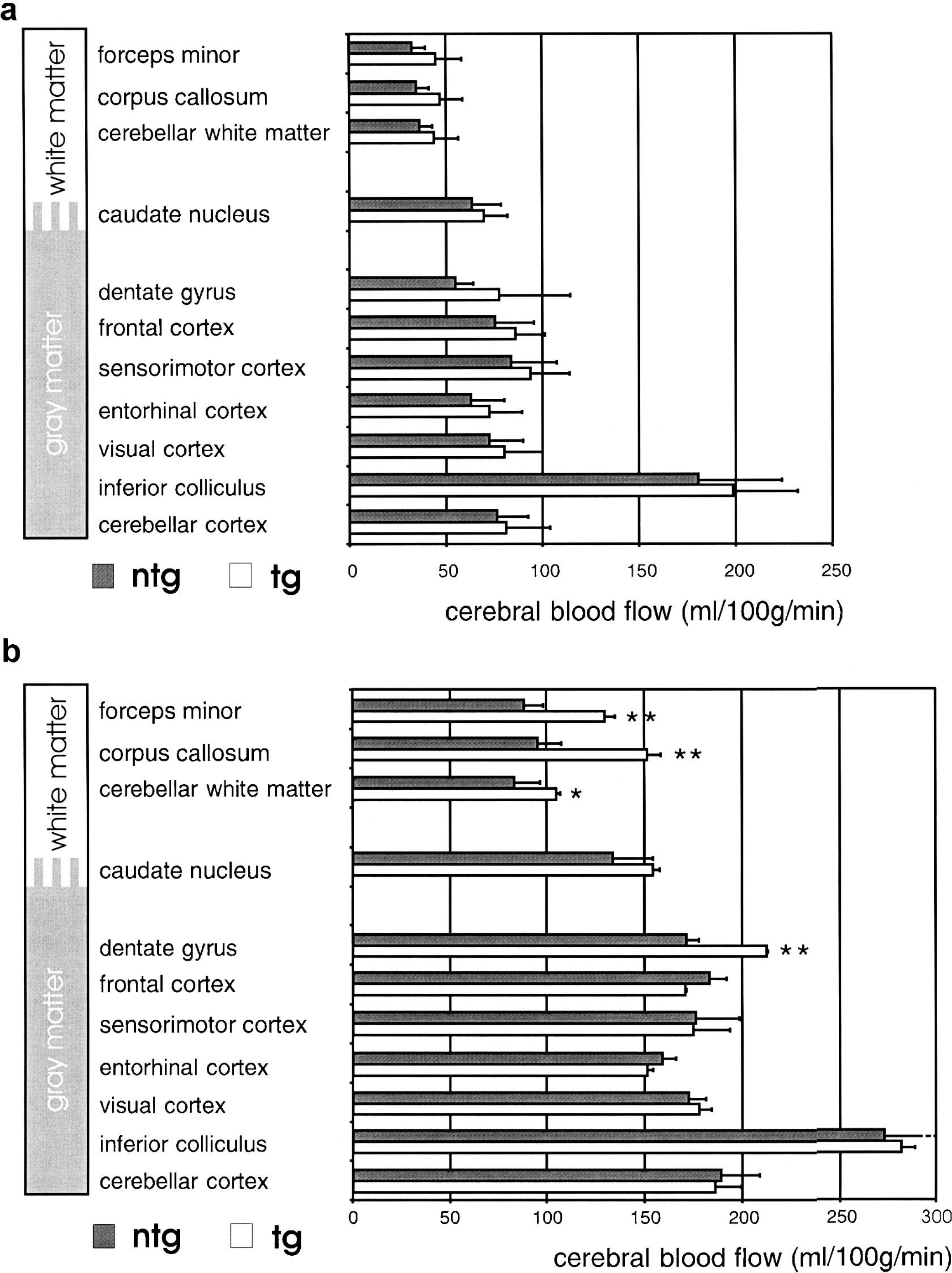
Measurement of local CBF during normocapnia (
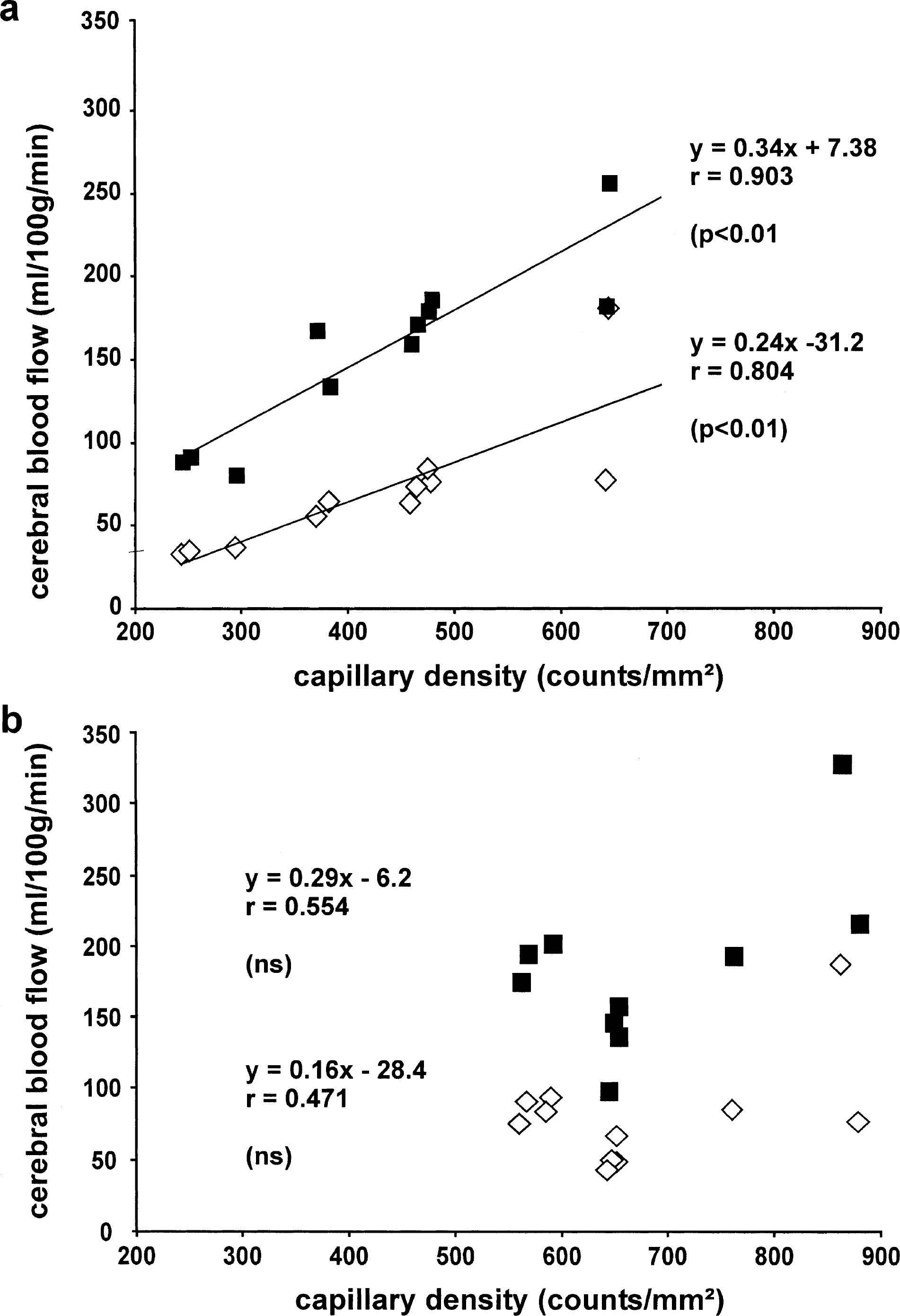
Correlation between capillary density and CBF. Each point represents the mean of six mice. CBF and capillary density were always measured in the same mouse. (
DISCUSSION
Transgenic, brain-specific overexpression of the human VEGF165 cDNA in mice results in increased density of morphologically normal and perfused capillaries, most prominently in white matter structures. During normocapnia, increased capillary densities were not associated with increased CBF. Even during extreme hypercapnia, the increase in CBF was much lower than expected from the increase in capillary densities. These findings indicate that one accountable mechanism might be a compensatory vasoconstriction during normocapnia. In addition, ineffective capillary network construction might also be responsible for the unexpected low effect of increased capillary density on CBF in transgenic mice.
VEGF, the most potent angiogenic factor, induces the formation of abnormal vessels when administered solely to the adult organism; these newly formed vessels are leaky, thin walled, and have an increased diameter (Carmeliet, 2000; Lee et al., 2000; Pettersson et al., 2000; Vogel et al., 2003). In brain tissue, leakiness results in severe brain edema, which is lethal at high VEGF dosages (Vogel et al., 2003). Transfection of the human VEGF165 cDNA into brain tissue resulted in increased CBF in the transfected area but also in dramatically decreased blood flow in adjacent areas, showing that such abnormal vessels have limited functional properties (Vogel et al., 2003). In contrast, the mouse model presented here with morphologically normal capillaries and a tight blood–brain barrier despite sixfold higher VEGF165 levels in the brain enabled us to elucidate the effect of increased capillary density independent of side effects such as abnormal vascular morphology or leakage.
Transgenic mice developed increased capillary densities that were, apart from the dentate gyrus, most apparent in white matter structures, despite transgenic VEGF165 expression being confined to gray matter. This might be explained by the fact that VEGF165 is a secreted growth factor with the capability to diffuse towards white matter areas. However, VEGF165 could have been transported via axons into the white matter (Ferrara, 2001; Schmechel et al., 1980), resulting in higher local concentration of VEGF165 in white matter. However, it is very difficult to measure the local VEGF165 concentrations in mouse brain.
The aim of the present study was to investigate the functional effects of increased capillary densities that are formed independent of an increased tissue demand. The unchanged metabolic rate despite increased angiogenesis in transgenic mice was proven by the measurement of the local CGU. During normocapnia, the increased capillary densities had no effect on CBF. During hypercapnia, CBF increased far less than expected from the increase in capillary density. Under normal conditions, capillaries contribute to approximately 30% of the flow resistance (Harper et al., 1984). Thus, at extreme hypercapnia, when upstream resistance vessels are maximally dilated (Reivich, 1964), a threefold increase in capillary density should result in a decrease of at least 50% in flow resistance with subsequent increase of CBF by approximately 100%. However, CBF was increased only by 20% to 30% in transgenic mice, and only in those brain structures where capillary density had at least doubled, resulting in a 30% reduction in single capillary blood flow in transgenic animals compared with controls. This fact raises the question as to compensatory mechanisms. Because the local CGU was not changed in transgenic mice, the lack of an increased CBF might be due to compensatory vasoconstriction during normocapnia. This is in line with other studies that showed direct dependence of CBF on the local oxygen demand of the brain tissue (Brown et al., 1985; Waschke et al., 1994). This compensatory effect could also be demonstrated by a reduction of the flow resistance after cleavage of the endothelial glycocalix, which resulted in a CBF increase only during extreme hypercapnia (Vogel et al., 2000). Similar to these findings, extreme hypercapnia in the present study resulted in a higher CBF only in brain structures showing more than doubled capillary densities, suggesting a partial contribution of vasoconstriction to the unexpected low CBF. However, this is not sufficient to explain the rather low increase in CBF during maximal vasodilation in face of a more than twofold increase in capillary density.
Two additional mechanisms could contribute to this observation. First, an insufficient flow capacity of upstream arteries and arterioles or the entire capillary network could prevent a higher CBF even during maximal vasodilatation. Under normocapnic conditions, approximately 50% of the cerebrovascular resistance is located in arterial vessels larger than 160 μm in diameter (Faraci et al., 1987; Faraci and Heistad, 1990). Because capillaries contribute to 30% of the vascular resistance (Harper et al., 1984), smaller arterioles (< 160 μm in diameter) account for an additional 20% of flow resistance. Therefore, in the brain the main vascular resistance (> 70%) is represented in arteries and arterioles, making it unlikely that the flow capacity of arterial vessels is a limiting factor.
However, one could argue that CBF values do not change much when capillaries contribute (only) approximately 30% to the total vascular resistance. However, during extreme vasodilation (PaCO2 > 130 mm Hg), the resistance vessels maximally dilate (Reivich, 1964) whereas capillaries hardly dilate (Duelli and Kuschinsky, 1993). Capillaries would then contribute to a much higher percentage of the total vascular resistance. Thus, a two- to threefold increase in capillary density would result in a decrease in flow resistance of at least 50% and consequently result in an increase in CBF of approximately 100%. However, CBF measured during extreme hypercapnia was only 20% to 30% higher in transgenic mice, and the flow per single capillary was reduced to 70% compared with nontransgenic mice. These findings indicate that additional mechanisms account for the attenuated CBF increase during hypercapnia in areas of increased capillary density.
A second explanation for this observation might be ineffective insertion of the additional capillaries into the preexisting network. After angiogenesis, microvascular networks have to be remodeled to establish an optimal flow distribution among all capillaries and an effective tissue supply. In this process, mechanical as well as metabolic stimuli appear to be involved (Bongrazio et al., 2000; Pfeifer et al., 1998; Pries et al., 2001). In contrast, in our transgenic mice no changes of the metabolic demand (unchanged local CGU) of the brain tissue were observed. The metabolic stimulus is therefore missing. Thus, the normal remodeling process might be disturbed, resulting in an ineffective integration of additional capillary segments into the preexisting microvascular network. Blood flow per single capillary is normally very similar in each brain structure despite different capillary densities (Klein et al., 1986; Kuschinsky, 1991), which holds true also for our nontransgenic mice during normocapnia as well as during extreme arteriolar vasodilatation induced by hypercapnia (Fig. 4). In contrast, flow per single capillary was reduced and the correlation between capillary density and CBF lost even under hypercapnia in transgenic mice. This strongly suggests a structural difference in the three-dimensional microvascular network construction between nontransgenic and transgenic mice. Different possibilities exist as to how additional capillaries can be inserted into preexisting networks as summarized in Fig. 5. First, an additional capillary could be inserted as an additional pathway between in- and outflow resulting in an increased flow capacity of the whole network (Fig. 5A). Second, the new capillary could bridge between two existing capillaries with equal blood flow direction without change in the network's flow capacity (Fig. 5B). Third, angiogenesis could also result in a mere elongation of existing capillaries leading to more coiled vessels. Such coiling has indeed been observed in the skin of mice overexpressing VEGF in keratinocytes (Detmar et al., 1998; Thurston et al., 1999). Elongation of capillaries would increase their flow resistance and thus reduce flow capacity of the microvascular network (Fig. 5C). However, it is very difficult to assess functional relevant construction changes of the highly complex three-dimensional capillary network of the brain (Yoshida and Ikuta, 1984; Motti et al., 1986) from two-dimensional sections. To answer this question, additional morphometric studies of the capillary network appear to be necessary.
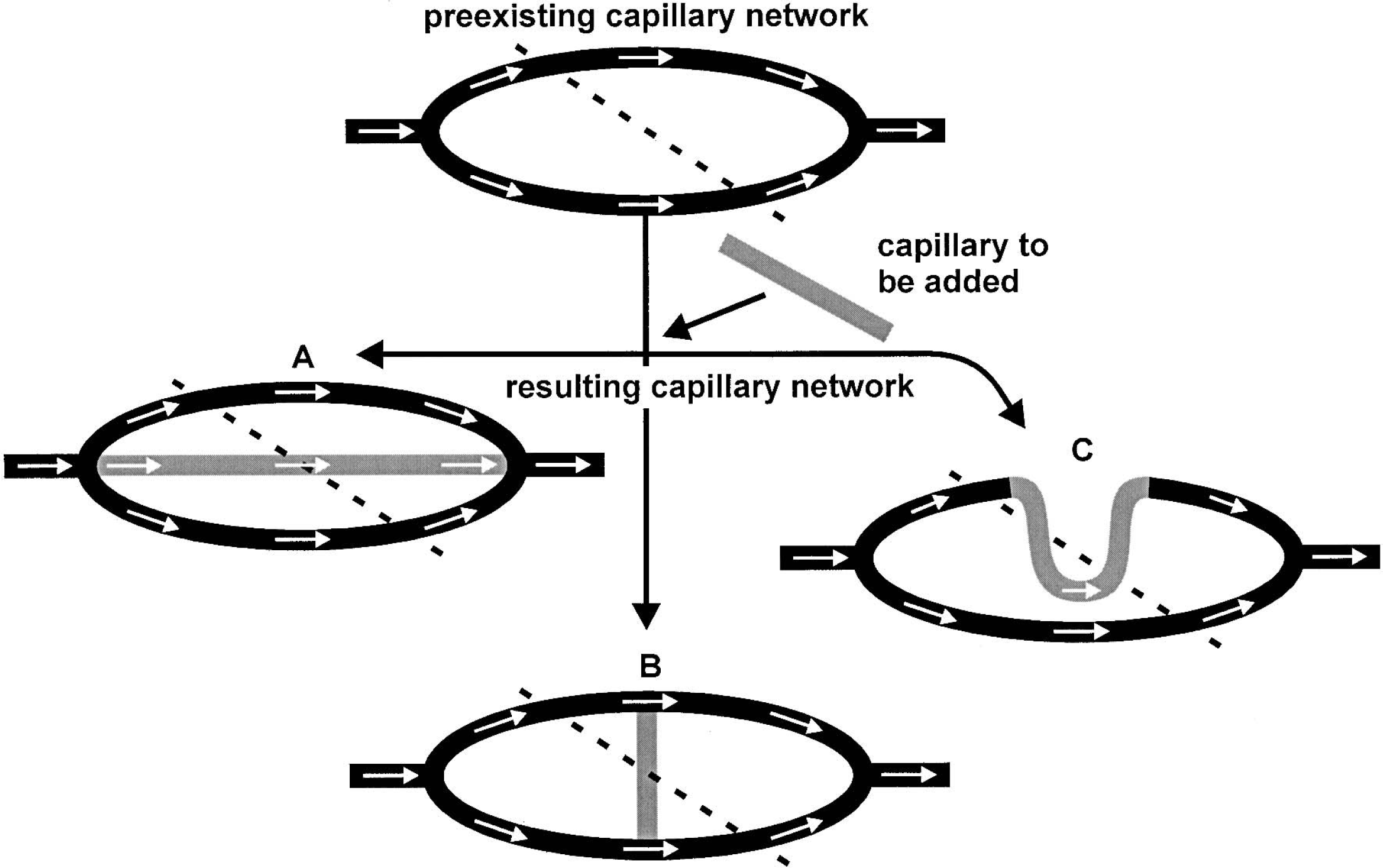
Illustration of the three (
In summary, the present study has shown that a drastic increase in density of normal and perfused capillaries due to transgenic overexpression of VEGF165 did not result in increased CBF during normocapnia. Even during extreme hypercapnia, which is paralleled by maximal dilation of larger brain arteries and arterioles, only minimally higher CBF values were measured. These findings indicate the presence of functional relevant differences in the microvascular network construction in VEGF-over-expressing mice, and suggest that additional vascular remodeling is required to establish a proper vascular network (Bruick and McKnight, 2001).
Footnotes
Acknowledgments:
The authors thank G. Breier, U. Deutsch, and J. G. Sutcliffe for the gift of plasmid DNA; G. Teichmann, M. Walker, and F. Müller-Holtkamp for generating transgenic mice; S. Lindlar, C. Büttner, M. Euler, H. J. Schoch, B. Saam, and R. Landwehr for excellent technical assistance; and O. Ogunshola for editing the manuscript.
