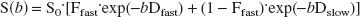Abstract
Diffusion magnetic resonance imaging (MRI) provides a surrogate marker of acute brain pathology, yet few studies have resolved the evolution of water diffusion changes during the first 8 hours after acute injury, a critical period for therapeutic intervention. To characterize this early period, this study used a 17.6-T wide-bore magnet to measure multicomponent water diffusion at high b-values (7 to 8,080 s/mm2) for rat hippocampal slices at baseline and serially for 8 hours after treatment with the calcium ionophore A23187. The mean fast diffusing water fraction (Ffast) progressively decreased for slices treated with 10-μmol/L A23187 (—20.9 ± 6.3% at 8 hours). Slices treated with 50-μmol/L A23187 had significantly reduced Ffast 80 minutes earlier than slices treated with 10-μmol/L A23187 (P < 0.05), but otherwise, the two doses had equivalent effects on the diffusion properties of tissue water. Correlative histologic analysis showed dose-related selective vulnerability of hippocampal pyramidal neurons (CA1 > CA3) to pathologic swelling induced by A23187, confirming that particular intravoxel cell populations may contribute disproportionately to water diffusion changes observed by MRI after acute brain injury. These data suggest diffusion-weighted images at high b-values and the diffusion parameter Ffast may be highly sensitive correlates of cell swelling in nervous issue after acute injury.
Many neuroprotective strategies have proven successful for treating animal models of ischemic stroke and traumatic brain injury, yet have failed to improve clinical outcome in human patients during clinical trials (Faden, 2001). The frequent disappointments in these studies have often been attributed, in part, to defects in clinical trial design wherein studies fail to rapidly assess and stratify potential subjects based on the severity, location, and complexity of their acute brain injuries (Faden, 2001). Appropriate subject selection is particularly important because brain injury patients represent a far more heterogeneous population compared to their corresponding animal models of disease (Statler et al., 2001). To address this problem, it has recently proven beneficial to incorporate diffusion-weighted magnetic resonance imaging (MRI) into clinical trials (Warach, 2001) because of its sensitivity to changes in nervous-tissue water diffusion that occur immediately after injury (Moseley et al., 1990). Diffusion-weighted MRI of stroke patients, for example, aids recognition of the “perfusion-diffusion mismatch,” which identifies penumbral volumes of nervous tissue that may be rescued from subsequent injury by prompt infusion of tissue plasminogen activator (Warach, 2002).
An improved understanding of water diffusion in nervous tissue and the acute water-diffusion changes that follow injury may further increase the sensitivity and specificity of diffusion-weighted MRI as a surrogate marker of acute brain injury. Unfortunately, previous clinical diffusion MRI studies have provided limited data from the first 24 hours after ischemic or traumatic brain injury (for example, Beaulieu et al., 1999). Although this information has proven useful for understanding the chronic progression of diffusion changes after brain injury, it is well beyond the optimal therapeutic windows when experimental interventions are most likely to succeed (Marler et al., 2000). Thus, studies that better resolve the acute temporal progression of diffusion changes after brain injury are needed to provide improved patient stratification based on injury severity and to monitor the effects of therapeutic interventions early after brain injury.
Unlike previous clinical studies of acute brain injury, recent studies have demonstrated that diffusion-weighted signal attenuation is nonmonoexponential in rat (Niendorf et al., 1996) and human brains (Mulkern et al., 1999) when measured with very strong diffusion-sensitizing gradients (high b-values). As a first approximation, a biexponential function may be used to describe water diffusion in nervous tissue by separating the MRI signal into contributions from fast and slow diffusing components. Shifts between these two components may correlate with changes in the relative size of the intracellular compartment (Niendorf et al., 1996; Thelwall et al., 2002). Although the biexponential model is an incomplete description of tissue water diffusion, it is more appropriate than the monoexponential fits currently employed clinically.
Continuous acquisition of water diffusion data for multicomponent analysis throughout the first 8 hours after an acute brain injury would prove extremely difficult in human or animal subjects because of their limited tolerance for long imaging times, and because high b-value diffusion weighting is difficult to obtain on relatively large subjects owing to current hardware limitations. Thus, to further investigate the value of diffusion MRI as a surrogate marker of acute brain injury, we measured water diffusion in perfused rat hippocampal slices using a 17.6-T magnet with strong diffusion-sensitizing gradients (1,000 mT/m). Unlike human and animal subjects, rat hippocampal slices tolerate long periods of investigation yet maintain the heterogeneous neuronal and glial cell populations (Aitken et al., 1995) that determine in vivo MRI contrast. Furthermore, studies of hippocampal slices are not confounded by anesthesia or movement and perfusion artifacts (Aitken et al., 1995). Previous studies have used diffusion-weighted MRI of rat hippocampal slices (Blackband et al., 1997) and have shown that slices perturbed by ouabain and N-methyl-
In the present study, water diffusion was monitored in rat hippocampal slices before and after treatment with different doses of the neurotoxin A23187. This carboxylic acid calcium ionophore rapidly diminishes calcium gradients across cell membranes (Pressman, 1976) as is known to occur in vivo after ischemic or traumatic injury (Choi 1988). Previous studies have shown that neuronal cultures treated with high doses of calcium ionophores, such as A23187, experienced rapid loss of calcium homeostasis and pathologic cell swelling (Chan et al., 1998; Gwag et al., 1999; Takadera and Ohyashiki, 1997). We hypothesized that this cellular response would be detectable in rat hippocampal slices using diffusion-weighted MRI and correlative histology. Unlike previous diffusion studies of nervous tissue injury, using A23187 allows this study to uniquely evaluate water-diffusion changes after calcium overload (Petersen et al., 2000), a downstream integrating event in many forms of acute brain injury (e.g., ischemic stroke or traumatic brain injury) (Choi, 1988; McIntosh et al., 1998). Results of this investigation indicated that the biexponential diffusion parameter Ffast and diffusion-weighted MRI at high b-values provide rapid and sensitive measures of A23187-induced tissue injury that are correlated with neuronal cell swelling and overt histopathology in selectively vulnerable regions of the rat hippocampal slice.
MATERIALS AND METHODS
Brain slice procurement
The University of Florida Institutional Animal Care and Use Committee approved the use of laboratory animals for this study. Rat hippocampal slice procurement has been described previously (Aitken et al., 1995). Briefly, male Long-Evans rats (250 to 350 g) were anesthetized with isoflurane and decapitated. The brain was removed and placed into ice-cold artificial cerebrospinal fluid (aCSF) (120-mmol/L NaCl, 3-mmol/L KCl, 10-mmol/L glucose, 26-mmol/L NaHCO3, 2-mmol/L CaCl2, 1.5-mmol/L KH2PO4, and 1.4-mmol/L MgSO4) gassed with 95% O2 and 5% CO2 to maintain a pH of 7.4. The aCSF osmolality was 300 ± 1 mOsm/kg as determined by Osmette A freezing point depression osmometer (Precision System, Natick, MA, U.S.A.). Both hippocampi were dissected and cut orthogonal to the septohippocampal axis into 500-μm-thick sections with a McIlwain tissue chopper within 10 minutes of decapitation. Hippocampal slices remained immersed in ice-cold aCSF for 1 hour after decapitation to minimize procurement-induced ischemic damage (Newman et al., 1992). Slices then were warmed gradually to room temperature (20°C) for the MRI experiment and placed into a multislice perfusion chamber (Shepherd et al., 2002).
Slices were perfused continuously with aCSF (2 mL/min) while the perfusion chamber was lowered into the magnet and during pilot image acquisition. Although perfusion was discontinued during all diffusion measurements (described below), calculations based on the published metabolic rates of nervous tissue (Magistretti, 1999) indicate that the typical 6-mg hippocampal slice consumes less than 0.2% of the glucose available in the perfusion chamber (containing 3.0 mL aCSF) during a 35-minute MRI acquisition. Further, it has been shown previously that intermittent cessation of aCSF flow does not affect slice viability for at least 8 hours after slice procurement (Shepherd et al., 2002).
Diffusion MRI of rat hippocampal slices
All MRI data were obtained at room temperature using a 15-mm birdcage coil interfaced to a Bruker 17.6-T vertical wide-bore magnet and console. Pilot multislice axial, sagittal, and coronal T1 and diffusion-weighted imaging sequences were used to locate the perfusion chamber, then to optimize the positions of axial magnetic resonance-defined slices through the center of the 500-μm-thick rat hippocampal slices. Diffusion-weighted images were acquired using a pulsed gradient spin-echo multislice sequence with a diffusion time of 14 milliseconds and an echo time of 30 milliseconds. This diffusion measurement consisted of a series of 8 diffusion-weighted images (128 × 64 matrix, 1.5-cm field of view, 2-second repetition, 30-millisecond echo time, 2 averages, δ = 3 milliseconds, Δ = 15 milliseconds) using diffusion gradients aligned perpendicular to the plane of the MRI images in linear strength increments from 0 to 940 mT/m. This resulted in 8 diffusion-weighted images with b-values between 7 and 8080 s/mm2 (including imaging terms). Image slice thickness was 300 μm with 117 × 234-μm in-plane resolution and the protocol required 34 minutes for completion. Additional pilot images were acquired between diffusion measurements to monitor for slice movement due to aCSF perfusion; data from hippocampal slices that moved during the experiment were rejected (n = 1).
Slice perturbation with A23187
Before slice procurement, rat hippocampal slices were randomly assigned to treatment by control aCSF, or aCSF containing 10–μmol/L A23187 or 50-μmol/L A23187. Hippocampal slices were exposed to higher doses of A23187 than previous studies in neuronal cultures (Mattson et al., 1991; Petersen et al., 2000) because the slice model was composed of high-density, heterogeneous cell populations studied at lower temperatures. Dimethyl sulfoxide (DMSO) was required to solubilize A23187 in aCSF, so control aCSF contained 0.1% DMSO. The additions of 0.1% DMSO with or without A23187 did not affect the pH or osmolality of the aCSF solution. Previous studies in our laboratories suggest that 0.1% DMSO does not affect calcium movement in slices as indicated by the absence of calcium-mediated calpain activation (Zhao et al., 1999). Each of the aCSF treatment solutions were gassed with 95% O2 and 5% CO2 throughout the experiment. All slices were perfused with standard aCSF during pilot scans and before initial baseline diffusion measurements were acquired. Slices then were perfused with the assigned treatment aCSF for 20 minutes, then perfusion was stopped and a post-treatment diffusion protocol obtained. Preliminary studies demonstrated that an initial perfusion duration of 20 minutes with treatment aCSF was sufficient for perfusate exchange. A cycle consisting of 6 minutes of perfusion with the assigned treatment aCSF followed by an additional diffusion measurement (34 minutes) was repeated serially until 12 hours after initial brain slice procurement (each complete cycle required 40 minutes).
The initial treatment period for slices occurred at slightly different times after brain slice procurement because of variations in the time required for hippocampal slice placement and pilot MRI scans (mean time delay from procurement to baseline diffusion measurement was 3.1 ± 0.4 hours). Therefore diffusion data from rat brain slices required assignment to time-bins for statistical comparisons. The baseline diffusion measurement for each slice was assigned to 0 minutes and subsequent data were assigned to time points increasing in 40-minute increments. Although data were reassigned to time points relative to the initial MRI measurement, diffusion data acquired more than 12 hours after slice procurement were excluded from the analysis.
Analysis of diffusion data
To analyze changes in the diffusion-weighted signal intensity in rat hippocampal slices after treatment with the various aCSF solutions, a region of interest (ROI) was drawn on the images to enclose the entire hippocampal slice. Previous studies demonstrated that diffusion-weighted water signal attenuation in rat hippocampal slices is nonmonoexponential at high b-values (Buckley et al., 1999). A biexponential equation (Eq. 1) was fitted to the diffusion-weighted signal attenuation in the rat hippocampal slice ROIs using the Levenberg-Marquandt nonlinear least-squares fitting routine:
where So is signal intensity without diffusion weighting, Ffast is the fraction of water with fast apparent diffusion coefficient (ADC), and Dfast and Dslow represent the ADCs of the fast and slow diffusing water components, respectively. Observed changes over time from baseline in the biexponential parameters of the three treatment groups (control, 10-μmol/L A23187, and 50-μmol/L A23187) were compared statistically using a two-way repeated-measures analysis of variance (ANOVA) with one factor repeated (Sigma Stat 2.03). If the ANOVA test detected a statistically significant difference among treatment groups at a particular time point, a Tukey multiple comparisons test was used to isolate treatments that differed significantly from one another. Statistical significance for all tests was defined as P < 0.05.
Histology of rat hippocampal slices
Because it proved difficult to consistently recover rat hippocampal slices used in the diffusion MRI experiments for subsequent study, three additional rats were required to provide slices for histologic correlation. These slices experienced the same experimental procedure as slices used in the diffusion MRI measurements and at 12 hours after procurement, the slices were gently removed from the perfusion chambers and immersion-fixed with 4% paraformaldehyde in phosphate-buffered saline. Some slices were fixed either immediately after procurement or before perfusion with treatment aCSF (4 hours after procurement) to serve as additional controls. In addition, a few slices that were recovered after the MRI experiments were fixed for histologic analyses to assess whether differences existed for slices treated in the magnet versus slices treated in the correlative protocol.
The fixed slices were embedded in paraffin wax, cut into 8-μm-thick sections, mounted onto slides and stained with hematoxylin and eosin (H&E). Most slices were sectioned parallel to the plane of the slice (orthogonal to the septotemporal axis of the rat hippocampus), but in some slices, sections were cut perpendicular to the plane of the tissue (cross section). These sections were used to determine whether cells at different depths from the tissue surface were affected differently by the various aCSF treatment solutions. Superficial and central planar sections, stained with H&E, also were compared to assess the potential variable effects of tissue depth on tissue response to A23187 treatment.
In all H&E-stained sections, morphologic characteristics of granule and pyramidal neurons from the dentate gyrus, CA1, CA2, and CA3 fields were noted. Nuclear changes such as pyknotic and eosinophilic nuclei, karyorrhexis, or karyolysis were noted. The cytoplasm of cells was observed for evidence of eosinophilia, microvacuolation, and loss of structure or fragmentation. The surrounding neuropil, in such regions as the stratum radiatum, were also inspected for vacuolization and other pathologic changes. These features were compared between the slices treated with control, 10-μmol/L A23187, or 50-μmol/L A23187 aCSF solutions for differences in injury-induced cellular morphology and pathology.
RESULTS
Diffusion-weighted MRI of rat hippocampal slices
Twenty-nine of the 35 rat hippocampal slices procured from 17 rats were included for the diffusion MRI aspects of this investigation. The other six slices were rejected because of perfusion chamber flooding (n = 3), susceptibility artifacts due to air bubbles in the perfusion chamber (n = 2), and slice movement (n = 1). In comparing the three treatment groups, there were no significant differences for the post-procurement time when the first baseline diffusion measurements were obtained (ANOVA, P = 0.265) or for the time when slices were subsequently treated (ANOVA, P = 0.393). Mean time to treatment for all slices (3.8 ± 0.5 hours) closely matched the time when correlative rat hippocampal slices were treated outside the magnet for histology studies (4 hours). Diffusion measurements were obtained on all 29 slices up to 6 hours after treatment, on 28 of 29 slices up to 7.3 hours after treatment, and on 20 of 29 slices 8 hours after treatment.
Preliminary high-resolution diffusion-weighted images were obtained of rat hippocampal slices to determine achievable signal-to-noise ratio (SNR) and spatial resolution per unit time. Such images (Fig. 1) exhibited the detailed laminar anatomy of the hippocampus, but required long acquisition times (4.5 hours per b-value measurement) without sufficient perfusion to maintain slice viability (Shepherd et al., 2002). For this study, such high spatial resolution was sacrificed for better temporal resolution of the water-diffusion changes that accompany slice perturbation with A23187 (4.2 minutes per b-value measurement). In Fig. 2, the lower-resolution images show the signal intensity changes that occur after treatment with 10-μmol/L A23187. Plotting the signal intensity of the hippocampal slice ROI versus b-value gave nonmonoexponential diffusion-weighted signal attenuation curves. Figure 3 shows that treatment with 10-μmol/L A23187 caused a progressive decrease in the rate of diffusion-weighted signal attenuation over the 8 hours. This change was most evident at b-values above 4000 s/mm2 (see box B). Similar results to those shown in Figs. 2 and 3 were also observed for slices treated with 50-μmol/L A23187.
A diffusion-weighted magnetic resonance image with 59-μm in-plane resolution (
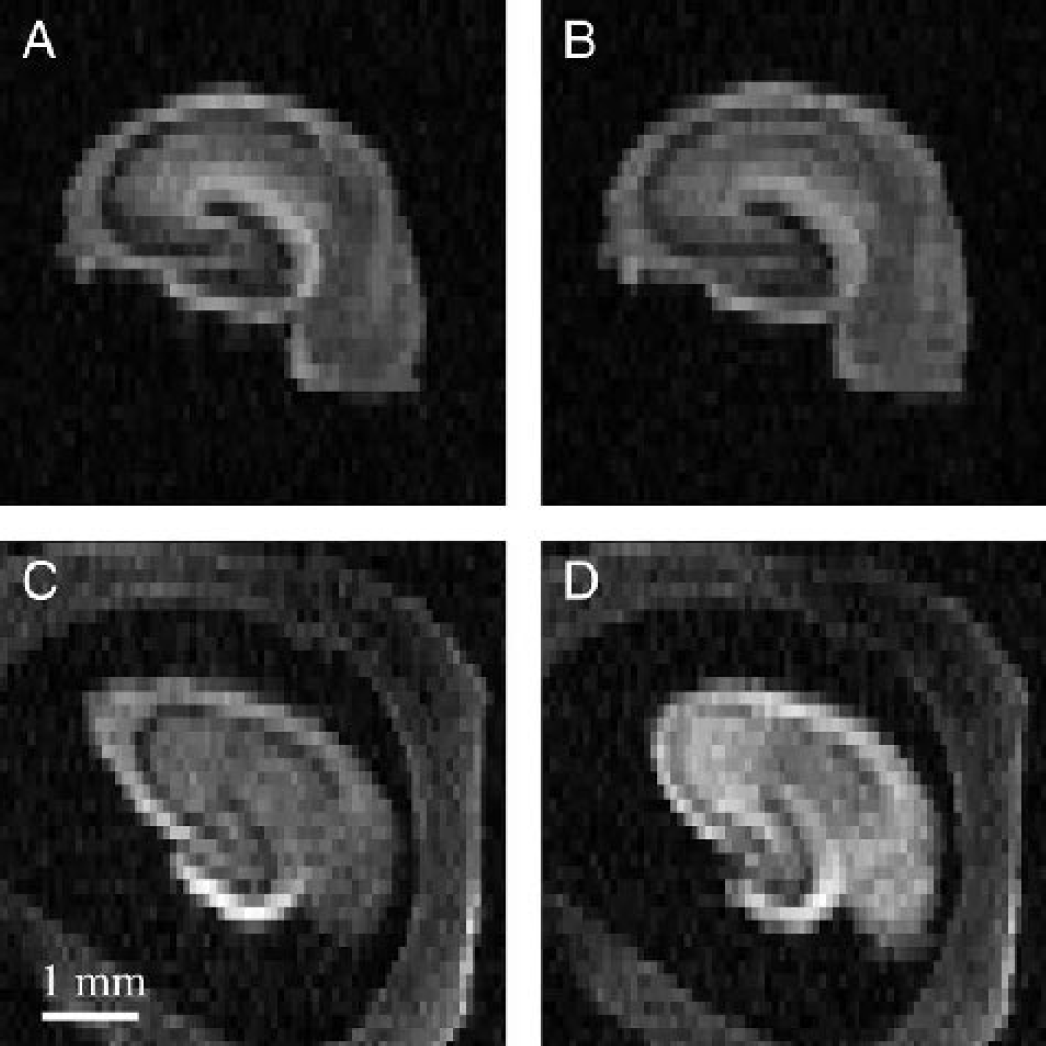
Typical diffusion-weighted images (b = 7,977 s/mm2) of rat hippocampal slices. Panels
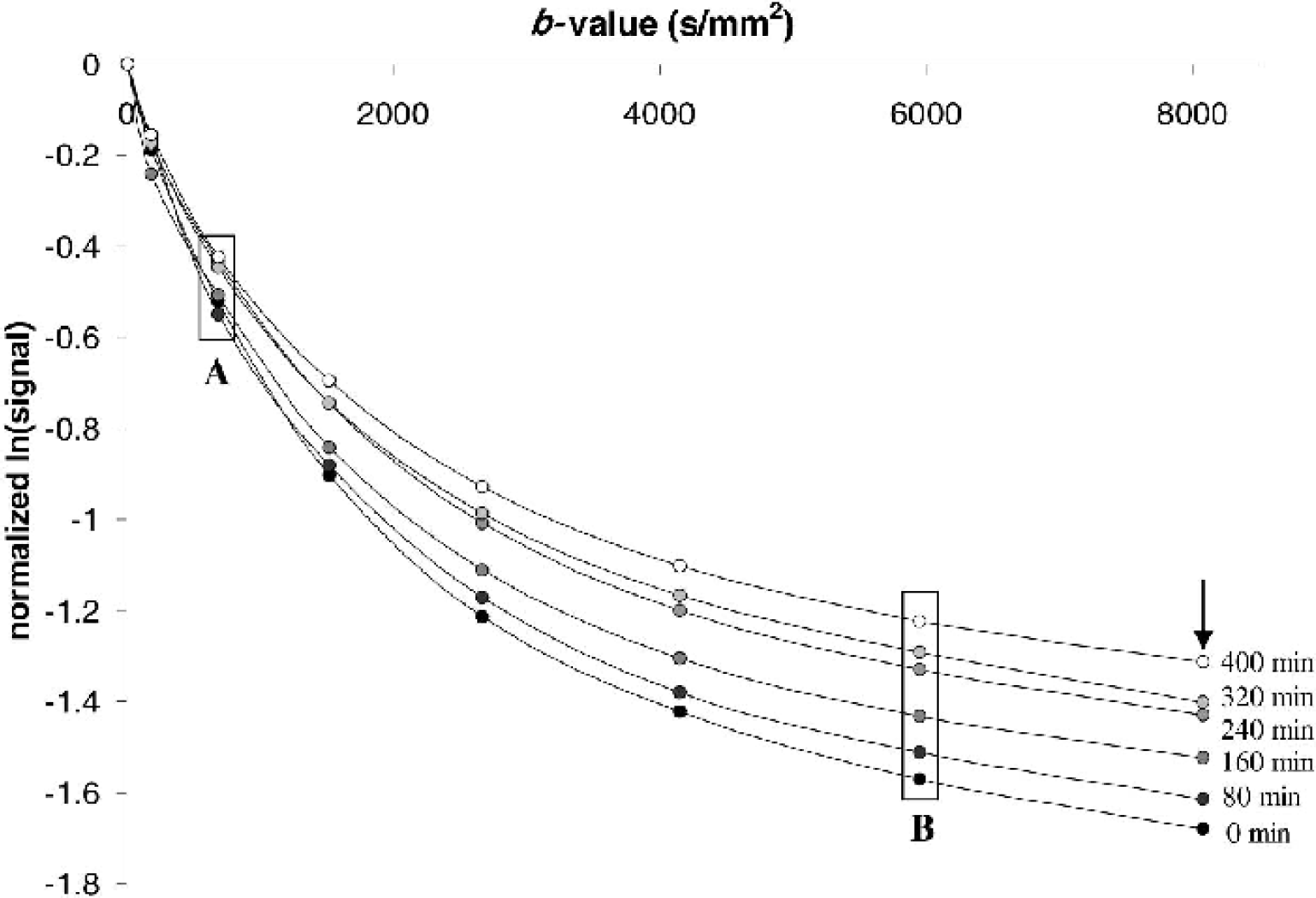
Typical semilog plot showing temporal changes in the diffusion-weighted signal attenuation curves for a rat hippocampal slice treated with 10-μmol/L A23187. Each point represents the log signal intensity for the slice ROI in a diffusion-weighted image normalized to the signal intensity of the first image (b = 7 s/mm2). Although diffusion data were collected every 40 minutes, only alternating data from baseline (0 minutes), 80, 160, 240, 320, and 400 minutes are shown for clarity. Comparison of boxes labeled A and B illustrates that the signal intensity changes after A23187 treatment are better resolved at higher b-values given sufficient signal-to-noise. The arrow indicates approximately where data from Fig. 2 (b = 7,977 s/mm2) would be plotted in the signal-attenuation curve.
Biexponential analysis of diffusion in hippocampal slices treated with A23187
The diffusion-weighted signal attenuation curves of rat hippocampal slices (Fig. 3) were well described by the biexponential function (Eq. 1) (R2 > 0.99). The biexponential-derived diffusion parameters for all 29 rat hippocampal slices before treatment (mean ± SD) were 0.601 ± 0.057 for Ffast, 1.14 ± 0.14 × 10−3 mm2/s for Dfast and 0.067 ± 0.010 × 10−3 mm2/s for Dslow. These values were comparable to previous reports of water diffusion in rat hippocampal slices (Buckley et al., 1999; Bui et al., 1999; Shepherd et al., 2002) and were qualitatively similar to biexponential diffusion parameters reported for in vivo human brain (Mulkern et al., 1999). There were small yet statistically significant differences in the baseline biexponential parameters Ffast and Dslow for slices assigned to the three different aCSF treatment groups (Table 1). Possible explanations for these differences are addressed in the Discussion.
Baseline diffusion characteristics for rat hippocampal slices in each of the three treatment groups

Data are mean ± SD.
Groups that were statistically different from one another as determined post-ANOVA by a Tukey multiple comparisons test (P < 0.05). DMSO, dimethyl sulfoxide, ANOVA, analysis of variance, Ffast, fast diffusing water fraction, Dfast, fast apparent diffusion coefficient, Dslow, slow apparent diffusion coefficient.
The biexponential water diffusion parameters for control slices before and immediately after treatment with aCSF containing 0.1% DMSO were not statistically different (ANOVA, P > 0.05). The mean Ffast of DMSO-treated slices decreased stepwise by approximately 5% only 9 hours after procurement. This change, although not statistically significant, represented a trend present in the data from most individual slices. For the three treatment groups, there were statistically significant interactions between time and treatment in the change from baseline for the diffusion parameter Ffast (ANOVA, P <0.001), but not for Dfast (P = 0.945) or Dslow (P = 0.870). Figure 4 depicts the mean percentage change from baseline in the biexponential parameters Ffast for slices treated with aCSF containing 0.1% DMSO, 10–μmol/L A23187, and 50-μmol/L A23187. Compared to vehicle-treated slices, the percentage change in the fraction of fast diffusing water (Ffast) for slices treated with 50-μmol/L A23187 was significantly different for all time points after baseline (Tukey, P <0.05). Changes from baseline for slices treated with 10-μmol/L A23187 were significantly different than vehicle-treated slices for all time points after 80 minutes (Tukey, P < 0.05). Although the dose of A23187 (10 or 50 μmol/L) predicted when the Ffast diffusion parameter of A23187-treated slices would first differ statistically from vehicle-treated slices, at no time point were data from the 10-μmol/L or 50-μmol/L A23187-treated slices statistically distinct from each other.
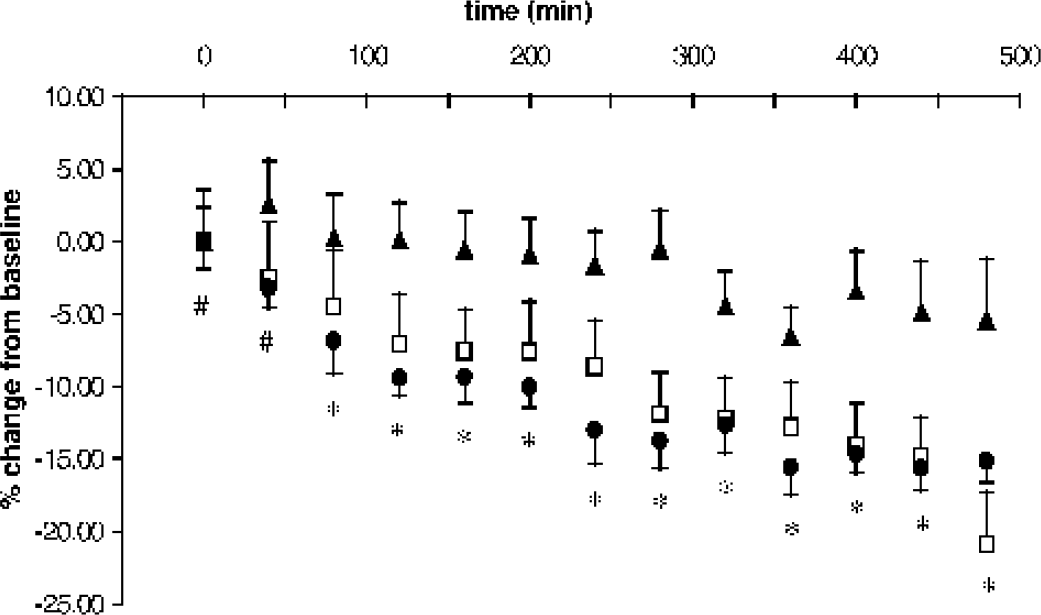
Mean percentage change from baseline over time for the fraction of fast diffusing water in rat hippocampal slices treated with aCSF containing 0.1% DMSO vehicle (▴; n = 9), 10-μmol/L A23187 (□; n = 10) or 50-μmol/L A23187 (• n = 10) (mean ± SEM). At early time points, slices treated with 50-μmol/L A23187 statistically differed from vehicle-treated slices (#) whereas at later time points slices treated with either 10–μmol/L or 50-μmol/L A23187 differed from vehicle-treated slices (*) (P < 0.05).
Histology of A23187-induced pathologic cell swelling
Correlative H&E histology of the rat hippocampal slices exhibited clear dose-related differences in particular hippocampal regions (Fig. 5). In rat hippocampal slices treated with A23187, susceptible neurons typically manifested pathologic change by intense microvacuolation of the neuronal perikaryon and stranded, slightly pyknotic nuclei. These features are typical oncotic changes that frequently lead to the appearance of necrotic cells at later time points than observable for this study (Majno and Joris, 1995). The described cellular changes were most prominent in aspects of the CA1 cell band (CA1a), but also present in the ends of the CA3 cell band (CA3a and CA3c) and the granule cell layer in the internal blade of the dentate gyrus. The changes were more severe in slices treated with aCSF containing 50-μmol/L A23187 than slices treated with 10-μmol/L A23187. Other regions of the hippocampus, such as CA2, appeared relatively unaffected by A23187, although the neuropil surrounding the affected pyramidal and granule cells did exhibit some staining pallor and vacuolation as well (Fig. 5).
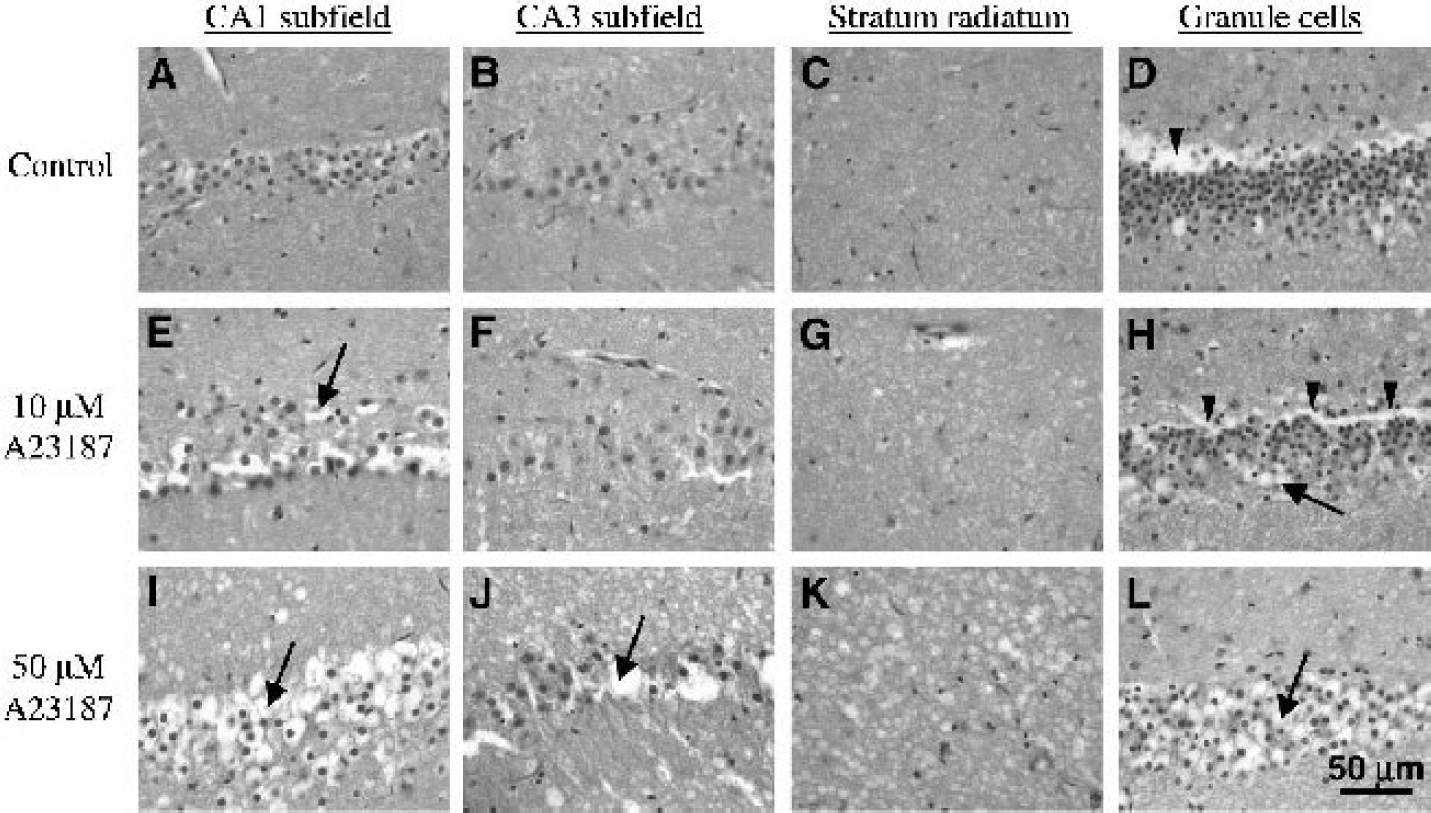
Correlative histology of rat hippocampal slices 8 hours after treatment with DMSO vehicle (
Similar to previous studies (Newman et al., 1992), comparison of H&E-stained cross sections and planar sections from different depths in the hippocampal slices did not suggest different reactions to A23187-treatment based on tissue depth. Additional control slices taken shortly after slice procurement (at 30 minutes after rat decapitation) and before perfusion with aCSF treatment solutions (at 4 hours after procurement) did not show differences from hippocampal specimens obtained by intracardiac perfusion of normal rats with 4% paraformaldehyde except for some minor pathology of the internal blade of the dentate gyrus (see Discussion). The histologic appearance of the few slices recovered from the diffusion MRI studies also did not differ from correlative slices with the same aCSF treatment assignment (data not shown).
DISCUSSION
Pyramidal neurons and granule cells in rat hippocampal slices treated with A23187 showed substantial cytoplasmic microvacuolation indicative of pathologic cell swelling. The neuropil apposed to such damaged neurons also appeared vacuolated. The neuronal pathology after A23187 treatment resembles early ischemic cell changes (Brierley et al., 1973) and electron microscopy studies have correlated such microvacuolation with mitochondrial swelling (Auer and Benveniste, 1997). Although pathologic oncotic swelling is clearly evident, it is difficult to determine whether some neurons ruptured during the experiment because cellular membranes rendered unstable by A23187 treatment might also rupture on fixation (Auer et al., 1985). These findings concur with previous descriptions of pathologic swelling in cultured neurons treated with the calcium ionophores ionomycin or A23187 (Takadera and Ohyashiki, 1997; Chan et al., 1998; Gwag et al., 1999) and are appropriate to the mechanisms of A23187 toxicity. A23187 initiates a rapid intracellular calcium overload in the neuropil that spreads to the neuronal perikaryon at higher doses (Gwag et al., 1999). This calcium overload then provokes mitochondrial permeability transition pore formation and pathologic cell swelling (Petersen et al., 2000).
Unlike previous neuronal cell culture studies (Chan et al., 1998; Petersen et al., 2000), A23187 treatment did not affect all neurons in the hippocampal slices equally. This anatomically selective pattern of pathology in slices after A23187 treatment reproduces the selective vulnerability of the CA1 and CA3 hippocampal regions to ischemic and traumatic injuries (Kotapka et al., 1994). Selective vulnerability of the hippocampus has been attributed to differences in N-methyl-
Vulnerable regions of the rat hippocampal slices also showed dose-related differences in the magnitude of pathological cell swelling induced by A23187 (see Fig. 5). These dose-related histopathologic differences were difficult to distinguish with the diffusion MRI measurements using the whole-slice ROI method because regions affected by A23187 in a dose-related fashion were volume-averaged with relatively unaffected regions. However, dose-related differences were noted after A23187 treatment when the Ffast of slices became statistically distinct from control slices. With either A23187 dose, the Ffast for slices was reduced 7% to 10% from baseline during the first 2 hours after treatment, but it then took an additional 6 hours of A23187 treatment to double this reduction.
The biophysical interpretation of Ffast reductions observed in rat hippocampal slices treated with A23187 is not trivial because the biexponential model of water diffusion in nervous tissue is too simplistic. Although the fast and slow diffusing components of water diffusion can not be directly attributed to the extracellular and intracellular compartments respectively, studies have shown that changes in the Ffast do correlate with perturbation-induced changes in the relative size of the intracellular compartment (Niendorf et al., 1996; Thelwall et al., 2002). There are several additional biophysical properties of nervous tissue that affect water diffusion and are unaccounted for by the biexponential model; for example, transmembrane water exchange rates, intracellular restriction, and extracellular tortuosity also influence the biexponential diffusion parameters obtained (including Ffast) (Thelwall et al., 2002). When neurons are pathologically injured by A23187 treatment, these additional biophysical features are also likely to be altered and influence the biexponential parameters obtained. Given sufficient time and SNR, future studies could use more complex analytical models, such as the one developed by Stanisz et al. (1998), to better describe the biophysical tissue properties of hippocampal slices altered by A23187-induced pathologic cell swelling.
Despite these limitations, the biexponential model does provide a more complete description of nervous-tissue water diffusion than the monoexponential ADC fits currently in clinical use, and it is reasonable to cautiously interpret changes in Ffast to correlate with changes in the relative size of the intracellular compartment. Previous clinical studies have shown decreased ADC in acute ischemic brain followed by an increase in ADC beyond normal values several days later (Beaulieu et al., 1999). These diffusion changes, noted in animal models of both ischemic and traumatic injury, may be due to cellular swelling followed by lysis (Assaf et al., 1997; Knight et al., 1994;). Because high doses of A23187 caused a similar yet accelerated pattern of cell swelling and lysis in neuronal cultures (Chan et al., 1998; Takadera and Ohyashiki, 1997), a biphasic pattern of Ffast changes was hypothesized to be observable for Ffast within the time span of this experiment on rat hippocampal slices treated with A23187. Thus, as cells swell, Ffast would decrease, but then as cells rupture, Ffast would be predicted to increase.
Because potential dose-related differences in Ffast were only observable during the first 80 minutes after A23187 treatment, the results of this study might suggest that there were a limited number of A23187-sensitive cell populations within the rat hippocampal slices. At high doses of A23187, these cells swell more quickly (reducing Ffast), but the A23187-sensitive populations become saturated such that the high and low doses of A23187 become indistinguishable at later time points. However, the correlative histology suggests that more neurons were recruited to pathologic swelling by the higher A23187 dose. Thus, a better explanation may be that in slices treated with 50-μmol/L A23187, 2 hours after treatment the Ffast increases due to lysis in the initially injured cell population may have been obscured by Ffast decreases due to neurons subsequently recruited to pathologic cell swelling. Thus, dose-related differences in A23187 injury may be obscured at later time points by volume averaging of not only healthy and injured neurons, but also by volume averaging of neurons at different stages of cellular injury (swelling and lysis).
Nonetheless, this study shows that the Ffast diffusion parameter may be used as a sensitive correlate of cellular swelling after acute brain injury in future clinical evaluations. However, compared to correlative histology, the diffusion MRI measurements failed to observe the appearance of substantial dose-related, region-specific pathologic changes in the rat hippocampal slices because the present MRI analysis did not examine individual anatomical regions. This also may explain the failure to observe a biphasic Ffast response in hippocampal slices treated with the higher dose of A23187. Because the SNR advantages of a 17.6-T magnet were used to make water diffusion measurements suitable for the biexponential model with high temporal resolution, the ROI volume chosen for this study (2 mm3) was comparable to typical MRI voxel volumes in clinical diffusion studies (Beaulieu et al., 1999). As such, this study highlights how potential variations in regional, cell subtype, and dose-related responses to acute injury may pass unobserved in diffusion measurements that sacrifice spatial resolution to obtain adequate signal-to-noise ratios in practical scan times, particularly when correlative techniques are not used. Clearly, this will continue to be a challenging problem clinically. Previous studies in human patients also have suggested that ADC changes in ischemic stroke may be heterogeneous and might reflect “different temporal rates of tissue evolution toward infarction” (Nagesh et al., 1998).
The time and hardware limitations in present clinical imaging protocols make it difficult to collect data sufficient even for biexponential analysis of water diffusion. Alternatively the diffusion data can be examined without biexponential analysis by simply comparing images of slices at particular diffusion weightings before and after A23187 exposure. A23187 treatment increased diffusion-weighted signal intensity for all rat hippocampal slices treated with either 10-μmol/L or 50-μmol/L A23187, and these changes were noted at all nonzero b-values. In Fig. 2, for example, diffusion-weighted slice signal intensity at b = 7,977 s/mm2 increased 42% after 8 hours treatment with 10-μmol/L A23187. Although the SNR is decreased at higher b-values, the increases in diffusion-weighted signal intensity between slice MRI images at baseline and at different time points after A23187 treatment were better resolved with increasing b-value (for example, see Fig. 3 boxes A and B). These results support previous reports that suggested stronger diffusion-weighting (higher b-values) may increase the tissue characterization capabilities of diffusion-weighted MRI in human patients when there is sufficient contrast-to-noise (Meyer et al., 2000; Sze and Anderson, 2000). This may be because high b-value diffusion-weighted signal intensity emphasizes water diffusion in the intracellular compartment where most of the initial pathologic injury to tissue occurs. Future studies will be required to further investigate the clinical merits of high b-value diffusion-weighted MRI.
There are some caveats to the interpretation of the diffusion MRI data presented. The comparison of baseline diffusion parameters for slices based on treatment group assignment (Table 1) revealed some significant pretreatment differences in the diffusion parameters Ffast and Dslow. Procurement conditions were unlikely to contribute significantly to these baseline differences because slices were procured under neuroprotective hypothermic conditions (see below). Instead, the initial diffusion differences may be attributed to differences in positioning the MR image location within the hippocampal tissue (see Materials and Methods). The undesired inclusion of aCSF within the MRI-defined slice can increase the amount of freely diffusing water present in the slice ROI, which then alters the biexponential diffusion parameters obtained. The study design controlled for such pretreatment differences by examining change in water diffusion in individual slices from baseline after treatment.
Brain slices have some limitations as models of in vivo nervous tissue. Rat hippocampal slices were procured at low temperatures to minimize procurement-induced injury before A23187 treatment (Newman et al., 1992; Aitken et al., 1995). In addition, water diffusion changes were measured in rat hippocampal slices treated with A23187 at room temperature instead of 37°C because this simplifies slice perfusion-chamber design and permits hippocampal slices sufficiently thick (500 μm) for MRI study (Aitken et al., 1995). The neuroprotective effects of hypothermia suggest that the measured diffusion responses in this tissue injury model may underestimate the responses of in vivo nervous tissue. In addition, the diffusion coefficient of unrestricted water will increase approximately 50% when heated from room temperature to 37°C (Harris and Woolf, 1980). This may limit direct extrapolation of the data presented; however, the study focused on relative changes that should be comparable between the slice model and in vivo subjects.
Despite the neuroprotective effects of hypothermia, some histopathology was notable in control slices 12 hours after slice procurement (e.g., CA1 and the internal blade of the granule cell layer). Although not statistically significant, there also was a 5% decrease in mean Ffast for control slices at the conclusion of the experiment. Further, the conditions under which brain slice experiments occur (e.g., slice procurement and intermittent aCSF perfusion) might enhance A23187-induced tissue injury (Newman et al., 1992). These findings indicate that brain slices can not perfectly model healthy and pathologic in vivo nervous tissue, yet over the duration of the present experiment, changes to control slices were of limited magnitude compared to the histopathology and water diffusion changes due to pathologic swelling induced by A23187 treatment. Thus, hippocampal slices provided a workable tissue model for comparing water diffusion in injured and viable nervous tissue. Brain slices tolerate long imaging periods in very-high-field magnets that enable data collection not possible in current clinical scanners. Thus, brain slice methodology will remain highly relevant in the future to developing a better understanding of the biophysical basis for water diffusion changes after acute brain injury.
CONCLUSION
This study successfully used diffusion-weighted MRI of rat hippocampal slices to study the acute temporal evolution of multicomponent water-diffusion changes after A23187 treatment. Brain slices provided a stable and highly controllable nervous tissue model. Data obtained from this model of acute brain injury suggest that the biexponential diffusion parameter Ffast may be a sensitive correlate of cellular swelling in nervous tissue and that diffusion changes after acute brain injury may be intravoxel volume-averaged summations of responses from anatomically or temporally distinct healthy and pathologically injured cell populations. These results suggest that regional analysis of A23187-treated hippocampal slices may improve our understanding of the unique and heterogeneous morphologic processes that follow acute cellular injury and contribute to improved clinical measurements of diffusion in human stroke and traumatic brain injury patients. In addition, data from this study concur with findings of previous clinical studies that diffusion-weighted images at high b-values may offer improved characterization of normal and pathologic nervous tissue. Future perturbation studies of rat hippocampal slices may be capable of reconciling complex, data-intensive mathematical models of nervous-tissue water diffusion with diffusion MRI data obtained from human patients with acute brain injuries.
Footnotes
Acknowledgments:
The authors thank Dr. Michael King for reviewing the manuscript and acknowledge the valuable technical assistance of Deborah Dalziel, Daniel Plant, and Barbara O'Steen.