Abstract
Stroke causes heterogeneous changes in tissue oxygenation, with a region of decreased blood flow, the penumbra, surrounding a severely damaged ischemic core. Treatment of acute ischemic stroke aims to save this penumbra before its irreversible damage by continued ischemia. However, effective treatment remains elusive due to incomplete understanding of processes leading to penumbral death. While oxygenation is central in ischemic neuronal death, it is unclear exactly what actual changes occur in interstitial oxygen tension (pO2) in ischemic regions during stroke, particularly the penumbra. Using the unique capability of in vivo electron paramagnetic resonance (EPR) oximetry to measure localized interstitial pO2, we measured both absolute values, and temporal changes of pO2 in ischemic penumbra and core during ischemia and reperfusion in a rat model. Ischemia rapidly decreased interstitial pO2 to 32% ± 7.6% and 4% ± 0.6% of pre-ischemic values in penumbra and core, respectively 1 hour after ischemia. Importantly, whilst reperfusion restored core pO2 close to its pre-ischemic value, penumbral pO2 only partially recovered. Hyperoxic treatment significantly increased penumbral pO2 during ischemia, but not in the core, and also increased penumbral pO2 during reperfusion. These divergent, important changes in pO2 in penumbra and core were explained by combined differences in cellular oxygen consumption rates and microcirculation conditions. We therefore demonstrate that interstitial pO2 in penumbra and core is differentially affected during ischemia and reperfusion, providing new insights to the pathophysiology of stroke. The results support normobaric hyperoxia as a potential early intervention to save penumbral tissue in acute ischemic stroke.
Keywords
INTRODUCTION
Acute ischemic stroke results in heterogeneous changes in cerebral blood flow and brain metabolism in the affected region. Ischemic penumbra is the initially viable tissue compromised by partially decreased cerebral blood flow and disturbed metabolism surrounding the severely damaged ischemic core (Astrup et al., 1981). Although the ischemic penumbra is a major target of acute stroke therapy (Fisher, 1997) and is being actively explored in research, its pathophysiology of oxygenation and metabolism has not been well understood. Positron emission tomography (PET) studies have demonstrated that the ischemic penumbra has an increased oxygen extraction fraction, reduced cerebral metabolic rate of oxygen (CMRO2), and increased glucose metabolic rate (Heiss et al., 1994; Kuge et al., 2001). Using autoradiographic methods, Ginsberg and co-workers have shown that increased glucose metabolic rate was a signature of the early penumbra, and that differential levels of blood flow in the penumbra and core were highly predictive of histological injury within the same focus (Belayev et al., 1997; Zhao et al., 1997). A recent study in a primate model showed that the CMRO2 level obtained using PET scanning could predict penumbral regions (Frykholm et al., 2000). One important reason for this reduced CMRO2 in penumbra is hypothesized to be decreased interstitial (or tissue) oxygen levels that are directly available for cells in the penumbra to utilize for metabolism. Oxygen plays a key role in the pathophysiology of cell death associated with brain ischemia or hypoxia: a lack of oxygen disturbs cerebral metabolism during the insult, while over-supply of oxygen has been concerned with increased generation of reactive oxygen species. Yet due to technical difficulties in measuring brain interstitial oxygen in vivo, it is presently unclear exactly what actual changes occur in interstitial oxygen levels in ischemic regions, particularly the penumbra, during cerebral ischemia and reperfusion. Determining these interstitial oxygenation changes in penumbra and core are therefore crucial to fully understanding the processes occurring in the penumbra. Furthermore, monitoring interstitial oxygenation will provide accurate values for evaluating emerging hyperbaric (Rusyniak et al., 2003) and normobaric (Singhal et al., 2002a; 2002b) oxygen therapies in acute ischemic stroke.
In this work, we have investigated the local interstitial oxygen levels in penumbra and core during ischemia, reperfusion and hyperoxic treatment, using a novel technique, electron paramagnetic resonance (EPR) oximetry. EPR oximetry is a recently developed technique for interstitial partial oxygen pressure (pO2) measurement in living animals (Liu et al., 1993; Dunn et al., 2000). Compared to other oxygen measurement methods such as Clark-type electrodes (Charbel et al., 1997), phosphorescence quenching (Tammela et al., 1996), and near infrared spectroscopy (Holzschuh et al., 1997), EPR oximetry has several advantages, such as repetitive and highly accurate measurement of localized interstitial (or tissue) pO2 in a non-invasive manner after initial stereotaxic implantation of the inert and microscopic paramagnetic material at a specific site in the brain. This implantation procedure is well characterized, and importantly, is known not to result in any localized long- or short-term changes in structure or function at the implantation site (Rolett et al. 2000; Dunn and Swartz, 2003). We report here our findings, in a rat intraluminal model of cerebral ischemia and reperfusion, that localized pO2 in the ischemic core and penumbra responds very differently not only to transient ischemia and reperfusion, but also to hyperoxia treatment.
MATERIALS AND METHODS
Animal preparation
The Laboratory Animal Care and Use Committee of the UNM HSC approved all experimental protocols. Male Sprague-Dawley rats, 310 to 330g, were from Charles River Laboratory (Wilmington, MA). Animals were maintained in a climate-controlled vivarium with a 12-hour light-dark cycle with free access to food and water.
For all surgical procedures, except hyperoxic treatment, 4.0% isoflurane in N2O:O2 (70%:30%) was used for anesthesia induction, and 2.0% for anesthesia maintenance. For hyperoxic treatment, N2 was used instead of N2O to eliminate compounding effects due to inspired fraction changes of N2O, and isoflurane concentration remained constant. Rats were anesthetized throughout pre-ischemia, ischemia and reperfusion. Physiological monitoring during the procedure comprised measurement and maintenance of core (rectal) temperature at 37.5 ± 0.5°C using a heating pad. Mean blood pressure was also monitored using Advanced Blood Pressure Monitor System (Harvard Apparatus, Holliston, MA, USA).
Induction of focal ischemia and reperfusion
Middle cerebral artery occlusion (MCAO) followed by reperfusion was conducted using an intraluminal model as previously described (Koizumi et al., 1986). Briefly, external carotid artery (ECA), internal carotid artery (ICA) and pterygopalatine artery (PPA) of ICA were exposed. A silicone rubber-coated monofilament nylon suture was inserted into the ICA via a slit on the ECA. The suture was advanced along the ICA to the extent of 18 to 19 mm from the bifurcation. Reperfusion was produced by gently withdrawing the suture until the suture tip reached the bifurcation and the incision closed. For all animals used in this study, successful MCAO was confirmed postmortem immediately after experiment (3 hours after reperfusion) by 2% 2,3,5-triphenyltetrazolium chloride (TTC) stain as previously described (Liu et al., 2002). Ischemia was further confirmed in vivo, in a subset of one-third of animals, by laser Doppler flowmetry (LDF) as described in the literature (Takagi et al. 1994; Nakai, et al. 1997). For LDF, a hole was made using a manual drill on the right prietal skull at a position of 0.5mm posterior, 4mm lateral to the bregma, with dura left intact. Focal cerebral blood flow was measured by stereotactic placement of DRT4 LDF (Moor Instruments, Wilmington, DE) probe over the hole. Of the 74 rats initially used in this study, about 90% had successful occlusion of MCA, and 70% (52 rats) had both successful occlusion and reperfusion. Only the latter were used for data analysis. Most of the excluded rats were due to subarachnoid hemorrhage upon reperfusion.
Implantation of EPR oximetry probe Lithium Phthalocyanine (LiPc)
Ischemic penumbra and core positions were pre-determined as described in detail in our previous reports (Liu et al., 2002; Liu et al., 2003). Briefly, by TTC stain of sectioned brain we compared the different infarction areas resulting from various durations of focal ischemia and reperfusion in five groups with 5 rats in each group. Group 1 was subject to 30 minute ischemia with 24 hour reperfusion; group 2 to 60 minute ischemia with 24 hour reperfusion; group 3 to 90 minute ischemia with 3 hour reperfusion; group 4 to 90 minute ischemia with 24 hour reperfusion; and group 5 to permanent ischemia for 24 hours. The differences in infarction area between those groups were considered as the penumbra, and the 24-hour permanent ischemia group was used to demonstrate the maximum ischemically compromised area after middle cerebral artery occlusion. This penumbra area was then converted to corresponding coordinates used for stereotaxic implantation according to brain atlas of Paxinos and Watson (1986). As shown in Fig. 1, A and D, LiPc crystals were implanted stereotaxically to the basal ganglial penumbra and core region. At penumbra position (Fig. 1A), although brain tissue was stained by TTC (red color), this part of brain tissue is ischemically compromised, and will become infarcted if reperfusion or ischemia continues for 24 hours, as shown by TTC stain in Fig. 1, B and C. For every animal, correct assignment of the implantation site was confirmed post- mortem by TTC staining. LiPc was a gift from Dr. Harold Swartz (NIH In Vivo EPR Center, Dartmouth College, NH). The use of LiPc in measuring tissue pO2 has been well characterized. LiPc is known to be very stable in tissue, and implantation of LiPc does not lead to any localized short- or long-term changes in structure or function at the implantation site (Rolett et al. 2000; Dunn and Swartz, 2003; Liu et al. 1995).
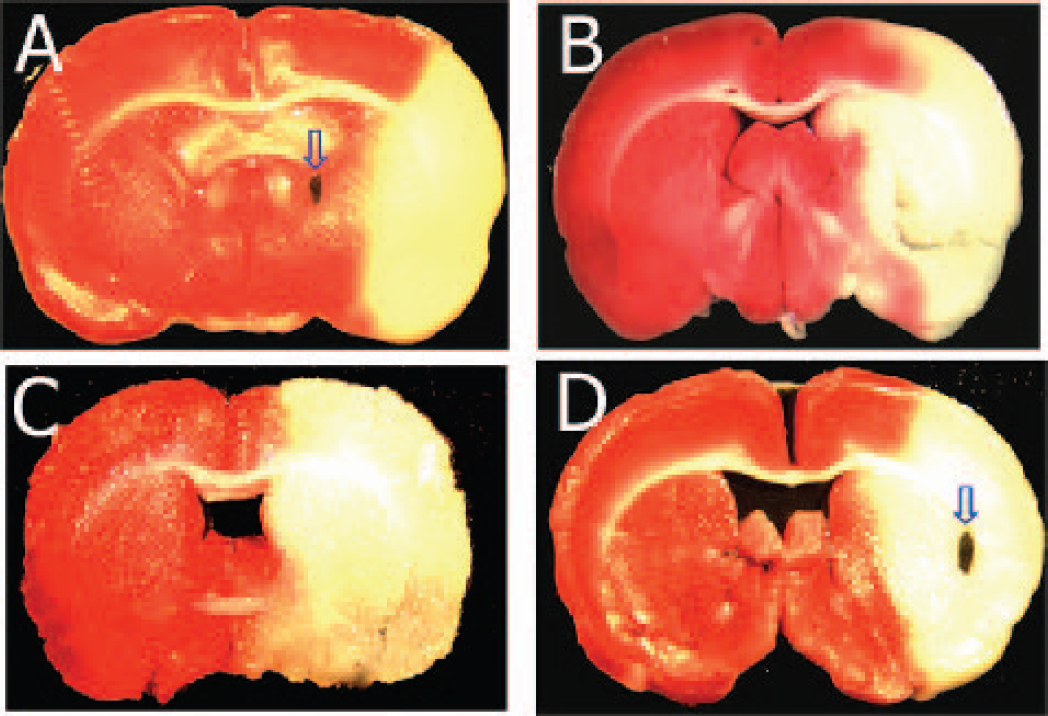
Triphenyltetrazolium chloride stain of rat brain sections after ischemia and reperfusion showing the areas considered to be ischemia core and penumbra. Red area represents non-ischemic region, and yellow bulk (absence of staining) region represents dead ischemic brain tissue. Arrows in A and D indicate LiPc crystal placement in penumbra and core respectively. A, D, MCAO 90 minutes and reperfusion 180 minutes. B, MCAO 90 minutes and reperfusion 24-hour; C, MCAO 24 hours without reperfusion. The area difference of yellow regions in A-C is penumbra.
LiPc crystals were implanted in 17 rats (including 6 for hyperoxic treatment) at penumbra position, 12 rats (including 3 for hyperoxic treatment) at core position and 5 rats at contralateral (left side) core position. Under anesthesia, a pin hole on the prietal skull was made at the position of 0.5 mm posterior, 2 mm lateral to bregma for penumbra, and 0.5 mm posterior, 5 mm lateral to bregma for core. A small LiPc crystal (approximate diameter 0.2mm) was placed down 5.5 mm for basal ganglial penumbra position and 4 mm for cortical core position using a microdialysis guide cannula with an inner diameter of 0.24mm (CMA microdialysis, Stockholm, Sweden). Rats were allowed to recover from implantation 48 to 72 hours before induction of cerebral ischemia.
Measurement of cerebral pO2 by EPR oximetry
For non-invasive measurement of local cerebral pO2 directly in the anesthetized rat, an external loop resonator was placed over the position where LiPc was implanted, and EPR spectrum was recorded using a Bruker EleXsys E540 EPR spectrometer equipped with a L-band bridge (Bruker Instruments, Billerica, MA). The resonator has advanced automatching and autotuning capabilities that correct for any slight animal movement. The EPR spectrum was acquired with a scan time of 30 seconds, and usually 10 scans were obtained and averaged to produce a signal-to-noise ratio of at least 5 to 1, which allows accurate fitting. The peak-to-peak line width of the spectrum was obtained via computer line-fitting (Fig. 2A), and converted to pO2 values according to a calibration curve for the oximetry probe LiPc used in the study. The linear calibration curve was obtained in vitro by measuring the EPR linewidth at various known pO2 values, as described before (Liu et al., 1993). Typical EPR acquisition parameters include microwave power 10 mW, microwave frequency 1.07 GHz, magnetic field center 380 G, scan range 2.0 G, and modulation amplitude less than one-third of EPR line-width. Interstitial pO2 was measured continuously throughout the experiment, and reported at the time points shown.
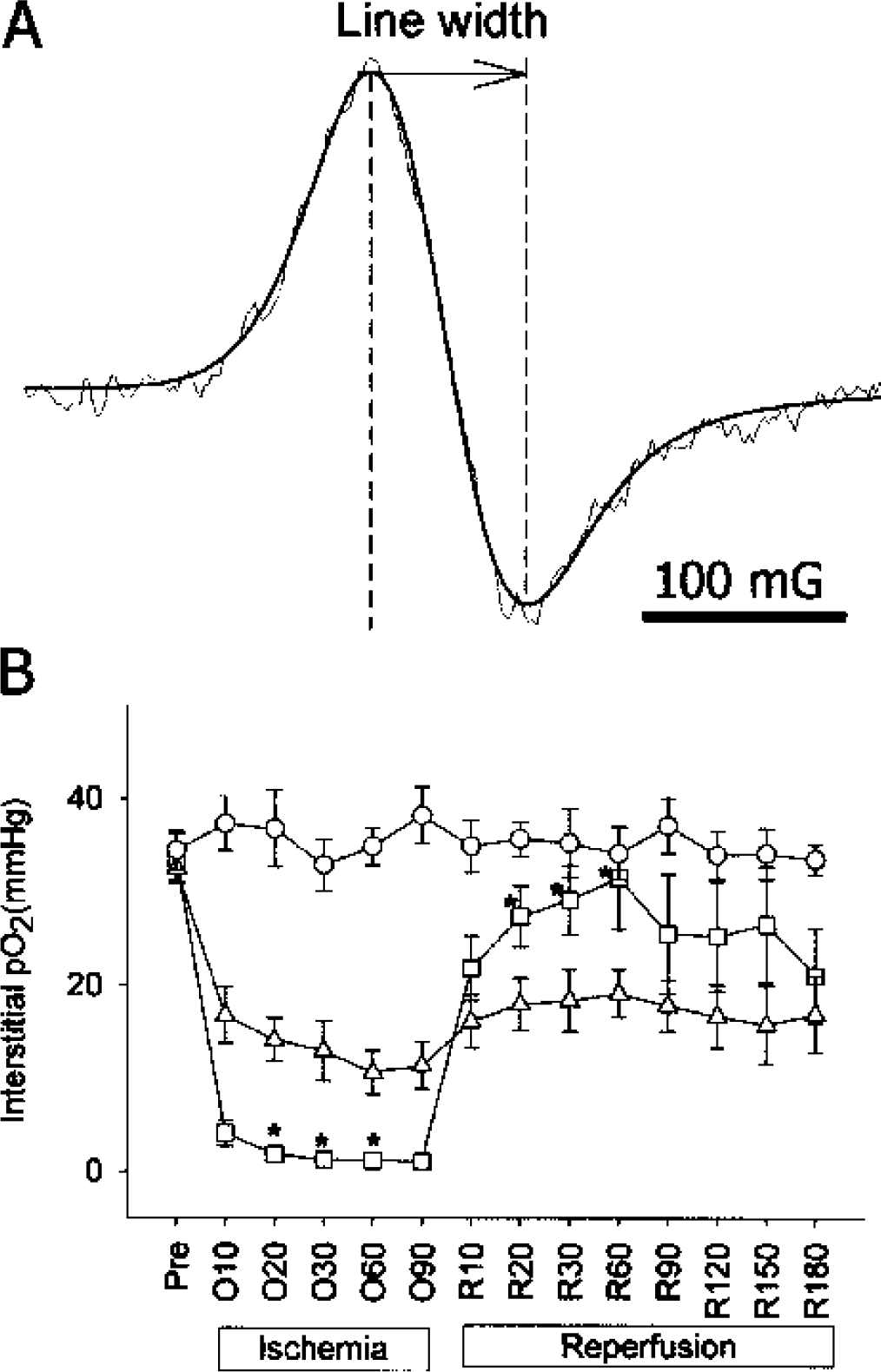
Interstitial pO2 levels at different positions during cerebral ischemia and reperfusion. A, typical EPR spectrum obtained in vivo during ischemia and computer simulation (smooth line); B, pO2 changes in basal ganglial penumbra (n = 11), ischemic core (n = 9) and contralateral (n = 5) during ischemia and reperfusion. Data was expressed as Mean ± SEM. Pre, pre-ischemia; O10-O90, 10 to 90 minutes after MCAO; R10-R180, 10 to 180 minutes after reperfusion. ○, contralateral side; ▵, penumbra; □, core. Asterisk indicates significant (P < 0.05) difference when compared with penumbra.
Effect of hyperoxic treatment on cerebral pO2
To measure the effect of hyperoxia treatment on cerebral pO2 in penumbra and core during ischemia and reperfusion, the oxygen content in the inspiration gas was raised from 30% (30%:70%, O2:N2) to 70% (70%:30%, O2:N2), and then returned to 30%. Interstitial pO2 was measured at 10-minute intervals. 6 rats (3 rats each for penumbra and core) breathed 70% oxygen during the period of 30 to 60 minutes after MCAO, and additional 3 rats during 30 to 90 minutes after reperfusion.
Measurement of cellular oxygen consumption rate in different regions of ischemic brain
Rats were euthanized by decapitation under anesthesia after 90-min ischemia and 1-hour reperfusion. Brain was removed into Dulbecco's modified Eagle's medium/F12 (DMEM/F12, Sigma, St. Louis, MO) and sliced. Tissue was sampled from core, penumbra and contralateral core positions as indicated in Fig. 4A. Cell suspensions from the isolated brain tissue were made using the technique of Brewer et al. (1997). Cells were resuspended in 7.5% dextran (MW 500,000) in DMEM/F12 at 2 × 106 cells/ml. Oxygen consumption rate of the suspended cells was then measured using the well established EPR oximetry method for cell cultures (James et al. 1995). A soluble oxygen probe, 4-Oxo-2,2,6,6-tetramethylpiperidine-d16-1-15N-oxyl (15N-PDT) (Cambridge Isotope Laboratories, Andover, MA, USA) was added to 100 μl cell suspension at a final concentration of 0.1 mM. The cell suspension containing 15N-PDT was drawn into a glass capillary tube and sealed at both ends with Critoseal. (Oxford Labware, St. Louis, MO, USA). Oxygen concentration in the medium was calculated from 15N-PDT EPR spectrum line-width according to a calibration curve. Spectra were recorded at 37°C using a Bruker E540 EPR spectrometer at X-band. EPR parameters were field center 3360 G; scan range 2.6 G; microwave frequency 9.05 GHz; microwave power 5 mW, and modulation amplitude 0.03 G.
Statistics
ANOVA for repeated measures and Sheffe multiple comparison were used for the analyses of pO2 changes during ischemia and reperfusion, pO2 response to hyperoxia intervention during ischemia. One-way ANOVA and Sheffe multiple comparison were used for analyses of pO2 changes during hyperoxia intervention in reperfusion period and oxygen consumption rates in core, penumbra and contralateral positions. Linear regression was used to obtain oxygen consumption rate. Significance was considered with P < 0.05.
RESULTS
CBF and BP monitoring of animals
During ischemia, LDF regional cerebral blood flow dropped to 18.8 ± 2.0% of the pre-ischemic level, and after reperfusion the blood flow was restored to 88.7 ± 5.6% of pre-ischemic level, which is consistent with the reported values in rats under similar conditions (Sugo et al. 2002; Westermaier et al. 2003). Mean blood pressure remained constant throughout the procedure within the range of 80 ± 14 mmHg.
pO2 in core and penumbra during ischemia and reperfusion
Pre-ischemic pO2 values in ischemic core and basal ganglial penumbra of the anesthetized rats were 33.4 ± 6.0 mmHg. ANOVA for repeated measures showed significant difference (P < 0.05) between the changing patterns of pO2 in penumbra, core and contralateral hemisphere, as shown in Fig. 2B. After MCAO, interstitial pO2 in both core and penumbra dropped rapidly in the first 10 minutes, thence the rate of decrease slowed, and reached their respective lowest levels at 1-hour post-occlusion. The interstitial pO2 values in penumbra were significantly higher than the corresponding values in the core, and were 10.7 ± 7.8 and 1.2 ± 0.7 mmHg at 1-hour after occlusion, respectively. After reperfusion, pO2 levels in both core and penumbra positions increased, but very differently. One hour after reperfusion core pO2 returned to near pre-ischemic levels, 31.6 ± 16.5 mmHg, whilst penumbral pO2 showed only partial recovery to a level of 19.1 ± 6.7 mmHg. At 20 to 60-min after reperfusion pO2 values in core were significantly (P < 0.05) higher than in penumbra. In contrast to the values in the occluded hemisphere, pO2 values in the contralateral hemisphere remained stable during the entire experiment (Fig. 2B).
Effect of hyperoxic treatment on pO2 levels in penumbra and core
To determine whether hyperoxic treatment could alter cerebral interstitial pO2 in the penumbra and core, inspired oxygen was increased from 30% to 70%. Fig. 3A shows that MCAO resulted in a rapid drop in both penumbral and core pO2. When 70% inspired oxygen was given to the animals during the time of 30 to 60-min during the occlusion period, there was a significant increase in penumbral pO2, reaching 49.4 ± 16.3% of pre-ischemic level. In contrast, core pO2 was not increased by the hyperoxia treatment during ischemia and remained at 8.4 ± 1.3% of the pre-ischemic level. During the last 30 minutes of ischemia, when inspired oxygen was returned to 30% oxygen, the increased pO2 in penumbra observed during inspiration of 70% oxygen, returned to values similar to previous values. ANOVA and Sheffe multiple comparison show significant (P < 0.05) difference between penumbra and core after 30-min hyperoxia.
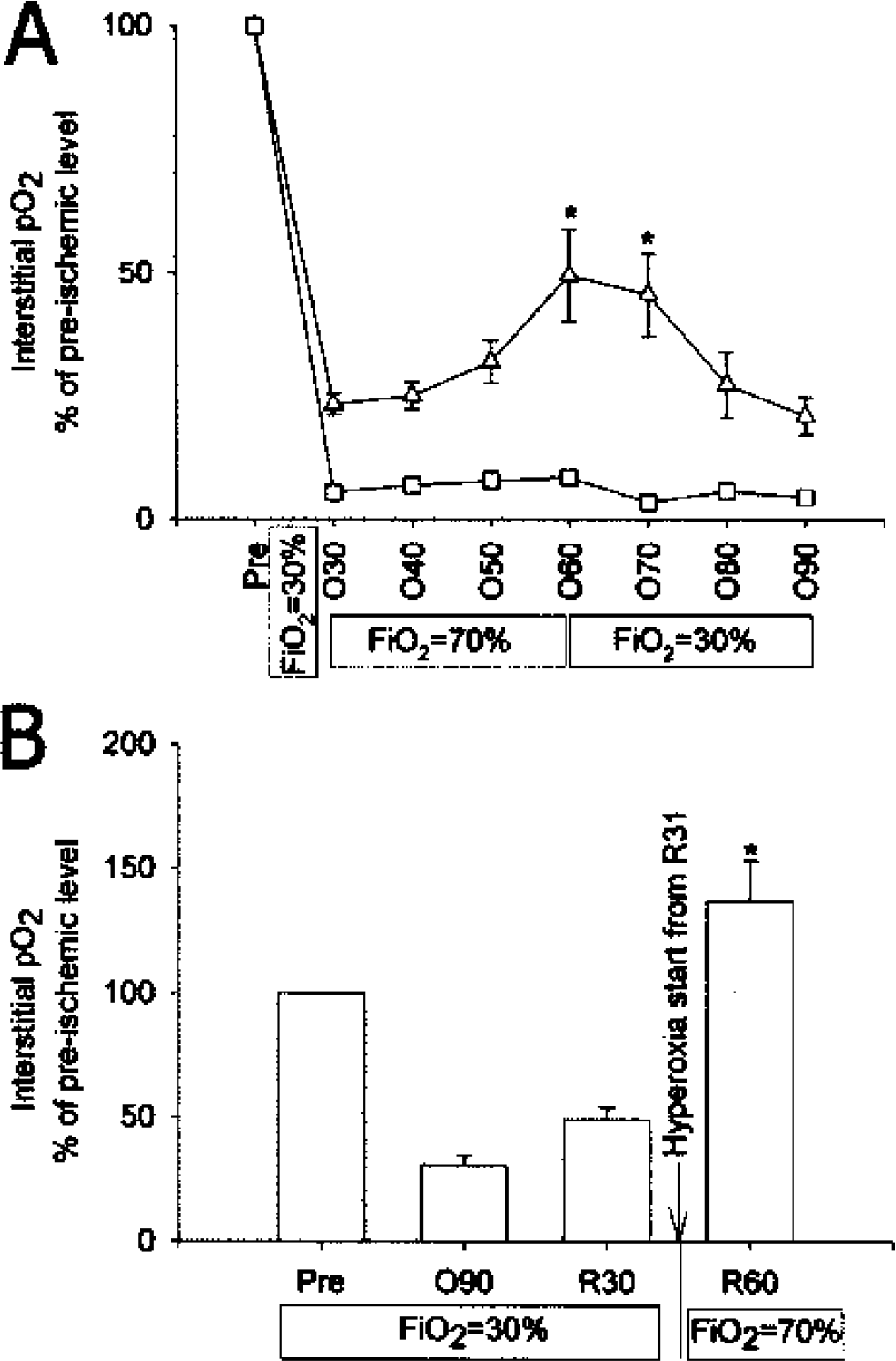
Different responses to hyperoxic treatment in penumbra and core. Data was expressed as Mean ± SEM. N = 3 for each group. FiO2, inspired oxygen fraction. A, Effect of hyperoxia during ischemia. Pre, pre-ischemia; O10-O90, 10 to 90 minutes after MCAO. ▵, penumbra; □, core. Asterisk indicates significant difference (P < 0.05) when compared with core of the same time point. B, Response of pO2 to hyperoxia in penumbra during reperfusion. Pre, pre-ischemia; O90, 90-min after MCAO; R30 and R60: 30-min, and 60-min after reperfusion respectively. Asterisk indicates significant difference (P < 0.05) when compared with each of the other groups. N = 3 for each group.
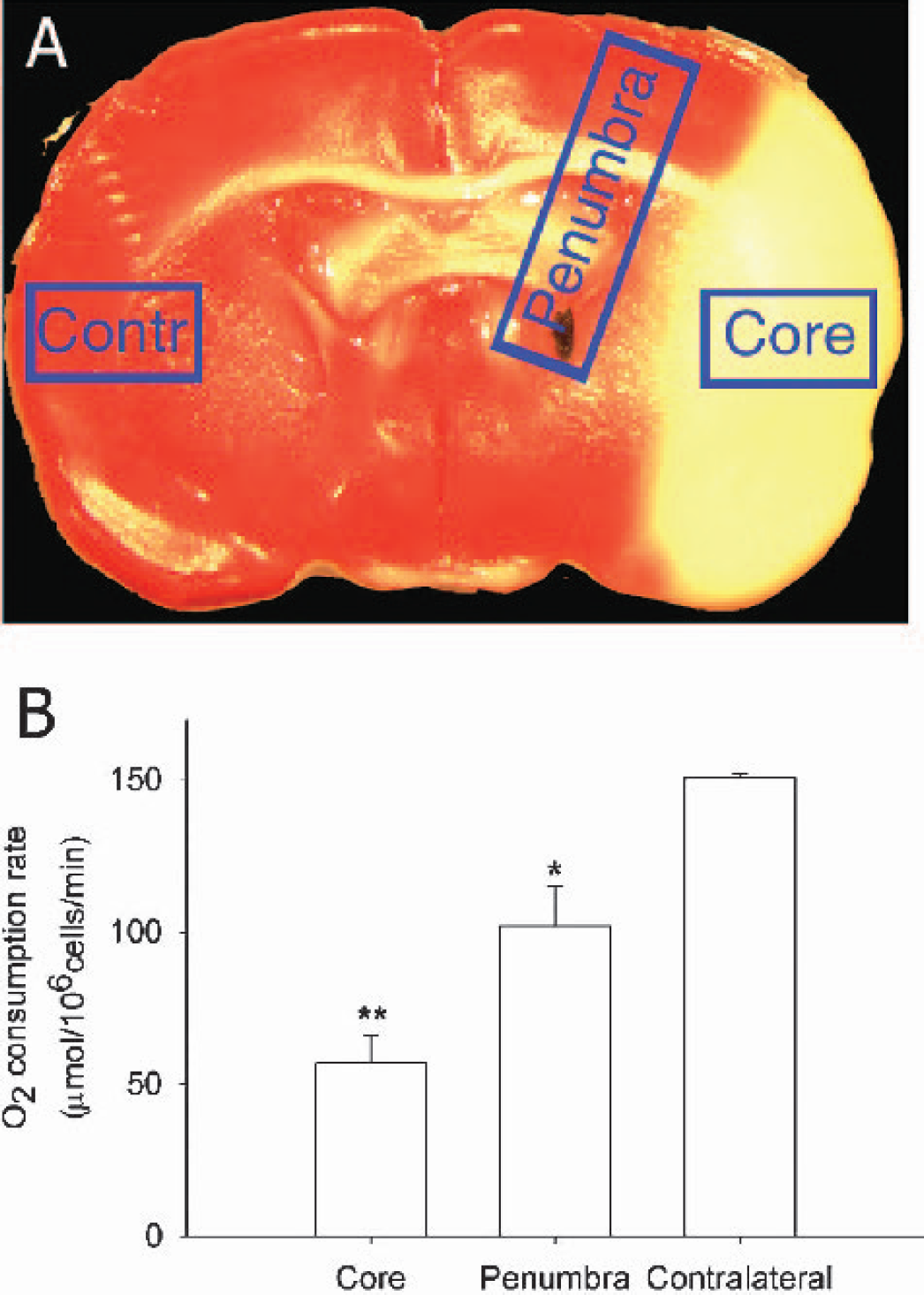
Different oxygen consumption rates of cell suspension from core, penumbra and contralateral side. A, TTC stain illustrating tissue-sampling sites for core, penumbra, and contralateral side (Contr). B, Oxygen consumption rates at different positions. Data was expressed as Mean ± SEM. N = 3 for each group. Asterisk indicates significant (P < 0.05) difference when compared with contralateral side. Double asterisks indicate significant difference when compared with penumbra (P < 0.05).
The positive response to hyperoxia treatment of the pO2 in penumbra during ischemia led us to test further whether hyperoxia treatment could also increase penumbral pO2 during reperfusion. As shown in Fig. 3B, while rats were breathing 30% oxygen, pO2 in penumbra dropped to a very low level after 90-min ischemia, and was only partially restored after 30-min reperfusion. However, when the inspired oxygen was increased from 30 to 70% oxygen, a dramatic increase in penumbral pO2 was observed, reaching 136.9 ± 28.3% of the control, pre-ischemic level (P < 0.05).
Cellular oxygen consumption rate in penumbra and core
To better understand the substantial differences between penumbral and core pO2 levels during MCAO and reperfusion, we utilized the well established EPR oximetry techniques for measurement of cellular oxygen consumption under cellular physiological conditions without the complication due to differences in blood flow and other in vivo conditions. The oxygen consumption rate was measured for cells collected from penumbra, core, and contralateral regions of ischemic rats after 90 min MCAO and 1 h reperfusion (as indicated in Fig. 4A). Fig. 4B shows that cells from ischemic core consumed oxygen significantly slower than cells from penumbra, while cell suspension from the contralateral hemisphere had the highest oxygen consumption rate. ANOVA showed significant overall difference between the three groups (P < 0.05). Sheffe multiple comparison showed significant (P < 0.05) differences of oxygen consumption rates between penumbra and core, between penumbra and contralateral hemisphere.
DISCUSSION
Using the unique capability of in vivo EPR oximetry to measure localized interstitial pO2 in living animals, we have provided both the absolute values and temporal changes of pO2 in the ischemic penumbra and core during cerebral ischemia and reperfusion. Our results demonstrate that pO2 in penumbra and core behaves significantly differently. During ischemia, as expected, blood supply to the ischemic core was interrupted, and residual oxygen was consumed rapidly by these cells, leading to a pO2 near zero. In the penumbra, on the other hand, there was a reduced but constant blood supply to the region (Dunn et al., 2001), and consequently, a lowered, but constant tissue pO2 level was maintained (Fig. 2B). An interesting, and unexpected, finding of the present study is that, upon reperfusion, the tissue pO2 levels in the core returned close to the pre-ischemic level, whilst penumbral pO2 increased but only to a significantly lower level than in the core. This is most likely related to differences in oxygen consumption and microcirculation status between core and penumbra during reperfusion.
Upon reperfusion, oxygen is delivered to the core with restored blood flow but, as shown in Fig. 4B, there is limited consumption of oxygen in ischemic core, because most of the cells in this region were either dead or damaged, as is evident by the lack of viable mitochondria by TTC stain after 180-min reperfusion (Fig. 1A). Therefore, interstitial pO2 was restored rapidly in core during reperfusion. The penumbra, on the other hand, is undergoing dynamic changes (Heiss et al., 1994) making the condition in penumbra very complicated. First, although lower than the contralateral, the oxygen consumption rate in penumbra is significantly higher than in the core (Fig. 4B). This finding is supported by results from PET studies that ischemic penumbra had increased oxygen extraction fraction, increased glucose metabolism but decreased oxygen metabolism as compared to baseline values, and that oxygen metabolism in the core was significantly lower than the penumbra (Heiss et al., 1994; Kuge et al., 2001; Frykholm et al., 2000). Second, as demonstrated in our previous study (Liu et al., 2002), blood flow in penumbra may not be completely restored upon reperfusion due to microcirculation stasis (or hypoperfusion) that is more severe in penumbra than in core. In addition, the decreased penumbral blood flow could be further compromised by increased cerebral edema due to reperfusion (Rosenberg, 1999). With the compromised circulation upon reperfusion due to microcirculation stasis, and with active penumbral oxygen consumption, the overall recovery of penumbral pO2 is incomplete during reperfusion. This slow and incomplete recovery of penumbral pO2 is implicated in the delayed death from reperfusion injury observed in this region (Kuroda and Siesjo, 1997). Thus, if left untreated, the incomplete recovery of penumbral pO2 will very likely contribute to the further deterioration of penumbra even if reperfusion occurs.
There have been limited and scattered reports in the literature on cerebral pO2 during ischemia and reperfusion in the core, but none in the penumbra. It was shown that interstitial pO2 had a good relationship with CBF, brain pH, and pCO2 changes during ischemia in primate (Harris et al., 1987) and hypoxemia in rabbits (Martinez-Tica et al., 1999). Using microelectrodes, Nakai et al (1997) found that pO2 in the cortical core decreased to 5% of the control during ischemia, and was restored to 160% of pre-ischemic level during reperfusion. In contrast, we did not observe this overshoot recovery of pO2 in core upon reperfusion; rather it was only restored close to pre-ischemic levels. This difference may be due to the different locations used for pO2 measurements, and/or the potential difficulties associated with microelectrodes for measurement during extended times in vivo. Recent studies have examined the ischemic thresholds for brain oxygen tension (Charbel et al., 1997), and brain oxygenation monitoring is being actively explored in clinical situations (Jodicke et al., 2003; Gupta et al., 2002; Doppenberg et al., 1998). Studies using PET have reported an increased oxygen extraction fraction and reduced CMRO2 in penumbra (Heiss et al., 1994; Kuge et al., 2001). This increased oxygen extraction fraction and reduced CMRO2 is consistent with our finding of decreased oxygen availability in penumbra.
EPR oximetry is an emerging technique that is well suited for the measurement of cerebral tissue oxygenation, and is based on the sensitivity of the linewidth of EPR oximetry probe to oxygen levels in the tissue immediately surrounding the surface of the material (Velan et al. 2000; Dunn and Swartz, 2003). The technique with particulate probe measures the localized interstitial, steady-state, oxygen level, which depends on, but is different from, the commonly reported cerebral blood flow or cerebral metabolic rate of oxygen (CMRO2). After initial stereotaxic implantation of the microscopic oximetry probe, the measurement of pO2 itself is totally non-invasive, permitting continuous and repetitive monitoring of pO2 accurately with high reproducibility and high temporal resolution (< 3 min). These advantages of EPR oximetry are reflected by the consistent absolute values of pO2 over nearly 5 hours for the control group as shown in Fig. 2B. The pO2 values of the control group obtained in this study are also very consistent with previous reports of cerebral tissue pO2 measurements made by EPR oximetry techniques (Hou et al. 2003; Liu et al. 1995; Dunn et al. 2000).
There is accumulating evidence indicating that oxygen therapy is safe and feasible (Rusyniak et al., 2003), and may be neuroprotective for acute ischemic stroke (Singhal et al. 2002b). Experiments by Singhal et al. (2002a; 2002b) showed that normobaric hyperoxia during focal cerebral ischemia-reperfusion salvaged ischemic brain tissue, with no increased oxidative stress being found to result from this hyperoxia treatment. A recent clinical study demonstrated that there was a slight increase of pO2 in ischemic patients during hyperoxia (Hoffman et al., 1997). Our results in the present study show that normobaric hyperoxia can indeed improve oxygenation in penumbra during both ischemia and reperfusion, supporting normobaric hyperoxia as a practicable early intervention for acute ischemic stroke. If high-risk stroke patients have access to oxygen therapy immediately after stroke, the patients may gain a widened therapeutic window through preserving penumbral tissue before further aggressive thrombolytic treatment. During reperfusion, breathing 70% oxygen was found to increase pO2 in penumbra to 136% of pre-ischemic level. Whether this improvement of pO2 in penumbra could be beneficial is beyond the design of this study and will be explored in future experiments.
In summary, our results provide absolute and accurate values of interstitial pO2 in deep brain tissue, and their temporal changes in both penumbra and core during cerebral ischemia and reperfusion, giving interesting insights about the pathophysiology of ischemic stroke. The methods developed in this study could also be used as an effective means to assess the potential value of neuroprotective strategies that are aimed at increasing oxygenation of the ischemic tissue.
Footnotes
Acknowledgment
We thank Prof. Harold Swartz for providing LiPc, Prof. Gary Rosenberg for valuable comments, and Bruker Biospin Co. for continued EPR support.
