Abstract
Post-ischemia angiogenesis and vascular plasticity help to restore blood flow to ischemic tissue and likely benefit long-term functional recovery. Physical activity has been shown to cause morphologic and functional effects, including promoting angiogenesis in normal or injured animals. A therapeutic effect of peripheral activity on central angiogenesis after cerebral ischemia, however, has not been studied. In the present study of whisker-barrel cortex ischemia in the mouse model, we tested the hypothesis that enhancing whisker activity and sensory input to the ischemic barrel cortex might promote post-ischemia cerebral angiogenesis. Three days after focal ischemia in adult mice, the whiskers corresponding to the ischemic barrel cortex were stimulated by two methods: (1) whiskers on the right side of the mouse face were trimmed away, so the left whiskers were overused by the animals, (2) left whiskers were manually stimulated to enhance input signals to the ischemic barrel cortex. Western blot analysis showed that whisker stimulation increased expression of the angiogenic factors vascular endothelial growth factor, basic fibroblast growth factor, Tie-1, angiopoietin-2 (Ang-2), and possibly Ang-1. Co-immunostaining with markers for proliferation (5-bromo-2-deoxyuridine (BrdU)) and vascular endothelial cells (Glut-1/CD-31) identified vessel proliferation in the penumbra region. Whisker stimulation increased BrdU-positive endothelial cells and vessels in this region 7 and 14 days after ischemia. Whisker stimulation also attenuated endothelial cell death and increased local cerebral blood flow. Our data suggest that appropriately enhanced peripheral activity and afferent signals to the ischemic cortex can promote post-ischemic angiogenesis, which may imply beneficial effects of specific physical therapy on long-term recovery from ischemic stroke.
Introduction
Stroke is a leading cause of death as well as disability in the United States and other countries (American Heart Association, Disease and Stroke Statistics—2005 Update, 2005). Ischemic strokes account for about 88% of all strokes. Cerebral ischemia results in massive cell death, which translates to paralysis, aphasia, and the inability to perform activities of daily living.
Angiogenesis, the formation of new blood vessels from the pre-existing vascular network, is a process likely involved in tissue repair after stroke. This process of vascular growth is essential in development, reproduction, and wound healing; it requires proliferation, activation, and migration of endothelial cells (Arsic et al, 2003; Ellsworth et al, 2003; Folkman and Shing, 1992). Several growth factors and their receptors have been implicated in controlling the highly regulated angiogenic process and are collectively known as angiogenic factors. Vascular endothelial growth factor (VEGF) and fibroblast growth factor (FGF) are well-known angiogenic factors, whereas the angiopoietins (Ang-1 and Ang-2) and their receptors (Tie-1 and Tie-2 receptors) are needed for maturation and stabilization of the newly formed vessels (Eliceiri and Cheresh, 2001; North et al, 2005; Scott et al, 2005; Zhang et al, 2002a, b).
Adult blood vessels undergo remodeling as a direct response to tissue demands. In ischemia, the ischemic tissue depends on collateral blood flow as an alternate blood supply immediately after injury (Hayashi et al, 2003; Risau, 1997). Angiogenesis, or proliferation of vascular endothelial cells, provides an enduring mechanism of restoring local blood flow and functional activity after ischemia. In ischemic stroke patients, the number of new vessels surrounding injured tissue correlated to longer survival (Krupinski et al, 1994). We have shown that microvascular proliferation and remodeling occur after cerebral ischemia in the rat barrel cortex, which increased blood flow and stimulated collateral growth (Wei et al, 2001). Recent animal studies have linked increased angiogenesis to improved performance in neurologic and behavioral tests (Kaya et al, 2005; Shyu et al, 2004; Sugiura et al, 2005; Zhang et al, 2005).
Compelling evidence suggests that peripheral stimuli and activity have significant ‘use-dependent’ impacts on the functional and morphologic alterations in the central nervous system and on the outcome of central nervous system disorders. For example, rearing and experience of young or adult animals in ‘enriched’ environments change cortical thickness and synaptic density and also increase capillary branches and capillary surface areas in the visual cortex (Sirevaag et al, 1988) and cerebellum (Isaacs et al, 1992). Use of the forelimbs is essential for an increase in the number of pyramidal cell dendritic branches contralateral to lesions in somatomotor cortex (Jones and Schallert, 1994; Kozlowski et al, 1996). Several weeks of an ‘enriched’ motor environment improved the behavioral motor tests of rats after middle cerebral artery (MCA) ligations (Johansson and Ohlsson, 1996). A recent study showed that whisker stimulation enhanced functional recovery from a unilateral ischemic lesion to the posteromedial barrel subfield that impairs the ability of rats to discriminate textures using their whiskers (Hoffman et al, 2003). In peripheral injury models, electrical stimulation, via several mechanisms including angiogenesis, has been suggested as a clinical approach for enhanced healing from wounds caused by venous insufficiency, diabetic neuropathy, and ischemia (Kloth, 2005). A modest regimen of electrical stimulation accelerates neovascularization, enhances angiogenesis, and improves the healing process in muscle tissues (Chekanov et al, 2002; Sheikh et al, 2005). Angiogenesis has also been reported in response to motor exercise in normal animals (Black et al, 1990; Kleim et al, 2002; Swain et al, 2003). A very recent study showed that prior exercise could pre-condition rats and reduce ischemic brain damage by enhanced neurotrophin expression and regional angiogenesis (Ding et al, 2005). However, a therapeutic effect of peripheral stimulation/activity on post-ischemic cerebral angiogenesis has not been explored.
The rodent whisker-barrel cortex has been targeted as an ischemic stroke model with the advantages of the defined whisker sensory pathway and feasible identification of corresponding morphologic and functional changes. Rats and mice use their whiskers for a variety of exploratory tasks including location of objects and discrimination of texture. Each barrel corresponds to a specified whisker on the contralateral face of the animal and receives sensory input upon activity or stimulation of that whisker (Woolsey et al, 1975; Woolsey and Van der Loos, 1970). Whiskers can be mechanically stimulated, either individually or in groups, to evoke strong responses in barrels (Simons, 1985). Clipping one side of whiskers forces the animal to intensify the use of the remaining whiskers for exploratory functions (Armstrong-James et al, 1994; McCasland and Woolsey, 1988). In the present investigation, peripheral stimulation-induced angiogenesis in the ischemic cortex was studied using the mouse model of whisker-barrel cortex stroke, modified from our previous rat model (Wei et al, 1995). We tested how two types of therapeutic whisker stimuli, forced overuse and rhythmic manual sweeps, may affect cell death, expression of angiogenic factors, proliferation of endothelial cells, formation of microvessels, and local blood flow in the corresponding post-ischemic barrel cortex of the mouse brain.
Materials and methods
Animals and Focal Ischemia of the Whisker-Barrel Cortex
The focal barrel cortex ischemic stroke was similarly induced as previously described in rats (Wei et al, 1995), with modifications for a few differences in the surgery procedures and anatomy in mice. Adult C57/BL6 mice weighing between 25 and 30 g and in the age range of 2.5 to 3 months were anesthetized with isoflourane. Focal ischemia of the barrel cortex was induced by permanent occlusion of 2 to 3 branches of the MCA supplying the right whisker barrel cortex. This was accompanied by 7-mins occlusion of the common carotid artery. During surgery and recovery periods, body temperature was monitored using a rectal probe and maintained at 37°C with a homoeothermic blanket in a ventilated incubator. The right femoral artery was cannulated in some of the animals for monitoring arterial blood pressure and for obtaining blood samples for arterial blood gases. Blood pressure was monitored with a Digi-Med blood pressure analyzer (Micro-Med Inc., Tustin, CA, USA). Arterial blood gases were determined before ischemia and 1 to 14 days after the onset of ischemia with a blood gas analyzer (Ciba Corning, Ramsey, MN, USA). Mice were killed by decapitation 7 or 14 days after ischemic stroke. The brain was immediately removed and preserved in OCT compound at −80°C until further processing. Before and after the surgery, animals were housed in standard mice cages (27 × 17 × 13 cm), at 3 to 4 mice per cage, having free access to food and water. The animal protocol was approved by the Institutional Animal Care and Use Committee (IACUC) at MUSC. Animal procedures followed institutional guidelines, which meet or exceed NIH standards.
Whisker Stimulation
Three days after ischemia, mice were randomly divided into three groups: (1) ischemic stroke-only group (control), (2) a group in which all right whiskers were trimmed to the skin, so the animals had to rely on their left side whiskers that have neuronal pathways across the brain stem to the ischemic barrel cortex on the right hemisphere, and (3) a group of mice that received manual stimulation of their left whiskers (Figure 1). The left whiskers in the third group were swiped in a rostro-caudal direction using a stick at a frequency of about 140 strokes per minute (∼ 2.3 Hz) for 15 mins, performed three times a day. During periods of stimulation, animals behaved normally and were allowed to move freely in the cage. Other than trimming and manual stimulation, no additional environmental manipulation was applied.
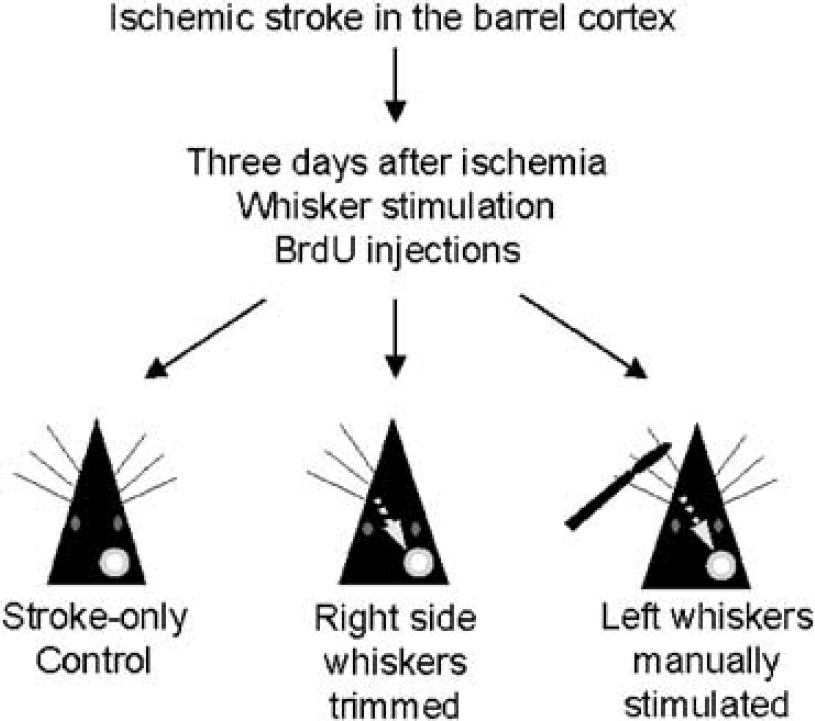
Experimental groups and whisker stimulation. Adult mice were subjected to permanent occlusion of MCA branches supplying the right barrel cortex. Three days after ischemia, all mice received BrdU injection (50 mg/kg, intraperitoneally, once/day). The mice were randomly divided into three groups: (1) ischemic stroke-only group as the control, (2) all right whiskers trimmed to enhance the whisker activity of the left face whiskers (trimmed group), and (3) direct manual stimulation of left whiskers for 15 mins at about 140 strokes per minute, three times per day (manually stimulated group). The left whiskers have corresponding neuronal pathways that, via the interneurons in the brain stem, lead to the right barrel cortex. The white circle represents the ischemic barrel cortex in the right hemisphere.
5-Bromo-2′-Deoxyuridine Administration
For labeling of cell proliferation, 5-bromo-2′-deoxyuridine (BrdU) (Sigma, St Louis, MO, USA) was administered to animals at a dosage of 50 mg/kg body weight by intraperitoneal injection. Injections began on day 3 after ischemia and continued once daily until killing.
TdT-Mediated dUTP Nick End Labeling Staining
A TdT-mediated dUTP nick end labeling (TUNEL) staining kit (DeadEnd™ Fluorometric TUNEL system, Promega, Madison, WI, USA) was used to assess cell death by catalytically incorporating fluorenscein-12-dUTP at 3′-OH DNA ends using the terminal deoxynucleotidyl transferase and recombinant enzyme. After fixing and pretreatment with ethanol/acetic acid and triton, the brain sections were incubated in an equilibration buffer as described in the kit. The TdT enzyme and nucleotide mix were then added at proportions specified in the kit for 75 mins at room temperature. The slides were washed with the provided wash buffer. To identify endothelial cell death, sections were then incubated with the Glut-1 primary antibody overnight. After phosphate-buffered saline (PBS) washes and incubation with Cy3-conjugated anti-rabbit IgG, brain slides were mounted for viewing and cell counting.
Microvessel Staining
Fresh frozen brains were sliced into coronal sections at 14-μm thick using a cryostat vibratome (Ultapro 5000, St Louis, MO, USA). Sections were completely air dried and subsequently fixed in 10% buffered formalin phosphate (Fisher Scientific, Pittsburgh, PA, USA). Brain sections were then processed in an ethanol:acetic acid (2:1) solution at −20°C for 10 mins, washed three times in PBS solution at pH 7.4, and incubated in 0.2% Triton X-100 for 5 mins. Slides were blocked in 1% fish gelatin (Sigma, St Louis, MO, USA) in PBS for 1 h at room temperature. Incubation with a primary antibody diluted in PBS was performed overnight at 4°C. The rabbit anti-Glut-1 (1:1,000, Chemicon, Temecula, CA, USA) and rat anti-CD-31 (1:400, Cymbus Biotechnology, Eastleigh, UK) antibodies were used to label endothelial cells. Primary antibodies were removed by PBS washes and replaced by secondary antibodies Alexa Fluor 488 anti-rabbit IgG and anti-rat IgG (1:200, Molecular Probes, Carlsbad, CA, USA) for 1 h at room temperature before rinsing again in PBS.
For double staining with BrdU, brain slides were postfixed in 10% buffered formalin phosphate and washed in PBS. Slides were then treated with 100% methanol and allowed to air dry. After re-wetting in PBS, sections were incubated in 2 N HCl for 1 h at 37°C. Sections were treated with 0.1 mol/L borate buffer at pH 8.4 for 10 mins, followed by a 45 mins incubation in 0.2% Triton X-100. After blocking in 1% fish gelatin, slides were incubated with mouse anti-BrdU (1:1,000, Roche, Basel, Switzerland) or rat anti-BrdU (1:400, Abcam, Cambridge, UK) antibodies overnight at 4°C followed by rinsing in PBS before being incubated with Cy3-conjugated goat antimouse IgG (1:500; Jackson ImmunoResearch, West Grove, PA, USA) or Texas Red goat anti-rat IgG (1:400, Abcam, Cambridge, UK) antibodies for 1 h at room temperature. After a final PBS rinsing, slides were mounted with ProLong Antifade mounting medium (Molecular Probes, Carlsbad, CA, USA) for photography and imaging analysis.
Cytochrome Oxidase Histology
Barrel pattern on the cortex was examined using the mitochondrial enzyme cytochrome oxidase (Dowling et al, 1996). Briefly, following videomicroscopy, mice were perfused with warm heparinized saline followed by 10% formalin for 15 mins. The brains were removed and immersed for 24 h in periodate-lysine-paraformaldehyde buffered to pH 7.4, then transferred to pariodate-lysine-paraformaldehyde 30% sucrose solution until they sank. The brains were sectioned parallel to the pial surface into 50 mm slices, and were stained for cytochrome oxidase activity. Serial sections were mounted on chromealum subbed slides, cleared in xylene, and coverslipped.
Western Blot Analysis of Angiogenic Factors
Tissue samples were taken from the penumbra region of brain and proteins were extracted using the NE-PER Nuclear and Cytoplasmic Extraction Kit (Pierce Biotechnology, Rockford, IL, USA). Protein concentration of each sample was determined using the Bicinchoninic Acid Assay (Sigma, St Louis, MO, USA). Proteins from each sample (50 μg) were separated by SDS-polyacrylamide gel electrophoresis in a Hoefer Mini-Gel system (Amersham Biosciences, Piscataway, NJ, USA) and transferred in the Hoefer Transfer Tank (Amersham Biosciences, Piscataway, NJ, USA) to a PVDF membrane (BioRad, Hercules, CA, USA). Membranes were blocked in 7% evaporated milk diluted in Tris-buffered saline containing 0.1% Tween 20 (TBS-T) at room temperature for at least 2 h, and then incubated with one of the following antibodies: VEGF (Santa Cruz, Santa Cruz, CA, USA), Tie-1 (Santa Cruz, Santa Cruz, CA, USA), Tie-2 (Santa Cruz, Santa Cruz, CA, USA), basic fibroblast growth factor (bFGF) (Santa Cruz, Santa Cruz, CA, USA), Ang-1 (Chemicon, Temecula, CA, USA), and Ang-2 (Chemicon, Temecula, CA, USA). A mouse β-actin antibody (Sigma, St Louis, MO, USA) was used for protein loading control. After primary antibody incubation, membranes were washed with TBS-T and incubated with alkaline phosphatase-conjugated anti-mouse or anti-rabbit IgG (Promega, Madison, WI, USA) antibodies for 2 h at room temperature. Finally, membranes were washed with TBS-T followed by three washes with TBS and signal was detected by the addition of 5-bromo-4-chloro-3-indolylphosphate/nitroblue tetrazolium (BCIP/NBT) solution (Sigma, St Louis, MO, USA).
Stereological Cell and Vessel Counting
For systematic random sampling in design-based stereological cell counting, every fifth brain section (70 mm apart) across the entire region of interest were counted. For multistage random sampling, six fields per brain section were randomly chosen under × 40 magnification of a light microscope or in confocal images (see figure legends for animal numbers of each experiment).
Laser Doppler Measurement of Local Cerebral Blood Flow
Cerebral blood flow was measured using laser-Doppler flowmetry (PeriFlux System 5000-PF5010 LDPM unit, Perimed, Stockholm, Sweden). The tip of a flexible 0.5 fiber optic probe was pointed to a selected MCA branch visualized through a cranial window with the dura intact over the right somatosensory cortex (see Figure 7). Visualization of the arteries was achieved by means of angiography and videomicroscopy (Wei et al, 2001). Briefly, under isoflourane anesthesia and epifluorescent illumination, 0.05 mL boluses of 1.3 mmol/L fluorescein isothiocyanate were injected into a branch of the right jugular vein. Dye transits through the arteries, capillaries, and veins of the cortex were observed and recorded through the microscope under a × 4 objective with a Quantex ICCD videocamera set to manual black and fixed gain settings on VHS tape. Vascular images were used for identifying blood vessels and processed with NIH Image 1.61.
Local cerebral blood flow (LCBF) was measured before, during, and after ischemic surgery. The cranial windows could be reopened after ischemia for fluorescein isothiocyanate angiographic videomicroscopy and LCBF measurements. Changes in LCBF after MCA occlusion were expressed as percentages of baseline values before ischemia.
Statistical Analysis
All analyses were performed using SAS, version 9.1. Analyzed data were graphed using GraphPad Prism 4.0. As a result of the hierarchical nested experimental design, the data were clustered. To properly account for this correlation, a Poisson regression using generalized estimating equations was used with the natural log of the count data for each slide, clustering on mouse and slide within mouse; counts within a slide were considered approximately independent of one another. For the time-series data (examining the counts at days 7 and 14), a profile analysis approach was used, which included time in the analysis. To assess whether the change in counts from day 7 to 14 was significantly different between treatment group and control, an interaction term was created as the product of the time variable and treatment group; the significance of these interaction effects in the final analysis was then assessed. Assessing the total number of vessels per field, proliferating vessels per field and cell death were considered as separate outcomes, so an a level of 0.05 was used for each. To account for multiple comparisons, an a level of 0.025 was used within each of these outcomes (Dunnett's procedure for all pairwise comparisons with control). Bar figures show the mean value and standard deviation (s.d.).
Results
Barrel Ischemic Stroke and Stimulation of the Whisker-Barrel Pathway
The sensory barrel cortex on the right hemisphere was subjected to permanent ligation of the MCA branches supplying the region (Wei et al, 2001, 1995). The focal ischemia resulted in restricted infarct and distinctive cell death in the targeted barrel cortex 24 h later. To test the effects of increased whisker activity on angiogenesis in the ischemic cortex region, animals were divided into three groups 3 days after ischemia. In addition to a stroke-only control group, mice in two other groups received additional manipulation to enhance whisker activity on the left face, sending afferent signals to the right side (ischemic) barrel cortex via the thalamocortical pathway. In one experimental group, all right side whiskers were trimmed away, so the animals had to solely rely on their left whiskers for searching. Previous investigations showed that whisker trimming increased activity in spared cortex within hours of trimming (McCasland and Woolsey, 1988), and single-unit recordings showed increased synaptic activity up to 30 days after trimming (Armstrong-James et al, 1994). In the third group, no trimming was performed but the left whiskers were directly stimulated by rhythmic sweeps at a frequency of 140 strokes per minute for 15 mins, performed three times a day until killing (Figure 1).
Barrel cortex ischemia induced a restrictive infarct in the right side of the brain cortex 24 h after ischemia (Figures 2A and 2B). Cytochrome oxidase staining revealed the disappearance of most barrel structures in the ischemic barrel cortex (Figures 2C and 2D). Three to seven days after ischemia, many TUNEL-positive cells were observed in the ischemic core and penumbra regions (Figure 2B). Double labeling with Glut-1, a specific marker for glucose transporter in brain vascular endothelial cells (Wei et al, 2001), indicated injured endothelial cells (Figures 3A–3F). In the penumbra region, TUNEL-positive endothelial cells were decreased by enhanced whisker activity induced by manual stimulation, although no reduction was seen with the whisker-trimmed group (Figure 3G).
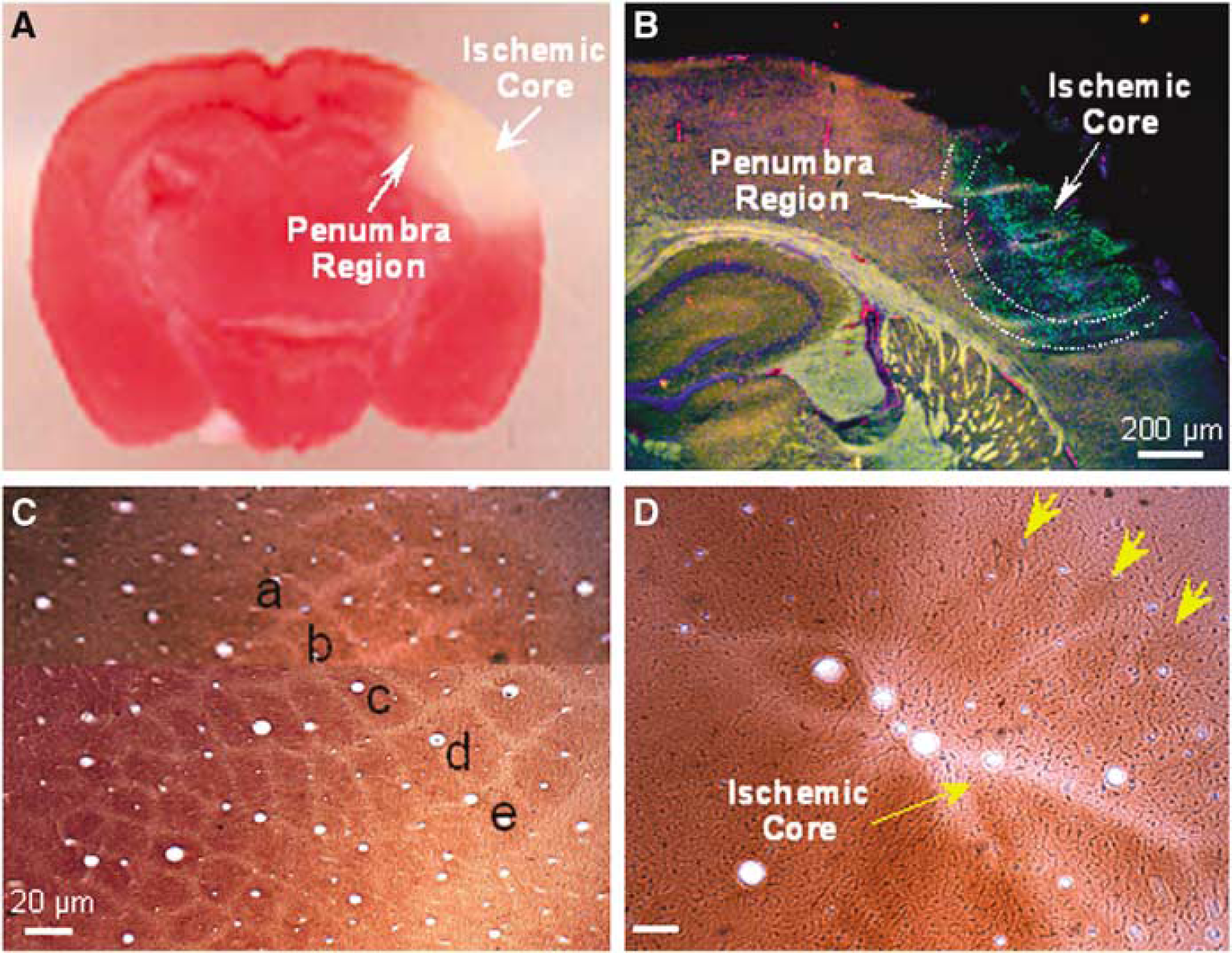
Ischemia-induced barrel cortex infarct formation and cell death. Middle cerebral artery branch ligations induced a focal ischemic infarct and cell death in the barrel cortex. (
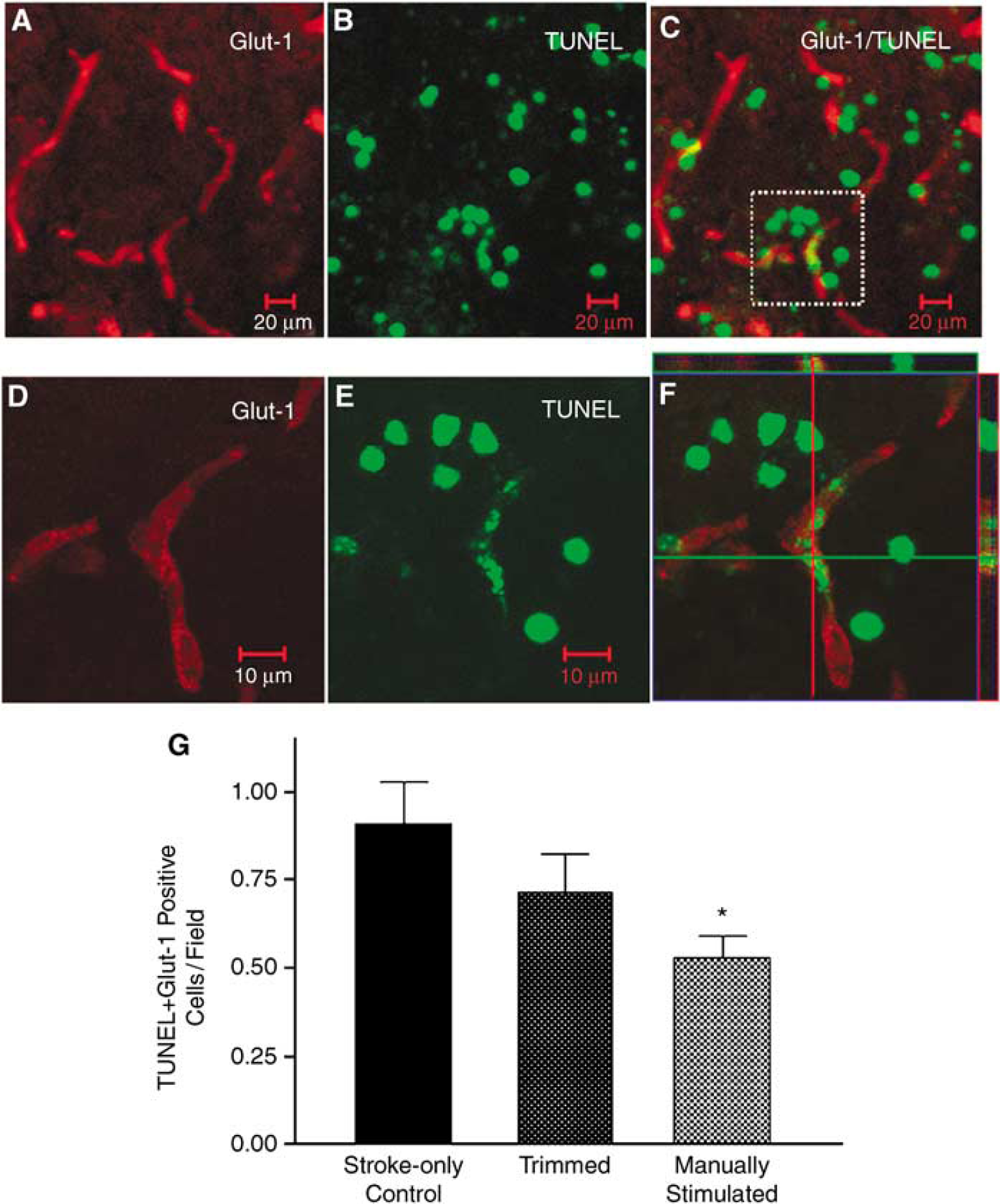
Ischemia-induced endothelial cell death. Using confocal microscopy, endothelial cells were identified using Glut-1 staining and cell death was assessed using TUNEL staining of DNA damage 7 days after ischemia. (
Whisker Stimulation and Angiogenic Factor Expression in the Ischemic Barrel Cortex
Expression of several angiogenic factors, VEGF, bFGF, Ang-1, Ang-2, Tie-1, and Tie-2, in the ischemic core/penumbra region was analyzed 7 days after ischemia by Western blot (Figure 4). Manual stimulation of whiskers led to significant increases of expression of all angiogenic factors tested except Tie-2 and Ang-1 compared with the stroke-only controls (Figure 4). Conversely, whisker trimming increased the expression of Tie-1 and bFGF, but not other factors (Figure 4). In the case of Ang-1 expression, both stimuli showed a noticeable trend of increasing this angiogenic factor, although the changes were not statistically significant (Figure 4).
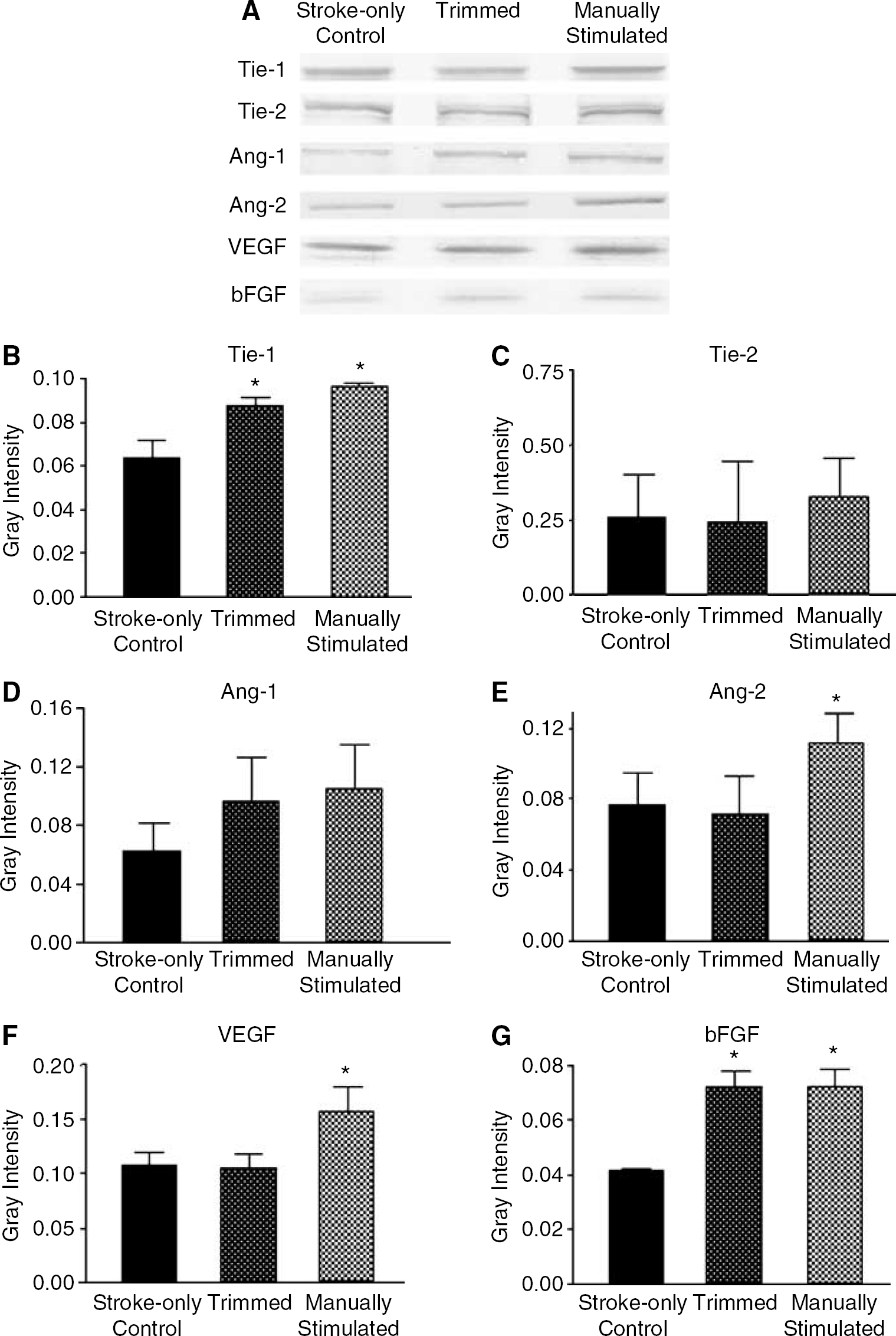
Expression of angiogenic factors in the penumbra region and effect of whisker stimulation. The protein levels of Tie-1, Tie-2, Ang-1, Ang-2, VEGF, and bFGF were detected using Western blot analysis. (
In immunohistochemical assessments 7 days after ischemia, Tie-1 immunoreactivity was barely detectable in the contralateral cortex, whereas positive staining was seen in the ipsilateral cortex including the ischemic core and penumbra regions. Tie-1 and VEGF double labeling almost exclusively stained microvessels but not large vessels (Figures 5A–5C).
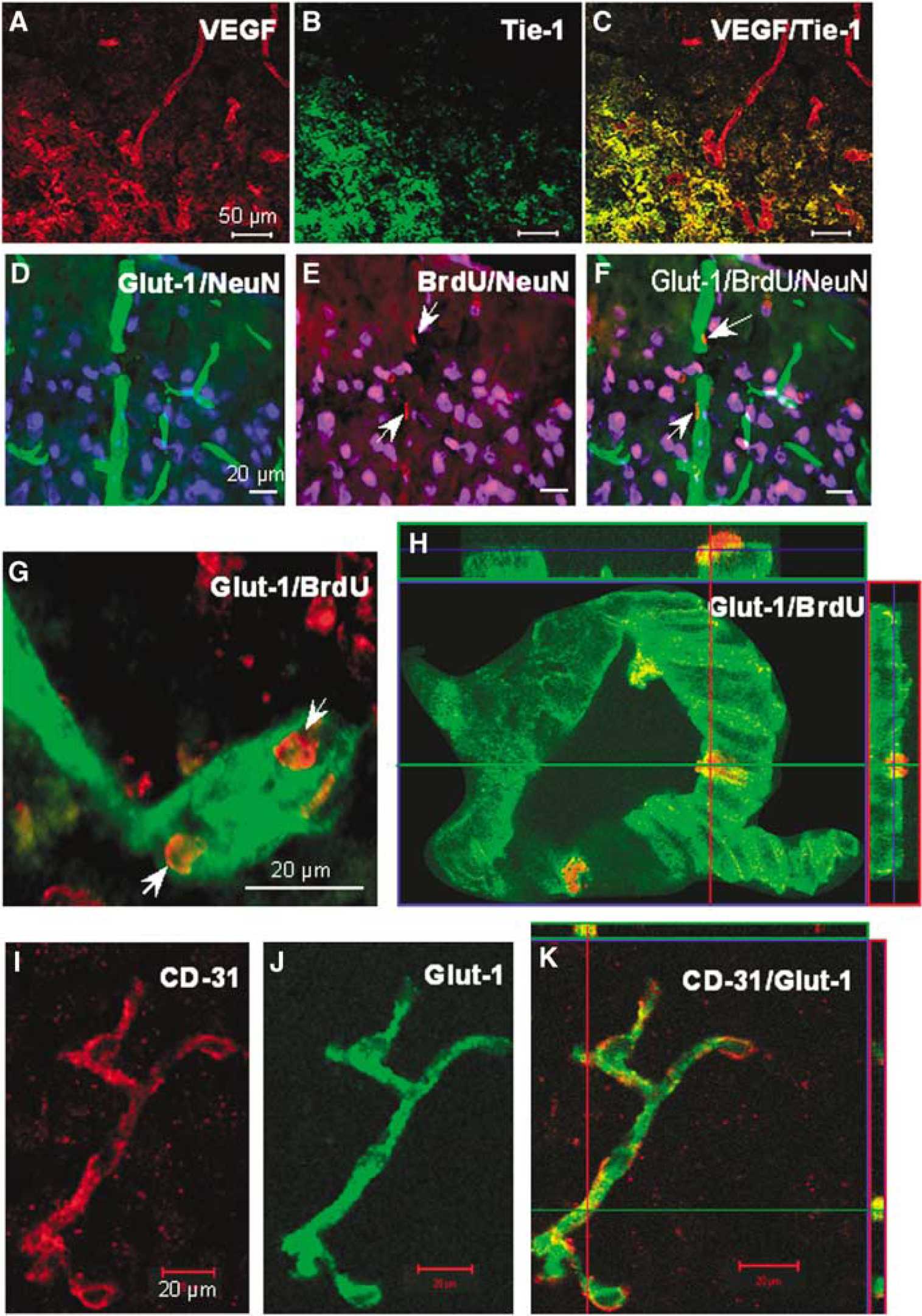
Endothelial cell proliferation and microvessel formation in the ischemic penumbra region. Immunohistochemical staining for angiogenesis and vessel formation 7 days after ischemia. (
Endothelial Cell Proliferation and Vessel Growth After Focal Cerebral Ischemia
Proliferating endothelial cells were detected by double labeling of Glut-1 and the proliferation marker BrdU. Little BrdU labeling was observed in blood vessels in the contralateral cortex or in the normal cortex adjacent to the ischemic penumbra. Some surface vessels and intraparenchymal microvessels in the penumbra expressed BrdU-labeled nuclei 7 and 14 days after ischemia (Figures 5D–5H).
CD-31 is a well-known marker specific for endothelial cells. We performed double labeling with anti-CD-31 and anti-Glut-1 to confirm the specificity of our Glut-1 labeling. Confocal microscopy showed that CD-31 and Glut-1 antibodies indeed labeled identical microvessels of the mouse brain tissue (Figures 5I–5K).
The hyperactivity induced by whisker trimming and manual stimulation increased Glut-1-positive endothelial cells/vessels (Figure 6). To quantify endothelial cell proliferation, Glut-1-positive microvessels co-labeled with BrdU were counted in the penumbra region. At 7 and 14 days after ischemia, both trimmed and manually stimulated groups showed significant increases in endothelial proliferation compared with stroke-only controls (Figure 7A). The total vessel number, indicated by Glut-1-positive staining, was also increased by whisker manipulations (Figure 7B). Interestingly, the vessel number in the penumbra of stroke-only mice increased from 7 to 14 days after ischemia, although the increase was significantly smaller than that in stimulated groups (Figure 7). Consistently, better recovery of LCBF in the stroke region of whisker-stimulated mice was detected 14 days after ischemia compared with that of mice without whisker stimulation (Figure 8). Meanwhile, physiologic variables of mice were not changed during 14-day experiments (Table 1).
Physiologic variables before and after ischemia
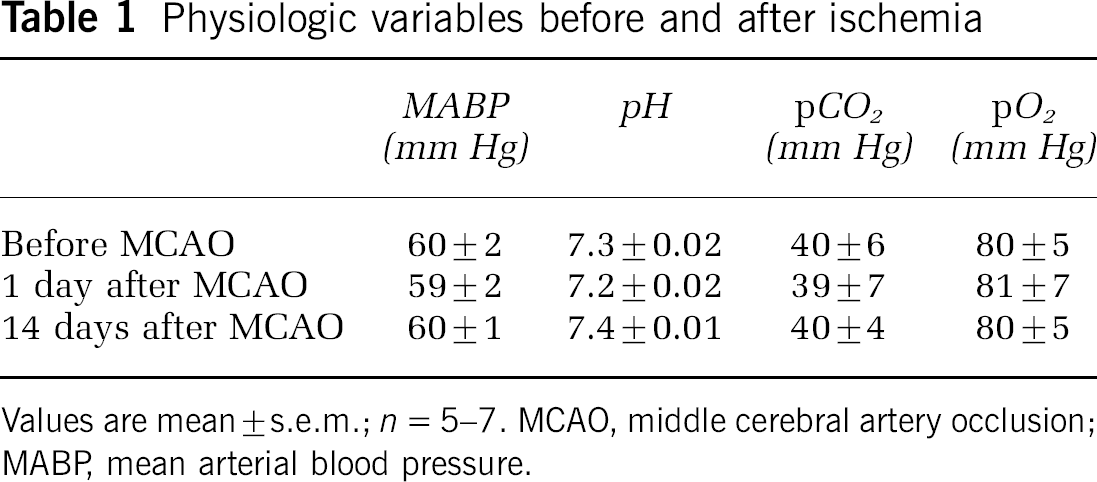
Values are mean ± s.e.m.; n = 5–7. MCAO, middle cerebral artery occlusion; MABP mean arterial blood pressure.
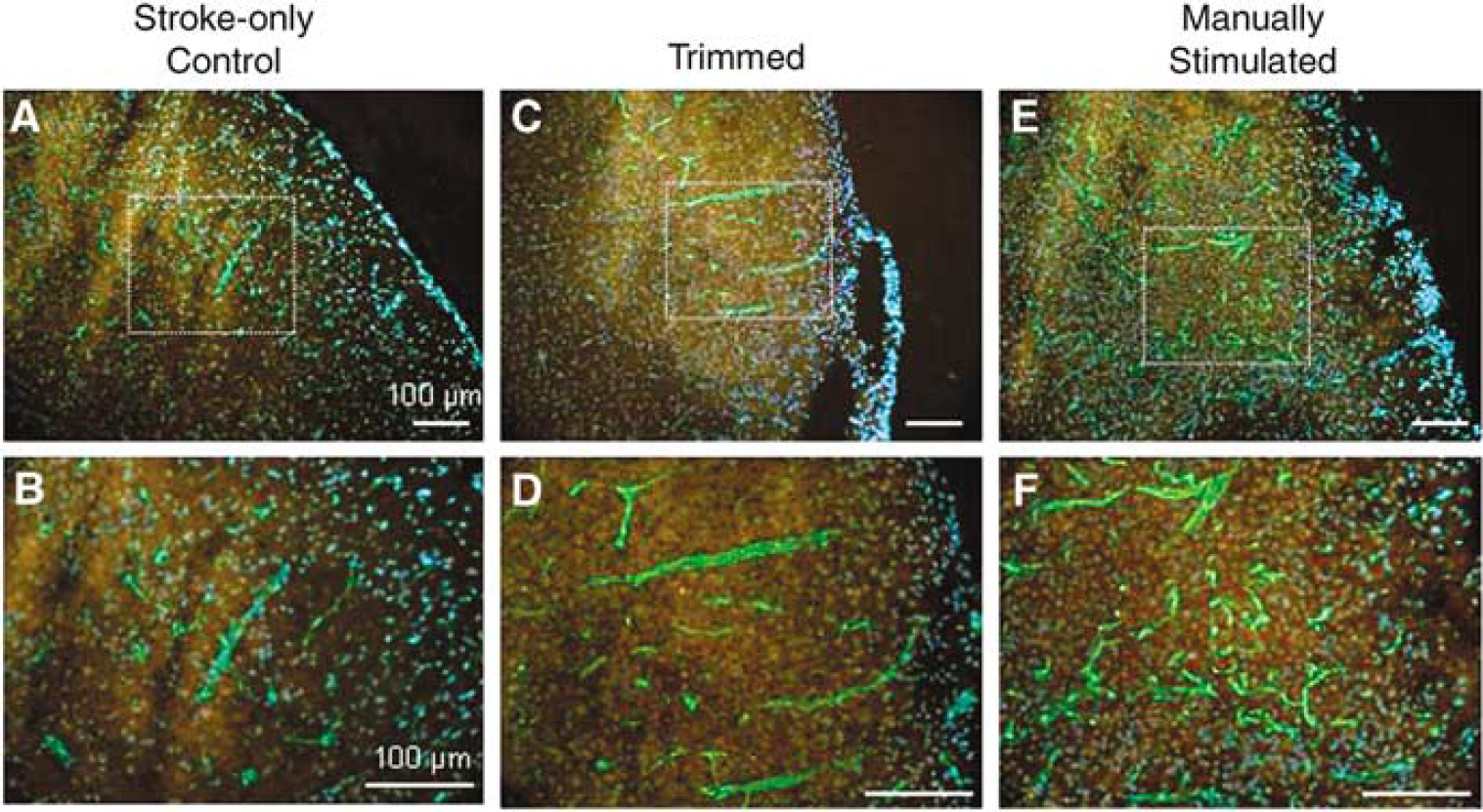
Vessel staining in ischemic penumbra and effect of whisker stimulation. Fluorescence microscopy showed Glut-1 staining (green) of microvessels in the penumbra region 7 days after ischemia. (
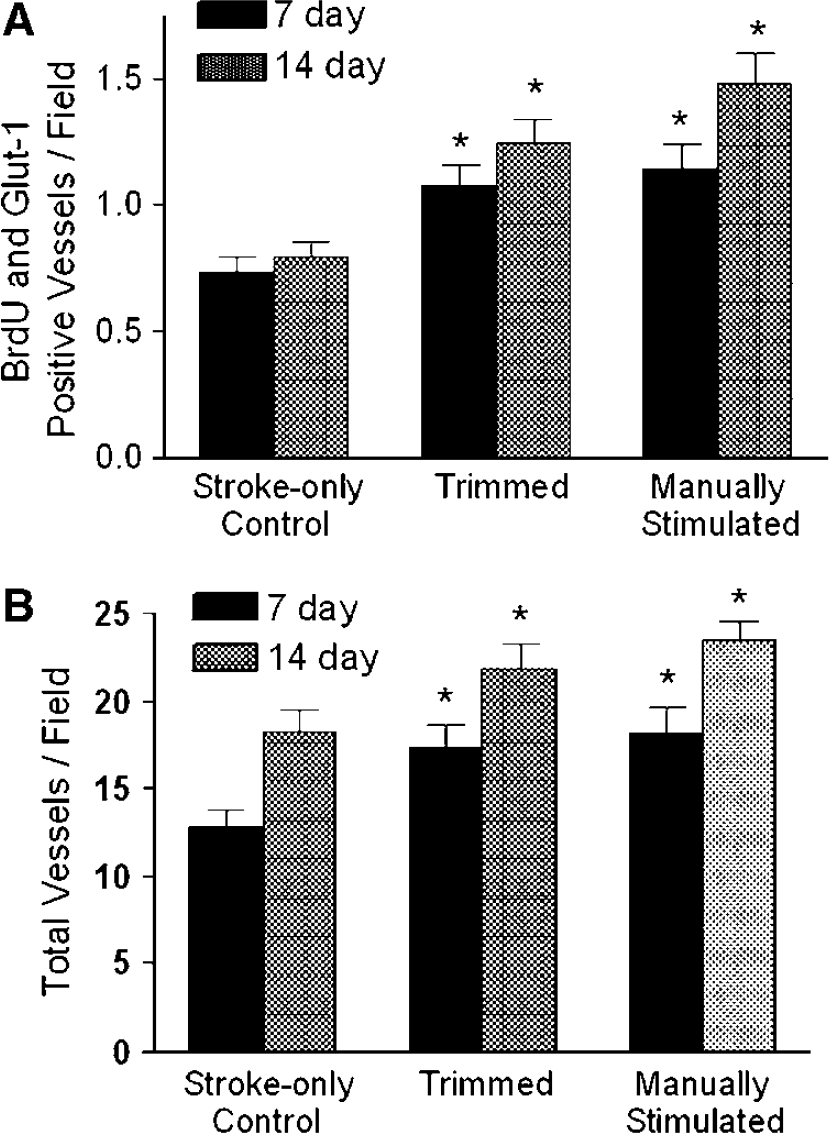
Vessel formation increased by whisker stimulation in the penumbra region. At 7 and 14 days after ischemia, blood vessel proliferation and the number of vessels were increased by whisker stimulation. (
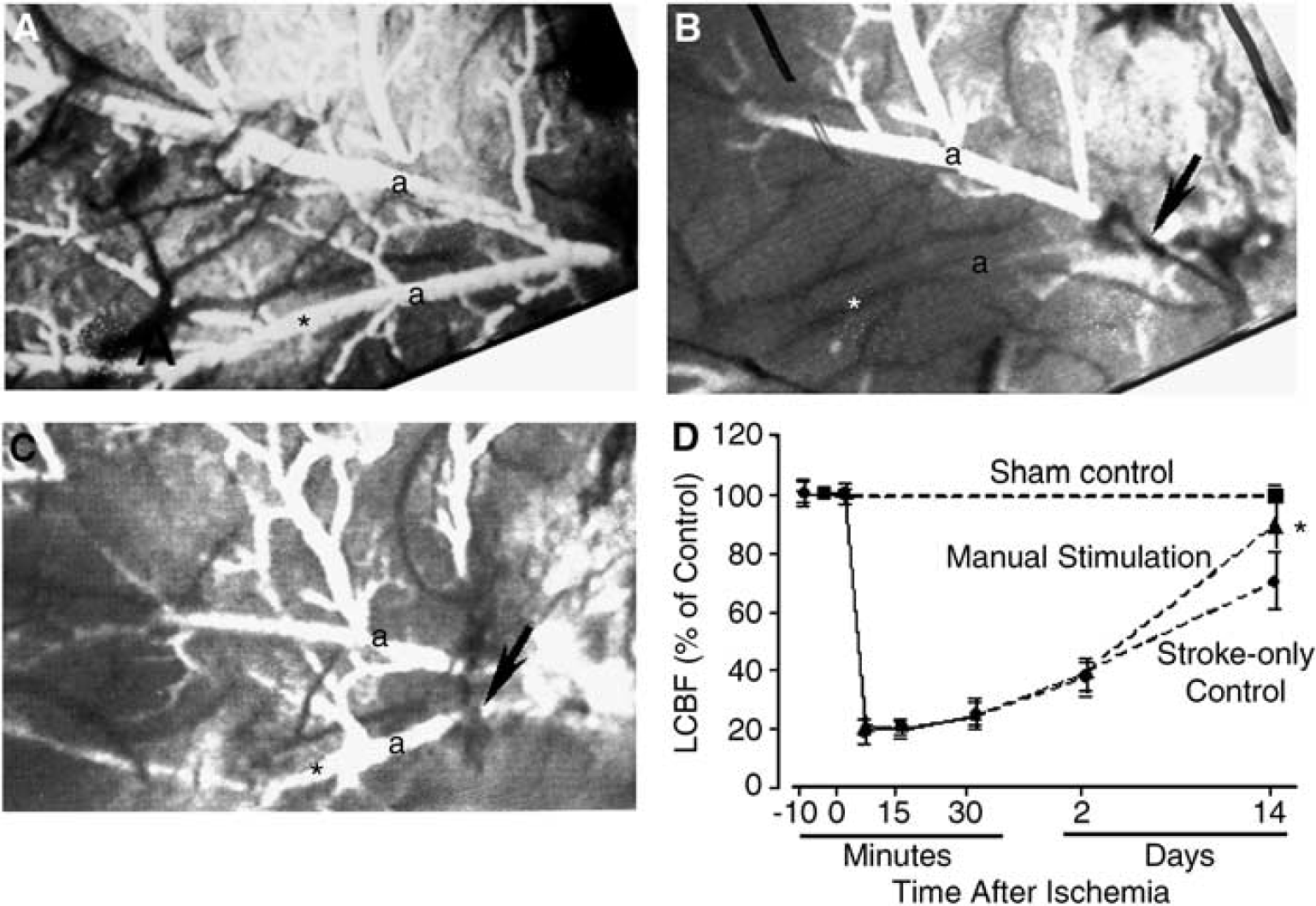
Enhanced LCBF promoted by whisker hyperactivity. Vascular alterations and LCBF in the ischemic and perifocal region of the ipsilateral hemisphere were observed and measured using videomicroscopy and laser Doppler flowmetry. (
Discussion
The present study shows that enhanced peripheral activity, due to forced overuse or passively evoked mechanical stimulation, can noticeably enhance the expression of angiogenic factors and angiogenesis in the penumbra region after focal cerebral ischemia. The increased angiogenesis is exhibited as enhanced proliferation of vascular endothelial cells and actual formation of new microvessels. The increased total number of vessels was likely a consequence of angiogenic activity after cerebral ischemia (Hayashi et al, 2003). To the best of our knowledge, this is the first study that directly shows the central angiogenic effect of peripheral stimulation after ischemic stroke.
As a cell death assay, TUNEL staining was used. Owing to the notion that increased neural activity may augment the metabolic burden of the fragile post-ischemic brain, we wondered whether the timing of stimulation might influence cell survival after stroke. In preliminary experiments, when whisker stimulation was started 1 day after ischemia, cell death was indeed exacerbated. However, beginning 3 days after ischemia, the adverse effect was no longer detected and, on the contrary, peripheral activity attenuated endothelial cell death by the stimulation protocol. This seemingly time-and/or strength-dependent effect may have important clinical significance and calls for further investigation.
The protective effect in this study was likely due to increased trophic factor expression and angiogenesis. Endothelial cell death accounted for only a small portion of total cell death, which was fully expected because there were fewer of these cells compared with neurons and glial cells. The protective effect of peripheral stimulation on vascular endothelial cells, together with proliferation of these cells, however, may be of significance in terms of promoting post-ischemic vascular recovery and faster restoration of local blood flow.
Previous studies showed that focal ischemia in mice resulted in increased expression of angiogenic factors, which reached peak levels in 3 to 7 days (Lin et al, 2000; Zhang and Chopp, 2002). Enhancing sensory input to the ischemic whisker barrel cortex further increased expression of angiogenic factors compared with the stroke-only control group 7 days after ischemia. Considering that retrograde thalamus neuronal cell death occurs following the barrel stroke (Wei et al, 2004), it was questioned how the afferent signals could be passed by the damaged thalamocortical pathway to the barrel cortex. Because angiogenic activity was observed in the penumbra region, it can be assumed that some thalamocortical connections to the peripheral barrel columns may be preserved via surviving cortical and thalamus neurons. This was supported by our observation that cytochrome oxidase labeling showed residual barrel structures after the focal ischemia (Figure 2D). In addition, some afferent singles may be passed by a compensated plasticity of nearby vertical and horizontal pathways (Finnerty et al, 1999) or intracortical projections, arising from peri-infarct cortex (Carmichael et al, 2001).
Western blot analysis showed enhanced expression of VEGF, bFGF, Ang-1, Ang-2, and Tie-1. Immunohistochemical labeling with Tie-1 and VEGF antibodies showed that expression of these factors is mainly localized to the penumbra region after ischemia. The expression of Ang-1 and Ang-2 coincides with proliferation of endothelial cells (Hayashi et al, 2003; Lin et al, 2000). Angiogenic factors have been shown to be important for sprouting of vessels (Hayashi et al, 2003; Lin et al, 2000; Risau, 1997), suggesting that their expression corresponds with the new vessel growth phase of angiogenesis. In our experiments, the whisker activity-induced expression of several key factors in angiogenesis occurred at a time when both endothelial cell proliferation and sprouting took place. Indeed, new vessel formation was confirmed at 7 and 14 days after ischemia, and enhanced LCBF was seen in mice subjected to whisker stimulation.
Between the whisker-trimmed mice and the mice that received manual stimulation, the latter increased expression of most angiogenic factors tested in the penumbra region. Trimming the right side whiskers, which forced the mice to overuse their remaining whiskers, showed less of an effect on angiogenic factor expression. Consistently, manual stimulation showed a greater effect in promoting angiogenesis. These data may implicate short but intensive peripheral afferent signals in producing stronger responses in the central nervous system than persistent but mild activity. Alternatively, different effects of self-controlled and passive movements of whiskers remain to be depicted. In this investigation, we cannot rule out the possibility that in whisker-trimmed animals, deficient afferent inputs might result in atrophic alteration of the contralateral barrel cortex, and this could affect gene expression and angiogenesis in the ipsilateral cortex, although increased expression of several angiogenic genes does not support this assumption.
Neurovascular plasticity is important for tissue remodeling after cerebral injury (Wei et al, 2005). Methods to promote neuronal and vascular plasticity often involve manipulation of growth factors. Administration or induction of VEGF after MCA occlusion in the mouse can decrease infarct size, increase angiogenesis and neurogenesis, and improve functional recovery (Chen et al, 2005; Kaya et al, 2005). Treatment of mice with recombinant adenoviruses expressing Ang-1 before cerebral ischemia reduced blood–brain barrier leakage and lesion volume (Zhang et al, 2002a, b). Intravenous infusion of FGF-18 after ischemia and reperfusion attenuated infarct volume and behavioral deficits, which was assumed to be a result of increased regional cerebral blood flow (Ellsworth et al, 2003). Thus, increases in angiogenic activity enhance neurovascular remodeling, which may contribute to improved functional recovery.
The results obtained in this study indicate that angiogenesis after focal cerebral ischemia in the mouse whisker barrel cortex can be enhanced by peripheral manipulation. It is in line with the opinion that an adequate amount and proper timing of physical therapy, such as exercise and peripheral stimulation, may promote brain tissue repair and functional recovery. Further understanding of the molecular and cellular mechanisms involved in the regulation of post-ischemic angiogenesis and neurovascular plasticity may lead to the development of more specific and efficient therapeutic approaches for stroke patients.
