Abstract
Adenosine 5′-triphosphate outflow increases after an ischemic insult in the brain and may induce the expression of P2X7 receptors in resting microglia, determining its modification into an activated state. To assess the effects of P2X7 receptor blockade in preventing microglia activation and ameliorating brain damage and neurological impairment, we delivered the P2 unselective antagonist Reactive Blue 2 to rats after middle cerebral artery occlusion. In sham-operated animals, devoid of brain damage, double immunofluorescence verified the absence of P2X7 immunoreactivity on resting microglia, astrocytes, and neurons, identified, respectively, by OX-42, glial fibrillary acid protein, and neuronal nuclei (NeuN) immunoreactivity. After ischemia, vehicle-treated rats showed monolateral sensorimotor deficit and tissue damage in striatum and frontoparietal cortex. Moreover, P2X7 immunoreactivity was
Introduction
Brain infarction caused by an acute ischemic insult elicits secondary inflammatory responses involving infiltration of hematogenous macrophages and reaction of microglial cells in the neighboring areas (Dirnagl et al, 1999; Stoll et al, 1998). Macrophages and activated microglia are committed to remove necrotic cell debris from the infarct area, but it is unclear whether they do have also detrimental effects by inducing postischemic neuronal damage on reversibly damaged neurons localized in the so-called penumbra zone surrounding the infarct area (Wang and Feuerstein, 2004). Blocking the invasion of macrophages was indeed found helpful in reducing stroke volume after transient middle cerebral artery occlusion (MCAo) in rat (see Stoll et al, 1998), suggesting that pharmacological treatment of the secondary inflammatory responses may constitute an adjunctive neuroprotective resource against postischemic brain damage. However, no attempts have been performed so far specifically addressed to modulate the reaction of microglia after an ischemic insult.
Hours or days after focal ischemia, microglial cells change or lose their ramified morphology, release various cyto- or chemokines (Hanisch, 2001; Le Feuvre et al, 2002), as well as cytotoxic molecules, reactive oxygen species, and nitric oxide (Chao et al, 1992; Colton and Gilbert, 1993), leading to the so-called process of activation (Bruce-Keller, 1999; Stoll et al, 1998). These events may contribute to the inflammatory response that follows an ischemic insult, hence inducing a secondary brain damage (see Dirnagl et al, 1999). The triggers responsible for such microglial activation are not well understood, although evidence indicates that one likely candidate is adenosine 5′-triphosphate (ATP) (Collo et al, 1997; Di Virgilio et al, 1999).
Adenosine 5′-triphosphate is the main cellular energy metabolite that plays an important role in the extracellular milieu of the central nervous system as a neuromodulator, neurotransmitter, and trophic factor (Burnstock, 1997; Latini et al, 1995; Neary, 2000; Ralevic and Burnstock, 1998). Extracellular ATP evokes responses through two subclasses of P2 receptors: P2X, coupled to ligand-gated ion channels, and P2Y, coupled to GTP-binding proteins (Burnstock and Kennedy, 1985). Seven distinct ionotropic P2X and eight metabotropic P2Y receptors have been cloned so far from mammalian species (Khakh et al, 2001).
Interest is growing on the role played by ATP on P2 receptors during hypoxic/ischemic events. Adenosine 5′-triphosphate outflow increases in the brain during ischemia induced by MCAo (Melani et al, 2005) and data in the literature support the idea that under such conditions ATP exerts a cytotoxic role (Amadio et al, 2002, 2005; Cavaliere et al, 2001, 2002; Ryu et al, 2002). Moreover, the wide-range P2 antagonist suramin was shown to possess neuroprotective actions in the model of focal ischemia in rat (Kharlamov et al, 2002). Furthermore, the transition of microglial cells from resting into activated state is marked by
In the present study, we investigated whether P2 receptor-blocking agents could modify the P2X7 receptor expression and/or the morphological process of microglial activation after MCAo in rat. In detail, the effects of Reactive Blue 2 (RB2), a sulfonic derivative of anthraquinone and unselective antagonist of P2X-P2Y receptors (Amadio et al, 2002; Cavaliere et al, 2001), were investigated: (a) on the severity of neurological impairment and extent of necrotic neuronal damage induced by permanent focal ischemia in rat; (b) on the expression of the P2X7 receptor subtype and the process of microglial activation in areas close to and distant from a brain infarction.
Materials and methods
Male Wistar rats (Harlan, Italy) weighing 270 to 290 g were used. They were housed in groups of three with free access to food and water and kept on a 12 h light/dark cycle. The guidelines of the European Community for animal experiments were followed. All efforts were made to minimize animal suffering and to reduce the number of animals used.
Cerebral Focal Ischemia Model
The animals were randomly divided into four groups: animals sham operated or with focal cerebral ischemia were treated either with vehicle or RB2. Focal cerebral ischemia was induced by permanent MCAo in the right hemisphere. The animals were anesthetized with 5.0% isoflurane (Baxter, Kista, Sweden) and spontaneously inhaled 1.0% to 2.0% isoflurane in air by way of a mask. Body core temperature was maintained at 37°C with a recirculation pad and K module and was monitored via an intrarectal type T thermocouple (Harvard, Kent, UK). The surgical procedure to occlude the middle cerebral artery (MCA) consisted of insertion of a 4-0 nylon monofilament (Ethilon, Johnson & Johnson, Somerville, NJ, USA), precoated with silicone (Xantopren, Heraeus Kulzer, Germany) mixed with a hardener (Omnident, Germany), via the right external carotid artery into the internal carotid artery to block the origin of the MCA (Longa et al, 1989; Melani et al, 1999). The sham operation was conducted by inserting the filament into the internal carotid artery and immediately withdrawing it. At the end of the surgical procedure, anesthesia was discontinued and the animals were allowed to free access to food and water.
Physiological solution or RB2 (10 and 100 mg/kg), dissolved in saline, was injected intraperitoneally 5 mins after sham operation or MCAo. The RB2 doses administered were calculated on the basis of those found protective in
Motor Behavior
To assess the efficacy of the ischemic procedures, the rats were clinically tested. Soon after awakening from anesthesia and 24 h later, our rats were evaluated for failure to extend fully the left paretic forepaw and for turning on the paretic side when pulled by the tail, as reported previously (Hunter et al, 2000). All MCA occluded rats showed an evoked circling to the left side, contralateral to the MCAo.
Moreover, approximately 1 h after MCAo and thereafter, the rats showed spontaneous turning behavior contralateral to the ischemic hemisphere: placed in a round cage, the rats with MCAo were unidirectionally turning along the perimeter of the cage, chasing their tail. Five separate counting periods of 3 mins each, separated by 15-min intervals, were made between the 3rd and 4th hour and 15 mins after MCAo by an expert operator (Melani et al, 2003). Values are reported as the mean numbers of complete rotations during the five counting periods.
Neurological Test
Neurological evaluation of motor and sensory functions was performed 24 h before and after MCAo or sham operation. The examiners had no knowledge of the procedure and treatments that the rat had undergone. Adherence to a predetermined time excluded behavioral changes based on circadian rhythm. The neurological examination consisted of six standardized tests (Garcia et al, 1995b; Melani et al, 1999, 2003): the final minimum score per each rat was 3, indicating the most severe neurological impairment, and the maximum 18, indicating completely normal sensor motor functions, depending on the sum of the scores reported in each of the six tests.
Histological Analysis
After the neurological evaluation, the rats were anesthetized with chloral hydrate (intraperitoneal) and decapitated. The brains were rapidly removed and fixed with Liquid of Carnoy (6:3:1 absolute ethanol, chloroform, and glacial acetic acid) and then processed as described by Melani et al (1999). Briefly, the brain was embedded in paraffin after dehydration in different concentrations of ethanol and xylol. Coronal sections (7
Statistically significant differences in the histological damage and in the neurological impairment among sham-operated, vehicle- and RB2-treated rats (10 and 100 mg/kg) were evaluated by 1-way ANOVA followed by
Immunohistochemistry
Reactive Blue 2 at the higher dose (100 mg/kg, dissolved in saline), or vehicle alone, was injected intraperitoneally at 5 mins after sham operation or MCAo. At 24 h after surgical procedures, the animals were deeply anesthetized with 35% chloral hydrate (0.5 mL intraperitoneally) and perfused transcardially with 50 mL of 0.1 mol/L sodium phosphate buffer and 1% heparin, and with 250 mL of 4% paraformaldehyde in 0.1 mol/L sodium phosphate buffer (pH 7.4). The brains were removed and overnight post-fixed in the same fixative solutions, then washed three times in phosphate-buffered saline (PBS) and cryoprotected in 30% sucrose-PBS at 4°C. Transverse sections, 40
The sections were washed in PBS-1% BSA three times for 5 mins each and then incubated for 2 h in a solution containing a mixture of the secondary antibodies in 1% normal donkey serum in PBS. The secondary antibodies used for double labeling were Cy3-conjugated donkey anti-rabbit IgG (1:100, Jackson Immunoresearch, West Baltimore Pike, PA, USA, red immunofluorescence) and Cy2-conjugated donkey anti-mouse IgG (1:100, Jackson Immunoresearch, green immunofluorescence). After rinsing, the sections were finally mounted on slide glasses, allowed to air dry and coverslipped with gel/mount™ anti-fading medium (Biomeda, Foster City, CA, USA).
Double-label immunofluorescence was analyzed by means of a confocal laser-scanning microscope (CLSM) (LSM 510, Zeiss, Arese, Milan, Italy) equipped with an argon laser emitting at 488 nm and a helium/neon laser emitting at 543 nm. Immunohistochemical results were evaluated on the sensor motor cortex and striatum of both hemispheres in four representative sections (Bregma + 1.7, + 0.2, −1.8, and −3.3, according to Paxinos and Watson, 1986; Davies et al, 1998). In the hemisphere with the infarct, three areas were defined: (a) the infarct zone characterized by a complete loss of NeuN and GFAP immunopositive glial cells; (b) a narrow zone with some neuronal loss that was marginal to the infarct area; and (c) the surrounding area free from any apparent neuronal damage. Colocalization of P2X7 receptor subtype with GFAP, OX-42, or NeuN on astrocytes, microglia, and neurons, respectively, was qualitatively rated as present or absent. For the P2X7 immunostaining intensity, the labeling was scored as follows: 0 = no labeling; 1 = pale staining intensity sufficient to detect the presence and the outline of the positive cells; 2 = intense staining sufficient to evaluate the characteristics of cell body and cellular processes; 3 = brilliant immunostaining with evident cell body and processes. Differences in immunostaining intensity and in the topographic distribution of P2X7-positive microglial cells between ischemic control animals and ischemic animals treated with RB2 were evaluated by means of the two-tailed Student's
Results
Severity of Neurological Deficit and Ischemic Brain Damage
All sham-operated rats (
Effect of RB2 (10 and 100 mg/kg intraperitoneally) on turning behavior after right permanent MCAo
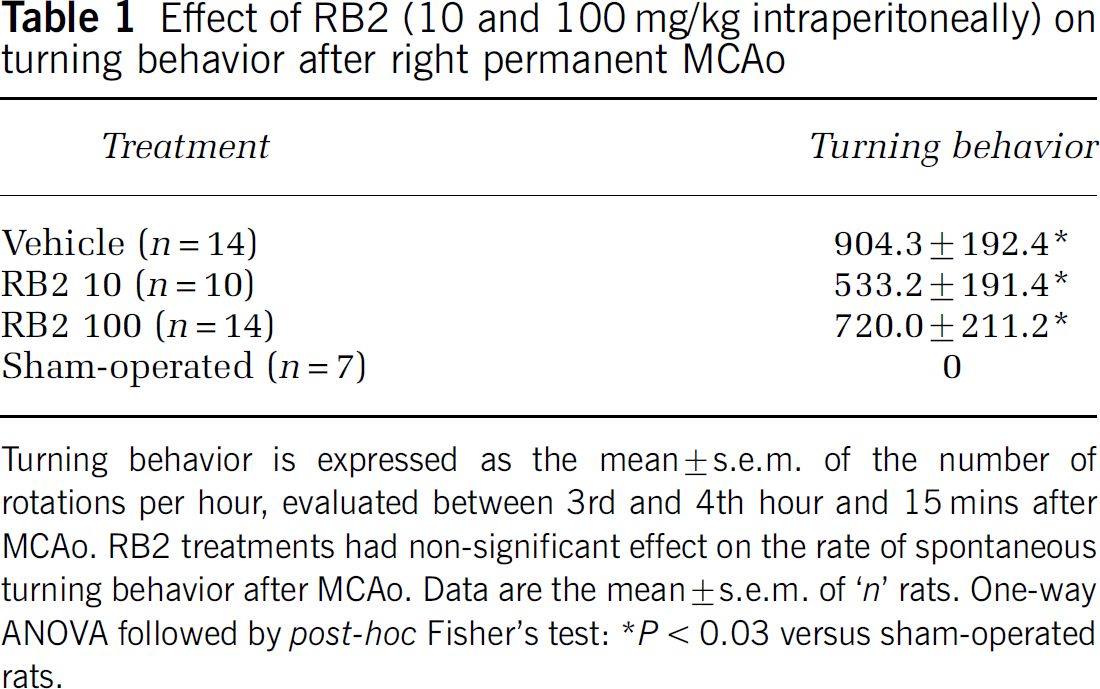
Turning behavior is expressed as the mean ± s.e.m. of the number of rotations per hour, evaluated between 3rd and 4th hour and 15 mins after MCAo. RB2 treatments had non-significant effect on the rate of spontaneous turning behavior after MCAo. Data are the mean ± s.e.m. of ‘
The neurological deficit was quantitatively evaluated 24 h after surgical operation and comparatively reported in Table 2 for all groups. Sham-operated rats showed a neurological score equal to 100% of the normal sensor motor functions, the same as before operation. Vehicle-treated rats after MCAo showed a clear impairment of sensory-motor functions with a neurological score reduced to 46%, in comparison with their own presurgical evaluations and with postsurgical evaluations of sham-operated rats. With respect to the vehicle-treated rats, the neurological score was significantly improved in the treated rats to 55% after RB2 10 mg/kg, and to 65% after RB2 100 mg/kg (Table 2).
Effect of RB2 (10 and 100 mg/kg intraperitoneal) on neurological deficit after permanent MCAo

Data are the mean ± s.e.m. of ‘
Rats were then killed and the ischemic brain damage was evaluated in the cortex and striatum by acetate cresyl violet staining (Figure 1). No tissue damage was found in the sham-operated rats (Figure 1A) or in the hemisphere contralateral to the ischemic side of each group. Definite ischemic tissue damage was instead found in the vascular territory supplied by the MCA, involving the striatum and the sensor motor cortex (Figure 1B). The lesioned area of the brain was detected as a pale zone lacking the acetate cresyl violet staining, and reflecting mainly the extent of unlabeled necrotic neurons 24 h after MCAo (Garcia et al, 1995a).
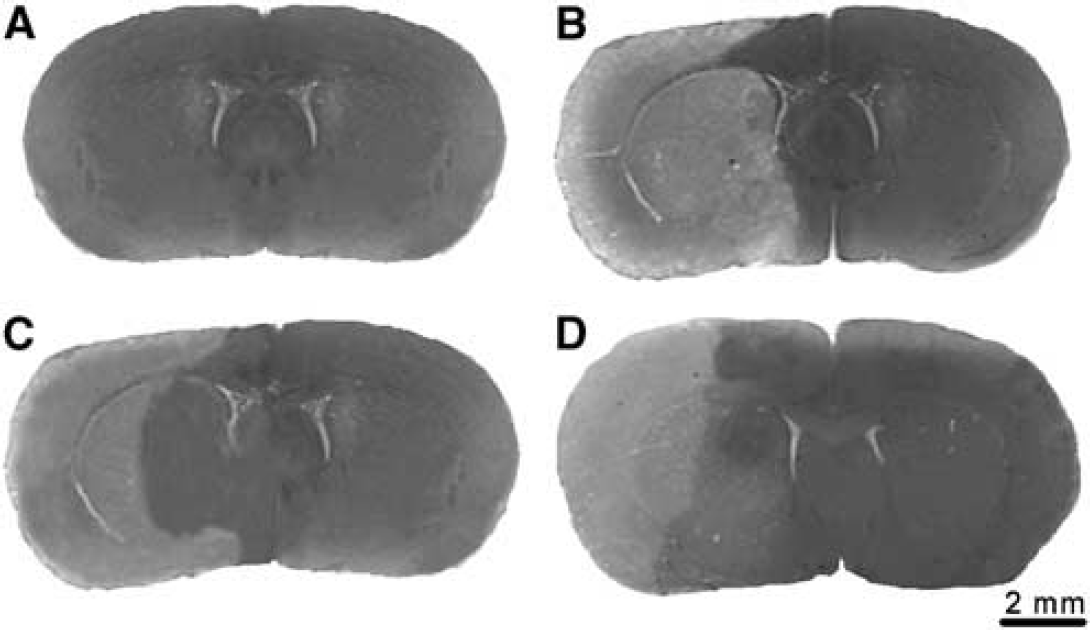
Cresyl violet stain in sham-operated and MCA occluded rat brains. Photomicrographs of brain coronal sections after sham operation or right permanent MCAo and acetate cresyl violet stain. (
In coronal sections, the necrotic area involved the frontal, parietal and piriform cortex and almost the entire caudate putamen; instead, the medial frontal (F2) and the cingulate cortex and, ventrally, the olfactory tubercle and the pallidum were spared. After administration of 10 mg/kg RB2, the extent of the ischemic pale area appeared reduced in the striatum, saving part of its medial region (Figure 1C). After administration of 100 mg/kg RB2, the extent of the pale ischemic area was reduced not only in the medial striatum but also in the sensor motor cerebral cortex, showing preservation of part of the piriform, ventrally, and dorsally of the frontal (F1 regions (Figure 1D). To evaluate the severity of the ischemic tissue damage, the volume of the infarct area was evaluated in vehicle- and RB2-treated rats. The extent of the tissue damage was 17.6 ± 1.7 mm3 in the striatum, and 46.0 ± 5.0 mm3 in the cerebral cortex of vehicle-treated rats; after RB2 treatment, we observed a dose-related effect on the infarct volume, which was significantly reduced by 29% only in the striatum, at the dose of 10 mg/kg, and by 43% in the striatum and by 49% in the frontoparietal cerebral cortex, at the dose of 100 mg/kg (Figure 2). A statistically significant correlation was found between the total (striatal plus cortical) infarct volume and the neurological score (
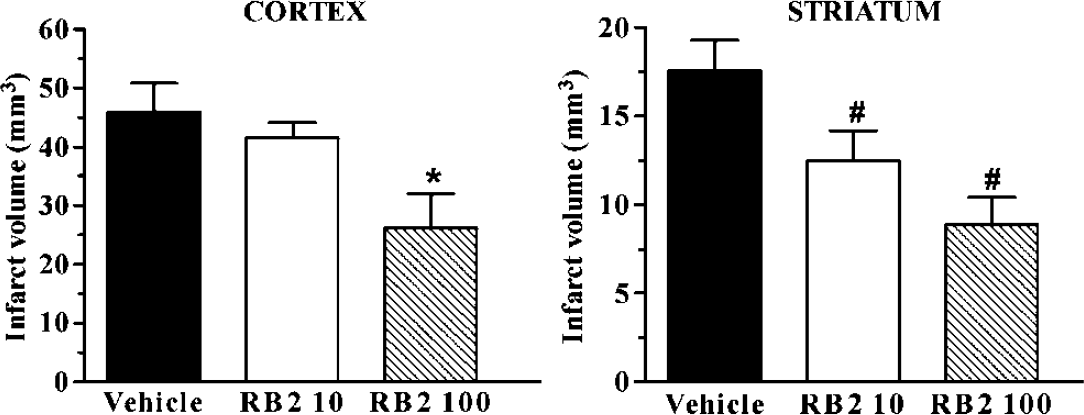
Effect of RB2 10 and 100 mg/kg on the volume of the ischemic brain damage. Cortical and striatal infarct volumes after permanent MCAo in vehicle-treated and RB2-treated rats. Reactive Blue 2 at 10 and 100 mg/kg intraperitoneally was administered 5 mins after MCAo. Infarct volumes are expressed in mm3. Data are the mean ± s.e.m. of ‘
Expression of the P2×7 Receptor Subtype
In sham-operated animals, NeuN immunohistochemistry was evaluated and showed positive NeuN of all neuronal types in the cerebral cortex and basal ganglia nuclei. Glial fibrillary acid protein and OX-42 immunoreactivity proved the presence of, respectively, astroglial cells and resting microglia, which were regularly scattered in the gray and white matter of both hemispheres. Conversely, in sham-operated animals, P2X7 immunoreactivity was not distinctly detectable in the cerebral cortex and striata of adult rat brain (data not shown), neither in vehicle (
On coronal sections of ischemic vehicle-treated animals (
The borders facing the infarct area were differently characterized in the cerebral cortex and the striatum. In the cerebral cortex, the infarct area was surrounded by a narrow zone (⩽
OX-42 and P2X7 immunoreactivities were both detectable in the infarct area, as well as in the surrounding undamaged tissues, nevertheless showing different morphological characteristics: in the infarct area (Figure 3A) extending over the frontal and parietal cortex, we distinguished sparse and small rod cells, likely macrophages; in the marginal zone (Figure 3B), small cells with short and thick processes, likely activated microglia. In the homolateral and even contralateral cingulate and medial frontal cortex and corpus callosum, we found small cells with ramified thin processes corresponding to the characteristic morphology of resting microglia (Figure 3C), showing, however, at the same time OX-42 and P2X7 immunoreactivity. We considered these last cells as reactive microglia, that is, in an intermediate state between the activated (with short thick processes, expressing P2X7 and OX-42 immunoreactivity) and the resting microglia (with ramified thin processes, OX-42 positive but without P2X7 immunoreactivity).
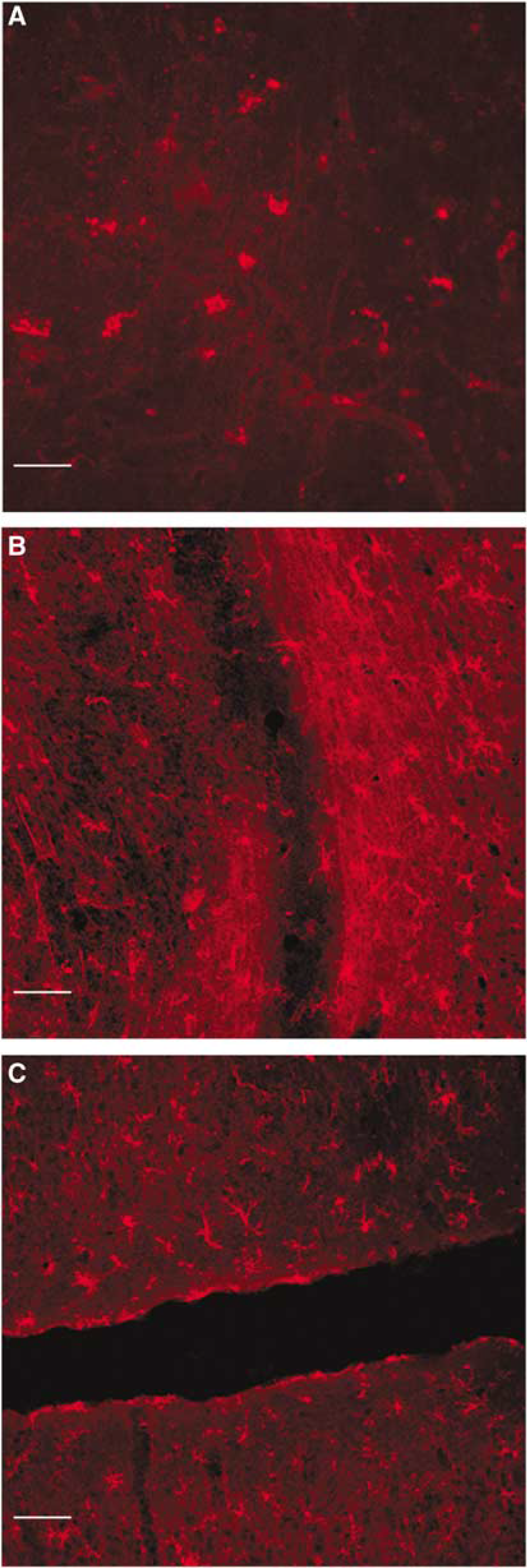
P2X7 immunoreactivity in and around a brain infarct. Fluorescence detection of P2X7 immunoreactivity 24 h after permanent MCAo in rat. (
While P2X7 immunoreactivity was clearly coexpressed with OX-42 in microglia, it did not colocalize with NeuN or with GFAP-positive cells in any brain area of the cerebral cortex and striata, (Figure 4). This suggests that P2X7 receptors are expressed in these areas only by activated or reactive microglial cells at 24 h after MCAo.
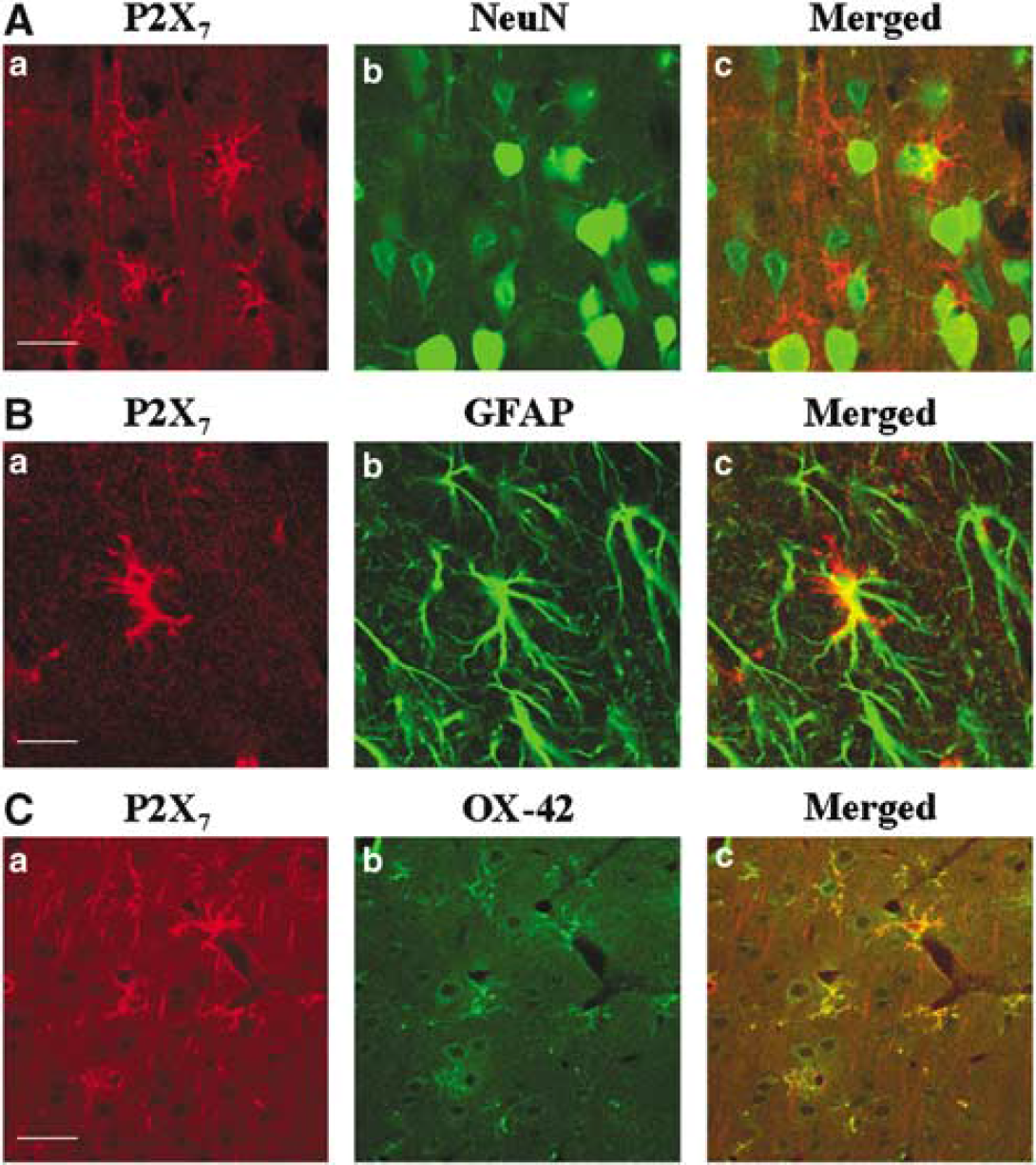
Localization of the P2X7 receptor in microglial cells, but not in astrocytes or neurons. Confocal images of double immunofluorescence were analyzed to characterize the localization of the P2X7 receptor (in red with Cy3 fluorescence) on neurons, astrocytes, and microglial cells (in green with Cy2 fluorescence) in the periinfarct area 24 h after MCAo. (
After focal ischemic necrosis and RB2 treatment (100 mg/kg intraperitoneally;
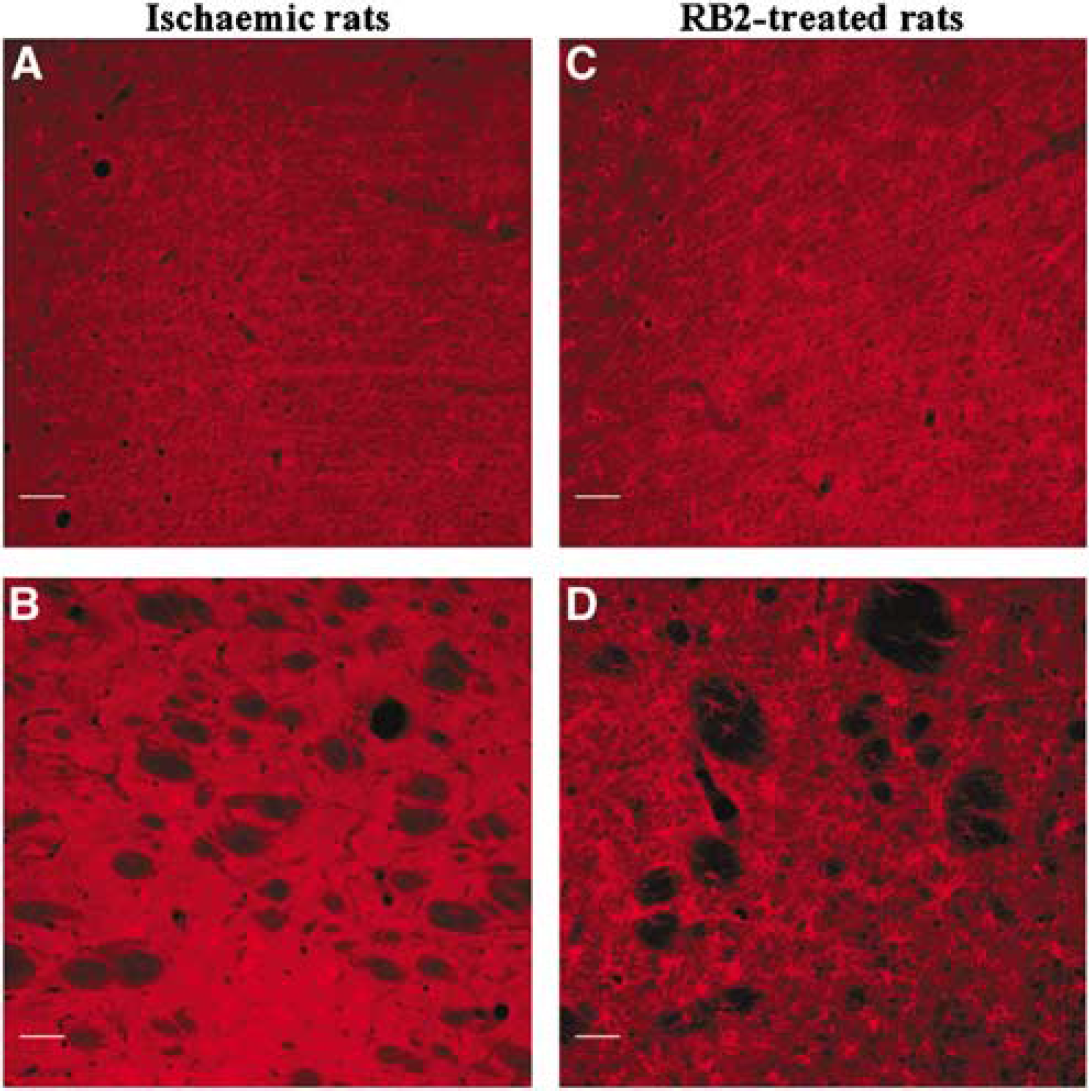
P2X7 immunoreactivity in remote areas from a brain infarct. Fluorescence detection of P2X7 immunoreactivity in the undamaged parietal cortex (
Discussion
The main finding of this paper is the recognition of a widespread
The topographic distribution of P2X7 receptors in microglial cells appeared by far larger than previously found at 24 h after cerebral ischemia (Collo et al, 1997; Franke et al, 2004). This can be explained by differences in immunohistochemical procedures, such as higher concentrations of primary and secondary antibodies, and/or the use of freezing microtome sections, which might have both improved the recognition of specific P2X7 immunoreactivity, and decreased the diffuse unspecific background staining. Finally, also the extent of the infarct volume could be an additional parameter conditioning the intensity of the response in the neighboring areas. Consistently, this precocious and extensive distribution of P2X7-positive microglial cells in areas remote from the brain infarct is in accordance with a previous integrated analysis of the progression of responses induced by permanent MCAo in the rat, clearly showing that the undamaged ipsilateral part of the medial frontal cortex and the equivalent contralateral zone contained activated microglia at 24 h (Davies et al, 1998).
Unexpectedly, after focal ischemia and RB2 treatment, the expression of the P2X7 in microglial cells was even larger than in vehicle-treated ischemic animals, involving also the entire frontal and parietal cortex and the caudate putamen in the contralateral undamaged hemisphere. In these contralateral areas, Davies et al (1998) have shown that activated microglial cells can be seen only 48 h after MCAo-dependent brain damage. Similar results have been observed
We have documented different aspects of the reaction of the P2X7-positive microglia, directed to a focal ischemic insult, morphologically and timely differentiated, at the primary site of the infarct as activated and amoeboid microglia, and in the remote projection areas as reactive microglia. These two steps of microglial activation sustain the concept of a modulatory role for ATP in focal brain ischemia (Boucsein et al, 2003) and regionally differentiated functions for microglial reactions (Block et al, 2005; Collo et al, 1997; Davies et al, 1998; Kreutzberg, 1996). A time-sustained increase in ATP outflow was recently described in the first hour after MCAo in the infarct area (Melani et al, 2005), so that high levels of ATP can likely boost the activation of microglial cells in the area of the primary damage (Boucsein et al, 2003), which in turn can produce inflammatory substances and induce detrimental effects on the neighbor tissue (Hanish, 2001; Hide et al, 2000; Sanz and Di Virgilio, 2000; Suzuki et al, 2004). Conversely, a self-sustained wave of low ATP release has been shown in the astrocytic network, spreading from the area of brain infarct to the remote projection areas (Boucsein et al, 2003; Charles, 1998; Schipke et al, 2002). This low level of ATP release may attenuate the reaction of microglia and the production of inflammatory cytokines outside the area of the focal ischemic insult (Boucsein et al, 2003; Suzuki et al, 2004), likely conferring to P2X7 reactive microglia-specific function in the reparative processes developing in the remote projection areas of the injured neurons. Consistently, it was recently shown that the basal activation of P2X7 receptor modulates mitochondrial ion homeostasis, increases cellular energy stores and cell survival, whereas a sustained stimulation of P2X7 receptor causes mitochondrial fragmentation and cell death (Adinolfi et al, 2005).
Previous studies failed to show any neuroprotective action of P2X receptor antagonists in mice, particularly oxidized ATP and pyridoxalphosphate-6-azophenyl-2′,4′-disulfonic acid, injected in the lateral ventricle immediately before and after transient MCAo, suggesting that P2X7 receptors are not primary mediators of experimentally induced neuronal death (Le Feuvre et al, 2002, 2003). There was, however, no proof that these compounds actually reached the target cells in a sufficient amount. Species differences and an effective diffusion of RB2 through a disrupted blood-brain barrier could explain the discrepancy between the present and previous studies (Le Feuvre et al, 2003). Even though the actions of RB2 appear to be complex (Amadio et al, 2002), our results suggest that RB2 limited the extent of the brain infarct probably acting on P2X7 receptors expressed by activated microglial cells, and probably exerting its protective effect by reducing their production of toxic inflammatory compounds.
Although the antagonist-induced P2X7 upregulation strongly supports the view that RB2 is an effective P2X7 targeting agent, its failure to prevent the microglial transition from resting into activated state and phagocytes in and around the acute infarct area suggests that ATP may act also through P2 receptors not blocked by RB2. Alternatively, ATP could boost the function of already activated microglial cells (Boucsein et al, 2003). However, further work is necessary to fully clarify the mechanisms of RB2 against the ischemic damage. Little, indeed, is known on the kinetics of RB2 injected
In conclusion, our data suggest that microglial cells expressing P2X7 receptors can be implicated in the process of tissue damage evolving in and around the infarct area, as well as in the defense and reparative processes consequentially developing in the ipsi- and contralateral undamaged but deafferented regions. The two processes, although regionally interconnected, are well differentiated in nature and in time. Both of them should be taken into account by future pharmacological approaches targeting P2 receptor expression and functions, for the prevention of the ischemic and postischemic brain damage.
Footnotes
Acknowledgements
The authors thank Mrs Laura Calosi for her collaboration in preparing histological specimens.
