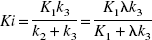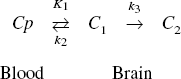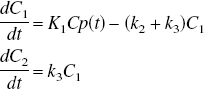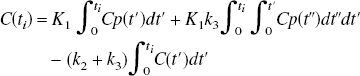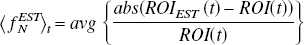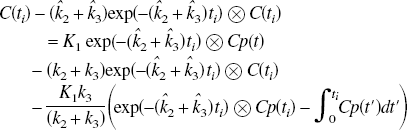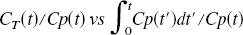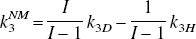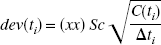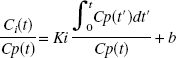Abstract
The construction of parametric positron emission tomography images of enzyme or receptor concentration obtained using irreversibly binding radiotracers presents problems not usually encountered with reversibly binding radiotracers. Difficulties are most apparent in brain regions having low blood flow and/or high enzyme or receptor concentration and are exacerbated with noisy data. This is especially true when minimal doses of radiotracer are administered. A comparison was recently reported of the irreversible monoamine oxidase A (MAO A) radiotracers [11C]clorgyline (CLG) and deuterium-substituted [11C]clorgyline (CLG-D) in the human brain using region of interest (ROI) analysis in which the authors observed an unexpected loss of image contrast with CLG-D compared with CLG. In order to more fully investigate patterns of binding of these irreversibly binding radiotracers, a strategy was devised to reduce noise in the generation of parametric images of the model term related to enzyme or receptor concentration. The generalized linear least squares (GLLS) method of Feng et al. (1995), a rapid linear method that is unbiased, was used for image-wide parameter estimation. Since GLLS can fail in the presence of large amounts of noise, local voxels were grouped within the image to increase the signal, and the GLLS method was combined with the standard nonlinear estimation methods when necessary. Voxels were grouped together depending on their proximity and whether they fell within a specified range of the time-integrated image. It was assumed that voxels meeting both criteria are functionally related. Simulations reflecting varying enzyme concentrations were performed to assess precision and accuracy of parameter estimates in the presence of varying amounts of noise. Using this approach, images were generated of the combination parameter λk3 (λ = K1/ k2, where K1 and k2 are plasma-to-tissue and tissue-to-plasma transport constants, respectively) that is related to enzyme concentration as well as images of the transport constant K1 for individual subjects. Reasonably high-quality images of both K1 and λk3 were obtained for CLG and CLG-D for individual subjects even with low injected doses averaging 6 mCi. While there were no differences in the K1 images, the λk3 images revealed the loss of contrast previously reported for CLG-D using the ROI analysis. This method should be generalizable to other tracers and should facilitate the analysis of group differences.
The enzyme monoamine oxidase, which occurs in two forms, MAO A and MAO B, catalyzes the oxidative deamination of amines from endogenous and exogenous sources (Shih et al., 1999). The two subtypes occur in different types of cells in the brain, with MAO B occurring predominately in glial cells and serotonergic neurons while MAO A occurs in catecholaminergic neurons as well as in glial cells. From a medical viewpoint, these enzymes are of interest for, among other things, their ability to regulate chemical neurotransmitters. Drugs inhibiting MAO have been used in the treatment of depression and Parkinson's disease. Two 11C-labeled positron emission tomography (PET) tracers have been used for imaging MAO: L-deprenyl (MAO B) and clorgyline (MAO A). Both of these compounds are irreversible inhibitors of MAO. They bind covalently to the enzyme, which also inactivates it. This binding also traps the 11C at the site of inactivation. While the graphical method of Patlak et al. (1985) is applicable to such irreversibly binding tracers and much easier to apply than compartment models, the influx constant, Ki, resulting from the Patlak analysis is a function of blood flow as well as the enzyme concentration, and both vary from one brain region to another. It is therefore necessary to separately evaluate the transport constants and model parameter related to enzyme concentration using some form of compartmental modeling. Another complicating factor is that tracer uptake for irreversible ligands is less sensitive to variations in enzyme level in brain regions having low blood flow and/or high enzyme concentration. In addition, parameter estimation is more difficult in the presence of noise, which is a greater problem in the construction of parametric images than in region of interest (ROI) analysis. Owing to the widespread distribution of MAO in the brain, it is advantageous to have an image map of the enzyme concentration, so these difficulties need to be addressed.
We report here a method for the construction and comparison of parametric images of MAO A in the human brain using [11C]clorgyline (CLG) and deuterium-substituted [11C]clorgyline (CLG-D). Deuterium substitution serves as a mechanistic tool for the investigation of the biochemical basis of the PET image. In the irreversible inhibition of MAO by CLG, the C-H bond on the methylene carbon of the propargyl group is cleaved in the rate-limiting step. When deuterium is substituted for hydrogen, the bond is more difficult to cleave and the rate of reaction is reduced, providing evidence that MAO catalysis is responsible for the irreversible trapping of C-11 in brain. We undertook this study in order to more thoroughly investigate unexpected differences in the magnitude of the deuterium isotope effect in different brain regions when these two tracers were compared using the ROI analysis (Fowler et al., 2001).
Two of the most important limiting factors for consideration in the construction of parametric images for CLG and CLG-D are (1) the signal-to-noise ratio at the voxel level and (2) the time involved in the parameter estimation process, which involves several thousand voxels per plane. The generalized least squares (GLLS) method is sufficiently rapid that images can be generated in a reasonable amount of time: on the order of minutes. It is, however, more sensitive to noise than the standard nonlinear estimation procedures. Low signal-to-noise ratio is a particular problem in the present study because the injected doses were low (6 to 7 mCi) in order to permit multiple studies in the same subject. In this study we compare, using computer simulations, the GLLS and nonlinear least squares (NLS) methods for the two-tissue compartment irreversible model in the presence of varying amounts of noise. Simulations reflecting varying enzyme concentrations were performed to assess precision and accuracy of parameter estimates. Results from the simulations indicate that the preferred model parameter for enzyme concentration is the combination parameter λk3 as opposed to k3 (λ = K1/k2, where K1 and k2 are plasma-to-tissue and tissue-to-plasma transport constants, respectively, and k3 is the model parameter directly proportional to enzyme concentration). Unlike K1 and k2, λ does not depend on blood flow.
THEORY
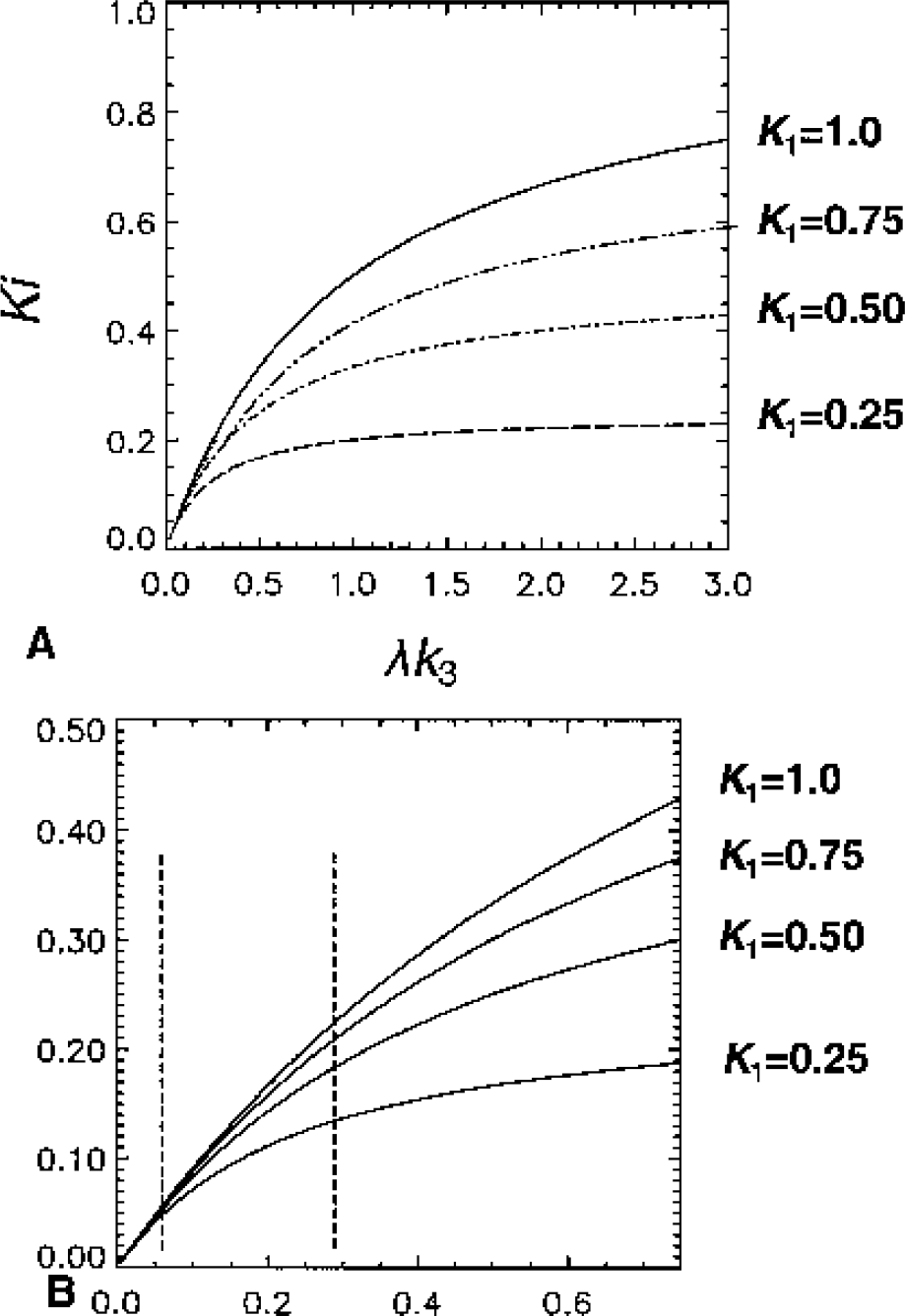
The uptake and binding of irreversible tracers such as CLG and [11C]L-deprenyl can be described by the two-tissue compartment model with three model parameters, K1, k2 and k3:
Analysis based on the direct application of these equations will be referred to as the nonlinear least squares (NLS) method. There is also a nonspecific binding component, which is assumed to be sufficiently rapid that it is incorporated into the model constants k2 and k3 (Mintun et al., 1984). With this assumption, the free ligand is a constant fraction of the free plus nonspecifically bound.
These equations can be solved explicitly through Laplace transform or numerically. The problem is to optimize the model parameters, which in this formulation requires an iterative solution using, for example, the Levenberg-Marquardt or Simplex methods (Press et al., 1990). These equations can also be written in a linearized form as (Blomqvist, 1984)
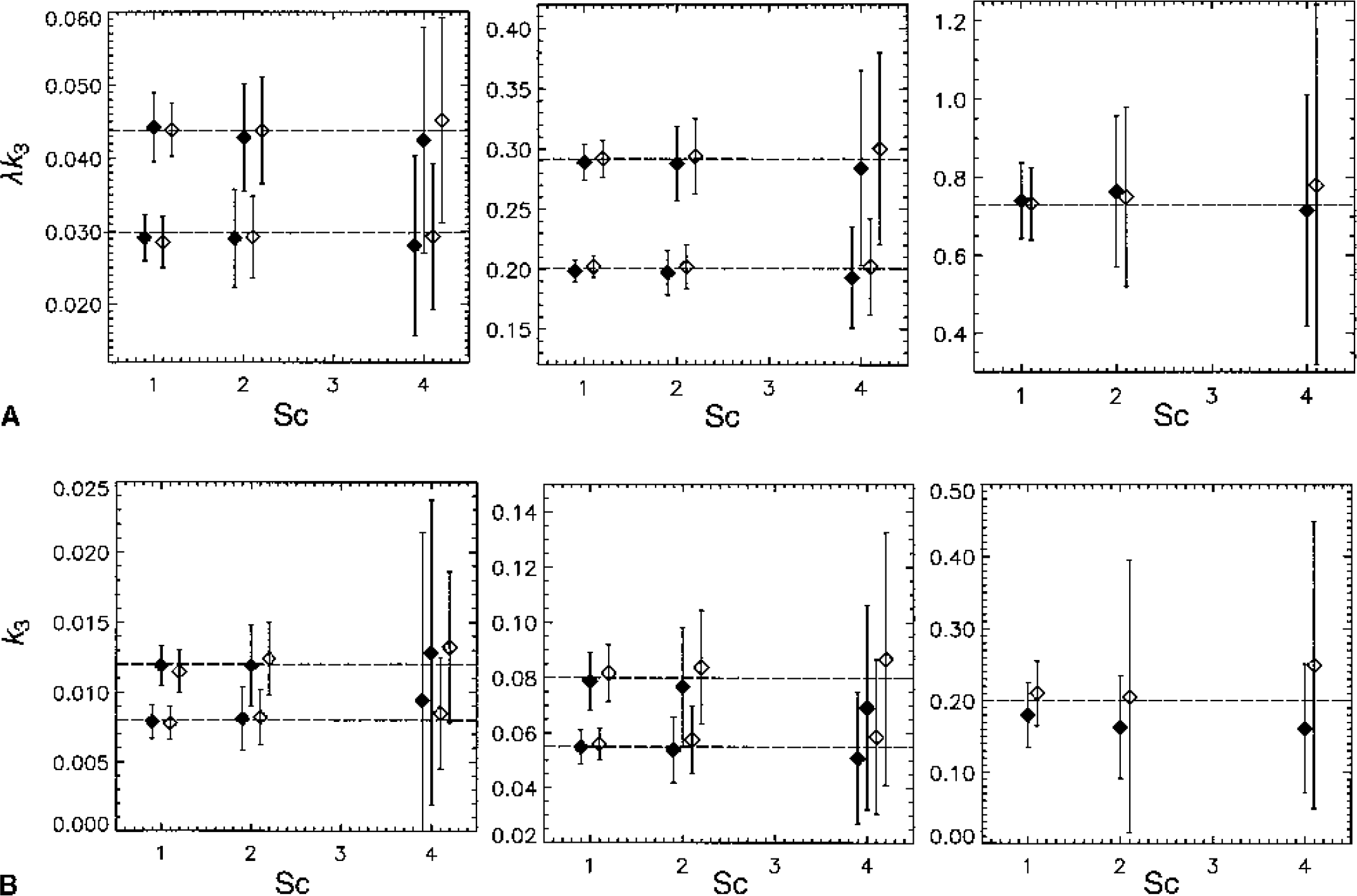
The ability to accurately estimate k3 has been found to depend on its value relative to the efflux constant k2 (Koeppe et al., 1996, 1999). When k3 » k2 (λk3 » K1), uptake depends primarily on K1 and is relatively insensitive to changes in k3 (uptake is flow limited). This can also be expressed in terms of Ki, which depends on K1 and λk3 (Ki = K1k3/(k2 + k3) = K1λk3/(K1 + λk3), where λ = K1/k2) as shown in Fig. 1A. Ki is used as a measure of uptake. Ki can also be obtained from a Patlak analysis (Patlak and Blasberg, 1985; Patlak et al., 1983) in which a plot of
A plot of Ki versus λk3 for K1 values of 0.25, 0.50, 0.75, and 1.0 mL · g−1 · min−1. The flow-limited nature of the uptake is indicated when Ki changes very little with changes in λk3.
MATERIALS AND METHODS
Computations
Solution of the differential equations Eq. 1 and optimization of model parameters were accomplished using routines in Numerical Recipes in C (Press et al., 1990). Numerical integrations in Eqs. 2, 3, and 5 were performed using the routine “qtrap” also from Numerical Recipe in C. A linear interpolation between time points was used for the concentration values associated with frame mid times for both ROIs and voxel approaches. Equal weights were used in all of the linear analyses. Parameter estimation for the NLS method was accomplished using a simplex method.
Simulations
Solution of the irreversible three-compartment model with the GLLS method was tested with simulations using transport constants K1 and k2 fixed at 0.31 mL · g−1 · min−1 and 0.085 min−1, respectively (typical of those found for CLG and CLG-D). Values of k3 used were 0.008, 0.012, 0.055, 0.08, and 0.20 min−1 to represent the spread of values over different regions applicable to both H/D clorgyline and H/D deprenyl. A measured CLG input function was used to generate the data using Eq. 1. Additive noise (dev) to simulate counting statistics based on 11C was generated using pseudo random numbers (xx) from a gaussian distribution with 0 mean and variance of 1.
Sc is a scale factor that determines the level of noise, Δti is the scan length, and C(ti) tissue radioactivity at time ti. Values used for Sc were 4, 2, and 1, ranging from a very high noise level (Sc = 4) to a low noise level (Sc = 1). At each noise level, 500 data sets were generated. Time points from t = 1 minute were used in the fitting process. The failure rate of the GLLS method increases with increasing noise. When it fails (provides negative values or the parameters exceed specified limits), the differential equations are solved directly using the NLS method but fixing the ratio λ = K1/k2 so that only two model parameters need to be determined.
Positron emission tomography studies
Preparation of CLG and CLG-D have been described previously (Fowler et al., 2001; MacGregor et al., 1988). PET scans were run on a whole body, high-resolution positron emission tomograph (Siemen's HR+ 4.5 × 4.5 × 4.8 mm at center of field of view) in 3D dynamic acquisition mode. Experimental details have been described (Fowler et al., 2001) and data reported here are from the five subjects used in those studies. Plasma samples were analyzed for CLG (or CLG-D) using the same solid phase extraction procedure described previously for deuterated [11C]L-deprenyl (Alexoff et al., 1995).
Parametric image generation
Analysis of image differences between [11C]clorgyline and deuterium-substituted [11C]clorgyline
The properties of the CLG and CLG-D images were compared by first scaling the D images such that an ROI from the thalamus (ROITH) was constrained to have the same value for both the H and D studies, that is, by scaling the D image by the factor ROITH(H)/ROITH(D). The H and scaled D images were then compared for potential differences; however, owing to the correlation between voxels, the application of statistical tests requires modification since the true number of independent data points is unknown. A value for the number of independent points was selected by reducing the number of image voxels by a factor np. Values of np from 20 to 100 were used so that for N voxels, the number of data points in the evaluation of significance was taken to be N/np, which gives 330 and 70 (for np = 20 and 100, respectively) as the number of independent data points for the thalamus plane. Images at the level of the thalamus and cerebellum were compared for differences in mean using a t-test and for different variances by means of the F-test using the reduced number of degrees of freedom (Numerical Recipes in C).
RESULTS
Simulations
Results of the simulations (n = 500) are shown in Figs. 2, 3, and 4. Figure 2A gives average values and standard deviation (SD) for λk3 versus noise level as determined by Sc values of 1, 2, and 4. Figure 2B gives results for k3 versus noise level. For clarity, symbols are shown slightly displaced from the Sc value actually used. The true values (λkT3 and kT3) are indicated by the dashed lines. For λkT3 these are 0.029, 0.044, 0.20, 0.29, and 0.73 mL · g−1 · min−1 (corresponding to kT3 = 0.008, 0.012, 0.055, 0.08, 0.20 min−1). The GLLS-NLS method slightly underestimates λk3 at the highest noise levels. The mean values from both NLS and GLLS-NLS, however, differ from the true value by less than 5%. For k3 there are larger differences; the GLLS-NLS method underestimates k3 by 20% for Sc = 4 with kT3 = 0.20 min−1. The NLS method tends to overestimate k3 with increasing noise, while the GLLS-NLS method tends towards an underestimation. The coefficient of variation (CV) for λk3 was less than for k3 as expected (Figs. 3A and 3B, respectively). The smallest CV at all noise levels occurs for λk3 = 0.20. The CVs were generally smaller for the NLS method except for Sc = 4 with kT3 = 0.20. The GLLS method failed for 20 of 500 (for kT3 = 0.008, Sc = 2), reverting to the NLS solution with λ fixed as described above. This number increased to 300 of 500 for kT3 = 0.20 (Sc = 4). As a result, the GLLS-NLS had a smaller SD (Fig. 2A) than the NLS method. This is due to the fact that when the GLLS method failed, the NLS method was implemented with λ fixed, thus reducing the number of parameters determined. Figure 4A shows the variation in mean value of λ with kT3 for Sc = 2 and Fig. 4B shows λ versus Sc for kT3 = 0.08 min−1. λ deviates from the true value as kT3 increases (Fig. 4A) and the CV increases as well (0.86 for = 0.20). λ also increases with increasing noise (Sc) (Fig. 4B). On the other hand, K1 estimates (Fig. 5) are only 3% below the true value when kT3 = 0.20 (Fig. 5). Parameter estimates of λ and k3 are negatively correlated r = −0.91 for Sc = 1 and r = −0.10 for Sc = 4 (P < 0.0005) consistent with a positive correlation between k2 and k3.
Average values and standard deviation (SD) for λk3 versus noise level (Sc) for nonlinear least squares (NLS) and generalized linear least squares (GLLS-NLS) methods. For clarity, symbols are shown slightly displaced from the Sc value actually used. Values determined by the NLS method are indicated by ⋄ and by ⧫ for the GLLS-NLS method. The dashed line indicates the true values of λkT3, which were 0.029, 0.044, 0.20, 0.29, and 0.73.
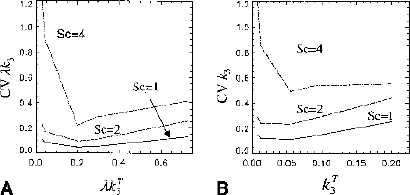
Coefficient of variation (CV) for λk3 as a function of noise level (Sc) and λkT3 for the generalized linear least squares–nonlinear least squares (GLLS-NLS) method.
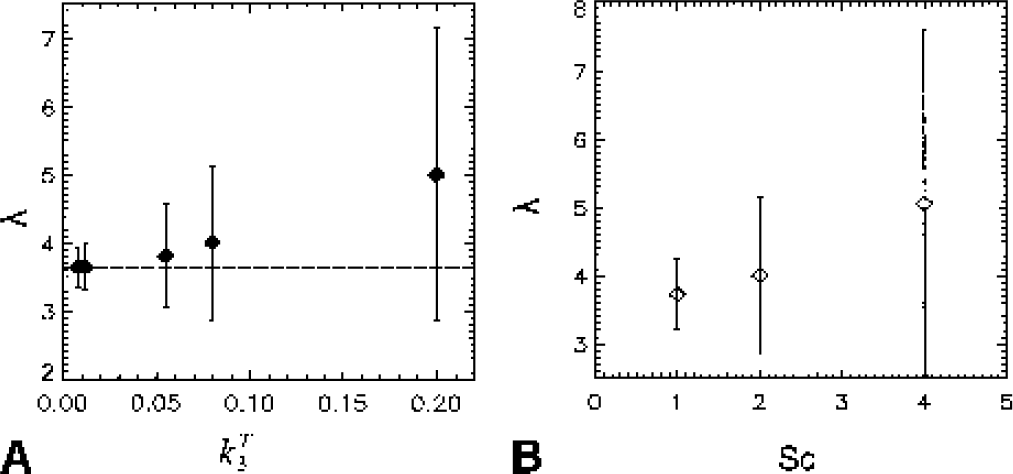
λ versus kT3 for one noise level (Sc = 2) for the generalized linear least squares–nonlinear least squares (GLLS-NLS) method. The estimated value of λ increases with increasing kT3.
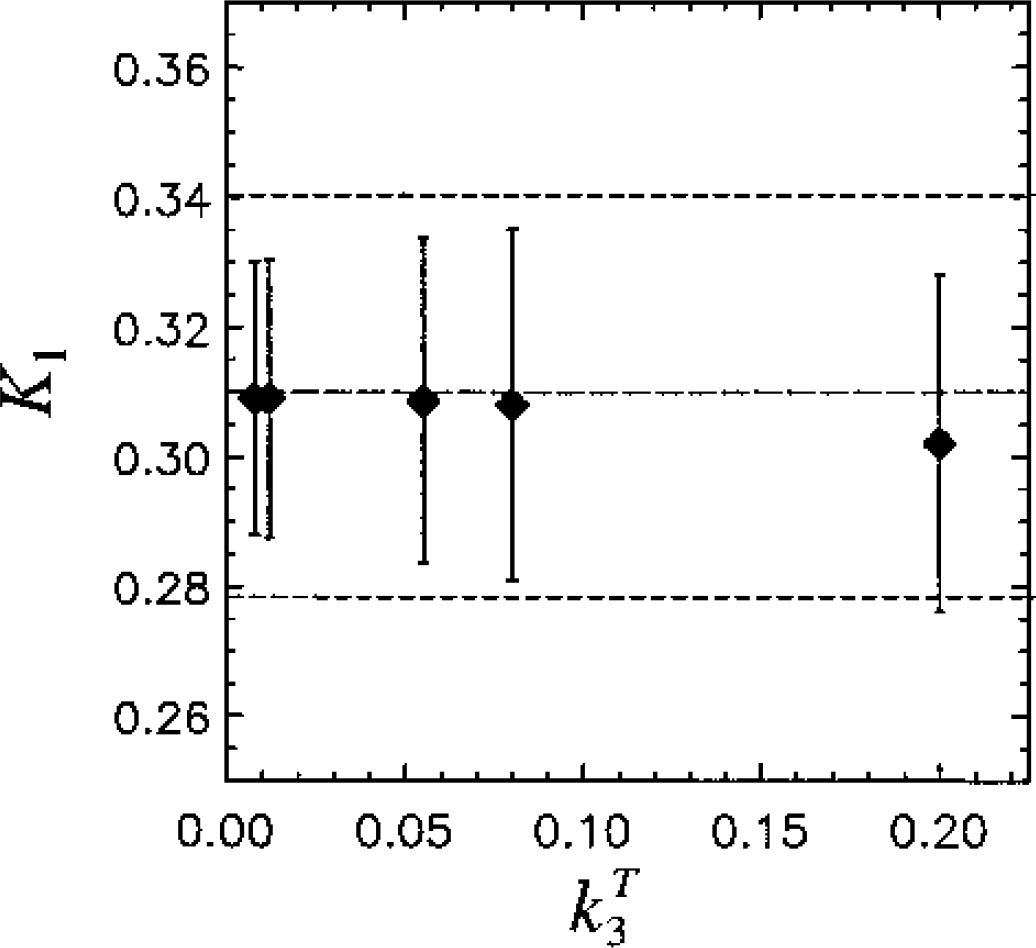
K1 versus kT3 for Sc = 2. Mean estimates of K1 deviate only slightly from the true value, kT3,as kT3 increases.
Images
Parametric images of K1 and λk3 are shown in Fig. 6 for three of the five subjects. Two planes are shown: one at the level of the thalamus and one at the level of the cerebellum. The λk3 images from CLG and CLG-D are displayed on the same scale by multiplying the deuterium images by the ratio of a thalamus region of interest for CLG to that of CLG-D for each subject. The λk3 images for CLG-D show less contrast than those of CLG. The K1 images are not scaled and are similar for both compounds with no loss of contrast in the D images. These images were generated by grouping voxels within a region of ±4 voxels and within 2.5% of the integrated image except in regions of low uptake. When the value of the scaled integrated images was <55, the acceptance was increased to ±5% to compensate for low counts. The average number of grouped voxels was in the range of 15 to 25. The number of voxels for which the NLS method was used varied considerably depending on the plane (related to count rate) and study. For example, planes at the level of the thalamus required the NLS method for −1.5% or less of the voxels while at level of the cerebellum this increased to 5% to 10%. Using the integrated image in two parts did not improve the image and required larger limits on number of voxels and acceptance region, ±5 voxels and 5.5%, respectively. The values with the two-part integration were within ±2% for the thalamus and cerebellum ROIs. The important point is that a sufficient number of voxels are required in order to generate reasonable λk3 images, and this number is larger in regions of lower counts.
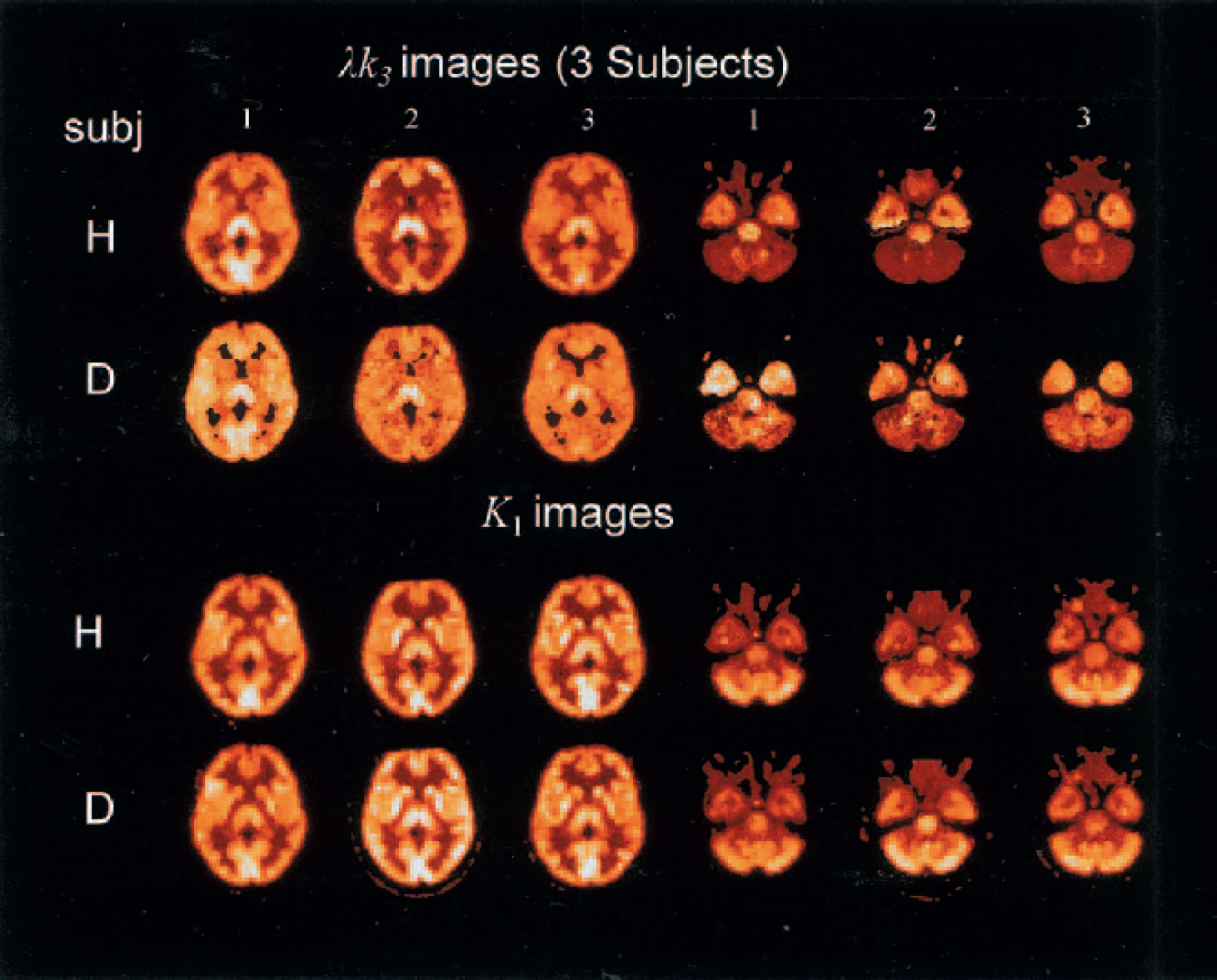
Parametric images of K1 and λk3 for three of the five subjects. Two planes are shown, one at the level of the thalamus (left) and one at the level of the cerebellum (right). The λk3 images from [11C]clorgyline (CLG) and deuterium-substituted [11C]clorgyline (CLG-D) are displayed on the same scale bymultiplying the deuterium images by the ratio of a thalamus region of interest for CLG to that of CLG-D for each subject. The λk3 images for CLG-D show less contrast than those of CLG. The K1 images are not scaled and are similar for both compounds with no loss of contrast in the CLG-D images.
Figure 7 illustrates a histogram of the λk3 values from the plane including the thalamus for all five subjects for the H and scaled D. The images were constrained to include the same number of voxels by using the same voxels from each image for which both exceeded a cutoff value. The average, median, and SD are given. The distribution of values in the thalamus (TH) plane appears to be broader for CLG than for the scaled CLG-D, which accounts for the greater contrast. For the cerebellum (CB; images on the right in Fig. 6) the distributions of λk3 for CLG-D are similar in width but with a higher median and average values than for CLG (data not shown). A t-test on the difference in means between CLG and scaled CLG-D for the TH plane found a significant difference in all cases but 1 for np = 100 (df = 70, P < 0.05, t ranged from −4.1 to −2). For three of five subjects, the F-test for differences in variances found CLG distribution significantly greater than the scaled CLG-D for np = 100 (P < 0.005, F ranged from 2.1 to 1.4). For the CB plane, a significantly greater mean for scaled CLG-D occurred in four of 5 cases for np = 50 (df = 75, P < 0.05, t ranged from −6 to −2). There was a significantly greater variance for CLG in 3 of the 5 subjects for np = 50 (df = 75, P < 0.05, F = 1.5 for all 3). The average difference in mean between scaled CLG-D and CLG was greater for CB image than for TH image, 0.241 ± 0.18 (CB) and 0.095 ± 0.13 (TH). In contrast, no significant difference in mean or variance was found for H and D distributions of K1 for np = 75.
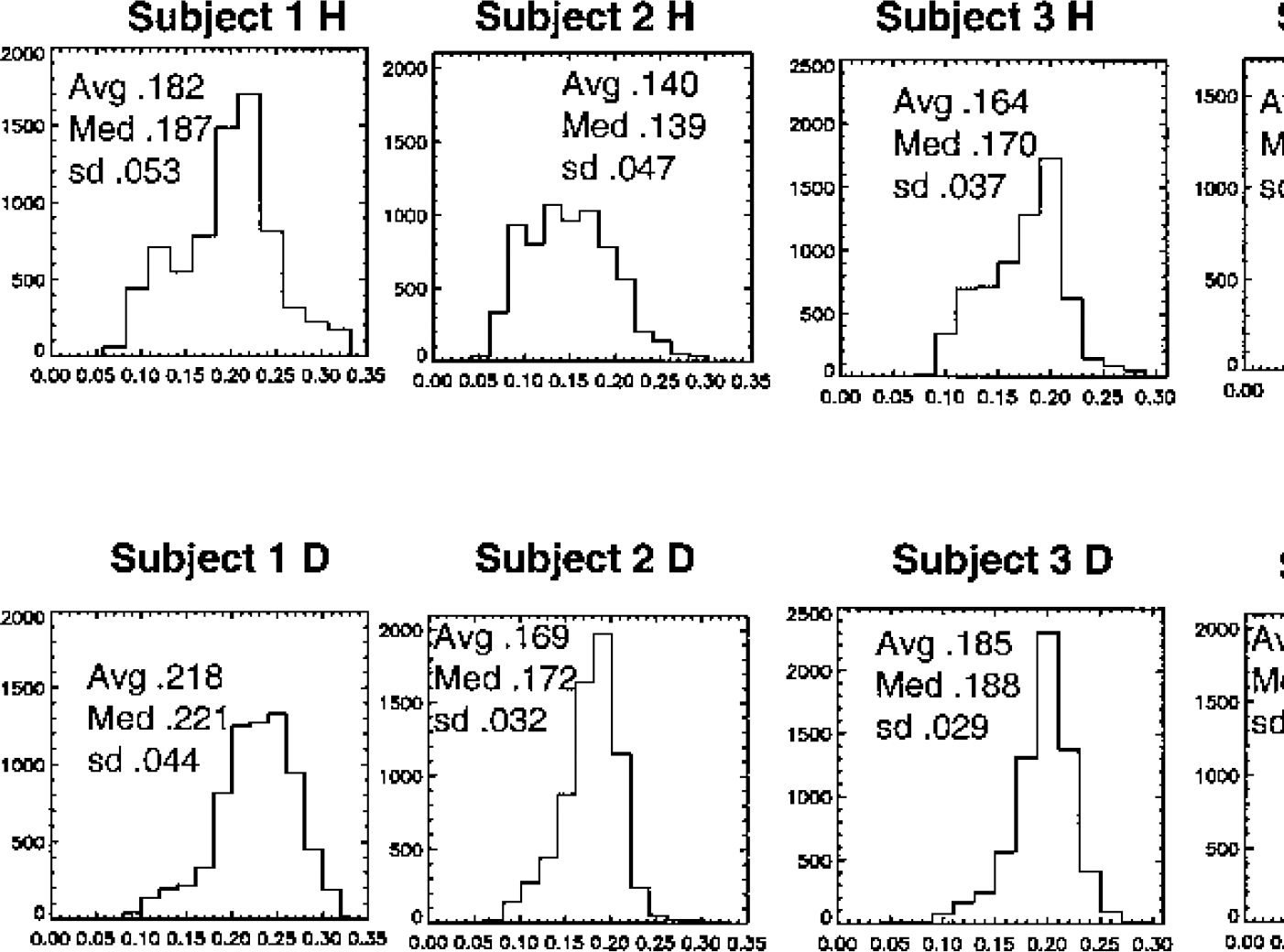
Histograms of λk3 from a plane containing the thalamus for [11C]clorgyline (CLG) and scaled deuterium-substituted [11C]clorgyline (CLG-D) for all five subjects. Images were constrained to have the same number of voxels. Note the broader distribution of values for CLG than for CLG-D. The average (Avg), median (Med), and standard deviation (sd) are shown.
Figures 8A and 8B illustrate the average CV, determined from the average and SD of voxels from the thalamus and cerebellum regions, for λk3 and k3 over all five subjects. Calculations of λk3 and k3 used both the SEV (the signal-enhanced voxel) and single voxel methods. The GLLS-NLS estimation method was used for both the SEV and single voxel, and for comparison the single voxel NLS estimation was also included. The average value from the SEV method (data not shown) was in good agreement with the traditional ROI value (using NLS). The CVs were considerably less for the SEV calculation of Xk3 compared with the single voxel calculation (P < 0.05, paired t-test). The CV for CLG was less than that for CLG-D and the CV for thalamus was less than that for cerebellum. For k3, the SEV CVs are considerable greater than for λk3 (P < 0.05, paired t-test). For CB using the single voxel GLLS-NLS method, the average value for λk3 was 6% less than that calculated using the ROI method for CLG-D and 4% less for CLG. For THL, the GLLS-NLS single voxel method was 3% less for CLG-D, with no difference found for CLG.
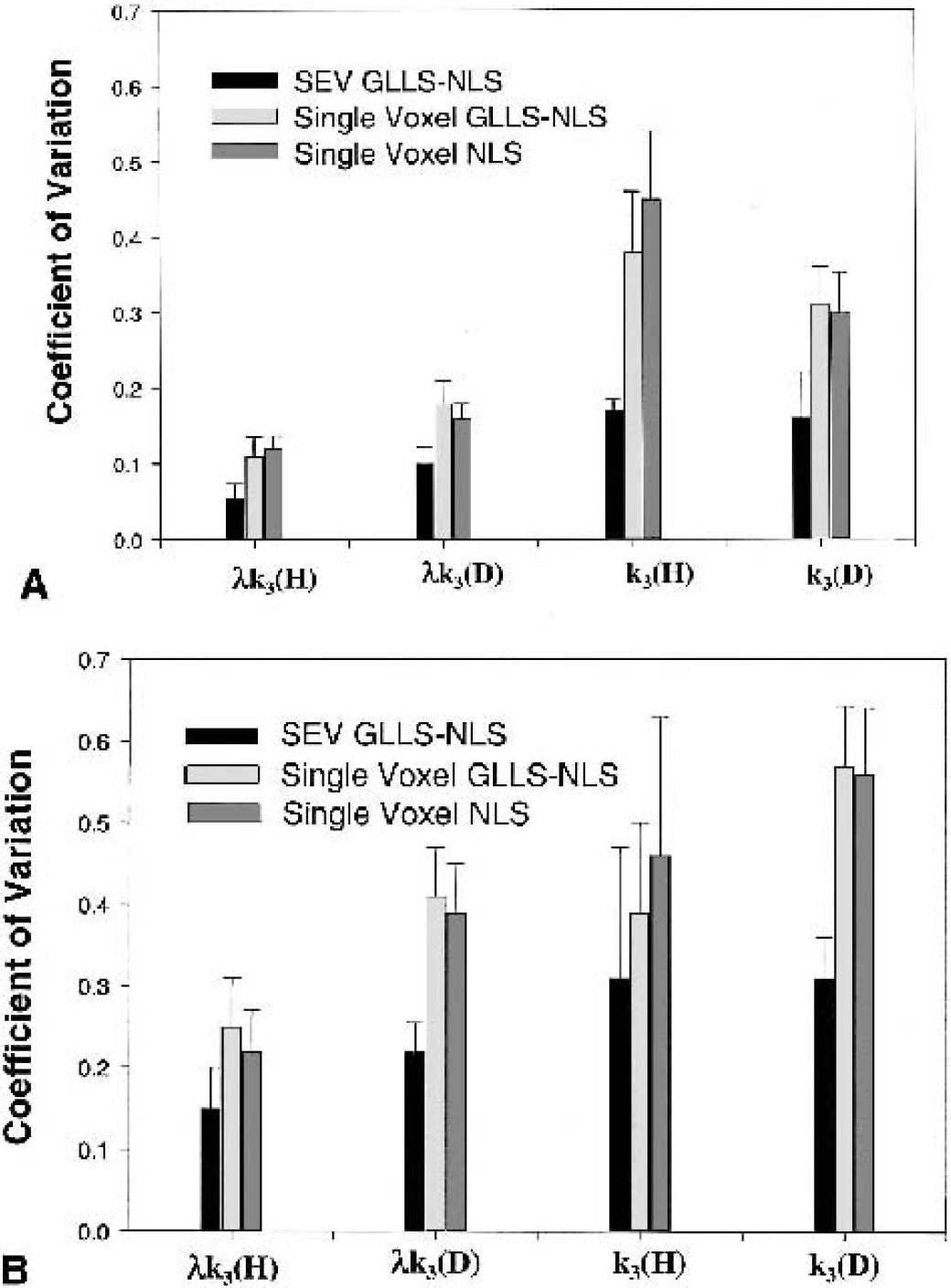
Coefficient of variation (CV) of λk3 and k3 from the thalamus region for the signal-enhanced voxel (SEV) and single voxel methods. The SEV method refers to the signal enhancement procedure applied to each voxel in the defined region of interest. The generalized linear least squares-nonlinear least squares (GLLS-NLS) method was used. For comparison, the CV from calculations based on a single voxel approach (no enhancement) for both GLLS-NLS and NLS are also presented. The average value for the thalamus region using the SEV method (data not shown) was in good agreement with the traditional ROI value. The CVs were considerably less for the SEV calculation of λk3, with the CV for CLG less than that for CLG-D. For k3 the CVs are considerable greater than for λk3.
Although it is not possible to directly relate the noise level in these images to that used in the simulations, a comparison of the quantity (averaged over time points t) given by
DISCUSSION
The simulation results indicate that the accurate estimation of the enzyme parameter depends on multiple factors. One factor is the amount of tracer in the tissue and the half-life of the isotope. Another is the sensitivity of the uptake curve to variations in the amount of enzyme, as illustrated in Fig. 1. The greatest sensitivity occurs for lower values of enzyme, but accuracy of parameter estimation is also dependent on having sufficient tracer in tissue to achieve a signal. The largest CVs were obtained for the lowest values of k3 (Fig. 3) at the highest noise level. At the high end when λk3 » K1 (or equivalently k3 » k2), uptake depends primarily on K1 and is relatively insensitive to changes in k3 or λk3, (the “flow-limited” case). The largest value of λk3 used in the simulations was 0.73 (k3/k2 = λk3/K1 = 2.4). From Fig. 1 this region is of lower sensitivity than λk3 = 0.20. This places some limits on the range of enzyme concentration that a particular tracer can accurately estimate. For deprenyl, K1 has values close to what would be expected for blood flow (0.60 mL · g−1 · min−1), and λk3 is on the order of 0.5 (white matter) to 2.0 or greater in thalamus, which is a region of lower sensitivity since λk3/K1 = 3.3 (Fig. 1A). The introduction of the deuterium (D deprenyl) reduces λk3 so that the maximum values are on the order of 0.4 to 0.5 with minimum values in the range 0.12, which puts λk3 in a region of greater sensitivity since λk3/K1 < 1 (Fig. 1B). For CLG the λk3/K1 ratio is on the order of 0.8 for thalamus and 0.45 for cerebellum. For CLG-D these ratios are 0.25 and 0.17 for thalamus and cerebellum, respectively. Koeppe et al. (1999) report that optimum values of λk3/K1 for the accurate estimation of k3 range from 0.1 to 0.3. From our simulations, the optimum value was 0.6 for k3 = 0.055 (λk3 = 0.20), for which the CVs for both λk3 and k3 go through a minimum (Fig. 3). The true minimum may actually occur between the values k3 = 0.055 and 0.012 (corresponding to λk3/K1 = 0.14). For larger values of λk3/K1, the effect of noise on parameter estimation is much greater. Note the larger CVs for λk3 = 0.73 (λk3/K1 = 2.35) than for λk3 = 0.20. Also at higher noise levels and larger values of kT3, the accurate estimation of k2 and k3 is much more difficult. As illustrated in Fig. 4A, there is a positive correlation between λ and kT3 (Sc = 2) so that the average value of λ increases with increasing kT3. The CV estimates of λ also increase with noise when kT3 is held constant. K1 values, on the other hand, show only a small decrease (<5%) as kT3 increases (Fig. 5). The combination parameter λk3 eliminates this correlation problem.
The range of values found for λk3 previously reported for deprenyl (Fowler et al., 1995) seem to be consistent with experimental measurements of MAO B from postmortem studies. Ratios of between 2.5 and 3 were estimated for the thalamus-to-cerebellum and basal-ganglia-to-cerebellum ratios (Fowler et al., 1995) based on data from Fowler et al. (1980), Glover et al. (1980), and Oreland et al. (1983). Values for MAO A appear to be in the same range. In comparison, a much greater difference is seen for the enzyme acetylcholinesterase (AchE), for which the experimentally measured trapping rates differ by 20 to 30 between the highest and lowest regions (Atack, 1986; Reinikainen et al., 1988). Koeppe et al. (1999) used the tracer PMP to measure AchE levels and found that the range of values across regions for k3 depended on the method used in the estimation procedure, a result of the loss of sensitivity at higher enzyme levels. An unconstrained procedure for estimating all three parameters gave estimates for the range of k3 values far lower than a constrained estimation in which k3 was calculated as λk3/4. This assumes that the true value of λ is 4 and constant across the brain. This last procedure is analogous to the use of the combination parameter λk3. The unconstrained procedure underestimated k3 in regions of high enzyme activity, whereas the constrained procedures gave values much closer to the reported range of enzyme activity, which lends support for the use of λk3 over k3.
Parametric images of k3 for CLG and CLG-D, in addition to being considerably more noisy than images of λk3, do not show the higher enzyme level characteristic of the MAO A in the thalamus, most likely because of the correlation problem between k2 and k3 (and the amount of noise since the ROI data show a greater amount of binding in thalamus). Aside from eliminating the correlation problem, a second reason for using λk3 is that it does not depend on nonspecific binding. Since the nonspecific binding is incorporated into the constants k2 and k3, the dependence of λk3 on nonspecific binding has been removed since it appears in both k2 and k3 and therefore cancels (Carson et al., 1997). However, λk3 is dependent on plasma protein binding (which is also rapid, similar to nonspecific binding) through K1. There is some indication that λ is somewhat larger for CLG-D than for CLG. Using a global ROI λD is greater than λH with an average ratio of 1.15 (D/H). Also λ for CB is somewhat larger than for TH (based on ROI data from Fowler et al., 2001) for CLG-D. The average ratio is 1.12 (CB/TH). For CLG the difference is less. These regional differences may be due to small differences in nonspecific binding, in which case λk3 is a better measure, since it does not depend on nonspecific binding.
Since the λk3/K1 ratios for CLG and CLG-D appear to be fairly close to the optimal range, this ensures adequate sensitivity of the model parameters to variations in enzyme level. The other factor contributing to the quality of the images is the noise associated with low counting statistics at the voxel level. Based on the simulations, the accuracy of parameter estimation and coefficients of variation of the values are improved by a reduction in noise. Also, because the GLLS method is a linear method, it is more sensitive to noise than the NLS method. The strategy of combining local voxels based on proximity and whether they fall within a specified range of the integrated voxel value improves the counting statistics. Furthermore, by meeting both criteria, the assumption is that the grouped voxels are likely to be functionally related. Although this will tend to smear out the signal, it reduces noise. Voxel grouping, along with the combination of the GLLS method with the NLS method as described, allows the rapid generation of reasonably good-quality images of the model parameters λk3 and K1, even though the injected radioactivity averaged only 6 to 7 mCi. For comparison, Koeppe et al. (1999) injected 16 to 32 mCi when studying the irreversible tracer [11C]PMP. Other strategies have been used for grouping voxels in order to improve the signal; for example, Kimura et al. (1999) have used a cluster analysis procedure averaging over voxels with similar concentration histories in a two-parameter model.
Figure 6 illustrates the loss of contrast in the CLG-D λk3 images compared with the CLG images. This is presumably due to a slow nonspecific binding component that cannot be separately evaluated apart from the MAO A binding and that is not diminished with D substitution. A rapid nonspecific binding component would be incorporated into the parameters λ (in k2) and k3 as the nonspecific binding fraction. In that case, it would not enter into λk3. The fact that λ is slightly higher for CLG-D would be consistent with a small increase in the rapid nonspecific binding. In spite of the fact that the images appear different, SPM analysis (SPM software MRC Cyclotron Unit, Hammersmith Hospital, London, 1995) of the two groups, the CLG versus the scaled CLG-D, did not find specific areas of significant difference, although differences were detected in the ROI analysis. As an alternative means of comparing the CLG and CLG-D images, histograms of λk3 values corresponding to planes at the level of TH and CB were used. The histograms of Fig. 7 for CLG and scaled CLG-D images illustrate the generally broader distribution of λk3 for the CLG images at the plane including the thalamus. The broader distribution is consistent with the increased contrast, whereas the CLG-D distributions are more peaked and narrower, consistent with a smaller range of values, and therefore have less contrast. For subjects 1, 2, and 3, the variance of the CLG distribution was significantly greater than for the scaled CLG-D distribution for a reduction factor (np) of 75 for which there would be 90 independent data points in the image (there are on the order of 7,000 voxels per image). The difference in variance is less for subjects 4 and 5. In general, subjects 1, 2, and 3 showed greater differences between CLG and CLG-D images than subjects 4 and 5. Also, the mean for the scaled CLG-D image is greater than for the CLG image in all subjects except 4. Since the scaling was based on the thalamus, which is one of the regions with the greatest amount of MAO A, this indicates that the reduction in binding for other regions is less. Similarly for the cerebellum plane, the scaled CLG-D distributions have larger averages than the CLG. In comparison, there is no significant difference in variance for the K1 images for any of the five subjects.
There is some question as to whether this additional binding is uniform. It certainly forms a larger fraction of the binding in the cerebellum (and white matter) than in the thalamus, which has a greater amount of MAO A. The relative contributions can only be calculated if the true isotope reduction factor is known. The non-MAO contribution can be calculated as (Fowler et al., 2001)
The nonuniform reduction in binding reported here was first observed visually on the time-summed images and confirmed in a ROI analysis of the data. In this work we have extended the analysis to parametric images of λk3 and analyzed differences in the distribution of values of the model parameter in two planes involving the thalamus and the cerebellum. This required developing a technique to increase the signal-to-noise ratio in these experiments with low injected radioactivity. The simple strategy of combining local voxels with similar integrated activity increased the signal and allowed the use of the GLLS method. When the GLLS method failed, the NLS method with λ fixed was used. This occurred more frequently in regions of low uptake. Taken together, these procedures allowed the generation of images of the combination parameter λk3, which is a measure of enzyme concentration.
Footnotes
Acknowledgments:
The authors thank Robert Carciello, Colleen Shea, Robert MacGregor, Victor Garza, Donald Warner, Noelwah Netusil, and Pauline Carter for advice and assistance in performing these studies. We also thank the people who volunteered for these studies.