Abstract
Experimental and clinical studies have provided evidence for spontaneous and therapeutically induced arteriogenesis after occlusion of major peripheral or cardiac vessels. Such evidence is lacking for the cerebrovascular system. In halothane-anesthetized rats, different degrees of brain hypoperfusion were induced by one- to four-vessel occlusion, that is, one or both common carotid arteries in combination with or without bilateral vertebral artery occlusion. The flow decline was monitored by laser Doppler flowmetry, the residual hemodynamic reserve by testing flow reactivity to ventilation with 6% CO2 and arteriogenesis by intravascular latex infusion and immunohistochemistry of vascular proliferation and monocyte adhesion. The optimum condition for induction of arteriogenesis was three-vessel (one carotid plus both vertebral arteries) occlusion, which led to reduction of blood flow to about 50% and complete suppression of CO2 reactivity, but no histologic injury. One week after three-vessel occlusion, the ipsilateral posterior cerebral artery significantly enlarged by 39%, and after 3 weeks by 72%, paralleled by the partial return of CO2 reactivity and the appearance of immunohistochemical markers of arteriogenesis. Three-vessel occlusion is a reliable model for the induction of arteriogenesis in the adult brain and is a new approach for exploring the potentials of arteriogenesis for the prevention of progressing brain ischemia.
Keywords
Experimental and clinical studies have provided evidence that therapeutically induced growth of collateral arteries and capillary networks may reduce ischemic injury in endangered territories of the peripheral and cardiovascular system (Arras et al., 1998; Buschmann et al., 2001; Schaper, 2001; Seiler et al., 2001; van Royen et al., 2001a, b ). Three different forms of vessel growth can be distinguished: vasculogenesis, angiogenesis, and arteriogenesis (Arras et al., 1998; Buschmann and Schaper, 1999; Ito et al., 1997a; Schaper and Buschmann, 1999). Vasculogenesis describes the formation of vessels by angioblasts during early ontogenesis (Risau, 1997). Angiogenesis produces capillary networks by sprouting or de novo growth, the main driving force being hypoxia, which via regulation of the hypoxia-inducible factor leads to increased transcription of angiogenic factors, such as vascular endothelial growth factor (VEGF) (Carmeliet et al., 1998; Deindl et al., 2001; Risau, 1997). Arteriogenesis refers to the adaptive growth of preexisting arteries in response to an increase of intravascular shear forces. Hemodynamically relevant obstruction or complete occlusion of a large artery causes redistribution of blood flow due to the change of pressure gradients across interconnecting arterioles (Ito et al., 1997b; Van Royen et al., 2001b). As a consequence, preexisting collaterals are recruited and intravascular shear stress increases. This process leads to the activation of endothelial cells, followed by an upregulation of cell adhesion molecules and the attraction of circulating monocytes and mononuclear cells (Arras et al., 1998; Ito et al., 1997a, b ). These cells release various chemokines, colony-stimulating factors, and growth factors, which contribute to the adaptive growth of the collateral vessels.
Recent experimental investigations have shown rapid proliferation of preexisting vessels after occlusion of a major coronary or peripheral artery, and significant improvement of blood supply by stimulation of collateral growth via infusion of monocyte chemoattractant protein-1 or granulocyte-macrophage colony-stimulating factor (Arras et al., 1998; Buschmann et al., 2001; Ito et al., 1997a; Seiler et al., 2001; van Royen et al., 2001a). These collateral pathways were able to restore tissue perfusion in the endangered territories (Buschmann and Schaper, 1999; Buschmann et al., 2001; Hoefer et al., 2001; van Royen et al., 2001a, b ), indicating the enormous potential for the treatment of peripheral and cardiovascular disease (Arras et al., 1998; Ito et al., 1997a; Schaper, 2001).
In the cerebrovascular system, similar studies have not been carried out. This is surprising because a multitude of extra- and intracranial collateral systems, such as the leptomeningeal anastomoses of Heubner, the anastomotic pathways via the ophthalmic artery, or the circle of Willis provide the chance of improving blood supply under conditions of slowly progressing vascular occlusion (Hossmann, 1993). In fact, there is evidence that the hemodynamic reserve of the cerebrovascular system correlates inversely with the severity of cerebral infarction and, in turn, seems to rely on the efficacy of the collateral circulation (Coyle and Jokelainen, 1983; Hendrikse et al., 2001; Kluytmans et al., 1998; Ringelstein et al., 1994). Amelioration of the cerebrovascular hemodynamic reserve by improving collateral cerebral circulation via induced arteriogenesis is therefore a promising approach for the prevention of cerebrovascular disorders.
In the present investigation we describe an animal model in which cerebral arteriogenesis can be investigated under controlled conditions. By comparing the effects of various combinations of extracranial vascular occlusions, we observed that the occlusion of one common carotid in combination with both vertebral arteries (three-vessel occlusion, 3-VO) induces hemispheric hypoperfusion that is not severe enough to produce histologic damage, but induces the shear stress required for the adaptive growth of the nonoccluded arteries.
MATERIALS AND METHODS
Animal model
Experiments were carried out with permission of state authorities according to the German Law for Protection of Animals, and the National Institute of Health Guidelines for Care and Use of Laboratory Animals (NIH Publication No. 85–23, revised 1996).
Forty-three adult, male Sprague-Dawley rats (weight 290 to 340 g) were used. For the development of an extracranial vascular occlusion model, 11 animals were randomly assigned to uni- or bilateral common carotid and vertebral artery occlusions in different combinations. This study led to the identification of a 3-VO model (left common carotid and bilateral vertebral artery occlusion) for induction of nonlethal brain hypoperfusion, which was subsequently used for the investigation of cerebral arteriogenesis.
To this end, four groups were compared: nonoperated controls (n = 8), animals studied immediately after 3-VO (n = 8), and animals investigated after 1 (n = 8) and 3 weeks survival (n = 8), respectively. At the end of experiments, animals were perfused either with colored latex for visualization of their cerebrovascular anatomy, or with 4% paraformaldehyde for histologic and histochemical evaluations.
Surgical methods
Anesthesia was induced with 3.5% to 4% halothane and maintained with 0.8% to 1.5% halothane in 70%/30% nitrous oxide/oxygen. Vascular occlusions were carried out in one session by first electrocoagulating both vertebral arteries, using a paravertebral access (Pulsinelli and Brierley, 1979; Pulsinelli et al., 1983), followed within the next hour by uni- or bilateral common carotid artery occlusion. The latter was done by ligation after ventral surgical midline incision under continuous monitoring of laser Doppler flow (see next section).
After vertebral artery coagulation, three animals (9%) died because of uncontrollable bleeding. In the other animals, wounds were closed and infiltrated with bupivacaine 0.25%. Thereafter, the animals were returned in pairs to their cages with free access to food and water, and were allowed to move freely.
Cerebral blood flow and cerebrovascular reactivity
Blood flow was measured by transcranial laser Doppler flowmetry (LDF; PeriSoft; PeriMed, Järfälla, Sweden). The skull above the frontoparietal cortex was bilaterally exposed, and laser probes were placed directly on the bone.
For the assessment of ischemia, flow measurements were started after vertebral artery coagulation and then continued for up to 1 h after uni- or bilateral carotid artery occlusion. Cerebral vascular reactivity was tested by recording blood flow during ventilation with 6% CO2. To this end, animals were tracheotomized, paralyzed with pancuronium bromide (0.2 mg · kg−1 · h−1), and mechanically ventilated with a small animal respirator. Body temperature was kept constant at 37°C, using a feedback-controlled heating pad, and blood gases were measured in arterial samples, drawn from one femoral artery. Arterial PCO2 was raised by connecting the input of the animal respirator for approximately 6 minutes to a certified gas bottle, containing 6% CO2, 30% oxygen and 64% nitrous oxide. The effect on blood flow was monitored by LDF and expressed as percent deviation from baseline per millimeter mercury increment in arterial PCO2. Measurements were repeated three times per animal, with enough recovery time to allow return of blood gases to the control range.
Postmortem cerebral angiograms.
Cerebrovascular anatomy was studied by a modification (Maeda et al., 1998) of the latex perfusion method of Coyle (Coyle, 1984; Coyle and Heistad, 1991; Coyle and Panzenbeck, 1990). In six animals of each group, the common carotid artery ligated for 3-VO was cannulated with polyethylene tubing (internal diameter 0.75 mm; SIMS Portex, Hythe, U.K.), and a lethal dose of papaverine hydrochloride (40 to 50 mg/kg) was intraarterially injected to produce maximal vasodilatation. Undiluted warm (37°C) latex (Chicago Latex Products no. 563) was mixed with a small amount of carbon black (10 μL/g, Bokusai; Fueki, Tokyo, Japan). Of this solution, 3.5 to 4 mL/kg was injected through the intracarotid catheter at 150 mm Hg pressure. Thoracotomy was performed, and the right atrium was incised to facilitate venous outflow. To harden the latex, animals were placed on crushed ice for 15 minutes. Subsequently, brains were carefully removed and fixed by immersion in 4% paraformaldehyde. To assess arteriogenesis, the external diameter was measured at five levels of both internal carotid arteries, anterior cerebral arteries, middle cerebral arteries, the posterior communicating artery, and both posterior cerebral arteries (PCA). Measurements were carried out using a stereo-zoom microscope with a calibrated eyepiece micrometer. Because no consistent differences were observed between the different levels of each vessel, measurements were combined into one mean value.
Histologic and histochemical analysis
For animals submitted to 3-VO, brains were examined histologically to exclude neuronal damage. Brains were frozen in liquid nitrogen and sliced at −20°C into coronal sections. Cryosections were thaw-mounted on poly-L-lysine–coated glass slides and stained with cresyl violet and RhoB antibody (RhoB[119]: SC-180; Santa Cruz Biotechnology, Santa Cruz, CA, U.S.A.). Degenerating neurons were identified by the terminal deoxynucleotidyl transferase-mediated 2′-deoxyuridine 5′-triphosphate-biotin nick end labeling (TUNEL) method (TUNEL-Kit; Boehringer-Mannheim, Mannheim, Germany). For the histologic evaluation of the PCA, cerebral vasculature was perfused in two animals of each group with 4% paraformaldehyde at 3 d after 3-VO, embedded in paraffin and sectioned for immunohistochemistry and hematoxylin–eosin staining. Proliferating cells were identified by antibody against Ki-67 antigen (Dako, Hamburg, Germany) after heat-induced epitope retrieval. Antibody recognizing ED-1 antigen of rat monocytes and macrophages (Acris, Hiddenhausen, Germany) was used to identify adventitial cells.
Data analysis.
All data are given as mean ± SD. Changes of laser Doppler flow are expressed as percentage of the mean baseline value. Group differences for hemodynamic, morphologic, and general physiologic measurements were analyzed for statistical significance by Student's t-test. Statistical significance was assumed for P < 0.05.
RESULTS
Induction of brain hypoperfusion
To establish a nonlethal model of brain hypoperfusion, common carotid and vertebral arteries were occluded extracranially in different combinations (Fig. 1). Monitoring of blood flow in parietal cortex by LDF revealed declining flow values in the following order: unilateral common carotid artery occlusion, unilateral common carotid artery occlusion in combination with bilateral vertebral artery occlusion (3-VO), bilateral common carotid artery occlusion, and bilateral common carotid artery occlusion in combination with bilateral vertebral artery occlusion (4-VO). Both bilateral common carotid artery occlusion models led to bilateral flow decline to less than 35% of control, which according to the threshold concept of brain ischemia is less than the flow rate required to preserve brain integrity. The unilateral common carotid artery occlusion models did not affect contralateral blood flow, but resulted in ipsilateral flow decline to about 55% of control, irrespective of the absence or presence of vertebral artery occlusion. However, with ongoing occlusion time, blood flow remained at the 50% level only in the 3-VO model, whereas it returned close to control within a few minutes when vertebral arteries remained patent (Figs. 2 and 3).
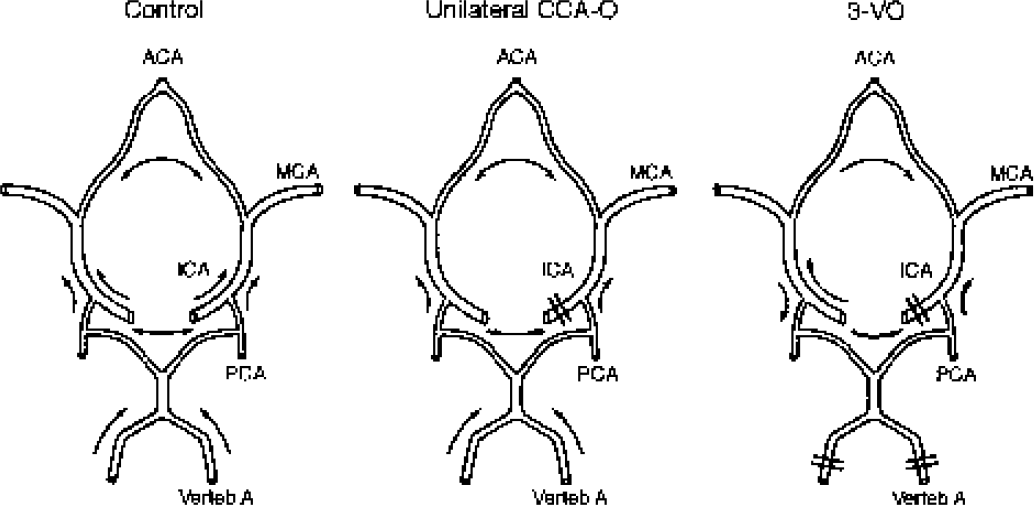
Arterial circle of Willis of the rat, representing different patterns of collateral blood flow under control conditions, after unilateral common carotid occlusion (CCA-O) and after occlusion of one common carotid and both vertebral arteries (three-vessel occlusion [3-VO]). ACA, anterior cerebral artery; MCA, middle cerebral artery; PCA, posterior cerebral artery; ICA, internal carotid artery; verteb A, vertebral artery.
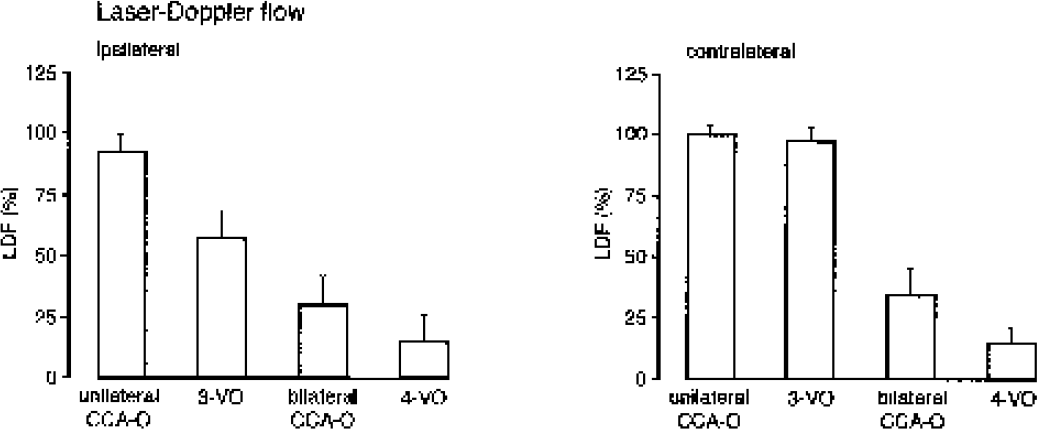
Measurement of cerebral blood flow by laser Doppler flowmetry (LDF) in the parietal cortex of unilateral common carotid artery occlusion (CCA-O), unilateral CCA-O in combination with bilateral vertebral artery occlusion (three-vessel occlusion [3-VO]), bilateral CCA-O, and bilateral CCA-O in combination with bilateral vertebral artery occlusion (four-vessel occlusion [4-VO]). Note reduction after 3-VO of ipsilateral blood flow by about 50%.
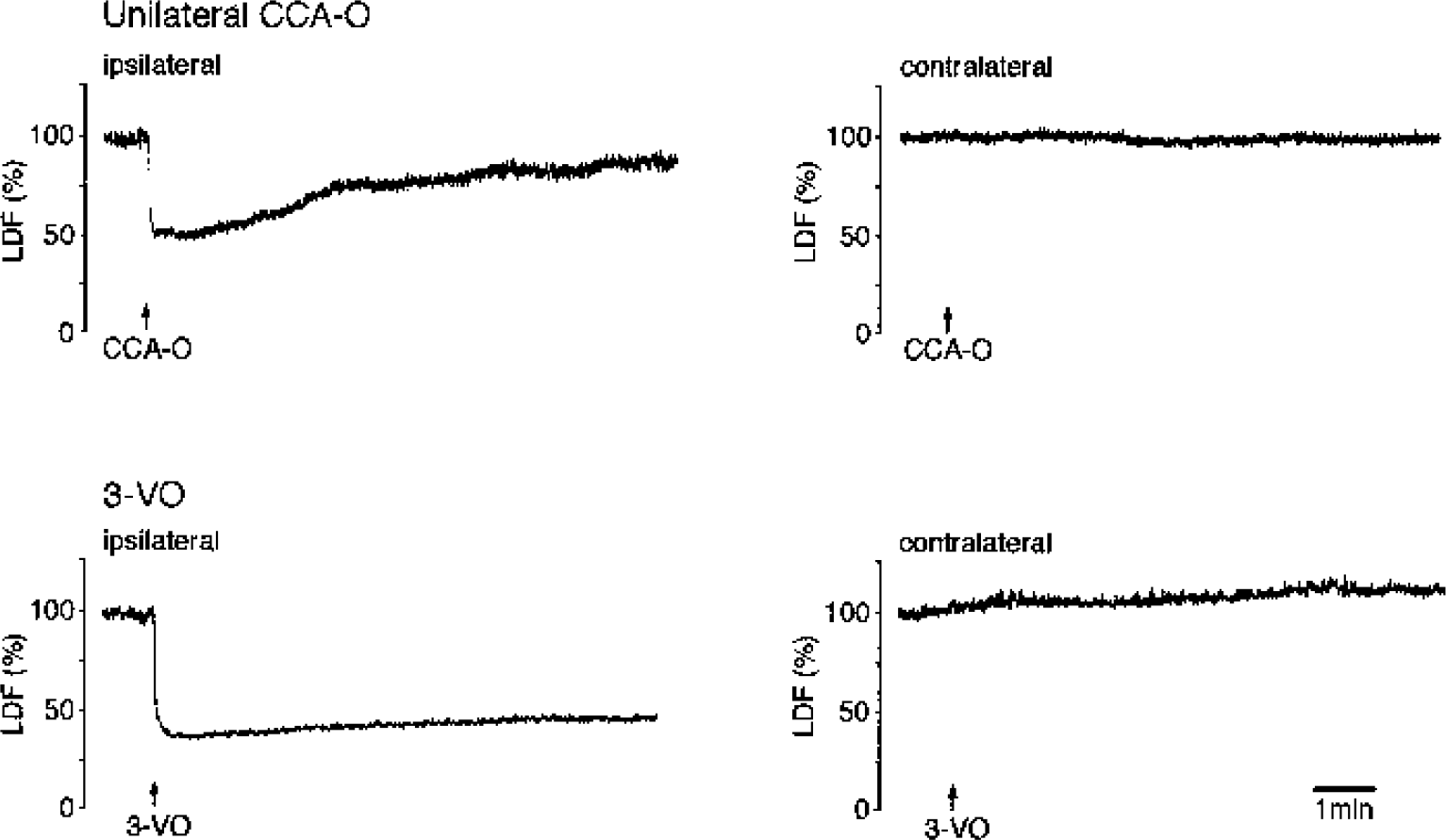
Representative recordings of cerebral blood flow by laser Doppler flowmetry (LDF) after unilateral common carotid artery occlusion (CCA-O) and three-vessel occlusion (3-VO) in the parietal cortex of the ipsilateral and contralateral hemisphere. In animals with unilateral CCA-O, cerebral blood flow returns to normal within a few minutes, whereas after 3-VO cerebral blood flow is permanently reduced.
To evaluate the morphologic outcome of the 3-VO model, histology was performed in four animals at 3 d after vascular occlusion. During the survival time, animals lost about 2% of their body weight, but none of them exhibited noticeable neurologic deficits. Histologic evaluation of cryostat sections using cresyl violet staining, RhoB immunohistochemistry, and TUNEL also failed to reveal any abnormalities. Even in the dorsolateral striatum and the selectively vulnerable CA1 sector of hippocampus, morphologic changes were absent, quite in contrast to middle cerebral artery occlusion, which was coprocessed as a positive control (Fig. 4). The 3-VO thus fulfills the requirement of a nonlethal brain hypoperfusion model and was further investigated for assessing cerebral arteriogenesis.
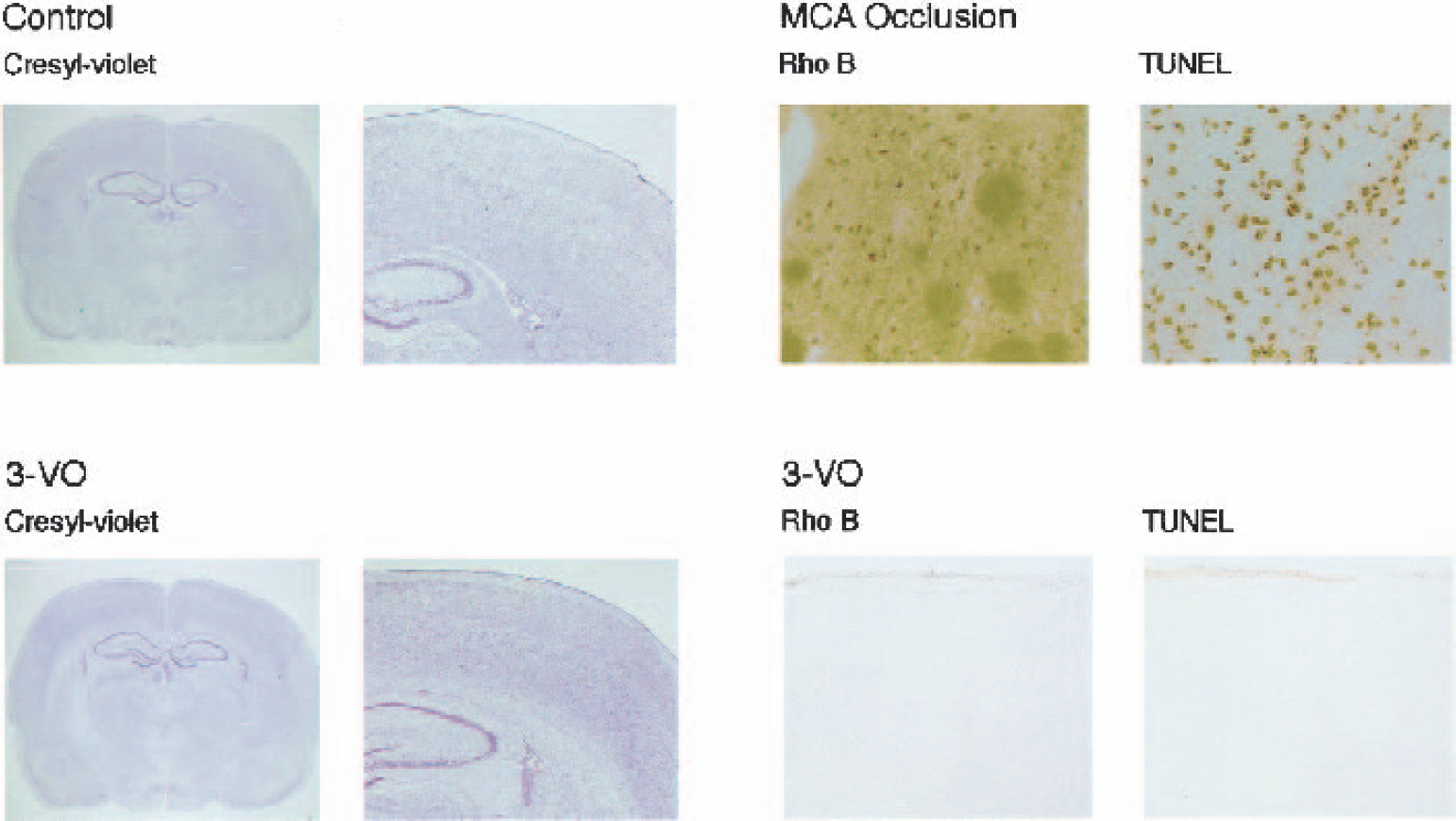
Morphologic evaluation of rat brain at 3 d after three-vessel occlusion (3-VO).
Cerebral arteriogenesis after three-vessel occlusion
The physiologic variables of animals submitted to this part of the study did not differ between groups (Table 1). There was also no significant difference in the initial LDF decline after vascular occlusion: 46.9 ± 8%, 39.7 ± 16%, and 51.0 ± 12% in experiments terminated immediately, 1 week or 3 weeks after onset of hyperperfusion, respectively (values refer to the hypoperfused hemisphere ipsilateral to the occluded common carotid artery).
General physiologic variables after three-vessel occlusion
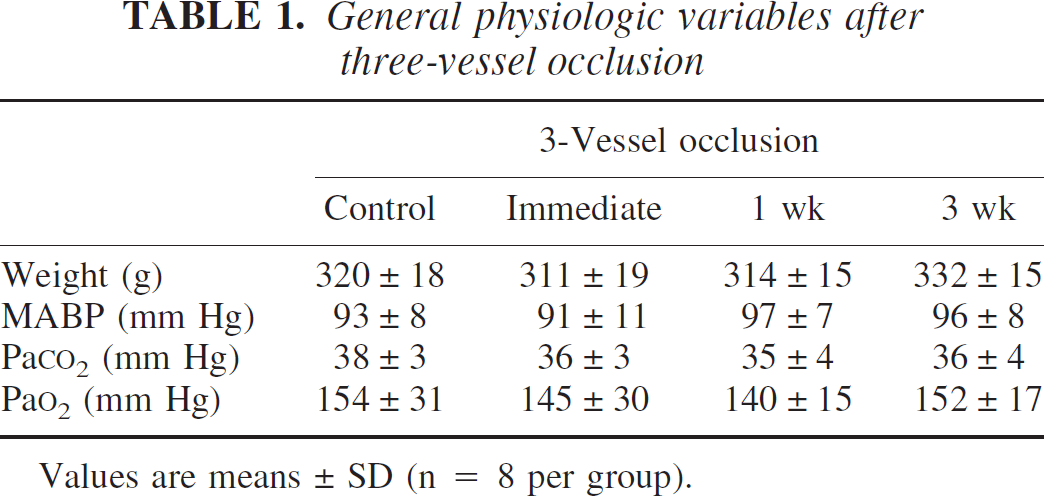
Values are means ± SD (n = 8 per group).
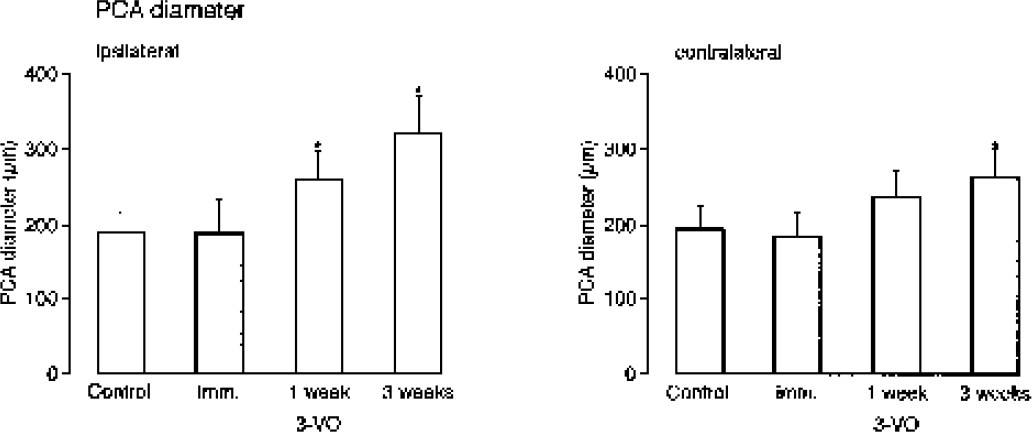
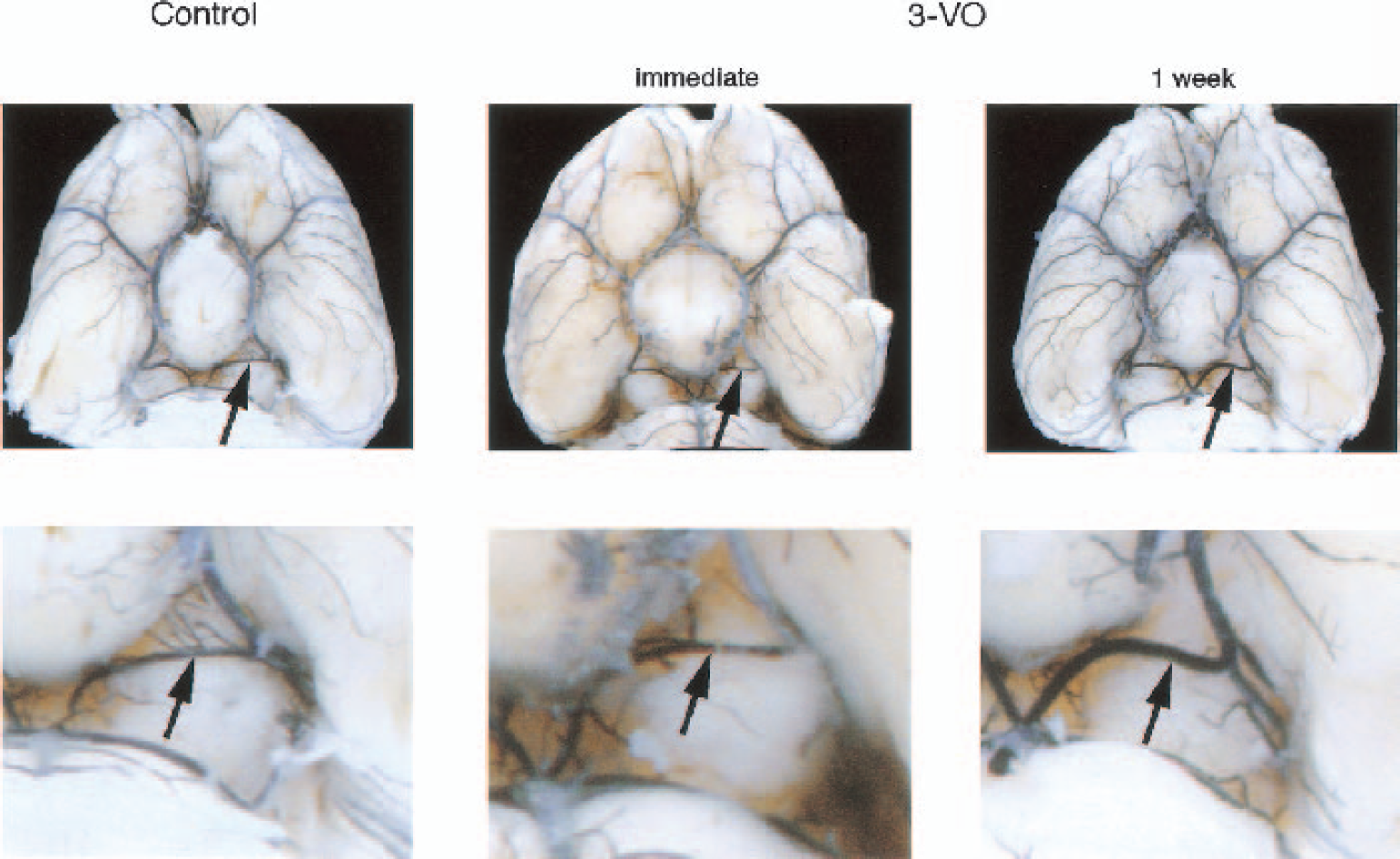
Induction of vascular growth by 3-VO was studied using the cerebrovascular latex perfusion method, which represents the maximal diameter to which the vessels are able to dilate after complete vasoparalysis and which reflects the anatomic size of the vessels (Fig. 5). The external diameter of the main supplying arteries of the brain was measured at the various survival times after 3-VO and was compared with nonischemic control animals (Fig. 6, Table 2). Immediately after 3-VO, no changes were observed, showing that the application of a high dose of papaverine before latex perfusion produced maximal vasodilation that was unaffected by intravital differences in vascular tone. With longer survival time, however, distinct increases in vessel size could be detected. The diameter of the ipsilateral PCA significantly increased from 187 ± 27 to 260 ± 37 μm (plus 39%) at 1 week, and further to 322 ± 50 μm (plus 72%) after 3 weeks after 3-VO. At this time, the ipsilateral anterior cerebral arteries also significantly enlarged, from 251 ± 37 to 322 ± 43 μm (plus 28%). The other supplying arteries did not change.
Changes in external vascular diameter (μm) of brain arteries after three-vessel occlusion
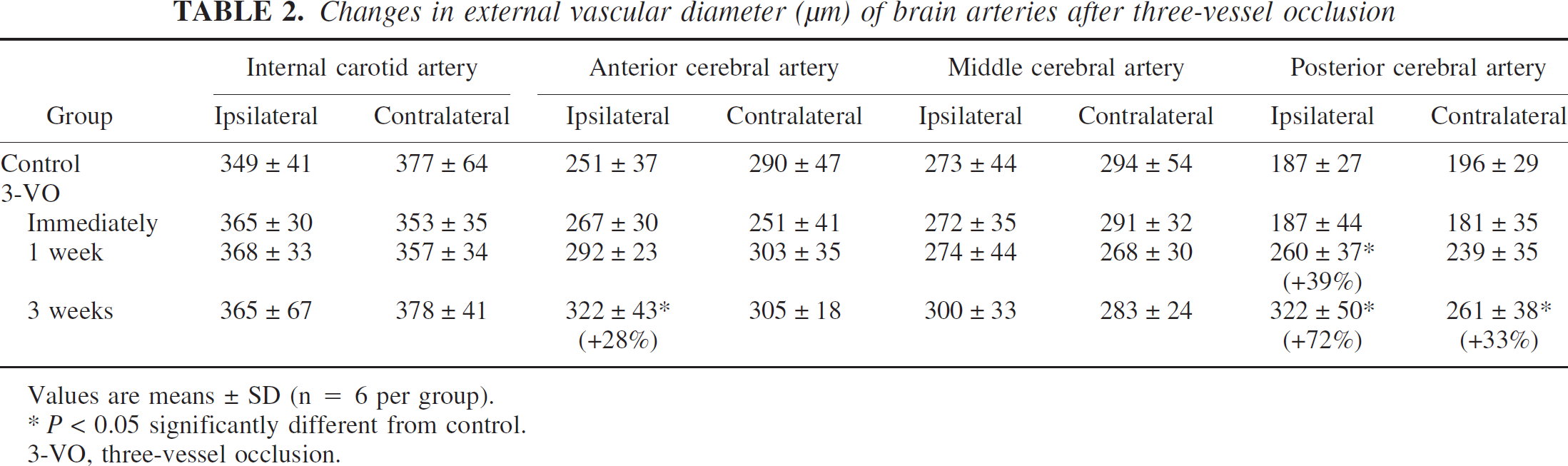
Values are means ± SD (n = 6 per group).
P < 0.05 significantly different from control.
3-VO, three-vessel occlusion.
Visualization of cerebral angioarchitecture by intravascular latex perfusion. Basal view of control brain and of brains inspected immediately or 1 week after three-vessel occlusion (3-VO). The arrows point to the ipsilateral posterior cerebral artery. Note absence of alterations immediately after three-vessel occlusion, but marked increase of vessel diameter after 1 week.
External diameter of posterior cerebral artery (PCA) in control rats and in rats studied immediately, 1 week, or 3 weeks after three-vessel occlusion (3-VO). Note gradual extension of vessel diameter in ipsilateral—and to a lesser degree also in contralateral—hemisphere. *P < 0.05 as compared with control.
As shown in peripheral vessels, arteriogenesis is associated with monocyte accumulation and proliferation of endothelial cells (Arras et al., 1998; Ito et al., 1997a, b ). Immunohistochemical evaluation of the proliferation marker Ki-67 and the monocyte–macrophage–recognizing ED-1 antigen revealed an increase of these arteriogenic markers in the enlarged ipsilateral, but not in the contralateral PCA at 3 weeks after 3-VO (7.4 ± 1.7 macrophages per field at 40× magnification in the ipsilateral PCA as compared with 2.7 ± 1.2 macrophages in sham-operated (P < 0.05) and 3.5 ± 1.1 macrophages in the contralateral PCA (P < 0.05); Fig. 7). In contrast, the hypoxia-dependent angiogenesis marker VEGF was unchanged, both in controls and in animals submitted to 3-VO (data not shown).
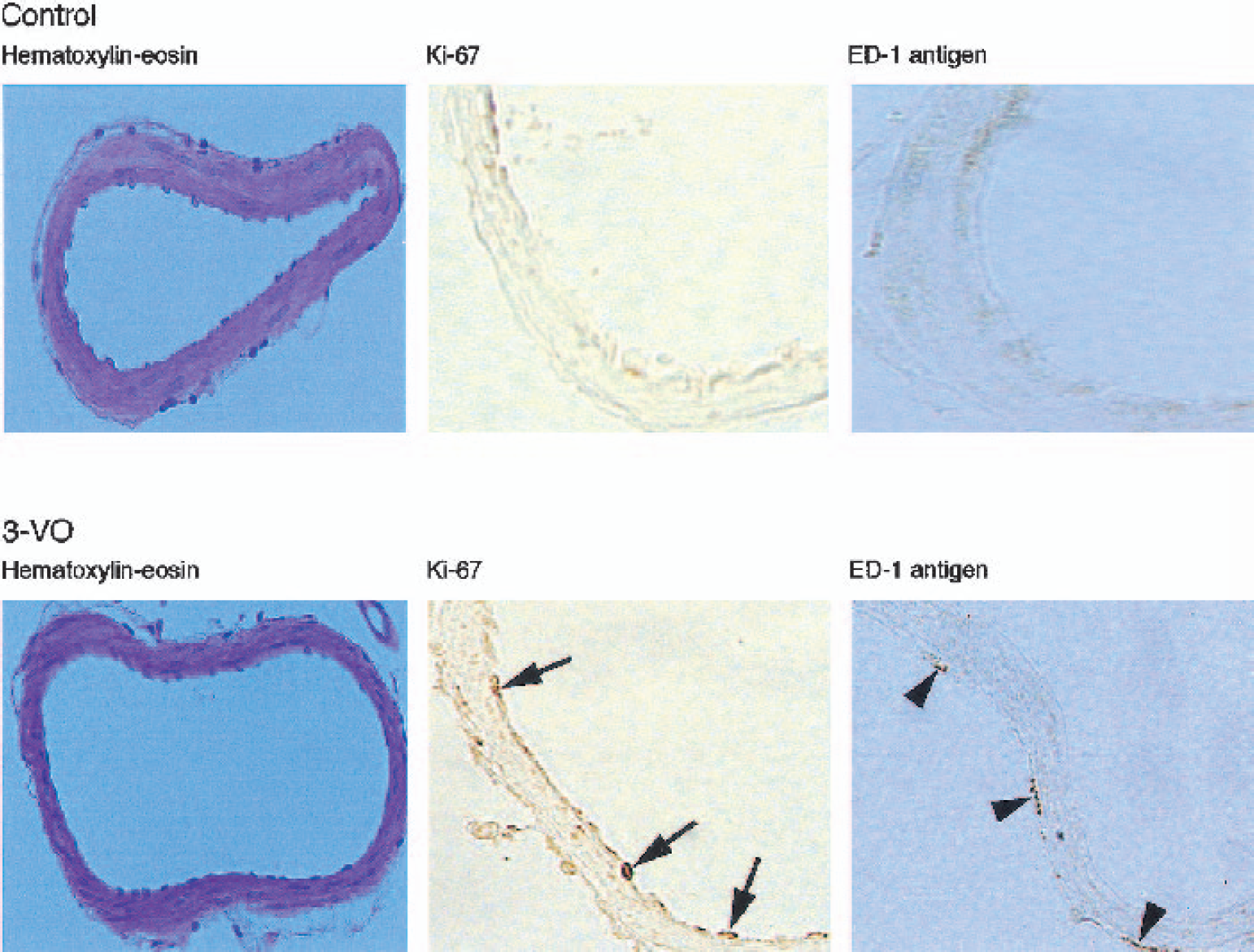
Cross-section of ipsilateral posterior cerebral artery of intact rat (control) and 3 weeks after three-vessel occlusion (3-VO). Note 3-VO–induced enlargement of vessel lumen, positive staining of endothelial cells with the proliferation marker Ki-67 (arrows), as well as positivity of adventitial cells to macrophagespecific ED-1 antigen (arrowheads).
The hemodynamic consequences of 3-VO were tested by measuring the alterations of blood flow during ventilation with 6% CO2. Arterial PCO2 increased similarly in all four groups: by 21 ± 7 mm Hg in the control animals and by 21 ± 9 mm Hg, 24 ± 4 mm Hg and 20.5 ± 6 mm Hg in the animals examined immediately or at 1 and 3 weeks after 3-VO, respectively (NS).
In the nonischemic controls, LDF increased during CO2 ventilation by 1.48 ± 0.3%/mm Hg PaCO2 in the left and by 1.1 ± 0.2%/mm Hg in the right hemisphere (no significant difference). Immediately after 3-VO, the cerebrovascular response was reduced to 0.07 ± 0.16%/mm Hg in the ipsilateral (left) and to 0.17 ± 0.3%/mm Hg in the contralateral (right) hemisphere (Fig. 8). After 1-week survival, this reduction did not improve (ipsilateral 0.06 ± 0.35%/mm Hg, contralateral 0.18 ± 0.6%/mm Hg), but after 3 weeks, CO2 reactivity returned to 0.48 ± 0.08%/mm Hg in the ipsilateral and to 0.3 ± 0.39%/mm Hg in the contralateral hemisphere. On the ipsilateral side, this value amounts to 32% of the control response and is significantly different from CO2 reactivity immediately after vascular occlusion (P < 0.05). Arteriogenesis thus markedly improves the hemodynamic capacity of the hypoperfused brain.
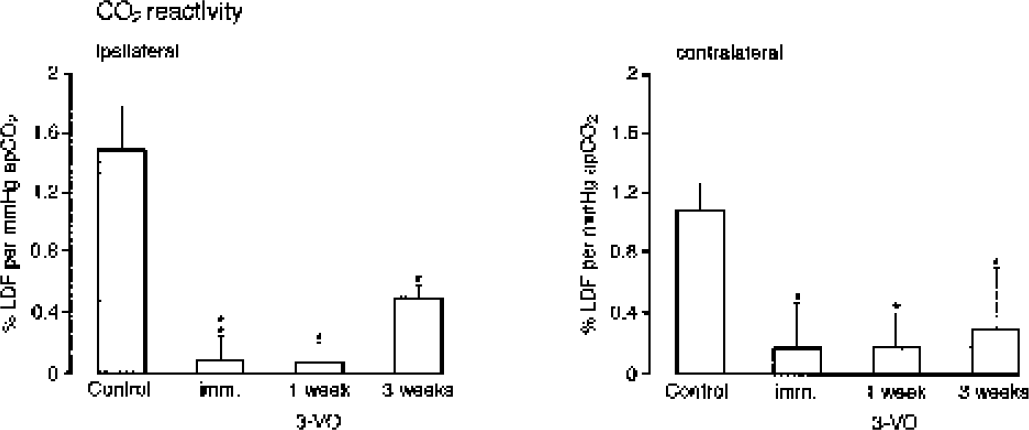
Carbon dioxide reactivity of cerebral blood flow in normal rat (control) and after different times (immediately, 1 and 3 weeks) after 3-vessel occlusion (3-VO). Blood flow was measured during ventilation with 6% CO2 by laser Doppler flowmetry (LDF) in the parietal cortex of the ipsilateral and contralateral hemispheres. CO2 reactivity is expressed as percent change of LDF per millimeter mercury increase of arterial PCO2. Note close to complete suppression of CO2 reactivity during the first week of 3-VO, followed by partial restitution after 3 weeks. *P < 0.05, as compared with control.
DISCUSSION
Most body organs, including the brain, exhibit powerful collateral systems that are able to compensate disturbances of blood supply by redistribution of flow from unobstructed vessels. In the brain, the most efficient collateral system is the circle of Willis, which provides a low-resistance connection between the four main supplying arteries, that is, the two carotid and the two vertebral arteries (Hossmann, 1993). After acute occlusion of one common carotid artery, cerebral blood flow returns to normal in most rat strains within a few minutes (Coyle and Panzenbeck, 1990; De Ley et al., 1985). Bilateral vertebral artery occlusion is also tolerated without lasting effects (Pulsinelli and Brierley, 1979), but occlusion of both common carotid arteries exceeds the collateral capacity of the circle of Willis and results in the decline of cerebral blood flow (Mitsufuji et al., 1996). This is confirmed by the present investigation, where bilateral common carotid artery occlusion reduced cerebral blood flow to about 30% of control. By combining bilateral vertebral with unilateral carotid artery occlusion (3-VO), the collateral system comes close to its limits. In an earlier study in Wistar rats, resting cerebral blood flow was not impaired but the hemodynamic reserve tested by acetazolamide application was distinctly reduced (Kawata et al., 1996). In the present study in which Sprague-Dawley rats were used, blood flow declined to about 50%, and the hemodynamic reserve was completely abolished, indicating that in this strain the collateral system is less efficient. However, even under this more critical condition, ischemia did not produce morphologic lesions, indicating that oxygen supply still remained above the threshold of structural integrity.
With ongoing ischemia time, the hemodynamic response in the 3-VO model gradually improved. As shown by latex angiography, the hemodynamic improvement was probably caused by the enlargement of the anterior and posterior cerebral arteries ipsilateral to the occluded carotid artery, pointing to arteriogenesis as the underlying mechanism.
In the postnatal organism, two types of vessel growth exist: angiogenesis, which involves the proliferation of endothelial cells and thus a sprouting of capillaries, and arteriogenesis, defined as the development of collateral arteries from preexisting arteriolar pathways, requiring the proliferation of endothelial and smooth muscle cells (Arras et al., 1998; Carmeliet and Collen, 1997; Ito et al., 1997a, b ; Risau, 1997; Schaper and Buschmann, 1999; Zhang et al., 2002). Whereas angiogenesis is observed mainly within the ischemic territory, arteriogenesis is usually temporally and spatially dissociated from this region (Buschmann and Schaper, 1999; Carmeliet et al., 1998). Collateral arteries grow in oxygen-rich tissue and, in contrast to angiogenesis, are able to supply blood from outside the risk region to prevent ischemic injury (Stepp et al., 2001). In fact VEGF, which is upregulated under conditions of ischemia, plays an important role in the scenario of angiogenesis by inducing capillary sprouting but is not a key player in the remodeling of larger collateral pathways associated with arteriogenesis (Carmeliet and Collen, 1997; Deindl et al., 2001). This may be because VEGF possesses unique target cell specificity for vascular endothelial cells, whereas collateral growth involves mitosis of both endothelial and vascular smooth muscle cells.
In peripheral vascular disease the spatial dissociation between angiogenesis and arteriogenesis is obvious: collateral growth may be found in the thigh, whereas clinical signs of ischemia are located in the foot. In the brain, such dissociations have not been explicitly described but may be concluded from published data. During focal brain ischemia, the angiogenic factor VEGF is upregulated within the ischemic territory (Lennmyr et al., 1998; Marti et al., 2000; Plate et al., 1999), whereas proliferation of collateral pathways occurs outside the ischemic region in the posterior part of the circle of Willis (Coyle and Heistad, 1991; Coyle and Panzenbeck, 1990; Hendrikse et al., 2001; Hillen et al., 1991).
Both arteriogenesis and angiogenesis contribute to the improvement of blood flow in the ischemic territory and, therefore, are of considerable therapeutic interest. However, to differentiate between the two mechanisms, experimental models are required in which these mechanisms can be studied separately. To the best of our knowledge, our 3-VO model presents for the first time brain arteriogenesis in the absence of ischemic tissue injury and in the absence of VEGF upregulation. The growth of the anterior and posterior cerebral arteries is therefore not mediated by signals from the ischemically damaged tissue but by the induction of pressure gradients across the circle of Willis with consecutive changes of blood flow across the preexisting collateral pathways.
The morphologic examination of the enlarged PCA revealed typical signs of adaptive arteriogenesis: activation of the endothelium, an enhanced proliferative index of Ki-67–positive endothelial cells, invasion of circulating monocytes, and an increased number of macrophages accumulating in the adventitia (Arras et al., 1998; Hoefer et al., 2001; Scholz et al., 2000). As a result of this controlled remodeling process, the vascular diameter of the PCA increased during the 3-week observation period by 72% and that of the anterior cerebral arteries by 28%. This substantial growth was reflected by the gradual improvement of the hemodynamic reserve, as assessed by the ventilation of animals with 6% CO2. The hemodynamic reserve describes the amount of flow impairment brain vessels may compensate by reducing the vascular resistance. At decreasing brain perfusion pressure—induced by constriction or occlusion of the main supplying arteries—brain arterioles dilate to maintain blood flow at a constant level (cerebrovascular autoregulation). A reduced hemodynamic reserve is equivalent to an increased risk of having ischemic injury under such conditions (Clifton et al., 1988; Kleiser and Widder, 1992; Miller et al., 1992). In fact, this has been confirmed by measurements of hemodynamic reserve for predicting the clinical outcome of patients with internal carotid artery occlusions (Markus and Cullinane, 2001; White and Markus, 1997; Widder et al., 1994). The gradual improvement of the hemodynamic reserve after 3-VO is, therefore, an impressive documentation of the functional importance of arteriogenesis and corroborates magnetic resonance angiographic observations, which show that spontaneous improvements of collateral flow reduce the prevalence of brain infarction (Hendrikse et al., 2001; Kluytmans et al., 1999).
A limiting factor of arteriogenesis as a self-adaptive response to impeding brain ischemia is the slowness of the process. At 3 weeks after 3-VO, CO2 reactivity was still markedly suppressed. This is in line with previous studies that revealed enlargement of the posterior cerebral artery only at 6 weeks after unilateral common carotid artery occlusion (Coyle and Heistad, 1991; Coyle and Panzenbeck, 1990). Obviously, such a late response is not able to prevent brain infarction during rapidly progressing cerebrovascular occlusion, but there are ways to accelerate this response. One of the first steps required for induction of arteriogenesis is the recruitment of circulating monocytes to the site of collateral growth (Arras et al., 1998; Ito et al., 1997a, b ), a process that can be stimulated by granulocyte–monocyte colony-stimulating factor (Buschmann et al., 2001; Seiler et al., 2001; van Royen et al., 2001a, b ). In another investigation, we provide evidence that systemic application of this factor accelerates collateral growth to such a degree that already 1 week after 3-VO, the hemodynamic reserve is fully restored (Buschmann et al., 2003). Therapeutically accelerated arteriogenesis may, therefore, be of interest to prevent infarction under conditions of uncompensated cerebrovascular disease.
Footnotes
Acknowledgments:
The authors thank Mrs. D. Schmiegelt and Mr. P. Janus for expert technical assistance, Mr. B. Huth and Mrs. I. Mühlhöver for preparing the figures, and Mrs. A. Lorig for copyediting the manuscript.
