Abstract
Activation dynamics of hippocampal subregions during spatial learning and their interplay with neocortical regions is an important dimension in the understanding of hippocampal function. Using the (14C)-2-deoxyglucose autoradiographic method, we have characterized the metabolic changes occurring in hippocampal subregions in mice while learning an eight-arm radial maze task. Autoradiogram densitometry revealed a heterogeneous and evolving pattern of enhanced metabolic activity throughout the hippocampus during the training period and on recall. In the early stages of training, activity was enhanced in the CA1 area from the intermediate portion to the posterior end as well as in the CA3 area within the intermediate portion of the hippocampus. At later stages, CA1 and CA3 activations spread over the entire longitudinal axis, while dentate gyrus (DG) activation occurred from the anterior to the intermediate zone. Activation of the retrosplenial cortex but not the amygdala was also observed during the learning process. On recall, only DG activation was observed in the same anterior part of the hippocampus. These results suggest the existence of a functional segmentation of the hippocampus, each subregion being dynamically but also differentially recruited along the acquisition, consolidation, and retrieval process in parallel with some neocortical sites.
Introduction
Although its importance in several forms of memory is well recognized, the precise dynamics of the hippocampal circuits in coding, consolidation, as well as retrieval of spatial memory in rodents is still a matter of debate (Dash et al, 2004; Eichenbaum, 2000; Nadel and Bohbot, 2001; O'Keefe and Nadel, 1978; Riedel et al, 1999, Squire, 1992). In this context, the notion that separate components of this structure could be engaged and differentially recruited at different stages of the learning process and on recall has been suggested (Nadel and Moscovitch, 1998) but not extensively explored. Previous evidence, notably provided by the description of place cells (Jung et al, 1994; Poucet and Buhot, 1994), suggested that the hippocampus does not participate in learning and memory tasks as a unitary structure but appears to display regional specialization. Based on the morphologic organization of afferent and efferent connections from the septum to the temporal lobe as well as on the results of lesion studies (Hughes, 1965; Stevens and Cowey, 1973), Moser and Moser (1998) have proposed that the anterior third of the hippocampus is functionally distinct from the posterior two-thirds. More recently, PET studies in humans have revealed the heterogeneous character of hippocampal activation (Lepage et al, 1998) and suggested that the anterior hippocampus is activated by encoding while its posterior part is engaged in retrieval (Greicius et al, 2003). Similarly, several studies have attributed distinct roles to hippocampal subregions, with the CA3 area being implicated in the acquisition of information in a new environment, while the CA1 and dentate gyrus (DG) areas being important in a familiar environment (Kesner et al, 2000; Lassalle et al, 2000; Lee and Kesner, 2002).
To date, few studies have provided a comprehensive picture presenting correlates of activation at the subregional level in the hippocampus during distinct phases of a spatial task. In this study, we have assessed the changes of metabolic activity in several segments along the entire anteroposterior axis to evaluate the extent of activation at different stages of learning and on recall of a spatial task in mice. Moreover, a parallel assessment of metabolic activity in the retrosplenial cortex and the amygdala was performed and evaluated in relation with changes in the hippocampus. Results obtained reveal a complex and evolving pattern of activation among hippocampal subregions throughout the learning process.
Materials and methods
Animals
Adult male mice of the C57BL/6J strain (IFFA CREDO, Lyon, France) were used throughout the study. They were group housed on arrival under a 12 h light/dark cycle (lights on at 0700). To minimize stress, mice were handled daily for 10 mins during the 2 weeks before testing. All the procedures described hereafter were conducted in conformity with the Swiss national institutional guidelines on animal experimentation, and approved by the local Committee for animal care.
Apparatus
The eight-arm radial maze (RAM) consisted of a circular central platform (16.5 cm in diameter) from which radiated eight arms made of transparent Plexiglas (6 × 6 × 50 cm). Arm access could be blocked with automated doors to prevent the reentry in already visited arms (working memory errors). Two wood barriers (1.5 × 1 × 6 cm) were placed in each arm at 0 and 20 cm from the entry to prevent the animal to see the bait from a distance and to increase the motivation for a visit. Arms were baited with ~ 10 µL of condensed milk diluted 1:1 with water.
Spatial Learning
We replicated a training schedule already described by Bontempi et al (1999). Starting 2 days before training, animals were food-deprived overnight. This procedure resulted in a stable weight loss not greater than 10% of their initial body weight. During the first 2 days, mice were familiarized with the experimental setup by a 10-min exposure to the maze with all arms baited. Thereafter, they were divided into five groups, according to the duration of training and, for the last group, testing for memory retrieval (respectively, 1 day, 3 days, 6 days, 9 days training and 9 days training plus a retention test 5 days after the last training day). Groups consisted of six animals, with the exception of the 6-day training group made of eight mice. Each group was assigned a different set of baited arms with sequence angles at 135°–90°–135° to test their reference memory. Each trial started by placing the animal in the central platform covered by a transparent Plexiglas lid and opening of all eight doors. One daily training session consisted of six consecutive trials separated by a 1-min rest in isolation. The same procedure was used for testing memory retention. After visiting an arm, the corresponding door was closed. Each trial was terminated when all three baited arms had been visited. The maximal time allowed per trial for each mouse was set at 5 mins, but they always found the three baited arms before this limit. Altogether, a training session lasted between 10 and ~ 25 mins, depending on the animal's spatial proficiency. Spatial learning performance was assessed by calculating the ratio between entries in correct arms versus total entries. To ensure that the metabolic effects observed were not caused by factors unrelated to the spatial learning process (e.g., motor activity), we performed a control experiment in which metabolic activity in the hippocampus was compared between animals that never saw the RAM (Quiet control, QC) and animals placed in the RAM with only the three baited arms opened (Untrained control) to prevent the development of a spatial strategy to accomplish the task. Animals underwent five sessions of three trials each spread over a period of 2 h and were killed at the end of the fifth session.
(14C)-2-deoxyglucose procedure: On the last training session, animals received 5 mins before testing an injection of (14C)-2-deoxyglucose intraperitoneally ((14C)2DG, 0.165 mCi/kg; Sokoloff et al, 1977). After testing, they remained in their cage until being killed. Control animals (QC) were brought to the testing room, but remained in their home cage after having received the (14C)2DG. Forty-five minutes after (14C)2DG injection, mice were killed under anesthesia, and their brain rapidly removed, frozen, and stored at −80°C until cutting. Ten series of four consecutives coronal sections (20 µm) were collected every 160 to 200 µm on coated slides, rapidly dried on a hot plate at 60°C, and then processed for autoradiography along with a calibrated (14C)-microscale on Kodak X-ray hyperfilm. After 2 weeks of exposure, films were developed and brain sections were stained with cresyl violet. Images of stained sections and their corresponding autoradiograms were digitized via a CCD camera coupled to an image analysis system (NIH image, USA). The outlines of the hippocampus and its subregions, the DG, the CA3 area, and the CA1 area, as well as the corpus callosum, the retrosplenial cortex, and the amygdaloid complex were drawn on the image of the sections and optical densities determined on the corresponding autoradiogram.
Data Analysis
Comparison of learning performance was performed using paired and unpaired t-test. Significance level was set at P < 0.05. Optical densities of the hippocampus (considered both as a whole and into its sub regions CA1, CA3, and DG) were assessed at 10 regularly spaced anteroposterior levels (160 to 200 µm). Optical densities were converted to (14C) tissue activity in nCi/g tissue (2DG uptake) based on the microscale standards and a cubic spline transformation. The anterior, intermediate, and posterior part of the hippocampus were determined according to the Atlas of Hof et al (2000) corresponding, respectively to, (–1.3; −2.1 mm), (–2.4; −2.7 mm), and (–3; −3.3 mm) from Bregma. For the global analysis of the hippocampus and its subregions, the mean 2DG uptake value of the region of interest was normalized using the corpus callosum as a reference. For the analysis at each level, 2DG uptake values were averaged from four consecutive sections and normalized to the mean 2DG uptake value of the corresponding corpus callosum. The mean relative 2DG uptake of each tested group was compared with one of the QC group using multifactor analysis of variance (ANOVA) followed by a Fisher's protected-least-significant-difference (PLSD) post hoc test with P < 0.05. Comparison of 2DG uptake values for the retrosplenial cortex and the amygdala were made between trained and QC groups using the Mann–Whitney test with P < 0.05.
Results
Animals trained in the eight-arm RAM displayed, after an initial delay of 1 to 3 days, a steady improvement of their performance stabilizing between 8 and 9 days of training, a level that they maintained on recall 5 days later (Figure 1). Moreover, examination of the curves showing the progression of spatial discrimination performance with days of training suggests that it comprises distinct phases. At the first training session on day 1, mice displayed around 50% of correct choices. Up to day 4 to day 5, their performance improved somewhat slowly, not being significantly different from performance at day 1. Then, between day 5 and 9, a steep improvement of their performance was observed, reaching up to 80% of correct choices at day 9 (paired t-test, t(5) = −6.446, P = 0.0013 versus day 1). In the recall group, when mice were tested 5 days after their last training session, their performance level was similar to the one displayed at day 9 (paired t-test, t(7) = −0.630, P = 0.5487).
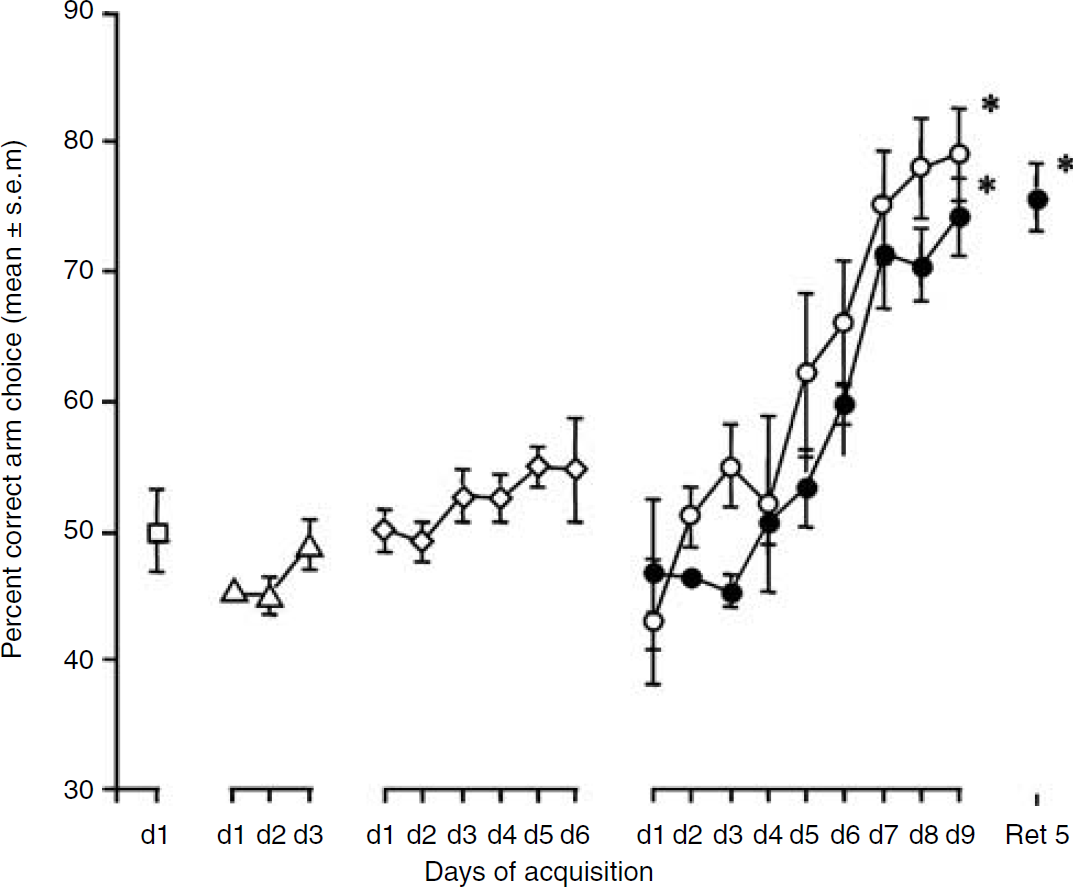
Spatial discrimination performance in groups undergoing either 1 day (open square), 3 days (open triangle), 6 days (open rhomboid), 9 days (open circle) or 9 days + 5 days retention (filled circle) training. *P < 0.05 as compared with the respective d***l.
A parallel analysis of metabolic activation using (14C)2DG autoradiography on coronal sections from animals at different stages of training revealed significant changes over the training period. Quite strikingly, important metabolic differences in each hippocampal subregion at different levels along the anteroposterior axis were observed with days of training as revealed by autoradiography (Figure 2). On a first qualitative evaluation, a clear increase of metabolic activity in both the anterior and the posterior part of the hippocampus was apparent at day 1 and day 9, whereas glucose utilization was found elevated only in the anterior part on recall after 5 days (Ret5) (Figures 2A and 2B). In contrast, no significant changes were detected in the somatosensory cortex, an area chosen as external reference since it is not considered to be involved in the learning process of the eight-arm RAM.
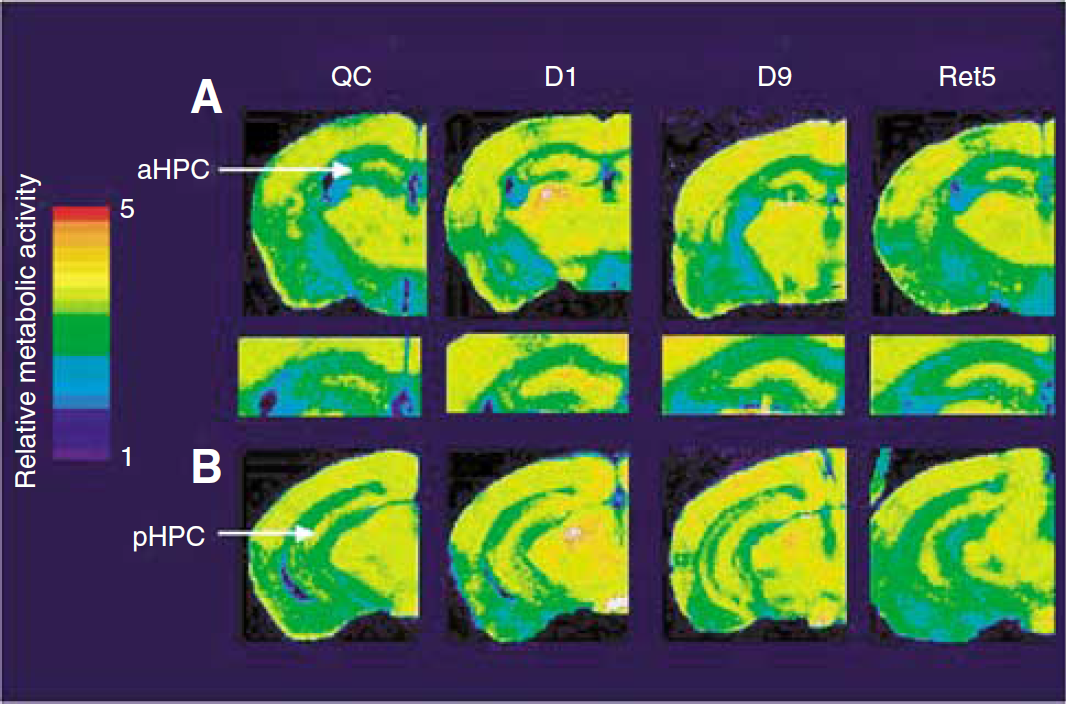
(14C)-2-deoxyglucose color-coded autoradiographs of coronal brain sections from mice either never trained (QC), trained 1 day (Dl), 9 days (D9) and trained 9 days and retested 5 days later (Ret5) in an eight-arm radial maze. The upper panel shows metabolic activity in the anterior part of the hippocampus with a magnified view of the hippocampus (
Characterization of the Metabolic Activation Pattern in the Hippocampus and Its Subregions During the Training Process
A quantitative analysis in the hippocampus considered as a whole revealed that at every training stage as well as during recall, the level of metabolic activity in trained animals was significantly elevated compared with QCs (ANOVA; F(5,33) = 4.521; P = 0.0030) (Figure 3A). In contrast, animals exposed to the RAM but not undergoing the training procedure (untrained group) did not display an increase in hippocampal metabolic activity compared with the animals of a QC group (data not shown). When the hippocampus was subdivided into its three major subregions, the CA1, CA3, and DG areas, glucose utilization in each of these areas was elevated from day 1 to day 9 in trained animals (Figures 3B to 3D). During recall however, glucose utilization was significantly enhanced only in the DG (P = 0.0146).
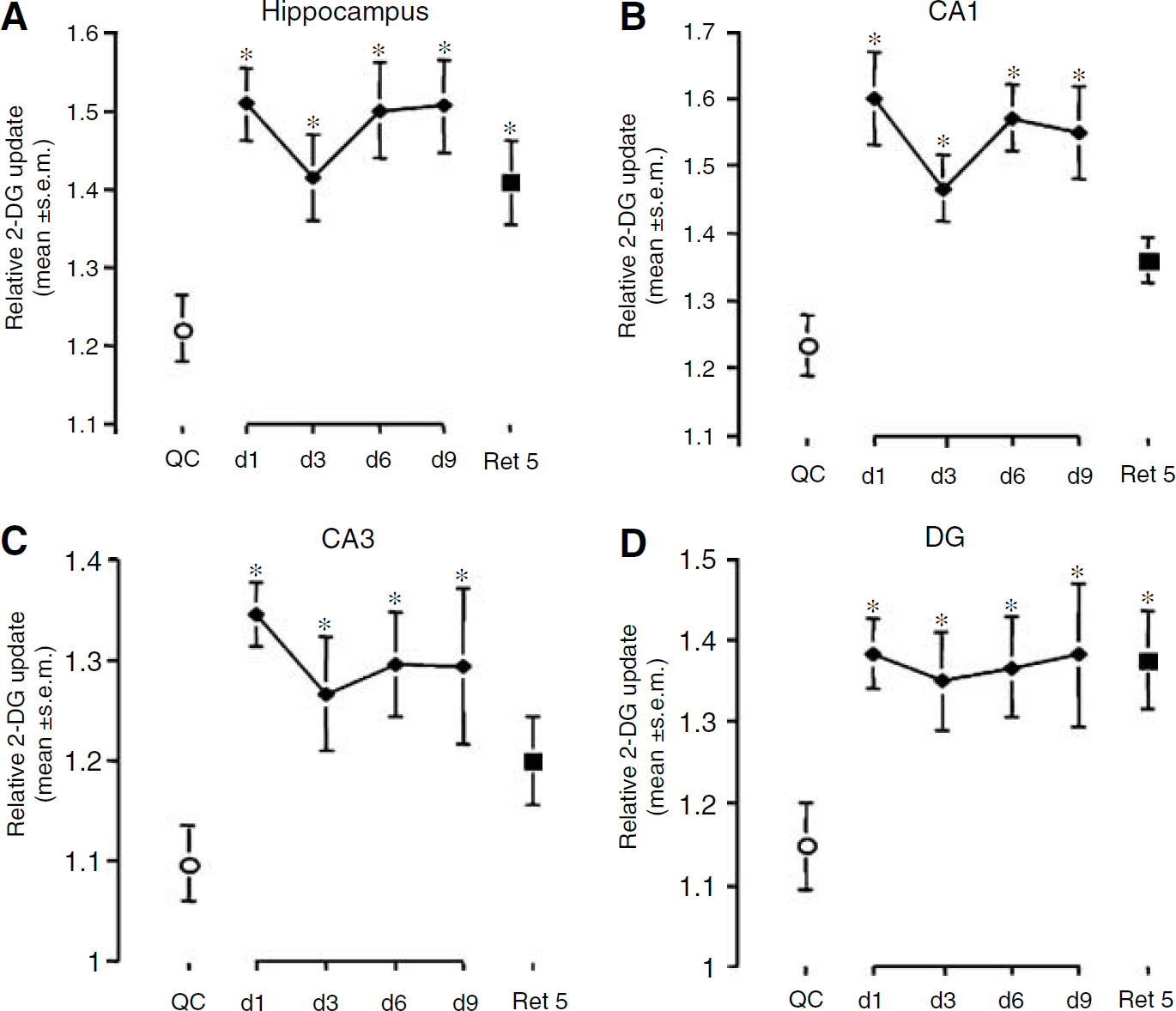
Changes in relative (14C)-2-deoxyglucose uptake in the whole hippocampus (
A more refined anatomic analysis based on segments taken along the entire anteroposterior axis indicated that at day 1 and day 3, a significant activation of the CA1 area occurred in a portion of the hippocampus extending from the intermediate region to its posterior part (F(2,33) = 10.8, P = 0.0032) (Figure 4). At day 6, the enhancement of 2DG uptake, while still involving the posterior part and the intermediate zone, extended further into the anterior area (F(1, 12) = 7.114, P = 0.0205). The CA3 area appeared to be significantly activated from day 1 to day 6 only within the intermediate portion of the hippocampus (F(3,19) = 4.173, P = 0.0198) (Figure 5). In the case of the DG, an activation of the anterior part occurred on day 1 and 3 (F(2,16) = 3.830, P = 0.0437), while at day 6, it was rather within the intermediate region that a significant enhanced glucose utilization was observed (F(1, 13) = 8.745, P = 0.0111) (Figure 6). At day 9, important modifications in metabolic activity took place in different areas. The significant activation of the CA1 area observed at day 6 was maintained at day 9. Activity in the CA3 area was increased both in the anterior and posterior zones and still involved the intermediate portion (F(1,11) = 5.845, P = 0.0341), while metabolic activity in the DG was increased from the anterior part to the intermediate section (F(1,11) = 12.482, P = 0.0047). When glucose utilization was analyzed in animals tested for memory retention (Ret5), a strikingly different pattern of activation was observed. Significant increases in metabolic activity were no longer observed in both CA1 and CA3 areas (Figures 4 and 5). In contrast, glucose utilization in the DG remained elevated within the same portion of the hippocampus as at day 9 of training (Figure 6), that is, encompassing the anterior and intermediate regions (F(1,11) = 7.002, P = 0.007).
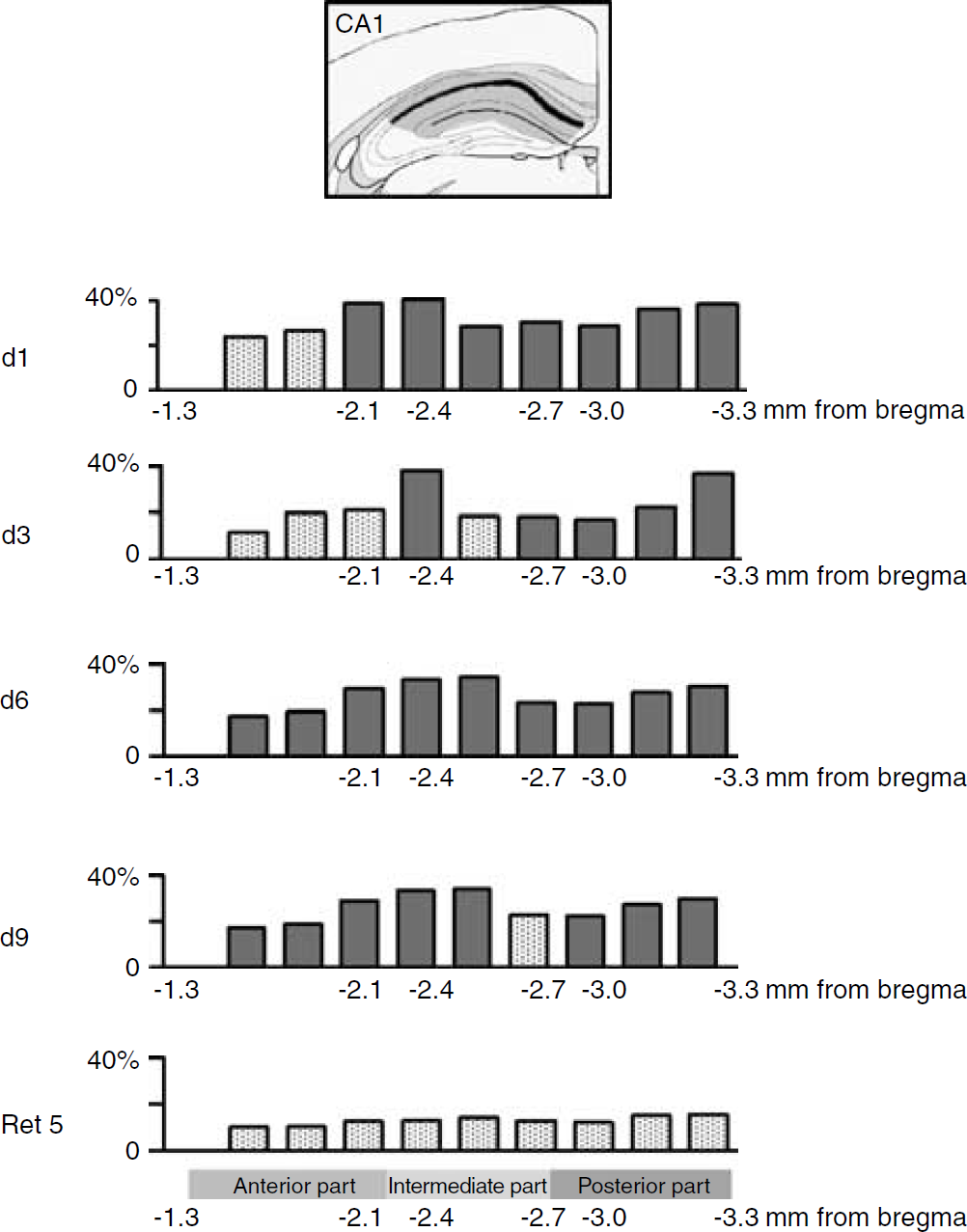
Percentage of relative changes in cerebral metabolic activity in the CA1 area of the hippocampus along the anteroposterior axis as a function of training time. Comparisons were made with relative metabolic activity of QC group. Significant changes are shown in filled column (Fisher ‘s protected-least-significant-difference (PLSD) post hoc test, P < 0.05).
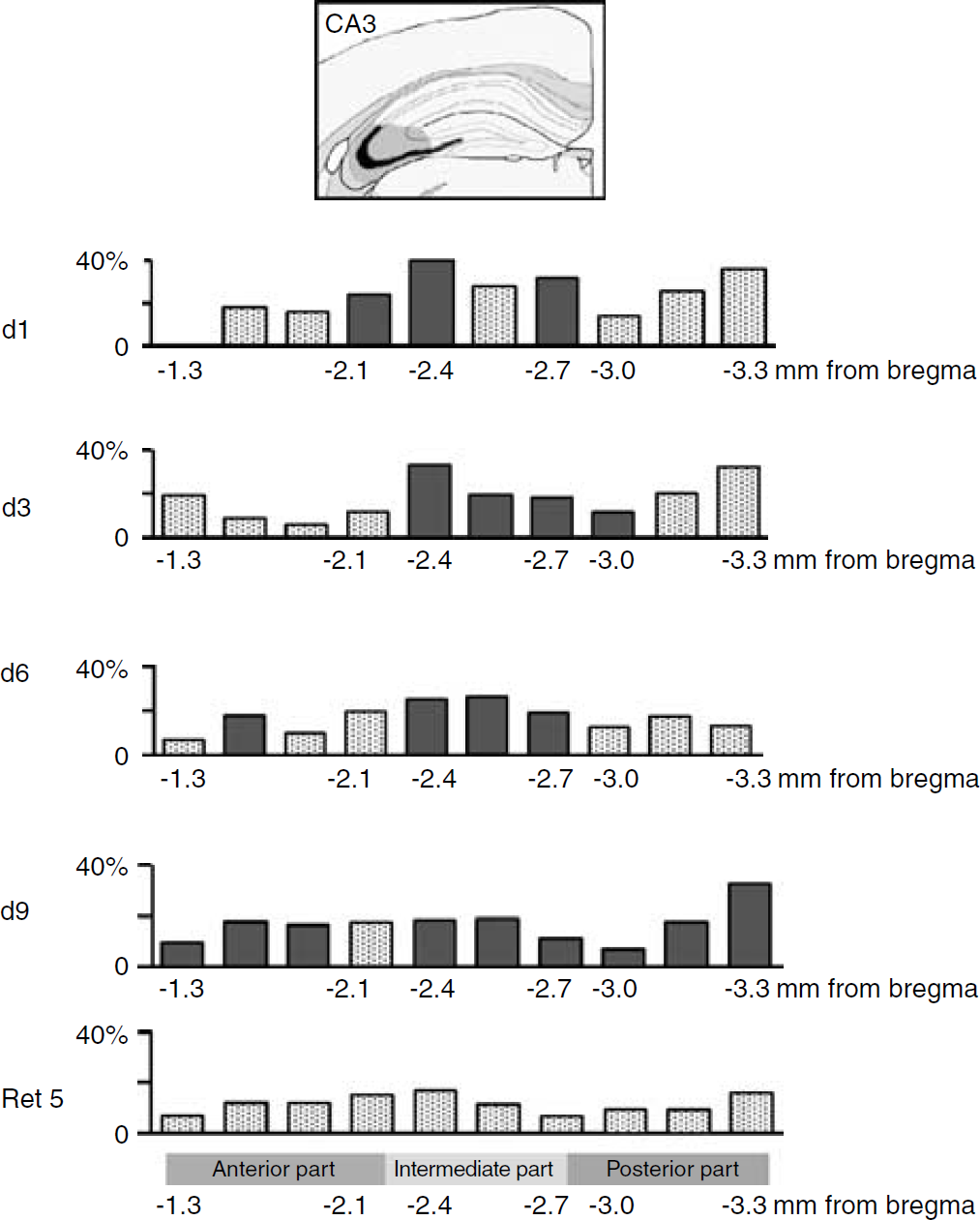
Percentage of relative changes in cerebral metabolic activity in the CA3 area of the hippocampus along the anteroposterior axis as a function of training time. Comparisons were made with relative metabolic activity of QC group. Significant changes are shown in filled column (Fisher ‘s protected-least-significant-difference (PLSD) post hoc test, P < 0.05).
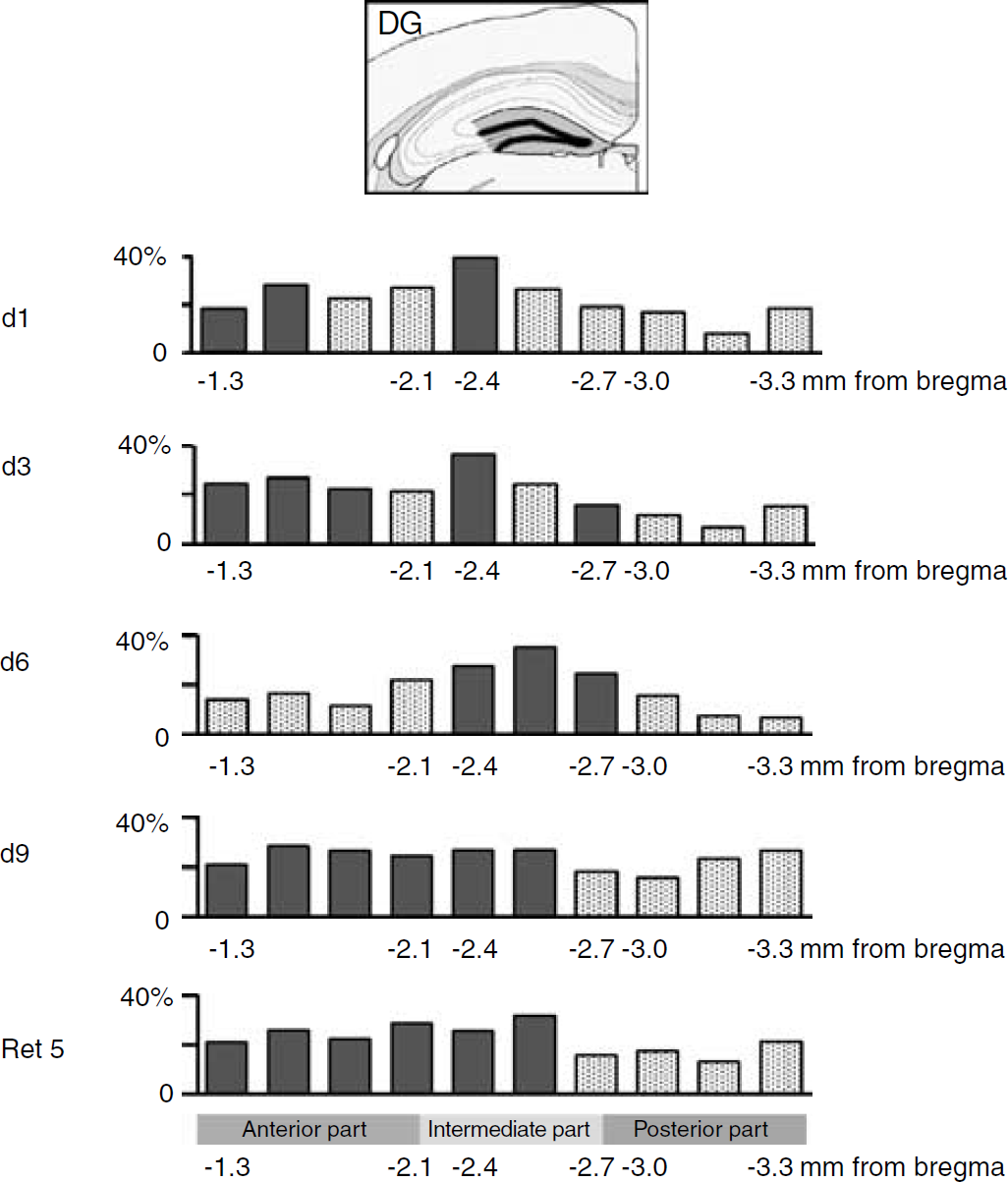
Percentage of relative changes in cerebral metabolic activity in the dentate gyrus (DG) of the hippocampus along the anteroposterior axis as a function of training time. Comparisons were made with relative metabolic activity of QC group. Significant changes are shown in filled column (Fisher ‘s protected-least-significant-difference (PLSD) post hoc test, P < 0.05).
Parallel Evaluation of Metabolic Activity in the Retrosplenial Cortex and the Amygdala
To test whether a time-dependent engagement of the neuronal circuitry involving regions other than the hippocampus (e.g., neocortex) was taking place during learning and/or on retrieval, metabolic activity was analyzed in the retrosplenial cortex and the amygdala. In the retrosplenial cortex, 2DG uptake was increased already on day 1 (P = 0.017, Figure 7A) and remained elevated up to the last day of training on day 9 (P = 0.0376, Figure 7 A). Although metabolic activity decreased when mice were retested 5 days later after their last training session, it never reached the level observed in QC mice. In contrast, analysis of glucose utilization in the amygdala indicated no significant difference throughout the entire training period including on retrieval (Figure 7B). This last observation suggests that for these mice, solving the RAM task under the present conditions does not involve an amygdaladependent component.
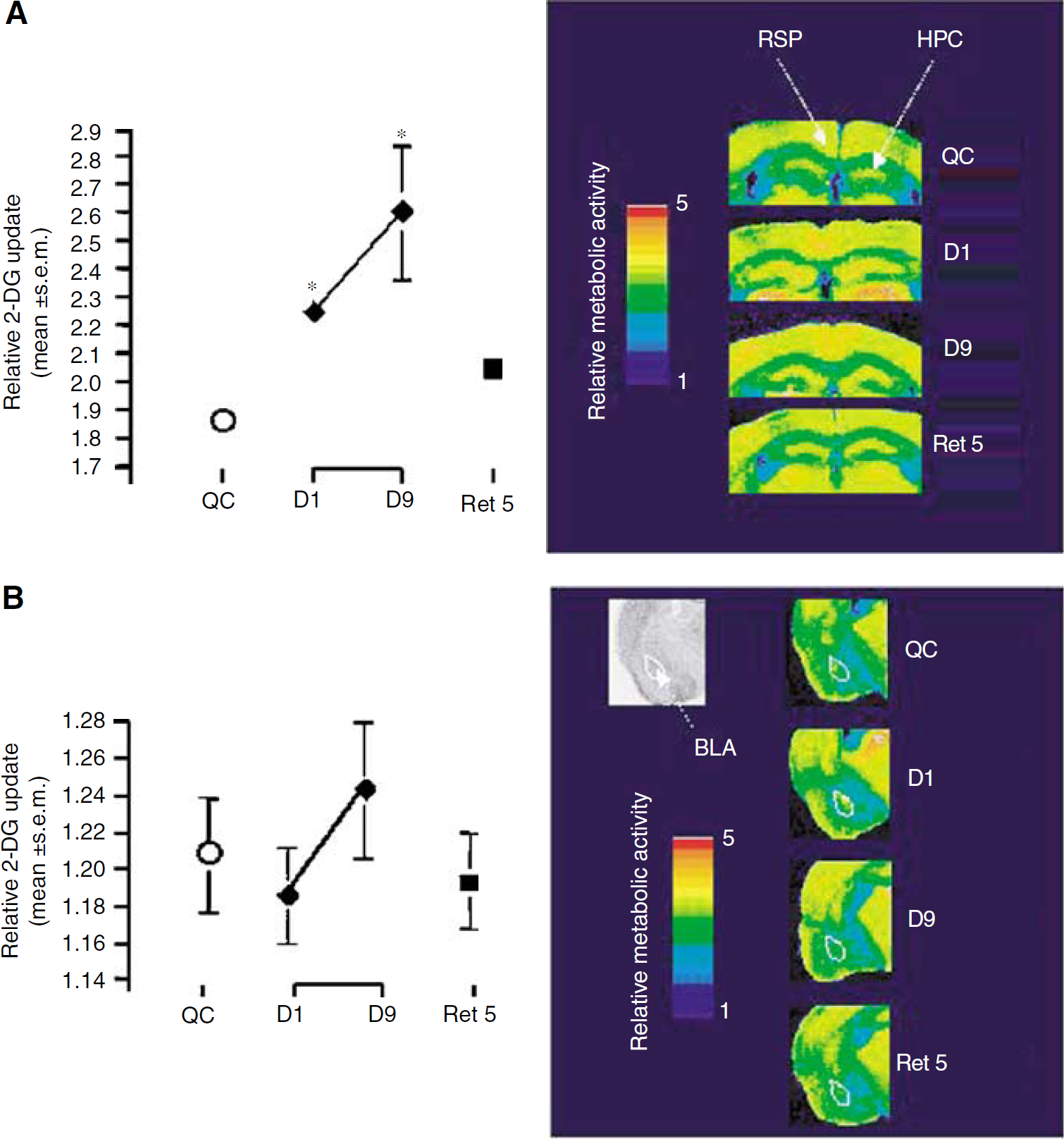
Changes in relative (14C)-2-deoxyglucose uptake and (14C)-2-deoxyglucose color-coded autoradiographs of coronal brain sections in the retrosplenial cortex (
Discussion
After its development in the late 1970s (Sokoloff et al, 1977), the 2DG autoradiographic method has proved to be one of the most suitable techniques to map brain activation in restrained as well as in freely moving animals. Recent advances in our understanding of its cellular and molecular bases suggest that the glucose utilization signal is a faithful (although indirect) reflection of glutamate-mediated synaptic activity (Bonvento et al, 2002; Pellerin and Magistretti, 1994). Over the years, the technique has been extensively used to unravel the involvement of specific brain areas in several paradigms involving somatosensory activation through various modalities (Coopersmith and Leon, 1984; Kennedy et al, 1976; Melzer et al, 1985; Schwartz et al, 1979). Furthermore, it has been applied to obtain functional maps of learningrelated activity such as in Pavlovian conditioning (Jones and Gonzalez-Lima, 2001), habituation (Gonzalez-Lima et al, 1989; Toga and Collins, 1981), or different working memory tasks (Friedman and Goldman-Rakic, 1988; Sybirska et al, 2000). The technique also offers the possibility to study the dynamic changes in the activation of a particular area along the different phases of a learning process. Taking advantage of this characteristic, we have monitored metabolic activity within the hippocampal formation along with the acquisition, consolidation, and retrieval of spatial information during a navigational task. Results obtained reveal to what extent the hippocampus and its different subregions participate to these different phases in parallel with some neocortical structures.
Acquisition of Episodic Informations
It is well established that the hippocampal formation plays a critical role in the acquisition and storage of informations expected to be integrated in episodic memory (Nadel and Moscovitch, 1998). While information pertaining to the episode (e.g., specific categories or attributes) would be encoded in neuronal assemblies located in various neocortical areas, the hippocampus was postulated to play a binding role to link the different elements of an episode distributed among these neocortical systems. More specifically, the hippocampus, by computing spatial relations, would provide a spatial scaffold (i.e., the contextual frame) onto which other elements constituting an episode can be associated. Such a particular operating mode for encoding an episode requires a concomitant activation of specific neuronal networks both within the hippocampus and in neocortical locations (Fernandez et al, 1998; Fink et al, 1996; Moscovitch, 1995). Our analysis of metabolic activity during the training period shows both an increase in 2DG uptake in the hippocampus as a whole and also within at least one neocortical structure, the retrosplenial cortex. As each trial can be assimilated to an ‘episode', changes in metabolic activity occurring simultaneously in the hippocampus and neocortical structures could be a reflection of the encoding of episodic information. Different neuronal ensembles in each location would contribute either to encode specific episodic features or create the contextual framework to bind them. Interestingly, a finer analysis within the hippocampus reveals that different subregions contribute in a heterogeneous manner to these neuronal networks, confirming the suggestion that the hippocampus participates in acquisition of spatial information not as a unitary structure but as an area divided in different functional components along its longitudinal axis (Hughes, 1965; Jung et al, 1994; Lepage et al, 1998; Moser and Moser, 1998; Poucet and Buhot, 1994; Stevens and Cowey, 1973). Indeed, recent studies provide support for the concept of functional heterogeneity along the anteroposterior axis within the hippocampus that may depend on distinctive information conveyed to separate parts of the hippocampus by inputs from different part of the entorhinal cortex (Hargreaves et al, 2005; Steffenach et al, 2005). Another interesting aspect highlighted by our subregional analysis of metabolic activity concerns the specific role of the CA1 area early during the acquisition phase. Initially, as mice were familiar with the RAM after 2 days of free exploration, a much larger portion of the CA1 area along the anteroposterior axis was activated as compared with the CA3 and DG, covering the intermediate and posterior parts of the hippocampus. This particular involvement of the CA1 area is consistent with the previous proposal that this area could be particularly important for acquisition of information in a familiar environment (Kesner et al, 2000; Lassalle et al, 2000; Lee and Kesner, 2002).
Consolidation
Memory consolidation is an important aspect of the learning and memory process that refers to a stabilization of the information after its initial encoding (Dudai, 2004; McGaugh, 2000; Squire, 1992). Several hypotheses have been proposed for its mechanism, inspired by the observation of retrograde amnesia in human subjects with lesions involving the hippocampus. One of these hypotheses, the ‘multiple-trace theory’ (Nadel and Moscovitch, 1998) postulates that each reactivation of an episodic memory results in the creation of a new hippocampally stored memory trace. Over time, as more traces are created, larger neuronal ensembles within the hippocampus would be activated. A test of this hypothesis requires analyzing activity in the hippocampal formation over its entire anteroposterior axis as performed here. Indeed, as the animal is repetitively exposed to the RAM task over the full training period, we observed a larger involvement of all three hippocampal subregions with time. Whether such extended activation of hippocampal areas reflects storage of new memory traces in the hippocampus proper cannot be deduced from our observations. However, even if it was the case, it could be only a temporary storage site as these areas are not reactivated on retrieval. Apart from the specific involvement of the hippocampus, it has been proposed that emotional arousal might play a critical role in memory consolidation, a component involving the basolateral nucleus of the amygdala (McGaugh, 2000). However, we did not observe metabolic activation of the amygdala during the learning period. Since animals were handled daily for 1 week prior training, this procedure might have contributed to reduce the importance of arousal in the present context.
From Episodic to Semantic Knowledge
From the acquisition of episodes sharing common features, it is possible to extract some knowledge that becomes independent of the context in which it was acquired and then could be applied to subsequent situations. This knowledge forms what is defined as semantic memory. In the context of spatial learning, navigational inferences can be made from the acquisition of spatial information through several distinct episodes (Eichenbaum, 2000). It was clearly shown that the hippocampus plays a critical role in creating associations among separated experiences necessary for making navigational inferences (Eichenbaum, 2000). Initially, we observed a constant performance from day 1 to day 3. Then a substantial improvement in performance starting from day 5 up to day 9 of training was observed. It is purported that such enhanced performance could be because of the establishment of a so-called cognitive map (O'Keefe and Nadel, 1978) with a complementary role of the dentate/CA3 and CA1 modules (Jacobs and Schenk, 2003). Such a map could be created by extraction of episodic information across the numerous trials of the task. Whether the cognitive map represents simple associations between navigational cues and locations, or a more elaborate representation of the spatial environment facilitating navigation cannot be distinguished with our protocol, although it has been suggested that for the eight arm RAM both mechanisms probably contribute to the formation of a cognitive map in animals with intact hippocampus (Hodges, 1996). Metabolic activities recorded during this period corresponding to the formation of the cognitive map reveal a gradual recruitment of each hippocampal subregions. Such an observation is consistent with the prominent role of the hippocampus in establishing spatial associations and creating semantic memories related to navigation that might require the involvement of larger neuronal networks within each hippocampal subregion along the anteroposterior axis (Eichenbaum, 2000). However, based on the fact that metabolic activity is concomitantly enhanced in the retrosplenial cortex, it is unlikely that the hippocampus is the sole component involved in this process. As pointed out previously, hippocampal–neocortical interactions are certainly crucial for extraction and storage of semantic information (Nadel and Moscovitch, 1998).
Retrieval
The role of the hippocampal complex in memory retrieval has been extensively discussed (Jarrard, 1995; Morris et al, 2003; Nadel and Moscovitch, 1997; Rosenzweig et al, 2003). Observations based on hippocampal damage in humans and animais, leading to retrograde amnesia, have suggested that newly formed memories are not stored permanently in the hippocampus (Anagnostaras et al, 1999; Kim and Fanselow, 1992; Nadel and Bohbot, 2001; Zola-Morgan and Squire, 1990). If they were, it would be expected that recall in an intact animal should lead to the reactivation of all the same hippocampal areas that were activated at the end of the consolidation phase. Consistent with the previous hypothesis, our data show the reactivation of only a portion of the DG, in contrast to extensive activation of all three hippocampal subregions after 9 days of training. Different theories have been proposed to account for the apparently transient involvement of the hippocampus in memory storage. One suggests that although the hippocampus is initially essential for memory formation and storage, with time, memory representation is transferred in distributed neocortical sites (Bontempi et al, 1999; Maviel et al, 2004; Squire, 1992). In support of this model, it was shown using a similar behavioral task and experimental approach as in our study that indeed hippocampal activation no longer occurs on recall after 25 days while neocortical activation develops (Bontempi et al, 1999). Moreover, it was more recently shown that specific neocortical areas such as the prefrontal, anterior cingulate, and retrosplenial cortices undergo molecular and structural changes consistent with the gradual establishment of extrahippocampal spatial maps (Maviel et al, 2004). In agreement with Bontempi et al (1999), we observed a reactivation of the hippocampus on recall at 5 days. Our finer analysis however revealed that only a specific subregion of the hippocampus (i.e., DG) is activated and within a particular section (i.e., the posterior part). Whether this particular area has acquired the capacity to coordinate the activation of neocortical sites or rather is necessary for reconstructing episodic memory because it contains specific traces cannot be ascertained. Moreover, in contrast to what Bontempi et al (1999) have reported, we observed in parallel to DG activation an involvement of the retrosplenial cortex at this stage. This observation, together with the activation of the retrosplenial cortex throughout the acquisition and consolidation process, does not support the idea of a simple transfer of memory representation from hippocampus to neocortex but rather underscores the dynamic hippocampal–neocortical interplay necessary for the establishment of spatial memory that might occur in several, discrete stages. Moreover, our study in general highlights the fact that information is probably distributed not only between separate brain areas (e.g., hippocampus and neocortex) but also within the same structure like the hippocampus. Such hippocampal heterogeneity, which probably reflects distinct neuronal ensembles exhibiting dynamic features, offers the possibility to recruit separate ensembles in networks to participate in the multiple operations of a complex learning and memory task.
Footnotes
Acknowledgements
We express our gratitude to E Bays for the invaluable technical assistance provided all along this study. We also thank Professor E Welker for sharing his expertise with deoxyglucose autoradiography and N Muller as well as F Rochat for technical assistance.
