Abstract
In this study, near-infrared spectroscopy was applied to examine whether cytochrome oxidase in the rat brain is inhibited by nitric oxide in vivo. During normoxia, intravenous NG-nitro-l-arginine methyl ester (l-NAME) administration significantly decreased the cerebral saturation of hemoglobin with oxygen but did not alter the cytochrome oxidase redox state. Anoxia significantly reduced the cytochrome oxidase. The time course of the recovery of the redox state during reoxygenation was not altered by l-NAME. The results suggest that in adult rats, cytochrome oxidase is not inhibited by nitric oxide, either in physiologic conditions or during reoxygenation after a brief anoxic period.
Nitric oxide (NO) is a competitive inhibitor of cytochrome oxidase, the terminal enzyme of the electron transport chain. In cultured synaptosomes, oxygen consumption can be inhibited by NO at physiologic concentrations, particularly at low oxygen tensions (Brown and Cooper, 1994). For this reason, it has been proposed that NO could be a physiologic modulator of the oxygen affinity of cytochrome oxidase in vivo (Clementi et al., 1999). This has been supported by the apparent decrease in the oxygen affinity of cytochrome oxidase in vivo compared with mitochondrial suspensions (Hempel et al., 1977; Kreisman et al., 1981) and the increase in whole body oxygen consumption during systemic inhibition of NO synthase (NOS) (Shen et al., 1994).
Measurements of CBF and CMRO2 during NO synthase inhibition in the rat (Horvath et al., 1994) and piglet (Greenberg et al., 1994) have shown decreases in CBF but no change in oxygen consumption at normocapnia. A more sensitive technique would be to measure the redox centers of cytochrome oxidase or the redox state of cytochrome c, which have been shown to become more reduced during inhibition in mitochondrial suspensions (Cassina and Radi, 1996) and isolated cardiomyocytes (Stumpe et al., 2001).
Using full spectral near-infrared spectroscopy, we observed in preliminary experiments that in the brain of adult rats, the copper A center (CuA) of the cytochrome oxidase is reduced during the reactive hyperemia after a brief period of anoxia (unpublished results). Previously it was shown that during reperfusion after ischemic hypoxia, NO tension increases to levels shown to inhibit oxygen consumption in vitro and that this release can be inhibited by NG-nitro-
The aim of this study is to determine in vivo whether NO inhibits cytochrome oxidase in the brain under normal physiologic conditions and during the reactive hyperemia after a brief period of anoxia.
MATERIALS AND METHODS
Animal housing and treatment conditions complied with European Union directive 86/609 on animal welfare.
Twelve male Sprague-Dawley rats (360 to 460 g) were anesthetized with isoflurane (1.5%), intubated, and ventilated with a 30:70 oxygen-to-nitrogen gas mixture. The left femoral artery was cannulated for arterial blood sampling and monitoring of the MABP and heart rate, and the left femoral vein for injection of
The near-infrared spectroscopy system and algorithms have been described previously (Springett et al., 2000). Near-infrared spectra were collected contiguously with a period of 50 milliseconds and 20 spectra were averaged to give a time resolution of 1 second. Absolute changes in the concentration of oxyhemoglobin, deoxyhemoglobin, and oxidized CuA ([CuA]) were calculated from the change in attenuation between 780 and 900 nm (Cope et al., 1989; Matcher et al., 1995) and normalized to the baseline optical pathlength measured from the 840-nm water feature using second differential analysis (Matcher and Cooper, 1994). Assuming an 85% water content, the absolute concentration of hemoglobin was obtained from second differential analysis and normalized to the pathlength obtained from the 740-nm water feature. The absolute deoxyhemoglobin and absolute changes in oxyhemoglobin and [CuA] were monitored online. Absolute oxyhemoglobin was back-calculated taking the value after terminal anoxia as zero. Total hemoglobin ([HbT]) was calculated as the sum of oxyhemoglobin and deoxyhemoglobin, and mean cerebral saturation of hemoglobin with oxygen (Smc
Differences between groups were evaluated using the Wilcoxon-Mann-Whitney midrank sum test. Differences within groups were evaluated using the Wilcoxon matched-pairs signed-ranks test.
RESULTS
Table 1 shows the physiologic data obtained before the pretreatment and posttreatment anoxic periods. No significant differences were found between groups at the onset of the experiment. Pretreatment near-infrared spectroscopy parameters were also comparable between groups (Table 2) except for [HbT], which was on average 10 μmol/L lower in the
Physiologic variables before the first (pretreatment) and second (posttreatment) anoxia
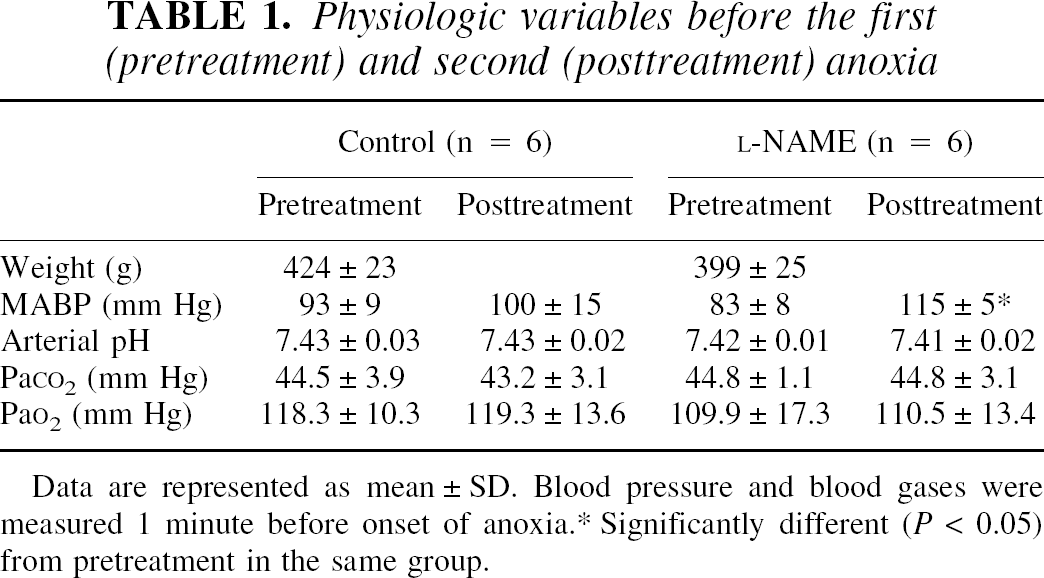
Data are represented as mean ± SD. Blood pressure and blood gases were measured 1 minute before onset of anoxia.
Significantly different (P < 0.05) from pretreatment in the same group.
During anoxia and reoxygenation, the changes in hemoglobin parameters and [CuA] showed a similar pattern in all animals. Figure 1 shows the mean time course of these changes for the control and
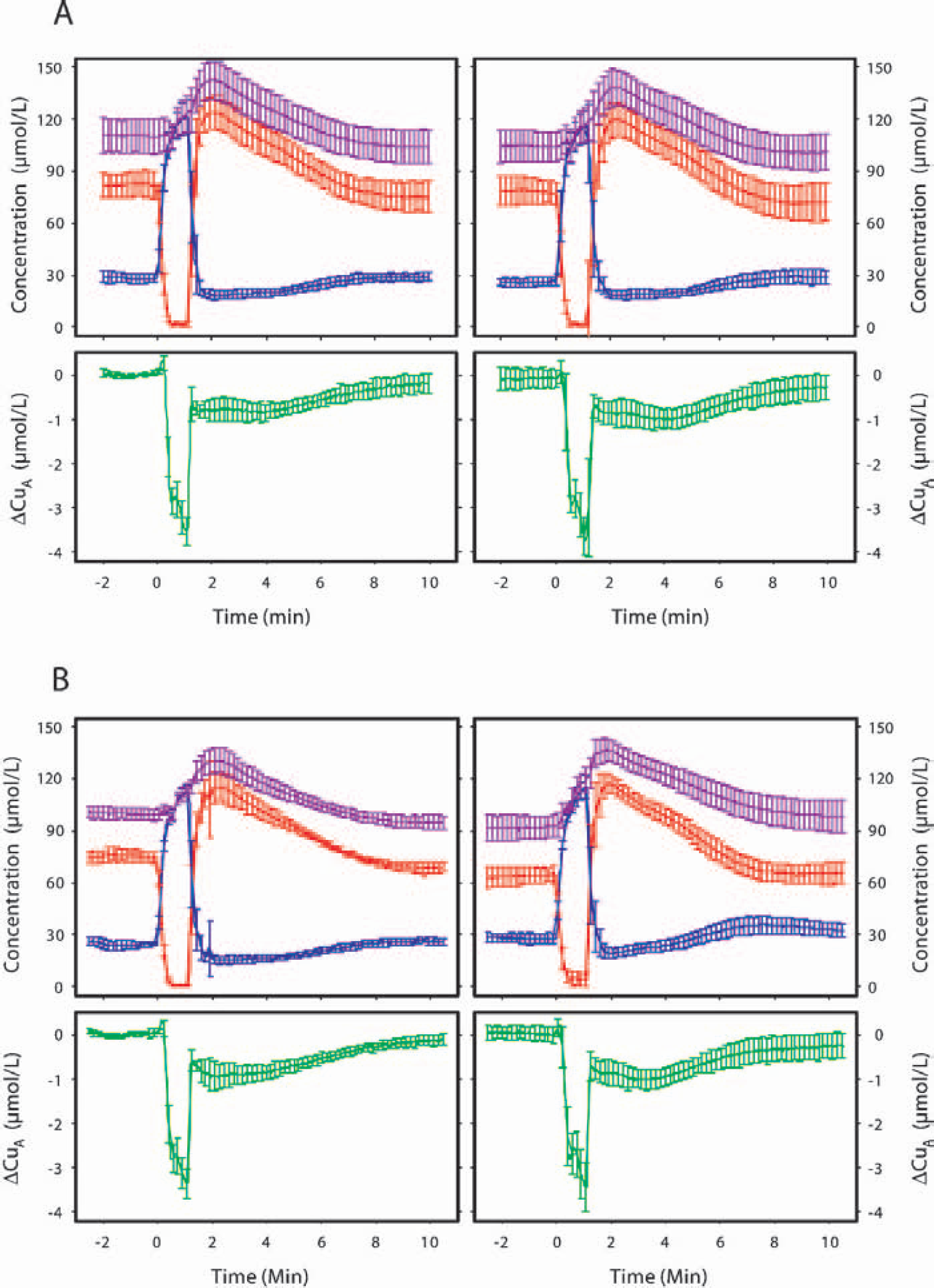
Mean time course (mean ± SD) of near-infrared spectroscopy variables during and after a 70-second period of anoxia for the control
In the control group, pretreatment and posttreatment values of arterial blood pressure, blood gases, [CuA] and brain hemoglobin parameters were comparable. Administration of
Near-infrared spectroscopy parameters 1 minute before and 3 and 10 minutes after onset of a 70-second period of anoxia
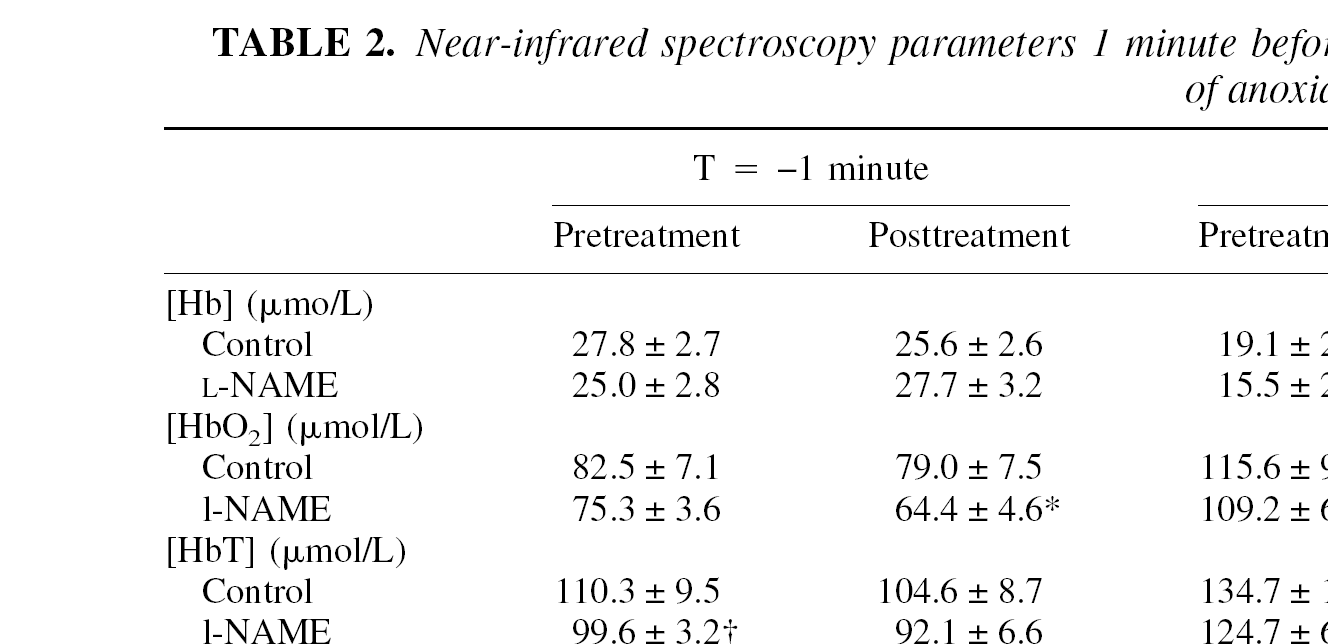
Data are represented as means ± SD. [Hb], deoxyhemoglobin concentration; [HbO2], oxyhemoglobin concentration; [HbT], total hemoglobin concentration; Smc
Significantly different (P < 0.05) from pretreatment in the same group.
Significantly different from pretreatment control group.
Table 3 shows the changes in [CuA] during and after anoxia. No significant differences within or between groups were observed. The decline in [CuA] during anoxia, plotted as a function of the Smc
Changes in concentration of oxidized Cu A (μmol/L) before, during, and after a 70-second period of anoxia
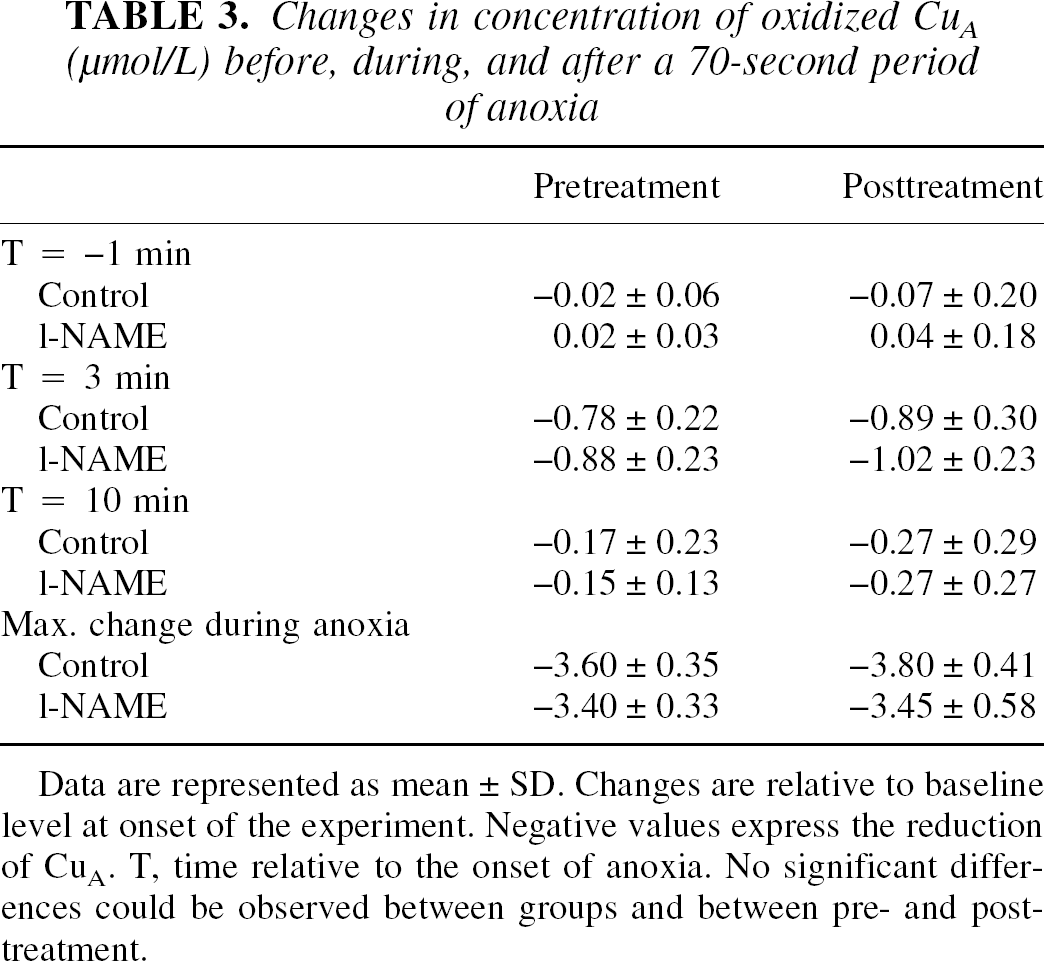
Data are represented as mean ± SD. Changes are relative to baseline level at onset of the experiment. Negative values express the reduction of CuA. T, time relative to the onset of anoxia. No significant differences could be observed between groups and between pre- and posttreatment.
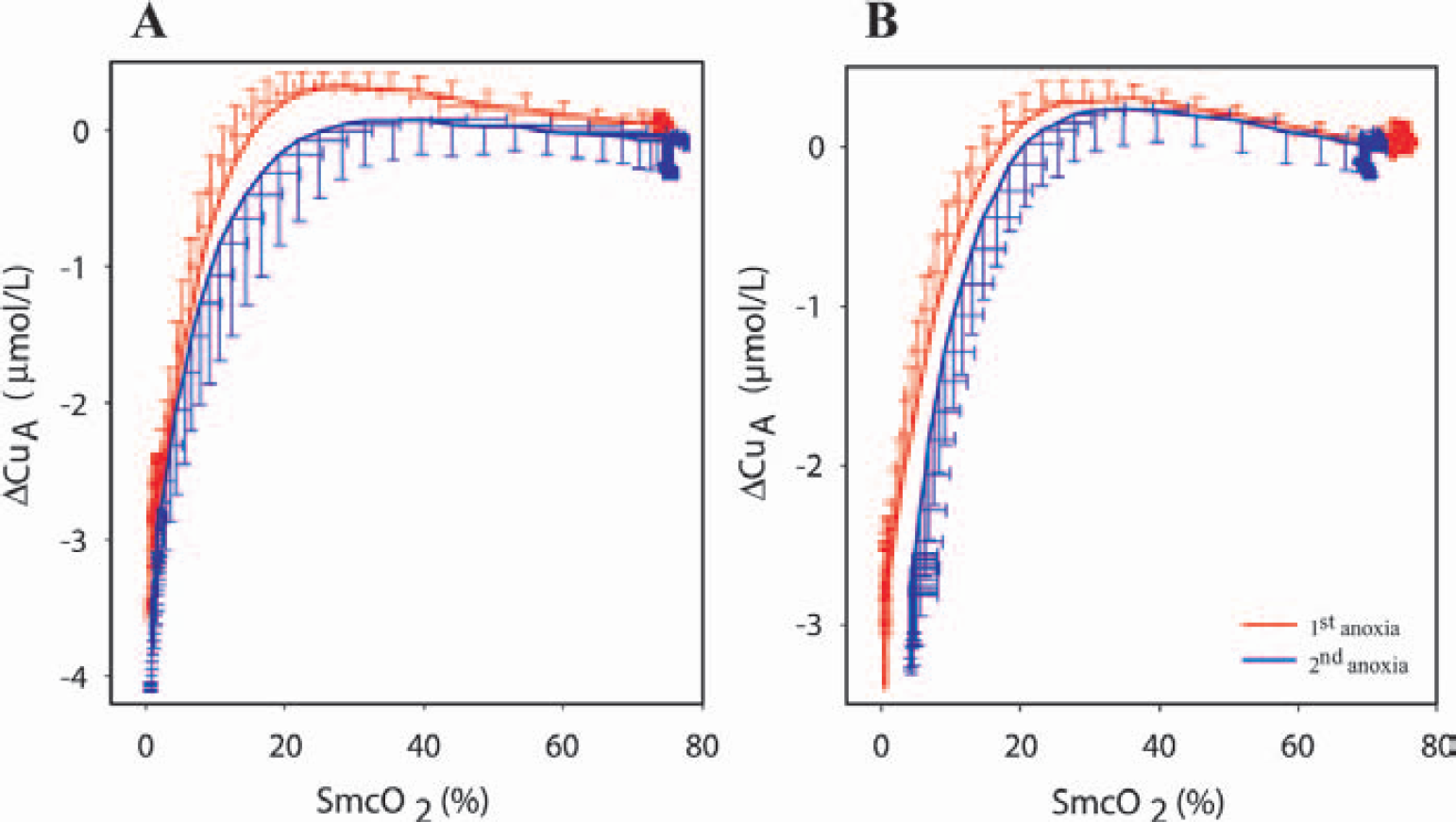
Changes in cytochrome oxidase redox state (Δ[CuA]) observed during anoxia and expressed as a function of cerebral hemoglobin saturation (Smc
DISCUSSION
In preliminary experiments, we observed a partial reduction of CuA during the hyperemic phase succeeding a brief period of anoxia (unpublished results). We postulated that during reoxygenation, NO is synthesized by constitutive NOS and contributes to the vasodilation and partial inhibition of cytochrome oxidase. Previously, it was shown that NO is synthesized during the reperfusion after middle cerebral artery occlusion (Malinski et al., 1993; Sato et al., 1994) and after 60 minutes of hypoxia (Fujisawa et al., 1999). In addition, in vitro experiments have shown that in isolated nerve terminals and mitochondria, NO clearly inhibits the respiration by competing with oxygen at the oxygen-binding site of cytochrome oxidase (Brown and Cooper, 1994; Brudvig et al., 1980; Cleeter et al., 1994). The present study shows that a reduction in oxidized CuA occurs reproducibly during and after a 70-second period of anoxia and that brain hemoglobin oxygenation and CuA redox state completely recover within 15 minutes after reoxygenation. Pretreatment with
From our data, no conclusions can be drawn regarding the question of whether NO derived from inducible NOS inhibits respiration in vivo. The model we apply should be inducible NOS free because upregulation of this enzyme can only be found several hours after an insult (Chatzipanteli et al., 1999; Ikeno et al., 2000). Therefore, although NO may inhibit cytochrome oxidase in vivo after prolonged insults such as hypoxia–ischemia, the data presented in this article would suggest that NO derived from constitutive NOS activation does not inhibit cytochrome oxidase either under normal physiologic conditions or during the reactive hyperemia after brief anoxia. Other factors that may affect the redox state of the cytochrome oxidase during reoxygenation include mitochondrial Ca2+ overload, O2 radicals, ionic shifts, acidosis, or simply an increased oxygen demand that is not matched by the oxygen delivery. Increased oxidative metabolism is likely to occur after a period of anoxia, because ionic shifts over the cell membrane have to be restored (for review, see Siesjo, 1981).
We conclude that, in adult rats, inhibition of NOS by
