Abstract
Inflammation can contribute to brain injury, such as that resulting from ischemia or trauma. The authors have previously shown that the cytokine interferon-beta (IFN-β) affords protection against ischemic brain injury, which was associated with a diminished infiltration of neutrophils and a reduction in blood–brain barrier (BBB) disruption. The goal of the current study was to directly assess the effects of IFN-β on neutrophil infiltration, with the use of an in vivo assay of neutrophil infiltration with relevance to ischemic brain injury. Intrastriatal injection of recombinant rat cytokine–induced neutrophil chemoattractant-1, a member of the interleukin-8 family (1 μg in 1 μL), triggered massive infiltration of neutrophils and extensive BBB disruption 6 hours later, as measured using immunofluorescence microscopy and magnetic resonance imaging in the rat, respectively. Depleting the animals of neutrophils before interleukin-8 injection prevented BBB disruption. Treatment with IFN-β (5 × 106 U/kg) almost completely prevented neutrophil infiltration and attenuated BBB damage. Gelatinase zymography showed matrix metalloproteinase-9 expression in the ipsilateral striatum after interleukin-8 injection. Both neutrophil depletion and IFN-β treatment downregulated matrix metalloproteinase-9. IFN-β has already been approved for human use as a treatment for the chronic inflammatory disorder multiple sclerosis. The potential value of IFN-β as a treatment that can attenuate acute brain inflammation is considered.
Keywords
The inflammatory response to acute insults such as cerebral ischemia or trauma can contribute to brain injury (Barone and Feuerstein 1999; DeGraba 1998; Iadecola and Alexander 2001). Hallmarks of the acute phase of the inflammatory response are the opening of the blood–brain barrier (BBB) and the infiltration of neutrophils. Cytokines orchestrate the response and, as such, constitute powerful intermediates through which the response may be altered to achieve therapeutic benefit.
We have previously shown that the cytokine interferon-beta (IFN-β) significantly reduced infarct volume after transient ischemic stroke in the rat (Veldhuis et al., 2002). The reduction in lesion volume was associated with a diminished infiltration of neutrophils and an attenuated disruption of the BBB. Because ischemic injury induces a plethora of cytokines and a complex inflammatory response, it is difficult to determine whether IFN-β directly prevents neutrophil influx or whether this effect is secondary to some other mechanism. In multiple sclerosis, the effect of IFN-β on monocyte and T-cell infiltration has been attributed to downregulation of adhesion molecules (Calabresi et al., 2001; Defazio et al., 2000; Floris et al., 2002; Lou et al., 1999) and matrix metalloproteinase (MMP)-9 expression (Galboiz et al. 2001; Stuve et al. 1997; Trojano et al. 1999; Waubant et al., 2001). The goal of the current study was to directly assess the effects of IFN-β on neutrophil infiltration, with the use of an in vivo assay of neutrophil infiltration with relevance to ischemic brain injury while avoiding the complexity of ischemia-induced inflammation. To this end, the α-chemokine cytokine-induced neutrophil chemoattractant-1, a member of the interleukin-8 (IL-8) family, was injected directly into the rat striatum. IL-8 is considered to be the prototypic and most potent neutrophil-attracting chemokine, attracting neutrophils to a wide variety of organ systems (Baggiolini et al., 1995; Mukaida et al., 1998a). After cerebral ischemia, IL-8 upregulation by glia and endothelium (Ehrlich et al., 1998; Stanimirovic and Satoh, 2000; Stanimirovic et al., 2001) precedes neutrophil influx (Yamagami et al., 1999; Yamasaki et al. 1995), and treatment with antibodies against IL-8 prevents influx and reduces ischemic damage (Matsumoto et al. 1997a,b; Mukaida et al., 1998b). In addition, the neutrophil chemotactic activity induced by production or injection of the key proinflammatory cytokines tumor necrosis factor-α (TNF-α) or interleukin-1β (IL-1β) has been attributed to IL-8 (Anthony et al., 1998; Tada et al., 1993). So far, a limited number of studies have reported on the injection of proinflammatory cytokines in a setting of acute neurodegeneration. These studies have mainly focused on IL-1β or TNF-α (Bernardes-Silva et al., 2001; Blamire et al., 2000; Liu et al., 1994; Rosenberg et al., 1995; Watts et al., 1989; Yamasaki et al., 1992). The effects of injection of α-chemokines such as IL-8 have been studied only in the mouse (Andersson et al., 1992; Bell et al., 1996).
In this study, we applied magnetic resonance imaging (MRI) because of its potential to detect acute tissue changes such as those occurring after ischemia, structural tissue abnormalities, and functional properties such as hemodynamics and BBB integrity. Immunofluorescence microscopy and gelatinase zymography were performed to confirm neutrophil infiltration and to obtain mechanistic information on the effects of IL-8 injection and IFN-β treatment. To assess the contribution of neutrophils to the consequences of IL-8 injection, these assays were also carried out in a group of animals that was depleted of neutrophils before the experiment.
MATERIALS AND METHODS
Animal model
Male Fischer rats (F344/Ico, Iffa-Credo Broekman, Someren, The Netherlands) aged 3 weeks were used. Animals had free access to standard laboratory chow and water. Anesthesia was induced by injection of a mixture of 0.5-mL/kg fentanyl citrate (0.315 mg/mL, intraperitoneally) and fluanisone (10.0 mg/mL, intraperitoneally), and 0.5-mL/kg midazolam (5.0 mg/mL, intraperitoneally). Body temperature was maintained at 37°C by means of a feedback-controlled heating pad and an infrared heating lamp. A small burr hole was drilled in the cranium over the left hemisphere, 2.5 mm lateral and 0.5 mm anterior of bregma. A 1-μL syringe was lowered into the right striatum to a depth of 4.0 mm. Recombinant rat IL-8 (cytokineinduced neutrophil chemoattractant-1; R&D Systems, Minneapolis, MN, U.S.A.; 1.0 μL, 1-μg/μL in PBS with 50-μg/μL BSA; n = 13) or vehicle (1.0 μL PBS with 50-μg/μL BSA; n = 2) was injected at a rate of 0.125 μL/min using a microdrive. After injection, the needle was left in situ for 2 minutes to avoid leakage of injection fluid from the needle tract. Between the end of the injection procedure and the start of the MRI measurements, the animals were kept in a temperature-controlled environment (31°C). Before the MRI measurements, the animals were reanesthetized using 5% halothane in N2O/O2 (70/30, volume/volume). In the scanner, anesthesia was continued using 1% to 1.5% halothane, and body temperature was maintained using a feedback-controlled water-filled heating pad. The Utrecht University Animal Experiment Ethical Committee approved all animal experiment protocols.
Recombinant rat IFN-β
The cloning and expression of the chromosomal gene encoding rat IFN-β in Chinese hamster ovary cells have been described elsewhere (Ruuls et al., 1996). The glycoprotein was purified to homogeneity by dye-ligand affinity chromatography and analyzed for endotoxin contamination. The biologic activity of each batch was determined in an antiviral bioassay (Olsson et al., 1994) and compared with the bioactivity of a laboratory standard preparation. Purified recombinant rat IFN-β was found to exert a bioactivity of approximately 5 × 107 U/mg of protein. IFN-β–treated animals received dosages on the basis of equal bioactivity per kilogram of body weight.
Magnetic resonance imaging
Magnetic resonance imaging experiments were performed using a 4.7-T horizontal bore spectrometer (Varian Instruments, Palo Alto, CA, U.S.A.) with a 220-mT/m gradient insert. Radio frequency-excitation and signal detection were accomplished by means of a Helmholtz volume coil (9-cm diameter) and an inductively coupled surface coil (2-cm diameter), respectively. A rapid spin-echo scout sequence was used for determination of the position of the animal in the magnet and subsequent slice planning (repetition time [TR] = 15 milliseconds, echo time [TE] = 600 milliseconds, matrix [M] = 64 × 64, field of view [FOV] = 40 × 35 mm2, number of transients [NT] = 1, acquiring 21 consecutive 1.0-mm-thick sagittal slices).
Experimental protocol
Six hours after injection of IL-8, the animals were transferred to the MRI scanner for perfusion imaging and assessment of BBB disruption. IL-8 injected animals were divided into three groups: (1) animals that received IL-8 alone (n = 4), (2) animals that received recombinant rat IFN-β before IL-8 injection (n = 4), and (3) animals that received neutrophil-depletion treatment before IL-8 injection (n = 3). To look for structural effects of IL-8 injection on brain parenchyma, two additional animals received IL-8 injection with no prior treatment, and at 24 hours, contrast-enhanced T1-weighted MRI and diffusion-weighted and T2-weighted datasets were acquired. Neutrophil depletion was induced by injection of rabbit antirat, antineutrophil serum (Accurate Chemical & Scientific, Westbury, NY, U.S.A.; 3 mL/kg, intraperitoneally) at 24 hours and again at 5 hours before IL-8 (Blamire et al., 2000). Animals treated with recombinant rat IFN-β (5 × 106 U/kg, intraperitoneally) received injections at 48, 24, and 0.5 hours before IL-8 injection. This treatment regimen was chosen based on maximal prevention of leukocyte infiltration reported in a rat model of multiple sclerosis (van der Meide et al., 1998). After the MRI experiments, the animals were killed and the brains were directly removed without perfusion or fixation and processed for immunohistology and gelatinase zymography.
Immunofluorescence microscopy
After the animals were killed, the brains were dissected, snap-frozen in the vapor phase of liquid nitrogen, and stored at −80°C. Cryostat sections (8 μm) were cut, and we analyzed the sections close to the center of the lesion (bregma + 0.5 mm), just avoiding the needle tract. Sections were melted onto gelatin-coated glass slides and dried in containers with silica gel. Slides were fixed with acetone (for 10 minutes) and incubated in PBS with 10% fetal calf serum to prevent aspecific binding. Thereafter, immunohistochemistry was performed. Infiltrated leukocytes were detected with the monoclonal antibody OX1, directed against CD45 (Biotinylated mIgG1; obtained from Serotec, Oxfordshire, UK). Neutrophil infiltration was determined by monoclonal antibody HIS48, which recognizes an antigen on all neutrophils (van Goor et al., 1991) (Biotinylated mIgM; BD Biosciences). Activated microglial cells were detected with the monoclonal antibody OX42, which recognizes the iC3b receptor (biotinylated mIgG2a) obtained from Serotec (Oxfordshire, UK). Intercellular adhesion molecule-1 (ICAM-1) expression was determined by the monoclonal antibody 1A29 (mIgG1; a gift from Dr. T. Tamatani, Tokyo Metropolitan Institute of Medical Science, Japan). Cerebral blood vessels were detected by an antibody directed against von Willebrand factor (rabbit antihuman, Dako, Denmark). Slides were incubated with the antibodies at appropriate dilutions (2.5 μg/mL) for 1 hour at room temperature. To detect primary antibodies, the following conjugates were used: streptavidin Alexa-488, goat anti–mouse Alexa-488, and donkey anti–rabbit Alexa-594 (all obtained from Molecular Probes, Eugene, OR, U.S.A.). Omission of the primary antibodies served as negative control. The sections were examined with a microscope (Nikon Eclipse E800), and recordings were made with a digital NIKON DXM1200 camera.
Gelatinase zymography
Data analysis
Blood–brain barrier disruption was determined from the increase in signal intensity (using ≥2 × SD compared with contralateral as the threshold) induced by the accumulation of Gadolinium-DTPA in the brain parenchyma (Blamire et al., 2000; Stone et al., 1995), using the Interactive Data Language software package (Research Systems, Boulder, CO, U.S.A.). Apparent diffusion coefficient and T2 maps were generated from the diffusion-weighted and T2-weighted magnetic resonance images by monoexponential fitting using the same software package. Gd-leakage images were constructed by subtracting the precontrast T1-weighted dataset from the postcontrast dataset (Neumann-Haefelin et al., 2002). Voxels in the ipsilateral hemisphere were considered pathologic if their signal intensity had increased more than a factor 1.6 (corresponding to a ≥2 × SD difference) from the mean signal intensity of the brain parenchyma in the contralateral hemisphere. The volume of affected brain tissue was calculated by multiplying the number of pathologic voxels in a slice by the voxel volume. The total lesion volume was obtained by summation of the lesion volumes for all slices. DSC-enhanced MRI data were processed as described previously (Dijkhuizen et al., 1997). Briefly, the signal intensity drop induced by passage of the contrast agent was converted to an increase in the transverse relaxation rate, 1/T2* (i.e., ΔR2*). Hemodynamic-parameter maps were calculated by pixel-by-pixel fitting of time plots of ΔR2* to a γ-variate function (Rosen et al. 1990). Relative cerebral blood volume (rCBV), mean transit time, and relative cerebral blood flow indices (rCBFi) were measured in the injected striatum and in the striatum on the contralateral side. Statistical analysis was carried out using SPSS 10.0 (SPSS Inc., Chicago, IL, U.S.A.). Reported P values correspond to two-tailed significance.
RESULTS
There were no differences between the treatment groups (control, IFN-β, and neutrophil depleted) in terms of increase in body weight between initial pharmacologic treatment and start of IL-8 injection, or in body weight at the start of IL-8 injection, as tested by one-way analysis of variance.
Effect of IL-8 injection on BBB integrity
The intravascular contrast agent Gd-DTPA cannot pass the intact BBB. To study the effect of IL-8 injection on BBB integrity, the leakage of Gd-DTPA into the brain parenchyma was determined from postcontrast T1-weighted images. Injection of IL-8 resulted in extensive BBB breakdown as demonstrated by the leakage of contrast-agent into the brain parenchyma (Fig. 1). In vehicle-injected animals, the mechanical trauma caused by needle insertion resulted in only limited leakage, directly around the needle tract. The T2-weighted and diffusion-weighted images acquired after 24 hours were normal, indicating that IL-8 injection caused no morphologic changes or edema formation (data not shown). The IL-8–induced opening of the BBB detected at 6 hours had normalized by 24 hours. By that time, only a small amount of Gd-DTPA leakage was detected, which was restricted to the region around the needle tract (data not shown).
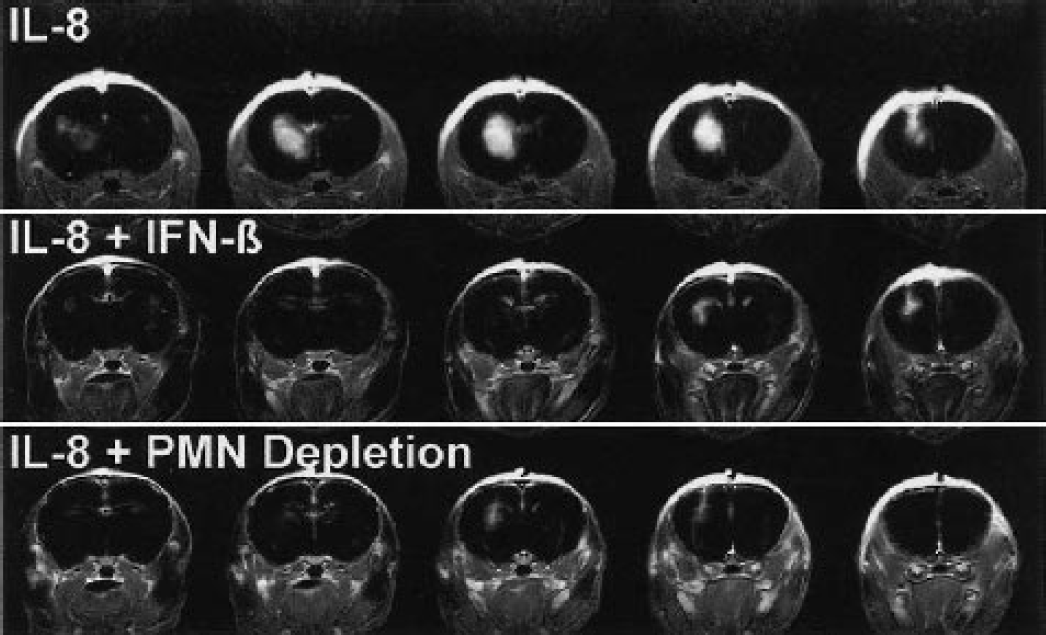
Blood–brain barrier integrity assessed 6 hours after intrastriatal injection of IL-8. Shown are postcontrast T1-weighted images of three animals, after subtraction of the precontrast datasets. Of each animal, five consecutive slices are shown in the rostrocaudal direction. The intravascular contrast agent Gd-DTPA cannot pass the intact BBB. At sites of BBB breakdown, the intraparenchymal presence of contrast agent is evidenced by increased signal intensity on T1-weighted images. IL-8, interleukin-8; IFN- β, interferon-beta; PMN, polymorphonuclear neutrophil.
Effect of neutrophil depletion and IFN-β treatment on IL-8–induced BBB disruption
Depletion of neutrophils before the experiment prevented the IL-8–induced breakdown of the BBB (Fig. 1). In those animals, only minimal leakage, directly around the needle tract, was seen. Figure 2 shows the total volume of brain tissue showing a ≥2 × SD increase in signal intensity, including the region around the needle tract, of all animal groups. Treatment with IFN-β potently protected the brain from the IL-8–induced BBB breakdown, and brought down the degree of Gd-DTPA–induced signal enhancement, almost to the level achieved by neutrophil depletion (Figs. 1 and 2).
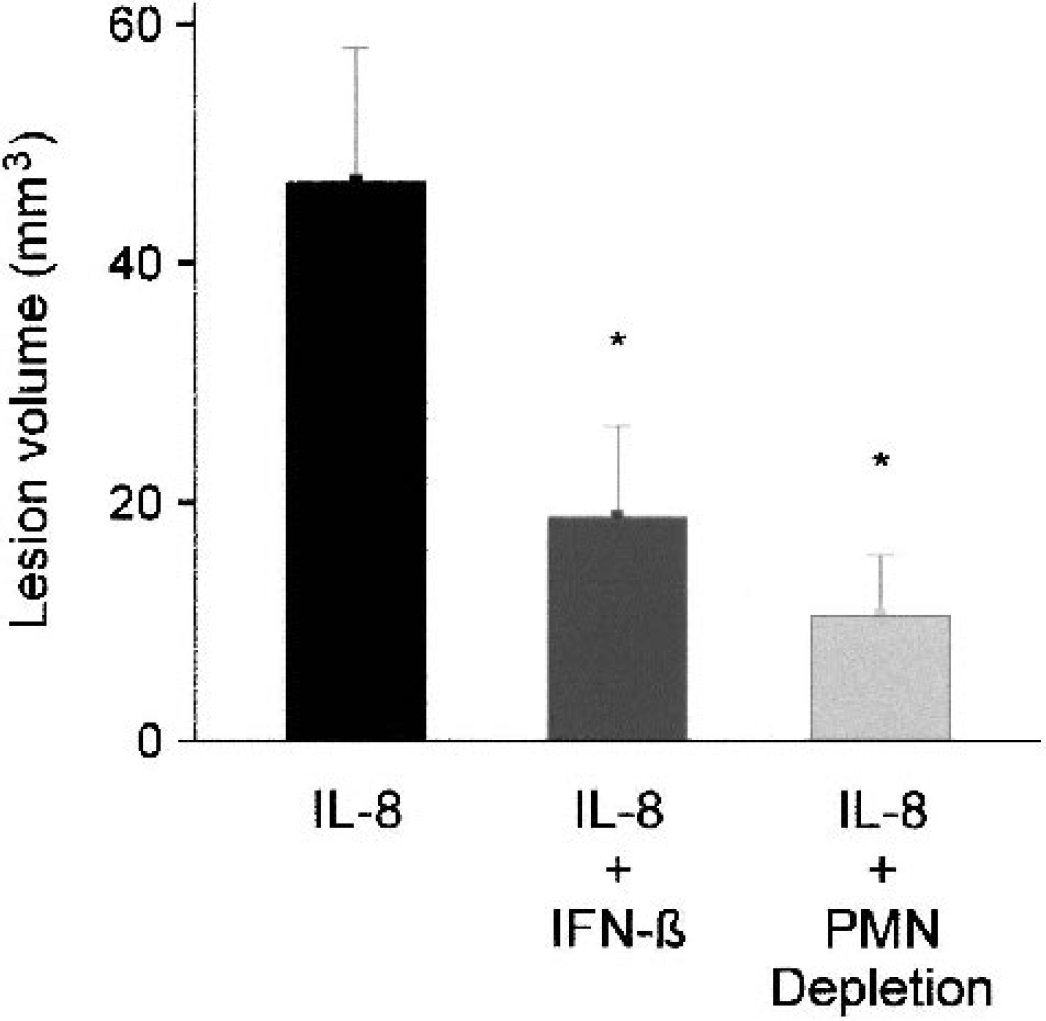
The total volume of brain tissue suffering from BBB disruption, including the region around the needle tract, as determined on the basis of a ≥2× SD increase in signal intensity on contrast-enhanced T1-weighted difference images (postcontrast – precontrast). Bars represent mean ± SD. *P < 0.05 versus IL-8 alone. IL-8, interleukin-8; IFN-β, interferon-beta; PMN, polymorphonuclear neutrophil.
Effects of IL-8 injection and therapeutic intervention on hemodynamic parameters
Because changes in rCBV have been reported after IL-1β–induced BBB breakdown (Blamire et al., 2000), we determined whether there were changes in hemodynamic parameters after IL-8 injection. In animals that received IL-8 alone, rCBFi in the injected striatum was slightly lower than in the untreated striatum, whereas after polymorphonuclear neutrophil depletion rCBFi was increased (Fig. 3A). After IFN-β treatment, IL-8 injection did not result in changes in cerebral blood flow (Fig. 3A). The changes in rCBFi could not be attributed to changes in either mean transit time or rCBV alone, but instead derived from slight variations in both. Changes in mean transit time and rCBV were only small and did not significantly differ among the treatment groups (Figs. 3B and 3C).
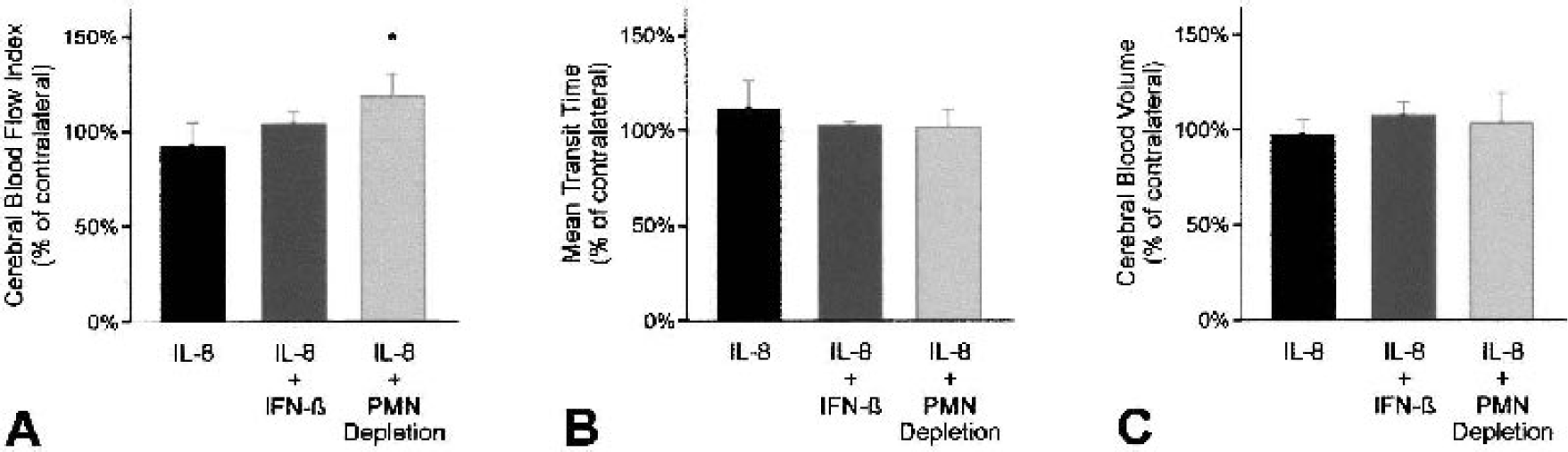
Effect of IL-8 injection, IFN-β treatment, and neutrophil depletion on hemodynamic parameters. Relative cerebral blood volume (A), mean transit time (B) and relative cerebral blood flow indices (C) were measured in the lesioned striatum 6 hours after injection and compared with the striatum on the contralateral side. Bars represent mean ± SD. *P < 0.05 versus IL-8 alone. IL-8, interleukin-8; IFN-β, interferon-beta; PMN, polymorphonuclear neutrophil.
Effect of IFN-β on neutrophil infiltration and cellular activation
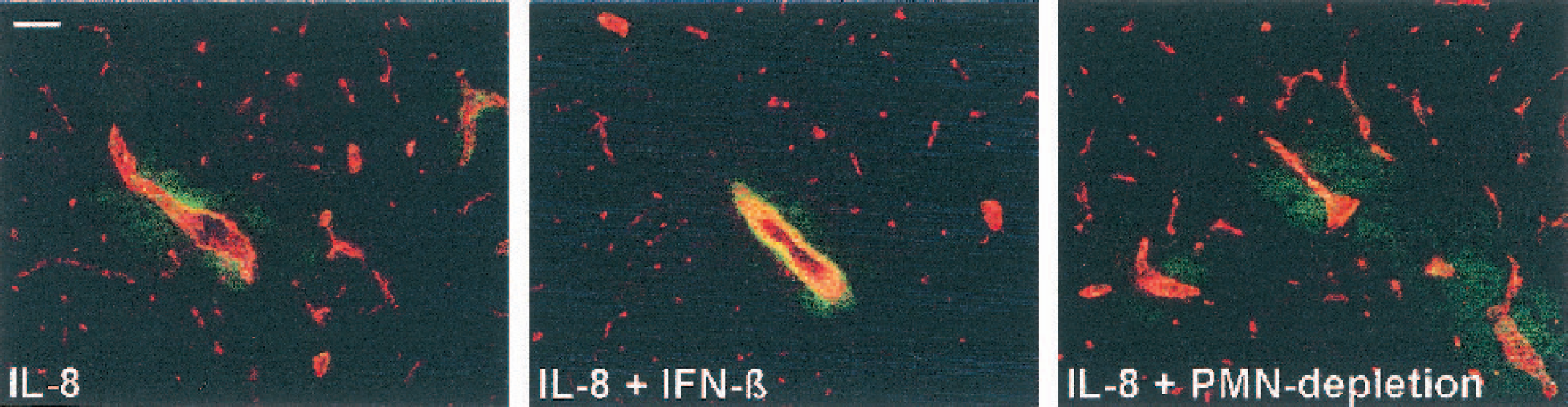
Immunofluorescent analysis was performed at the 6-hour time point to study the presence of neutrophils and analyze ICAM-1 expression and microglial activation in relation to the BBB breakdown detected by the MRI measurements. Intrastriatal injection of IL-8 resulted in massive infiltration of neutrophils, as identified by HIS48. Neutrophils were present in the perivascular space of cerebral blood vessels and in the brain parenchyma (Fig. 4). Furthermore, infiltrated cells expressed both CD45 (present on all leukocytes) and HIS48 epitope (present on neutrophils only), showing that all infiltrated cells were neutrophils (data not shown). In the animals that received IL-8 injection after neutrophil depletion, no cellular infiltrates were observed in the brain (Fig. 4). Similarly, treatment with IFN-β almost completely prevented the infiltration of neutrophils into the brain. Although in these animals careful scanning did reveal vessels with neutrophils, these were never found beyond the perivascular space (Fig. 4B).
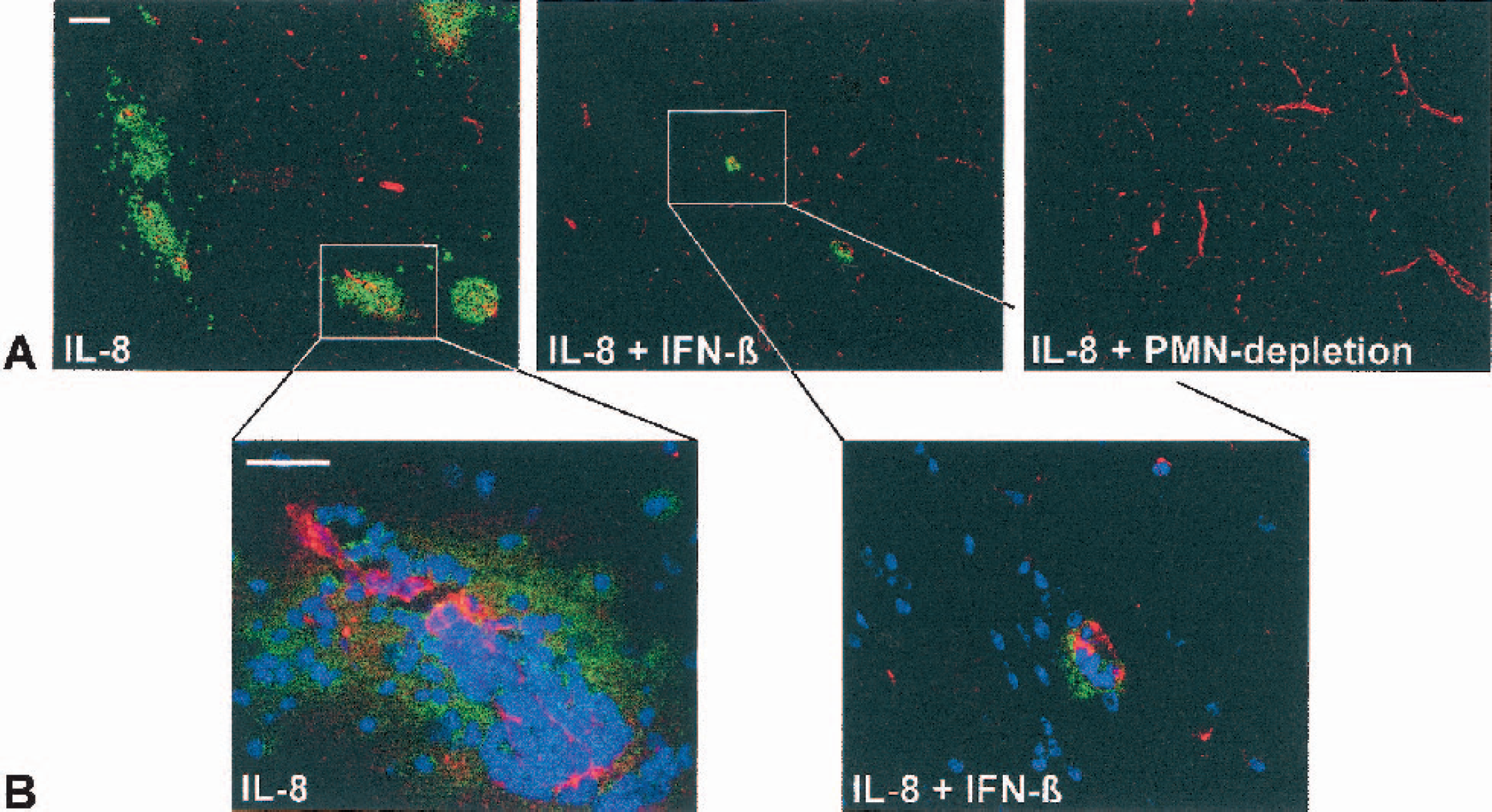
Effect of IFN-β treatment and neutrophil-depletion on IL-8–induced neutrophil infiltration. (A) Shown are sections through the striatum for all three treatment groups. HIS48-positive neutrophils (in green) are present around von Willebrand factor–positive blood vessels (in red) in the striatum 6 hours after injection of IL-8 alone (IL-8), but not after neutrophil depletion (IL-8 + PMN-depletion) or IFN-β treatment (IL-8 + IFN-β). Scale bar = 500 μm. (B) High-magnification image of the vessels indicated in panel
To investigate whether the observed effect of IFN-β was associated with a reduced activation of brain endothelial or microglial cells, ICAM-1 and iC3b receptor expression were analyzed. IL-8 injection resulted in a clear upregulation of ICAM-1 on the cerebrovasculature in the ipsilateral striatum (Fig. 5). No upregulation of ICAM-1 was detected in the contralateral hemisphere (data not shown). Neither polymorphonuclear neutrophil depletion nor IFN-β treatment prevented the upregulation of ICAM-1 (Fig. 5). Staining for the iC3b receptor (CD11b) showed that the influx of neutrophils and the breakdown of the BBB after IL-8 injection were not associated with microglial activation, as judged by the unchanged expression of CD11b (data not shown).
Expression of ICAM-1 (in green) on von Willebrand factor-positive vascular endothelium (in red) in the striatum 6 hours after IL-8 injection. Neither IFN-β treatment nor neutrophil depletion prevented the IL-8–induced upregulation of ICAM-1 expression. Scale bar = 250 μm. IL-8, interleukin-8; IFN-β, interferon-beta; PMN, polymorphonuclear neutrophil.
MMP-9 activity after IFN-β treatment
During extravasation into the brain parenchyma leukocytes not only have to traverse the tight endothelial layer, but also the basement membrane of the blood vessel endothelium. To this end, infiltrating leukocytes produce MMPs. Because the effect of IFN-β on monocyte and T-cell infiltration in patients with multiple sclerosis has been attributed to downregulation of MMP-9 expression (Galboiz et al., 2001; Stuve et al., 1997; Trojano et al., 1999; Waubant et al., 2001), and because MMP-9 inhibition in stroke reduces infarct size (Romanic et al., 1998), we performed gelatinase zymography, after gelatin-Sepharose purification, to assess the levels of MMP-2 and MMP-9 after IL-8 injection. The assay showed two bands for MMP-9: one at 92 kd, which corresponds to pro–MMP-9, and another at 120 kd, which has been reported to correspond to MMP-9 complexed to lipocalin (Yan et al., 2001). Lipocalin is coexpressed in neutrophils with pro–MMP-9 and enhances its direct activation (Tschesche et al., 2001). In the lesion homogenates of all animals, a clear band of pro–MMP-2 (72 kd) and a weak band of active MMP-2 were observed. The effect of neutrophil depletion or IFN-β treatment was not mediated by a decrease in MMP-2 expression (Fig. 6). After IL-8 injection, a clear band of pro–MMP-9 was observed. Neutrophil depletion before IL-8 injection resulted in a decrease in pro–MMP-9 gelatinase activity (Fig. 6). A similar reduction in MMP-9 activity was seen in IL-8–injected rats treated with IFN-β. In both treatments groups, the band at 120 kd was absent (Fig. 6).
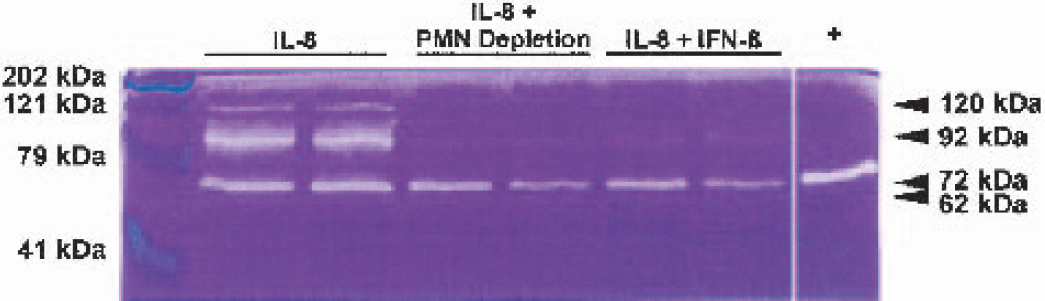
Gelatin zymogram showing upregulation of MMP-9 6 hours after intrastriatal IL-8 injection. IFN-β treatment or neutrophil depletion prevented the upregulation of the MMP-9 bands at 92 and 120 kd. Zymography was performed in duplicate. The lane labeled ‘+’ contains pro–MMP-2 from the SK-N-SH human neuroblastoma cell line. IL-8, interleukin-8; IFN-β, interferon-beta; PMN, polymorphonuclear neutrophil.
DISCUSSION
The mechanism of action of IFN-β as a treatment for the chronic inflammatory disease multiple sclerosis is incompletely understood. However, at least part of the benefit derived from its application is ascribed to reducing the influx of monocytes and T-cells into the brain (Floris et al., 2002; Yong et al., 1998). Recent experiments with IFN-β in an experimental stroke model suggested that the cytokine might also be of use as a treatment of brain insults that are accompanied by acute inflammation (Veldhuis et al., 2002). Therefore, in this study we have investigated the effect of IFN-β on IL-8–induced neutrophil infiltration into the rat brain, in a setting relevant to ischemic brain injury. The principal findings are that injection of IL-8 into the rat brain results in neutrophil-dependent BBB disruption, and that IFN-β prevents neutrophil infiltration and attenuates BBB disruption, presumably via an effect on MMP-9 release from neutrophils. These findings and their implications are discussed below.
Intracerebral IL-8 injection results in neutrophil influx and BBB disruption
After ischemia or trauma, proinflammatory cytokines are produced, which results in recruitment of neutrophils to the lesion area, breakdown of the BBB, and secondary tissue destruction. The present data show that 6 hours after injection of the chemokine IL-8 into the rat striatum, large numbers of neutrophils have entered the perivascular space and are infiltrating the brain parenchyma. This infiltration was accompanied by extensive damage to the BBB. Furthermore, BBB disruption did not occur after IL-8 injection in neutrophil-depleted animals. These findings of infiltration and neutrophil-dependent BBB disruption within hours after injection are in agreement with results from other studies inducing neutrophil recruitment by injecting the pleiotropic inflammatory mediator IL-1β (Bernardes-Silva et al., 2001; Blamire et al., 2000). However, IL-1β mediates many effects apart from inducing neutrophil influx (Rothwell and Luheshi, 2000; Wang and Shuaib, 2002). Even though IL-1β may be produced after injection of IL-8 (Matsukawa et al., 1995), the changes in rCBV and apparent diffusion coefficient seen after injections of IL-1β (Blamire et al., 2000) were not detected after injection of IL-8 in the current study. Blamire et al. (2000) were careful to point out that the IL-1 β–induced changes in apparent diffusion coefficient also occurred in neutrophil-depleted animals and may be related to direct cytotoxic effects of the injected cytokine. In addition, they have shown that the influx induced by IL-1β injection is mediated by IL-8 (Anthony et al., 1998). Intracerebral injection with TNF-α, another pleiotropic inflammatory cytokine relevant to ischemic injury (Barone et al., 1997; Wang and Shuaib, 2002), was also shown to induce both apparent diffusion coefficient and rCBV changes and induce BBB permeability (Sibson et al., 2001; Sibson et al., 2002). Although minor neutrophil infiltration occurred in those studies, injection of TNF-α into gliomas resulted in extensive neutrophil influx, which also contributed to IL-8 production (Tada et al., 1993). Thus, by using IL-8 in the current setup, we attempted to stimulate neutrophil recruitment with as few side effects as possible, enabling the direct assessment of the effect of IFN-β treatment on this influx. We did, however, detect a small decrease in rCBFi in the ipsilateral striatum after IL-8 injection. The cause for this decrease is unclear, but may be related to neutrophil recruitment, presumably reflecting their largescale attachment to the cerebral microvascular endothelium (Mori et al., 1992).
IFN-β prevents IL-8–induced neutrophil influx and attenuates BBB disruption
The current data show that IFN-β strongly limits the IL-8–induced influx of neutrophils into the brain. Therefore, IFN-β can directly influence neutrophil infiltration across the BBB. A tentative mechanism underlying the observed inhibition of neutrophil infiltration would be the prevention of ICAM-1 upregulation (Der et al., 1998). This mechanism has been suggested to play a role in the effect of IFN-β against multiple sclerosis (Trojano et al., 2000), although observations to the contrary have also been made (Corsini et al., 1997). We demonstrated that after IL-8 injection, ICAM-1 expression was upregulated in the ipsilateral striatum. However, neither IFN-β treatment nor neutrophil depletion prevented the upregulation of ICAM-1. Subsequent immunofluorescence analysis of the brains of animals used in the previously discussed stroke study (Veldhuis et al., 2002) showed that the protection afforded by IFN-β against transient ischemia was also not mediated by an effect on ICAM-1 expression (W. B. Veldhuis, unpublished observations, 2002). Thus, IFN-β prevented the influx of neutrophils without preventing the upregulation of membrane-bound ICAM-1. Increased shedding of membrane-bound adhesion molecules, thereby increasing the soluble fraction of the adhesion molecule in the blood stream, has been reported for both vascular cell adhesion molecule and ICAM after IFN-β treatment in multiple sclerosis (Calabresi et al., 2001; Kallmann et al., 2000; Trojano et al., 1999). The soluble form of adhesion molecules may inhibit the adhesion of leukocytes to endothelial cells by binding to their leukocyte counterreceptors in the bloodstream. This mechanism may have contributed to the observed reduction in neutrophil influx reported here after IL-8 injection in IFN-β–treated animals.
IFN-β reduces MMP-9 expression
Interferon-beta may impede the access of inflammatory cells to the brain parenchyma not only via an effect on adhesion molecule expression, but also via downregulation of proteolytic MMP-9 activity (Galboiz et al., 2001; Stuve et al., 1997; Trojano et al., 1999; Waubant et al., 2001). In the current study, we show that IFN-β directly prevents neutrophil-mediated BBB disruption. Using gelatinase zymography, the BBB disruption depended on both the presence of neutrophils and the expression of MMP-9. After cerebral ischemia, IL-8–induced neutrophil chemotaxis is central to the acute phase of the inflammatory response. The finding that IFN-β reduces MMP-9 in this setting is of putative therapeutic relevance, because it has been shown that knocking out or inhibiting MMP-9 reduces ischemic brain injury in the mouse (Asahi et al., 2000) and rat (Romanic et al., 1998). Moreover, MMP-9 is also directly associated with the risk of hemorrhagic transformation after stroke in general (Montaner et al., 2001) and after tissue plasminogen activator treatment in particular (Sumii and Lo, 2002). Tissue plasminogen activator is currently the only treatment available for acute ischemic stroke patients, and its application is limited by this risk of hemorrhagic transformation. MMP-9 is probably more important in this respect than MMP-2, because in the rat, MMP-2 was shown to be constitutively present in the brain and transient (1-hour) ischemia resulted in a greatly enhanced MMP-9 activity, whereas MMP-2 only very slightly increased during the first days (Planas et al., 2001). In a murine stroke model, MMP-2 activity was not increased after ischemia, and knocking out MMP-2 had no effect on brain injury after permanent or transient (2-hour) ischemia (Asahi et al., 2001).
Importantly, in neutrophil-depleted animals MMP-9 expression was also diminished, whereas no difference in MMP-2 expression was observed. This suggests that neutrophils are the predominant source of MMP-9 in this setting. This suggestion and its relevance to cerebral ischemia are directly confirmed by the results from a recent study in which bone marrow cells from MMP-9 knockout mice were transplanted into irradiated wild-type mice or wild-type cells were transplanted into MMP-9 knockouts, after which the mice were subjected to ischemia (Shah et al., 2002). Mice transplanted with MMP-9–knockout cells showed less BBB injury, smaller infarct volumes, and reduced neutrophil infiltration, whereas transplantation with wild-type cells did not afford any protection. Thus, we propose that IFN-β prevents the release of MMP-9 from neutrophils, thereby preventing their infiltration and preventing the escalation of inflammation.
It has also been shown that IFN-β directly reduces MMP-9 mRNA levels in nonimmune cells; e.g., in human astroglioma and fibrosarcoma cell lines and primary astrocytes stimulated with TNF-α (Ma et al., 2001). In addition, IL-8 may directly induce MMP-9 (Cross and Woodroofe, 1999), which may be prevented by IFN-β. However, given the absence of MMP-9 expression in neutrophil-depleted animals, these mechanisms most likely do not underlie the effects of IFN-β in the current setting.
Finally, we cannot exclude the possibility that the effect on MMP-9 was secondary to an as yet undefined effect of IFN-β on neutrophil activity or chemotaxis. Regarding the latter possibility, the only identified direct effect of IFN-β on neutrophil chemotaxis is the inhibition of IL-8 mRNA expression (Oliveira et al., 1992). Although this is of possible relevance to IFN-β treatment in a setting of ischemia, it is unlikely to explain the results in the current assay, where IL-8 was exogenously administered.
CONCLUSION
We have shown that IFN-β inhibited IL-8–induced neutrophil pleocytosis and BBB damage in the rat brain, and that this effect was presumably mediated by reducing MMP-9 release from neutrophils. The latter may be of relevance to thrombolytic therapy in acute ischemic stroke because MMP-9 is directly associated with the risk of hemorrhagic transformation, both after stroke in general (Montaner et al., 2001), and after tissue plasminogen activator treatment in particular (Sumii and Lo, 2002).
Interferon-beta may thus be used as a (co)treatment in disorders where acute inflammation contributes to brain injury, particularly brain ischemia (Matsumoto et al., 1997b; Veldhuis et al., 2002), but also for example traumatic brain injury (Kossmann et al., 1997), or lipopolysaccharide-induced meningitis (Dumont et al., 2000). Because IFN-β has already been approved for other human use, it may be tested with relative rapidity in a clinical trial for its efficacy in these disorders.
