Abstract
The distribution of brain iron is heterogeneous, but the mechanism by which these regional differences are achieved and maintained is unknown. In this study, the authors test two hypotheses related to brain iron transport. The first is that there is regional variability in the profile of proteins associated with iron transport and storage in the brain microvasculature. The second hypothesis is that the iron status of the brain will dictate the response of the protein profile in the microvasculature to changes in systemic iron status. The profile analysis consists of transferrin (iron transport), ferritin (iron storage), transferrin receptor (iron uptake), and divalent metal transporter 1 (release of iron from endosomes). An additional protein involved in cellular iron efflux, ferroportin, was not detected in brain microvasculature. The results show that there are significantly higher levels of these proteins in the microvasculature from each area of the brain compared to a whole brain homogenate, but no regional differences within the microvasculature. The levels of ferritin observed in the microvasculature indicate that the microvascular endothelial cells have significant iron storage capacity. There are no significant changes in the regional protein profiles in response to systemic iron manipulation when brain iron status was normal. In contrast, in Belgrade rats, whose brain is iron deficient, the expression of both divalent metal transporter 1 and transferrin receptor was increased compared with control in almost all brain regions examined, but not transferrin or ferritin. These findings indicate that regional brain iron heterogeneity is not maintained by differences in microvascular iron-management protein levels. The results also indicate that brain iron status dictates the response of the microvascular protein profile to systemic iron manipulation.
Iron is abundant in the brain and has a distinct pattern of distribution. Some brain regions such as the substantia nigra and deep cerebellar nuclei are particularly enriched in iron relative to other regions (Koeppen, 1995; Sourander and Hallgren, 1958). In general, the concentration of iron in regions of the brain associated with motor functions is greater than those not directly involved in motor activity (Drayer et al., 1986; Spatz, 1922). Iron participates in numerous catalytic reactions and regulatory processes in neural cells, such as ATP production, myelin and dopamine synthesis. The access of iron to the brain is limited by the presence of a blood-brain barrier. There are a number of unanswered questions regarding iron acquisition by the brain: (1) how a particular brain region becomes enriched with iron; (2) how iron acquisition by the brain is regulated; and (3) the site of regulation of brain iron acquisition.
Regulation of brain iron acquisition has not been directly addressed either at a regional or whole brain level. Regional loss and acquisition of iron was recently demonstrated in an animal model in which dietary iron levels were manipulated (Erikson et al., 1997; Pinero et al., 2000) and regional accumulation of iron is a consistent observation in a number of neurodegenerative disorders. It seems logical to begin to address the question of regulation of brain iron acquisition at the level of the blood-brain barrier. The barrier properties of brain vascular endothelial cells present a need for active transport of compounds into the brain. Indeed, there is a high density of transferrin receptor expression on the brain microvasculature in the adult human brain (six to 10 times higher than the expression on the brain parenchyma; Kalaria et al., 1992). Presumably, there is a control mechanism to regulate this transport. However, if there is a regulatory mechanism, there must also be a signal that controls the regulatory mechanism. What is the source of the signal? Signals could be sent from neural cells to the vasculature to indicate specific iron needs for that cell or group of cells. This signal may modulate the level of iron transport proteins expressed in the vasculature endothelia, or possibly trigger a release of iron stored in the endothelial cells. Alternatively, changes in plasma iron may be the signal to alter endothelial expression of ironmanagement proteins.
In this study, we test two hypotheses. The first hypothesis is that there is innate variability in the profile of microvascular iron transport and storage proteins from each brain region. The second hypothesis is that changes in the protein profile of the microvasculature in response to changes in systemic iron status will be dictated by brain iron status and may be region specific. The results of these studies will provide insight into the site of regulation of brain iron acquisition and the source of the regulatory input (brain versus plasma). Two animal models, the Sprague-Dawley and the Belgrade rat, were used to test these hypotheses. The Belgrade rat has a mutation in the divalent metal transporter 1 (DMT1) that results in a hypochromic, microcytic anemia, despite nearly 100% transferrin saturation (Fleming et al., 1998). At the cellular level, the effect of the DMT1 mutation is that an increased amount of transferrin-bound iron is recycled from the endosome to the extracellular space (Garrick et al., 1993). We have previously shown that the amount of histochemically detectable iron in the brain of Belgrade rats is decreased (Burdo et al., 1999), an observation supported by magnetic resonance imaging analysis (Zywicke et al., 2002). The Belgrade rats present the opportunity to study the protein profile in the brain microvasculature from an iron deficient brain and the alterations in that profile when plasma transferrin is capable of delivering sufficient iron. This model differs from the systemic iron-deficient paradigm in the Sprague-Dawley rat where insufficient plasma iron is available while the brain iron status is normal. The outcome of these studies will provide the data necessary to generate hypotheses regarding the source of the signals controlling the protein profile in the microvasculature and ultimately brain iron uptake.
MATERIALS AND METHODS
Adult Sprague-Dawley rats (Harlan, Indianapolis, IN, U.S.A.) or rats that were homozygotic or heterozygotic for the DMT1 mutation (Belgrade rats) were obtained from Michael Garrick (State University of New York, Department of Biochemistry, Buffalo, NY, U.S.A.) for this study. The animals were treated in one of three ways: fed a normal-iron rat chow, made acutely iron deficient, or iron supplemented. Iron supplementation was accomplished by injecting iron-dextran (Sigma, St. Louis, MO, U.S.A.; 5 mg i.p.) 7 days before death. Iron deficiency was achieved by feeding the rats an iron-poor rat chow (product number TD 80396, Harlan-Teklad, Indianapolis, IN, U.S.A.) and weekly tail vein bleeding until hematocrit levels decreased to a value of approximately 30%. This approach was taken to test the effects of an acute change in systemic iron status on the protein profile. Belgrade b/b rats were either similarly iron supplemented or fed a normal-iron rat chow. No iron deficient treatment was used for the b/b rats because of their anemic condition.
The rats were killed by an overdose of sodium pentobarbital (120 mg/kg i.p.). The brains were removed, the meninges separated from the brain, and the brains dissected into five regions: cortex, cerebellum, striatum, thalamus, and hippocampus. Each individual brain region was one sample (N = 1), and analysis was based on a sample size of n = 4 for each group. Each of the brain regions was placed in a 1:5 (wt:vol) ratio of microvessel isolation buffer (MVB). consisting of 147 mmol/L NaCl, 4 mmol/L KCl, 3 mmol/L CaCl2, 1.2 mmol/L MgCl2, 15 mmol/L HEPES (pH 7.4), and 0.5% bovine serum albumin (BSA), 5 mmol/L glucose, PMSF, aprotinin, and leupeptin added fresh before homogenization. The tissue was homogenized, using 20 strokes with a 0.25-mm clearance Teflon-glass homogenizer. The homogenate was centrifuged at 1,000g at 4°C for 10 minutes. The pellet was resuspended in 5 volumes of MVB containing 17% dextran and spun at 3000g for 15 minutes. After the spin, the lipid fraction was removed, the sides of the tube were dried, the pellet resuspended in MVB containing 0.5% BSA and triturated with a pipette to remove clumps.
The material was then filtered through a 118-μm nylon mesh, the eluant collected, and filtered through a 40-μm mesh to separate out contaminating nuclei. The microvessels were washed off of the 40-μm mesh with BSA-free MVB, pelleted at 1,000g for 15 minutes, and resuspended in Tris-edetic acidsucrose buffer consisting of 20 mmol/L Tris, 1 mmol/L edetic acid, and 255 mmol/L sucrose (titrated to pH 7.4). The samples were examined microscopically to analyze the purity of the preparation. Samples used in these studies were those that achieved at least 95% purity. The remaining homogenate, minus the microvessels, was saved to determine iron content.
After isolation, the microvessels were sonicated for 30 pulses to increase access to membrane-bound proteins and treated for 1 hour at room temperature with solubilization buffer. The microvessel solubilization buffer consists of 50 mmol/L Tris (pH 7.4), 2% sodium dodecyl sulfate, and 0.1% mmol/L edetic acid. Total solubilized protein levels were then assayed using the NI protein assay (GenoTechnology, St. Louis, MO, U.S.A.).
To determine how the iron treatments affected the microvascular profile of proteins related to iron transport, an immunoblot assay was performed. Five micrograms of total protein from each microvasculature sample was loaded per well of the slot-blot apparatus (Bio-Rad, Fremont, CA, U.S.A.) under vacuum. The protein was captured onto an Optitran 0.2-μm nitrocellulose membrane (Schleicher & Schuell, Keene, NH, U.S.A.). The blot was rinsed in dH20 and blocked for 45 minutes with 5% dried milk in phosphate-buffered saline, then incubated in one of the primary antibodies of interest overnight at 4°C. The next day, the blot was incubated with the appropriate secondary antibody for 1 hour at room temperature. For primary antibodies raised in rabbit, we used goat anti-rabbit immunoglobulin G secondary antibodies conjugated to horseradish peroxidase (Sigma). For primary antibodies raised in mouse, we used rabbit anti-mouse immunoglobulin G secondary antibodies conjugated to horseradish peroxidase (Sigma). After incubation with the secondary antibody, the blot was rinsed and then incubated with LumiGlo reagent (KPL, Gaithersburg, MD, U.S.A.) for 1 minute. The blot was then blotted dry and exposed to radiographic film. Band density on the radiographic film was quantified densitometrically. The point of reference for comparison of the relative amounts of protein in each preparation was whole brain homogenate from a Sprague-Dawley rat that had not undergone any manipulation of its iron status. The same sample was used as the point of reference for all assessments in this study.
Antibodies
The primary antibody against DMT1 was raised in rabbits against rat peptide by QCB Laboratories (Hopkinton, MA, U.S.A.). A complete description of the antibody production and its specificity has been detailed previously (Burdo et al., 2001). This polyclonal antibody was raised against a fragment of the protein from the third extracellular domain, and is reactive against both positive and negative iron responsive element forms of the protein. David Haile provided the primary antibody against ferroportin (also known as IREG1 and MTP); a complete description of the antibody production and its specificity has been detailed previously (Abboud and Haile, 2001). The monoclonal primary antibody against transferrin receptor was obtained from Zymed Laboratories (South San Francisco, CA, U.S.A.). Dr. John Beard (Pennsylvania State University, University Park, PA, U.S.A.) provided the primary antibody against liver ferritin. The polyclonal primary antibody against transferrin was obtained from ICN Biomedicals (Aurora, OH, U.S.A.).
Iron measurements
The tissue remaining after the microvessels were removed (see previous section) was collected for quantification of iron. The total protein in each homogenate was determined using a Bradford assay, and iron content was determined using atomic absorption spectrophotometry. Because only the protein profiles differed only in the Belgrade rats with treatment, iron values for brain homogenates were only determined in these animals.
Statistical analyses
A one-way analysis of variance (ANOVA) and post-hoc Bonferroni analysis were applied to the microvascular iron transport protein and iron values using Analyse-It for Microsoft Excel, with statistical significance set at P < 0.05.
RESULTS
The amount of DMT1, transferrin, transferrin receptor, and ferritin was elevated in the microvasculature isolated from each brain region compared with the amount found in total brain homogenate (Figs. 1A–1D). In contrast, ferroportin was not detected in the microvasculature, but is present in the total brain homogenate (Burdo et al., 2001). In the Sprague-Dawley rats, the amount of DMT1 (Fig. 1A), ferritin (Fig. 1B), transferrin (Fig. 1C), or transferrin receptor (Fig. 1D) did not differ statistically in the microvasculature isolated from any of the brain regions and the values obtained were not affected by either an increase or decrease in plasma iron levels.
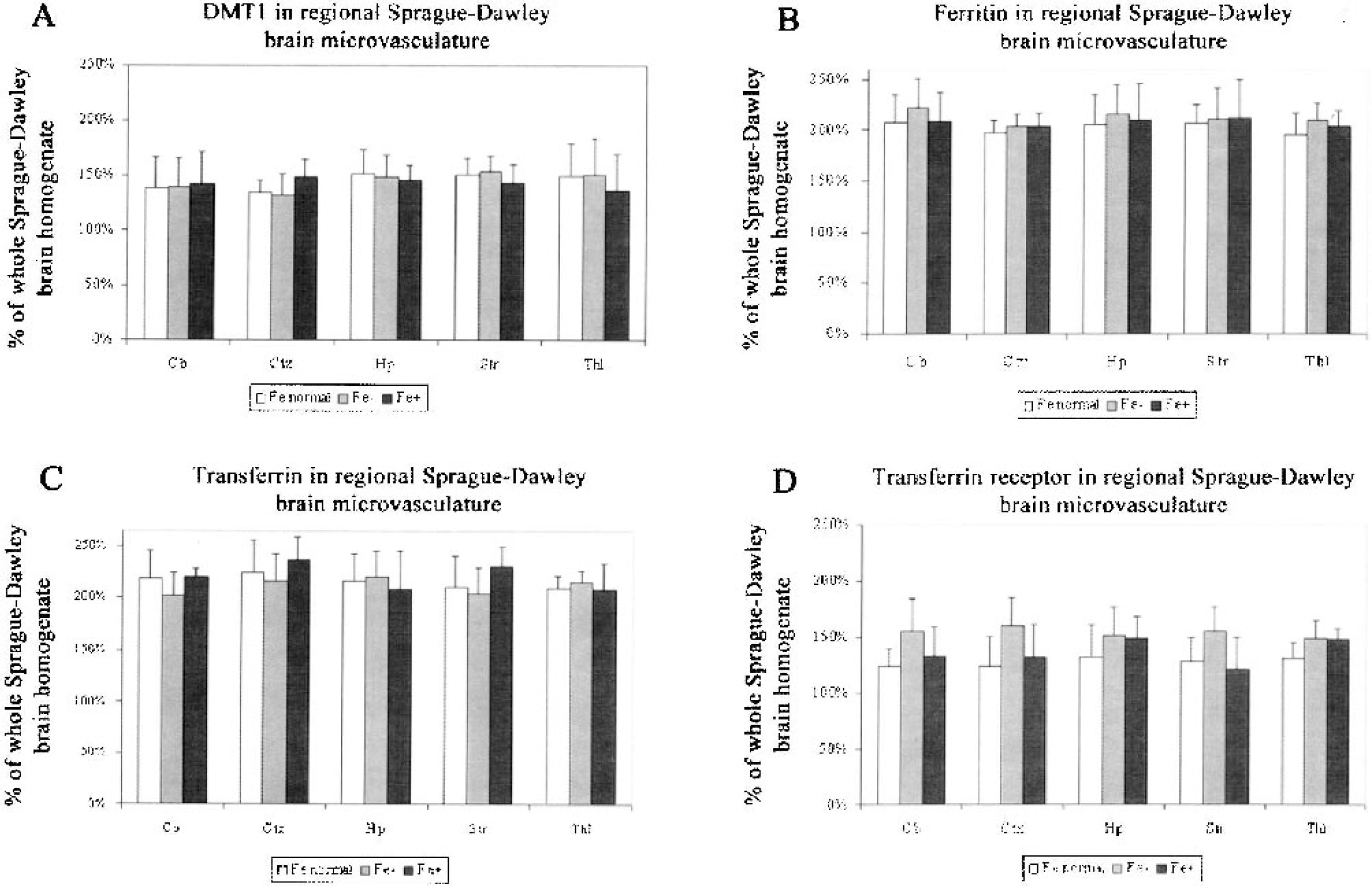
Levels of iron-metabolism proteins in Sprague-Dawley regional brain microvasculature. Divalent metal transporter 1 (DMT1) (
The levels of proteins in the microvasculature from the Belgrade rats were much more responsive to systemic iron manipulation than the acute model with normal brain iron status. DMT1 was significantly decreased in the microvasculature from all brain regions of rats that were both heterozygotic and homozygotic for the Belgrade mutation compared with the amount of DMT1 in the total brain homogenate used as a relative standard for both the Sprague-Dawley and Belgrade rats (Fig. 2A). This observation is not due to the inability of the antibody to recognize the mutant DMT1 protein (Burdo et al., 2001). There were no differences in the regional levels of DMT1 in the microvasculature within the b/b or +/b brain. Ferritin (Fig. 2B) and transferrin (Fig. 2C) levels in the b/b were elevated compared with that found in the standard total brain homogenate, but also did not differ among the various brain regions. Transferrin receptor (Fig. 2D) was increased in the microvasculature of the b/b rat compared with total brain homogenate. Similar to the Sprague-Dawley rats, ferroportin was not detected in the microvasculature of Belgrade rats (data not shown, but see Burdo et al., 2001). Under standard conditions, there was more DMT1 in the microvasculature from each brain region in the +/b rats compared with b/b rats (Fig. 2A). There was no difference between b/b and +/b in the relative amounts of transferrin (Fig. 2B) or ferritin (Fig. 2C) within the microvasculature from the different brain regions. Transferrin receptors were increased in the b/b rats in each brain region compared with +/b rats (Fig. 2D).
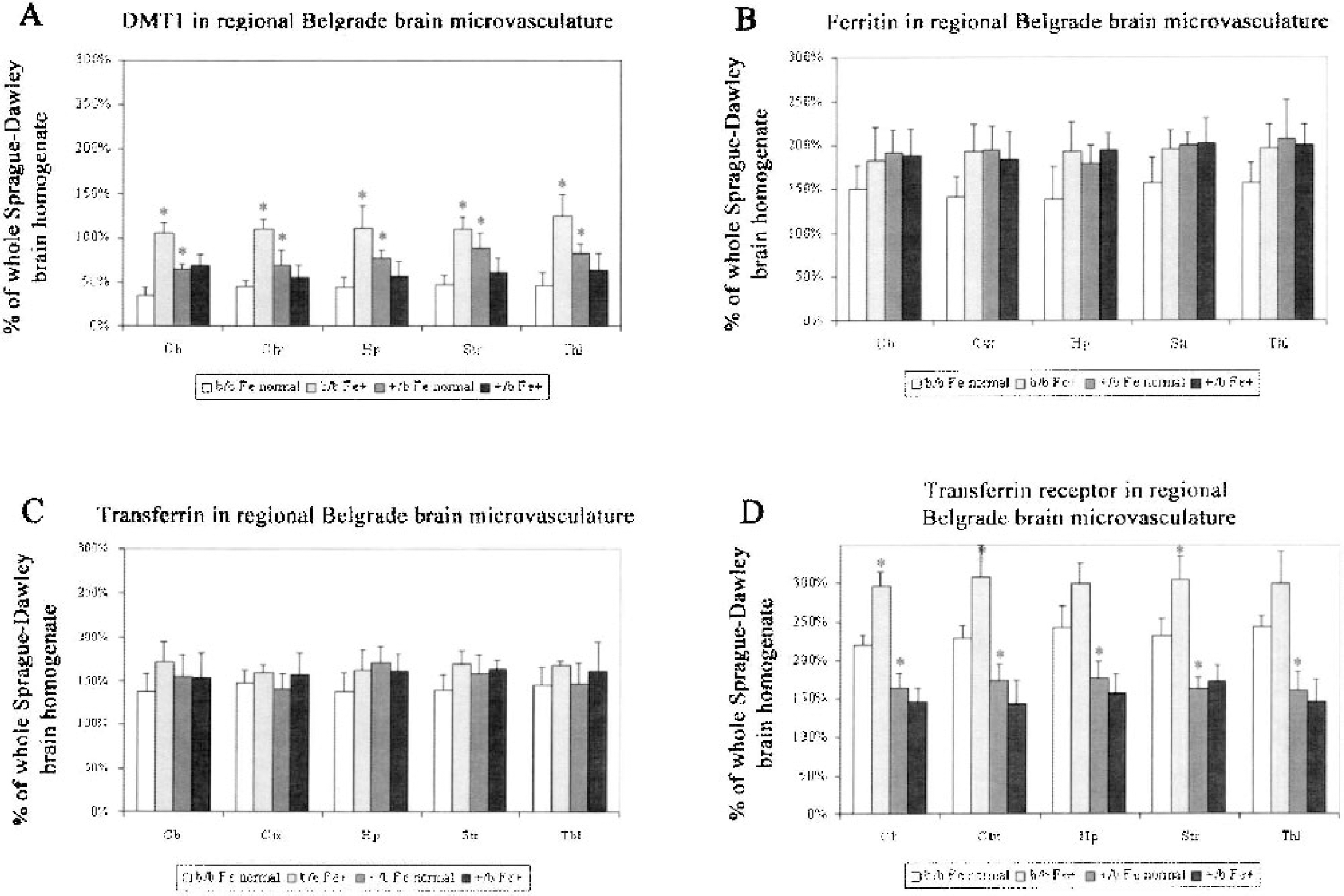
Levels of iron-metabolism proteins in Belgrade regional brain microvasculature. Divalent metal transporter 1 (DMT1) (
When the b/b rats were supplemented with iron, the levels of DMT1 detected in the vasculature increased by an average of 175% in all brain regions (Fig. 2A). Iron supplementation had no statistically significant effect on the levels of ferritin (Fig. 2B) or transferrin (Fig. 2C) in the b/b or +/b rats. Ferritin levels did increase in each brain region in the b/b rats by an average of 40%, but this increase was slightly outside the range of significance (P = 0.08). Iron supplementation was associated with a significant increase in transferrin-receptor expression in each brain region (Fig. 2D), except for the 45% increase in the hippocampal and thalamus microvasculature that did not reach statistical significance. Iron supplementation in the +/b rats had no effect on the levels of transferrin receptor in any of the brain regions.
The amount of iron was determined in the brain homogenates after the microvessels had been removed. The cerebellum had significantly more iron than the other brain regions examined in both the b/b and +/b rats (Fig. 3). However, the levels of iron differed between the two groups of rats in only the striatum and thalamus, where the levels of iron were less in the b/b rats in both areas. Iron supplementation had both region-specific and group-specific effects. A significant increase in iron content occurred in the cerebellum after iron supplementation in the +/b rats but not the b/b rats. This was the only brain region in which an increase in iron content was seen with supplementation in the +/b group. Iron supplementation increased brain iron levels in the cortex and thalamus of the b/b rats.
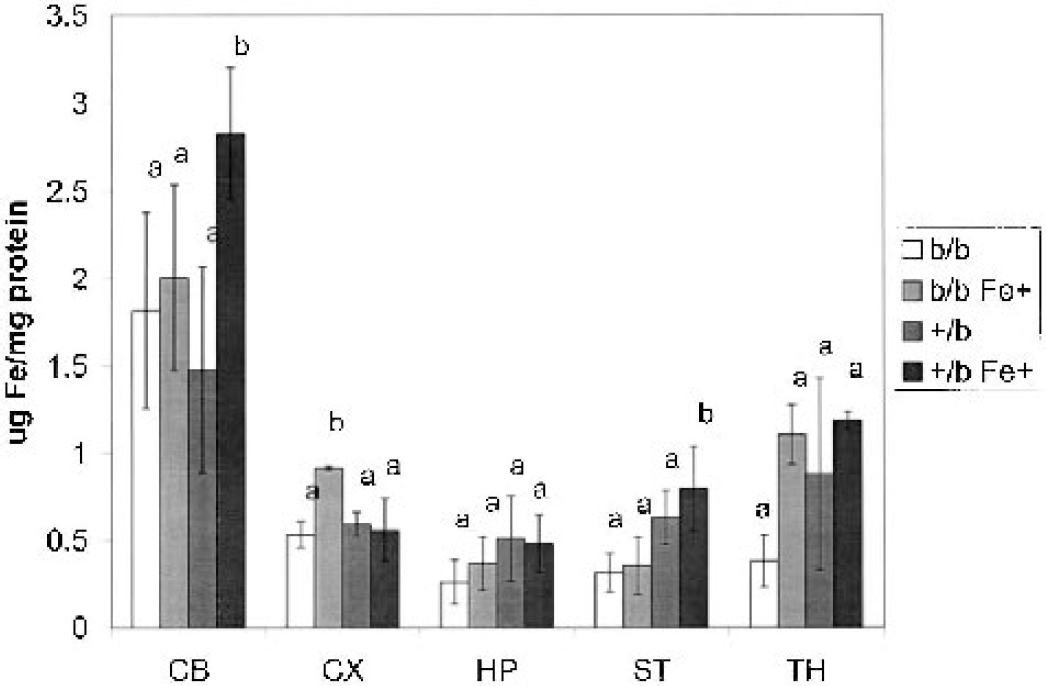
Regional brain iron concentrations in Belgrade rats. The concentration of iron was determined in each of the brain regions after removal of the microvasculature. The same brain regions were examined as indicated in the legend for Fig. 1. The data are reported as the mean and standard deviation for each group of animals. The letters indicate the iron concentration within each region that are similar (same letter) or that differ statistically (different letter). The data were analyzed with ANOVA and Scheffé post hoc pairwise analysis.
DISCUSSION
The results of this study reveal that the concentration of iron transport, uptake, and storage proteins in the endothelial cells of the brain microvasculature far exceed that found in total brain homogenates. This observation reflects the high iron requirement of the endothelial cells to meet the metabolic demands of transport. The relatively high level of ferritin in the microvasculature indicates that these cells have significant iron storage capacity, and the high levels of DMT1, a protein associated with release of iron from endosomes, indicates there is considerable removal of iron into the cytoplasm of endothelial cells. These two observations are compelling reasons to consider the endothelial cells as active reservoirs of iron for the brain rather than passive conduits. The data are also consistent with our observations that there is both a transferrin-mediated and non-transferrin-mediated iron efflux system from endothelial cells (Burdo et al., 2003). A salient observation in this study is the lack of detectable ferroportin (also called MTP) in the brain microvasculature, which is consistent with our previous report (Burdo et al, 2001) and indicates that the process of iron efflux from the endothelial cells is different from enterocytes (Abboud and Haile, 2000; McKie et al., 2000). The results of this study also reveal that there is considerable similarity in the protein profile for iron transport and storage proteins in microvasculature isolated from different regions of the rat brain that are known to have dissimilar iron levels. Furthermore, there is little change in the profile when systemic iron levels are increased or decreased in the Sprague-Dawley rat. The similarity in the protein profile from the different brain regions suggests that the mechanism of iron enrichment in each brain region may be based on delivery of iron from another source, such as the CSF.
The possibility that CSF can deliver iron to the brain has not been extensively studied. Iron uptake by the choroid plexus has been shown in a number of studies (Crowe and Morgan, 1992; Morris et al., 1992). Transferrin mRNA is enriched in the choroid plexus (Dickson et al., 1985) and this transferrin is presumably secreted to deliver iron, but transferrin secretion has only been demonstrated in an in vitro model (Tsutsumi et al., 1989). Some have argued that very little iron is extracted from the CSF, but this hypothesis is based on a single pass of a bolus injection (Moos and Morgan, 1998), which may have overwhelmed any transport system. Nonetheless, CSF-delivered iron is most likely responsible for global brain iron uptake and not maintenance of the heterogeneity of iron distribution in the brain. If CSF were primarily responsible for regulation at the regional level, it would be inconsistent with the high density of transferrin receptors and DMT1 in the microvasculature.
If there are no regional differences in iron metabolism protein levels in the microvasculature, the question arises as to how the regional differences in brain iron content are established and maintained. Regional variation in vascularization may be one possibility. Regional differences in rat brain capillary density have been reported, and this density is positively correlated with brain glucose utilization rate (Borowsky and Collins, 1989). Significant regional differences in blood volume and hematocrit have also been reported in the rat (Cremer and Seville, 1983).
Another possibility is that the microvascular protein profiles are established developmentally in concert with endothelial cell iron concentrations. Brain iron content changes regionally during development in the rat (Chen et al., 1995; Erikson et al., 1997). Nonheme iron content of rat brain microvasculature declines during early development (Moos, 1995), and iron regulatory protein 1 and 2 and DMT1 expression in the rat brain microvasculature increases concomitantly during development (Siddappa et al., 2002). The timing of these changes in the context of the development of the blood-brain barrier indicates dynamic regulation of iron at the site of the endothelial cells. It is possible that once adult brain iron status is reached, iron content of the endothelial cells lining the blood vessels is stabilized and subsequently so are the levels of proteins involved in iron management in the regional microvasculature. The protein profile is then expressed at a consistent concentration effective at maintaining the existing brain iron concentration.
The lack of change in the Sprague-Dawley iron protein profiles when systemic iron status is altered is consistent with a report that acute peripheral iron modification in adult rats does not affect brain iron status (Dallman and Spirito, 1977). However, it is possible that intracellular redistribution of DMT1 or transferrin receptor may dynamically regulate the amount of iron transported into or out of the microvascular endothelial cells without a change in profile. Less than 20% of the transferrin receptors located within cultured porcine microvascular endothelial cells participate in endocytosis of transferrin (van Gelder et al., 1995); thus, more than 80% of these receptors may be activated by the cell during an iron deficient state to increase iron transport into the brain. Redistribution of these proteins between active and inactive pools within the cell presumably would not change the immunochemical detection of these proteins. This change in intracellular distribution could occur as a result of loss of iron in the endothelial cell, but the loss of iron in the endothelial cells would presumably result from increased release of iron held in reserve to the brain (Burdo et al, 2003). Thus, we are still left with the idea that there is a signal from the brain that informs the endothelial cells about its iron status.
We were able to directly test the concept that low brain iron would result in a different response to systemic iron status using the Belgrade rats (Burdo et al., 1999; Zywicke et al., 2002). The regional quantitative analysis in this study revealed lower than normal concentrations of iron were specific to the striatum and thalamus of the b/b rats compared with control. However, compared with normal Sprague-Dawley rats and +/b rats, b/b rats have a higher expression of transferrin receptors in all regions of microvasculature studied. This transferrin receptor increase may be a compensatory response to the limited cellular iron uptake associated with the defect in DMT1 or a compensatory response necessary to achieve or attempt to achieve normal iron concentrations for each region. Why iron concentrations were able to achieve normal status in some regions (cerebellum and cortex) but not others (striatum and thalamus) in the b/b rats is not known. Future studies will directly relate protein profiles to regional iron uptake to address this void in understanding. That the increase in transferrin receptors in the nonsupplemented b/b microvasculature was not coupled to an increase in DMT1 is consistent with the concept that the increase in transferrin receptor is directly related to iron transport into the brain and consistent with data from our cell culture model that iron and transferrin transport across the endothelial cell are impacted differently by iron status (Burdo et al., 2003). The significant increase in DMT1 in microvasculature from all brain regions and transferrin receptors in the b/b rat cerebellum, cortex, and striatum after iron supplementation was different from that seen in both the supplemented and nonsupplemented +/b rats. Transferrin receptor levels in the b/b Fe+ hippocampus and thalamus increased relative to the unsupplemented group but did not reach statistical significance. Consistent with the increase in transferrin receptors was an increase in ferritin in the microvasculature of the b/b rat in each brain region. Although none of these increases in ferritin reached the 95% confidence level, all of them reached 92% confidence so a definite and consistent trend can be identified. Thus the ferritin, DMT1, and transferrin receptor data together support the idea that iron levels were increasing in the b/b endothelial cells after iron supplementation. The increase in iron concentration after supplementation in the b/b rats in a brain region is also not simply due to a lower starting point compared with control. The starting iron concentration in the striatum was lower than control but did not increase with supplementation, whereas the concentration in the thalamus started lower than control and increased with supplementation. The concentration of iron increased in the cortex with supplementation but the starting iron concentration was similar to normal. The differences in the response of the transferrin receptor and DMT1 in the brain microvasculature to iron supplementation in the three different groups examined in this study provides compelling evidence that there is a signal from the brain that is the controlling factor in the regulation of the protein profile of the microvasculature in response to changes in systemic iron status and to the amount of iron that is transported to the brain.
These findings in the Belgrade rat also have provided a potential insight into the human disease, restless legs syndrome (RLS). RLS is a sensory disorder characterized by akathisia, an uncontrollable, irresistible urge to move the limbs, which can be moderated or alleviated in a significant number of cases through iron supplementation. Even though many sufferers have normal plasma iron measurements, brain iron deficiency is prevalent among these patients (Allen et al., 2001 Earley et al., 2000) and systemic iron supplementation is an effective therapy (Hening et al., 1999). A possible explanation for the clinical findings in RLS is that iron supplementation in RLS patients has a similar effect on the microvasculature of RLS patients as that of the b/b rats; namely, an increase in microvascular levels of DMT1 and transferrin receptor because of an iron deficiency in the brain. This increase in iron transport protein levels could allow for greater transport of iron into the endothelial cells and brain, moderating the iron deficiency and alleviating symptoms of the disease.
In summary, we have found that iron transport proteins are enriched in the rat brain microvasculature, and that levels of these proteins do not respond to peripheral iron modification in the Sprague-Dawley rat but do respond when the brain is iron deficient. There is a lack of regional variability in iron transport and storage proteins of the microvasculature, despite the significant differences in iron concentration among brain regions. This observation suggests that a set point for regional iron concentration is attained during development and then a steady-state protein profile is established to provide a consistent supply of iron. Small fluctuations in iron availability that do not require changes in receptor concentration may be handled by recruitment of additional transferrin receptors to the plasma membrane. These findings have important implications for identifying windows of opportunity for therapeutic intervention for the treatment of iron deficiency, the study of brain iron metabolism, and neurologic diseases in which excess iron accumulates.
Footnotes
Acknowledgements
Sharon Menzies passed away on August 18, 2003 at the age of 38. The authors, her friends and family mourn her loss.
