Abstract
Neuronal death after ischemia-induced brain damage depends largely upon the activation of the N-methyl-D-aspartate (NMDA) excitatory glutamate receptor that is a target for many putative neuroprotective agents. Whereas the NMDA receptors mediate ischemic brain damage, blocking them is deleterious in humans. Here, the authors investigated whether nitrous oxide or xenon, which are gaseous anesthetics with a remarkably safe clinical profile that have been recently demonstrated as effective inhibitors of the NMDA receptor, may reduce the following: (1) ischemia-induced brain damage in vivo, when given after occlusion of the middle cerebral artery (MCAO), a condition needed to make these potentially neuroprotective agents therapeutically valuable; or (2) NMDA-induced Ca2+ influx in cortical cell cultures, a major critical event involved in excitotoxic neuronal death. The authors have shown that both nitrous oxide at 75 vol% and xenon at 50 vol% reduce ischemic neuronal death in the cortex by 70% and further decrease NMDA-induced Ca2+ influx by 30%. In addition, xenon at 50%, but not nitrous oxide at 75 vol%, further decreases ischemic brain damage in the striatum (a subcortical structure that is known to be resistant to neuroprotective interventions). However, at a higher concentration (75 vol%), xenon exhibits potentially neurotoxic effects. The mechanisms of the neuroprotective and potentially neurotoxic effects of nitrous oxide and xenon, as well as the possible therapeutic implications in humans, are discussed.
Neuronal death after a major brain insult, such as ischemic stroke or trauma, is thought to depend largely upon the activation of the N-methyl-D-aspartate (NMDA) excitatory glutamate receptor. Blocking the NMDA receptor may therefore contribute to decrease brain damage produced by ischemic stroke (Simon et al., 1984; Dingledine et al., 1999). However, despite the beneficial effect of this class of agents in animal models of stroke and other major brain insults, the clinical development of several NMDA receptor antagonists has had to be abandoned because these compounds possess an intrinsic neurotoxicity (Olney et al., 1989; 1991) and fail to reach the site of brain injury (Arrowsmith et al., 1998).
In contrast with volatile anesthetics that mainly act at the γ-aminobutyric acid type A (GABAA) receptor (Krasowski and Harrison, 1999), nitrous oxide and xenon, which are anesthetic gases with a remarkably safe clinical profile, have been shown to be effective inhibitors of the NMDA receptor (Jevtovic-Todorovic et al., 1998; Franks et al., 1998; Yamakura and Harris, 2000). Given that both nitrous oxide and xenon readily cross the blood-brain barrier and have low blood/gas solubility that is advantageous in terms of rapid inflow and washout, conditions that may favor treatment and reduce risk of neurotoxic side effects, it has been suggested that they may possess neuroprotective properties. Accordingly, possible neuroprotection by nitrous oxide and xenon was first addressed in recent studies that showed that these anesthetic gases prevent excitotoxic neuronal death in cortical cell cultures (Wilhelm et al., 2002) and further reduce neuronal degeneration induced by the intraperitoneal injection of N-methyl-DL-aspartate (Jevtovic-Todorovic et al., 1998; Wilhelm et al., 2002). Although xenon has been suggested as a possible neuroprotectant in acute stroke (Altschuler, 2001), yet this has never been demonstrated either clinically or experimentally in animal models of brain diseases.
Here, the authors studied whether nitrous oxide or xenon may reduce the following in rats: (1) ischemia-induced brain damage produced by middle cerebral artery occlusion (MCAO), when administered after the MCAO period (a condition prerequisite for the therapeutic viability of these potentially neuroprotective agents); or (2) NMDA-induced Ca2+ influx in neuronal cell cultures, which is thought to be a major critical event involved in excitotoxic neuronal death (Goldberg and Choi, 1993). Alternatively, to make interspecies and interagents comparisons of inhalational anesthetics, a reliable and useful index is the minimum alveolar anesthetic concentration (MAC) that prevents purposeful movement to noxious stimulation. As the MAC of xenon is not available yet in rats, the authors further determined it to allow comparison of nitrous oxide and xenon action.
MATERIALS AND METHODS
All experiments were performed within the framework of the French legislation that governs the use of animals for biomedical experimentation.
Ischemia-induced brain damage
Adult male Sprague-Dawley rats (n = 24) weighing 280 to 300 g (CERJ, Le Genest-St-Isle, France) were subjected to transient cerebral ischemia for 90 minutes by the intraluminal MCAO method (Longa et al., 1989). Briefly, rats were anesthetized (in the absence of nitrous oxide) in an anesthesia box with 4% halothane completed by oxygen. Then, the animals were intubated and maintained under general anesthesia with 1 to 1.5% halothane (completed by oxygen in the absence of nitrous oxide) throughout the surgical intervention. A femoral artery was cannulated for continuous arterial pressure monitoring and gas analysis. A midline neck incision was made, and the right common carotid artery was exposed. After coagulation of the branches of the external carotid artery, a nylon thread (0.18 mm diameter) with a distal cylinder (3 mm long and 0.38 mm diameter) was inserted in the lumen of the external carotid artery, directed into the internal carotid artery up to the origin of the middle cerebral artery, and removed 90 minutes later to restore blood flow. Arterial pressure, blood gases, and rectal temperature were maintained within their physiologic range throughout the procedure.
Gas treatment after MCAO
Fifteen minutes after the MCAO period (i.e., after blood flow was restored), awake freely moving rats were treated for 3 hours, in a closed chamber (10 L vol) fitted with viewing windows to allow behavioral observation, to either air (control rats, n = 7), nitrous oxide at 75 vol% (n = 6), or xenon at 50 vol% (n = 6) or 75 vol% (n = 5) of medical grade (with the remainder being oxygen). When connected to oxygen and nitrous oxide or xenon, gas mixtures were obtained using calibrated flowmeters. After a 3-minute flush at a flow rate of 5 L/min, the flow rate was reduced to 1 L/min for the remaining 3 hours. This situation maintained carbon dioxide levels less than 0.03% and humidity between 60 and 70%. Physiologic parameters were not monitored because the animals were treated in a closed chamber (in addition, neither nitrous oxide at 75 vol% nor xenon at 50 vol% or 75 vol% produced anesthesia, so the animals showed locomotor activity and movement [see Discussion]).
Histologic analysis
The rats were killed 24 hours after MCAO, and the brains were removed and frozen in isopentane for histologic analysis. Coronal brain sections (20 μm) were crystat-cut and subsequently stained with cresyl violet and analyzed with an image analyzer (Biocom, Paris, France). The lesioned areas were delineated by the pallor of histologic staining in necrotic tissue and then compared with that of healthy tissue using a blinded procedure. Regions of interest were determined through the use of a stereoataxic atlas for the rat brain (Paxinos and Watson, 1998).
Cell cultures
Mixed cortical cultures containing both neuronal and glial cells were prepared from fetal mice at 15 to 16 days gestation (Rose et al., 1993). Cerebral cortices were dissected in media stock (MS; Eagle's minimal essential medium augmented with 2 mM glutamine and 25 mM glucose). Then, dissociated cortical cells were transferred to MS supplemented with 5% fetal bovine and 5% horse serum for trituration and plated in glass petri dishes that had been previously coated with poly-D-lysine and laminin. Cultures were kept at 37°C in a humidified atmosphere containing 5% CO2. The medium was changed twice weekly. Experiments were performed on cortical cultures after 12 to 14 days in vitro.
Measurement of NMDA-induced Ca2+ influx in neuronal cell cultures
Intracellular free Ca2+ in cortical neurons was measured by using fura-2 fluorescence videomicroscopy. Cell cultures were loaded with fura-2 for 30 minutes at room temperature (22°C) in 5 μM fura-2/am plus 0.1% pluronic F-127 (Molecular Probes, Leiden, the Netherlands) and incubated for an additional 30-minute period in a HEPES-buffered saline solution. Experiments were made on the stage of an inverted microscope equipped with a 75 W mercury lamp and a 40×, 1.3 N.A. epifluorescence oil immersion objective. Fura-2 (ex: 340,380 nm, em: 510 nm) ratio images were acquired with a CCD camera and digitized (256 × 512 pixels) with Metafluor 4.11 software (Universal Imaging Corporation, Chester, PA, USA).
Changes in intracellular free Ca2+ in cortical neurons were induced by rapid NMDA stimulations of 1.25 to 100 μM for 30 seconds each. During the experiments, the cells were continuously perfused at a rate of 2 ml/min using a peristaltic pump with a HEPES-buffered saline solution, previously saturated with either nitrous oxide at 75 vol%, xenon at 50 vol%, or xenon at 75 vol%, as needed (for each experiment, n = 3, cell number = 135-150). All experiments were performed at room temperature (22°C) with the pH set at 7.4.
Measurement of the MAC of nitrous oxide and xenon
Adult male Sprague-Dawley rats weighing 280 to 300 g (CERJ, Le Genest-St-Isle, France) were placed in a Perspex cylinder that could be rotated; the animals were compressed with either nitrous oxide (n = 6) or xenon (n = 6) in a hyperbaric chamber fitted with three viewing windows. To avoid compression rate-dependent distortion of anesthetic potency (Abraini et al., 1998), nitrous oxide and xenon were introduced in the hyperbaric chamber at a compression rate of 0.04 atm/min. The total pressure of nitrous oxide or xenon minus the oxygen partial pressure (see below) at which the animals exhibited loss of righting reflex, a reliable index of anesthetic potency, corresponded to the MAC (Lever et al., 1971). The oxygen level was maintained at 0.23 ± 0.02 atm. The carbon dioxide level was kept less than 0.03% using soda lime, and the humidity was maintained between 60% and 70% using silicagel.
Data presentation and statistical analysis
Data were expressed as means ± SEM and analyzed using nonparametric statistics. Between-group comparisons of in vivo data were carried out using a Mann-Whitney U-test. The effects produced by nitrous oxide and xenon on the NMDA-induced Ca2+ influx in cortical neurons were fitted by the logistic equation using nonlinear least squares regressions; within-group comparisons of NMDA-induced Ca2+ influxes were analyzed using a Friedman (F) analysis of variance. The level of significance was set at P < 0.05.
RESULTS
Effect of nitrous oxide and xenon on ischemia-induced brain damage
Nitrous oxide at 75 vol% and xenon at 50 vol% reduced cortical infarction by about 70%, as compared with control rats treated with air (Fig. 1A) (U-test = 0, P = 0.0045). Xenon at 50 vol%, but not nitrous oxide at 75 vol%, further reduced striatal infarction by approximately 50%, as compared with control rats (Fig. 1B) (U-test = 0, P = 0.0045). In contrast, xenon at 75 vol% showed an ambigous effect, producing a trend toward reduction in cortical infarction and a trend toward increase in striatal infarction, both of which failed to reach statistical significance (Fig. 1A and Fig. 1B).
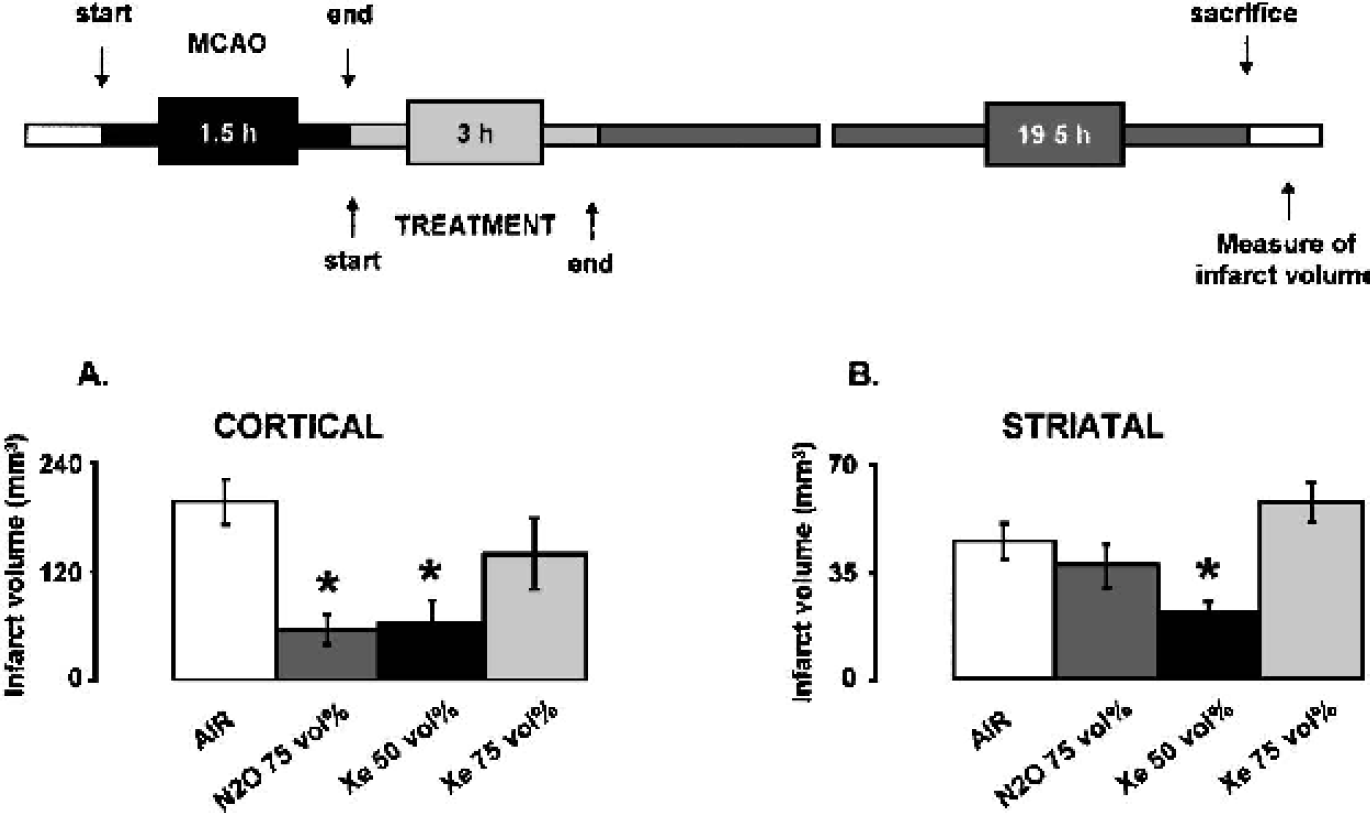
Effects of nitrous oxide and xenon on ischemic brain damage. After a transient MCAO of 90 minutes duration was completed, rats were exposed for 3 hours to air (control animals), nitrous oxide at 75 vol%, xenon at 50 vol%, or xenon at 75 vol% of medical grade (with the remainder being oxygen).
Effect of nitrous oxide and xenon on NMDA-induced Ca2+ influx in neuronal cell cultures
Both nitrous oxide at 75 vol% (Fig. 2A) or xenon at 50 vol% (Fig. 2B) reduced the NMDA-induced Ca2+ influx in neuronal cell cultures by approximately 30% (F = 21, P = 0.0001). In contrast, as described previously in the present authors' in vivo experiments, xenon at 75 vol% (Fig. 2C) also had a bivalent effect, reducing the Ca2+ influxes induced by low doses of NMDA (1.25-12.5 μM; F = 12, P = 0.0001) and potentiating those induced by high doses of NMDA (50-100 μM; F = 6, P = 0.014). Representative NMDA-evoked Ca2+ influxes in cortical neurons treated with air, nitrous oxide at 75 vol%, xenon at 50 vol%, or xenon at 75 vol% are illustrated in Fig. 2D–Fig. 2F.
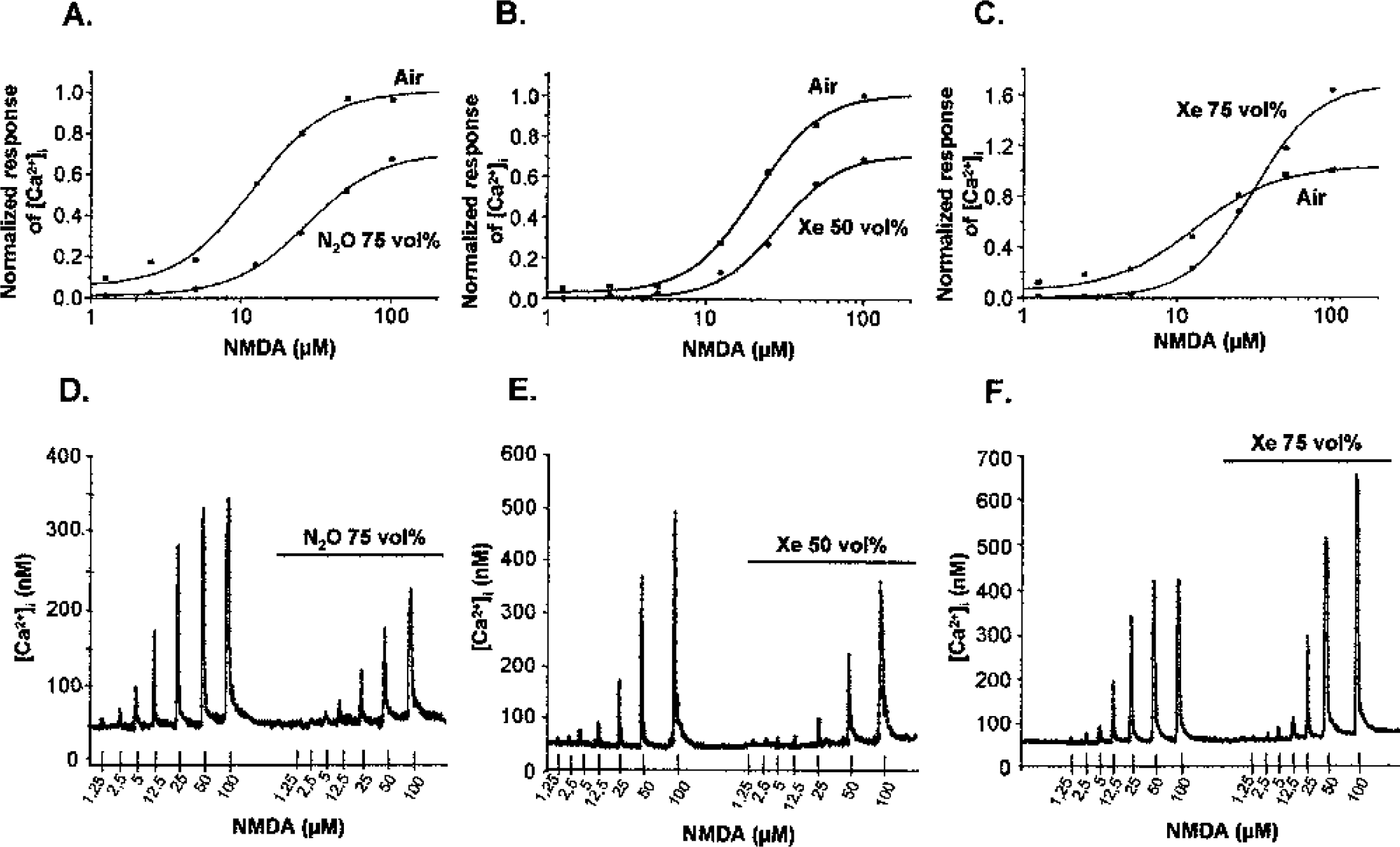
Effects of nitrous oxide and xenon on the NMDA-evoked Ca2+ influx in cortical neurons. The cells were continuously perfused with a buffered saline solution, previously saturated with either nitrous oxide at 75 vol%, xenon at 50 vol%, or xenon at 75 vol% as required.
Determination of the MAC of nitrous oxide and xenon
To obtain loss of righting reflex in rats, the authors found that nitrous oxide and xenon were effective at respectively 1.28 ± 0.029 atm (i.e., 128 ± 2.9 vol%) and 0.86 ± 0.023 atm (i.e., 86 ± 2.3 vol%), which would indicate that the potency of xenon is 1.48-fold higher than that of nitrous oxide (data not shown; U-test = 0, P = 0.0039). Given that the MAC of nitrous oxide and xenon is, respectively, 105 vol% and 71 vol% in humans (Cullen et al., 1969; Hornbein et al., 1982), these data indicate that the MAC ratio of nitrous oxide and xenon is similar in both rats and humans.
DISCUSSION
Neuroprotection by nitrous oxide at 75 vol% and xenon at 50 vol%
Both nitrous oxide at 75 vol% and xenon at 50 vol% (i.e., at equipotent narcotic concentrations corresponding to approximately 0.6 MAC) reduce cortical brain damage in vivo and decrease NMDA-evoked Ca2+ influxes in neuronal cell cultures, which is a process that is recognized as a major critical event involved in excitotoxic neuronal death (Goldberg and Choi, 1993). Xenon at 50 vol%, but not nitrous oxide at 75 vol%, further reduces striatal brain damage in vivo.
The neuroprotective action in vivo of nitrous oxide at 75 vol% and of xenon at 50 vol% administered after MCAO may account for their pharmacologically antagonistic properties at the NMDA receptor (Jevtovic-Todorovic et al., 1998; Franks et al., 1998; Yamakura and Harris, 2000) because both gases have no effect on the α-amino-3-hydroxy-5-methyl-4-isoxazoleproprionic acid (AMPA) receptor (Yamakura and Harris, 2000). Concordant with these data are recent studies that have demonstrated that nitrous oxide and xenon prevent excitotoxic neuronal death produced by either glutamate or NMDA stimulation in cortical cell cultures (Wilhelm et al., 2002) and further reduce neuronal degeneration produced by the intraperitoneal injection of N-methyl-DL-aspartate (Jevtovic-Todorovic et al., 1998; Wilhelm et al., 2002).
In addition, both nitrous oxide and xenon readily cross the blood-brain barrier and have low blood/gas solubility that offers advantages in terms of rapid inflow and washout. Therefore, it is likely that the ability of both agents to reduce neuronal death when administered after MCAO accounts for their rapid access to the ischemic core. In this way, the efficacy of xenon at 50 vol%, but not of nitrous oxide at 75 vol%, at reducing neuronal death in the striatum (a subcortical structure that is notoriously difficult to protect against stroke because of its lack of collateral vasculature when compared with the cortex) may account at least partly for the lower blood/gas partition coefficient of xenon compared with nitrous oxide (0.12 vs. 0.46; see (Goto et al., 1998).
As well as their pharmacologic action, both nitrous oxide and xenon can produce physiologic effects on thermoregulation and cerebral circulation and metabolism, which may contribute, at least partly, to their neuroprotective action. General anesthetics are known to produce hypothermia in a dose-dependent manner, a condition that is neuroprotective by itself in various stroke models. However, in the present study, the neuroprotective effects of nitrous oxide and xenon were found at subanesthetic concentrations corresponding to 0.6 MAC, which did not produce suppression of locomotor activity, as assessed by the observation of the behavior of the freely moving animals. Based upon these facts, it is probable that thermoregulation was preserved in these experimental conditions, so that it is highly unlikely that the neuroprotective effects of nitrous oxide at 75 vol% and of xenon at 50 vol% result from an inhibitory action on thermoregulation. Support for this is the lack of neuroprotective action of xenon at the higher and more narcotic (but not sedative) concentration of 75 vol% (0.87 MAC) in the present study. With regards to the effects of nitrous oxide and xenon on cerebral blood flow and metabolism, the information obtained from computed tomography studies with xenon-133 as a tracer suggests that xenon may offer advantages over nitrous oxide in terms of circulatory and metabolic stability. Nitrous oxide increases cerebral blood flow and metabolism and disrupts cerebral vasoreactivity (Field et al., 1993; Reinstrup et al., 1994) that would contraindicate its use for the treatment of clinical situations associated with pathologic increases in intracranial pressure, such as stroke. In contrast, xenon at concentrations up to 70 vol% produces no significant changes in cerebral blood flow and metabolism (Yao et al., 1992; Whitehurst et al., 1994; Frietsch et al., 2001) and, further, maintains vasoreactivity (Schmidt et al., 2002). Therefore, it is possible that the advantages of xenon over nitrous oxide in terms of metabolic and circulatory stability may have also contributed, in addition to the lower blood/gas partition coefficient of the former, to the neuroprotective action of xenon in the striatum when used at 50 vol%.
In vivo and in vitro dual effects of xenon at 75 vol%
Xenon at a more narcotic concentration of 75 vol% shows no neuroprotective effect and exhibits a dual effect, producing both a trend toward increase in striatal infarction and a trend toward reduction in cortical infarction that did not reach statistical significance; in a similar way, xenon at 75 vol% further reduces Ca2+ influxes in neuronal cell cultures induced by low doses of NMDA and potentiates those induced by high doses of NMDA.
Taken together with the effects of xenon at 50 vol%, these data suggest that, depending upon its concentration and the level of the glutamatergic input at the NMDA receptor, xenon may exhibit both neuroprotective and neurotoxic properties, as is known for other NMDA receptor antagonists (Olney et al., 1989; 1991). According to the MAC ratio of xenon and nitrous oxide, it should be noted that xenon at 75 vol% can be considered equipotent to nitrous oxide at 111 vol%, a concentration not far from that of 117 vol% at which nitrous oxide exhibits neurotoxic effects related to its NMDA receptor antagonistic activity (Jevtovic-Todorovic et al., 1998). This potentially neurotoxic effect of xenon at 75 vol%in vivo appears in contradiction with recent data that have suggested that xenon, uniquely among anesthetics with known NMDA antagonistic actions, may have neuroprotective properties without a coexisting neurotoxicity (Nagata et al., 2001; Ma et al., 2002; Wilhelm et al., 2002). However, whereas these studies only evaluated the neuroprotective action of xenon at 75 vol% on neuronal changes induced by systemic injection of N-methyl-DL-aspartate or ketamine (a noncompetitive antagonist of the NMDA receptor) in the arcuate nucleus, the present authors used an integrative and more complex model of severe ischemic brain damage. Therefore, the discrepancies between these studies and the present data might be because of the different models of brain injury used and, as suggested above, to their relative severity in terms of excitotoxicity. In addition and in contrast with its lack of effects on circulatory functions and metabolism at concentrations below 70 vol%, xenon at 79 vol% (a concentration equivalent to 117 vol% nitrous oxide) increases cerebral blood flow that contraindicates its clinical use at high concentrations for the treatment of cerebrovascular diseases (Schmidt et al., 2001). Such an adverse increase in cerebral blood flow may be the reason that explains the authors' in vivo data, which demonstrate that xenon at 75 vol% tends to increase cerebral damage in the striatum, a structure that does not benefit from a collateral vasculature.
Possible therapeutic implications
To make interspecies comparisons of inhalation anesthetics, a reliable and useful index is the MAC. The MAC of xenon is only 71 vol% in humans (Cullen et al., 1969). Thus, if xenon at 86 vol% in rats is equivalent to 71 vol% xenon in humans, then 75 vol% xenon in rats will be equivalent to 62 vol% xenon in humans. Therefore, although xenon when used as a general anesthetic under ‘physiologic’ conditions has virtually no side effects (Luttropp et al., 1994; Lynch et al., 2000), its latent ambivalent and potentially neurotoxic character when used at 75 vol% in the rat suggests that it may not be prudent to use it in patients at concentrations higher than 60 vol% for treating cerebral stroke or other brain pathologies that involve excitotoxicity. In contrast, xenon at 50 vol% decreases neuronal death in both the striatum and the cortex when administered as post-treatment after MCAO in the rat, and it reduces NMDA-evoked Ca2+ influxes in neuronal cell cultures; accordingly, xenon at an appropriate concentration (approximately 40 vol%) could be considered a practical therapy for the treatment of stroke in humans.
The major obstacle to the widespread clinical use of xenon is the excessive cost of production. Combining nitrous oxide and xenon, which have remarkably safe clinical profiles, may be a rational formula to obtain optimal subcortical neuroprotection while minimizing the risk of adverse side effects and reducing the cost of treatment. Finally, it is probable that a similar approach could be used to treat other brain diseases, in which excitotoxicity is pivotal.
