Abstract
Individual inhibition of nitric oxide (NO) synthase and cytochrome P450 (CYP) epoxygenase activity attenuates cortical functional hyperemia evoked by whisker stimulation. The objectives of the present study were to determine (1) if administration of epoxygenase inhibitors attenuates cortical functional hyperemia by using a different modality of sensory activation (i.e., electrical stimulation of the rat forepaw), (2) if epoxygenase inhibition has an additive effect with NO synthase inhibition on the flow response, and (3) the cellular localization of the epoxygenase CYP2C11 in cerebral cortex. In six groups of anesthetized rats, the cortical surface was superfused for 90 minutes with (1) vehicle; (2) 1-mmol/L Nω-nitro-L-arginine (L-NNA), to inhibit NO synthase activity; (3) 20-μmol/L N-methylsulfonyl-6-(2-propargyloxyphenyl)hexanamide (MS-PPOH), a substrate inhibitor of P450 epoxygenase; (4) MS-PPOH plus L-NNA; (5) 20-μmol/L miconazole, a reversible inhibitor at the heme site of P450 epoxygenase; and (6) miconazole plus L-NNA. The percent increases in laser-Doppler perfusion over primary sensory cortex during 20-second forepaw stimulation were reduced by 44% to 64% in all drug-treated groups. The addition of L-NNA to MS-PPOH produced no additional reduction (64%) compared with MS-PPOH alone (64%) or L-NNA alone (60%). The addition of L-NNA to miconazole also produced no additional reduction in the flow response. In situ hybridization of CYP2C11 mRNA showed localization in astrocytes, including those adjacent to blood vessels. Thus, activity of both epoxygenase, presumably localized in astrocytes, and NO synthase is required for generating a complete cortical hyperemic response evoked by electrical forepaw stimulation. The lack of additional blood flow attenuation with the combination of the NO synthase and the distinct epoxygenase inhibitors suggests that the signaling pathways do not act in a simple parallel fashion and that other mediators may be involved in coupling cortical blood flow to neuronal activation.
Dynamic control of cerebral blood flow (CBF) to neuronal activation is a highly integrated response involving multiple signaling molecules. In experimental models using whisker stimulation, increases in CBF in whisker barrel cortex are attenuated by adenosine antagonists (Dirnagl et al., 1994), nitric oxide synthase (NOS) inhibitors (Dirnagl et al., 1993; Irikura et al., 1994; Ma et al., 1996), soluble guanylyl cyclase inhibition (Lindauer et al., 1999), cyclooxygenase-2 inhibition and gene deletion (Niwa et al., 2000), and antagonists of calcium-activated potassium (KCa) channels (Gerrits et al., 2002). Pial arteriolar dilation during sciatic nerve stimulation also is attenuated by an adenosine antagonist (Ko et al., 1990). However, the magnitude of the attenuation of the vascular response in all of these studies was approximately 40% to 50%. Even with combined use of an adenosine antagonist and a NOS inhibitor, approximately 40% of the baseline CBF response to whisker stimulation is sustained (Dirnagl et al., 1994). Thus, there may be other mechanisms involved in mediating functional hyperemia.
Epoxyeicosatrienoic acids (EETs) represent a set of lipid-signaling molecules that appear to be involved in the coupling of CBF to neuronal activation. EETs are synthesized from arachidonic acid by specific cytochrome P450 (CYP) enzymes possessing epoxygenase activity (Roman, 2002). One such epoxygenase, CYP2C11, has been identified in cultured rat astrocytes (Alkayed et al., 1996b). Application of glutamate to cultured astrocytes increases EETs in the astrocytes and media, and the increase is inhibited by the epoxygenase inhibitor miconazole (Alkayed et al., 1997). Likewise, the cortical CBF response to glutamate (Alkayed et al., 1997) and the striatal CBF response to N-methyl-D-aspartate (Bhardwaj et al., 2000) are reduced by epoxygenase inhibitors such as miconazole acting at CYP heme and by N-methylsulfonyl-6-(2-propargyloxyphenyl)hexanamide (MS-PPOH), a selective epoxygenase substrate inhibitor (Wang et al., 1998). More recently, we reported that miconazole and MS-PPOH reduce the CBF response to whisker stimulation (Peng et al., 2002). EETs can dilate cerebral arteries (Ellis et al., 1990; Gebremedhin et al., 1992; Leffler and Fedinec, 1997) by a mechanism involving the opening of KCa channels (Alkayed et al., 1996b; Gebremedhin et al., 1992). In cortical brain slices, afferent stimulation leads to increases in astrocyte calcium and subsequent arteriolar dilation by a mechanism dependent on activation of metabotropic glutamate receptors (Zonta et al., 2003). Based on this evidence, we postulate that astrocytes may serve to integrate local neuronal activity by activation of astrocytic metabotropic glutamate and possibly other receptors, leading to increased calcium, mobilization of arachidonic acid, and release of EETs from astrocyte processes, which then act to open KCa channels on cerebral arterioles (Harder et al., 1998; Harder et al., 2002).
The objectives of the present study were threefold. First, cell-specific expression of CYP2C11 was examined in rat cerebral cortex by in situ hybridization. Second, the role of epoxygenase activity was evaluated in a different model of functional activation using electrical stimulation of the forepaw to determine if MS-PPOH and miconazole are effective in reducing functional hyperemia in a different region of somatosensory cortex with a different modality of activation. Electrical forepaw stimulation has been characterized as a model of functional hyperemia (Ances et al., 2000, 2001a,b; Silva et al., 2000). Third, the effect of combined inhibition of epoxygenase and NOS was investigated to determine whether there is an additive effect of inhibiting epoxygenase and NOS pathways on the reduction of the CBF response to forepaw stimulation. One possibility is that nitric oxide (NO) derived from neurons during activation acts strictly on vascular smooth muscle to cause dilation by a mechanism independent of an epoxygenase mechanism based in astrocytes. Alternatively, NO and EETs may converge on common mechanisms that interact at the level of vascular smooth muscle or astrocytes.
MATERIALS AND METHODS
Nonradioactive in situ hybridization
A unique fragment of CYP2C11, corresponding to 1,502 to 1,840 bp of published CYP2C11 cDNA sequence, was isolated from a full-length CYP2C11 cDNA with primers 5'-GAAAAGCACAATCCGCAGTCTGA-3' and 5'-AGCCTCGTAAAAAGCGGGCAAAT-3' by polymerase chain reaction and was subcloned into the EcoR1 site of plasmid pCRII, which contained phage T7 and sp6 promoters. The Protein and Nucleotide Facility at the Medical College of Wisconsin sequenced insert DNAs. Antisense and sense riboprobes for CYP2C11 were generated by in vitro transcription, using sets of restriction enzymes and T7 or Sp6 RNA polymerases, and labeled with digoxigenin according to the manufacturer's instructions (Roche Molecular, Indianapolis, IN, U.S.A.).
Frozen 10-μm sections of adult male rat cortex were cut on a cryostat and fixed with 4% paraformaldehyde. After wash with 0.1-mol/L phosphate buffer (pH 7.4), sections were treated with triethanolamine plus 0.25% acetic anhydride acid (v/v) for 10 minutes. Sections were then washed for 5 minutes in 2xSSC (1xSSC contains 150-mmol/L NaCl and 15-mmol/L sodium citrate, pH 7.0). The sections were placed in a humidified chamber, covered for 30 minutes at 50°C with 50 μL prehybridization solution (containing 50% formamide, 5xSSC, 5xDenhardt's reagent, and 100 μg/mL denatured sheared herring sperm DNA), and probed with the labeled riboprobes (30 ng/mL) prepared in fresh hybridization solution for 18 hours at 55°C. The sections were washed three times with 2xSSC at room temperature and incubated in 10-mmol/L Tris buffer (pH 8.0) containing 0.5-mol/L NaCl and 30 μg/mL RNase A for 60 minutes at 37°C to destroy RNA:DNA hybrids and single-strand RNA. The sections were rinsed with 2xSSC, 1xSSC, and 0.5xSSC sequentially for 5 minutes at room temperature and then washed with 0.1xSSC for 60 minutes at 70°C.
The sections were washed with 0.1-mol/L phosphate buffer and blocked in carrageenan mix (containing 0.25% carrageenan and 0.3% Triton X-100 in 0.1-mol/L phosphate buffer) for 1 hour at room temperature. A digoxigenin-labeled CYP2C11 probe was localized by incubation with anti–digoxigenin-POD (horseradish peroxidase-conjugate antibodies; Roche Molecular) diluted at 1:100 in blocking mix followed by wash in phosphate-buffered saline (pH 7.4) and TSA Plus alkaline phosphatase deposition (NEN Life Science Products, Boston, MA, U.S.A.) according to the manufacturer's protocol. Immunoreactivity was visualized by treating sections with the alkaline phosphatase substrate BCIP and nitroblue tetrazolium for 1 to 3 hours in darkness at room temperature. Some sections were continuously immunolabeled with glial fibrillary acidic protein (GFAP), an astrocyte marker, using antibodies against GFAP (Chemicon, Temecula, CA, U.S.A.), followed by secondary antibodies conjugated with tetramethylrhodamine isothiocyanate (TRITC). Sections were viewed and photographed with a Nikon Eclipse 600 microscope (Nikon Inc., Melville, NY, U.S.A.), equipped for both light and epifluorescence and connected with a digital camera.
Electrical forepaw stimulation
The Johns Hopkins University Animal Care and Use Committee approved all procedures. Male Wistar rats weighing approximately 300 g were anesthetized with isoflurane during the surgical procedure. The lungs were ventilated via a tracheostomy with approximately 25% O2 and 1% to 1.5% isoflurane, adjusted to prevent muscle movement. A femoral artery and vein were catheterized, and mean arterial blood pressure was monitored. Rectal temperature was maintained near 37°C with a warm blanket and heating lamp. Arterial pH, PCO2, and PO2 were measured with a blood gas analyzer (Chiron Diagnostics, Halstead, Essex, U.K.), and hemoglobin concentration was measured with a hemoximeter (OSM3, Radiometer, Copenhagen, Denmark).
An approximate 3 × 3-mm region of skull was thinned by careful drilling for placement of a laser-Doppler flow (LDF) probe (Moor Instruments, Oxford, U.K.) over the left somatosensory cortex in the vicinity of the forepaw representation (approximately 2–4 mm lateral and 1 mm anterior to bregma). All inhibitor drugs were superfused subdurally over the cortical surface through a PE-10 catheter tapered to approximately 200 μm and inserted approximately 1 mm through a small incision in the dura, as described elsewhere (Alkayed et al., 1997; Peng et al., 2002). This catheter was situated approximately 3 mm dorsal and posterior to the flow probe site, with the catheter tip directed at the recording site. A second incision in the dura was made approximately 2 or 3 mm ventral and anterior to the recording site for passive drainage of cerebrospinal fluid (CSF) during superfusion. Superfusion of dye indicated coverage over the cortical surface underneath the recording site. Isoflurane was discontinued and anesthesia was maintained with μ-chloralose (50 mg/kg, intraperitoneal, plus 40 mg/kg per hour, intravenous) for the remainder of the experiment. One hour after discontinuing isoflurane inhalation, artificial CSF (Peng et al., 2002) was infused through this subdural catheter at a rate of 5 μL/min. The superfusion continued with either vehicle or a single drug regimen in six groups of rats: (1) artificial CSF with a 0.5% ethanol used as a vehicle for MS-PPOH and miconazole (n =7); (2) 1-mmol/L Nω-nitro-L-arginine (L-NNA; n = 6) to inhibit NOS activity (Irikura et al., 1994); (3) 20-μmol/L MS-PPOH (n = 6); (4) 20-μmol/L MS-PPOH plus 1-mmol/L L-NNA (n = 7); (5) 20-μmol/L miconazole (n = 8); and (6) 20-μmol/L miconazole plus 1-mmol/L L-NNA (n = 8).
The right forepaw was stimulated with 150-μs pulses of 2 mA at a rate of 2.9/s for a 20-second period through subcutaneous electrodes. Previous work showed that somatosensory evoked potentials are reproducible over 2 hours with these stimulation parameters (Peng et al., 2002). LDF was averaged over 1-second intervals for a 60-second baseline recording period, 20 seconds of stimulation, and a 60-second recovery period. For each animal, time-gated averaging of the LDF response was performed on three trials approximately 3 or 4 minutes apart after 90 minutes of drug superfusion. This exposure duration is sufficient for inhibiting cortical NOS activity and functional hyperemia (Irikura et al., 1994). The average percent change from the 60-second baseline for each animal was used for determining the mean and standard deviation of the group on a second-by-second basis (n = number of rats). Mean arterial blood pressure was also recorded and averaged at 1-second intervals during the stimulation period.
Analysis of variance and the Dunnett test were used to compare physiologic parameters with the CSF control group. The percent change in LDF averaged over the 20-second stimulation period for each rat was subjected to analysis of variance, and comparisons among the six groups were made with the Newman-Keuls multiple range test. A significance level of P>0.05 was used. Data are presented as means ± SD.
RESULTS
Expression of CYP2C11 mRNA in rat cortex
The generated riboprobe corresponded to a 200-bp fragment at the 3' coding region of CYP2C11 cDNA, a most unique area in CYP2C11. Its sequence has 98% identity to the published CYP2C11 sequence in the gene bank. It only matches the sequence of CYP2C11 and has very low homology to any other sequence, allowing detection of a signal specific to CYP2C11. The labeling was only detectable with TSA Plus (NEN Life Science Products), a system developed to work in combination with the conventional in situ hybridization protocol to enhance sensitivity. The pattern of expression resembled that of glial cells. Double-labeled rat brain cortical sections revealed that virtually all CYP2C11 mRNA (Fig. 1A) was localized in GFAP-immunoreactive astrocytic cell bodies (Fig. 1B). CYP2C11 mRNA could also be detected in astrocyte next to vessels (Fig. 1A) onto which astrocytes send foot processes (Fig. 1B). The expression of CYP2C11 mRNA could not be detected in astrocyte processes or in endothelial cells. Sections exposed to the sense riboprobe showed no staining (Fig. 1C).
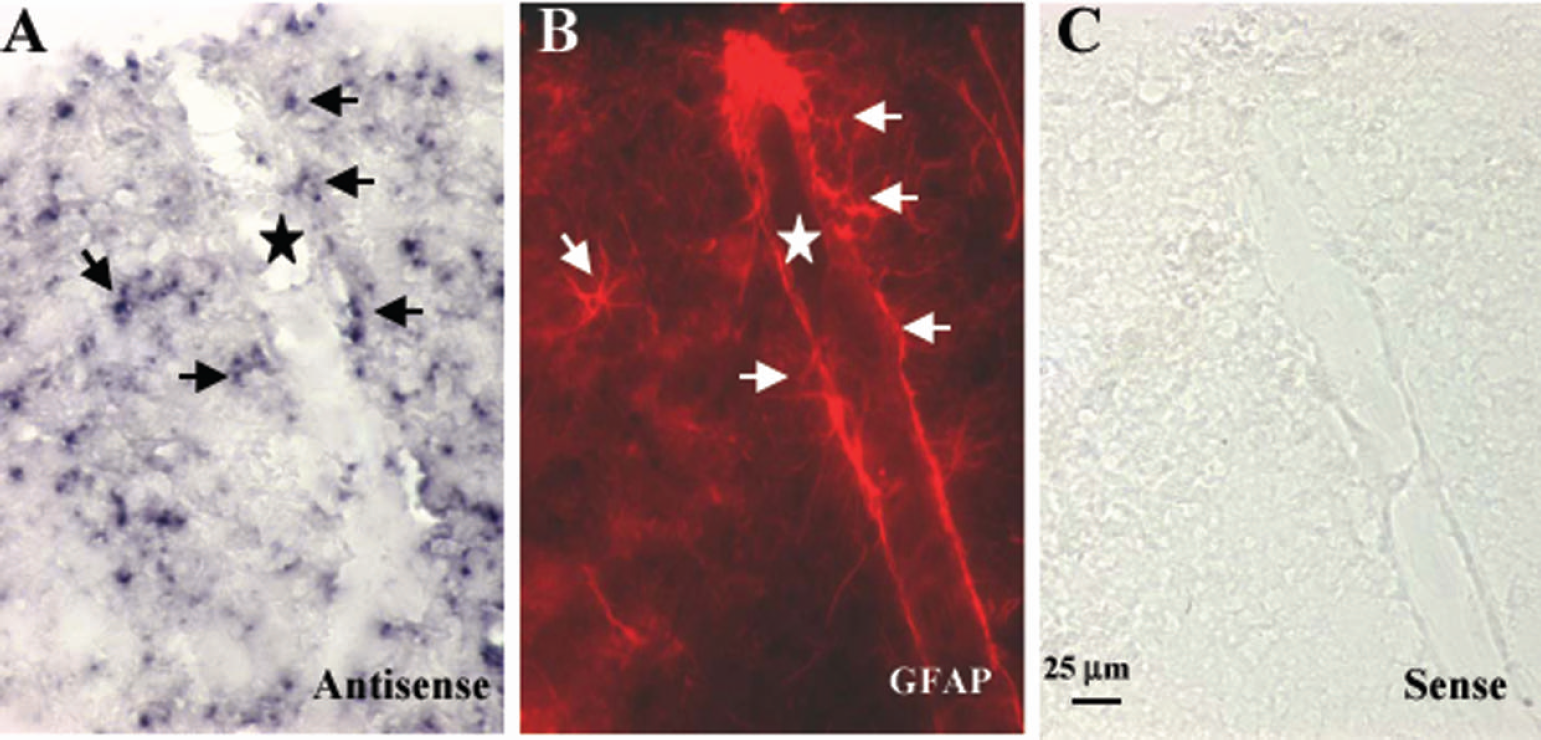
In situ hybridization of CYP2C11 mRNA in rat cortex. Shown are representative photographs of cortical sections hybridized with antisense probe (
Response to forepaw stimulation
Arterial pH, PCO2, PO2, and hemoglobin concentration were within the normal physiologic range before stimulation in all groups (Table 1). Rectal temperature and mean arterial blood pressure were not different among groups. Electrical forepaw stimulation did not produce a significant change in mean arterial blood pressure (Fig. 2).
Mean arterial blood pressure during the 60-second baseline before forepaw stimulation, the 20-second period of stimulation, and 60 seconds after stimulation in groups with cortical superfusion of CSF vehicle, L-NNA, MS-PPOH, MS-PPOH plus L-NNA, miconazole, and miconazole plus L-NNA. Values are means ± SD averaged among rats at 1-second intervals.
Physiologic variables before stimulation
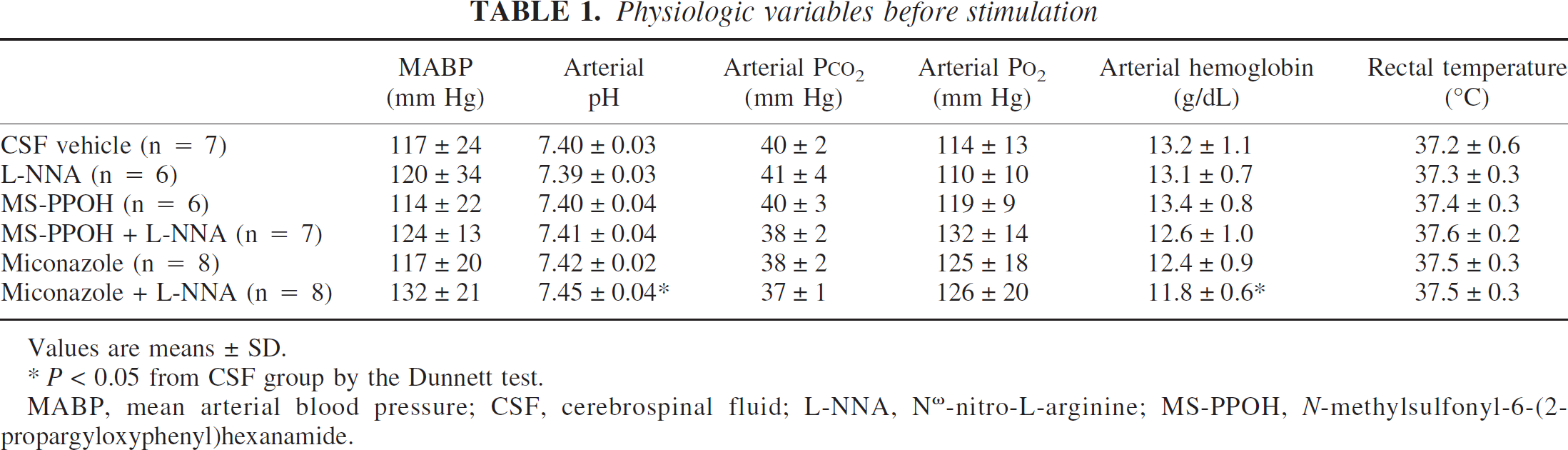
Values are means ± SD.
P>0.05 from CSF group by the Dunnett test.
MABP, mean arterial blood pressure; CSF, cerebrospinal fluid; L-NNA, Nω-nitro-L-arginine; MS-PPOH, N-methylsulfonyl-6-(2-propargyloxyphenyl)hexanamide.
The change in baseline LDF was analyzed over the 90-minute period of drug superfusion. Compared with the CSF time control group, LDF in the unstimulated state was lower in the groups receiving L-NNA, MS-PPOH, MS-PPOH plus L-NNA, and miconazole plus L-NNA (Table 2).
Percent changes in laser-Doppler flow
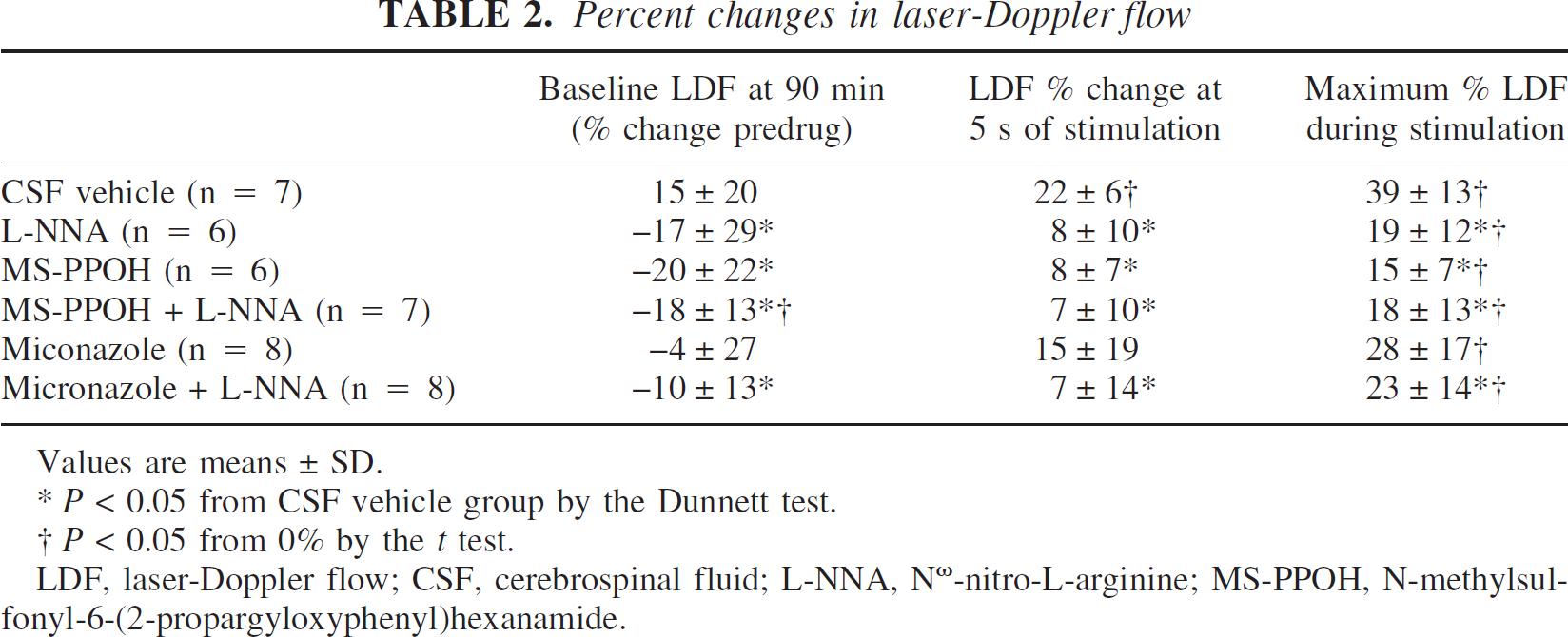
Values are means ± SD.
P>0.05 from CSF vehicle group by the Dunnett test.
† P>0.05 from 0% by the t test.
LDF, laser-Doppler flow; CSF, cerebrospinal fluid; L-NNA, Nω-nitro-L-arginine; MS-PPOH, N-methylsulfonyl-6-(2-propargyloxyphenyl)hexanamide.
In the control group receiving subdural CSF superfusion with vehicle, LDF began to increase significantly during the third second of stimulation and increased rapidly to 22% ± 6% by the fifth second of stimulation (Fig. 3). LDF continued to increase during the remaining period of stimulation, but at a more gradual rate. The maximum LDF increase was 39% ± 13%. In the group receiving cortical superfusion of L-NNA, both the initial increase at 5 seconds and the maximum increase in LDF were attenuated (Table 2).
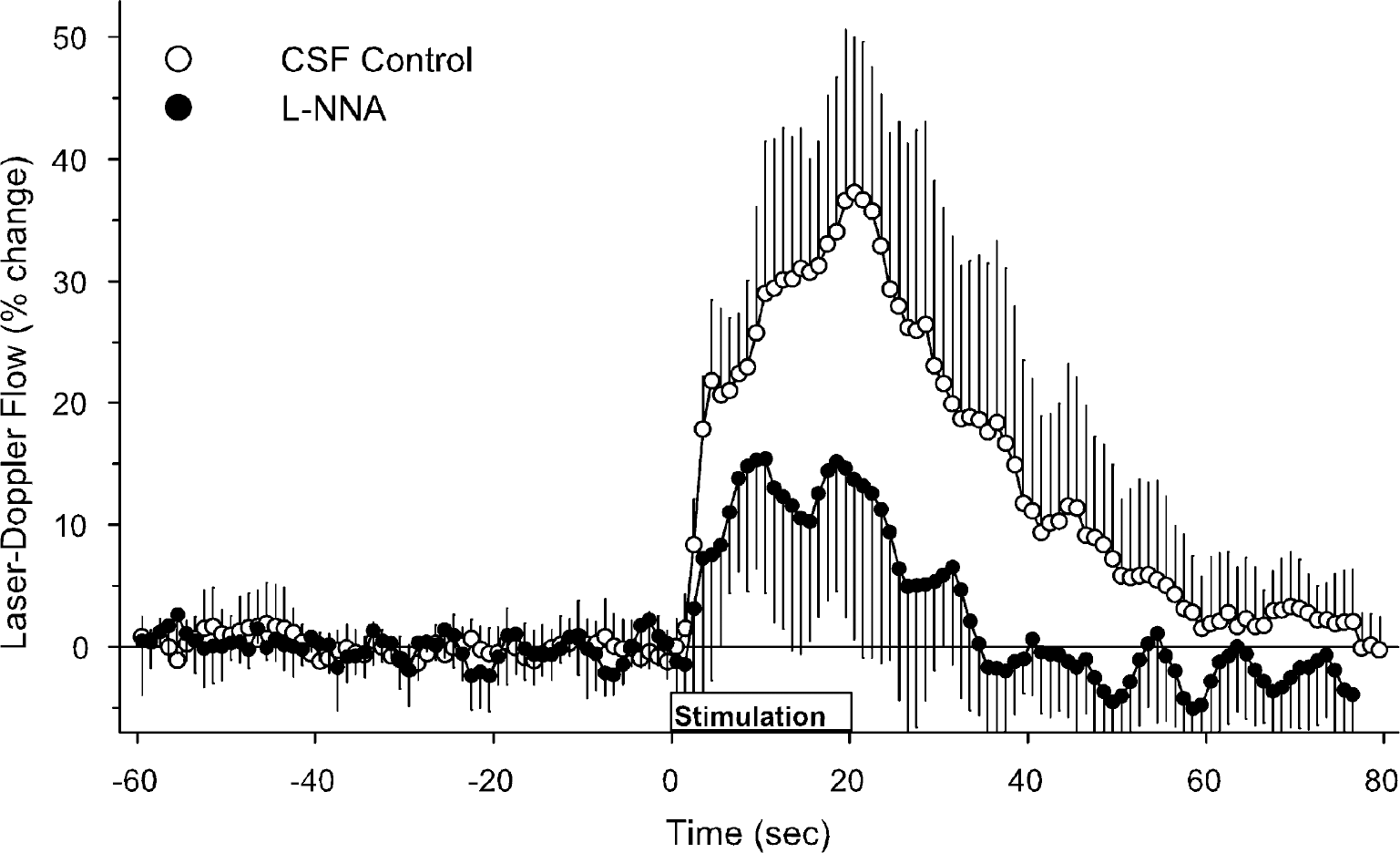
Laser-Doppler flow during the 60-second baseline before forepaw stimulation, the 20-second period of stimulation, and 60 seconds after stimulation in groups with cortical superfusion of CSF vehicle (open circles) and L-NNA (filled circles). Values are expressed as a percent change from baseline and are averaged among rats at 1 second intervals (means ± SD).
With MS-PPOH treatment, the LDF response was reduced throughout the stimulation period (Fig. 4). Both the early rapid response and the maximum increase were blunted (Table 2). When MS-PPOH was combined with L-NNA, the time course of the LDF response overlapped with the response seen with MS-PPOH treatment alone (Fig. 4).
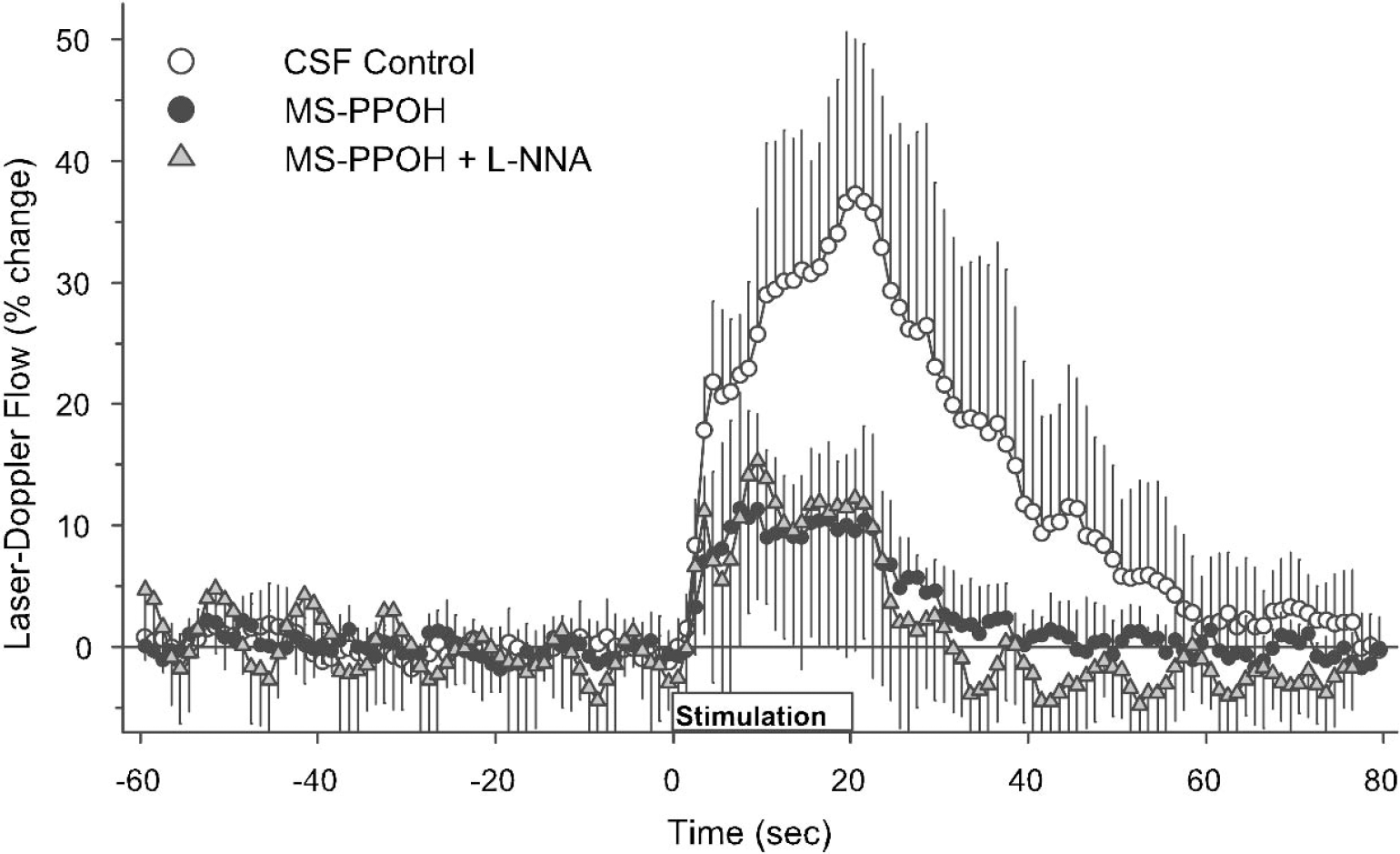
Laser-Doppler flow during the 60-second baseline before forepaw stimulation, the 20-second period of stimulation, and 60 seconds after stimulation in groups with cortical superfusion of CSF vehicle (open circles), MS-PPOH (filled circles), and MS-PPOH plus L-NNA (shaded triangles). Values are expressed as a percent change from baseline and are averaged among rats at 1-second intervals (means ± SD).
The time course of the LDF response with miconazole superfusion (Fig. 5) was similar to that of MS-PPOH. Although the LDF response at 5 seconds of stimulation was not statistically different from the control group, the percent increase was also not significantly different from zero because of variability in the onset of the response (Table 2). With miconazole plus L-NNA superfusion, the temporal response largely coincided with that seen with miconazole treatment alone (Fig. 5). The early response at 5 seconds and the maximum response were significantly less than the responses in the control group.
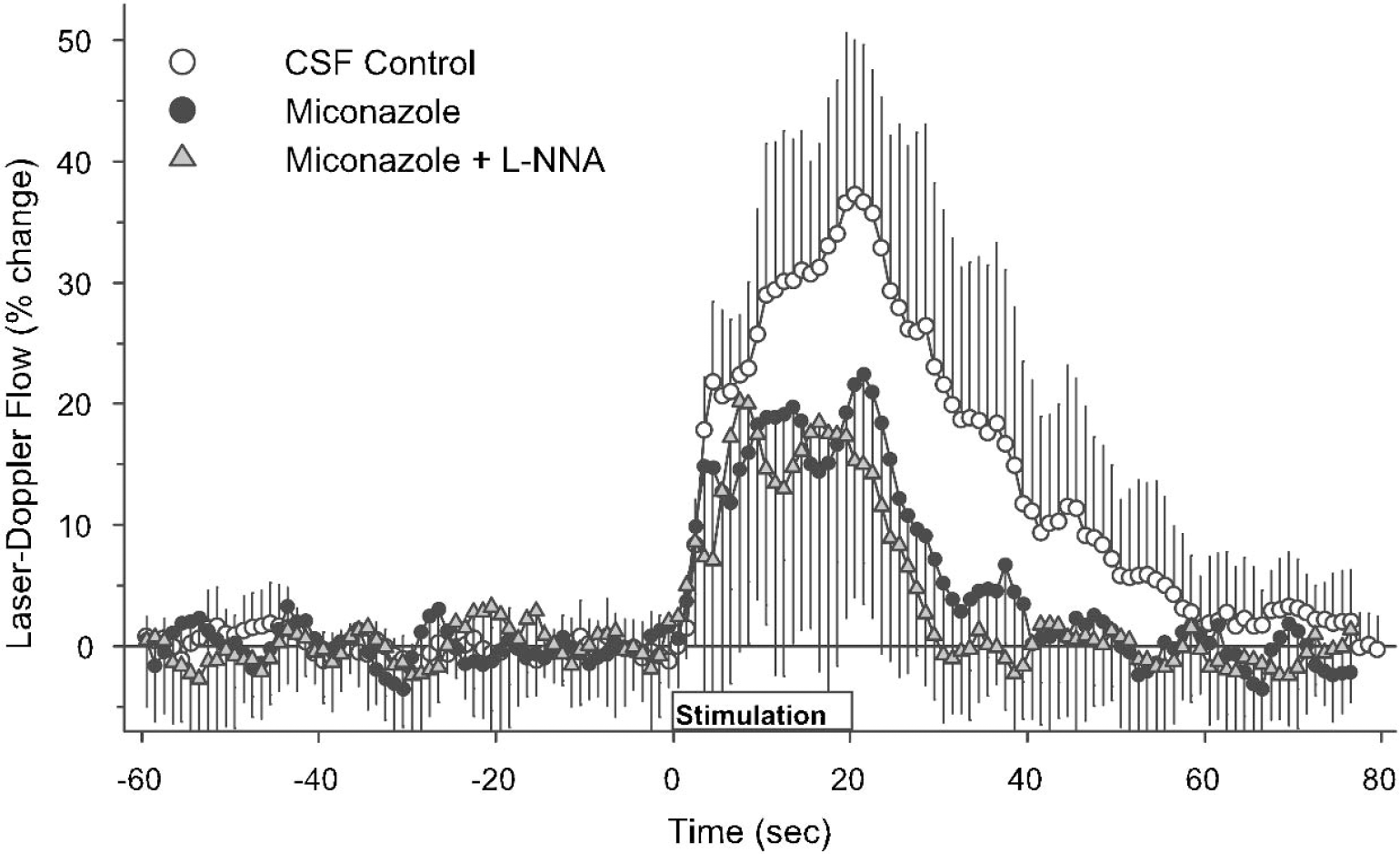
Laser-Doppler flow during the 60-second baseline before forepaw stimulation, the 20-second period of stimulation, and 60 seconds after stimulation in groups with cortical superfusion of CSF vehicle (open circles), miconazole (filled circles), and miconazole plus L-NNA (shaded triangles). Values are expressed as a percent change from baseline and are averaged among rats at 1-second intervals (means ± SD).
The percent LDF response in each animal was averaged over the 20-second stimulation period, and this average value was subjected to analysis of variance (Fig. 6). All drug-treated groups were significantly different from the control group but not from each other. Thus, combined treatment of MS-PPOH or miconazole with L-NNA was not different from single-drug treatment.
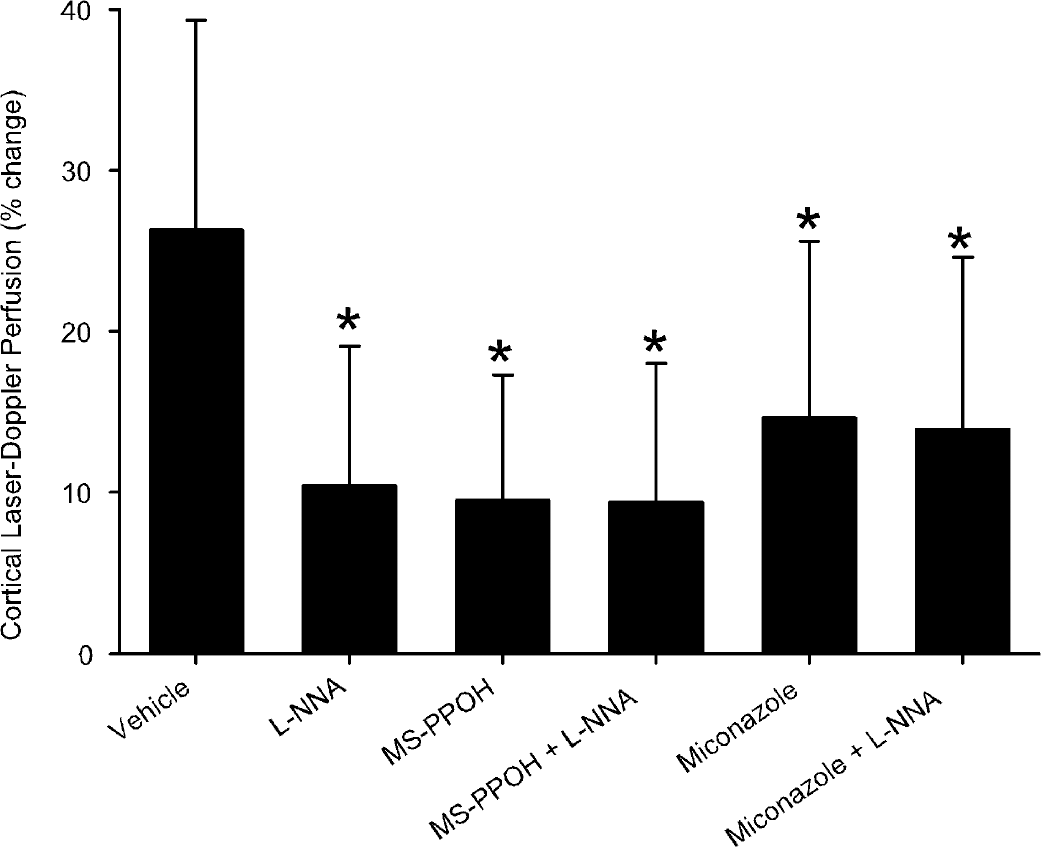
Laser-Doppler flow expressed as a percent change from baseline averaged over the 20-second period of forepaw stimulation in groups with cortical superfusion of CSF vehicle, L-NNA, MS-PPOH, MS-PPOH plus L-NNA, miconazole, and miconazole plus L-NNA. Values are means ± SD. *P>0.05 versus CSF vehicle group.
DISCUSSION
The major new findings of this study are that (1) CYP2C11, an epoxygenase previously found in cultured astrocytes, was expressed in astrocytes in rat cerebral cortex, including perivascular astrocytes; (2) both epoxygenase and NO synthase contribute to functional hyperemia in forelimb somatosensory cortex, as has been described previously in whisker barrel cortex (Dirnagl et al., 1993; Irikura et al., 1994; Peng et al., 2002); and (3) combined administration of epoxygenase and NOS inhibitors did not produce additional suppression of the blood flow response.
The overall level of CYPs in the brain is approximately 0.5% to 2% of that in liver microsomes (Hedlund et al., 2001). Epoxygenase activity is present in brain slices (Amruthesh et al., 1992) and in cultured astrocytes (Alkayed et al., 1996b), and CYP2C11 mRNA measured by reverse-transcription polymerase chain reaction is present in cultured astrocytes (Alkayed et al., 1996b). Homology among the different CYP2C family isoforms is exceedingly high (de Morais et al., 1993). Their mRNA levels are usually not abundant enough for detection by Northern blot analysis (Hedlund et al., 2001). The discrepancy among the somewhat widespread staining with antibodies and the low catalytic activity and mRNA level in the brain raises a question of how to detect a cell-type specific expression of CYP2C11 in the brain. By means of nonradioactive in situ hybridization with the TSA Plus signal-enhancing system (NEN Life Science Products) and a riboprobe specific for a unique area of the CYP2C11 sequence, CYP2C11 mRNA was detected in rat brain that was most consistent with astrocytic expression, as indicated by GFAP immunoreactivity. Moreover, the expression was detected in astrocytes situated in a close vicinity of blood vessels, but not in endothelial cells. Therefore, our results in intact cerebral cortex confirm cell culture work by showing specific expression of CYP2C11 mRNA in astrocytes in the brain. The close proximity of some of these positively labeled astrocytes to blood vessels is consistent with the concept that astrocytes may release vasodilatory substances such as EETs and thereby serve to integrate excitatory neuronal signaling for controlling blood flow in localized microcirculatory units.
Electrical stimulation of the forepaw has been used extensively for investigating the dynamics of the CBF response to functional activation in the transient response of tissue oxygenation (Ances et al., 2001a, b; Silva et al., 2000). The present results showing a 40% to 60% reduction of the LDF response in forelimb somatosensory cortex is in general agreement with results obtained in whisker barrel cortex with similar concentrations of MS-PPOH and miconazole (Peng et al., 2002) and L-NNA (Irikura et al., 1994). Therefore, the contribution of epoxygenase and NOS pathways to functional hyperemia appears to be ubiquitous for different regions of S1 somatosensory cortex and for different modalities of activation.
Activation of glutamate receptors can produce an increase in NO production within a selective neuronal population expressing neuronal NOS. NO is presumed to freely diffuse to nearby arterioles and produce localized vasodilation through stimulation of guanylyl cyclase in smooth muscle. Part of the vasodilation resulting from an increase in cyclic guanosine monophosphate can be mediated by opening of KCa channels (Paterno et al., 1996). In addition, NO may exert a direct effect on the β-subunit of the KCa channel (Wu et al., 2002). Vasodilation by EETs is also mediated by activation of KCa channels (Alkayed et al., 1996b; Gebremedhin et al., 1992). Because the reduction in the CBF response to whisker stimulation after NOS inhibition can be restored by application of a cell-permeant analog of cyclic guanosine monophosphate (Lindauer et al., 1999), a minimal amount of cyclic guanosine monophosphate may be required for permitting vasodilation by EETs. Thus, one potential explanation for the lack of additional inhibition of functional hyperemia by the combined use of epoxygenase and NOS inhibitors may be related to a nonredundant convergence of both pathways on the KCa channel. Another possibility is that EETs may function as a modulator of the CBF response and that a minimal amount of EETs may be required for its full expression.
In addition to activating guanylyl cyclase, NO donors have been found to produce cerebral vasodilation by a mechanism that involves inhibition of 20-HETE formation by CYP ω-hydroxylase activity (Sun et al., 2000; Yu et al., 2002). 20-HETE produces vasoconstriction by closing KCa channels (Harder et al., 1994; Lange et al., 1997). Thus, another potential explanation for the interaction of NOS inhibition with EETs is that loss of NO may permit an increase in 20-HETE formation, which might then counteract the opening of KCa channels by EETs.
The lack of a complete suppression of the CBF response to forepaw stimulation by the inhibitors used in this study implies that other mediators either normally play a role in the response or take on a greater role when the NOS and epoxygenase pathways are inhibited. This supposition assumes that the NOS and epoxygenase activities were completely inhibited. Although enzyme activities were not measured and dose-responses were not performed in the present study, a 1-mmol/L dose of L-NNA applied for at least 60 minutes has been shown to produce near-maximal inhibition of NOS catalytic activity (Irikura et al., 1994). With whisker stimulation, a dose of 5-μmol/L MS-PPOH produced a 28% inhibition of the flow response, whereas a dose of 20 μmol/L produced a 69% inhibition of the response by 60 minutes that was not reduced further by an additional hour of superfusion (Peng et al., 2002). Miconazole at a dose of 20 μmol/L produced a comparable inhibition of the flow response. Higher doses of MS-PPOH and miconazole were not tested because of concerns regarding nonspecific effects (Dudek et al., 1995; Wang et al., 1998). Thus, a limitation of the present study is the uncertainty of whether epoxygenase activity is completely inhibited throughout the cortical layers with the 20-μmol/L dose that was superfused for 90 minutes.
The reduction in the evoked CBF response with MS-PPOH and miconazole is unlikely to be due to a reduction in neuronal activation because neither drug was found to reduce somatosensory-evoked potentials during an electrical forepaw stimulation (Peng et al., 2002). Others have reported either a decrease (Ngai et al., 1995, 1998) or no change (Lindauer et al., 1996) in somatosensory-evoked potentials after NOS inhibition. If NOS inhibition decreases neuronal activation in the forepaw stimulation model, then it is possible that the lack of additional inhibition of the blood flow response with combined NOS and epoxygenase inhibition could be partly explained by a decrease in the stimulus for activating epoxygenases.
These epoxygenase inhibitors were found to have no effect on NOS catalytic activity at the concentrations used in the present study (Alkayed et al., 1996a; Bhardwaj et al., 2000). Moreover, neither of these drugs reduce cerebral vasodilation produced by NO donors or the adenosine agonist 2-chloroadenosine (Alkayed et al., 1996a; Bhardwaj et al., 2000; Peng et al., 2002). Therefore, MS-PPOH and miconazole are unlikely to be acting directly on NOS, or indirectly by interfering with NO or adenosine vasodilatory pathways.
The CBF response to whisker stimulation is considerably greater in the unanesthetized state (Nakao et al., 2001), where the effects of NOS inhibition on the CBF response appear to be much smaller (Gotoh et al., 2001). Thus, one limitation of the present study is the use of anesthesia, which may alter the interaction of the epoxygenase and NOS pathways on the functional hyperemia response.
In summary, the present study shows that CYP2C11 is expressed in astrocytes in rat cortex, that both NOS and epoxygenase activities are required for the full expression of the CBF response to functional activation in anesthetized rats, and that NOS and epoxygenase pathways do not act in a simple parallel fashion. The residual CBF response after combined inhibition implies that other mediators are also involved in the coupling of CBF to neuronal activation in cortex.
