Abstract
Matrix metalloproteinases (MMPs) are activated in focal cerebral ischemia. The activation of MMP-9 is involved in blood—brain barrier breakdown and tissue remodeling. The MMPs are released to the extracellular space, but the form and fate of secreted enzymes in brain are unknown. Using microdialysis in vivo, the authors studied whether ischemia-induced MMP-9 in brain tissue was related to free MMP-9 in the extracellular fluid. A microdialysis probe was placed into the right striatum and microdialysis was initiated 24 hours later in controls (n = 7). One hour prior to microdialysis, a group of rats (n = 7) was subjected to 1-hour occlusion of the right middle cerebral artery, followed by reperfusion. Dialysates were collected at discrete time points up to 24 hours, and subjected to zymography and Western blot analysis. The MMP-9 was released after ischemia and accumulated in the extracellular space at 24 hours (P < 0.05). Free MMP-9 forms include mainly the 95-kd proform, and, to a lesser extent, dimers and cleaved active forms (70 kd), but not the 88-kd form found in tissue. Probe implantation and microdialysis increased free MMP-9 in the dialysate. This increase was concomitant with neutrophil infiltration after the mechanical lesion, as myeloperoxidase was found by means of Western blot analysis in the brain hemisphere subjected to microdialysis (P < 0.005), and immunohistochemistry revealed the presence of myeloperoxidase stain surrounding the site of probe implantation. The results suggest that certain forms of MMP-9 are released and accumulate in the extracellular space after brain injury, and that vascular alterations and neutrophil recruitment elicit MMP-9 activation in the brain after focal ischemia and trauma.
Acute cerebral ischemia alters the cellular environment and affects neural cell viability through multiple alterations. Both the extracellular matrix and the extracellular fluid are part of the neural cell environment. The former maintains cells and tissues together and is a dynamic space suitable for intercellular communication. It consists of a complex network of macromolecules, mainly made of proteoglycans, that intensely interacts with cell membranes and blood vessels and that has regulatory and modulator properties (Bovolenta and Fernaud-Espinosa, 2000;Yamaguchi, 2000).
The extracellular matrix of the brain is not very extensive, compared with that in other tissues, because of the high cellular density and abundance of cell processes, but it is needed for the formation of perineuronal nets (Celio et al., 1998), in synaptic clefts (Yamaguchi, 2000), and around the endothelial wall of blood vessels (Yurchenco and Schittny, 1990). The basal lamina, which is located between the outer vessel wall and the surrounding astrocytic processes, is a specialized matrix that gives structural support to blood vessels and ensures integrity of the blood—brain barrier. Several zinc-and calcium-dependent matrix metalloproteinases (MMPs), like MMP-2 and MMP-9 (gelatinase A and B, respectively), degrade components of the basal lamina, causing disruption of the blood brain—barrier (Mun-Bryce and Rosenberg, 1998;Rosenberg et al., 1992, 1995). Microvascular basal lamina antigens disappear after cerebral ischemia (Hamann et al., 1995) and there is evidence that MMP-9 and MMP-2 are involved in blood—brain barrier breakdown after ischemia (Gasche et al., 1999;Rosenberg et al., 1998). Matrix metalloproteinases increase in the brain after ischemia in animal models (Fujimura et al., 1999;Gasche et al., 1999;Heo et al., 1999;Planas et al., 2000, 2001;Romanic et al., 1998;Rosenberg et al., 1994, 1996, 2001) and in humans (Clark et al., 1997). Increase of MMP-9 in brain is associated with hemorrhagic transformation (Heo et al., 1999), and increase of MMP-9 in plasma is predictive of hemorrhagic transformation after cardioembolic stroke (Montaner et al., 2001). Pharmacologic inhibition of MMPs reduces thrombolytic-induced hemorrhage after thromboembolic middle cerebral artery (MCA) occlusion in rabbits (Lapchak et al., 2000), decreases infarct volume after focal ischemia in rodents (Asahi et al., 2000;Romanic et al., 1998), and prevents oxidative stress-associated blood—brain barrier disruption after transient MCA occlusion in mice (Gasche et al., 2001). Also, deletion of the MMP-9 gene reduces infarct volume after ischemia (Asahi et al., 2000).
Matrix metalloproteinases contribute to signaling between the extracellular environment and cells, and to tissue degradation after brain lesion (Bruno et al., 1998;Lukes et al., 1999;Mun-Bryce and Rosenberg, 1998), as they degrade myelin basic protein (Gijbels et al., 1993) and other neural proteins and peptides (Backstrom and Tökés, 1995). Furthermore, MMPs are involved in various diseases of the central nervous system (Kieseier et al., 1999;Lukes et al., 1999;Yong et al., 2001). Cells synthesize MMPs as proforms, with the catalytic side hidden from substrates. Upon activation, they can suffer proteolytic cleavage, which exposes their active sites (Springman et al., 1990;Woesser, 1991). Matrix metalloproteinase 9 is released from the producer cells to degrade components of the extracellular matrix, although the extracellular release of MMP-9 in the brain following ischemia is unknown. In the present study, we aimed to test whether MMP-9 is released and accumulates in the extracellular fluid after ischemia in vivo, to determine its time course, and to identify the released MMP forms. This information will contribute to a better understanding of the mechanism of MMP-9 activation in the brain, which will lead to further designs of inhibitory drugs. Microdialysis is a tool for sampling interstitial compound concentration within the local brain tissue environment where the microdialysis probe is inserted (Adell and Artigas, 1997). We used in vivo microdialysis in the rat to measure MMP-9 in dialysates as an indicator of the release to the extracellular fluid within the 24 hours that follow MCA occlusion with reperfusion.
MATERIALS AND METHODS
In vivo brain microdialysis
Male Sprague-Dawley rats (280–320 g) obtained from Iffa-Credo (Lyon, France) were kept in a 12-hour light-dark cycle and allowed free access to food and water. Animal work was conducted in compliance with the Spanish legislation regarding the protection of animals used for experimental and other scientific purposes, and in accordance with the directives of the European Union on this subject. Rats were subjected to cerebral microdialysis following the method of Adell and Artigas (1998), with modifications. The day before ischemia, the rats were anesthetized with 4% halothane (Fluothane; Zeneca) in a mixture of 70% N2O and 30% O2 through a face mask. They were placed on a stereotaxic frame (Kopf Instruments) and anesthesia was maintained with 1.5% to 2% halothane. After making a cranial midline skin incision, a microdialysis probe (MAB2; Microbiotech, Stockholm, Sweden) was implanted into the right striatum through a burr hole at the following co-ordinates: 1.2 mm anteroposterior, 2.5 mm lateral, and 6 mm ventral to bregma (Paxinos and Watson, 1986). The membrane on the probe was made of polyether sulphone and had an outer diameter of 0.6 mm and a length of 3 mm. The probe was fixed to the skull with two miniature screws and dental cement. The wound was sutured and the animal was allowed to recover for 24 hours. Thereafter, one-hour transient MCA occlusion was carried out in the hemisphere ipsilateral to probe implantation (n = 7), and reperfusion was allowed. Rats were allowed to recover from anesthesia for 30 minutes, placed in a plastic cage with a mounted liquid swivel system (Instech Laboratories, Plymouth Meeting, PA, U.S.A.) and microdialysis was initiated. In a group of control rats, microdialysis was initiated without any intervention, other than probe implantation the day before (n = 7). Microdialysis was carried out in the awake, freely moving rat by pumping artifical CSF containing 125 mmol/L NaCl, 2.5 mmol/L KCl, 1.18 mmol/L MgCl2, and 1.26 mmol/L CaCl2 at a constant rate (1 μL/min) and samples were collected as 10-minute fractions. In preliminary experiments (n=4), perfusion and fraction collection was continuously carried out for about 10h, but this procedure led to continuous draining and did not allow for significant accumulation of MMPs in the dialysates. Before dialysate collection, an initial wash with artificial CSF (10 μL/min) was performed 30 minutes after 1-hour ischemia (i.e., 24 hours after probe implantation). Microdialysis was then carried out discontinuously for 40 minutes (as four 10-minute fractions) at 0.5, 5.5, 10.5, and 24 hours after the initial 10-minute wash. Thus, the first dialysate was taken after 1 hour of reperfusion. Thereafter, certain rats subjected to microdialysis and controls (rats not subjected to any surgical procedure) were anesthetized, killed, and the ipsilateral and contralateral cortex and striatum were removed. Tissues and microdialysis samples were immediately frozen in liquid nitrogen and kept at −80°C until further analysis.
Focal cerebral ischemia
Focal cerebral ischemia was produced by transient intraluminal occlusion of the MCA according to the method of Longa et al. (1989), with modifications as previously reported (Justicia et al., 2001). Briefly, rats were anesthetized with 4% halothane in a mixture of 70% N2O and 30% O2. After tracheal intubation for controlled ventilation, anesthesia was maintained with 1% to 1.5% halothane. The left femoral artery was cannulated to monitor blood pressure, and body temperature was maintained at 37.5°C with a heating pad controlled by a thermoregulatory system connected to a rectal probe. A 2.6-cm length of 3–0 monofilament nylon suture heat-blunted at the tip was introduced into the external carotid artery through a puncture. In addition, both common carotid arteries were clamped to minimize collateral circulation, as reported elsewhere (Soriano et al., 1997). After 50 minutes of ischemia, the clip on the left common carotid artery was released and reperfusion was visually assessed. Ten minutes later the filament was gently removed and the clip on the right common carotid artery was released. Following surgery, rats were allowed to recover spontaneous breathing and were kept in their cages with free access to food and water.
Gel zymography
Gel zymography was carried out with dialysate samples (10 μL) or brain tissue samples previously homogenized and subjected to extraction of gelatinase activity following the method of Zhang and Gottschall (1997), with modifications (Planas et al., 2001). Gels containing 10% acrylamide and porcine gelatin (1 mg/mL) were prepared. Samples were loaded in zymography loading buffer containing 80 mmol/L Tris-HCl (pH 6.8), 4% sodium dodecyl sulphate (SDS), 10% glycerol, and 0.01% bromophenol blue. A known amount of MMP-9 and MMP-2 standard (CC073; Chemicon International) was loaded in one lane of each gel. SDS/PAGE was carried out at 25 mA at 4°C. Gels were briefly washed in distilled water and then three times for 15 minutes each using 150 mL 2.5% Triton ×100 at room temperature, before incubation in 250 mL buffer containing 50 mmol/L Tris-HCl (pH 7.5), 10 mmol/L CaCl2, and 0.02% NaN3 for 42 hours at 37°C. After the incubation period, gels were stained in 150 mL 0.1% amido black (naphthol blue black; Sigma) in a 1:3:6 mixture of acetic acid:methanol:distilled water for 1 hour at room temperature. Gels were then unstained in 150 mL 1:3:6 acetic acid:methanol:distilled water four times for 30 minutes each, and were then washed in distilled water for 20 minutes.
Western blot
The presence of MMP-9 protein in dialysates was studied by Western blot analysis. Dialysate samples (10 μL) were mixed with loading buffer (as for zymography) and were loaded in 10% polyacrylamide gels for electrophoresis. Proteins were then transferred to a membrane (Immobilon-P, Millipore) that was incubated overnight at 4°C with the primary antibody, mouse monoclonal antibody against MMP-9 Ab2 (MAB 13420; Chemicon) diluted 1:150. In brain tissue, myeloperoxidase (MPO) expression was studied by Western blot analysis in the same rats used for microdialysis. For this study we used an aliquot of crude tissue homogenates, as MPO is tightly bound to membranes. Protein content in these fractions was determined (Bradford assay; Bio-Rad). Samples were diluted with loading buffer containing DTT, heated at 100°C for 5 minutes, and loaded on a denaturing 10% polyacrylamide gel for electrophoresis. Proteins were transferred to a membrane incubated with mouse monoclonal antibody against MPO (Menarini Diagnostics) diluted 1:500. A rabbit polyclonal antibody against actin (Sigma) diluted 1:10,000 was used as a control for protein gel loading. Secondary antibodies were peroxidase-linked antimouse or antirabbit Ig (Amersham), diluted 1:2,000. The reaction was developed with a chemiluminescence reagent containing luminol.
Immunohistochemistry
At 48 hours after probe implantation (24 hours after MCA occlusion) rats were perfused with 4% paraformaldehyde and their brains were postfixed overnight with the same fixative, embedded in paraffin, and cut into 5-μm thick coronal sections using a microtome. Paraffin was removed, endogenous peroxidases were blocked with methanol-H2O2, and unspecific-binding sites were blocked with 10% normal goat serum for 2 hours. Sections were then incubated with a rabbit polyclonal antibody against MPO (diluted 1:800; A 0398; Dako) for 2 hours at room temperature, biotinylated goat antirabbit antibody (diluted 1:200; Vectastain; Vector) for 1 hour, and the avidin-biotin complex (diluted 1:100; ABC kit; Vector Laboratories) for 1 hour. The reaction was developed with 0.05% diaminobenzidine and 0.03% H2O2.
Data analysis
Gels were scanned with a Kodak camera (DC-120) and analyzed with appropriate software (Kds1D; Kodak). In order to compare data from different gels, raw band intensity values of the samples were normalized by calculating the ratio between their net band intensity and net band intensity of an MMP-9 standard in each gel. We then applied two-way ANOVA by treatment (ischemic or control) and time (of sample collection), followed by the Bonferroni post hoc test. Values are expressed as mean ± SEM of net band intensity measures. The MPO data from Western blot analyses were also measured (expressed as percent of mean control), and analyzed with one-way ANOVA and the Bonferroni test.
RESULTS
Matrix metalloproteinases in the tissue and dialysates 24 hours after middle cerebral artery occlusion/reperfusion
Zymography in gelatinolytic extracts from brain homogenates revealed that MMP-9 and MMP-2 are expressed in the control rat brain (Fig. 1A). This technique allowed us to detect several bands, mainly one MMP-9 proform band of 95 kd and one intermediate form of 88 kd, together with one form of MMP-2 that was approximately 65 kd and the most intense band in the gel. In addition, MMP-9 forms dimers found at 210 kd. Band intensity for MMP-9 increased at 24 hours after ischemia in extracts of brain homogenates (Fig. 1A). The in vivo microdialysis technique revealed several bands in the dialysates, even in control rats not subjected to ischemia (Fig. 1B). Bands detected in the dialysate were 95-kd MMP-9 and dimeric MMP-9 forms, and, to a lesser extent, MMP-2, whereas the 88-kd form of MMP-9 found in tissue was not detected in the dialysate (Fig. 1B). At 24 hours after MCA occlusion/reperfusion, MMP-9 bands in the dialysate were more intense than in the corresponding timed controls (Fig. 1B). The presence of a 95-kd MMP-9 proform in the extracellular space was also evidenced by Western blot analysis of the dialysate samples (Fig. 1C).
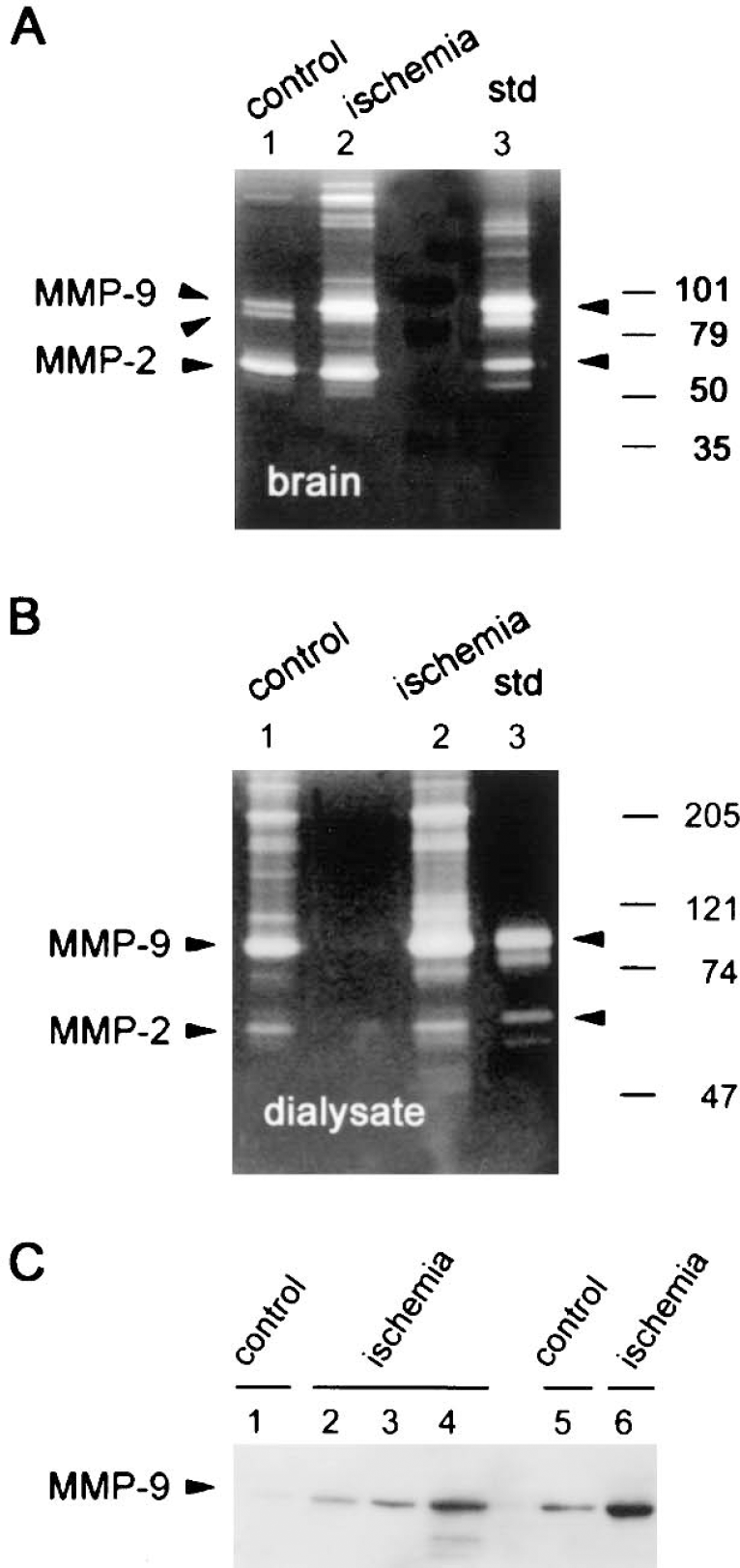
Gel zymography
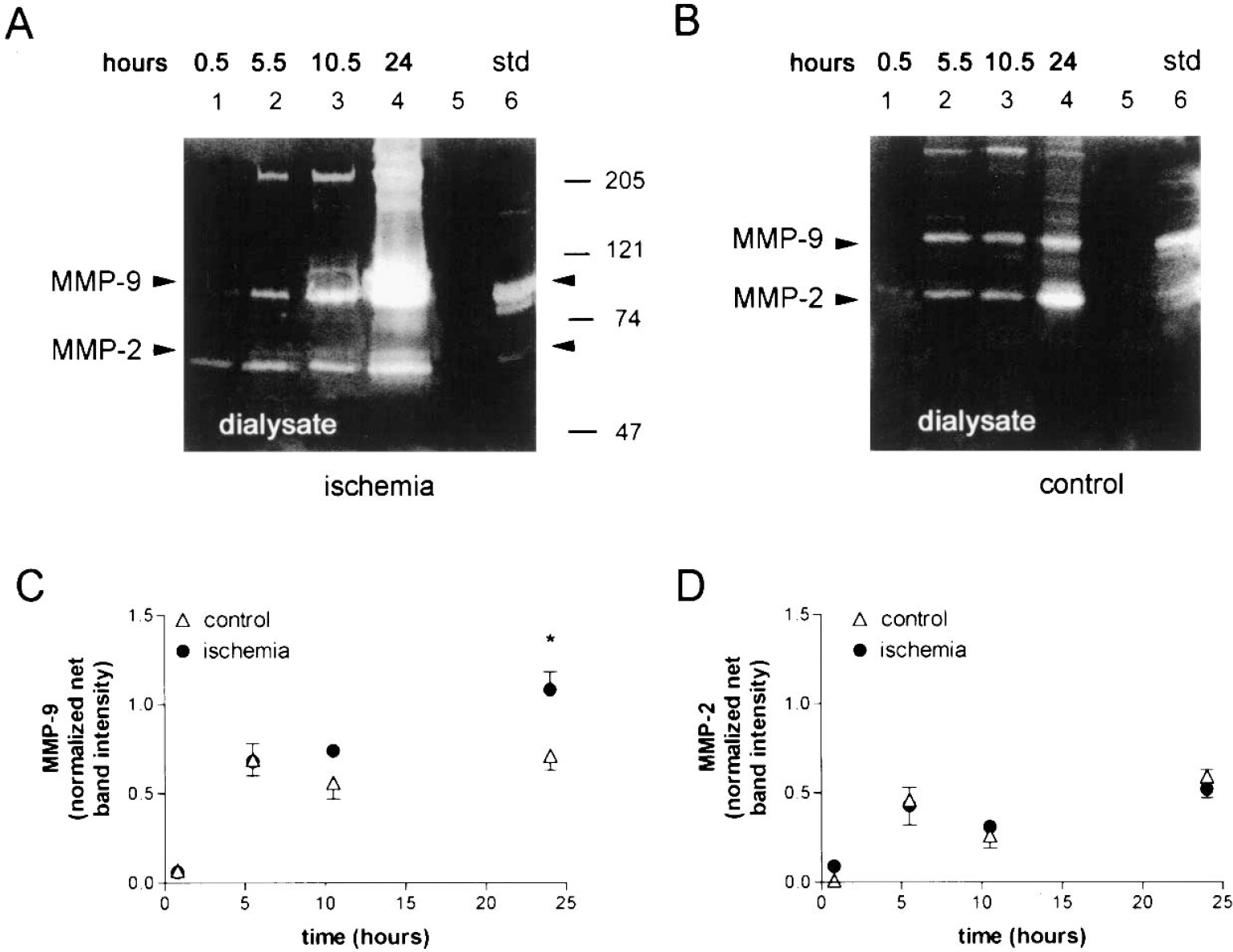
In vivo brain microdialysis: time course
Zymographic analysis of dialysates showed bands corresponding to MMP-9 and MMP-2 proforms in ischemic (Fig. 2A) and control (Fig. 2B) rats. Matrix metalloproteinases were found in the dialysates of all ischemic rats (six), whereas no signal was detected in two out of six controls. These two control rats were not considered in the statistical analysis in order to find differences between controls and ischemic rats showing MMP-9. The intensity of the 95-kd MMP-9 band after ischemia was higher than in controls and accumulated at 24 hours, which was the longest time point studied here (Figs. 2A and 2B). A two-way ANOVA, by treatment (ischemic versus controls) and time, showed a significant difference after ischemia (F1,28 = 5.68, P < 0.05), and the Bonferroni post hoc test showed that ischemia increased 95-kd MMP-9 at 24 hours (P < 0.05) compared with controls. Western blot analysis also showed that the abundance of free MMP-9 protein increased from the beginning of microdialysis (Fig. 1C, lane 2: 0.5 hours, and lane 3: 10.5 hours) to 24 hours (Fig. 1C, lanes 4 and 6) after ischemia, and to a lesser extent in controls (Fig. 1C, lanes 1 and 5).
Source of matrix metalloproteinase-9
We investigated whether neutrophils had infiltrated the tissue by examining MPO expression at the end of the study (i.e., 48 hours after probe implantation). Indeed, we found that nonischemic rats subjected to probe implantation and microdialysis showed MPO expression in homogenates of the ipsilateral, but not the contralateral, hemisphere, to an extent comparable with that seen in the corresponding ischemic rats (Fig. 3A). Measures of MPO net band intensity in Western blots followed by one-way ANOVA showed that, compared with the contralateral hemisphere, the ipsilateral hemisphere had a 7-fold and 10-fold increase in control (P < 0.05) and ischemic rats (P < 0.01), respectively.
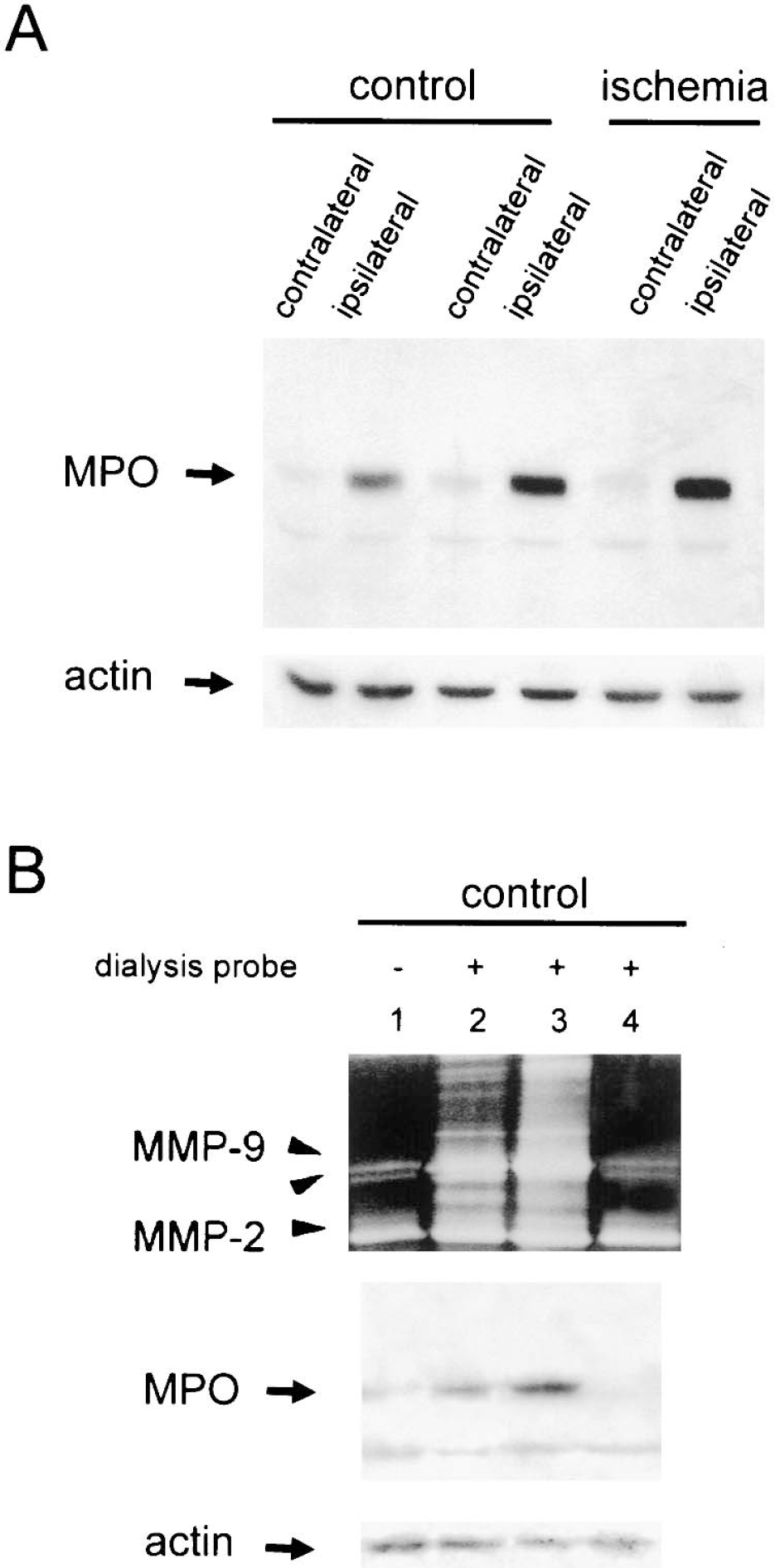
We examined MMPs in the brains of control (nonischemic) rats subjected to microdialysis and compared the dialyzed hemisphere with the nondialyzed hemisphere and with that of nonoperated controls. Microdialysis largely increased 95-kd MMP-9 proform and MMP-9 dimers in the brain (Fig. 3B), an effect that might be attributable to the mechanical injury caused by the intracerebral probe. This effect was concomitant with the appearance of MPO expression in brain tissue (Fig. 3B). We investigated whether brain MMP-9 had increased in the two control rats subjected to microdialysis that showed no MMP-9 in the dialysates. We found no MMP-9 increase in these brains and no MPO was detected (Fig. 3B), suggesting that those rats suffered little traumatic damage after probe implantation.
In rats showing MMP-9 in dialysates, immunohistochemistry against MPO showed the massive presence of neutrophils in the area surrounding the site of microdialysis probe implantation (Figs. 4A and 4B, control; Figs. 4C and 4D, ischemia). Infiltration of isolated neutrophils within the parenchyma was exclusively seen in rats subjected to ischemia (Figs. 4E and 4F).
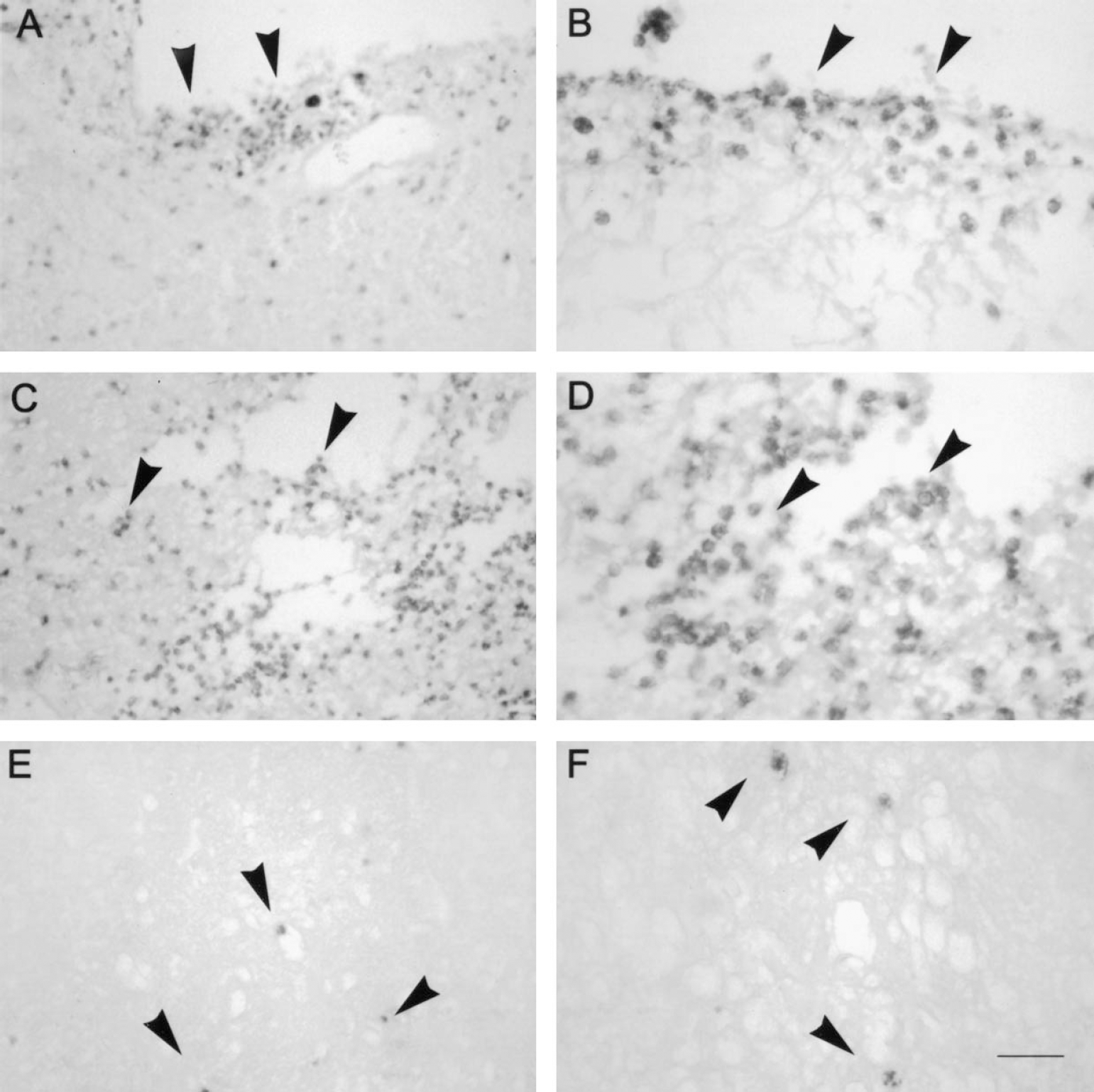
Immunohistochemistry against myeloperoxidase (MPO; 48 hours after microdialysis probe implantation; i.e., 24 hours after ischemia) shows neutrophil accumulation (dark cells) in the area surrounding the site of probe implantation (arrows in
Free and bound matrix metalloproteinase-9 forms
The 88-kd MMP-9 form detected in tissue homogenate was not observed in brain dialysates (Fig. 1). Before zymography, brain homogenates were subjected to extraction of gelatinolytic activity (see Materials and Methods), whereas brain dialysates were directly analyzed. A similar procedure was used for dialysate samples to rule out that the 95-kd form had been cleaved to the 88-kd form in vitro during extraction. However, the 88-kd MMP-9 form was not detected in the extracted dialysate (data not shown). Therefore, this form was not soluble in the extracellular fluid, and is likely associated with cells or bound to the matrix. In addition to the 95-kd MMP-9 proform, which was the major MMP-9 form in the dialysate, and the dimeric forms, a minor band of lower molecular weight (approximately 70 kd) was observed at 24 hours when the fraction corresponding to the proform was very high (Figs. 1B and 2).
DISCUSSION
Matrix metalloproteinases contribute to basement membrane degradation and blood—brain barrier breakdown, and play many other roles (Mun-Bryce and Rosenberg, 1998). Here we show, by in vivo microdialysis, that 95-kd MMP-9 progressively accumulated in brain dialysates from the striatum during reperfusion after focal ischemia. This finding suggests that MMP-9 is released and accumulates in the extracellular space after ischemia. Matrix metalloproteinase 9 was also detected in the dialysates of some control brains, although this does not necessarily imply that MMP-9 is normally free in the extracellular space. Indeed, we have evidence suggesting that the mechanical lesion caused by microdialysis probe implantation is responsible, at least in part, for 95-kd MMP-9 accumulation in the dialysates from controls. First, the brain hemisphere subjected to microdialysis in controls showed increased intensity of MMP-9 zymographic bands compared with the nonoperated hemisphere and with rats not subjected to probe implantation. Second, we found the presence of MPO expression (Wright et al., 1987) in the control brains subjected to microdialysis at 48 hours after probe implantation, and MPO-stained neutrophils surrounded the site of probe implantation. Two out of six controls showed no MMP-9 in the dialysates, and no raise in MMP-9 levels was detected in the brains of those rats, which did not show MPO expression. These findings support the idea that vascular damage and neutrophil infiltration are involved in MMP-9 release to the extracellular space after brain injury. Indeed, injury caused by probe implantation disrupts the blood—brain barrier in the vicinity of the probe, which leads to vasogenic edema (Dykstra et al., 1992). This appears to be a transient alteration and the integrity of the blood—brain barrier may recover after 1 hour of probe implantation (Benveniste et al., 1984;Terasaki et al., 1992). However, the blood—brain barrier remains permeable to small molecules for a few days after probe implantation, although it recovers earlier for permeability to proteins (Morgan et al., 1996). A priori, we decided to carry out microdialysis 24 hours after inserting the probe as the barrier should be at least partially recovered. However, the initial lesion facilitated capillary damage and neutrophil recruitment.
It is likely that a mechanical lesion caused by the microdialysis probe induces MMP-9 accumulation in the extracellular space at the time ischemia was induced (24 hours after probe implantation). Then ischemia would progressively contribute to extracellular MMP-9 accumulation for the following 24 hours, as a significant increase of free MMP-9 was found in the dialysates of rats subjected to ischemia compared with control dialysates. Previous studies using reverse transcription polymerase chain reaction showed increased MMP-9 mRNA production after ischemia (Asahi et al., 2000), showing that ischemia triggers de novo synthesis of MMP-9 proform. Similarly to MMP-9 expression, infiltration of neutrophils peaks 24 to 48 hours after cerebral ischemia (Garcia et al., 1994) and traumatic brain injury (Royo et al, 1999). Neutrophils are rich in MMP-9 (Kjeldsen et al., 1992), and MMP-9 labeling has been detected by immunohistochemistry in infiltrated neutrophils after ischemia (Romanic et al., 1998;Rosenberg et al., 2001). Furthermore, blockade of neutrophil infiltration reduces MMP-9 accumulation in the brain after ischemia (C. Justicia and A.M. Planas, unpublished data, 2002). Therefore, it is feasible that neutrophils themselves are a source of extra-MMP-9 expression and activity in the brain and contribute to the induction of MMP-9 expression in the tissue, as has been found in the myocardium following ischemia/reperfusion (Lindsey et al., 2001). However, the detection of infiltrating neutrophils in the tissue subjected to microdialysis does not fully rule out other sources of MMP-9.
Early increases in MMP-9 proform have been reported in brain tissue homogenates as soon as 2 to 4 hours after permanent MCA occlusion in mice (Asahi et al., 2000;Gasche et al., 1999). Likewise, MMP-9 is enhanced after 3 to 4 hours of reperfusion following 1-hour MCA occlusion in mice (Fujimura et al., 1999;Gasche et al., 2001) and rats (Planas et al., 2000). These early increases in MMP-9 are further enhanced at 10 to 15 hours and at 24 hours in permanent ischemia in rats (Romanic et al., 1998) and mice (Asahi et al., 2000), and after ischemia/reperfusion in rats (Planas et al., 2001;Rosenberg et al., 1998). We detected significant accumulation of MMP-9 only in the extracellular fluid from 12 to 24 hours after ischemia, but not earlier. However, we may have failed to detect low levels of MMP-9 for two reasons. First, microdialysis can induce neutrophil infiltration to the damaged striatum and MMP-9 release. Second, we probably obtained only a fraction of what is in the extracellular fluid in the dialysate, as we do not know the recovery of the technique. These constraints prevented us from determining the exact time at which ischemia-induced MMP-9 release was initiated. However, the finding of accumulation of MMP-9 in the extracellular space at 24 hours is robust and consistent with the results reported for tissue homogenates.
In addition to the increase in the 95-kd MMP-9 proform, a MMP-9 form of lower molecular weight (88 kd), which was taken as an active MMP-9 form (Zhang et al., 1998), has been described in brain tissue (Planas et al., 2000, 2001;Zhang and Gotshall, 1997;Zhang et al., 1998). In ischemia, the 88-kd form increases during the first hours of reperfusion in mice (Fujimura et al., 1999) and rats (Planas et al., 2000), and after permanent ischemia in mice (Gasche et al., 1999). However, the MMP forms that we found in the dialysates did not include the 88-kd form, thus implying that it is not soluble in the extracellular fluid. This form may be trapped in the extracellular matrix or it may bind to the cells, as there is evidence that certain MMP-9 forms are associated with cytoskeletal proteins or other structural proteins (Nelson and Siman, 1989).
In contrast to MMP-9, no major accumulation of MMP-2 due to ischemia was detected in the dialysate at 24 hours. Previous examinations of brain tissue homogenates in rats showed no major MMP-2 increases at 24 hours, but persistent activation was found at 4 to 5 days after focal ischemia/reperfusion (Planas et al., 2001;Rosenberg et al., 1998) and permanent focal ischemia (Romanic et al., 1998) associated with macrophagic cells.
In conclusion, ischemia induced progressive MMP-9 release into the extracellular space and accumulation by 24 hours. Mechanical injury caused by microdialysis triggered release of MMP-9 that was accompanied with the presence of neutrophils. The major form of MMP-9 in the dialysates was the 95-kd proform and dimeric forms, whereas the 88-kd form observed in the tissue was not found in dialysates. We propose that vascular alterations and neutrophil infiltration contribute to MMP-9 release and accumulation in the extracellular space of the brain after mechanical injury and focal ischemia.
