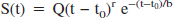Abstract
Functional imaging studies typically give prominence to positive responses. However, negative changes that accompany activation-induced positive responses are not yet clearly understood. The authors investigated the origin of sustained negative CBF responses that accompanied positive CBF changes. Measurements were made in the rat somatosensory cortex in response to whisker stimulation using laser-Doppler imaging. Flux images indicative of CBF were obtained at rest and during whisker stimulation with a spatial resolution of 200 μm. Large and intermediate blood vessels in the cortical surface exhibiting high flux values were clearly resolved. This greatly reduced the contamination of the tissue pixel volume with macroscopic blood vessels. Regions that responded positively to whisker stimulation were from areas with intermediate to low baseline flux and distinctly away from high flux areas. Stimulation-induced change in signal intensity was the largest in pixels with low baseline flux, presumably from tissue and microvessels. Simultaneously, a sustained decrease in signal intensity was observed in regions with high baseline flux values. The temporal coherence, macrovascular origin, lesser trial-to trial variability, and complete absence of the negative CBF response in the microvascular regions suggest that it may be purely hemodynamic in nature.
Sensory stimulation leads to an increase in electrical activity within neuronal populations resulting in local vasodilation. This leads to a change in blood flow and oxygenation in the activated regions (Woolsey et al., 1996). Localization of neuronal activity is inferred from such changes, which forms the basis of functional imaging techniques (Raichle, 1987). Although functional imaging studies have extensively observed positive responses and their underlying mechanisms have been well characterized, negative signal changes in response to neuronal activity are infrequent and have not received much prominence. Negative responses can occur independently in the absence of positive responses or simultaneously with the positive responses. In functional magnetic resonance imaging, the blood oxygenation level-dependent (BOLD) and flow signal changes may be positive under awake conditions and negative to the same stimuli, in the same subject, under certain physiologic conditions such as slow-wave sleep or sedation (Altman and Bernal, 2001; Born et al., 2002; Martin et al., 2000). The negative BOLD response has been related to a decrease in rCBF, leading to the hypothesis that certain energy-consuming cognitive processes active during sleep conditions cease during stimulus presentation, resulting in a negative response (Born et al., 2002). Several positron emission tomography and functional magnetic resonance imaging studies have reported the phenomenon where a simultaneous and sustained negative signal shift is observed in regions adjoining the activation-induced positive response (Allison et al., 2000; Brefczynski and DeYoe, 1999; LeBihan and Dohi, 1995; Shmuel et al., 2002; Shulman et al., 1997). Using visual and language tasks, Shulman et al. (1997) have shown that fixed foci in different regions in the cerebral cortex consistently display a decrease in CBF. However, Brefczynski and DeYoe (1999) obtained patterns of positive and negative signal change in the visual cortex that varied spatially with directed visual attention. Subsequently, the positive and negative activation were shown to be temporally coherent and dynamically dependent on the stimulus size (Saad et al., 2000). Although these studies hypothesize a possible neuronal origin for the observed negative responses that accompany positive signal changes, others attribute it to both neuronal and nonneuronal factors including passive hemodynamics. Harel et al. (2002) have observed negative BOLD and cerebral blood volume (CBV) responses in the suprasylvian gyrus containing higher-order visual areas, contrary to positive BOLD and CBV changes in the adjacent primary visual cortex during visual stimulation. These results support the notion that reallocation of cortical blood resources could overcome a local demand for increased CBF induced by increased neural activity. Shmuel et al. (2002) have recently shown that the negative BOLD, flow, and oxygen consumption were sustained and coupled to the positive response through a reduction in neuronal activity and passive CBF changes. Despite insights into the hemodynamic and neuronal contribution to negative functional responses, the exact mechanism and the compartment that gives rise to it has not yet been elucidated.
In the present study, the spatiotemporal characteristics of CBF changes were measured using laser-Doppler imaging (LDI) in response to whisker stimulation. The primary somatosensory barrel field (S1BF) is a unique region in rodents in which neuronal and metabolic activity change in response to whisker movement (Woolsey and Van Der Loos, 1970). The layer IV of this region contains dense cell structures, which are termed barrels. Their spatial pattern corresponds to the large vibrissae along the face (Armstrong et al., 1992; Ito, 1981; Simons, 1978). Its anatomy and physiology is well suited for studies on localized neuronal activity and activity-flow coupling studies (Cholet et al., 1997; Dirnagl et al., 1994; Wang et al., 1993). Our results show that simultaneous positive and negative LDI signal changes occur in the somatosensory barrel field during whisker stimulation. Although the positive signal shifts originated predominantly from the microvascular regions, the negative CBF shifts were confined to certain large blood vessels. The absence of negative responses in the microvascular regions and the spatiotemporal analysis of the positive and negative LDI responses suggest a passive hemodynamic change “perfusion steal” that may lead to the negative CBF signals adjoining positively activated regions.
MATERIALS AND METHODS
Surgical preparation
Seven male Harlan Sprague-Dawley rats (250–300 g; Indianapolis, IN, U.S.A.) were initially anesthetized with urethane (1.2 g/kg, intraperitoneal) and supplemented with 20% of the initial dose subsequently at later times if blood pressure increased to a tail pinch during the protocol. Body temperature was monitored with a rectal probe and maintained at 37.0° ± 0.5°C using a homeothermic feedback heating system (Baxter K-MOD100; Gaymar Industries, Inc., Orchard Park, NY, U.S.A.). The femoral arteries were cannulated with PE50 tubing for MABP measurements and blood gas sampling. The animal was subsequently tracheotomized and mechanically ventilated with room air. Gallamine triethiodide (Sigma, St Louis, MO, U.S.A.), a muscle relaxant, was administered in a single intraperitoneal dose of 250 mg/kg at the start of ventilation to prevent spontaneous breathing of the rats. Arterial blood pressure, end-tidal CO2, and inspired oxygen concentration were continuously monitored (POET II; Criticare Systems, Inc., Waukesha, WI, U.S.A.). The physiologic parameters with room air ventilation were as follows: PaO2 = 92.4 ± 8.5 mm Hg, PaCO2 = 36.2 ± 3.5 mm Hg, pH = 7.4 ± 0.06, and MABP = 93.9 ± 7.5 mm Hg. The study protocol followed institutional guidelines and was approved by the Animal Research Center of our institution.
The head was secured to an adjustable stereotaxic frame and the scalp was retracted from the frontoparietal cortex region on the intact skull by a midline incision. The temporalis muscle was disconnected from the skull. An area of 5 × 5 mm (centered 2 mm posterior and 5 mm lateral to the bregma) on the skull enclosing the whisker barrel cortex was thinned to translucency on either hemisphere (Gerrits et al., 1998). The underlying vasculature was visible when washed with saline. During measurements in the contralateral cortex, the stereotaxic frame was adjusted to provide a tilt of 30 degrees on the midline axis. This aligned the surface of the whisker barrel cortex normal to the laser beam.
Laser-Doppler imaging
Laser-Doppler imaging was carried out using the Moor LDI device (Moor Instruments, Sussex, U.K.). The device uses a set of plane mirrors to direct the beam from a low power (2 mW ± 20%, 632.8 nm) He-Ne laser onto the tissue surface and also collects the scattered Doppler shifted beam from the tissue, which is focused onto a photodetector. The mirror controlled by a motor enables the laser beam to scan in a raster pattern across the surface of the tissue and also capture the light scattered from the tissue. This is focused by a lens (50-mm diameter, f ± 100 mm) onto a photodetector, which substantially increases the light-collecting area. The Doppler shift is processed to build up a color-coded flux image of blood flow. The Moor LDI image can be generated in a matrix of 256 × 256 pixels covering an area of 5 × 5 cm2 with a maximum distance of 1 m between the head of the scanner and the tissue surface imaged. The highest achievable spatial resolution is determined by the beam diameter (100 μm). The in-plane resolution of the measurement depends upon the distance between the imaged tissue and the scanner head. In the present study the distance between the LDI scanning head and the thinned dura was 21 cm, thus improving the spatial resolution by almost four times compared with previous microimaging studies using LDI (Ances et al., 1999). In the present configuration, the moor LDI device is sensitive to a tissue depth of 0.5 mm (Jakobsson and Nilsson, 1993).
Images were initially obtained with a larger field of view but lower temporal resolution. Both hemispheres over the somatosensory cortex were covered to detect activation in the contra-lateral cortex in response to whisker stimulation. The location of Bregma in LDI pixel coordinates was also recorded. Subsequently, to improve temporal resolution, images were obtained over the contralateral cortex only where the image field of view completely covered the thinned skull window and the field of view was centered to the square window. Because the field of view did not include the bregma in the contralateral images, the thinning of the skull was always performed with a square geometry with its center 5 mm lateral and 2 mm posterior to the bregma. Flux images of the contralateral cortex were obtained with a matrix of 34 × 34 pixels covering a scan area of 0.7 × 0.7 cm leading to an in-plane resolution of 206 × 206 μm2. Images were obtained at a rate of 8 s/image and a dead time of 4 seconds between each image with a distance of 21 cm between the laser and the imaged surface. Twenty-two images were obtained during each stimulation protocol consisting of whisker stimulation for 24 seconds in two epochs separated by rest. The total time between consecutive images was 12 seconds.
Experimental stimulation
The right vibrissae were cut to a length of approximately 3 cm from the face and fitted through a mesh screen fixed to a solenoid-driven actuator arm while the left vibrissae was completely trimmed. The screen was positioned approximately 1 cm from the rat's face to ensure facial hair is not stimulated during whisker activation. A timing circuit controlled the solenoid driven actuator arm with an electrical transistor transistor logic (TTL) pulse output that was used to mark the stimulation period extent during data collection. A frequency of 8 Hz was used during stimulation because maximal flow changes have been reported to occur around this frequency range (Gerrits et al., 1998).
Image processing and statistical analysis
The Moor LDI Data analysis software package was used to process images during experiments. Image time-series data from specific regions of interest was used to detect activation. The flow related signal derived from the Doppler spectrum was processed via an A/D converter in the imaging system. Before the image was reconstructed, the signal was compensated for noise effects and color-coded in arbitrary flux units (Essex and Byrne, 1991). The image data were imported and analyzed using MATLAB (MathWorks, Natick, MA, U.S.A.). An idealized square-wave function representing the “on/off” cycle of the whisker stimulation was used as the reference waveform and cross-correlated with every pixel time course of the signal. Pixels with correlation coefficients greater than 0.4, which corresponds to a statistical significance of P < 0.05 after a Bonferroni correction, were considered active.
Because noise levels were relatively higher in pixels with high flux than in pixels with low flux, a bootstrap resampling technique was applied. This method was used to obtain the confidence interval of LDI parameters of interest. Bootstrap resampling is an empirically based method in which a large number of simulated datasets are generated with the same distribution as the original data sets. These simulated data sets have the same size as the original data set and are generated from the existing measurements with repetition (Efron and Tibshirani, 1993). The data are assumed to be a vector yobs of n independent observations. To estimate the confidence interval for θ (yobs) (correlation coefficient in the present case), the observations were sampled randomly with replacement from yobs to obtain a bootstrap dataset Y*. The boot samples of the time series and reference waveform were generated and cross-correlated on a pixel-by-pixel basis. This leads to the bootstrap version of the statistic of interest θ* = θ (Y*). This procedure was repeated 100 times to obtain an estimate of the bootstrap distribution. No prior assumptions regarding the nature of the data set need to be made. The mean and standard deviation of the distribution of correlation coefficients in each pixel in the image were determined. Activation maps were obtained using a 95% confidence interval of the correlation coefficient distribution corresponding to a statistical significance of P < 0.05.
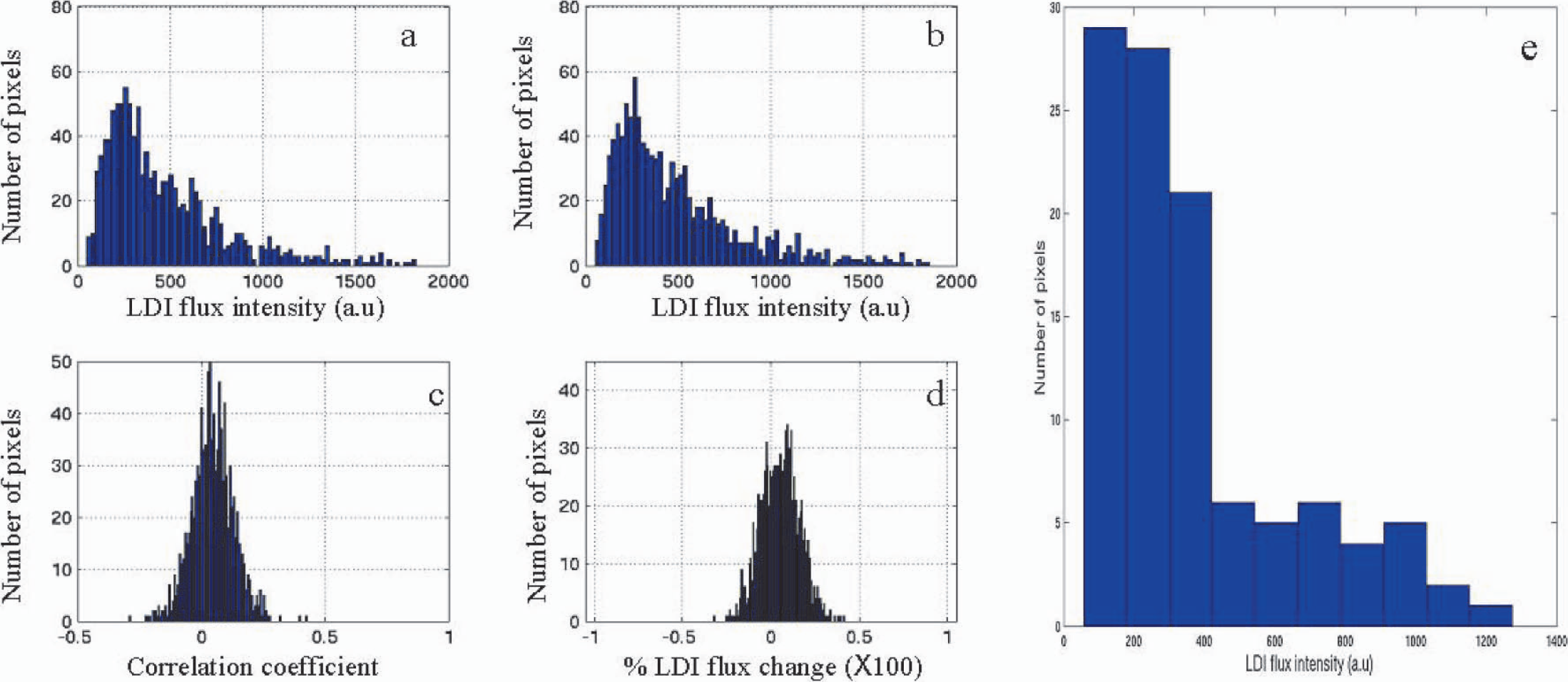
The activation-induced LDI response was modeled to a γ-variate function by nonlinear regression using AFNI (Cox, 1996). The γ-variate function is defined as:
Where S(t) is the LDI flux intensity; Q, r, and b are fit constants; t is the time after the start of whisker stimulation; and t0 is the onset time, the time between the start of whisker stimulation and 10% change in peak signal response. The experimental data was fit to the nonlinear model on a pixel-by-pixel basis after convolving it with the idealized square-wave reference function. The onset time (t0), the time to maximum signal (tmax), and the percent change at signal maximum were obtained for every pixel.
Regions of overlap between any two single trials were computed using the correlation coefficient images by creating a binary mask with a threshold of +0.3 for the positive and −0.3 for the negative responses. Overlapped regions were detected on a pixel-by-pixel basis using a logical AND operation on the two chosen masks. The percent overlap between any two trials was calculated as the number of active pixels in the resulting mask after an AND operation divided by the number of active pixels in the resulting binary mask after an OR operation between the same trials.
RESULTS
Laser-Doppler images were initially obtained with a rectangular field of view of 0.4 × 1.8 cm, covering the thinned skull windows on either hemisphere. Twenty-two sequential laser-Doppler images were obtained at rest and during whisker stimulation performed in two epochs. Images in the time series during rest and whisker stimulation were respectively averaged. Fig. 1a shows an average color-coded laser-Doppler flux image obtained from an anesthetized rat during rest. Pixels within the thinned skull window showed higher average flux values when compared with pixels lying over the normal skull. Large surface vessels showing high flux values were also resolved. Fig. 1b shows the difference image after subtracting the average whisker stimulation image and the average rest image from a single experiment. Activation in the contralateral cortex was observed in the difference image with a cluster of pixels showing a relatively higher flux difference. Large signal change seemed to emerge from pixels with low or intermediate flux values (perhaps capillaries and arterioles) and well within the area of the thinned skull window. The whisker barrel cortex was 5 mm lateral to the bregma making a steep angle in relation to the horizontal plane of the bregma; hence, the activated region was not normal to the laser beam. However, the spatial extent of activation in the contralateral cortex was highly reproducible in all animals imaged.
Typical laser-Doppler images obtained from an anesthetized rat covering the whisker barrel cortex on either hemisphere. Images are color coded in arbitrary perfusion units. A series of 22 flux images were obtained one every 14 seconds. Baseline images during rest (images 1–6, 9–14, and 17–22) were obtained with intermittent whisker stimulation (images 7–8 and 15–16). (
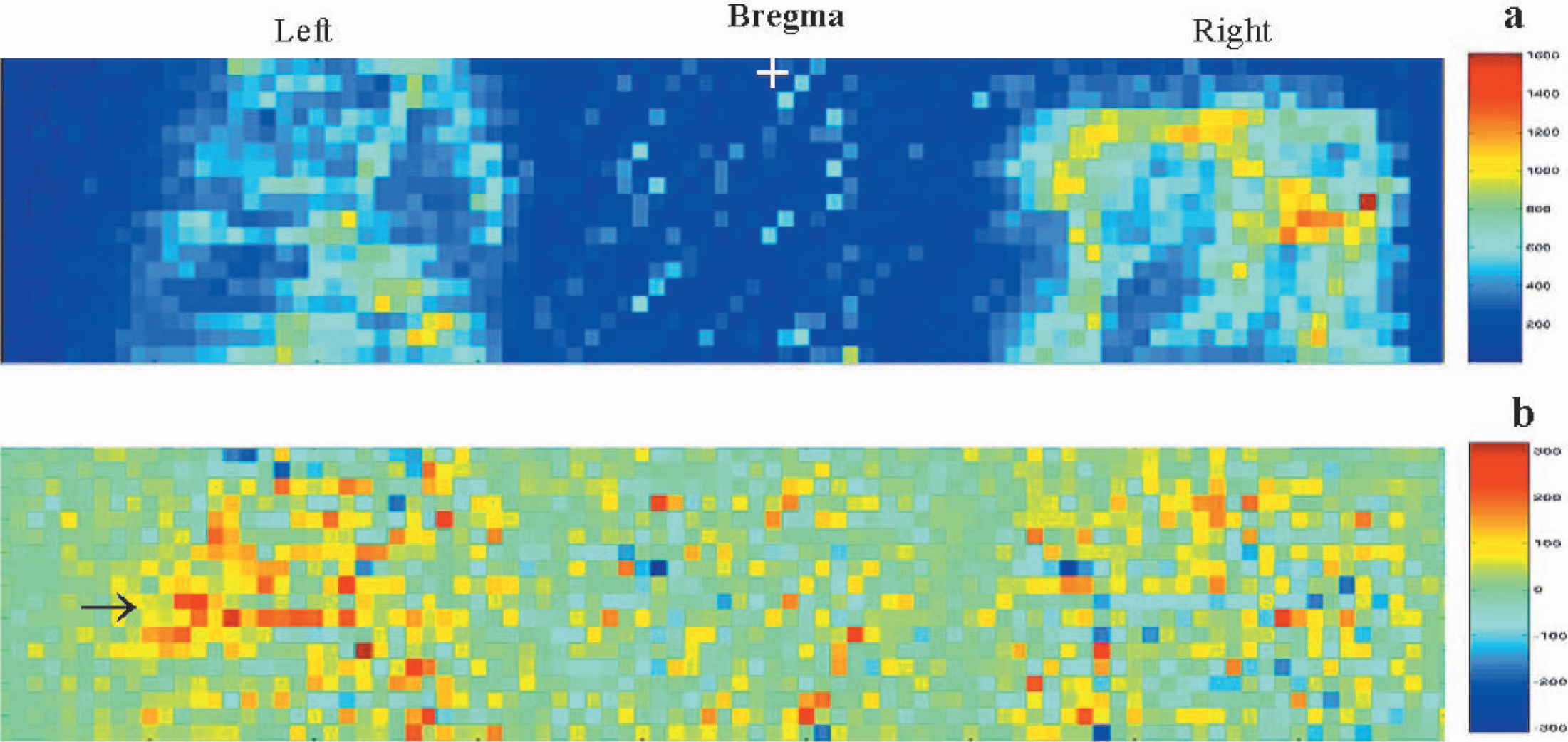
After obtaining measurements over the thinned skull area that included both hemispheres, rats were subsequently imaged only in the contralateral cortex. Selective imaging of the contralateral cortex not only enabled the tissue surface to be normal to the scanning laser beam, but also improved temporal resolution from 18 to 12 seconds owing to a smaller field of view. To enable this, the rat was tilted by 30 degrees on the stereotaxic frame along the midline axis such that the surface of the thinned skull window over the contralateral cortex was normal to the laser beam. All images in the contralateral cortex were obtained with the rat oriented in this position. Fig. 2a shows the high-resolution flux image obtained over the contralateral cortex during rest after averaging the baseline images in a single experimental trial. Pixels having the highest flux values were from the underlying large blood vessels from the cortical surface. With an in plane resolution of 206 μm, typical surface vessels were clearly discerned while intermediate vessels were partially resolved. Figs. 2b, 2c, and 2d show images of the difference between the average rest and activated images, correlation coefficients and percent change in LDI signal, respectively, from a single experimental trial. Activated regions were manifested as clusters of pixels showing high percent change, which clearly coincided with pixel clusters showing high correlation coefficients. These were primarily from regions with small baseline flux values. A small number of pixels displayed a negative percent change and negative correlation with whisker stimulation. They were usually localized to regions having high baseline flux values (>800 rpu) (Figs. 2a and 2d). Figs. 2e and 2f show the activation maps obtained after hard thresholding the correlation coefficient image at 0.4 and after bootstrap re-sampling, respectively. A 95% confidence interval of the correlation coefficient distribution was used after cross-correlating the bootstrapped data so that both maps (Figs. 2e and 2f) correspond to a significance level of P < 0.05.
Laser-Doppler images obtained from a single experimental trial in an anesthetized rat over the contralateral cortex. Twenty-two flux images were obtained over the contralateral cortex every 8 seconds with rest and intermittent whisker stimulation. (
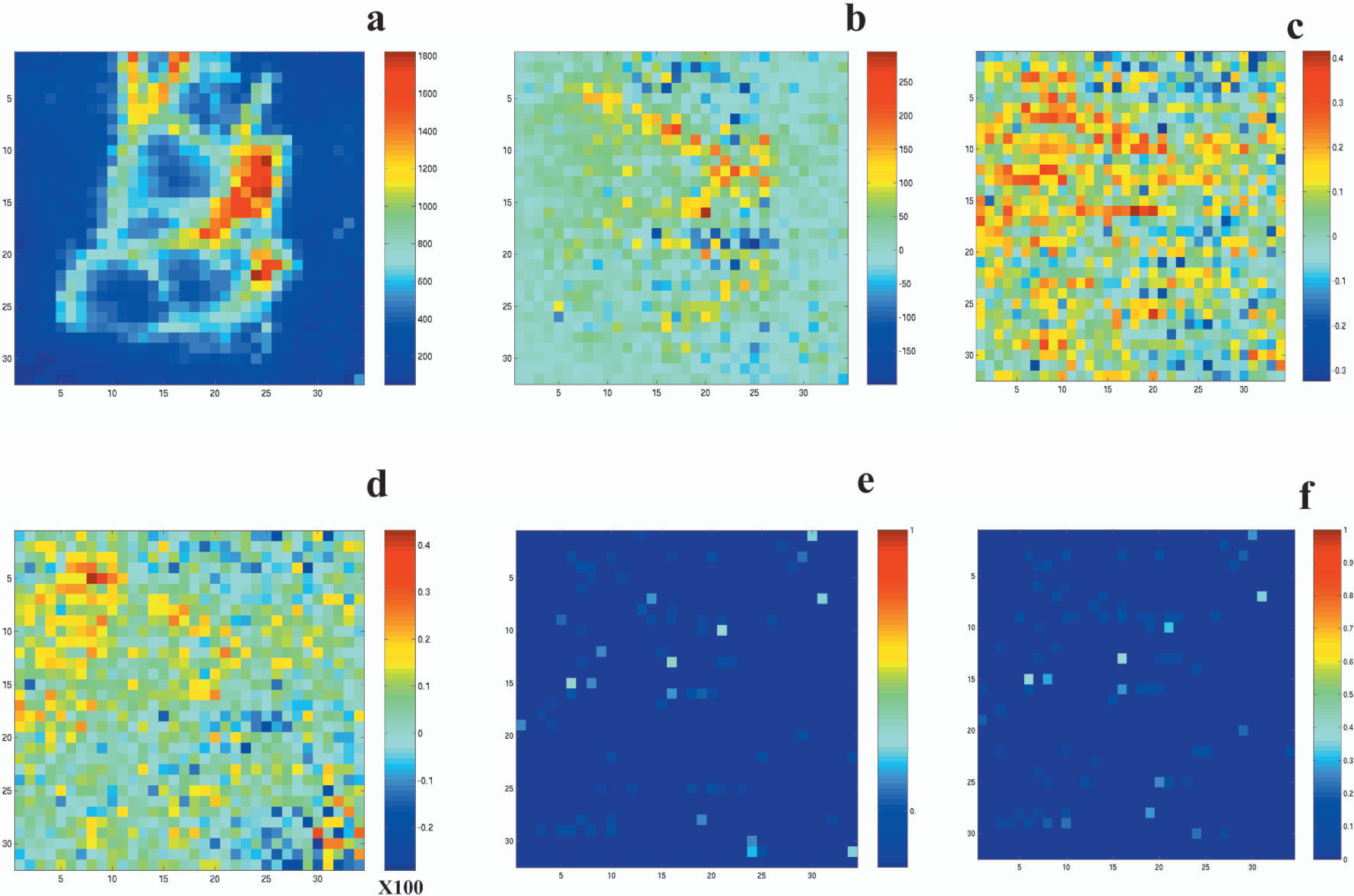
Averaging of images across animals was avoided because of inherent limitations in the accuracy of the field of view and apparent variations in the vascular anatomy of the cortex. However, activation was highly reproducible across animals. Figs. 3a and 3b show the distribution of flux values in the images at rest and during activation obtained in the contralateral cortex averaged over multiple experimental trials in all animals. Figs. 3c and 3d show the average distribution of correlation coefficients and percent change in LDI flux signal induced by whisker stimulation, respectively. Percent change in LDI flux in response to whisker stimulation varied from −20% to 40%. Median change in flux induced by whisker stimulation was 7% ± 2% from the images obtained over the contralateral cortex over all animals. Fig. 3e shows the distribution of baseline flux values from pixels having greater than 20% change in laser-Doppler flux during whisker stimulation. Pixels with robust positive responses were primarily from microvascular regions (<400 rpu).
Average distribution of pixel values across the image field of view from the contralateral cortex. (
Figure 4 shows the average signal time course during three whisker stimulation experiments from a typical rat. Different regions of interest were examined according to their baseline flux values and correlation coefficients in response to whisker stimulation. Regions showing positive responses (ROI 1) usually had low baseline flux (Fig. 4a). The grand average baseline flux value of pixels showing positive CBF response to whisker stimulation over all animals was 415 ± 30 rpu. However, certain pixels having high baseline flux values (>800 rpu) and negative correlation coefficients (ROI 2) showed negative CBF changes during whisker stimulation (Fig. 4b). Activation-induced negative responses were consistently observed in similar pixel locations in multiple experiments in a single animal. Fifteen to twenty percent of overall pixels had large flux values (>800 rpu), and majority of them showed minimal or no significant change in CBF during whisker stimulation. Fig. 4c shows the average signal time course obtained from three whisker stimulation experiments in a typical rat from pixels having baseline flux values >800 rpu. Though the noise levels were different, the flow in both high-flux and low-flux pixels were stable over the complete period of measurement. Fig. 4e shows the typical time course of laser-Doppler flux in a low-flux and high-flux pixel, respectively.
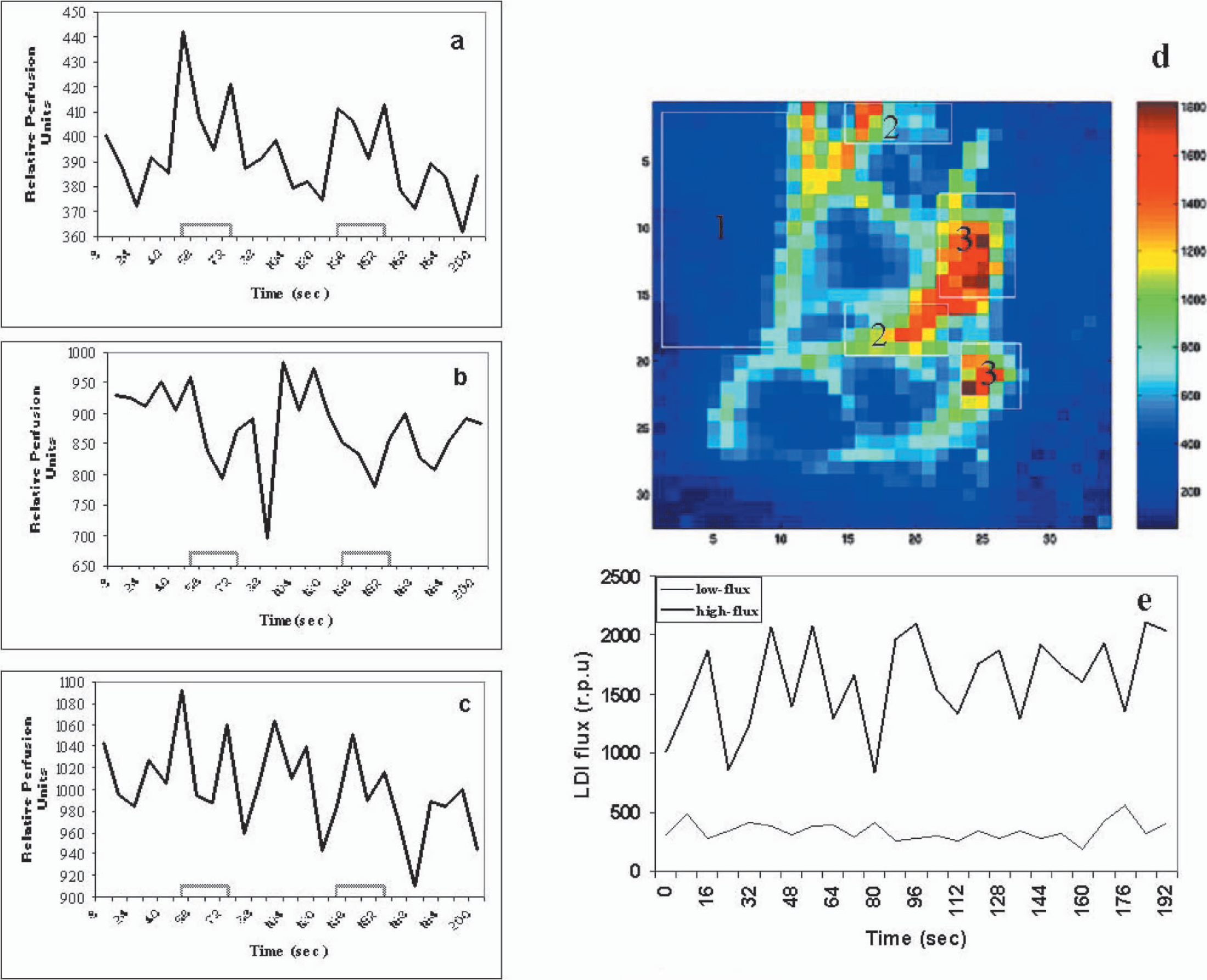
Average time course of the laser-Doppler flux signal and reference waveform from different regions of interest (ROI) from a typical whisker stimulation experiment. (
To quantify the temporal dynamics of the CBF response, the signal time series was modeled to a γ-variate function on a pixel-by-pixel basis, and parameters such as the signal onset time t0, time to peak tmax, and percent change in peak LDI flux signal were estimated from the nonlinear fit. The average amplitude of the negative response was approximately one third, but temporally coherent with respect to the positive response (Table 1). Fig. 5 shows typical time courses of the laser-Doppler signal from pixels showing positive and negative responses respectively and the nonlinear fit function. To control for any confounding effect of anesthesia on the regional cerebrovascular reactivity, hypercapnia was induced in the rats by ventilation with 2% or 5% CO2. Hypercapnia induced a large flux change throughout the field of view with the largest flux change in pixels with high baseline flux values (intermediate and large vessels). Fig. 6 shows the typical CBF response from a rat in the whisker barrel cortex region as a result of hypercapnia induced by either 2% CO2 or 5% CO2 ventilation.
Dynamics of the LDI signal change in response to whisker stimulation in anesthetized rats estimated from the nonlinear fit to the hemodynamic response on a pixel-by-pixel basis

LDI, laser-Doppler imaging.
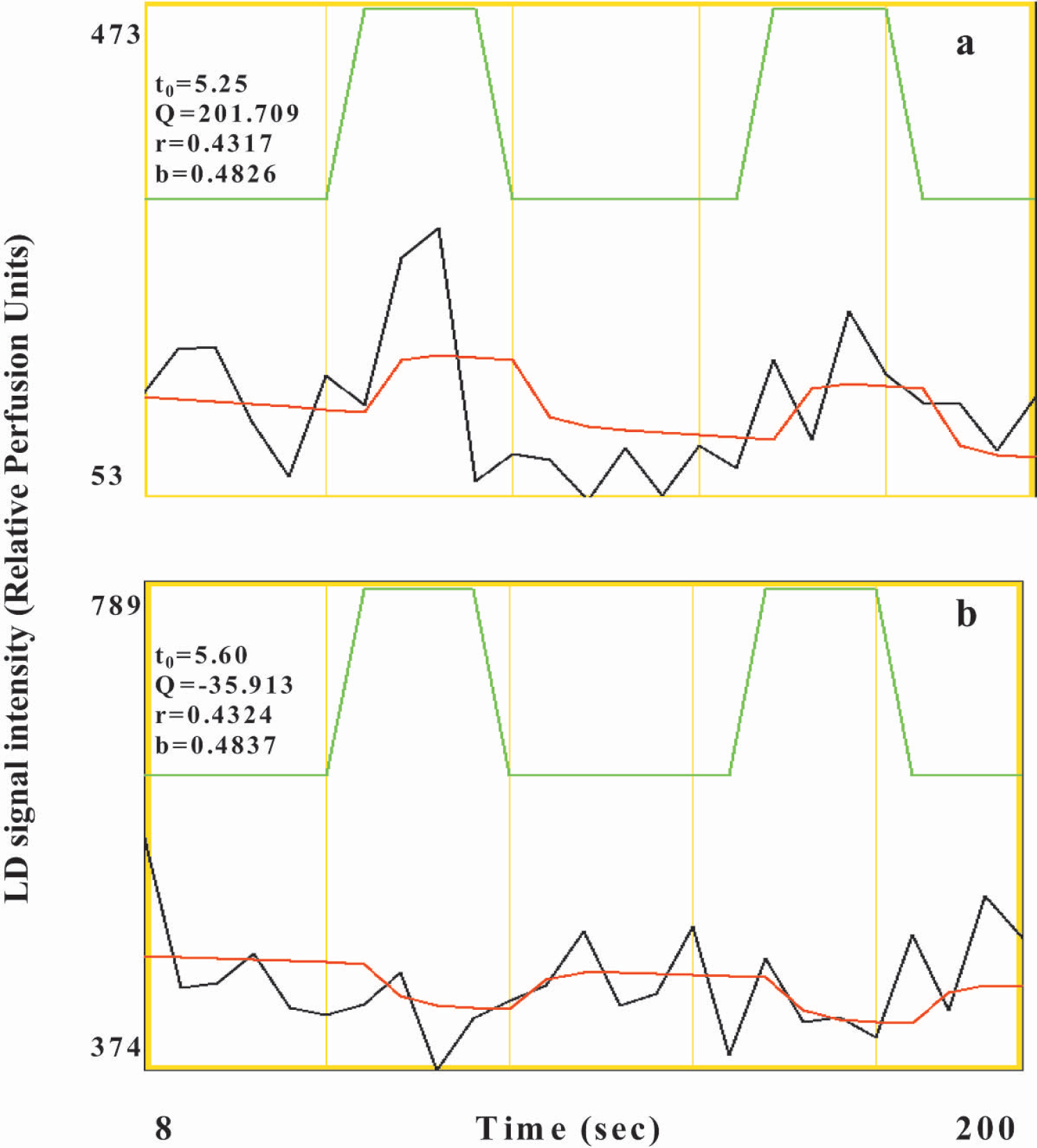
Representative laser-Doppler (LD) signal time course (black), nonlinear fit function (red), reference waveform (green), and the associated fit constants from typical pixels in the contralateral cortex of an anesthetized rat showing a positive response (
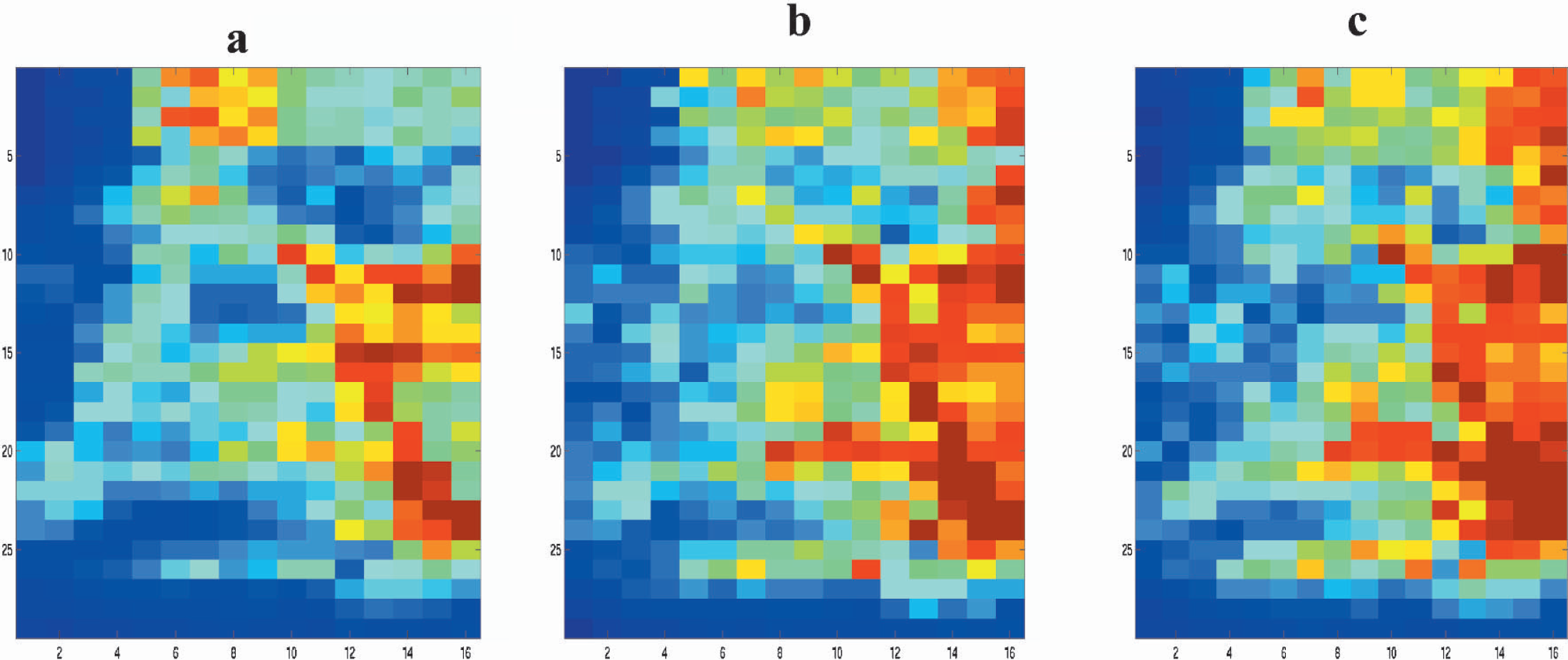
Effect of hypercapnia on the laser-Doppler flux in the somatosensory cortex. (
DISCUSSION
A variety of presently available imaging techniques such as positron emission tomography, functional magnetic resonance imaging, and optical imaging of intrinsic signal have been used to evaluate the mechanisms and localization of neural activation (Ances et al., 1999; Hess et al., 2000; Jones et al., 2001; Vanzetta and Grinvald, 1999; Yang et al., 1996). These imaging modalities use CMRglc, CBF, CBV, or deoxygenated hemoglobin HbR changes to obtain functional maps indicative of neuronal activity and are often used in combination to study the dynamics of neurovascular coupling (Hess et al., 2000; Jones et al., 2001; Malonek et al., 1997). LDI is one such technique where regional hemodynamic changes can be measured from the Doppler shift of the scattered beam of laser directed onto a particular area of tissue to be imaged. In the present study, the spatiotemporal characteristics of CBF change during whisker stimulation were measured in the rat somatosensory cortex using LDI. Our results show simultaneous positive and negative LDI signal changes in the somatosensory barrel field.
Priming is a form of implicit memory that manifests as a facilitation of reprocessing recently experienced stimuli. Thus, an increased efficiency of a given mental process decreases the activity required, which is called the “priming” hypothesis. Active mental processes during rest periods may inflate the estimated baseline signal. Thus, BOLD and flow responses can be positive under awake conditions and negative to the same stimuli in the same subject under certain physiologic conditions such as slow-wave sleep or sedation (Altman and Bernal, 2001; Born et al., 2002). The negative BOLD response is related to decreases in rCBF in the activated areas and hypothesized to be due to certain energy-consuming cognitive processes active during sleep conditions that cease during stimulus presentation (Born et al., 2002). This is called the “baserate” hypothesis, which predicts a decrease in neuronal activity prior to any change in rCBF. The hemodynamic phenomenon like “perfusion-steal” cannot account for negative BOLD or flow responses in conditions that support the priming or the baserate hypothesis. Such a phenomenon is possible when regions adjoining positive signal shifts show a simultaneous negative response. Negative BOLD signal changes have been shown to be accompanied by an increase in spike activity, and it has also been proposed that blood stealing may occur from less active regions into demanding areas adjacent to the less active regions (Raichle, 1998). This mechanism may occur actively or passively by constricting one branch of a feeding artery and dilating another, which has been suggested for the barrel cortex in rodents (Woolsey et al., 1996). If such a process exists, it is expected that the dynamics of the CBF in the region showing a decrease will be temporally coherent to the CBF dynamics in the region showing an increase. From our results, negative CBF responses almost always occurred in regions adjoining positive responses and were temporally coherent (Table 1), lending support to the perfusion-steal phenomenon. Further, no negative responses were observed in the microvascular regions, suggesting that a reduction in neuronal activity may have led to the signal decrease.
Because the vascular architecture over the cortex varied across animals, averaging of images across animals was avoided. The spatial pattern of activation in response to whisker stimulation could be determined from a single experiment by correlation analysis of the signal time series. Test-retest studies showed the spatial extent of activation to be highly reproducible within a single animal. The spatial location of the center of activation observed in the present study was 2.2 mm posterior and 6 mm lateral to the bregma. This is in agreement with previous electrophysiologic studies and somatosensory cortical maps in stereotaxic coordinates (Chapin and Lin, 1984; Dowling et al., 1996; Hall and Lindholm, 1974; Rousche et al., 1999). The amplitude and temporal characteristics of the signal change in response to whisker stimulation using LDI was similar to that observed by earlier workers using a single LDF probe (Gerrits et al., 1998). In the present study, the spatial resolution was high enough (206 μm) to resolve macroscopic and intermediate blood vessels. Hence, the pixel volume from low flux regions represented the activation-induced tissue flow change with minimal contamination from macroscopic blood vessels. Positive responses originated primarily from pixels with intermediate to low baseline flux and distinctly away from macroscopic blood vessels. The percent CBF change from pixels over large vessels was smaller than that obtained over intermediate and small vessels. This result is in concurrence with that obtained earlier using the rat forepaw model at a relatively lower spatial resolution (Ances et al., 1999); further, our data show that at high spatial resolution, activation-induced CBF change is indeed negligible in large pial vessels, as assumed by earlier workers (Lauritzen and Fabricius, 1995). Large vascular structures surrounding the activated regions form certain definite geometries that seem to demarcate the whisker barrel region in the resting flux images. In all cases, activity in response to whisker stimulation almost always occurred within these large vascular structures. Such unique vascular geometries have earlier been used to locate the whisker barrel region in the exposed cortex (Chapin and Lin 1984; Dowling et al., 1996). The vascular structure in the whisker barrel cortex contains large superficial arteries that penetrate radially into the cortical layers. These arteries usually feed blood to smaller vasculature in the deeper cortical layers. Cox et al. (1993) showed that large vessels were present only in the superficial layers of the cortex (>150 μm), whereas the barrel cortex at a depth of about 300 μm had vessels with diameter in the range of less than 50 μm. These superficial arteries showing high baseline LDI flux values were clearly resolved in the images and were highly reactive to hypercapnia (Fig. 6). Almost 90% of the high flux pixels were very sensitive to hypercapnia. These included the pixels that showed negative CBF response to whisker stimulation, indicating that they were predominantly arteries.
Activation was always accompanied by a sustained negative flux change in certain large vessels (Fig. 2). The negative CBF change was reproducible spatially within experiments in all animals imaged. The average distribution of activity in the images over the contralateral cortex show negative flux changes in approximately 10% of the total pixels in the image field of view (Fig. 3c). The average distribution of flux values during rest indicates pixels with high perfusion rates in the range of 800 rpu and above formed approximately 15% to 20% of the total number. Further, pixels showing negative responses during whisker stimulation were primarily from regions with high baseline flux values. These regions also had the highest flux change during hypercapnia (Fig. 6). Our results suggest that during stimulation, a net decrease in blood flow results in certain large vessels that may possibly feed arterioles and capillaries in the activated region and not a confounding effect of anesthesia. Heterogeneity of capillary flow has been shown in many situations, which could cancel the net flow change. We therefore analyzed the stability of LD flux in high and low flux pixels. The laser-Doppler flux was stable throughout the measurement time in both low-flux and high-flux pixels (Fig. 4e). Further, with unchanged physiologic setting, the flux did not fluctuate (i.e., low-flow pixels remained low-flow pixels and high-flow pixels remained high-flow pixels). Thus, microcirculatory heterogeneity does not seem to contribute to the negative flow responses. These results are consistent with those of Harel et al. (2002), who observed a positive BOLD and CBV change in the primary visual cortex during visual stimulation, whereas a prolonged negative BOLD and CBV change was detected in the adjacent suprasylvian gyrus containing higher-order visual areas. Their results support the notion that reallocation of cortical blood resources could overcome a local demand for increased CBF induced by increased neural activity. Shmuel et al. (2002) have also observed a similar coupling in negative and positive BOLD signal responses in the human visual cortex. By combining BOLD and CBF data, they modeled the oxygen consumption in the cortex to imply that a reduction in neuronal activity in addition to an independent hemodynamic component leads to the negative response. Our study did not correlate neuronal activity with the hemodynamic response. Further studies with simultaneous recording of increased or decreased neuronal activity with CBF are required to precisely answer the neuronal control of the negative CBF change.
Trial-to-trial variations are often observed in optical responses to the same whisker deflection stimulus that cannot be controlled (Tanaka et al., 2000). The same group (Yazawa et al., 2001) later showed that these trial-to-trial variations have a physiologic origin and reflect some features of the functional organization of the whisker barrel cortex. The neuronal activity in a single barrel is heavily influenced by activity from neighboring barrels in the mature cortex. Thus, neural responses will be prone to variations between each trial. However, variation in the activation-induced response would also depend on the signal-to-noise ratio with larger variability induced by a lesser signal-to-noise ratio. We compared the activation pattern in response to whisker stimulation between two random trials in the same animal. Each combination showed high spatial variability with approximately 12% ± 4% mean overlap between the positively activated and 21% ± 7% overlap in the negatively activated pixels. The coefficient of variation for various two-trial combinations for the positive and the negative responses were 37.0% and 18.3%, respectively. Although a component of the high variability could have been induced by a poor signal-to-noise ratio, approximately 2 in the present case, the variability across trials in the negative response was significantly lesser when compared to the positive response. This occurred despite a relatively lower SNR in the negatively responding pixels when compared with those showing a positive response. This indicates that the negative response is less likely to be under direct neuronal control.
The origin of negative CBF changes from a few selective medium and large vessels, and its spatial proximity, temporal coherence to the positive response, and lesser trial-to trial variability strongly suggest a decrease in CBF in feeder vessels to responding arterioles and capillaries in the activated regions.
Footnotes
Acknowledgements
The authors thank Dr. A. G. Hudetz for valuable suggestions, Dr. V. E. Sharoyan for assistance with the experiments, and Timothy Lee at Moor Instruments, Inc., for assisting with the use of the Moor LDI device.