Abstract
To investigate changes in dopamine release in the striatum during motor exercise in human subjects with and without striatal dopamine denervation, eight healthy subjects and eight patients with Parkinson disease (PD) were measured during unilateral foot extension/flexion movement using positron emission tomography with [11C]raclopride. Five subjects in each group were later scanned in the resting condition. Estimation of binding potential (k3/k4) of [11C]raclopride was based on Logan plot method. Significant reductions in [11C]raclopride k3/k4 were found in the dorsal putamen contralateral to the exercise side in the healthy group and ipsilaterally in the PD group. Spearman rank correlation analysis showed that [11C]raclopride k3/k4 correlated inversely with the decrease in performance (velocity and motion range) in the dorsal putamen contralaterally in the healthy group and ipsilaterally in the PD group. These results suggest that simple but laborious motor exercise (motor stimulation) generates significant dopamine release in the dorsal striatum contralateral to the motor execution in humans. Lack of the crossed pattern and ipsilateral increase in dopamine release in the dorsal striatum during the unilateral limb movement may reflect the pathophysiology for hypokinetic and insufficient coordinating movement in PD.
Keywords
Recent imaging studies indicate that mental manipulation with and without movement can change significant levels of dopamine release in the entire striatum (de la Fuente-Fernandez et al., 2001; Koepp et al., 1998). The bilateral dopamine activation has also been found in subjects after long-distance walking with fatigue (Ouchi et al., 2001). Although the dopamine release reported thus far is associated with mental activities, it remains unclear that motor tasks could increase dopamine release in the striatum or to what extent the striatum could be affected by the motor performance. It was shown that the basal ganglia serve motor control functions such as scaling or focusing of movements (Alexander and Crutcher, 1990; Alexander and DeLong, 1985; Mink and Thach, 1991). Dopamine contribution to this motor function is easily understood in view of motor impairment in parkinsonism. Thus, comparison of the normal condition with the dopamine-depletion state seen in Parkinson disease (PD) also allows us to study the dynamic aspect of pathophysiology in PD by taking advantage of D2 and −3 receptors ligand [11C]raclopride ([11C]RAC), the binding of which reportedly varies depending on synaptic endogenous dopamine level
A recent somatotopic mapping study showed that the somatotopic organization of foot representation resides in the striatum (putamen) more dorsally and more rostrally to the hand area (Maillard et al., 2000). The presence of this functional segregation pattern coincides with a known observation on PD that the nigral dopaminergic projections to the putamen are more affected than the medial striatum (Kish et al., 1988). These findings lead to the possible assumption that striatal dopamine release induced by unilateral motor task would present a region-specific alteration in [11C]RAC binding in the striatum in normal condition and different responses in PD. The present study was designed to shed light on the effect of motor exercise on regional dopamine release in the striatum
MATERIALS AND METHODS
Patients
Eight healthy, right-handed (right-footed) subjects (male/female: 6/2; mean age ± SD: 58.0 ± 13.3 y) and eight age-matched, right-handed (right-footed) drug-naive PD patients (5/3; 61.4 ± 7.5) rated at stage 1 to 2 on the Hoehn and Yahr scale participated in the motor task study. Five subjects in each group participated in the resting condition PET study performed within one month of the motor task study. Clinical assessment of each PD patient was performed with the Unified Parkinson's Disease Rating Scale and the Mini-Mental State Examination (Table 1). Magnetic resonance imaging (MRI) study disclosed no abnormalities in the brain of each participant. Diagnostic
Clinical characteristics of patient with Parkinson disease
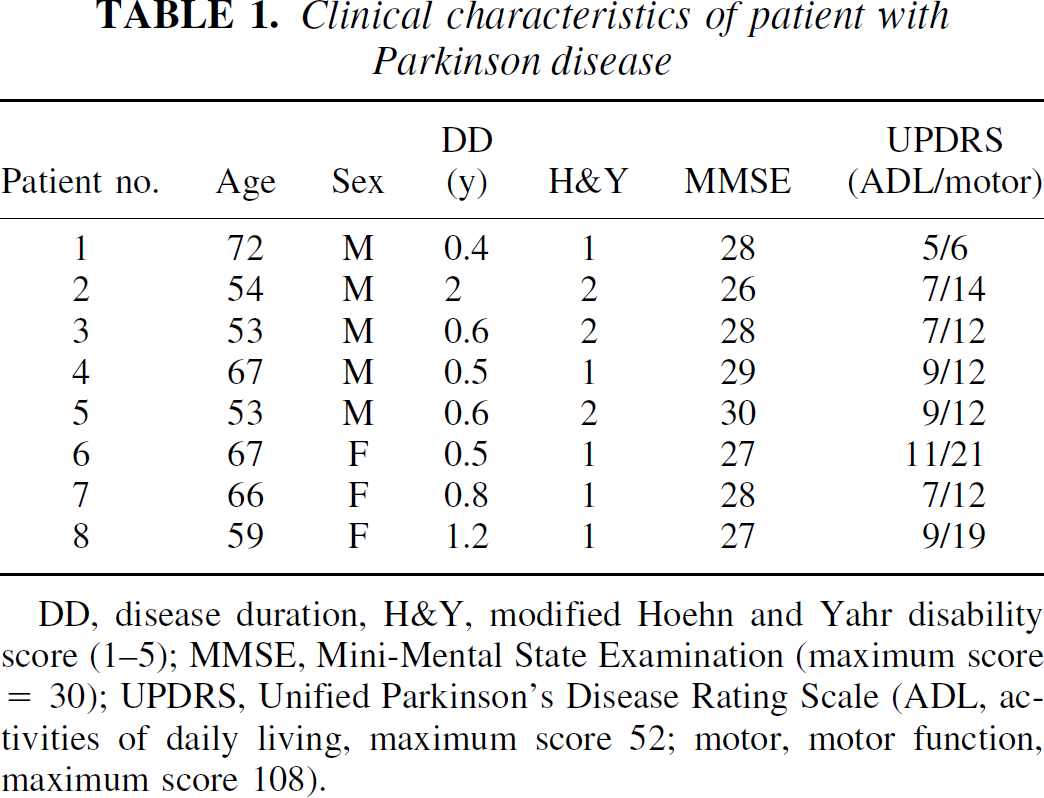
DD, disease duration, H&Y, modified Hoehn and Yahr disability score (1–5); MMSE, Mini-Mental State Examination (maximum score = 30); UPDRS, Unified Parkinson's Disease Rating Scale (ADL, activities of daily living, maximum score 52; motor, motor function, maximum score 108).
Task performance
Healthy subjects were instructed to perform right foot sequential extension/flexion movement at their own speed (close to 1 Hz) for 62 minutes in the supine position. Patients with PD were required to move the foot on the hypokinesic side at their own pace (close to 0.5 Hz). During the performance, time course changes of the following physiologic parameters were monitored: arterial blood pressure, pulse rates, and Pa
Magnetic resonance imaging and positron emission tomography procedures
The procedure of MRI study was reported elsewhere (Ouchi et al., 1998). Briefly, MRI with three-dimensional-mode data acquisition was performed just before PET measurement using a static magnet (0.3 T MRP7000AD; Hitachi, Tokyo, Japan) in order to determine areas of the striatal nuclei for setting regions of interest (ROIs). In reference to measures of tilt angle and spatial coordinates obtained in the procedure for determining the intercommissural (AC-PC) line on each subject's sagittal MR images, a PET gantry was set parallel to the AC-PC line by tilting and moving the gantry for each study, which permitted the reconstruction of PET images parallel to the AC-PC line without reslicing (Ouchi et al., 1999).
Positron emission tomography was performed using a high-resolution brain PET scanner (SHR2400; Hamamatsu Photonics K.K., Hamamatsu, Japan) with a capability of the gantry tilting from −20 to +90 and moving vertically and with in-plane spatial resolution of 2.7 mm at full width at half maximum (Yamashita et al., 1990). The gantry was set parallel to the AC-PC line determined by MRI (as described in the preceding paragraph), and a 20-minute transmission scan for attenuation correction was performed with a Ge/6 Ga source.
In PET study, a thermoplastic face mask designed for radio-surgery was used to fix the head to the same place between the two separate PET measurements (task and rest). Serial scans (time frames: 4 × 30 s, 20 × 60 s, 8 × 300 s) and periodic arterial blood sampling were performed for 62 minutes after a slow bolus injection (taking 1 minute) of a 370-MBq dose of [11C]RAC. To determine radioactive metabolites, additional arterial blood samples were drawn at 1, 5, 20, and 30 minutes after injection and analyzed using thin-layer chromatography and a storage-phospher-screen bioimaging analyzer (BAS-1500; Fuji Film, Tokyo, Japan). The free metabolite-corrected plasma activities were fitted to a sum of three exponentials by the nonlinear least squares method with the nonweighted Gauss-Newton algorithm.
Region of interest data analysis and statistics
As shown in Fig. 1, irregular ROIs (64 ∼ 800 mm2) were drawn bilaterally over the ventromedial striatum (head of the caudate), the inferolateral part (ventral putamen), and the superodorsal part (dorsal putamen) of the striatum and the cerebellum on the MR images (Mai et al., 1997; Ouchi et al., 1999) and transferred onto the corresponding [11C]RAC PET images using image-processing software (Dr View; Asahi Kasei Co., Tokyo, Japan) on a SUN workstation (Hypersparc ss-20; SUN Microsystems, CA, U.S.A.) (Ouchi et al., 1999). One PET image that integrated data of 6.5-mm thickness (Yamashita et al., 1990) as a volume radioactivity contained the functional information of the striatum at least 6.5 mm deep in the z direction. Thus, in the present study we regarded the ROI value on a single PET image as a regional (volume) value of ligand uptake. Binding potential, Bmax‘/kd (or k3/k4), was estimated using the following equation and the nonlinear least squares fitting method: Bmax‘/kd = (target tissue Vd)/(cerebellum Vd) −1, where each Vd (distribution volume) was obtained by the Logan graphical method (Lammertsma et al., 1996; Logan et al., 1990, 1994). Although age-related reduction was reported in D2 receptors (Antonini et al., 1993), the correction for aging for PET results was not performed because no significant difference in age was observed between the PD and the healthy groups (
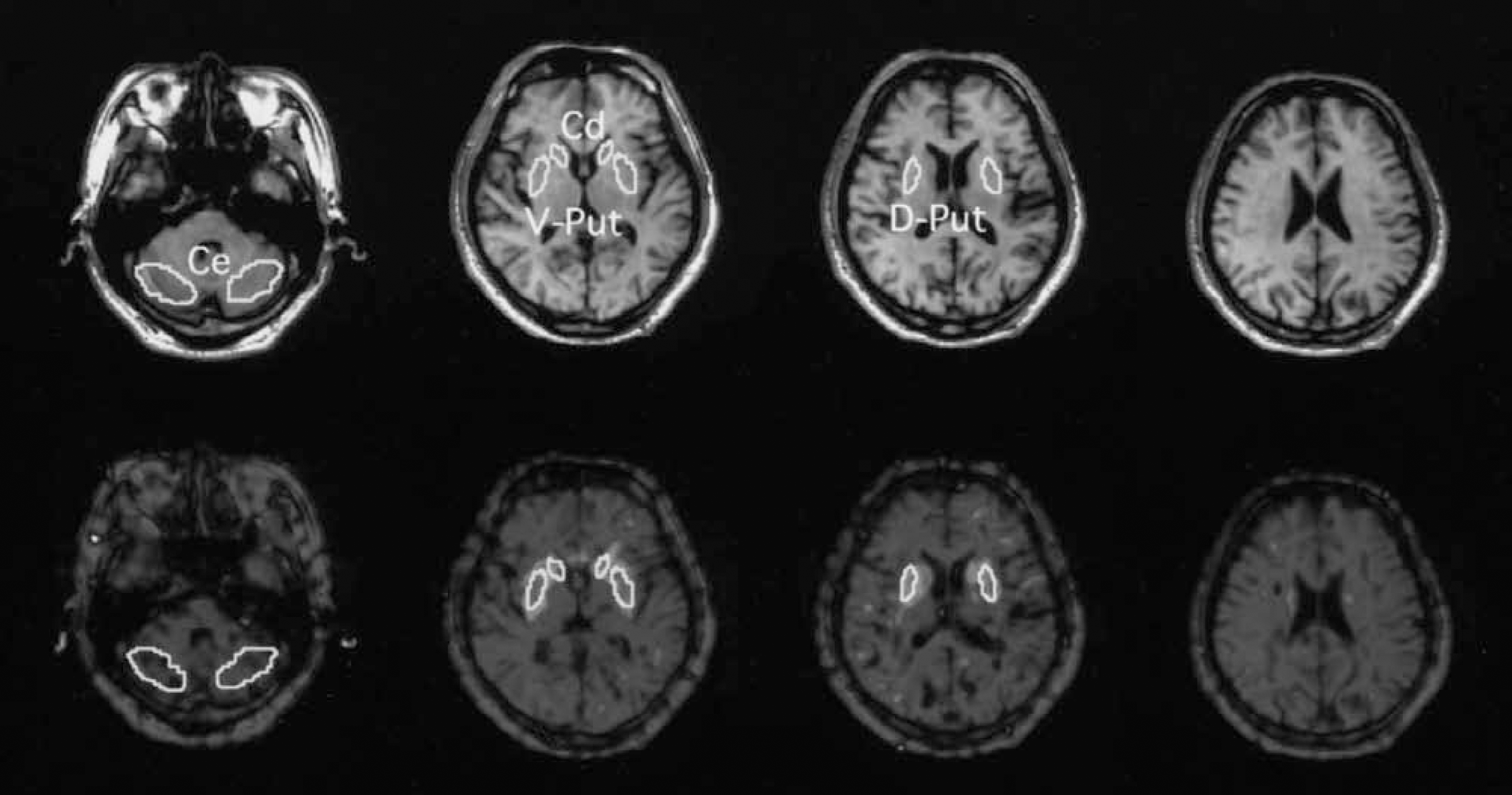
Setting of regions of interest (ROIs). Irregular ROIs, drawn bilaterally on the concerned regions on the magnetic resonance images (upper row), were placed on the corresponding positron emission tomography images (bottom row). Ce, cerebellum; Cd, caudate; V-Put, ventral putamen; D-Put, dorsal putamen.
Statistical parametric mapping analysis on [11C]raclopride image
To investigate brain regions showing positive correlation between reduction in receptor binding (increase in dopamine release) and increase in burden of performance, a voxel-wise-based correlation analysis was made using statistical parametric mapping (SPM99; Wellcome Department of Cognitive Neurology, London, U.K.) (Friston et al., 1995). Based on the Logan graphical method yielding DV values, the image was generated from a measured DV image (Drevets et al., 1999). With SPM, spatial normalization of the DV image to the standard stereotaxic brain atlas (Talairach and Tournoux, 1988) was performed using transformation parameters for early integrated images of [11C]RAC from 0 to 20 minutes after injection (Ito et al., 1999). After image normalization, symptom-(task-)induced hemispheric side difference in the PD group was effaced by flipping images so that the side of activation contralateral to the exercising foot was set on the left in all PD images. After smoothing images with an isotropic Gaussian kernel of 8 mm,
RESULTS
Physiology and performance
There were no significant changes in physiologic parameters (mean arterial blood pressure, pulse rate, Pa
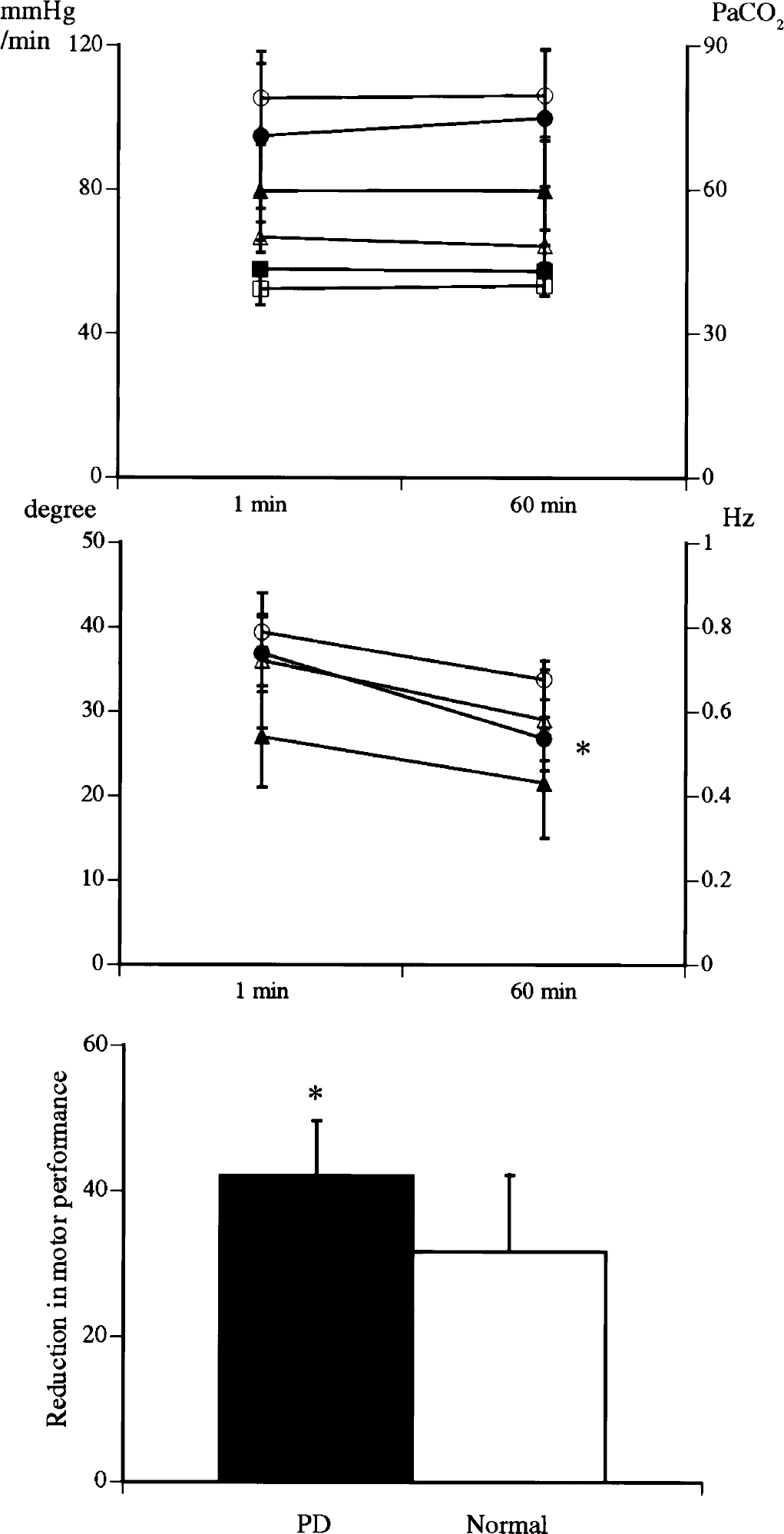
Changes in mean arterial blood pressure (
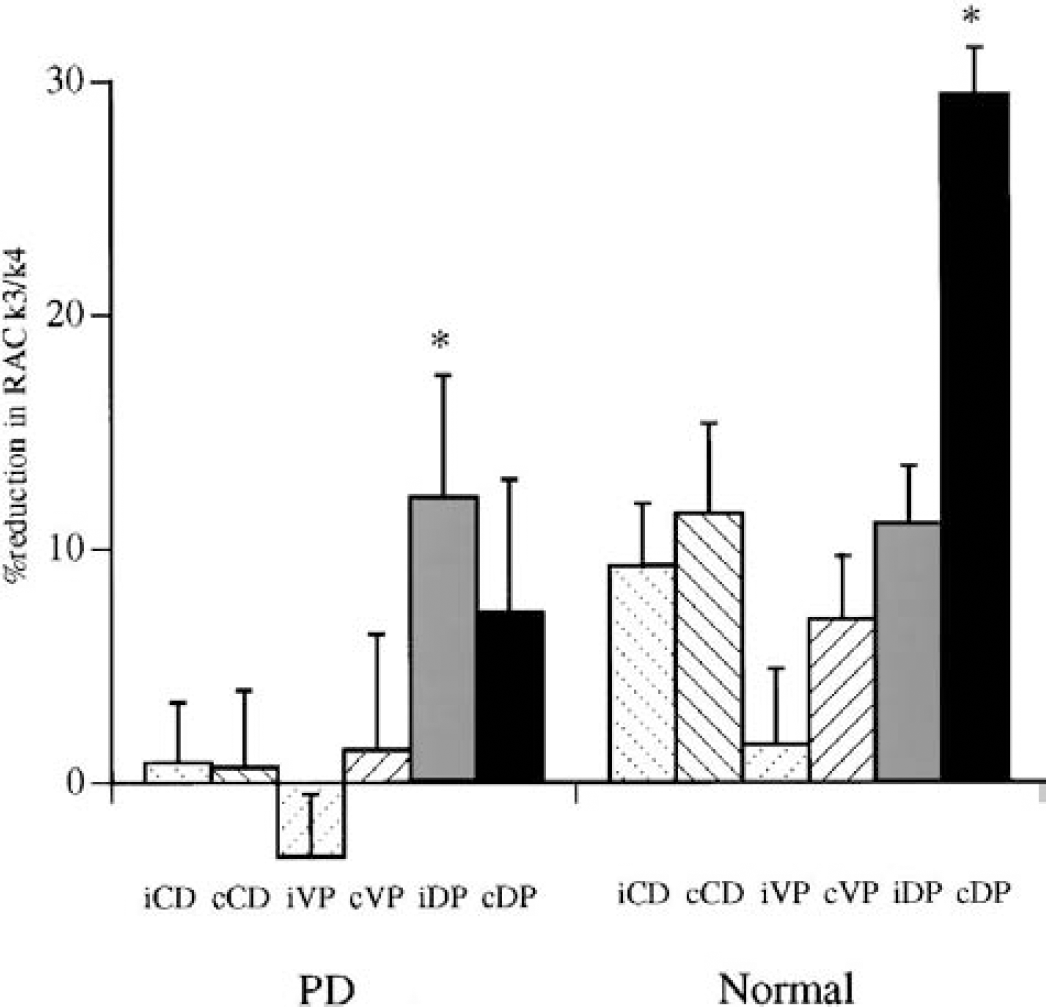
The magnitude of percentage of reduction in [11C]raclopride k3/k4 during motor exercise compared with the resting condition. An asterisk denotes a significant difference by repeated measures ANOVA (
Levels of binding potentials for [11C]raclopride in exercise and resting conditions
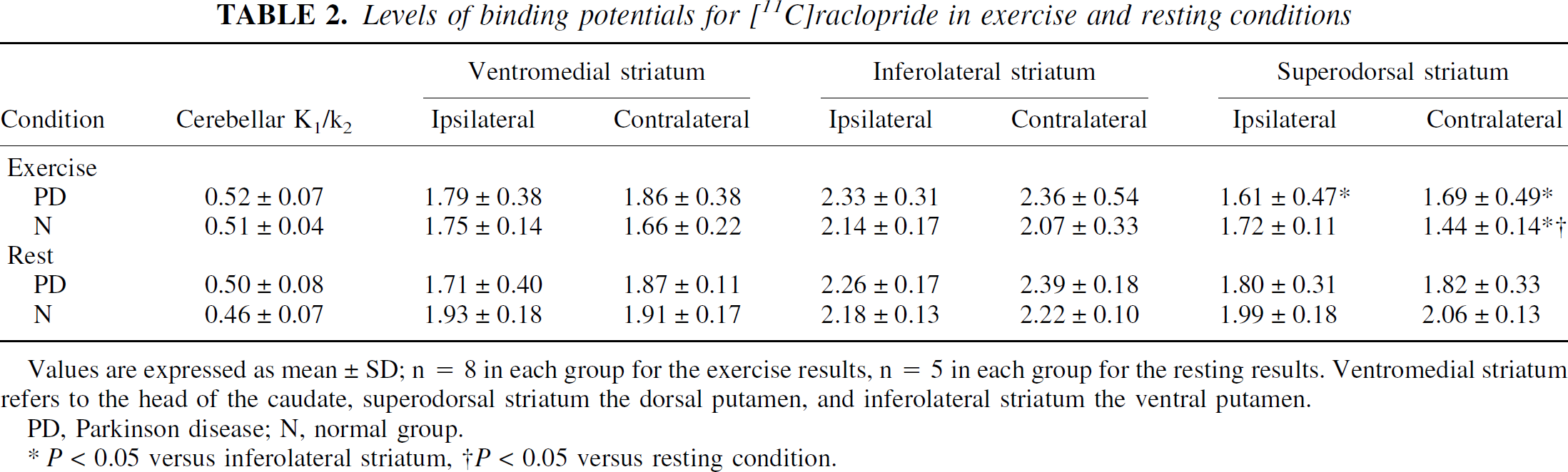
Values are expressed as mean ± SD; n = 8 in each group for the exercise results, n = 5 in each group for the resting results. Ventromedial striatum refers to the head of the caudate, superodorsal striatum the dorsal putamen, and inferolateral striatum the ventral putamen.
PD, Parkinson disease; N, normal group.
Spearman rank correlation analysis showed that [11C]RAC k3/k4 values correlated inversely with the performance reduction in the dorsal putamen contralateral to the motion side in healthy group (
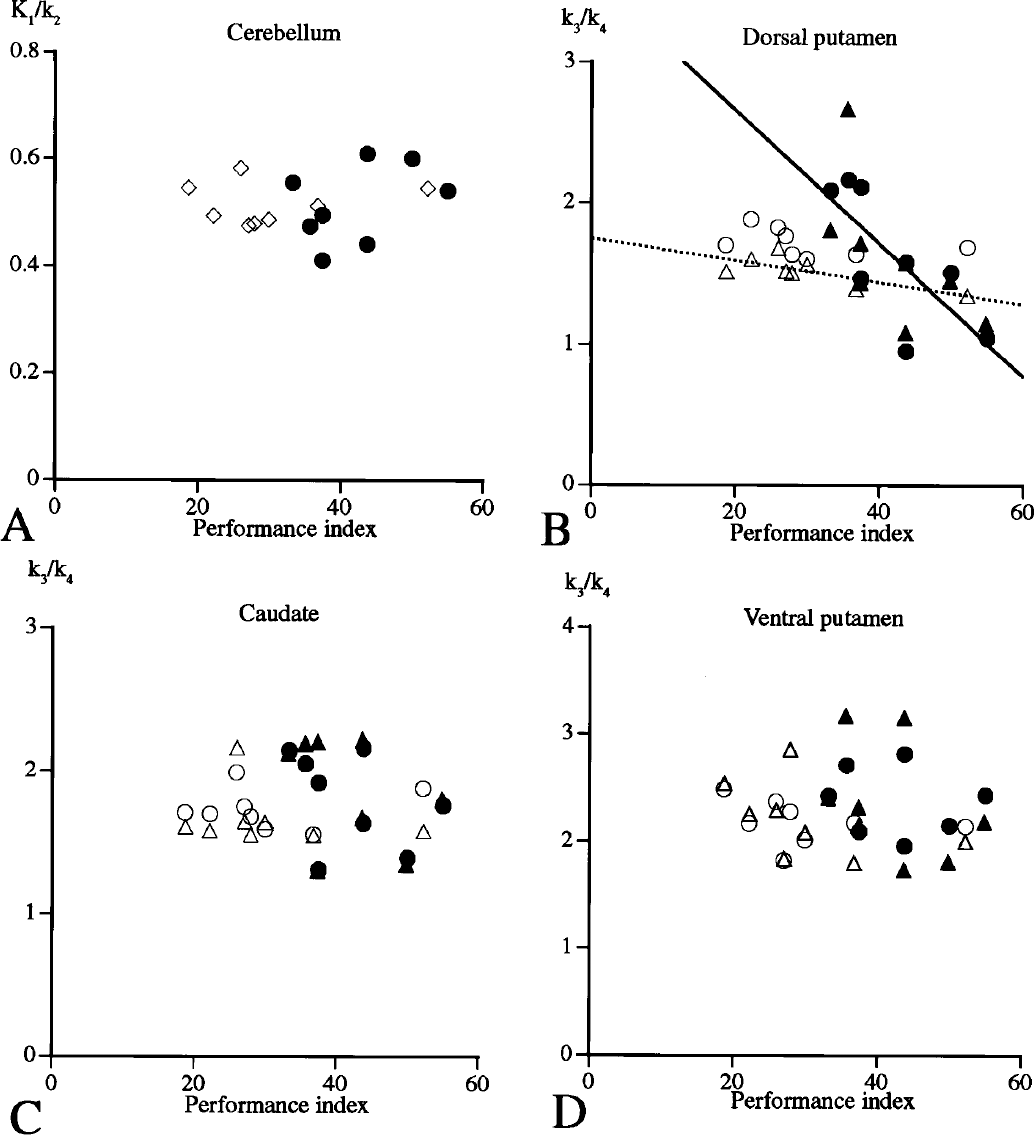
Spearman rank correlation between [11C]raclopride binding potential and performance index in the cerebellum, ventromedial caudate, superodorsal striatum, and inferolateral striatum (○: normal/ipsilateral, Δ: normal/contralateral, ●: PD [Parkinson disease]/ipsilateral, ▲: PD/contralateral). In
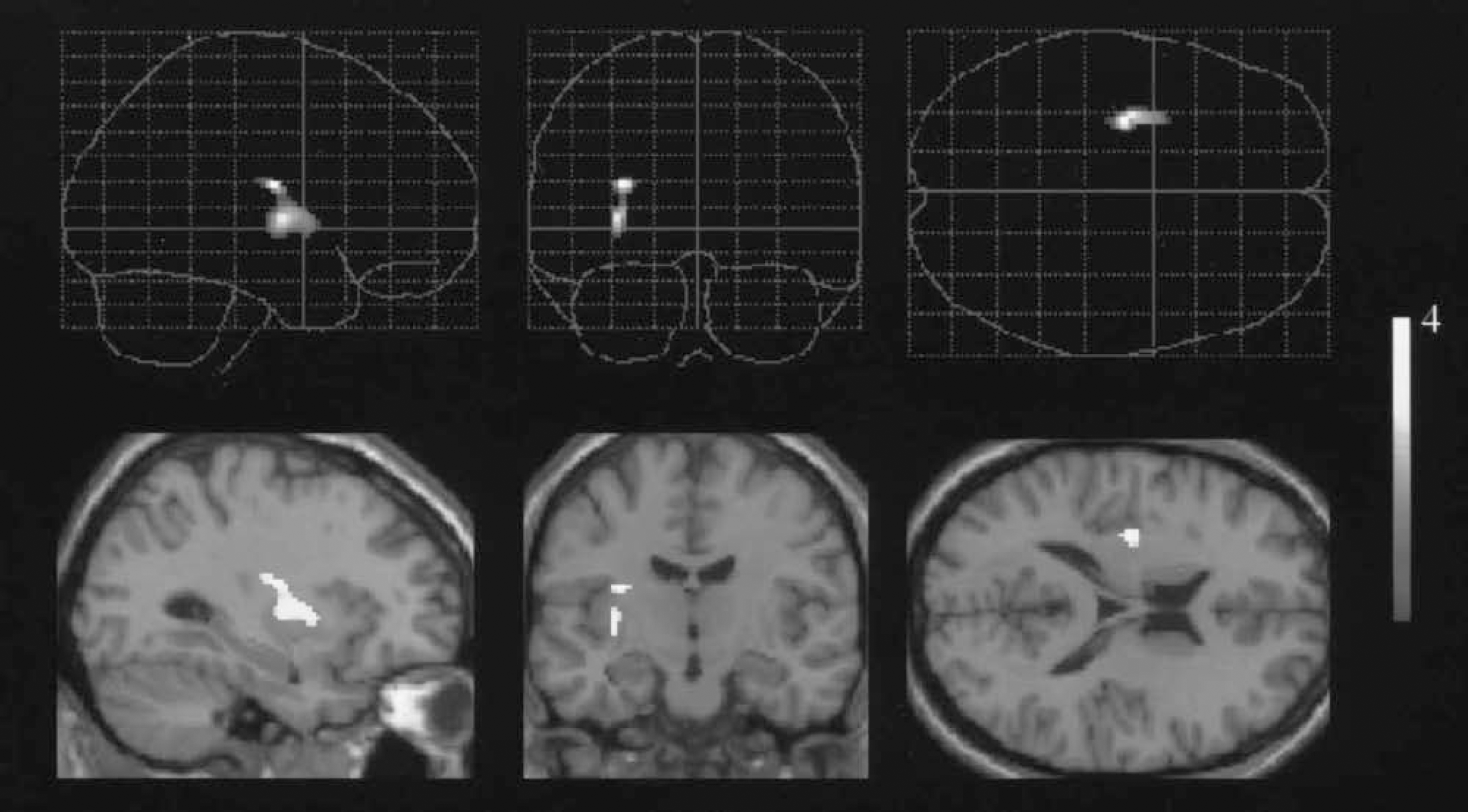
Brain regions showing significant reduction in [11C]raclopride availability correlated with magnitude of performance decline during the foot movement in normal subjects. (Superodorsal putamen: [x, y, z, = −27, −15, 16, respectively], Z = 3.61; ventrolateral putamen: [x, y, z, = −29, −12, 2, respectively], Z = 3.55.) Each top row: three-dimensional glass brain views; each bottom row: detected regions superimposed on MRI scan.
DISCUSSION
The present PET study performed during simple motor task is the first to investigate the
One caveat for interpretation of this motor-effect theory was the difficulty of differentiating stress (mental activity) from motor exercise applied in the present study. Almost all the animal experiments showing dopamine release by exercise (Heyes et al., 1988; Speciale et al., 1986) concurred with the finding that more or less stress induces dopamine release (Wise, 1980). However, the motor-effect theory is supported by the lateralized dopamine activation by unilateral motor exercise in the present study and by previous findings on functional connectivity that the dorsal striatum preferentially receives inputs from motor, sensory, and premotor cortices, the ventral striatum receiving afferent inputs from limbic cortices (Brooks, 1995; Graybiel et al., 1994). This leads to the possible assumption that dopamine release in the entire striatum hints at emotional activities or mental operations, as shown in other studies (de la Fuente-Fernandez et al., 2001; Koepp et al., 1998). Furthermore, the dorsal striatum encircles the somatotopic organization of foot representation revealed by a functional MRI (Maillard et al., 2000). Because the distribution volume for [11C]RAC is reportedly not dependent on regional CBF (Logan et al., 1994), which is a central biological parameter for the fMRI and [15O]H2O PET activation studies, the present quantified changes in [11C]RAC binding are probably not related to exercise-induced changes in regional CBF. Taken together, motor effect on dopamine release is likely present in the present study.
In PD, the level of [11C]RAC binding was higher in the affected striatum (and even nonaffected striatum) at rest, which is consistent with results from other reports (Rinne et al., 1990; Sawle et al., 1993), presumably caused by deafferentation-induced upregulation of D2 receptors. Therefore, lack of contralateral dopamine release in the PD group in the present study may be explained not by simply mechanistic effects by the following reason: that the more severely affected putamen is incompetent to generate sufficient dopamine release. In contrast, as shown in Fig. 3, exercise-induced dopamine release was greater in the ipsilateral dorsal striatum in comparison with the baseline condition. A previous PET-CBF activation study showed that bilateral premotor cortices were activated by a unilateral hand movement after unilateral pallidotomy (Ceballos-Baumann et al., 1994), indicating compensatory functional recovery in the basal ganglia-motor cortical circuit. Histologically, afferent fibers from the dorsal part of the putamen project somatotopically to the lateral parts of the substantia nigra, which relates to the motor circuit system, and fibers from the caudate project to the rostral third of the nigra, which relates to limbic and association cortical system (Szabo, 1980; Hedreen and DeLong, 1991; Parent and Hazrati, 1994). This suggests that activation in the motor cortex or the secondary motor cortical regions would affect downwards the substantia nigra, which would in turn activate the bilateral dopaminergic systems (the more intact side could afford to release more dopamine). In the present study, a close observation of patient's behavior showed that unilateral movement of the affected limb in five of eight PD patients was accompanied irregularly by minimal movement of a clinically intact limb. The limb's movement was, however, so small in magnitude and so brief that the movement-induced activation was likely negligible. These findings suggest that the ipsilateral dorsal striatum dopamine activation in PD may not be related to the motor-induced mechanical effect, but to the compensatory effect of the dopaminergic system in maintaining the movement of an affected limb.
The present correlation of reduction in [11C]RAC k3/k4 with performance deterioration in the dorsal striatum contralaterally in the healthy group and ipsilaterally in the PD group was intriguing. If the theory of stress on increase in striatal dopamine release during exercise was primary, [11C]RAC k3/k4 in the more ventral and medial striatum (meso-limbo-cortical projection system) might have been a response. Actually, there was such a tendency in the PD ventromedial striatum (Fig. 4C). This quantitative change was also confirmed by correlation analysis of SPM (Fig. 5). The present correlation result is in line with the idea that dorsal striatal dopamine release is related to sensorimotor coordination and response selection (Robbins and Everitt, 1992). A failure in this correlation in the PD group in SPM might be due to the magnitude of change not being high enough to be beyond the threshold of
In conclusion, motor exercise
Footnotes
Acknowledgements
The authors thank Fumitoshi Nakamura, Toshihiko Kanno, and Shuji Nobezawa (Hamamatsu Medical Center) for their technical support.
