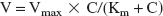Abstract
The mechanism that removes homovanillic acid (HVA), an end metabolite of dopamine, from the brain is still poorly understood. The purpose of this study is to identify and characterize the brain-to-blood HVA efflux transporter at the rat blood–brain barrier (BBB). Using the Brain Efflux Index method, the apparent in vivo efflux rate constant of [3H]HVA from the brain, keff, was determined to be 1.69 × 10–2 minute–1. This elimination was significantly inhibited by para-aminohippuric acid (PAH), benzylpenicillin, indoxyl sulfate, and cimetidine, suggesting the involvement of rat organic anion transporter 3 (rOAT3). rOAT3-expressing oocytes exhibited [3H]HVA uptake (Km = 274 μmol/L), which was inhibited by several organic anions, such as PAH, indoxyl sulfate, octanoic acid, and metabolites of monoamine neurotransmitters. Neurotransmitters themselves did not affect the uptake. Furthermore, immunohistochemical analysis suggested that rOAT3 is localized at the abluminal membrane of brain capillary endothelial cells. These results provide the first evidence that rOAT3 is expressed at the abluminal membrane of the rat BBB and is involved in the brain-to-blood transport of HVA. This HVA efflux transport system is likely to play an important role in controlling the level of HVA in the CNS.
Keywords
Homovanillic acid (HVA) is a major metabolite of dopamine, and is produced by oxidation and deamination of dopamine by monoamine oxidase and catechol-O-methyl transferase. Abnormalities in cerebral dopamine function have been implicated in many mental and neurologic disorders such as parkinsonism and various types of seizures. In these disorders, CSF levels of HVA have been reported to be variable (Le Witt and Galloway, 1990; Giroud et al., 1990). Patients with uremic encephalopathy show markedly elevated HVA concentrations in the CSF (Sullivan et al., 1978). Moreover, in Reye's syndrome with encephalopathy, the HVA concentration in the CSF is also elevated (Shaywitz et al., 1979), whereas the dopamine concentration in the caudate nucleus is normal (Lloyd et al., 1977). These observations raise the possibility that accumulation of HVA in the brain may cause encephalopathy.
The CNS is believed to possess an efflux system that prevents the accumulation of HVA in the brain, because HVA concentration in the brain was increased when probenecid or octanoic acid (a fatty acid that is increased in the blood of patients with Reye's syndrome and medium-chain acyl-CoA dehydrogenase deficiency), was administered peripherally (Emanuelsson et al., 1987; Kim et al., 1992). Moreover, microdialysis findings suggested that the brain-to-blood efflux transport rate of HVA is faster than the blood-to-brain HVA influx rate across the blood–brain barrier (BBB) (Morrison et al., 1999). Thus, after metabolic conversion of dopamine to HVA, the efflux transport of HVA from the brain should play an important role in controlling the HVA level in the brain, and a decrease of the HVA brain-to-blood efflux rate might lead to an increase of HVA level in the brain. Therefore, clarification of the properties of HVA efflux transport from the brain across the BBB and identification of the responsible transporter are key issues for improving our understanding of dopaminergic system homeostasis in the brain.
The BBB, which is formed by the tight intercellular junctions of brain capillary endothelial cells, has been shown to regulate strictly the transfer of substances between the circulating blood and the brain (Pardridge, 1999). Recently, it has been revealed that the in vivo efflux transport of organic anions such as indoxyl sulfate (Ohtsuki et al., 2002) and 17β-estradiol-
Rat organic anion transporter 1 was identified as a para-aminohippuric acid (PAH) transporter (Sekine et al., 1997; Sweet et al., 1997). Subsequently, rOAT2 (Sekine et al., 1998) and rOAT3 (Kusuhara et al., 1999) have also been cloned. Organic anion transporters mediate the transport of many endogenous and exogenous compounds and play an important physiologic function in determining the tissue distribution of organic anions. It is still unknown, however, whether OATs can mediate HVA transport and are involved in HVA efflux transport at the BBB in vivo.
The purpose of the present study is therefore to identify the transporter that mediates HVA efflux transport from the brain across the BBB by using the Brain Efflux Index (BEI) method (Kakee et al., 1996). In addition, we investigated the biophysical characteristics of the transporter by using a Xenopus laevis oocyte expression system, and its localization at the BBB by means of immunohistochemical analysis.
MATERIALS AND METHODS
Animals
Adult male Wistar rats, weighing 230 to 280 g, were purchased from Charles River Laboratories (Yokohama, Japan). Mature female X laevis were purchased from Hamamatsu Kyozai (Hamamatsu, Japan) and maintained in a controlled environment as described by Goldin (1992). All experiments were approved by the Animal Care Committee, Graduate School of Pharmaceutical Sciences, Tohoku University.
Reagents
The pGEM-HEN vector was a kind gift from Dr. T. Abe (Tohoku University, Japan). [carboxyl-14 C]Inulin ([14C]inulin, 1.92 mCi/g) was purchased from ICN Biochemicals (Costa Mesa, CA, U.S.A.). [ring-3H]Homovanillic acid ([3H]HVA, 12.5 Ci/mmol) was synthesized and purified by IZOTOP (Budapest, Hungary). para-Aminohippuric acid, probenecid, benzylpenicillin potassium salt, cimetidine, indomethacin, valproic acid sodium salt, octanoic acid sodium salt, 5-hydroxyindoleacetic acid (5-HIAA), quinolinic acid,
Brain Efflux Index study
In vivo brain efflux experiments were performed by use of the intracerebral microinjection technique (Kakee et al., 1996). In brief, a Wistar rat was anesthetized by intramuscular injection of ketamine–xylazine (1.22 mg/kg xylazine and 125 mg/kg ketamine) and was placed in a stereotaxic frame (SR-6; Narishige, Tokyo, Japan), which determines the coordinates of the rat brain coinciding with the parietal cortex area 2 (Par2) region. The applied solution (0.50 μL) containing 160 nCi of [3H]HVA and 8 nCi of [14C]inulin, which acts as a reference compound, in an extracellular fluid buffer (122 mmol/L NaCl, 25 mmol/L NaHCO3, 3 mmol/L KCl, 1.4 mmol/L CaCl2, 1.2 mmol/L MgSO4, 0.4 mmol/L K2HPO4, 10 mmol/L
The apparent BBB efflux rate constant (keff) was determined from the slope given by fitting a semilogarithmic plot of (100-BEI), that is the percentage of remaining test substrate in the ipsilateral cerebrum, versus time, using the nonlinear least-squares regression analysis program MULTI (Yamaoka et al., 1981).
To characterize the efflux transport system at the BBB, the BEI value of [3H]HVA at 40 minutes was determined in the presence or absence of several inhibitors.
Expression of rat organic anion transporter 3 in Xenopus laevis oocytes
Capped complementary RNA (cRNA) of rOAT3 was transcribed from NotI-linearized pGEM-HEN containing rOAT3 complementary DNA with T7 RNA polymerase as described (Deguchi et al., 2002). Defolliculated oocytes were injected with 50 nL of water or the capped rOAT3 cRNA (10 ng/50 nL) and incubated in Barth's solution (88 mmol/L NaCl, 1 mmol/L KCl, 0.33 mmol/L Ca(NO3)2, 0.4 mmol/L CaCl2, 0.8 mmol/L MgSO4, 2.4 mmol/L NaHCO3, and 10 mmol/L HEPES) containing 50 μg/mL gentamicin and 2.5 mmol/L pyruvate, pH 7.4, at 18°C. After incubation for 3 d, uptake experiments were performed.
Uptake by oocytes
Before the uptake experiment, oocytes were preincubated with 500 μL of ND96 solution (96 mmol/L NaCl, 2 mmol/L KCl, 1.8 mmol/L CaCl2, 1 mmol/L MgCl2, and 5 mmol/L HEPES, pH 7.4) for 20 minutes at 20°C. The uptake experiment was initiated by replacing the ND96 solution with 200 μL of the same solution containing 200 nmol/L [3H]HVA (0.5 μCi), and terminated by addition of ice-cold ND 96 solution after incubation for 1 h at 20°C. Oocytes were washed four times with ice-cold ND96 solution and solubilized in 5% sodium dodecyl sulfate solution, and the accumulated radioactivity was determined in a liquid scintillation counter (LS-6500; Beckman Coulter).
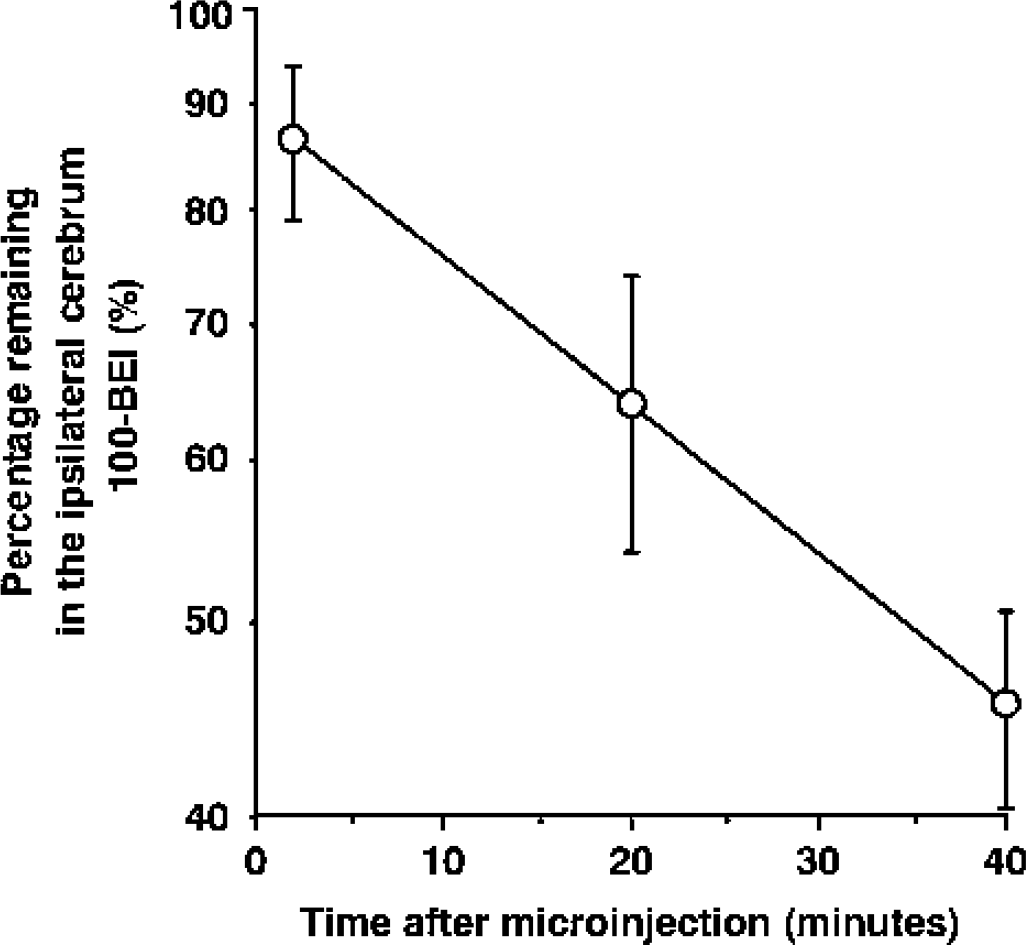
The kinetic parameters for the uptake of HVA by rOAT3 were estimated from the following Eq. 3,
where V is the uptake rate of the HVA [pmol/(h · oocyte)], C is the HVA concentration in the medium (mmol/L), Km is the Michaelis-Menten constant, and Vmax is the maximum uptake rate [pmol/(h · oocyte)]. To obtain the kinetic parameters, the equation was fitted to the rOAT3-specific transport rate, which was obtained by subtracting the transport rate in water-injected oocytes from that in rOAT3-expressing oocytes, by an iterative nonlinear least-squares method using the MULTI program (Yamaoka et al., 1981). The input data were weighted as the reciprocal of the observed values, and the Damping-Gauss Newton Method algorithm was used for fitting. The fitted line was converted to the V/C versus V form (Eadie-Scatchard plot).
For the inhibition study, uptake of 200 nmol/L [3H]HVA by rOAT3 was measured in the presence or absence of 1 mmol/L unlabeled compounds in ND96 solution. The specific uptake was obtained by subtracting the uptake into water-injected oocytes from the uptake into rOAT3-expressing oocytes.
Data analysis
All data are presented as the mean ± SD. An unpaired, two-tailed Student's t test was used to determine the significance of differences between two group means. One-way analysis of variance followed by the modified Fisher's least-squares difference method was used to assess the statistical significance of differences among means of more than two groups.
Anti-rat organic anion transporter 3 antibody and Western blot analysis
Antiserum against an oligopeptide (KTKQESEAEKAS) corresponding to amino acid residues 516 to 527 of rOAT3 was raised in rabbit (Sigma Genosys Japan, Ishikari, Japan). The immunogloblin fraction was purified using protein-A Sepharose (Amersham Biosciences, Piscataway, NJ, U.S.A.). Immunoglobulins specific to rOAT3 oligopeptide were affinity-purified using a HiTrap NHS-activated HP Column (Amersham Biosciences) carrying rOAT3 oligopeptide.
The membrane protein fraction of rat kidney, small intestinal epithelium, brain, and brain capillary–rich fraction for rOAT3 was prepared using a reported procedure (Hosoya et al., 2000). The protein samples were boiled for 3 minutes and subjected to sodium dodecyl sulfate–polyacrylamide gel electrophoresis (50 μg per lane). The separated proteins were electrotransferred to nitrocellulose membranes (Toyo Roshi, Tokyo, Japan), and the membranes were treated with blocking solution (Block Ace; Dainippon Pharmaceutical, Osaka, Japan) for 1 h at room temperature and with diluted affinity-purified anti-rOAT3 antibody (1 μg/mL) for 16 h at 4°C. Membranes were washed 4 times with 0.1% Tween 20/phosphate-buffered saline and then incubated with horseradish peroxidase–conjugated goat anti–rabbit immunoglobulin G (IgG; 1:5,000; Kirkegaard Perry Laboratories, Gaithersburg, MD, U.S.A.) for 1 h at room temperature. Immunoreactivity was visualized with an enhanced chemiluminescence kit (Supersignal west pico chemiluminescent substrate; Pierce, Rockford, IL, U.S.A.).
Immunostaining analysis
Adult Wistar rats were anesthetized with an intramuscular injection of ketamine and xylazine before perfusion through the common carotid artery with 4% paraformaldehyde in 0.1 mol/L phosphate buffer. The cerebrum from each animal was isolated and immersed in 10% sucrose/0.1 mol/L phosphate buffer solution. Sagittal sections (10-μm thick) were cut from the frozen cerebrum using a Leica cryostat (CM1900; Leica, Heidelberg, Germany) and were mounted onto silanized glass slides (DAKO, Carpinteria, CA, U.S.A.). The sections were incubated with 10% goat serum (Histofine; Nichirei, Tokyo, Japan) for 30 minutes at room temperature. The primary antibody (1 μg/mL rabbit anti-rOAT3 antibody or 0.5 μg/mL mouse anti–P-glycoprotein antibody (C219; Signet Laboratories, Dedham, MA, U.S.A.) or 6 μg/mL mouse anti-glial fibrillary acidic protein (GFAP) antibody (G-A-5; Sigma) was applied to the sections, which were incubated for about 36 h at 4°C. For single staining of rOAT3, this was followed by incubation for 1 h at room temperature with Alexa 488–conjugated goat anti–rabbit IgG (1:200; Molecular Probes, Eugene, OR, U.S.A.) as a secondary antibody. Then, nuclei were stained with 6.6 μmol/L propidium iodide. For double staining, Alexa 546–conjugated goat anti–mouse IgG (1:200; Molecular Probes) was used as a secondary antibody for anti–P-glycoprotein antibody or anti-GFAP antibody. Sections were mounted with coverslips using Vectashield mounting medium (Vector Laboratories, Burlingame, CA, U.S.A.). The immunofluorescence-stained sections were viewed using a confocal laser microscope (TCS SP; Leica).
RESULTS
Efflux transport of homovanillic acid from the rat brain across the blood–brain barrier
Figure 1 shows the time profile of the remaining percentage of [3H]HVA in the ipsilateral cerebrum after microinjection into the Par2 region of the brain. [3H]Homovanillic acid was eliminated in a time-dependent manner, and the apparent efflux rate constant, keff, was determined to be 1.69 × 10–2 ± 9.60 × 10–5 minute–1 (mean ± SD) from the slope given by fitting a semilogarithmic plot of (100-BEI) values versus time (Fig. 1). No radioactivity associated with this efflux transport process was detected in the contralateral cerebrum, cerebellum, or CSF (data not shown), suggesting the operation of a selective efflux transport process across the BBB. [14C]Inulin, used as an impermeable marker, however, remaining in the brain did not change during the 40 minutes after microinjection (data not shown).
Time course of [3H]homovanillic acid (HVA) in the ipsilateral cerebrum after intracerebral microinjection in the presence of [14C]inulin as an internal reference. A mixture of [3H]HVA (160 nCi) and [14C]inulin (8 nCi) dissolved in 0.50 μL of extracellular fluid buffer was injected into the parietal cortex area 2 region of the brain. The solid line was obtained with the nonlinear least-squares regression analysis program, MULTI. Each point represents the mean ± SD (n = 3 to 4). BEI, Brain Efflux Index.
Inhibitory effect of several compounds on [3H]homovanillic acid efflux transport from the brain
To characterize the HVA efflux transport process at the BBB in vivo, several compounds were coadministered with [3H]HVA and their inhibitory effects on the [3H]HVA efflux transport were investigated (Table 1). para-Aminohippuric acid, at a concentration of 100 mmol/L in the injection solution, significantly inhibited [3H]HVA efflux transport (by 39.4%). Substrates for rOAT3 such as benzylpenicillin (Kusuhara et al., 1999) at 100 mmol/L, indoxyl sulfate (Deguchi et al., 2002) at 100 mmol/L, and cimetidine (Kusuhara et al., 1999) at 50 mmol/L also significantly reduced [3H]HVA efflux transport (by 78.6%, 49.9%, and 36.7%, respectively). In contrast, indomethacin, which has a high affinity for rOAT1 and 2 (Apiwattanakul et al., 1999; Morita et al., 2001), did not have a significant effect on [3H]HVA efflux transport at the concentration of 1 mmol/L (Table 1). These results suggest that rOAT3 at the BBB participates in HVA efflux transport from rat brain.
Effect of coadministration of several compounds on [3H]homovanillic acid efflux from rat brain
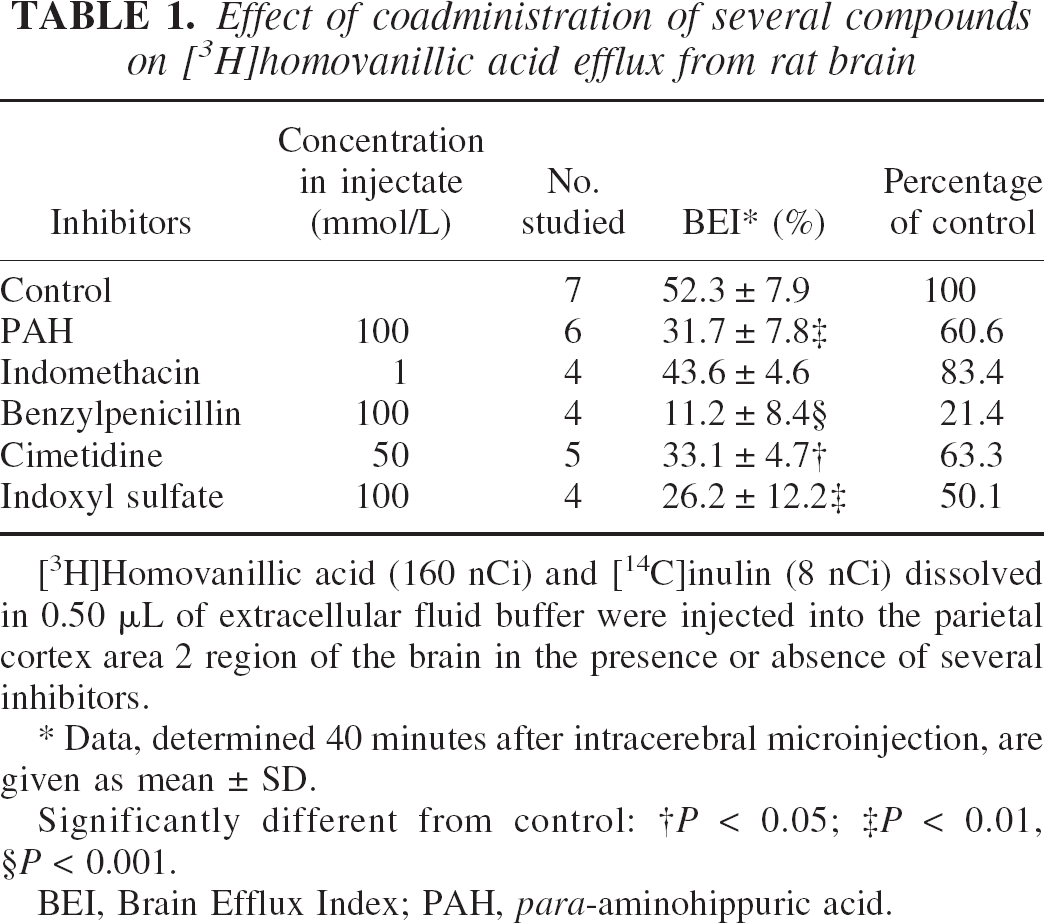
[3H]Homovanillic acid (160 nCi) and [14C]inulin (8 nCi) dissolved in 0.50 μL of extracellular fluid buffer were injected into the parietal cortex area 2 region of the brain in the presence or absence of several inhibitors.
Data, determined 40 minutes after intracerebral microinjection, are given as mean ± SD.
Significantly different from control:
P < 0.05
P < 0.01
P < 0.001.
BEI, Brain Efflux Index; PAH, para-aminohippuric acid.
Uptake of homovanillic acid by rat organic anion transporter 3–expressing oocytes
To elucidate the involvement of rOAT3 in HVA transport at the BBB, an X laevis oocyte expression system was used. [3H]Homovanillic acid was taken up by oocytes injected with cRNA derived from rOAT3 complementary DNA. The uptake rate of [3H]HVA in rOAT3 cRNA-injected oocytes at 60 minutes was 203 ± 47 pmol/(h · oocyte), whereas that in water-injected oocytes was 73.3 ± 25.8 pmol/(h · oocyte) (P < 0.01). This rOAT3-mediated [3H]HVA uptake was linear during at least 120 minutes (data not shown). Moreover, the rOAT3-mediated uptake of HVA showed saturable kinetics, followed the Michaelis-Menten equation (Fig. 2), and gave a single straight line on the Eadie-Scatchard plot (Fig. 2, inset). The estimated Km and Vmax values were 274 ± 100 μmol/L and 41.3 ± 12.5 pmol/(h · oocyte), respectively. These results indicate that rOAT3 transports HVA. Table 2 summarizes the effect of various compounds on [3H]HVA uptake by rOAT3-expressing oocytes. Several inhibitors of rOAT3, such as probenecid, PAH, benzylpenicillin, cimetidine, and estrone sulfate inhibited the rOAT3-mediated uptake of [3H]HVA by more than 75.5%, whereas indomethacin inhibited it by 59.1%.
Effect of several compounds on [3H]homovanillic acid uptake by Xenopus laevis oocytes injected with rat organic anion transporter 3 complementary RNA
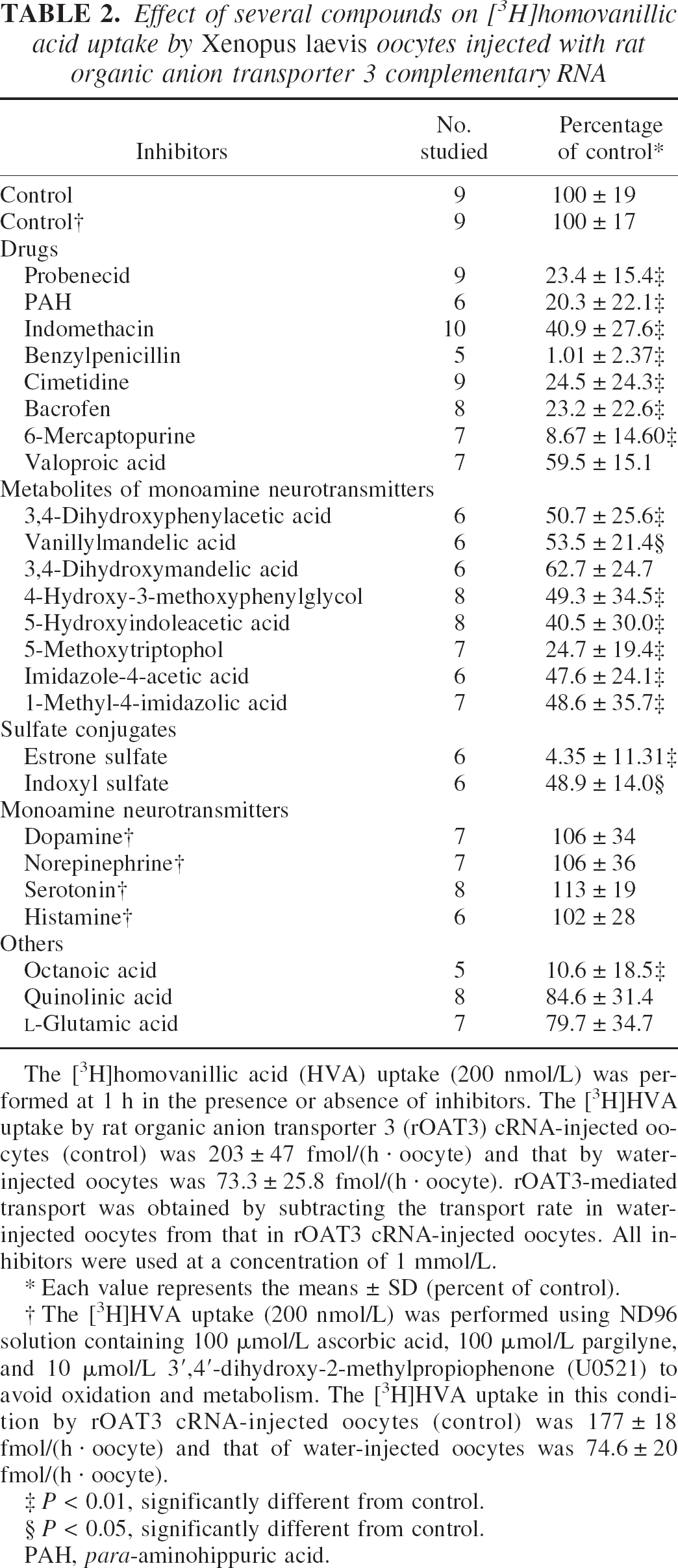
The [3H]homovanillic acid (HVA) uptake (200 nmol/L) was performed at 1 h in the presence or absence of inhibitors. The [3H]HVA uptake by rat organic anion transporter 3 (rOAT3) cRNA-injected oocytes (control) was 203 ± 47 fmol/(h · oocyte) and that by water-injected oocytes was 73.3 ± 25.8 fmol/(h · oocyte). rOAT3-mediated transport was obtained by subtracting the transport rate in water-injected oocytes from that in rOAT3 cRNA-injected oocytes. All inhibitors were used at a concentration of 1 mmol/L.
Each value represents the means ± SD (percent of control).
The [3H]HVA uptake (200 nmol/L) was performed using ND96 solution containing 100 μmol/L ascorbic acid, 100 μmol/L pargilyne, and 10 μmol/L 3′,4′-dihydroxy-2-methylpropiophenone (U0521) to avoid oxidation and metabolism. The [3H]HVA uptake in this condition by rOAT3 cRNA-injected oocytes (control) was 177 ± 18 fmol/(h · oocyte) and that of water-injected oocytes was 74.6 ± 20 fmol/(h · oocyte).
P < 0.01, significantly different from control.
P < 0.05, significantly different from control.
PAH, para-aminohippuric acid.
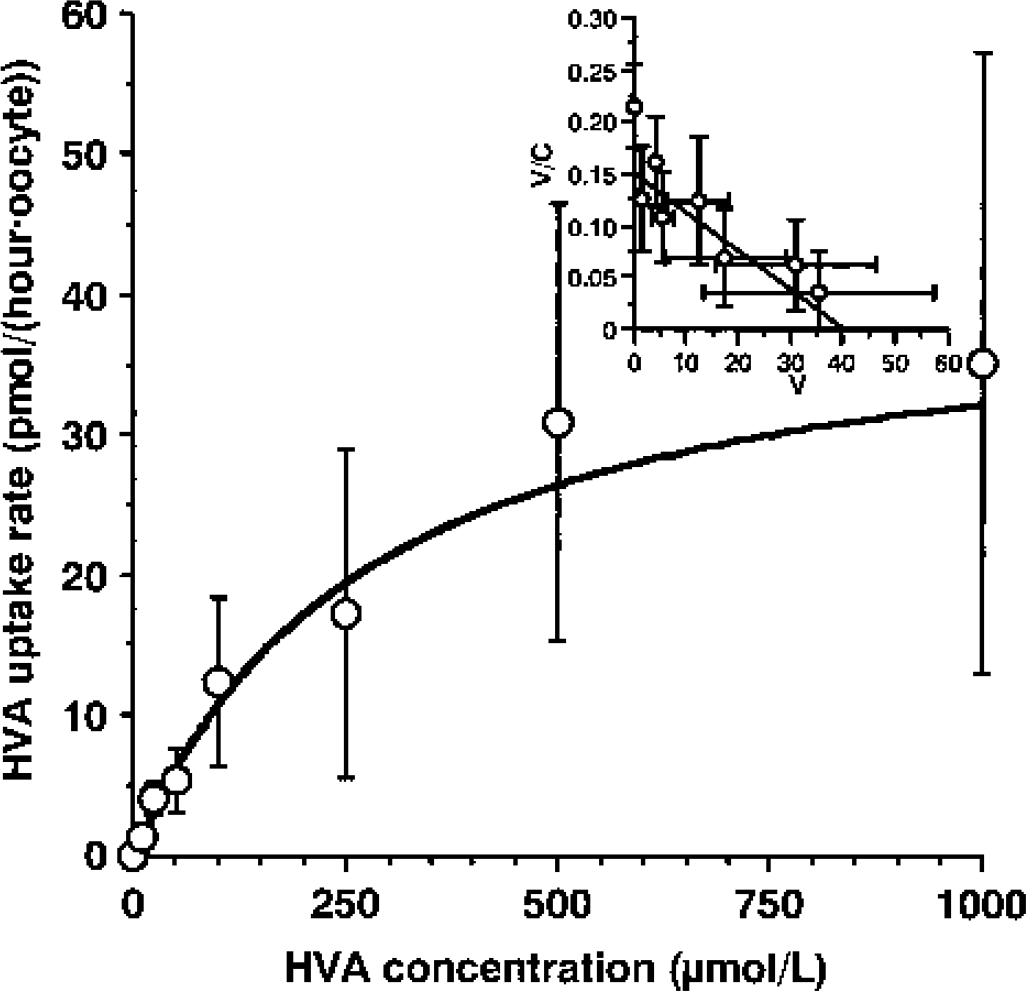
Concentration dependence of rat organic anion transporter 3 (rOAT3)–mediated uptake of homovanillic acid (HVA). The uptake of [3H]HVA by water-injected or rOAT3-injected Xenopus laevis oocytes was measured at the indicated concentration after incubation for 1 h. rOAT3-mediated transport was obtained by subtracting the transport rate in water-injected oocytes from that in rOAT3 complementary RNA–expressing oocytes.
Metabolites of neurotransmitters, such as 3,4-dihydroxyphenylacetic acid derived from dopamine, vanillylmandelic acid, 3,4-dihydroxymandelic acid and 4-hydroxy-3-methoxyphenylglycol derived from norepinephrine and epinephrine, 5-HIAA and 5-methoxytryptophol derived from serotonin, and imidazole-4-acetic acid and 1-methyl-4-imidazolic acid derived from histamine, inhibited the uptake of [3H]HVA in rOAT3-expressing oocytes by up to 75.3%. Monoamine neurotransmitters, however, such as dopamine, norepinephrine, serotonin, and histamine, and neurotoxic endogenous compounds, such as quinolinic acid and
Expression of rat organic anion transporter 3 protein in rat brain
The expression of rOAT3 protein in rat brain and brain capillary-rich fraction was determined by Western blot analysis using anti-rOAT3 antibody (Fig. 3). A single band at approximately 50 kd in rat brain and two bands at approximately 72 and 50 kd in rat brain capillary–rich fraction were detected. These bands had the same molecular weights as those in rat kidney, used as a positive control. In contrast, rat small intestinal epithelial cells, used as a negative control, showed no immunoreactive signals. These results suggest that rat brain and brain capillaries express rOAT3 protein.
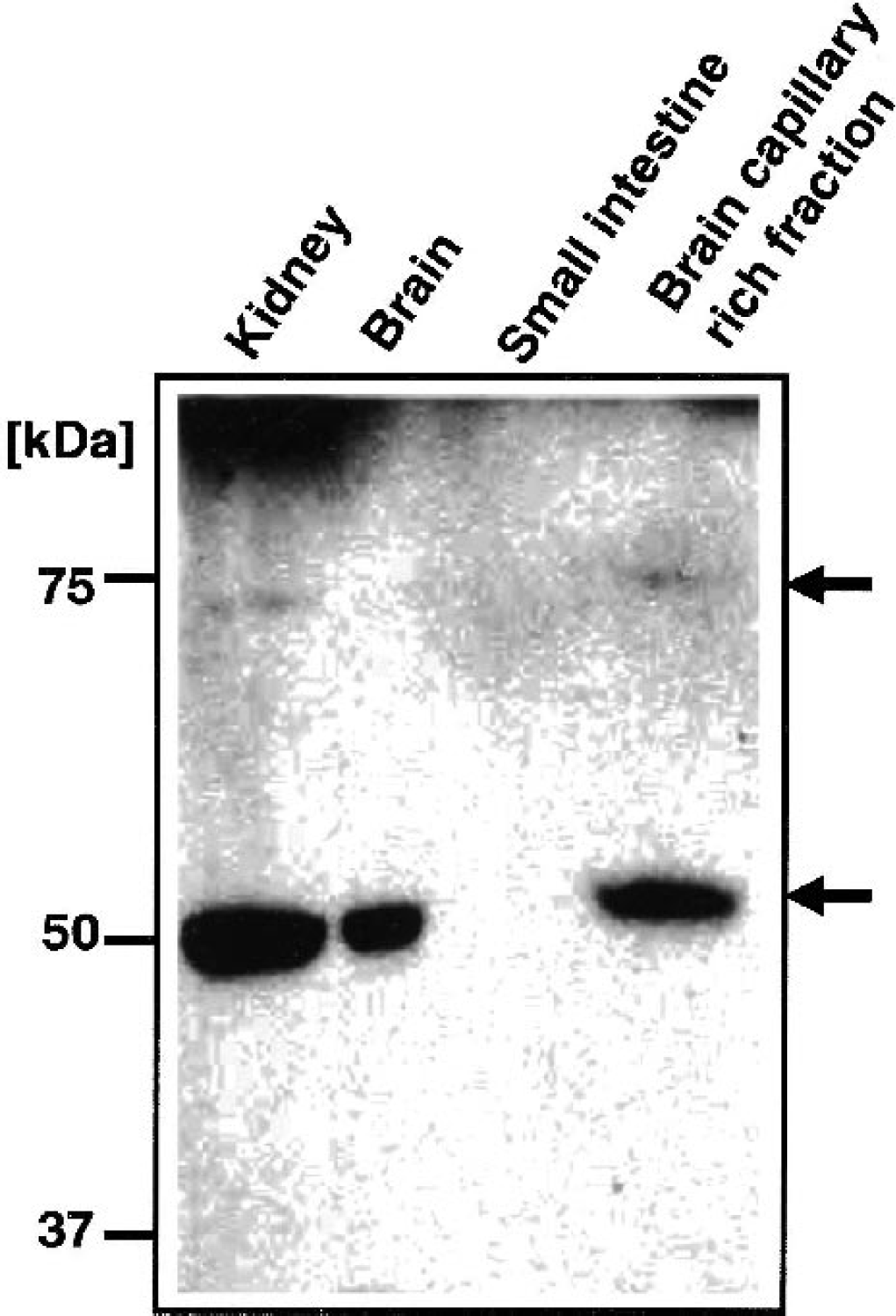
Western blot analysis of rat organic anion transporter 3 (rOAT3) with anti-rOAT3 antibody in rat kidney, small intestine epithelial cells, brain, and brain capillary. The two bands detected at 72 and 50 kd are indicated by arrows on the right-hand side.
Localization of rat organic anion transporter 3 protein in rat brain capillaries
The localization of rOAT3 in rat brain was examined immunohistochemically by using anti-rOAT3 antibody. The brain capillaries in cerebral cortex were stained with anti-rOAT3 antibody (green; Figs. 4A and 4B). Green fluorescence was observed on the outer side of endothelial cell nuclei (red; Figs. 4A and 4B). Furthermore, the rOAT3 immunoreactivity (green; Fig. 4C) was localized on the outer side of immunostaining with anti–P-glycoprotein antibody (C219), which reacts with the luminal membrane of capillary endothelial cells (red; Fig. 4C). In contrast, the rOAT3 immunoreactivity (green; Fig. 4D) was localized on the inner side of GFAP staining, which is an astrocyte marker (red; Fig. 4D). No significant fluorescence was observed in brain capillaries stained with normal rabbit IgG (data not shown). These results support the idea that rOAT3 is localized on the abluminal side of the BBB. In addition, some neural cells, which were negative to GFAP staining, also appeared to be stained with anti-rOAT3 antibody (data not shown). This suggests that rOAT3 is expressed in neural cells, except for astrocytes, although the cell types were not identified.
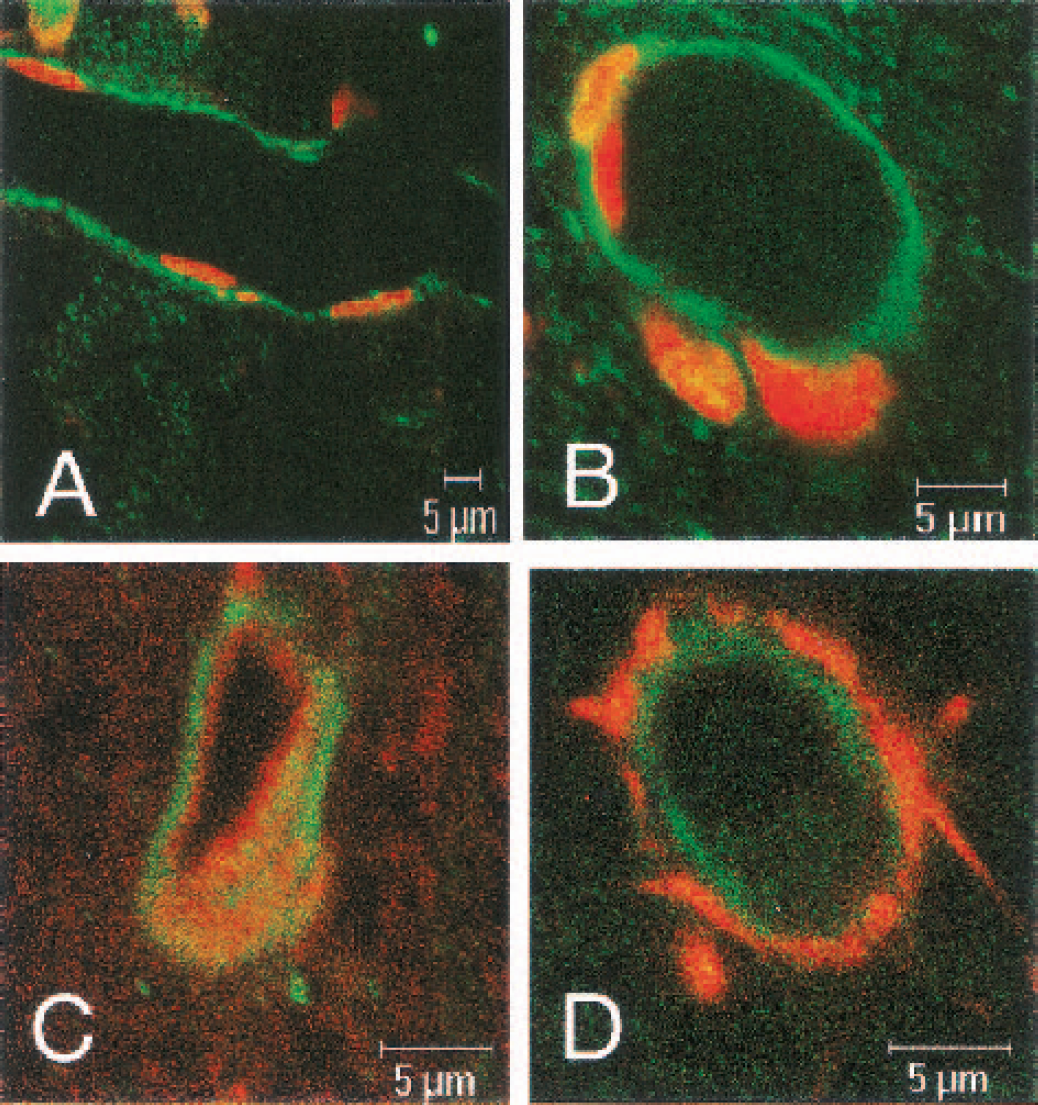
Confocal immunofluorescence microscope images of singly
DISCUSSION
The present study shows that rOAT3 is expressed at the abluminal membrane of rat brain capillary endothelial cells and is responsible for brain-to-blood efflux transport of HVA, an end metabolite of dopamine, across the BBB in vivo.
Using the BEI method, we showed that [3H]HVA was eliminated from the brain across the BBB with a half-life of 41.0 minutes (Fig. 1). The HVA efflux transport was significantly inhibited not only by organic anion compounds, such as PAH, benzylpenicillin, and indoxyl sulfate, but also by the organic cation compound cimetidine (Table 1). para-Aminohippuric acid is a common substrate for rOAT1, rOAT2, and rOAT3 and has inhibitory effects similar to those of probenecid on these transporters (Sekine et al., 1997, 1998; Sweet et al., 1997; Kusuhara et al., 1999). Benzylpenicillin and cimetidine are relatively specific inhibitors for rOAT3 among the OAT family members (Nagata et al., 2002; Feng et al., 2001), and indoxyl sulfate is a substrate of rOAT3 (Deguchi et al., 2002; Ohtsuki et al., 2002). Furthermore, [3H]HVA is taken up by rOAT3-expressing oocytes in a saturable process, as shown in Fig. 2, and this [3H]HVA uptake was inhibited by PAH, benzylpenicillin, cimetidine, and indoxyl sulfate as was the case in the in vivo study (Tables 1 and 2). Rat organic anion transporter 3–mediated [3H]HVA transport was also significantly inhibited by probenecid and octanoic acid. This result is in good agreement with previous reports showing that the HVA concentration in the brain was increased by systemic administration of probenecid or octanoic acid (Emanuelsson et al., 1987; Kim et al., 1992). These results suggest that rOAT3 is involved in the HVA efflux transport across the BBB.
Indomethacin is a potent inhibitor of rOAT1 (Ki = 10 μmol/L; Apiwattanakul et al., 1999) and rOAT2 (Ki = 0.37 μmol/L; Morita et al., 2001). In this study, its Ki value for rOAT3 was estimated to be about 1 mmol/L, because indomethacin (1 mmol/L) inhibited rOAT3-mediated [3H]HVA uptake by 59.1% (Table 2). Although the inhibitors administered to the cerebrum gradually diffuse from the point of injection, an average concentration of indomethacin in the area reaching the [3H]HVA is calculated to be about 33 μmol/L with reference to the dilution factor (i.e., 30.3) reported previously (Kakee et al., 1996). Indomethacin has no significant inhibitory effect in the BEI study (Table 1) because the cerebral concentration is sufficient to inhibit rOAT1 and rOAT2, but not rOAT3. Therefore, rOAT3 is suggested to be responsible for the HVA efflux transport at the BBB, and it seems that the involvement of rOAT1 and rOAT2 in the efflux transport is relatively small.
Recently, we reported the detection of rOAT3 mRNA by reverse transcription–polymerase chain reaction in the capillary-rich fraction of rat brain and in conditionally immortalized rat brain capillary endothelial cells (TR-BBB) (Ohtsuki et al., 2002). The protein expression of rOAT3 and its membrane localization in brain capillary endothelial cells, however, are unknown. Figure 3 shows the expression of rOAT3 protein in the brain capillary-rich fraction. Two bands at 72 kd and 50 kd were detected both in brain capillary-rich fraction and kidney, where rOAT3 is reported to be expressed (Hasegawa et al., 2002). The same immunoreactivity pattern was reported in rOAT3-expressing LLC-PK1 cells, which are porcine renal proximal tubular epithelial cell line, and the kidney using another anti-rOAT3 antibody (Hasegawa et al., 2002). The difference in the molecular size may be because of the difference in the degree of glycosylation. This result suggests that rOAT3 is expressed in brain capillary endothelial cells. The crude membrane fraction of the brain, however, has a strong band at 50 kd (Fig. 3). Part of this immunoreactivity probably reflects rOAT3 expression in neural cells other than brain capillaries.
Figure 4 shows the first evidence that rOAT3 protein is expressed in rat brain capillaries and that it is mainly localized at the abluminal membrane. Rat organic anion transporter 3 may therefore be involved in the HVA uptake step from the brain interstitial fluid into endothelial cells at the abluminal membrane of BBB. Polarized localization of transporters at the brain capillary endothelial cells controls the transport direction across the BBB. It is conceivable that multidrug resistance–associated proteins (MRPs) on the luminal membrane of the BBB are involved in the efflux transport of HVA from the cells to the circulating blood, because MRP1, 2, 4, 5 and 6, which are the primary active transporters mediating the flux of organic anions from the intracellular to the extracellular compartment, are expressed at the bovine and/or rat BBB (Zhang et al., 2000; Miller et al., 2000). Regarding luminal membrane transport, luminal localization of rOAT3 could not be ruled out yet because it has been reported that the expression of GLUT1 at the abluminal side is fourfold greater than that at the luminal side (Farrell and Pardridge, 1991). To clarify these issues, further investigations, such as electron microscope studies, are necessary.
As shown in Table 2, rOAT3-mediated uptake of [3H]HVA was significantly inhibited by many metabolites derived from dopamine, norepinephrine, epinephrine, serotonin, and histamine, suggesting that these monoamine neurotransmitter metabolites undergo brain-to-blood efflux transport by rOAT3, as does HVA. The HVA concentration in the normal rat brain is reported to be between 3.6 and 7.5 μmol/L (Kopin, 1985), and other metabolites have similar brain concentrations (e.g., 3,4-dihydroxyphenylacetic acid: 4.9 to 9.2 μmol/L [Kopin, 1985]). The Km value of HVA transport by rOAT3 is 274 μmol/L (Fig. 2), and the estimated affinity constants of rOAT3 for the transport of other metabolites are about 1 mmol/L because 1 mmol/L concentration of these metabolites exhibited about 50% inhibition of HVA uptake by rOAT3 (Table 2). Therefore, in relation to the brain concentrations of metabolites, the affinity constants for rOAT3 would be high enough so that the brain-to-blood efflux transport at the BBB would not be saturated. This seems to be the case even under conditions where the metabolite levels are increased to several-fold greater than normal, as in the prodromal phase of schizophrenia (Sumiyoshi et al., 2000).
The regional concentration of neurotransmitters and their metabolites vary widely, depending on the region and density of the neurons. However, rOAT3 is likely to distribute at the capillaries over a wide region of the brain to eliminate various metabolites, because rOAT3 would be involved in the efflux transport of not only HVA but also other anionic monoamine metabolites and is capable of transporting the metabolites in high concentration. In the present immunohistochemical analysis, rOAT3 immunoreactivity was mainly observed in the cerebral cortex, which is rich in brain capillaries (Fig. 4). Nevertheless, it is still possible that there is a regional expression of rOAT3 in the brain.
Unlike the metabolites, monoamine neurotransmitters themselves, such as dopamine, norepinephrine, serotonin, and histamine, did not inhibit [3H]HVA uptake by rOAT3 (Table 2). This result suggests that rOAT3 does not mediate neurotransmitter transport. Our previous report showed that norepinephrine and serotonin transporters are expressed in the abluminal membrane of mouse brain capillary endothelial cells (Wakayama et al., 2002). Moreover, GAT2/BGT-1, the γ-aminobutyric acid transporter, is localized at the mouse BBB and plays a role in brain-to-blood efflux transport of γ-aminobutyric acid (Takanaga et al., 2001). Thus, the efflux transport of neurotransmitters seems to be mediated by neurotransmitter transporters.
In patients with Reye's syndrome and medium-chain acyl-CoA dehydrogenase deficiency, octanoic acid concentrations in the blood increase dramatically with concomitant increases in the brain because of the transport of octanoic acid across the BBB (Oldendorf, 1973). The HVA concentration in the CSF is elevated in patients with Reye's syndrome (Shaywitz et al., 1979). In rats, after intraperitoneal administration of octanoic acid, the concentrations of HVA and 5-HIAA in the caudate nucleus were increased to 154% and 150%, respectively (Kim et al., 1992). These observations suggest that increased plasma and brain levels of octanoic acid could induce elevation of brain HVA levels in patients. Recently, hOAT3 was isolated as a human homolog of rOAT3. The transport properties and tissue distribution of hOAT3 are similar to those of rOAT3 (Cha et al., 2001), suggesting that hOAT3 may also be expressed at the human BBB. As shown in Table 2, octanoic acid (at 1 mmol/L concentration) significantly inhibited rOAT3-mediated HVA uptake by 90%, and the plasma level of octanoic acid in severe cases of Reye's syndrome reaches about 1 mmol/L (Mamunes et al., 1975). Therefore, based on our present results, we propose that increased octanoic acid in plasma and brain of patients inhibits the brain-to-blood efflux transport of HVA mediated by OAT3. Uremia is also associated with increases of brain HVA level and plasma levels of uremic toxins, such as indoxyl sulfate. We have shown that indoxyl sulfate is eliminated from the brain by rOAT3 at the BBB, and that rOAT3-mediated indoxyl sulfate transport is inhibited by HVA (Ohtsuki et al., 2002). In the present study, indoxyl sulfate inhibited the efflux transport of [3H]HVA from the brain in vivo (Table 1) and the rOAT3-mediated HVA uptake (Table 2) as efficiently as did octanoic acid. The combined effects of the compounds described could account for the accumulation of neurotransmitter metabolites in the brain that is observed in Reye's syndrome and uremia with encephalopathy.
Homovanillic acid is excreted from plasma to urine in the kidneys after its efflux from the brain. A previous study has shown that rOAT3 is involved in renal efflux transport of indoxyl sulfate from plasma (Deguchi et al., 2002). Thus, rOAT3 also seems to be involved in HVA efflux transport in the kidney. The HVA concentration in human plasma (69.4 nmol/L) is lower than the Km value of HVA transport by rOAT3 (274 μmol/L) (Kopin, 1985), suggesting that the HVA transport system in the kidney is not saturated. Therefore, it is reasonable that HVA concentration in blood and urine is widely used as an indicator of dopaminergic neuronal activity in the brain. However, rOAT3-mediated HVA uptake is inhibited by many kinds of compounds, including drugs such as baclofen and 6-mercaptopurine (Table 2). This result suggests that there are interactions between HVA, drugs, and endogenous compounds that influence the rOAT3-mediated transport. Thus, when organic anion drugs are administered or in disorders such as Reye's syndrome, peripheral HVA concentration may not accurately reflect dopaminergic neuronal activity in the CNS.
In conclusion, this study provides the first evidence that rOAT3, expressed at the abluminal membrane of the rat BBB, is involved in efflux transport of HVA from the brain. These results throw light on one of the final steps among the mechanisms that maintain homeostasis of the dopaminergic neuronal system in the CNS.
Footnotes
Acknowledgments:
The authors thank Dr. T. Abe for supplying the pGEM-HEN vector for protein expression in X laevis oocytes, Dr. H. Asaba for technical assistance, and Ms. N. Funayama for secretarial assistance.