Abstract
The P2X7 receptor for extracellular ATP is the main candidate, among P2 receptors, inducing cell death in the immune system. Here, we demonstrate the direct participation of this receptor to cell damage induced by oxygen/glucose deprivation, in the ex vivo model of organotypic hippocampal cultures. By pharmacological and immunological approaches, we show that P2X7 is rapidly and transiently up regulated in hippocampal areas eliciting metabolism impairment. Moreover, the P2 antagonists 2′,3′,-dialdehyde ATP and reactive blue 2 prevent both up regulation of this receptor and hypoxic/hypoglycemic damage. By confocal laser microscopy, we show that P2X7 is present at the synaptic level of fibers extending from the CA1–2 pyramidal cell layer throughout the strata oriens and radiatum, but absent on oligodendrocytes, astrocytes or neuronal cell bodies. Colocalization of P2X7 is obtained with neurofilament-L protein and with synaptophysin, not with myelin basic protein, glial fibrillary acidic protein or a marker for neuronal nuclei. P2X7 up regulation and diffuse cellular damage are also induced by 3′-O-(4-benzoyl) benzoyl-ATP, an agonist selective but not exclusive for P2X7.
In summary, our study demonstrates that P2X7 not only directly participates to the hypoxic/hypoglycemic process, but also owns specific phenotypic localization. We do not exclude that it might serve as a sensor of dysregulated neuronal activity and ATP release, both occurring during oxygen/glucose deprivation.
ATP is now regarded as one of the most ubiquitous neuromodulator co-released in the extracellular space together with other neurotransmitters such as glutamate or acetylcholine and acting as an intercellular signaling molecule (Burnstock and Williams, 2000;Fields and Stevens, 2000;Vizi, 2000).
The receptors for extracellular ATP are classified into P2X and P2Y subclasses. P2Y are G-protein coupled metabotropic receptors possessing slow excitatory responses. P2X are ionotropic receptors responsible for fast excitatory neurotransmission (North and Barnard, 1997). These last are in turn classified by electrophysiological properties and molecular homology into seven different subunits (P2X1–7) (Khakh et al., 2001), which are functional only when assembled in heteromeric and oligomeric structures. For example, P2X2 can dimerize with P2X3, and P2X1 (Nicke et al., 1998) can form active receptors with P2X5 (North, 2002). On the contrary, P2X7 is the only receptor to be functional in its homomeric form (North and Surprenant, 2000). The P2X7 is also considered the most cytotoxic among P2 receptors. Although it is widely expressed in a variety of different tissues (Collo et al., 1997), its role on cellular toxicity and inflammation was thoroughly studied in the immune system (Di Virgilio et al., 1995;Di Virgilio et al., 1999). Stimulation of P2X7 with ATP induces a plethora of biological responses comprising intracellular Ca++ overloading and K+ efflux, all culminating with apoptotic and necrotic cell death (Schulze-Lohoff et al., 1998;Chen and Swanson, 2003). Recently, it was also suggested that activation of P2X7 regulates the release of neurotransmitters in astrocytes and neurons (Duan et al., 2003;Sperlagh et al., 2002) and the cleavage and release of IL-1 in macrophages and microglia (Sanz and Di Virgilio, 2000;Le Feuvre et al., 2002; 2003). These events leading to cell death comprise phosphorylation and activation of specific kinases (Parvathenani et al 2003;Humphreys et al., 2000;Communi et al., 1999) and induction of transcription factors (Ferrari et al. 1997) and caspase-1 (Ferrari et al. 1999; Shulze-Lohoff et al., 1998).
In contrast, little is still known about the exact function of the P2X7 in neurons and its involvement in neurodegenerative insults and stroke, although preliminary results were reported. For instance, during an ischemic event ATP may reach high extracellular levels, being exocytotically released from various cell types including neurons (Agteresch et al., 1999). Moreover, Collo and her colleagues (1997) demonstrated that P2X7 mRNA expression increases in the penumbra of ischemic damage;Amadio et al. (2002) reported that P2X7 mRNA and protein are up regulated in cerebellar granule neurons and in hippocampal organotypic cultures following ATP stimulation;Cavaliere et al. (2002) showed that P2X7 protein augments as a consequence of metabolism impairment in CNS neurons. Since in a previous work we described that P2X2 and P2X4 are up regulated, respectively in neurons and in microglia, by transient global ischemia in the gerbil (Cavaliere et al., 2003), in this work we analyze the participation of the P2X7 receptor to oxygen/glucose deprivation, using the ex vivo model of organotypic primary hippocampal cultures.
METHODS
Organotypic cultures
Organotypic hippocampal slice cultures were prepared using a modification of the method by Stoppini (1991). Briefly, Wistar rat pups (8–10 days old) were sacrificed and brains removed. Hippocampi were excised, cut on a Mc Ilwain tissue chopper (400 μm) and the slices separated into cold HBSS (0.185 mg/ml CaCl2, 0.1 mg/ml MgSO4, 0.4 mg/ml KCl, 0.06 mg/ml KH2PO4, 8 mg/ml NaCl, 0.05 mg/ml Na2HPO4, 0.35 mg/ml NaHCO3, 1 mg/ml glucose; Sigma, MI-Italy). Four slices were plated on each Millicell CM culture inserts (Millipore Rome, Italy) and kept in organotypic maintenance medium (50% MEM, 25% HBSS, 25% heat-inactivated horse serum, supplemented with 4.5 mg/ml glucose, 1 mM glutamine) at 37°C, 100% humidity and 5% CO2. The medium was changed every 3–4 days and experiments performed after 12–14 days in vitro.
Oxygen/glucose deprivation and quantification of cell death
For oxygen/glucose deprivation studies, the Millicell CM inserts with organotypic slices were placed for 40–60 minutes at 37° C in a N2 saturated environment in 1 ml of Glucose-Free Medium (GFM-Earle's Balanced Salt Solution; Sigma, Mi-Italy) added with 5 μg/ml propidium iodide (PI), previously saturated with 95% N2, for the analysis of cell death. The GFM was then replaced with the organotypic maintenance medium and the cultures were kept with 5 μg/ml PI under normoxic conditions for different lengths of time, before evaluating cell death. For normoxic conditions, the medium consisted of GFM supplemented with 1 mg/ml of glucose.
The extent of cell death was quantified by propidium iodide incorporation measured with a fluorescence microscope, using the densitometry software Scion Image for Windows (Scion corporation, Maryland, USA), obtained from the NIH public domain. In detail, regions of interest (ROI) from each experimental image were outlined and pixel/inches were directly measured, following the software procedures directly provided by the manufacturer. Results are expressed as mean pixel/inches ± SEM (n = 3).
Total protein extraction from organotypic cultures
Organotypic cultures were maintained for 40 minutes under oxygen/glucose deprivation. After different lengths of post-treatment time, four organotypic slices were extracted in RIPA buffer (PBS supplemented with 1% NP-40, 0.5% sodium deoxycholate, 0.1% SDS, 0.5 μM PMSF, 10 μg/ml leupeptin, all from Sigma, Mi-Italy) and homogenized. They were maintained for one hour on ice, sonicated and centrifuged at 4°C at 10000x g for 10 minutes. Protein quantification was performed in the supernatants by Bradford colorimetric assay (Biorad, Mi-Italy).
Western blot
Equal amount of total protein from each sample (50 μg) was separated by SDS-PAGE on a 12% polyacrylamide gel and transferred over-night onto a nitrocellulose membrane Hybond C (Amersham, Mi-Italy). The filters were pre-wetted in 5% non-fat milk in TBS-T (10 mM Tris pH 8, 150 mM NaCl, 0.1% Tween 20) and hybridized for three hours with anti-P2X1,4,7 antisera (1:200) (Alomone, Jerusalem, Israel). All antisera were immunodetected with an anti-rabbit HRP conjugated antibody and developed by ECL chemioluminescence (Santa Cruz, Mi, Italy). Quantification of the specific bands was performed in a linear range of detection, by using Kodak 1D 3.5.3 software.
Immunofluorescence studies
Confocal microscopy.
Organotypic hippocampal slices were used for immunofluorescence labeling. Each slice was fixed for 2.5 hours at RT in 4% para-formaldehyde, three times washed in PBS and then incubated in a mixture of primary antisera for 24 hours in 0.4% Triton X-100 in 1% albumin bovine serum in PBS. Rabbit anti-P2X7 (1:100, Alomone, Jerusalem-Israel) was used alone or in combination with mouse anti-MBP (myelin basic protein, 1:400, Boehringer Mannheim, Mi-Italy), mouse anti-GFAP (glial fibrillary acidic protein, 1:400, Sigma, Mi-Italy), goat anti-NFL (neurofilament-L protein, 1:100, Santa Cruz, Mi-Italy) or mouse anti-synaptophysin (1:100 Sigma, Mi-Italy). The secondary antibodies were Cy2-conjugated donkey anti-rabbit IgG (1:100, green immunofluorescence) or Cy3-conjugated donkey anti-mouse IgG (1:100, red immunofluorescence) or Cy3-conjugated donkey anti-goat IgG (1:100, red immunofluorescence). All antibodies were from Jackson Immunoresearch, West Baltimore Pike PA, USA. The sections were washed three times in PBS for five minutes and then incubated for 2 hours with the secondary antibodies. After rinsing, the slices were mounted on slide glasses, allowed to air dry and cover slipped with gel/mount™ anti-fading medium (Biomeda, Foster City CA, USA).
Double label immunofluorescence was analyzed by means of a confocal laser scanning microscope (CLSM) (LSM 510, Zeiss, Mi-Italy) equipped with an argon laser emitting at 488 nm and a helium/neon laser emitting at 543 nm.
Statistical analysis.
Values were normalized as specified in each figure legend. Statistical differences were evaluated by one-way analysis of variance (ANOVA), followed by post hoc test (HSD Tukey). A value of P < 0.05 is considered significant.
RESULTS
In this work, we adopted organotypic slice cultures to study the direct participation of the P2X7 receptor to the cellular damage caused by oxygen/glucose deprivation, using both pharmacological and immunological approaches. We deprived the organotypic hippocampal slices of oxygen/glucose for 40 minutes respectively in the absence or presence of the specific P2X7 antagonist 2′,3′,-dialdehyde ATP (oxATP) (Fig. 1A) (Murgia et al., 1993). Under these conditions, a localized CA1–3 cellular damage (Frantseva et al., 2002;Pringle et al., 1997) was obtained in about 20 hours post-treatment (Fig. 1A). As visualized by propidium iodide incorporation, whereas the simultaneous addition of 100 μM oxATP during oxygen/glucose deprivation only partially reduced the cellular damage in the CA1–3 hippocampal region (data not shown), oxATP completely sustained cell viability when provided 1 hour before and during hypoxia/hypoglycemia (Fig. 1A).
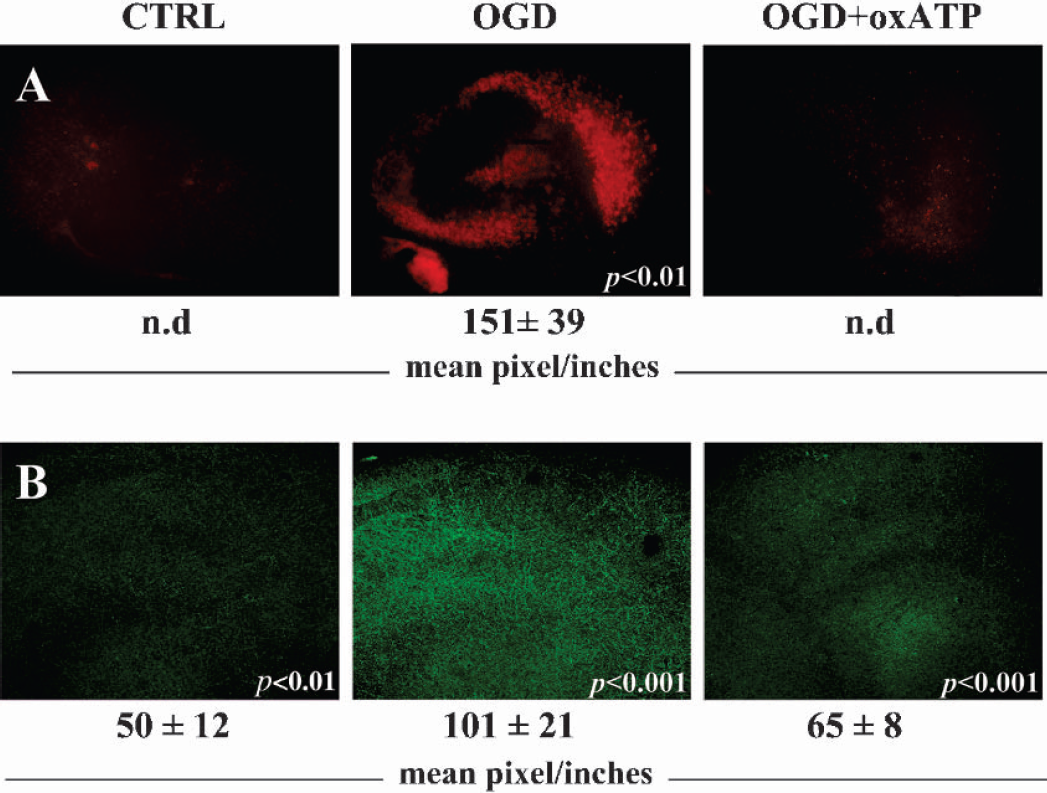
The P2X7 antagonist oxATP protects from hypoxic/hypoglycemic hippocampal injury and prevents P2X7 protein up regulation. Hippocampal organotypic cultures at 12–14 DIV were maintained for 40 minutes under oxygen/glucose deprivation (OGD), in the simultaneous presence or absence of 100 μM 2′,3′,-Dialdehyde ATP (oxATP). Control cultures (CTRL) were maintained in the presence of 1 mg/ml glucose. Cellular damage was visualized 20 hours later with a fluorescence microscope, by propidium iodide incorporation (A). Immunofluorescence for P2X7 receptor was performed in 1 hour after oxygen/glucose deprivation and visualized at 20X magnification (B). The results were analyzed by using Scion Image and are representative of three independent experiments. Intensities are expressed as mean pixel/inches ± SEM (n = 3), where n.d. (not detected) represents a signal below threshold level. Statistical differences in OGD were calculated vs CTRL, whereas in OGD+oxATP they were calculated vs OGD.
We also studied the P2X7 protein expression following metabolism impairment. In 1 hour after the insult (Fig. 1B), the P2X7 immunofluorescent signal homogeneously increased (about two fold) in the treated slices compared to the control, whereas the antagonist oxATP reduced this effect by about 70% (Fig. 1B). When observed at higher magnification, immunofluorescence was present especially on parallel fibers running across the pyramidal cell layer at the CA1–2 region (Figs. 3B and 4). Confirming this up regulation, Western blot analysis showed a strong transient induction of the P2X7 protein occurring in 1 hour post-treatment (Fig. 2A). This decreased to control level in 20 hours, when the cellular damage was instead maximally induced (Fig. 1A). When added before and during oxygen/glucose deprivation, 100 μM oxATP not only reduced the cellular damage (Fig. 1A), but also partially prevented (about 55%) the induction of the P2X7 protein (Fig. 2B). The broadly effective P2 antagonist Reactive Blue-2 (Cavaliere et al., 2001a;Cavaliere et al., 2001b) completely inhibited such up regulation (Fig. 2B).
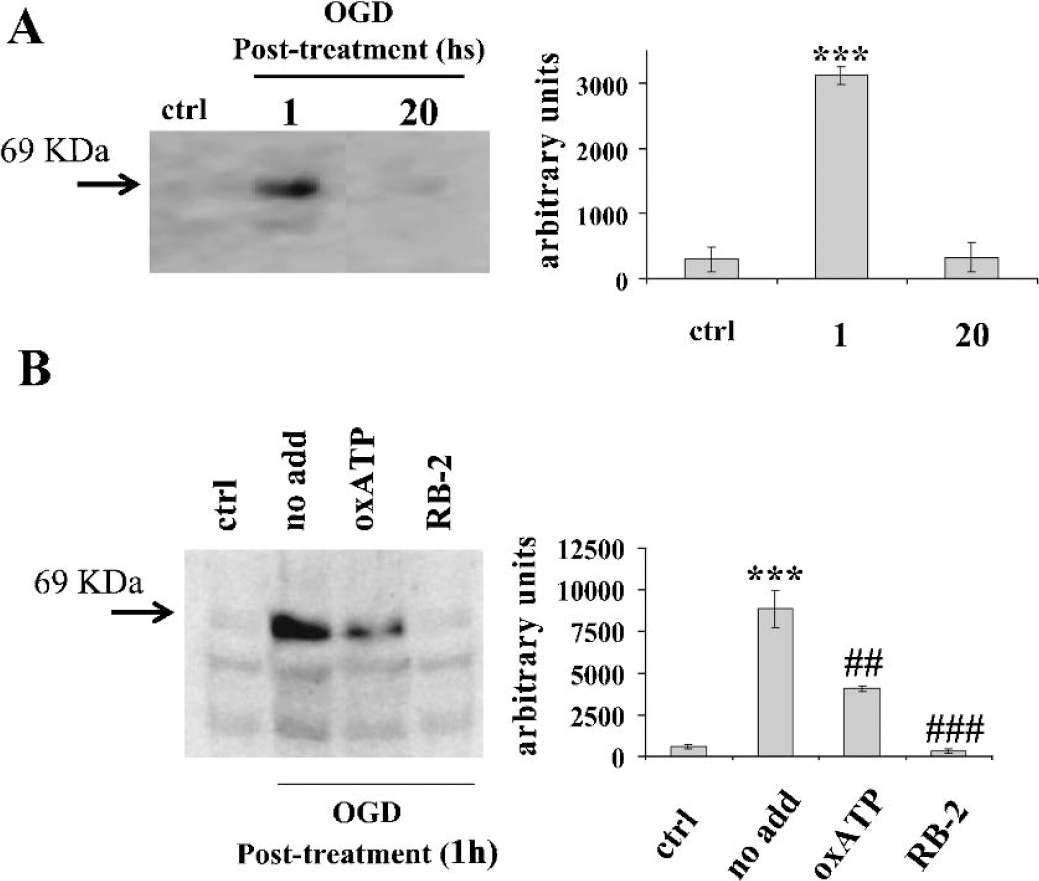
Expression of P2X7 receptor after hypoxia/hypoglycemia: Western blot analysis. Hippocampal organotypic cultures at 12–14 DIV were maintained for 40 minutes under oxygen/glucose deprivation (OGD). Total protein was extracted after different lengths of time (A) and analyzed by Western blot in the absence (no add) or presence of 100 μM oxATP or 100 μM reactive blue 2 (RB-2) (B). Similar results were obtained in three independent experiments. Bands were quantified with Kodak Image Station software, and expressed as arbitrary units. Statistical differences were calculated by one-way ANOVA test followed by Tukey's post hoc test. Statistical differences were evaluated as follow: in (A), OGD post treatment was compared to ctrl; in (B), no add was compared to ctrl; both oxATP and RB-2 were compared to no add. (***P < 0.001; ###P < 0.001; ##P < 0.01).
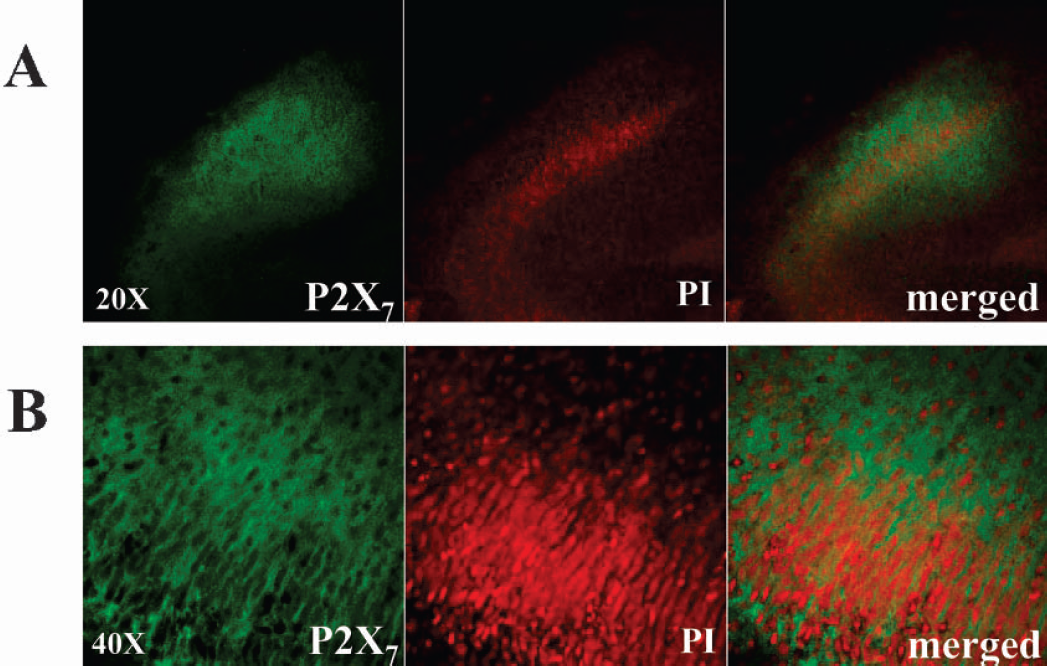
Up regulation of P2X7 protein occurs in 1 hour posttreatment in the CA1–2 damaged region. Organotypic slices were subjected to oxygen/glucose deprivation for 40 minutes and to 1 hour post-treatment, in the presence of 5 μg/ml of propidium iodide. The slices were then fixed and stained with anti-P2X7. Results are representative of at least three independent experiments.
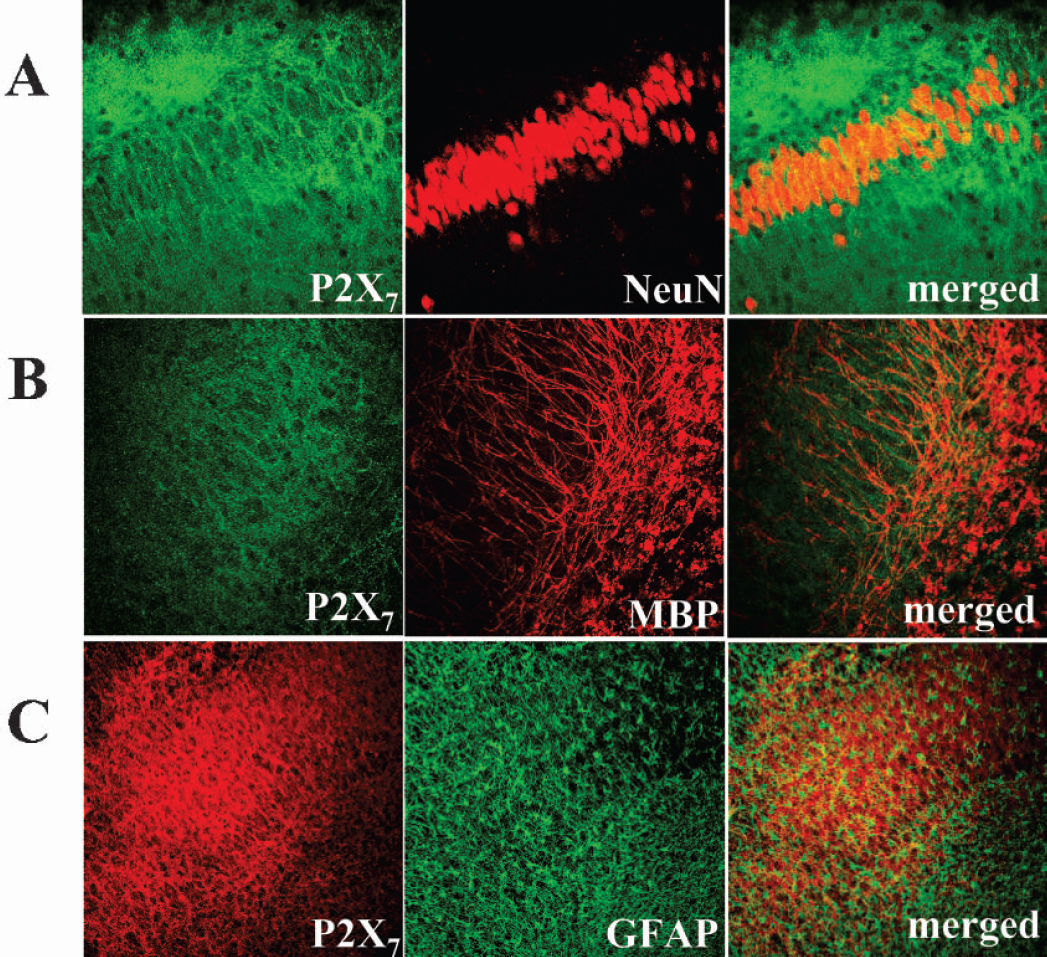
Cellular localization of P2X7 receptor protein. After 40 minutes of oxygen/glucose deprivation and 1 hour post-treatment, hippocampal organotypic cultures were double stained with P2X7 and several cellular markers. The CA1–2 region was visualized by CLSM. Antibodies against NeuN were used for labeling neuronal nuclei; against myelin basic protein (MBP) for oligodendrocytes, and against glial fibrillary acidic protein (GFAP) for astrocytes. Results are representative of three independent experiments.
We localized the P2X7 signal by performing double labeling with propidium iodide. In one hour after the insult, the incorporation of propidium iodide starting in the CA1–2 region overlapped with the P2X7 staining (Fig. 3A; 20X magnification). Nevertheless, ultra structural analysis at higher magnification did not reveal colocalizing signals between P2X7 and cellular damage (Fig. 3B; 40X magnification). In one hour after oxygen/glucose deprivation, the P2X7 immunopositive signal was present on green punctuated fibers extending throughout the CA1-CA2 pyramidal cell layer, but was not coincident with the nuclear staining observed with propidium iodide (Fig. 3B merged view). At a cellular level, we demonstrated that the P2X7 positive signal did not colocalize with neuronal nuclei (Fig. 4A), with oligodendrocytes (Fig. 4B), or with astrocytes (Fig. 4C). We instead described both neuronal localization of P2X7 on fibers sprouting from the pyramidal cell layer to the strata oriens and radiatum (Figs. 1B, 3A, 4A) as well as positive double staining with neurofilament-L protein (NFL) (Fig. 5A). Moreover, as shown in Fig. 5B, double immunofluorescence demonstrated strong colocalization between P2X7 and the synaptic marker synaptophysin.
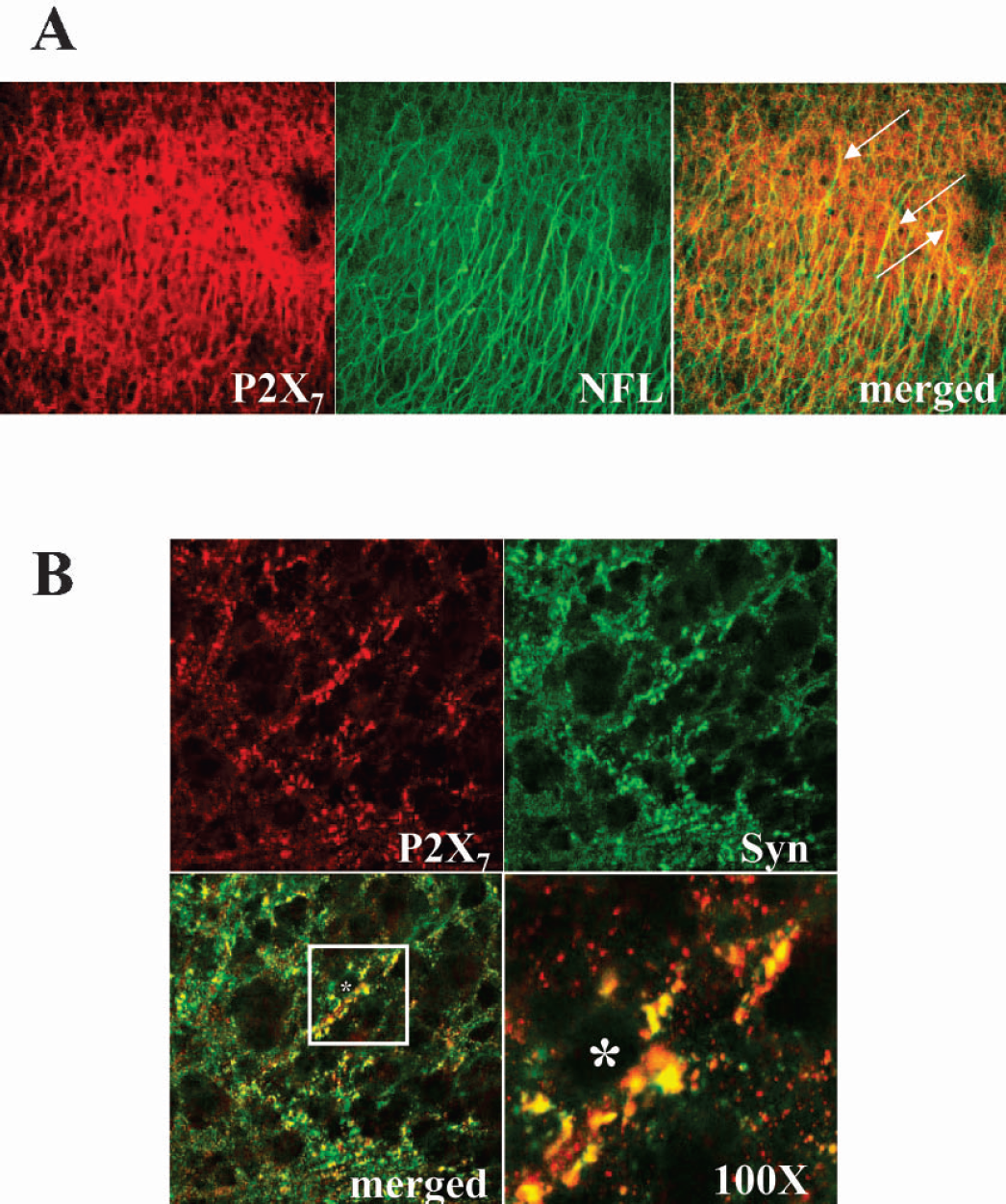
Sub-cellular localization of P2X7 receptor protein. After oxygen/glucose deprivation, the hippocampal slices were fixed, double stained with P2X7 and either neurofilament-L protein (NFL) (A) or synaptophysn (Syn) (B), and CA1–2 region was visualized by CLSM (40X magnification). In (A), the arrows show a marked colocalization of P2X7 and NFL; in (B), the white square on the merged panel is enlarged at 100X magnification and the asterisk indicates the neuronal bodies. Results are representative of three independent experiments.
In order to strengthen the correlation between P2X7 and cell death, we subjected hippocampal slices to 3′-O-(4-benzoyl) benzoyl ATP (BzATP) and then tested cellular damage and P2X7 protein expression. Although very limited subtype-specific P2 ligands yet exist and most available agonists/antagonists generally bind to both P2X and P2Y subclasses (Burnstock, 2002), the compound BzATP is a P2 agonist considered most specific for P2X7 although activating a few additional P2X receptors (P2X1,3,4) (North and Surprenant, 2000;Evans et al., 1995). Therefore, we kept the organotypic hippocampal cultures with 500 μM BzATP for 20 hours, in the presence or absence of the P2X7 specific antagonist ox-ATP. We observed diffuse cell death without specific damage across the entire hippocampal region, and a reduction by oxATP of about 40% in the propidium iodide incorporation (Fig. 6A). Under these conditions, also the P2X7 protein expression was induced by BzATP (about 100%) and prevented by oxATP (60% reduction), as demonstrated by Western blot analysis and quantification (Fig. 6B). In parallel, we observed about two/three fold increase in the P2X1 protein expression, but this was not prevented by oxATP (Fig. 6B) thereby confirming the specificity of oxATP on P2X7. The additional targets of BzATP, P2X3 and P2X4, were not modulated in protein expression during and after the insult (data not shown).
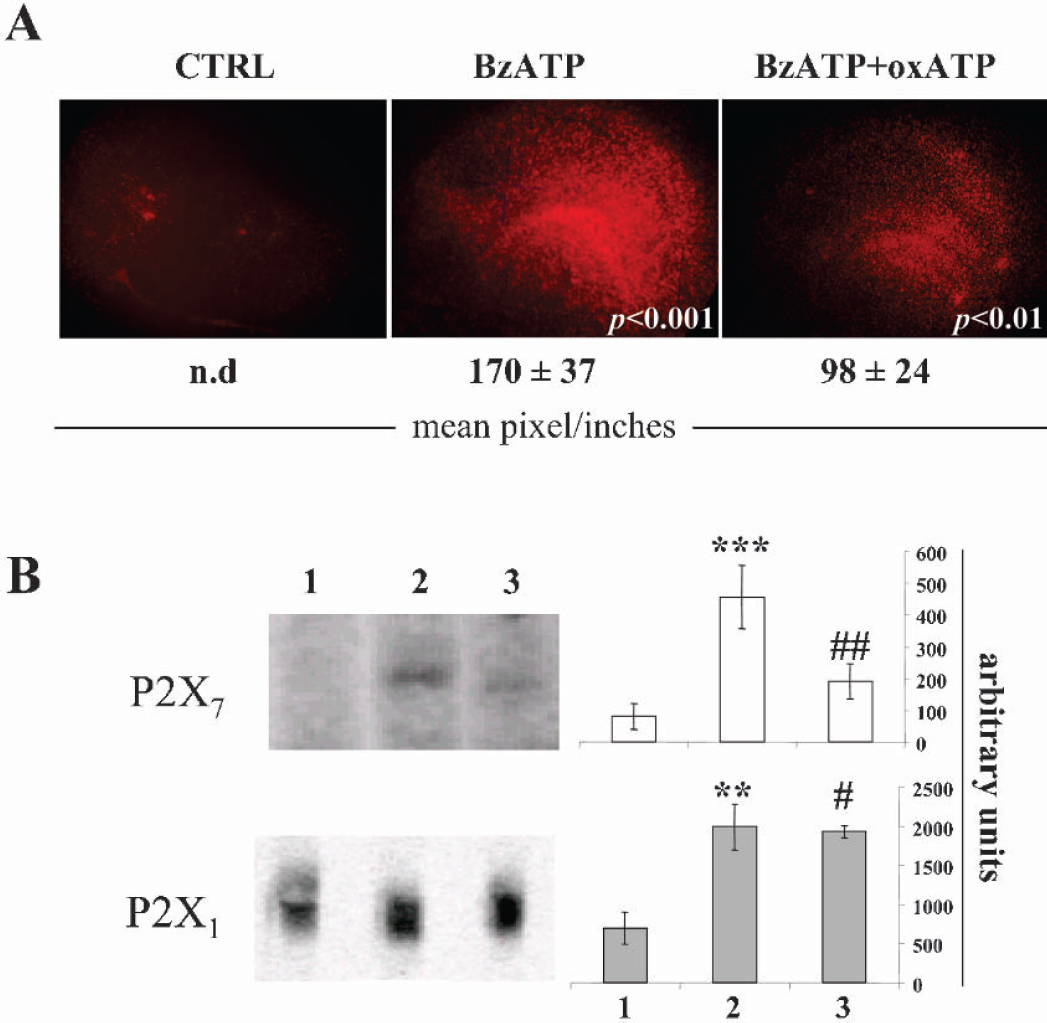
BzATP-induced damage and modulation of P2X target receptors. (A) Hippocampal organotypic cultures at 12–14 DIV were maintained for 20 hours in maintenance medium, in the presence of 500 μM BzATP and with or without 100μM oxATP. Cellular damage was visualized with a fluorescence microscope by propidium iodide incorporation. Results are representative of three independent experiments and were analyzed by using Scion Image. Intensities are expressed as mean pixel/inches ± SEM (n = 3), where n.d. (not detected) represents a signal below threshold level. Statistical differences in BzATP were calculated vs CTRL, whereas in BzATP+oxATP they were calculated vs BzATP. (B) Organotypic cultures were homogenized, total protein was extracted and P2X1,7 was detected by Western blot and analyzed by Kodak software. Statistical differences were calculated in sample 2 (BzATP) vs 1 (CTRL), and in sample 3 (BzATP+oxATP) vs 2 (BzATP). (***P < 0.001; **P < 0.01; ##P < 0.01; #P < 0.05).
DISCUSSION
The use of organotypic slice cultures represents one of the most convenient ex vivo system to study features and mechanisms of cerebral hypoxia/hypoglycemia (Pringle et al., 1997; Pellegrini-Gianpietro et al., 1999). The slices preserve organotypic structures and functions, neuronglia interactions and activation of numerous pathways, such as immune and inflammatory responses (Coltman et al., 1995; Czapiga and Colton, 1999). Moreover, they represent an efficient biological model to overcome the in vivo impediments of the blood brain barrier. Our previous work demonstrated, both in organotypic cultures and in vivo, the direct participation of the extracellular ATP receptors, particularly P2X2 (over expressed in neurons) and P2X4 (over expressed in microglia), to ischemic injury (Cavaliere et al., 2003). Whereas little is still known about a direct role of P2X7 on metabolic impairment, indirect experimental evidence is provided by the outflow of ATP from damaged cells after seizure and stroke (Nieber et al., 1999;Phillis et al., 1993;Burnstock 2002), by the expression of P2X7 in different cell types, tissues and in the penumbra of ischemic damage (Collo et al., 1997;Di Virgilio et al., 1995;Di Virgilio et al., 1999), by the increase of microglial P2X7 mRNA and protein during cerebral ischemia in a rodent model (Collo et al., 1997), and by the activation and modulation of inflammatory processes through several P2 receptors comprising P2X7 (Di Virgilio 1995;North, 2002). With the present work, we now recognize the ionotropic P2X7 receptor as an important element in the mechanisms of cellular damage induced by oxygen/glucose deprivation. On this topic, Le Feuvre and co-workers recently established a distinction between in vitro and in vivo pro-inflammatory role of P2X7 following ischemic insults (Le Feuvre et al., 2002; 2003). Although they demonstrated that activation of P2X7 in primary in vitro cultures of microglia and macrophages directly regulates the release of IL-1 (Brough et al., 2002), they sustained that pathways additional or parallel to P2X7 receptor could mediate IL-1 secretion in in vivo ischemia. In fact, deletion of P2X7 in knock out mice does not apparently affect neuronal damage induced in the cortex-striatum by medial cerebral artery occlusion (Le Feuvre et al., 2002), and a selective antagonist of IL-1 receptor reduces the ischemic damage also in P2X7 knock out mice (Le Feuvre et al., 2003).
Our ex vivo model of hippocampal organotypic cultures now establishes that the insult from oxygen/glucose deprivation goes also through the neuronal P2X7 receptor. Pharmacological data integrate and support biochemical and immunological evidence on this regard. We have first demonstrated that during the early phases of post-hypoxia/hypoglycemia, before the onset of cell loss, the P2X7 protein is rapidly and transiently activated. Moreover, P2X7 can be considered an early marker of a specifically localized cellular damage. In fact, up regulation occurs in hippocampal areas coincident with cellular damage and specific (oxATP) or widespread (RB-2) P2 receptor antagonists either reduce or block simultaneously this activation and cellular damage. Second, we have proved that despite a temporal gap between P2X7 induction and cell loss, the topological and pharmacological correlation between P2X7 expression and cell death is very specific. Whereas hypoxia/hypoglycemia induce cell death in the restricted CA1–3 hippocampal region, BzATP causes a more largely diffuse cellular damage in the entire hippocampal area. Furthermore, the selective P2X7 antagonist oxATP totally prevents cell death, but only partially reduces the cell damage caused by BzATP. In this case, the less localized insult and the consequent less efficient protection would suggest that pathways additional to P2X7 might be involved. Indeed, our results show that P2X1 is already a likely candidate, being up regulated by BzATP but not inhibited by oxATP. This is consistent with the concept that factors additional to ATP, such as IL-1 (Brough et al., 2002) or TNFα (Hide et al., 2000), and receptors additional to P2X7, such as P2X2 and P2X4 (Cavaliere et al., 2003), might be actively involved. Indeed, if metabolic impairment certainly occurs through the induction and activity of the P2X7 receptor, we do not exclude that complementary redundant receptors might be simultaneously recruited or compensatory mechanisms might be triggered (Cavaliere et al., 2001a, 2001b).
In this work, we also characterized the cellular and sub cellular distribution of P2X7 in hippocampal sections after oxygen/glucose deprivation. Not only this receptor specifically takes part in the metabolic impairment, it also owns specific phenotypic targets. The P2X7 signal is in fact present in fibrillary and parallel structures rising from the damaged region to the strata oriens and radiatum. Whereas absent in oligodendrocytes, astrocytes and neuronal cell bodies, it is instead localized in neurites at the synaptic level. This confirms the work by Sperlagh et al (2002) and Armstrong and colleagues (2002), which demonstrates the presynaptic localization of P2X7 in the CA1-CA3 region. Therefore, we could speculate that P2X7 present at the presynaptic membrane might serve as a sensor of dysregulated neuronal activity and ATP release, which both occur during oxygen/glucose deprivation. These events might in turn detrimentally over stimulate the P2X7 receptor, which is already known to require high doses of ATP to be activated (North and Surprenant, 2000). This would also explain the strong up regulation of P2X7 occurring only during the early stages of the hypoxic/hypoglycemic insult, when ATP is maximally released (Juranyi et al., 1999). Changes in receptor number are actually a well-studied mechanism for activity-dependent changes in synaptic efficacy.
Another important issue emerging from our study is the involvement of P2X7, such as P2X2 either in neurodegenerative events or in regenerative efforts. Since P2X7 seems not to be present on neuronal cell bodies, on differentiated cells such as astrocytes and oligodendrocytes present at the injury site, it is likely that its up regulation and presence at synaptic terminals could be coincident with the neuronal activity occurring through the injury. This may in turn be related to the ability of the ischemic tissue to undergo intercalary modulation toward both degeneration and regeneration. The most appealing question is now to understand in which of these opposite situations exactly the P2X7 receptor fills in, if not in both.
Finally, ischemic injury still maintains unanswered fundamental questions. The challenge remains not only to understand all the propagating mechanisms, but also to identify all the players. Our work contributes to reveal that the P2X7 receptor directly takes part to the metabolic impairment and that the cellular events, which are observed ex vivo to be mediated by the P2X7 during hypoxia/hypoglycemia, could contribute to the remodeling of the cellular functions seen in vivo.
Footnotes
Acknowledgment
The professional style editing of Mrs. Catherine Wrenn is gratefully acknowledged. We thank Dr. C. Sgobio for support on statistical analysis of the data.
