Abstract
Recent blood oxygenation level–dependent (BOLD) functional magnetic resonance imaging studies have shown a reduction of cerebral activation during aging, which may be associated with age-related changes of the cerebral vascular system. The authors used a global hypercapnic breath-holding challenge to define nonneuronal contributions to a significantly reduced activation in the primary sensorimotor cortex during finger tapping in a group of old (n = 6; mean age 65 years) compared with a group of young (n = 6; mean age 27 years) subjects. Within significantly activated voxels in both groups during finger tapping, the mean BOLD signal amplitudes were significantly smaller in the group of older subjects for both tasks. In those voxels showing significant activation only in young subjects during finger tapping, the response to hypercapnia was also greatly diminished in older subjects. The attenuated hypercapnic BOLD signal response in older subjects within this region suggests that age-dependent changes of the cerebral vasculature may alter the neuronal–vascular coupling. In older subjects, cerebral vessels may not react as effectively in response to a vasodilating stimulus, which will lead to differences in the number of voxels that pass a criterion threshold despite similar neuronal activation.
With respect to most neurodegenerative and vascular diseases occurring in the last decades of life and against the background of an increasing population mean age in industrial countries, the scientific and socioeconomic interest in the physiology and pathophysiology of aging has grown substantially. In the past few years, evidence has accumulated that normal aging is associated with structural and functional changes in different neuronal systems. Several positron emission tomography (PET) studies have shown age-associated declines of cerebral blood flow (CBF) and cerebral blood volume (CBV) (Frackowiak et al., 1980; Leenders et al., 1990; Martin et al., 1991; Melamed et al., 1980; Pantano et al., 1984), as well as a decrease of the cerebral metabolic rate of glucose (Kuhl et al., 1982) and cerebral metabolic rate of oxygen (CMRO2)(Marchal et al., 1992; Yamaguchi et al., 1986). Various functional imaging methods have identified different cortical neuronal activation patterns during sensory (D'Esposito et al., 1999; Hesselmann et al., 2001; Huettel et al., 2001; Mattay et al., 2002; Mehagnoul-Schipper et al., 2002; Taoka et al., 1998) or cognitive stimuli (for a review see Grady and Craik, 2000) in young and elderly subjects. Since it seems plausible that the aging brain might need to mobilize more resources to produce the same output as a younger one, the recruitment of new brain areas during sensorimotor activation or cognitive perturbations in older subjects has been attributed to compensatory mechanisms for age-related cell changes in some instances (Grady and Craik, 2000). In contrast, a decreased activation has been associated with a neuronal loss in other cases (Grady and Craik, 2000).
Principally, these studies have used functional magnetic resonance imaging (fMRI) techniques to clarify the impact of aging on regional brain activation patterns (D'Esposito et al., 1999; Hesselmann et al., 2001; Huettel et al., 2001; Mattay et al., 2002; Mehagnoul-Schipper et al., 2002; Ross et al., 1997; Taoka et al., 1998). In this context, the utility of fMRI is directly related to the degree to which the relation between MR signal changes and underlying neuronal activation can be established. With respect, however, to blood oxygenation level– dependent (BOLD) fMRI, which is based on neuronal– vascular coupling, differences in the regional microvasculature anatomy and resting CBV values may interfere with the accurate assessment of the timing and location of neural activation.
Recently, several fMRI studies have been performed to assess the temporal and spatial characteristics of the fMRI hemodynamic response (HDR) in healthy young and old subjects during sensorimotor (D'Esposito et al., 1999; Hesselmann et al., 2001; Taoka et al., 1998) or visual activation (Huettel et al., 2001; Ross et al., 1997). Whereas HDR waveforms were similar between young and elderly subjects for the visual and sensorimotor cortices in these studies, the spatial extent of activation at a given threshold was significantly reduced in the older subjects. Assuming a comparable neuronal activity during sensorimotor or visual stimulation in young and old subjects, the visualization of “true” areas of neuronal activation with BOLD fMRI in normal aging might be limited by other nonneuronal mechanisms. In fact, a number of specific age-related changes of the cerebral vasculature, such as changes of the microcirculation (Hajdu et al 1990; Mann et al 1986), as well as an age-related decline in cerebrovascular reactivity to carbon dioxide or acetazolamide have been observed (Kastrup et al., 1998a; Reich and Rusinek 1989; Yamamoto et al., 1980). These factors could be responsible for alterations of the neuronal–vascular coupling, so that younger and older subjects differ in the number of voxels that pass a criterion threshold, despite similar neuronal activation.
In a recent study, we used a breath-holding maneuver to assess the regional variability of the BOLD response to a global, vascular challenge (Kastrup et al., 1999). In good agreement with biophysical models describing determinants of BOLD signal contrast (Bandettini and Wong, 1997; Boxerman et al., 1995a, b ; Ogawa et al., 1993), those regions that possess high capillary densities also exhibit high BOLD signal changes. Under the assumption that hypercapnia and neuronal activation cause similar hemodynamic events, a voxelwise comparison between activation and hypercapnic-induced signal changes may offer a promising approach defining nonneuronal contributions to altered activation patterns during aging. Therefore, the present study was designed to detect age-related vascular differences that contribute to altered BOLD fMRI signal responsiveness during an equivalent neuronal input.
MATERIALS AND METHODS
Fifteen healthy, native-German participants were divided into groups of young (3 men and 4 women, aged 22 to 31 years; mean age = 27 years) and old (4 men and 4 women, aged 54 to 82 years; mean age = 65 years) subjects. The younger volunteers consisted of neurology department staff members and medical students, whereas the older volunteers were recruited from the community. The older subjects were carefully selected and each underwent a detailed interview, as well as a general physical and neurologic examination. We excluded subjects with any history of neurologic, psychiatric, cardiac, or pulmonary diseases, cardiovascular risk factors such as hypertension, diabetes mellitus, hyperlipidemia, or smoking. Additionally, subjects were excluded if they were taking any centrally active drugs. All volunteers had to be able to hold their breath for at least 30 seconds to perform the breath-holding task. All subjects were asked to refrain from caffeine at least 6 h before their fMRI study.
Informed consent was obtained from all subjects in accordance with the Institutional Review Board of the University of Tübingen. All participants were right-handed as determined by means of a standardized inventory with a lateralization index > 70% as an inclusion criterion.
All images were obtained using a 1.5-T whole-body scanner (Siemens Vision, Siemens, Germany). A standard radio-frequency (RF) head coil was used and the heads of all subjects were secured by means of foam rubber, to minimize movement artifacts. For functional imaging, volume data consisting of 28 parallel axial slices (thickness = 4 mm, gap = 1 mm) were obtained across the complete brain using an echo planar imaging sequence (64 × 64 matrix, 192-mm field of view, TE = 39 milliseconds, TR = 3 seconds, α = 90°)(Klose et al., 1999). In all subjects, two scanning sessions for finger tapping and another two scanning sessions for breath holding were performed, each including five initial dummy scans for equilibration of T1 saturation effects. The order of the four blocks was counterbalanced across subjects. High-resolution images obtained with a T1-weighted three-dimensional turbo-flash sequence (MPRAGE; 128 sagittal slices, thickness = 1.5 mm, 256 × 256 matrix, 256-mm field of view, TE = 4 milliseconds, TR = 9.7 milliseconds) served as the anatomic reference for functional images.
As a hypercapnic challenge, all subjects performed alternating periods of breath holding and self-paced breathing at 30-second intervals after normal expiration (Kastrup et al., 1998b). The time to stop and start breathing was indicated by acoustic stimuli.
For motor activation, all subjects were instructed to place their right index finger on a response button designed for application within the MR scanner. Pacing signals were generated by manual editing (Cool edit 2000; Syntrillium Software Corporation, Phoenix, Arizona, U.S.A.) of a recorded “natural” sound (stroke of a pen against the desk). To improve audibility within the MR scanner environment, a double-spike (spaced 2 milliseconds apart) was superimposed on the initial “excitation” phase (about 3 milliseconds) of broadband noise, followed by a dampened signal (duration about 10 milliseconds, spectral energy distribution centered at 3 kHz). The auditory stimuli were well discernible at a comfortable loudness level against the background of scanner noise.
Using the same experimental setup, we had recently found an optimal performance of finger tapping at a frequency of 3 Hz in young volunteers (unpublished data, Riecker, 2002), so we decided to use this frequency in the present study. In line with our previous data, Blinkenberg et al. (1996) also showed an optimal performance for finger tapping in the frequency range of 2 to 4 Hz. To prevent anticipation of the frequency or the timing of the acoustic stimuli, a total of six different frequencies (2 to 6 Hz) were presented in a randomized fashion and jittered by interspersing gaps, so that the onset-to-onset intervals amounted to 12 to 24 seconds.
Subjects pressed the response button according to the acoustical trigger stimuli, which were applied via headphones simultaneously to both ears. During execution of the motor task, subjects did not report signs of physical exhaustion at any frequency. Subjects were briefly instructed and were allowed to practice the motor activation task several times before scanning. Performance was recorded by a standard personal computer so that each single signal from the response button could be verified. According to visual control, movements of the nondominant hand did not take place.
To delineate eventual contributions of sensory processing to the hemodynamic responses of this finger-tapping experiment, additional fMRI measurements during passive listening to the same acoustic stimuli without any motor response had been performed previously (Ackermann et al., 2001). Altogether, the experiment comprised 90 click trains (6 rates × 15 trials).
The fMRI data were transformed to an ANALYZE-compatible format. Signal analysis and statistics were performed with the SPM99 package (Wellcome Institute of Cognitive Neuroscience, London, U.K.) (Friston et al., 1995). After defining the anterior and posterior commissural line, all functional images were realigned using the first remaining scan of each session as a reference. During this spatial preprocessing procedure, the time series data were sync interpolated.
Data sets with more than 2-mm motion in any direction were excluded from further analysis. Despite all our efforts to prevent head movement by securing all volunteers with pillows and foam padding in the head coil, two old and one young subject had to be excluded from further analysis owing to head movement, leaving six subjects in each group. The averaged fMRI scans obtained from each subject were coregistered with the corresponding anatomic data. As a prerequisite to normalization to a standard stereotactic space, the anatomic data sets were coregistered with the T1 template and the calculated transformation matrix was applied to the functional images. Before statistical analysis, a gaussian filter with 10-mm full-width half-maximum was applied to the functional images. The time series were bandpass filtered, removing information at <0.1 Hz and >0.4 Hz.
In a first step, single-subject contrast images were created for both finger tapping and breath holding by modeling the different tasks as stimulus functions applying the general linear model. In a second-level (random effects) analysis, the individual contrast images were used to create group maps for direct comparison and subtraction of young and old activation maps. Besides comparing global activation we performed an additional region of interest (ROI) analysis by identifying the central sulcus region on the standard brain template of the Montreal Neurological institute (provided by SPM99) and defining an ROI encompassing the primary sensorimotor region (Fig. 1). This ROI was used in all subjects and contained a total of 2,733 voxels. All subjects showed significant activation during both tasks in this region.
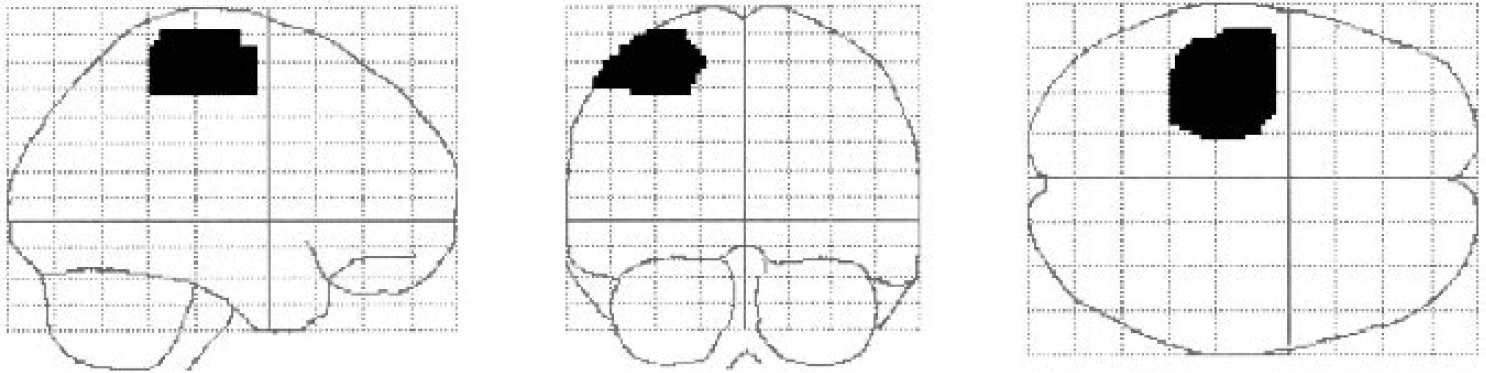
Region of interest of the primary sensorimotor cortex (total of 2,733 voxels) used for further analysis.
The average individual BOLD signal responses during both tasks within the entire ROI, as well as within those regions demonstrating age-related differences of activation, were determined. In addition, we also selected the single voxel within this ROI with the largest t-value for each group during finger tapping for further analysis. The individual HDR waveforms during finger tapping and breath holding were obtained for this single voxel. For the ROI, estimates of the noise variance and signal variance were used to calculate the signal-to noise ratios of suprathreshold voxels.
All steps of statistical analysis were set at a threshold of T>3.10 (P < 0.001) at voxel level. To correct for multiple comparisons across volume, an extension threshold was introduced at cluster level (P < 0.05 corrected, k>59). For anatomic localization of significantly activated areas, the fMRI maps were superimposed on transverse sections of the structural MR images averaged across all subjects.
We performed a one-way analysis of variance to look for significant differences across the two groups on task accuracy measured as the number of button presses relative to the number of acoustic stimuli presented. Additionally, a comparison of the within-group variability of the mean BOLD signal amplitudes between young and old subjects was performed with an F test and subsequently a one-way analysis of variance to look for significant differences across the two groups, respectively.
RESULTS
The analysis of the motor task accuracy rates for button presses demonstrated no significant difference (P = 0.8) between both groups. All subjects were able to hold their breath as prescribed by the study protocol. Consistent with previous fMRI and PET studies (Calautti et al., 2001; Mattay et al., 2002; Sabatini et al., 1993; Sadato et al., 1996) a typical pattern of activation during the motor task was observed, which mainly included the contralateral primary sensorimotor and parietal cortex, ipsilateral cerebellum, and supplementary motor areas. For the entire brain, the spatial extent of activation at identical thresholds was significantly larger in young than in old subjects (6,498 vs. 3,164 voxels, respectively). Similarly, younger subjects had significantly more activated voxels in the ROI of the primary contralateral sensorimotor cortex (567 vs. 286 voxels, respectively). The spatial pattern of activation with emphasis on the primary sensorimotor cortex for the group maps of young and old subjects during 3-Hz finger tapping is shown in Fig. 2. Table 1 summarizes the number of activated voxels and corresponding t-values of the left supplementary motor region, left primary sensorimotor cortex, and right cerebellum during finger tapping and hypercapnia in young and old subjects. Notably, the t-value of the highest activated voxel within the primary sensorimotor cortex was 35.40 in the group of young subjects compared with 22.38 in the group of old subjects.
Number of activated voxels and corresponding t-values of the left supplementary motor region, left primary sensorimotor cortex and cerebellum during finger tapping and hypercapnia in young and old subjects
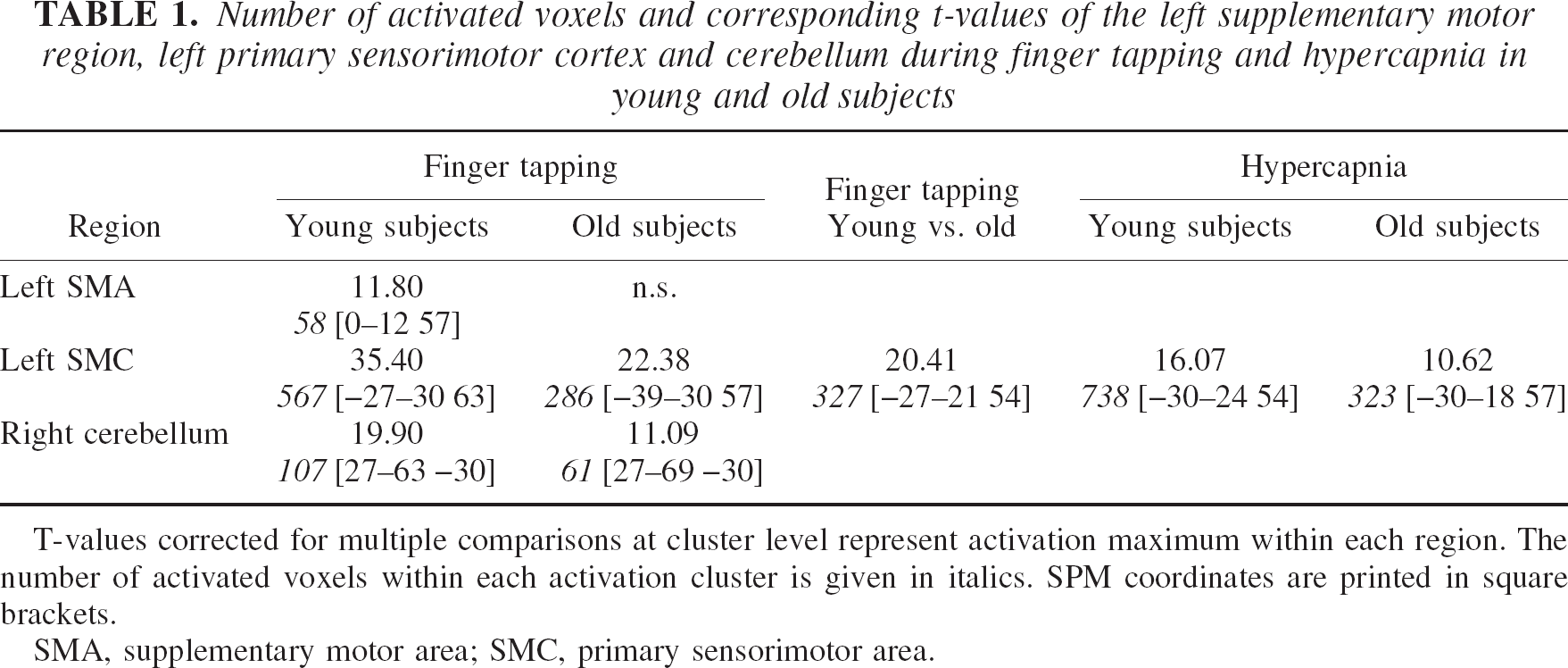
T-values corrected for multiple comparisons at cluster level represent activation maximum within each region. The number of activated voxels within each activation cluster is given in italics. SPM coordinates are printed in square brackets.
SMA, supplementary motor area; SMC, primary sensorimotor area.
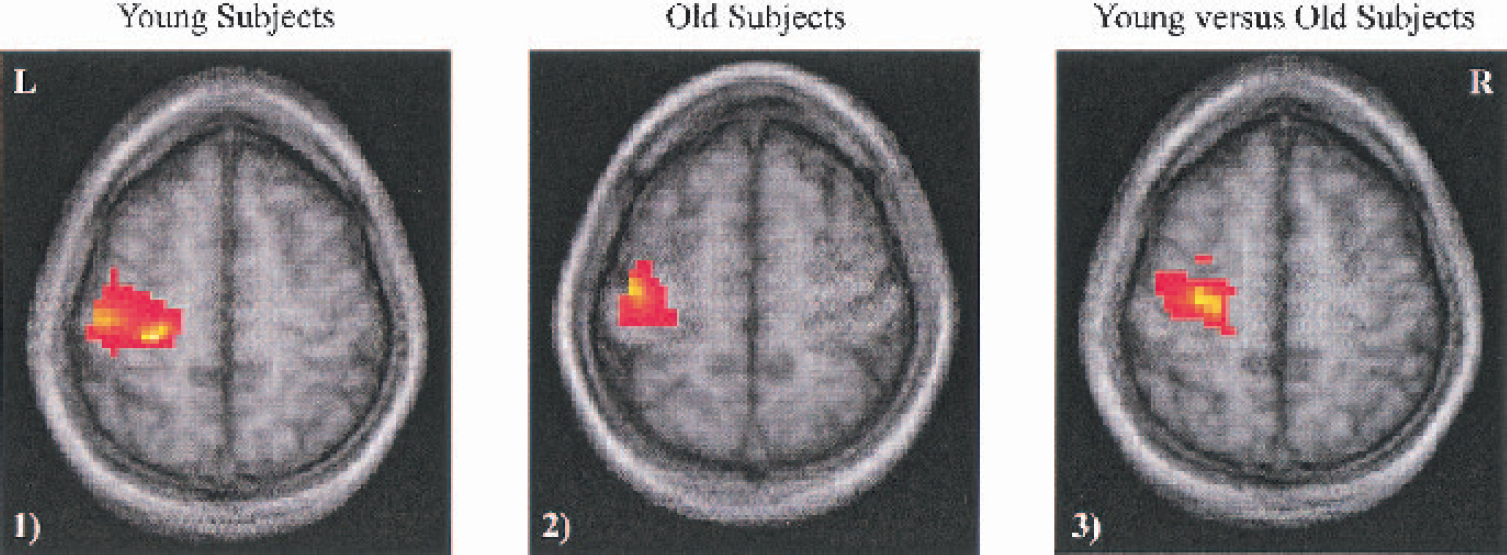
A representative slice of the primary sensorimotor cortex from the group maps showing significant areas of activation during a finger-tapping task with the dominant hand in (1) young and (2) old subjects, as well as significant (P < 0.05 with correction for multiple comparisons) regions of greater activation in the younger subjects (3). In this comparison, none of the activation differences was greater in older subjects. See Table 1 for exact coordinates of significantly activated voxels.
Repeated challenges of breath holding induced a global rise in BOLD signal intensities, which was more pronounced in young compared with old subjects globally (40,493 voxels in young subjects vs. 12,044 voxels in old subjects), as well as in the ROI of the primary sensorimotor cortex (738 voxels in young subjects vs. 323 voxels in old subjects).
The individual HDR waveforms during finger tapping and breath holding for the single voxel with the largest t-value for each group within the ROI of the primary sensorimotor cortex are displayed in Fig. 3. With respect to the amplitudes of the HDR, a comparison of the within-group variability between young and old subjects showed no significant difference (P = 0.5 for finger tapping and P = 0.3 for breath holding). A subsequent one-way analysis of variance showed significantly smaller HDR amplitudes in the group of old subjects compared with the group of young subjects during finger tapping (4.8 ± 0.9 vs. 7.2 ± 0.9, respectively; F = 20.3, P < 0.01), as well as during hypercapnia (8.1 ± 0.9 vs. 12.7 ± 0.7, respectively; F = 93.2, P < 0.01).
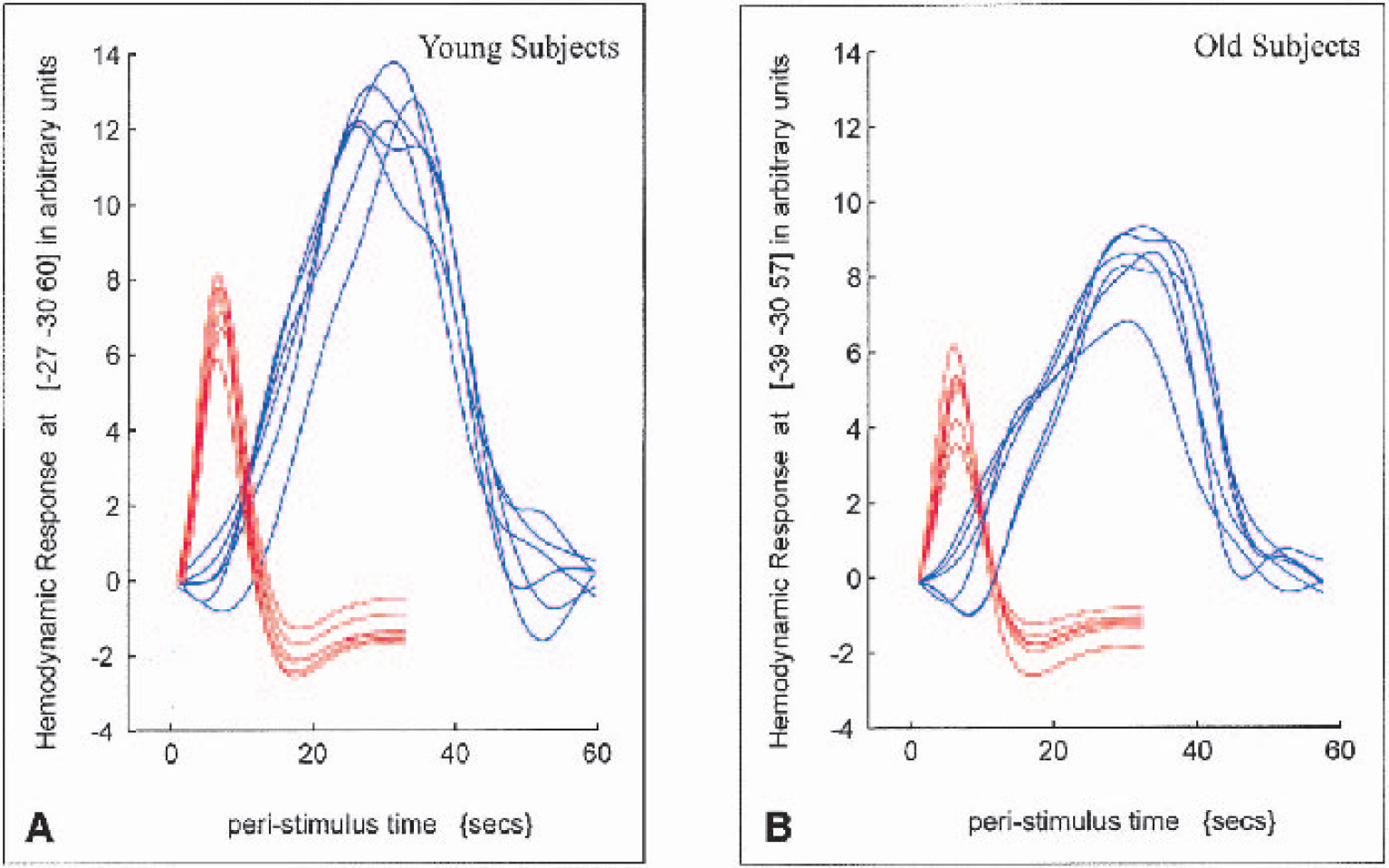
Individual, trial-averaged hemodynamic response waveforms (arbitrary units) from young
The mean BOLD signal amplitudes of significantly activated voxels in both groups within the ROI of the primary sensorimotor cortex during finger tapping and hypercapnia are summarized in Fig. 4. Similar to the HDR waveform analysis at single-voxel level, a one-way analysis of variance showed significantly smaller mean BOLD signal amplitudes in the group of old subjects for both finger tapping (3.7 ± 0.5 vs. 7.8 ± 0.8 in young subjects; F = 125, P < 0.01) and hypercapnia (4.2 ± 0.7 vs. 11.2 ± 1.0 in young subjects; F = 180, P < 0.01). A comparison of the mean BOLD signal amplitudes during finger tapping and hypercapnia showed an attenuation of the BOLD signal changes, indirectly indicating an increase in oxidative metabolism during finger tapping (Kastrup et al., 2002).
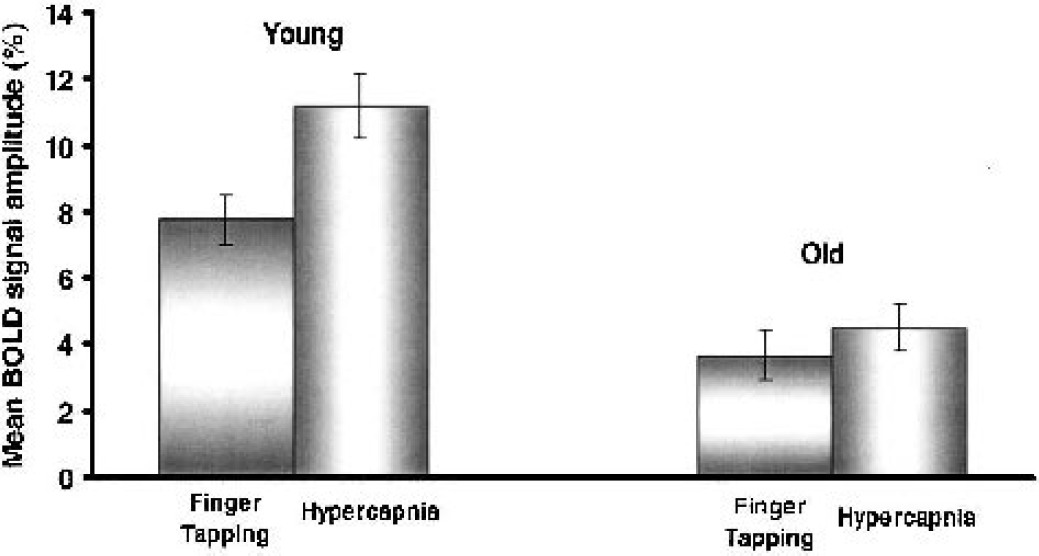
Mean blood oxygenation level–dependent (BOLD) signal amplitudes of significantly activated voxels in both groups within the region of interest of the primary sensorimotor cortex during finger tapping and hypercapnia for young and old subjects. A comparison of the mean BOLD signal amplitudes during finger tapping and hypercapnia shows an attenuation of the BOLD signal changes, indirectly indicating an increase in oxidative metabolism during finger tapping. A one-way analysis of variance revealed significantly smaller (P < 0.01) mean BOLD signal amplitudes in the group of older subjects for both finger tapping (3.7 ± 0.5 vs. 7.8 ± 0.8 in young subjects) and hypercapnia (4.2 ± 0.7 vs. 11.2 ± 1.0 in young subjects).
The signal-to-noise ratios within the ROI of the primary sensorimotor cortex were significantly greater in the young subjects during both finger tapping (0.91 vs. 0.51 in old subjects; F = 9.1, P < 0.05) and hypercapnia (0.94 vs. 0.55 in the old subjects; F = 6.1, P < 0.05). Since the noise magnitudes were not significantly different in both groups (finger tapping: F = 0.85, P = 0.4; hypercapnia: F = 0.27, P = 0.6) the increased signal-to-noise ratios during both tasks were mainly attributable to the greater signal amplitudes in the group of young subjects compared with the older group.
The mean BOLD signal amplitudes of both groups during hypercapnia of those regions showing significant activation in young but not in old subjects during finger tapping are shown in Fig. 5. There was a significantly attenuated mean BOLD signal response in old subjects within this region during hypercapnia.
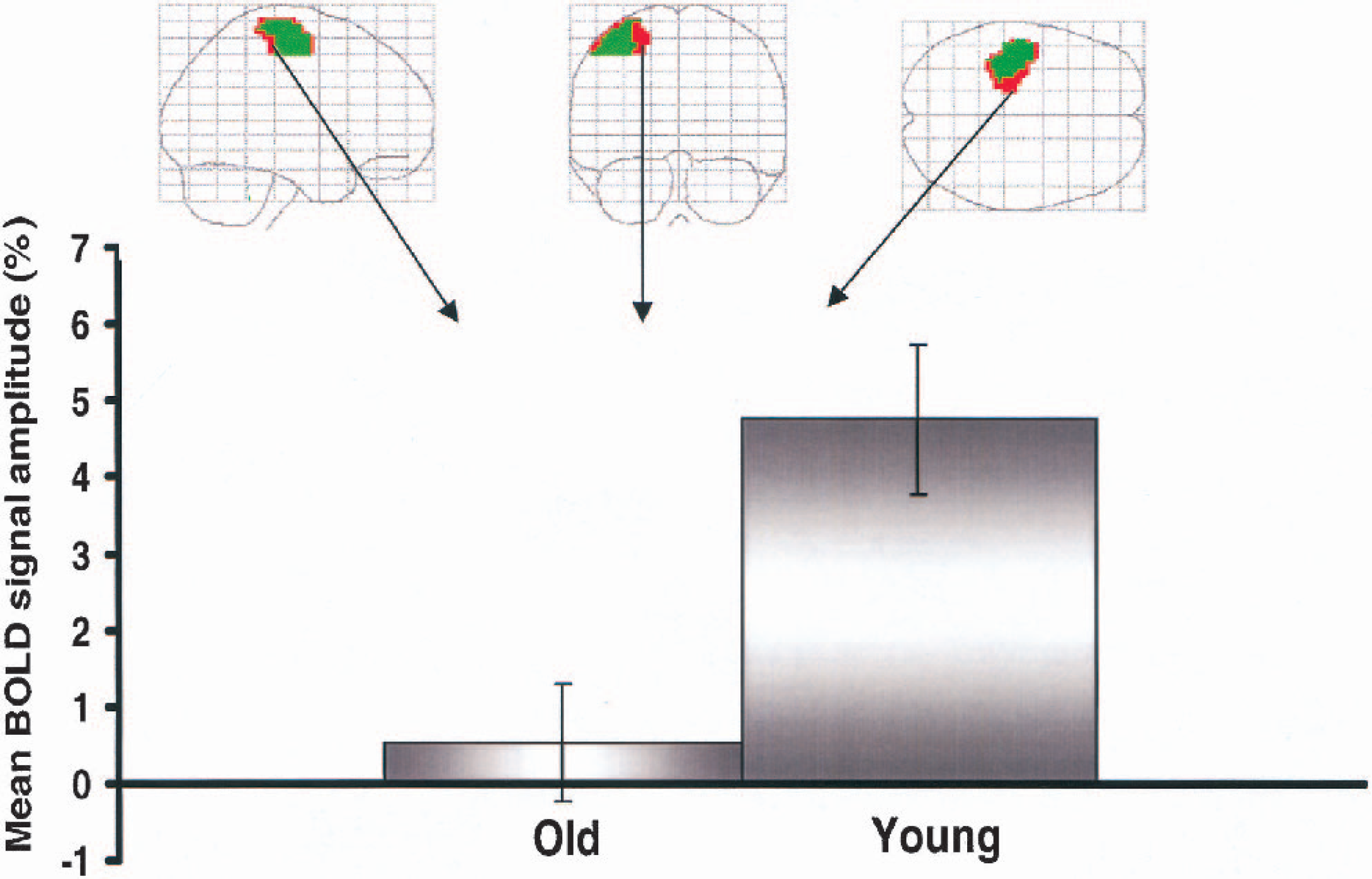
Mean blood oxygenation level–dependent (BOLD) signal amplitudes of both groups during hypercapnia of those regions showing significant activation in young but not in old subjects during finger tapping (red). Within this region there is clear attenuation of the mean BOLD signal response in the older subjects.
DISCUSSION
In this study we used a global hypercapnic challenge to define nonneuronal contributions to altered age-related activation patterns during focal brain stimulation. In view of the fact that there is no neuropathologic evidence of a significant neuronal loss in the primary sensorimotor cortex during aging (Long et al., 1999), as well as a similar neuronal activation during a simple finger-tapping task within this region (D'Esposito et al., 1999; Sailer et al., 2000), this cortical area seemed well suited to advance our knowledge of the systematic influences of normal aging on focal brain activation and cerebral hemodynamics. Since older people require progressively longer reaction times as movement complexity increases (Light and Spirduso 1990; Smith et al., 1999) and error rates increase with higher stimulation frequencies (Blinkenberg et al., 1996), we chose to study a simple, externally paced finger-tapping task. Despite a comparable performance during this task, the spatial extent of global and regional activation of the primary sensorimotor cortex was significantly smaller in old compared with young subjects. Similar observations have been made in recent fMRI studies during sensorimotor (D'Esposito et al., 1999; Hesselmann et al., 2001; Mehagnoul-Schipper et al., 2002; Taoka et al., 1998) and visual stimulation (Huettel et al., 2001). In contrast to these studies, Mattay et al. (2002) recently found a greater extent of global activation during a 0.6-Hz finger-tapping task in subjects older than 50 years of age compared with a group of younger subjects (<35 years of age). Instead of performing a pure motor task, however, in that study the subjects were required to press a button with a single finger according to a visually presented complex scheme, and additional cognitive processes may have obscured these results. Nonetheless, although not a focus of the present study, the recruitment of additional cortical areas during a simple motor task has also been observed in previous functional imaging (Calautti et al., 2001) and electrophysiologic studies (Sailer et al., 2000) and has generally been interpreted as a compensatory mechanism of the aging brain.
A major finding of the present study is that the reduced spatial extent of activation in the primary sensorimotor cortex of older subjects during a simple finger-tapping task can at least partially be attributed to a diminished vascular reactivity to local brain activation in normal aging. The significantly reduced vascular response to hypercapnia in the older subjects in those regions showing significant activation during finger tapping in younger subjects indicates that age-dependent changes of the cerebral vasculature may alter the neuronal–vascular coupling, so that younger and older subjects differ in the number of voxels that pass a criterion threshold, despite similar neuronal activation.
It is a well-known fact that the wall of the cerebral arterioles undergoes atrophy during aging and there is a decrease in the distensibility of the cerebral vessels, which is mainly based on alterations of their composition with a disproportionate reduction of the distensible elements (Hajdu et al., 1990). Therefore, cerebral vessels in older subjects may not react as effectively in response to dilator stimuli such as hypercapnia or those released during neuronal activation. In support of this notion, a decreased global and regional cerebrovascular reactivity to hypercapnia in aging has been found in numerous studies (Kastrup et al., 1998a; Reich and Rusinek 1989; Yamamoto et al., 1980). Moreover, a decreased vasoreactivity of pial arterioles to the main mediators of neurovascular coupling during aging has been found (Jiang et al., 1992; Mayhan et al., 1990).
Although our major findings are in good agreement with recent biophysical models of the BOLD signal contrast describing a significant contribution of the microvascular system to the regional BOLD signal responsiveness (Bandettini and Wong 1997; Boxerman et al., 1995a, b ; Ogawa et al., 1993), a variety of other factors could have also contributed to the smaller spatial extent of activation in the older subjects. Since we excluded all subjects (two old and one young subject) with significant head motion, and in line with previous fMRI studies (D'Esposito et al., 1999; Huettel et al., 2001), it is unlikely that this factor greatly influenced the present findings. At a given criterion threshold the number of suprathreshold voxels is dependent on the signal-to-noise ratio, which has also been shown to be greater in younger than in older subjects during sensorimotor (D'Esposito et al., 1999; Hesselmann et al., 2001) or visual stimulation (Huettel et al., 2001; Ross et al., 1997). Principally, an increased signal-to-noise ratio in young compared with old subjects may be related to a high signal magnitude, a low noise magnitude, or a combination of both. Whereas previous studies have found significantly greater noise levels in older than in younger subjects (D'Esposito et al., 1999; Huettel et al., 2001), our data support the notion that the increased spatial extent of activation in young volunteers mainly reflects a higher signal magnitude during task activation in this group. Similarly, significant reductions of mean BOLD signal changes during aging have recently been described during finger tapping and visual stimulation (Hesselmann et al., 2001; Ross et al., 1997). Furthermore, using near-infrared spectroscopy, Hock et al. (1995) found a significantly attenuated increase in oxyhemoglobin in a group of old compared with a group of young subjects during a calculation task.
Our finding of significantly decreased BOLD signal amplitudes during finger tapping in a single maximally activated voxel, however, stands in contrast to a recent comprehensive study of D'Esposito et al. (1999). Using bilateral button-press events, these authors studied the HDR of single randomly selected suprathreshold voxels in the motor cortex in a group of young and old subjects. Whereas the number of suprathreshold voxels was also significantly higher in the group of young subjects in this study, the HDR amplitudes were not significantly different in both groups. Similarly, Huettel et al. (2001) recently showed comparable response amplitudes in single maximally activated voxels, as well as a ROI encompassing the primary visual cortex during a brief (500-millisecond) checkerboard stimulation in a group of young and old subjects. These conflicting results can be attributed at least in part to methodological differences, mainly the use of a short-lasting block paradigm of 6 seconds in our study. Irrespective of these discrepancies, the impact of an event-related versus a block design might have tremendous implications for the design of future studies in older subjects and should be clarified in further studies. Nonetheless, it should also be stressed that the finding of a significantly decreased spatial extent of BOLD activation in older subjects is a common feature of all of these studies.
Principally, it could be speculated that the initial hemodynamic response to a functional challenge is characterized by rather nonspecific elevations of the BOLD signal within the first seconds, which are not subject to age-related changes. In favor of this speculation, Janz et al. (2000) studied BOLD signal changes during various short-lasting visual stimulation paradigms. Although these paradigms were known to produce significantly different hemodynamic responses when applied in a block design, the BOLD signal changes during short stimulus application were nearly identical. In a further study by Fransson et al. (1999), subsecond visual stimulation led to a similar oxygen-sensitive MRI response as commonly observed for longer stimulation duration. While frequency-dependent changes in CBF during extended periods of finger tapping have been well documented (Sabatini et al., 1993; Sadato et al., 1996), Glover (1999) showed similar BOLD responses during brief periods of finger tapping with frequencies varying from 1 to 4 Hz. Therefore, all of these studies give rise to the suspicion that the initial hemodynamic response to a functional challenge is dominated by very rapid (within seconds, possibly within hundreds of milliseconds) and nonspecific elevations of CBF within the first seconds.
It should be emphasized that our results and conclusions are drawn from a small sample as well as a very simplistic model based on several assumptions. As discussed previously, we believe that our simple motor task elicited a comparable neuronal response in young and old volunteers. This assumption, however, may not apply to other activation tasks, so that our results cannot be generalized to other cortical areas without restriction. Without simultaneous electrophysiologic monitoring of neuronal activity, age-related reductions of BOLD signal changes could also reflect alterations of the cerebral capillary morphology that have occurred secondary to a “true” loss of nerve cells.
Although there is no neuropathologic evidence of a significant neuronal loss in the primary sensorimotor cortex during aging (Long et al., 1999), cortical atrophy is an important possible confound in BOLD fMRI studies dealing with elderly persons. Although we did not perform a quantitative structural volumetry to assess the effects of age-related cortical atrophy on the HDR, Johnson et al. (2000) found no correlation between the cortical atrophy and BOLD fMRI signal changes in activated language regions during a semantic task in a recent comprehensive study.
The mechanisms responsible for the coupling of CBF to hypercapnia or focal brain activation are incompletely understood. Based on the criteria of vasoactivity on cerebral vessels and accumulation in the extracellular space during neuronal activity, several putative chemical mediators have been postulated, namely H+ ions, K+, and adenosine (Betz, 1972; Kuschinsky and Wahl, 1978; Villringer and Dirnagl, 1995). H+ ions are derived from lactate and CO2 as intermediate and end products of local energy metabolism, and similar mediators of hypercapnia and neuronally induced vasodilation have been proposed. Nevertheless, age-related changes of these mediators may be different for hypercapnia or neuronal activity.
Similar to other research reports that used global, hypercapnic challenges to estimate changes in CMRO2 during neuronal activation with fMRI (Davis et al., 1998; Kastrup et al., 2002), our results are based on the assumption that hypercapnia and neuronal activation cause comparable hemodynamic events. In good agreement with this assumption, the mean BOLD signal amplitudes of activated voxels within the sensorimotor cortex during finger tapping (r = 0.55) correlated with the mean BOLD signal amplitudes during hypercapnia in this study. Proportional BOLD signal changes during hypercapnia and focal neuronal activation have also been observed in other recent studies (Bandettini and Wong, 1997; Davis et al., 1998).
Since the BOLD contrast depends on multiple physiologic parameters such as CBV, CBF, and CMRO2 (Bandettini and Wong, 1997; Boxerman et al., 1995a, b ; Ogawa et al., 1993), it is difficult to extract a single parameter from the observed signal changes. In this context, the well-known age-associated declines of CBF and CBV (Frackowiak et al., 1980; Leenders et al., 1990; Martin et al., 1991; Melamed et al., 1980; Pantano et al., 1984), as well as cerebral metabolic rate of glucose (Kuhl et al., 1982) and CMRO2 (Marchal et al., 1992; Yamaguchi et al., 1986), could differentially influence the extent of BOLD activation.
Despite these shortcomings, our findings have important implications for future BOLD fMRI studies. To correctly define “true” areas of neuronal activation with BOLD fMRI, alterations of the neurovascular coupling have to be taken into account. Irrespective of their cause, the altered BOLD activation patterns during aging underscore the importance of using age-matched controls. While this study comprised healthy volunteers, it should be understood that the presence of major risk factors for arteriosclerosis such as hypertension or diabetes could alter the microvascular reactivity in addition to age-associated changes (Pineiro et al., 2002). In this context, a hypercapnic challenge might become useful to correctly interpret the spatial extent of activation with BOLD fMRI.
Footnotes
Acknowledgments:
The authors are grateful to Dr. Frank Rodden for his excellent technical support, and they also thank the volunteers who participated in the study.
