Abstract
Phosphorylation of cyclic adenosine monophosphate (AMP) response element binding protein (CREB) was examined immunohistochemically in the corpus callosum of the rat brain at various time points after 90-minute focal cerebral ischemia. Focal ischemia was induced by occlusion of the middle cerebral artery (MCA) using the intraluminal suture method. Sham animals showed that numerous oligodendrocytes (OLGs) constitutively express unphosphorylated CREB. Local cerebral blood flow (lCBF) measured by the14 C-iodoantipyrine method was reduced from 44.2 ± 15.4 (mL 100 g−1 min−1) to 18.4 ± 3.8 and from 53.9 ± 14.4 to 4.8 ± 4.5 in the medial and the lateral regions of the corpus callosum, respectively, during MCA occlusion (MCAO). After release of the MCAO, lCBF recovered to the control level in each region. The medial region of the corpus callosum showed a marked increase in phosphorylated CREB-positive OLGs at 3.5 hours of recirculation, and it remained increased until 2 weeks of recirculation as it gradually declined. The activation of CREB phosphorylation in the OLGs was accompanied by expression of antiapoptotic protein bcl-2, normal staining with cresyl violet, and negative TUNEL (terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling) staining. Myelination detected by immunostaining with anti-myelin basic protein (MBP) antibody and anti-myelin associated glycoprotein (MAG) antibody remained normal in the medial region of the corpus callosum. The lateral region of the corpus callosum showed a significant but only transient increase in phosphorylated CREB-positive OLGs at 3.5 hours of recirculation, which was followed by a rapid decrease during the subsequent recirculation period. Expression of bcl-2 was suppressed in this region, and demyelination became apparent. These findings suggest that signal transduction through CREB phosphorylation may be closely associated with survival of OLGs and maintenance of myelination in the corpus callosum after cerebral ischemia.
Oligodendrocytes (OLGs), a major cellular component of white matter, are the only myelin-forming cells in the brain, and thus play a pivotal role in the proper execution of neural functions (Hof et al., 1999). Recent studies have revealed that OLGs are susceptible to ischemic stress (Mandai et al., 1997; Pantoni et al., 1996; Petito et al., 1998; Shibata et al., 2000), hypoxia combined with hypoglycemia (Lyons and Kettenmann, 1998), and stimulation of α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid/kainate receptors (McDonald et al., 1998). Pantoni et al. (1996) have reported that pathologic changes in OLGs and myelin precede the appearance of necrotic neuronal damage in the permanent ischemia model in the rat brain.
Developing OLGs express elevated levels of the transcription factor cyclic adenosine monophosphate (cAMP) response element binding protein (CREB) (Sato-Bigbee and Yu, 1993; Sato-Bigbee et al., 1994). Analysis of CREB expression in OLGs isolated from rat brain at different postnatal ages has shown it to be maximal at 14 days, suggesting that it plays a crucial role in the developmental period that immediately precedes peak myelination (Sato-Bigbee and Yu, 1993; Sato-Bigbee et al., 1994). CREB synthesis inhibition experiments have indicated that CREB is one of the mediators of the cAMP-dependent stimulation of OLG differentiation (Sato-Bigbee and DeVries, 1996). CREB becomes activated through phosphorylation of its serine residue (Ser133) and initiates transcription of various genes containing the CRE motif in their promoter regions (Shaywitz and Greenberg, 1999). CREB phosphorylation in OLGs can be up-regulated in a protein kinase A (PKA)-dependent fashion at all developmental stages, whereas the capacity for calcium-dependent and mitogen-activated protein kinase (MAPK)-dependent phosphorylation seems to be lost in mature OLGs (Sato-Bigbee et al., 1999; Wegner, 2000). Proliferation of OLG precursor cells induced by neurotrophin-3 recently has been shown to be dependent on CREB phosphorylation through the signaling pathways involving MAPK and protein kinase C (PKC) (Johnson et al., 2000). Implication of cAMP in the maturation of OLGs and in the expression of several genes, such as myelin basic protein (MBP), P2, P0, proteolipid protein (PLP), and myelin-associated glycoprotein (MAG), has been extensively described (Anderson and Miskimins, 1994; Bharucha et al.,1993; Jensen et al., 1993; Ye et al., 1992). However, although the myelin oligodendrocyte glycoprotein (MOB) gene contains the CRE motif in its promoter region (Jaquet et al., 1999), other myelin protein genes, such as MBP, P2, P0 and MAG, lack the CRE motif in their promoter regions (Li et al., 1994; Sato-Bigbee and De Vries, 1996), suggesting that CREB may regulate expression of MBP, P2, P0, and MAG indirectly or through expression of MOG. Nevertheless, CREB is now considered to be an important mediator of myelination and cell process outgrowth in OLGs (Sato-Bigbee and DeVries, 1996; Wegner, 2000).
Persistent activation of CREB phosphorylation has been reported to be closely associated with neuronal survival in the cerebral cortex during the postischemic recirculation period after focal cerebral ischemia (Tanaka et al., 1999a, 1999c) or after hypoxic-ischemic stress in immature rats (Walton et al., 1996). The cerebral white matter is affected in most cases of acute ischemic stroke, and experimental data suggest that the cerebral white matter and OLGs are highly vulnerable to ischemia (Pantoni et al., 1996; Shibata et al., 2000). Although activation of CREB phosphorylation in OLGs after focal cerebral ischemia has been described briefly (Irving et al., 2000), the chronologic and topographic features of CREB phosphorylation in the corpus callosum and its relation to OLG survival and myelination have never been reported. In the current study, the authors therefore examined the chronologic and topographic changes in the phosphorylation state of CREB in OLGs in the corpus callosum after 90-minute occlusion of the middle cerebral artery (MCA) in the rat.
MATERIALS AND METHODS
The protocol described here was in accordance with the NIH Guide for Care and Use of Laboratory Animals and received prior approval as meeting the Animal Experimentation Guidelines of Keio University School of Medicine by the Laboratory Animal Care and Use Committee.
Animals and surgical procedures
Adult male Sprague-Dawley rats (Japan Laboratory Animals, Tokyo, Japan) weighing 280 to 320 g were divided into two groups: an ischemia group (n = 41) and a sham group (n = 6). The animals in the ischemia group were anesthetized with 1.0% to 1.5% halothane in 30% oxygen/70% nitrogen during the operation. A temperature probe (TD-300; Shibaura Electronics, Tokyo, Japan) was inserted into the rectum, and a heat lamp was used to maintain the rectal temperature at 37.0°C to 37.5°C. The MCA was occluded by the intraluminal filament technique, as described previously (Suzuki et al., 1999; 2000; Tanaka et al., 1999b). Briefly, a 3–0 nylon monofilament suture (Matsuda Ikakogyo, Tokyo, Japan) was inserted through the right external carotid artery into the internal carotid artery to obstruct the origin of the right MCA. The length of suture inserted beyond the origin of the internal carotid artery was 19 mm.
After the intraluminal filament had been fixed in position, the neck incision was closed with a silk thread. The animals were allowed to recover from the anesthesia, and 1 hour after MCA occlusion (MCAO), the extent of their neurologic deficits was graded as severe, moderate, mild, or no deficit based on the neurologic examination method devised by Bederson et al. (1986). Briefly, animals consistently circling toward the left (paretic) side were graded as 3. Rats consistently showing a reduced resistance to lateral push toward the left side with no circling movement were graded as 2. Rats with any amount of consistent forelimb flexion and no other abnormalities were graded as 1. Rats with no neurologic deficits were assigned a grade of 0. Only animals with grade 2 or 3 were used for further study as the ischemia group (n = 37), because these animals were expected to invariably reveal a large infarct area in the territory of the MCA (Bederson et al., 1986).
After 90 minutes of MCA occlusion, the rats were anesthetized again with the same anesthetic combination as described above, and the intraluminal suture was carefully removed. The animals were allowed to survive for 3.5 hours (n = 7), 12 hours (n = 6), 24 hours (n = 8), 48 hours (n = 8), 7 days (n = 4), or 14 days (n = 4) with free access to food and water. In the sham group, only the neck incision was made to expose the right common, external, and internal carotid arteries. The other procedures were identical to those used in the ischemia group.
At the end of the recirculation period or 24 hours after sham surgery, the brains were perfusion-fixed with 4% paraformaldehyde in 0.1 mol/L phosphate buffer as described previously (Suzuki et al., 1999, 2000). The fixed brains then were frozen in powdered dry ice, and 20-μm-thick coronal sections were prepared on a cryostat (Cryocut 1800; Leica Instruments GmbH, Nussloch, Germany). The sections were thaw-mounted onto silane-coated slides and stored at −30°C.
Immunohistochemistry
Immunohistochemistry was performed using an avidin-biotinylated enzyme complex system (Vectastain ABC Elite Kit, Vector Laboratories, Burlingame, CA, U.S.A.), as reported previously (Tanaka et al., 1999a, 1999c). Briefly, after preincubation, the sections were incubated overnight with affinity-purified rabbit polyclonal anti-rat phosphorylated CREB antibody or anti-rat CREB antibody (Upstate Biotechnology, Lake Placid, NY, U.S.A.), each at a 1:100 dilution. The former antibody is specific for phosphorylated CREB in which the Ser133 residue is phosphorylated (Ginty et al., 1993), whereas the latter antibody binds to CREB regardless of its phosphorylation state. After completion of the primary incubation, the slides were rinsed in phosphate-buffered saline and subsequently incubated with the biotinylated secondary antibody (anti-rabbit goat antibody) and 1% normal goat serum for 90 minutes. Sections then were rinsed and incubated with avidin-biotinylated peroxidase conjugate. Finally, they were developed in 0.02% diaminobenzidine with 0.02% H2 O2. Negative control staining was performed with normal rabbit serum as a substitute for the primary antibody. The specificity of the immunoreactivity of the anti-phosphorylated CREB antibody has already been examined, as reported previously (Tanaka et al., 1999a). Adjacent sections were stained with cresyl violet for conventional histologic examination.
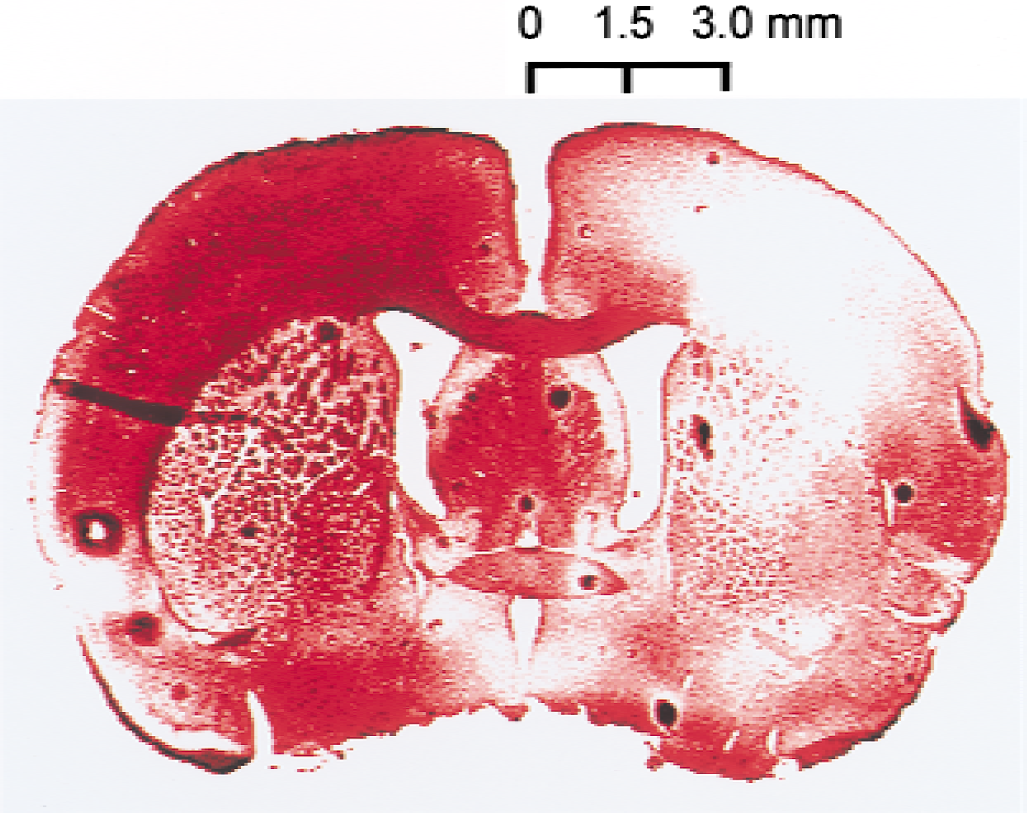
Absolute immunoreactive cell counts (per 0.024 mm2 = 154 μm × 154 μm) were made in the coronal section located 0.20 mm anterior to the bregma with an ocular micrometer (Nikon, Tokyo, Japan) attached to a light microscope. The regions of interest (ROIs) were set in the medial region (1.5 mm from the midline) and the lateral region (3.0 mm from the midline) in the corpus callosum on both sides according to a rat brain atlas (Paxinos and Watson, 1997), as shown in Fig. 1. The location of these ROIs was determined based on changes in local cerebral blood flow (CBF) during MCA occlusion, as described below. In the current model, the medial region of the corpus callosum on the ischemic side belongs to the outer border zone of severe ischemic area, whereas the lateral region of the corpus callosum on the ischemic side locates in the severe ischemic area. Counting was performed on three consecutive slices, and the mean value was calculated.
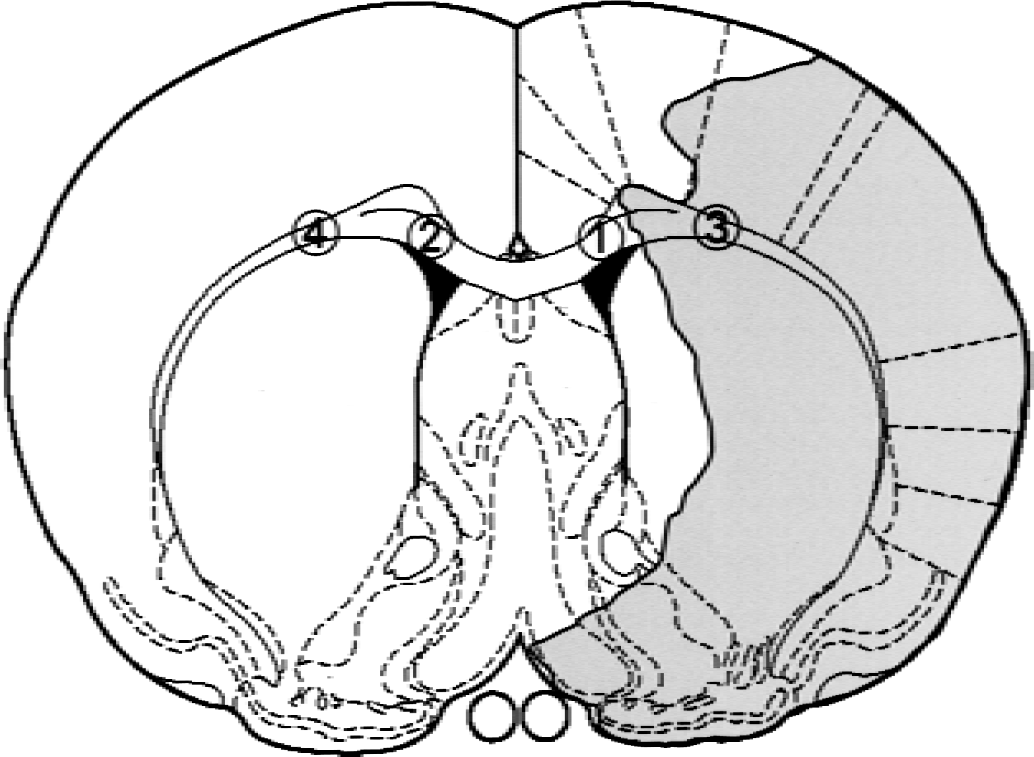
Regions of interest set in the coronal brain slice located 0.2 mm anterior to bregma based on the rat brain atlas by Paxinos and Watson (1997). Shaded area represents severe ischemic regions during occlusion of the middle cerebral artery. 1 and 2: medial regions in the corpus callosum; 3 and 4: lateral regions of the corpus callosum.
In parallel with the above staining procedures, the tissue was immunohistochemically examined for expression of GST-π, a specific marker of the cell body of mature OLGs (Tansey and Cammer, 1991; Shibata et al., 2000), MBP, MAG, bcl-2, and glial fibrillary acidic protein (GFAP), a specific marker of astrocytes. The authors used rabbit polyclonal antibodies raised against GST-π (MBL, Nagoya, Japan) at a dilution 1: 200, MBP (Nichirei, Tokyo, Japan), MAG (courtesy of Dr. Y. Matsuda, National Center for Neurology and Psychiatry, Tokyo, Japan, and Dr. H. Asou, Tokyo Metropolitan Institute of Gerontology, Tokyo, Japan) at a dilution 1:500, bcl-2 at a dilution 1:100 (PharMingen, San Diego, CA, U.S.A.), and GFAP (Dako, Glostrup, Denmark) at a dilution 1:300. The sections stained with the anti-GST-π antibody were permeabilized with 0.1% Triton X-100/phosphate-buffered saline at room temperature for 10 minutes before blocking. The brain slices immunostained for bcl-2 were incubated in a jar containing 10 mmol/L citrate buffer, pH 6.0, at 98°C for antigen enhancement before blocking. The specificity of this antibody has already been described, and it has been successfully applied to the study of ischemia (Krajewski et al., 1995). Negative control staining was performed with normal rabbit serum as a substitute for the primary antibody. Immunoreactivity was visualized by an avidin-biotinylated enzyme complex system and diaminobenzidine. Microglia were histochemically identified by using isolectin-B4 from Griffonia simplicifolia seeds (GSAI-B4; Sigma, St. Louis, MO, U.S.A.), as described previously (Suzuki et al., 1999).
To confirm the localization of phosphorylated CREB to oligodendrocytes, the authors performed double-immunostaining with an oligodendrocyte marker, anti-adenomatous polyposis coli antibody (mouse monoclonal anti-adenomatous polyposis coli [anti-APC] antibody (Ab-7), Oncogene, Boston, U.S.A.) (Bhat et al., 1996; Liu et al., 2001), and anti-phosphorylated CREB antibody (rabbit polyclonal antibody; Upstate Biotechnology). After incubation with the above primary antibodies for 16 hours, sections were incubated for 2 hours with Texas Red-conjugated anti-rabbit IgG antibody (Amersham, Buckinghamshire, U.K.) and FITC-conjugated anti-mouse IgG antibody (Amersham). Sections were examined by fluorescent microscopy (Eclipse E-800, Nikon).
For detection of in situ DNA fragmentation, terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling (TUNEL) staining was performed using an in situ cell death detection kit (Boehringer Mannheim, Mannheim, Germany). Incorporated deoxyuridine triphosphate was visualized using diaminobenzidine. Negative controls for TUNEL staining were prepared by omitting terminal deoxynucleotidyl transferase. Double-staining of TUNEL and phosphorylated CREB also was performed using the above kit and antibody. TUNEL staining and phosphorylated CREB were visualized with FITC and Texas Red, respectively.
Western blot analysis
Western blot was performed to confirm the specificity of the antibodies, which were used for immunostaining of phosphorylated CREB (molecular weight = 43 kDa) and bcl-2 (molecular weight = 26 kDa) in the current study. Animals were decapitated under deep anesthesia with pentobarbital after the sham operation (n = 3) or 24 hours of recirculation after 90-minute MCAO as described above (n = 3). Samples were taken from the motor area of the frontoparietal cortex (periischemic area) on the operated side. Tissues were homogenized in a Teflon-coated Potter homogenizer containing buffer (0.32 mol/L sucrose, 1 mmol/L EDTA, 5 mmol/L Tris-HCl (pH 7.4), 0.1 mmol/L phenylmethylsulfonylfluoride, 10 μg/mL leupeptin, 1.25 μg/mL pepstatin A, 1 mmol/L beta-mercaptoethanol, 0.1 mol/L Na3 VO4, 50 mmol/L NaF, 2 mmol/L sodium pyrophosphate). The homogenates then were centrifuged at 920×g at 2°C for 10 minutes. The resultant pellet (P1) was used as the crude nuclear fraction. The supernatants were further centrifuged at 10,500×g for 60 minutes at 2°C, and the pellets (P2 + P3) were collected. P1 and P2 + P3 were suspended with a homogenization buffer. Protein concentration was determined using a Protein Assay Kit (Bio-Rad Laboratories, Hercules, CA, U.S.A.) with bovine serum albumin as the standard. The pellets were subjected to 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis in the buffer system of Laemmli, and protein bands were transferred from the gel to a nitrocellulose membrane (Hybond ECL; Amersham) using a transfer apparatus and a blotting buffer including 0.192 mol/L glycine and 5% methanol (pH 8.4). The membrane then was incubated with 1:500 dilution of an affinity purified rabbit polyclonal antibody against phosphorylated CREB (Upstate Biotechnology) or with 1:500 dilution of an affinity purified rabbit polyclonal antibody against bcl-2 (PharMingen). The membrane was next reacted with horseradish peroxidase-linked (HPL) anti-rabbit antibody (Amersham). Protein bands were detected by an enhanced chemiluminescence method (ECL kit; Amersham) and exposed to Kodak X-OMAT film (Eastman Kodak, Rochester, NY, U.S.A.).
Measurement of cerebral blood flow
Cerebral blood flow was measured by the14 C-iodoantipyrine method in this ischemia model, as described in earlier reports (Tanaka et al., 1999b). Briefly, 1.1 MBq of 4-iodo-N-methyl-[14C]antipyrine ([14C]IAP; specific activity = 1.9 GBq/mmol; NEN Life Science Products, Boston, MA, U.S.A.) dissolved in 0.7 mL of saline was infused at a constant speed through the venous catheter for a period of 45 seconds after the sham surgery (n = 4), at the end of MCAO (n = 4), or the end of the 3.5-hour recirculation period (n = 4). Simultaneously, arterial blood samples (20 μL) were collected every 3 to 5 seconds into capillary pipettes to count timed radioactivity. At the end of the infusion period, the animals were decapitated, and the brains were quickly removed and frozen by immersion in liquid Freon 22 (Asahi Glass, Tokyo, Japan). The frozen brains were cut into 20-μm-thick coronal sections on a cryostat and then exposed to x-ray film (Hyperfilm-βmax; Amersham, Buckinghamshire, U.K.), together with calibrated14 C-embedded methylmethacrylate standards, for 7 days. Quantitative densitometric analysis of the autoradiograms was performed using a computerized digital image analysis system (Inquiry; Loats Associates, Westminster, MD, U.S.A.).
Data analysis
All data are expressed as means ± SD. The data were tested by two-way analysis of variance followed by post hoc analysis using the modified t-test with Bonferroni correction for multiple simultaneous comparisons. P < 0.05 was considered significant.
RESULTS
Cerebral blood flow
Table 1 summarizes the CBF data for each of the regions in the corpus callosum. The medial region (at 1.5 mm from the midline) on the right (operated side) side showed a moderate (58.4%) but statistically significant reduction in CBF during MCAO, followed by slight overshoot recovery during the recirculation period. The lateral region (at 3.0 mm from the midline) on the right side exhibited a more marked (91.9%) reduction in CBF during MCAO that was followed by good recovery during the recirculation period. Neither the medial nor the lateral region on the left (nonoperated) side showed any significant changes in CBF during the experiment.
Local cerebral blood flow (mL 100 g −1min−1) in the corpus callosum

Values are means ± SD. MCAO, middle cerebral artery occlusion.
P< 0.05 versus sham.
P< 0.05 versus the medial region.
Representative autoradiograms of CBF are shown in Fig. 2. The lateral region of the corpus callosum on the occluded side became severely ischemic, whereas the medial region of the corpus callosum was spared any marked decrease in CBF. Cerebral blood flow was restored to the normal range during the recirculation period.
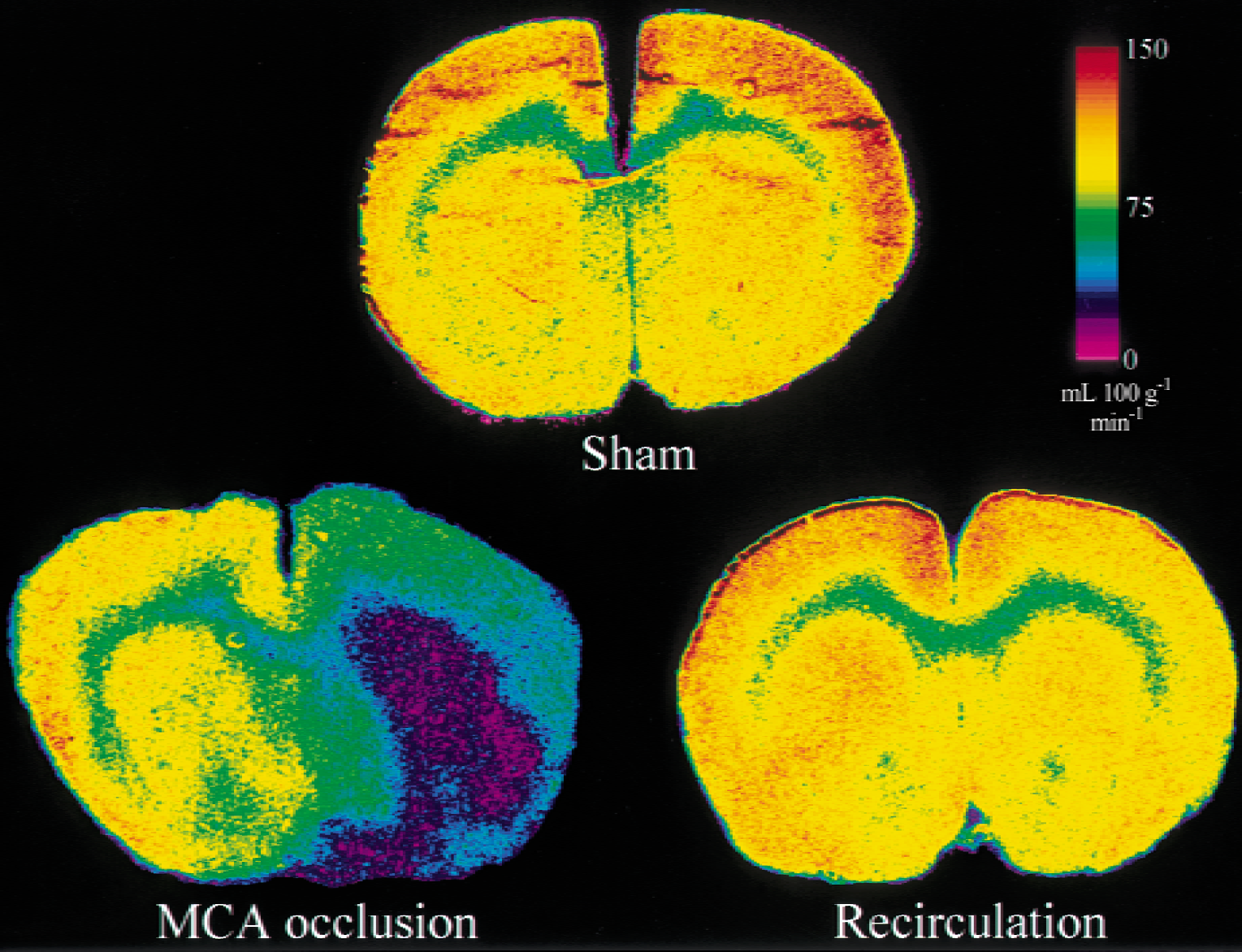
Representative color-coded autoradiograms of cerebral blood flow obtained from a sham animal
Western blot analysis
Figure 3 is a representative Western blot of samples from the sham animal (lane 1) and the periischemic area (lane 2), showing that the antibodies used in the current study specifically detect phosphorylated CREB at 43 kDa and bcl-2 at 26 kDa, respectively. Expression of phosphorylated CREB and bcl-2 was enhanced in the periischemic area compared with the sham animal.
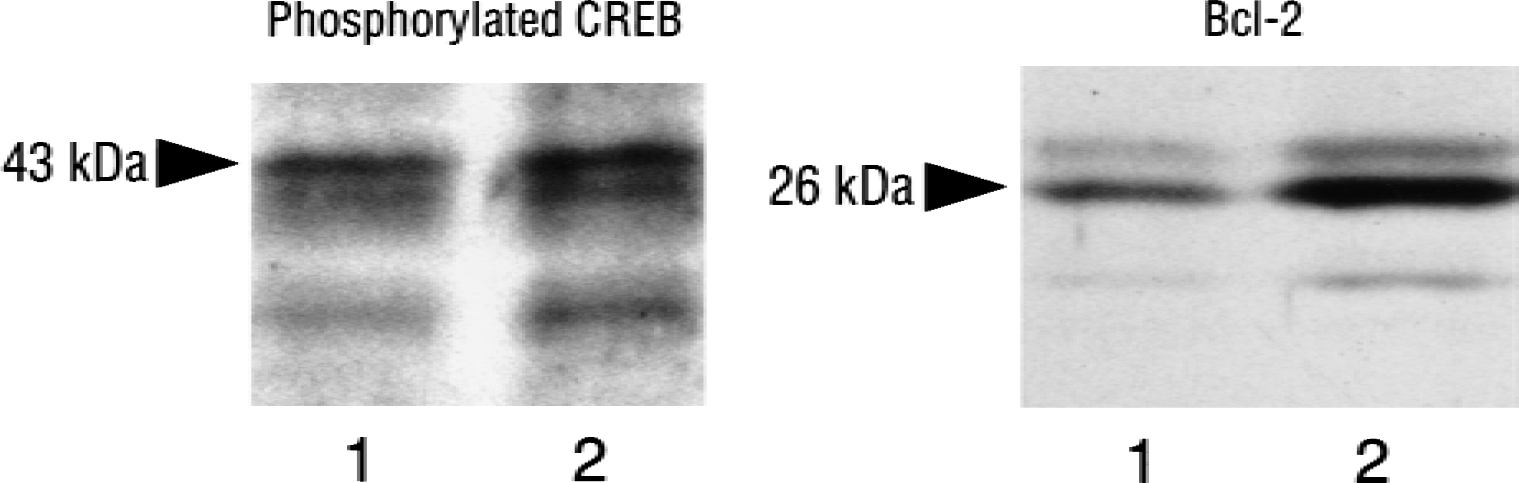
Representative Western blot of samples taken from the motor area of the frontoparietal cortex of a sham animal (lane 1) and an animal subjected to 24 hours of recirculation after 90-minute middle cerebral artery occlusion (lane 2, periischemia area). The left and right columns show the phosphorylated cyclic adenosine monophosphate response element binding protein (CREB) and bcl-2, respectively.
Histologic evaluation
Sham group.
As shown in Fig. 4A, cresyl violet staining in the corpus callosum revealed a number of cells with characteristic morphologic features consistent with those of OLGs, including a compact, rounded appearance and arrangement in beadlike rows with sparse cytoplasm. Indeed, as shown in Fig. 4B, these cells were immunopositive for GST-π, a specific marker of the cell body of mature OLGs. CREB was found to be constitutively expressed in OLGs (Fig. 4C), although virtually no phosphorylation of CREB was detected, as shown in Figs. 4D and 5.
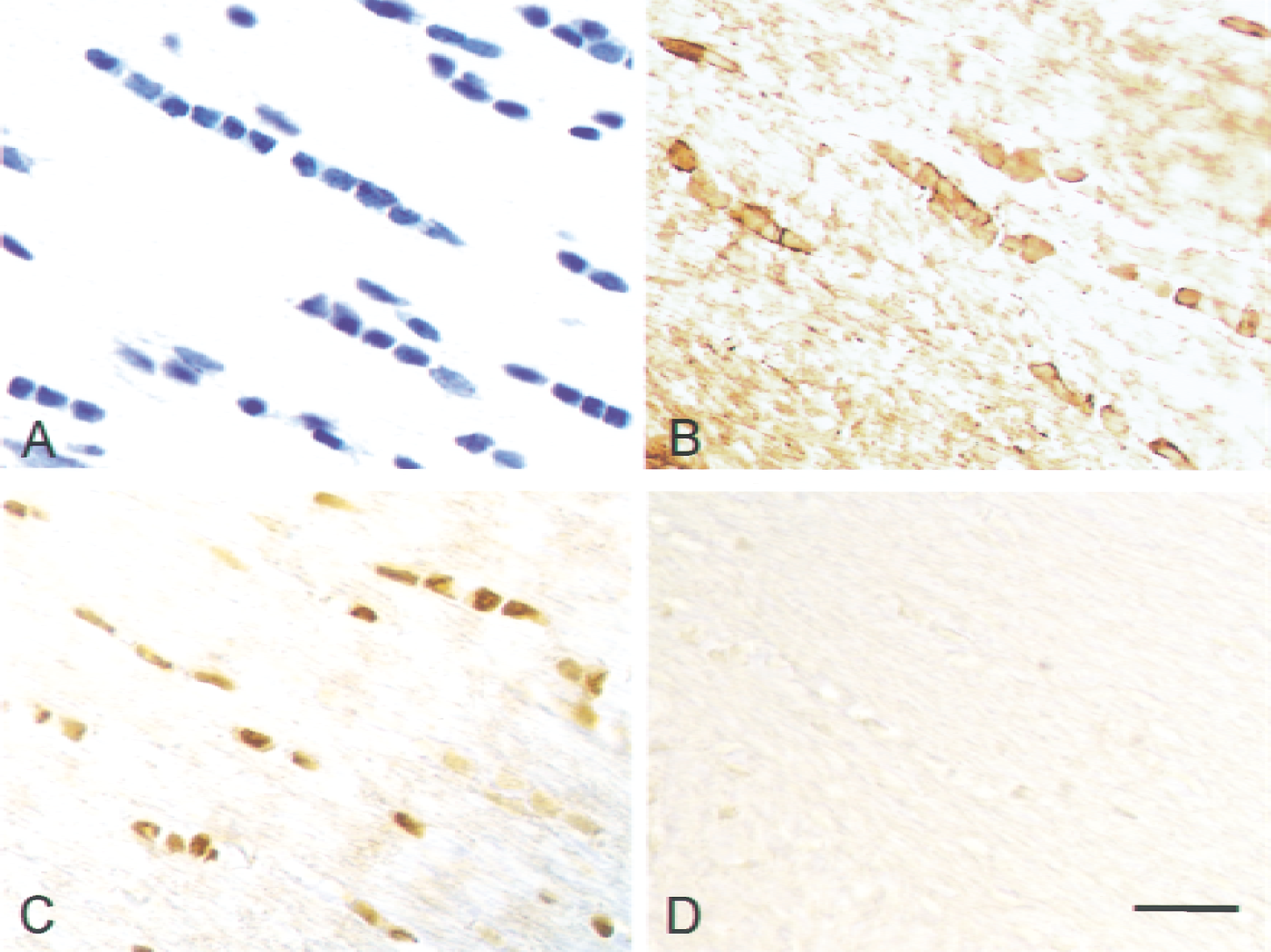
Representative photomicrographs of the corpus callosum from a sham animal obtained by staining with cresyl violet

Representative photomicrographs showing immunoreactivity with anti-phosphorylated cyclic adenosine monophosphate response element binding protein antibody in the lateral region
Postischemia group.
The representative photomicrographs in Fig. 5 show the immunoreactivity for phosphorylated CREB in the corpus callosum in each recirculation period. At 3.5 hours of recirculation, marked increases in immunoreactive cells were observed on the ischemic side, especially in the medial region, and the nonischemic side also showed a mild increase in immunopositive cells. According to the immunostaining pattern (compact, rounded appearance in beadlike rows) and parallel staining with cresyl violet, anti–GST-π antibody, anti-GFAP antibody, and GSAI-B4, the observed immunoreactivity was concluded to be present exclusively in the nuclei of OLGs. After 12 hours of recirculation, the lateral region on the ischemic side showed a rapid decrease in the number of immunopositive cells, whereas a significant increase in immunopositive cells was still observed in the medial region until 48 hours of recirculation. The medial region on the nonischemic side still showed a few immunopositive cells until 48 hours of recirculation with a gradual decline in the number of immunoreactive cells. At each time point, the immunoreactive cells were concluded to be exclusively OLGs based on their immunostaining pattern, parallel staining with cresyl violet, anti–GST-π antibody, anti-GFAP antibody, and GSAI-B4, and the following double-immunostaining.
Figure 6 shows representative photomicrographs of double-immunostaining with anti-phosphorylated CREB antibody and an oligodendrocyte marker, anti-APC antibody, in the medial region of the corpus callosum on the ischemic side. The anti-APC antibodies have been known to stain the cell bodies of OLGs (Bhat et al., 1996; Liu et al., 2001). As clearly shown in Fig. 6, expression of phosphorylated CREB was localized exclusively in the nuclei of OLGs at each time point.
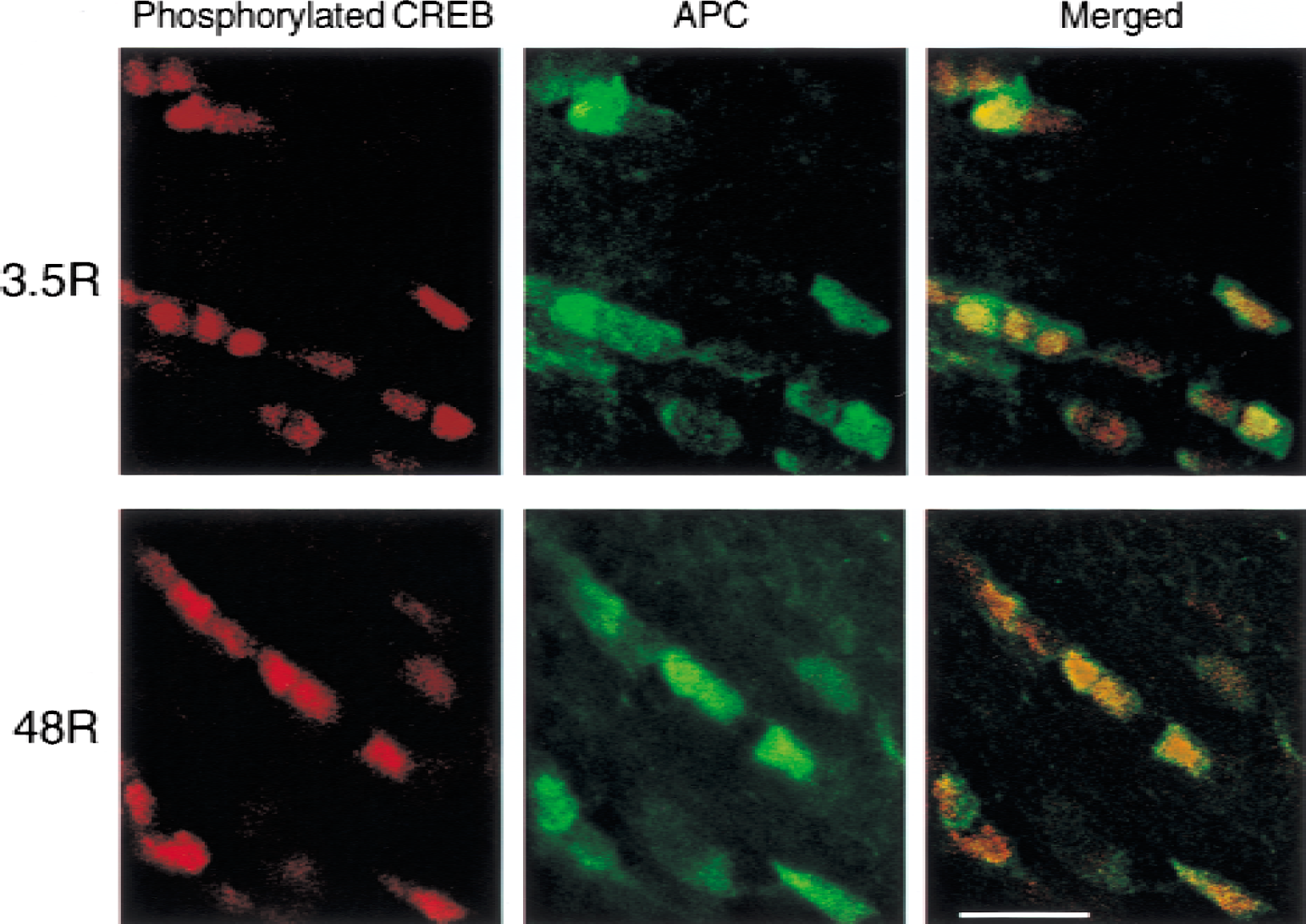
Representative photomicrographs of double-immunostaining with anti-phosphorylated cyclic adenosine monophosphate response element binding protein (CREB) antibody, and anti-adenomatous polyposis coli (APC) antibody, a marker of oligodendrocyte cell body, in the medial region of the corpus callosum on the ischemic side after 3.5 (
Figure 7 shows representative photomicrographs obtained from the medial region of the corpus callosum on the ischemic side at 48 hours of recirculation. Cresyl violet staining (Fig. 7A) revealed that normal OLG morphology was maintained in this region. The OLGs were immunopositive for phosphorylated CREB (Fig. 7B) as well as bcl-2 (Fig. 7C), but completely negative for TUNEL staining (Fig. 7D). By contrast, intact OLGs disappeared based on staining with cresyl violet and anti–GST-π antibody in the lateral region on the ischemic side at 48 hours of recirculation, and there was no definite immunoreactivity for phosphorylated CREB or bcl-2.
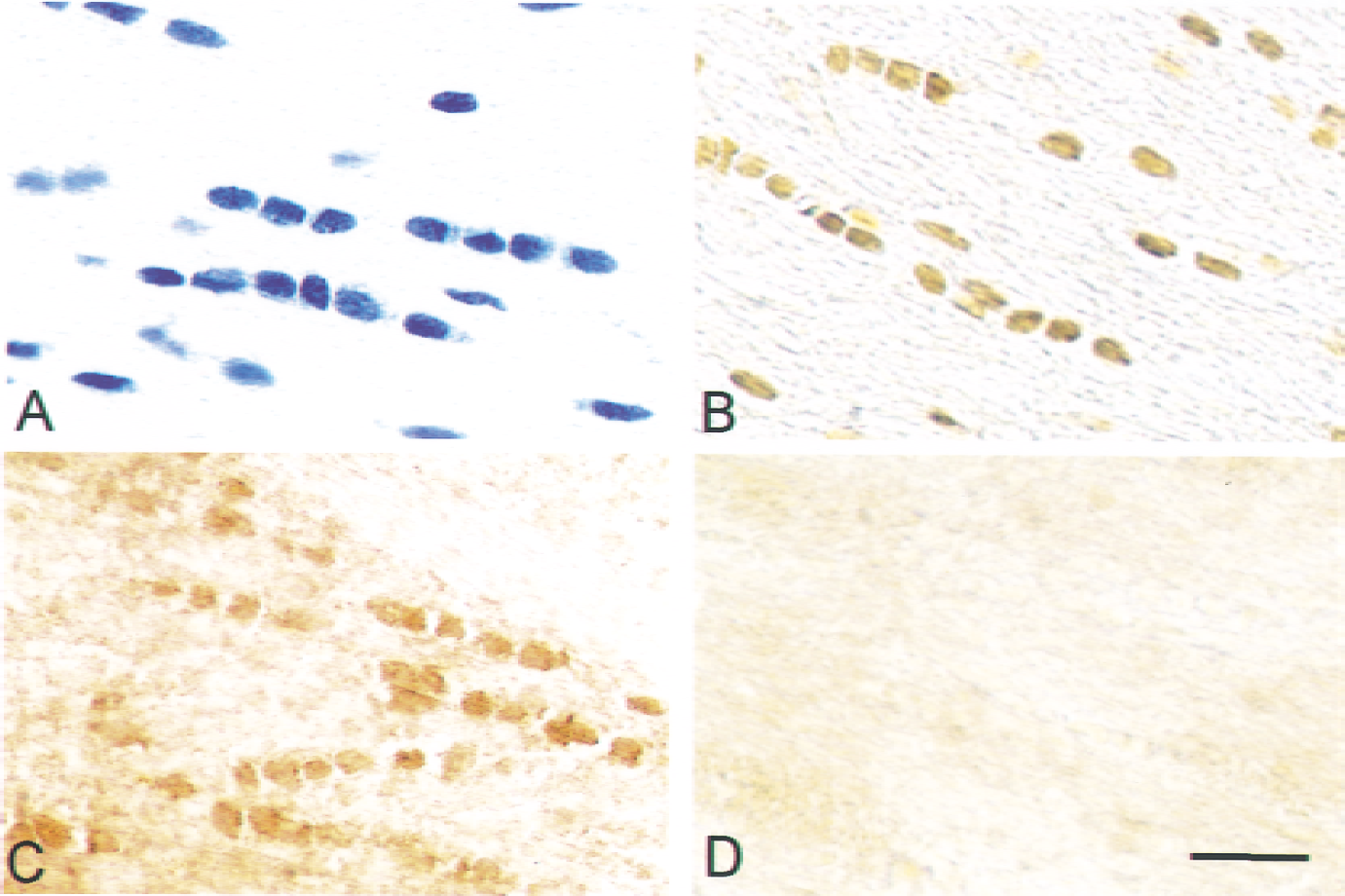
Representative photomicrographs in the medial region in the corpus callosum on the ischemic side obtained after 48 hours of recirculation after 90-minute middle cerebral artery occlusion. Sections were stained with cresyl violet
Figure 8 shows representative photomicrographs of double-immunostaining with TUNEL and anti-phosphorylated CREB antibody in the medial and lateral regions of the corpus callosum on the ischemic side after 48 hours of recirculation. In the medial region, phosphorylated CREB-positive cells were clearly detected without any positive TUNEL staining. In the lateral region, there were no phosphorylated CREB-positive cells, whereas positive TUNEL staining was clearly noted.
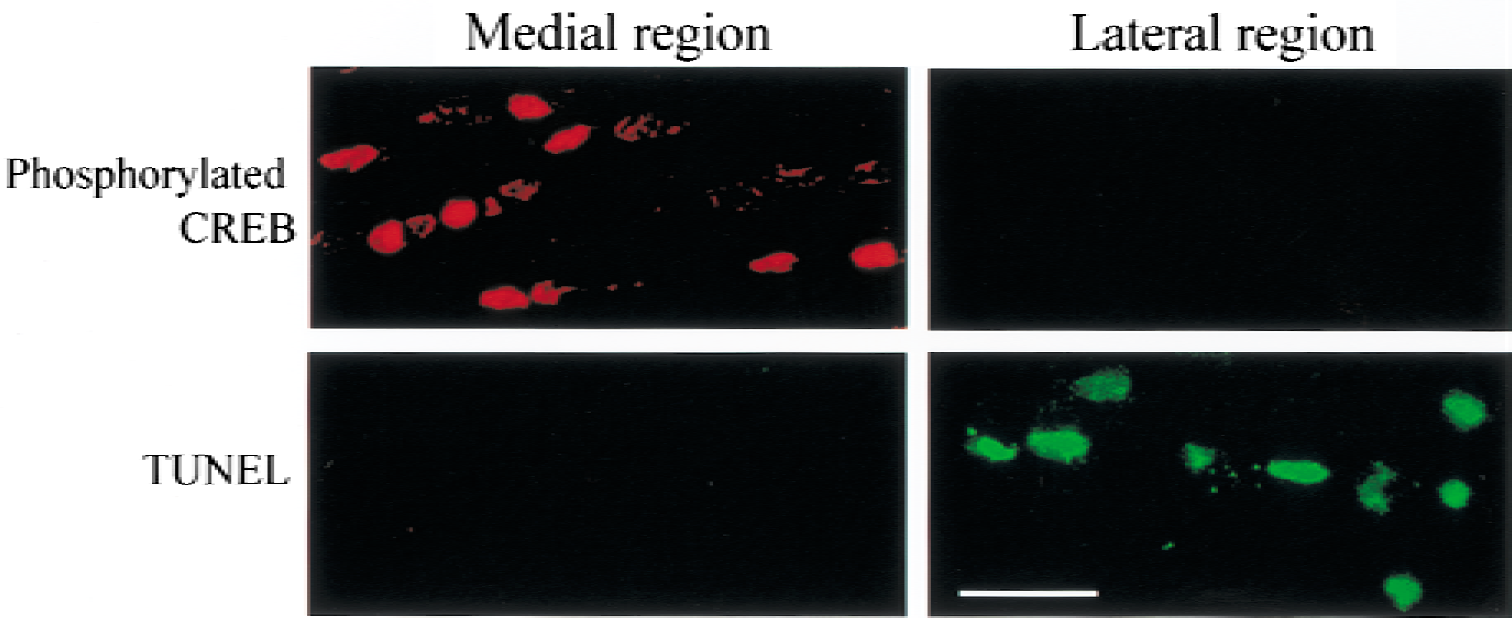
Representative photomicrographs of double-staining with TUNEL and anti-phosphorylated cyclic adenosine monophosphate response element binding protein (CREB) antibody in the medial and lateral regions of the corpus callosum on the ischemic side after 48 hours of recirculation after 90-minute middle cerebral artery occlusion. Scale bar = 20 μm.
Figure 9 summarizes the number of phosphorylated CREB-positive cells in the corpus callosum at different time points throughout the experiment. At 3.5 hours of recirculation, a marked increase in immunoreactive cells was found in the medial region on the ischemic side, whereas the other regions showed a moderate increase. The number of immunopositive cells was significantly greater on the ischemic side than on the nonischemic side in each region, but the increase in the number of immunopositive cells was followed by a gradual decline during the subsequent recirculation period. At 12 hours of recirculation, the lateral region on the ischemic side no longer showed a significant increase in the number of immunoreactive cells as compared with the sham group. At 24 hours of recirculation, the number of immunopositive cells in the lateral region on the ischemic side had decreased to almost the sham level, whereas the medial regions on both sides, especially on the ischemic side, still maintained a significant increase in the number of immunopositive cells. Such a significant increase in the number of immunoreactive cells in the medial region on the ischemic side persisted until 2 weeks of recirculation.
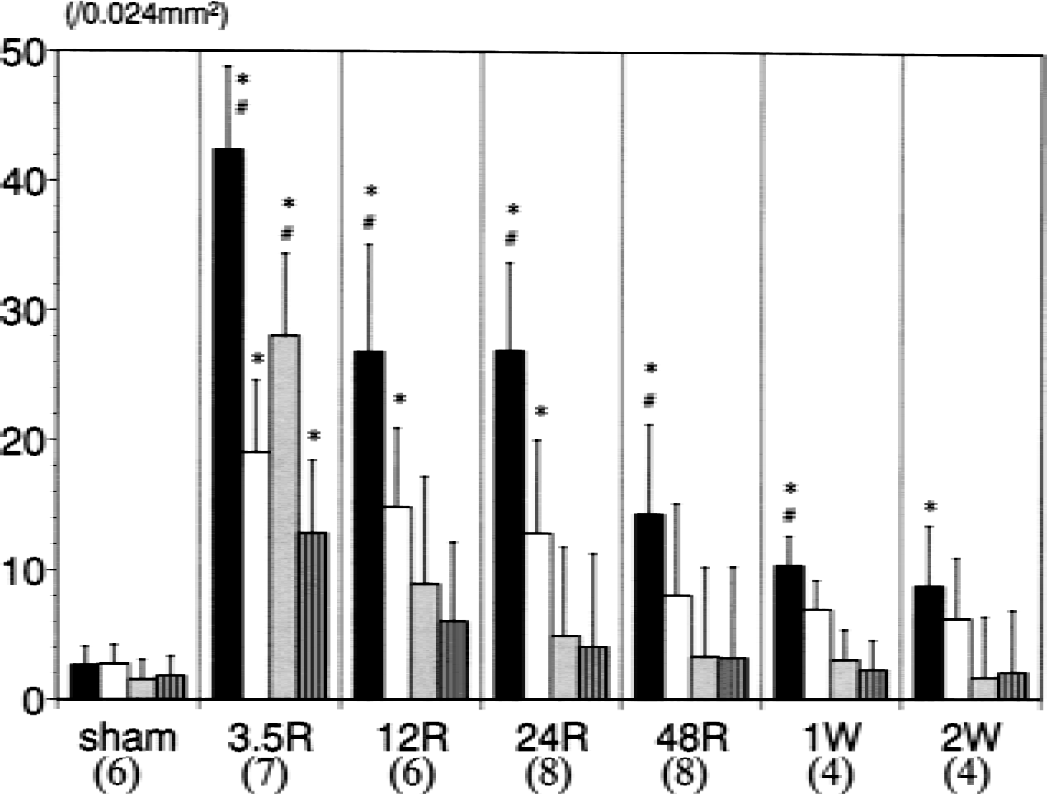
Numbers of cells immunopositive for staining with anti-phosphorylated cyclic adenosine monophosphate response element binding protein antibody at each time point. The solid, open, shaded, and striped bars represent the right medial region, left medial region, right lateral region, and left lateral region, respectively, of the corpus callosum. The labels 3.5R, 12R, 24R, 48R, 1W, and 2W denote 3.5, 12, 24, and 48 hours, 1, and 2 weeks, respectively, of recirculation after 90-minute middle cerebral artery occlusion. The number of animals analyzed for each group is shown in parentheses below the figure. * P < 0.05 compared with the sham group. #P < 0.05 compared with the left side.
Figure 10 shows a representative macroscopic photograph of immunohistochemical staining with anti-MBP antibody at 2 weeks of recirculation. The staining intensity was clearly decreased in the cerebral hemisphere on the ischemic side, including the lateral region of the corpus callosum, whereas the medial region showed normal staining intensity, suggesting that myelination was maintained in this region. Similar findings also were obtained with anti-MAG antibody.
Representative macroscopic photograph showing immunoreactivity with anti-myelin basic protein antibody obtained from an animal exposed to 2 weeks of recirculation after 90-minute middle cerebral artery occlusion. Viewer's right is the occluded side of the brain.
Figure 11 shows representative photomicrographs with higher magnification showing phosphorylated CREB-positive cells and expression of myelin basic protein in the medial and lateral regions of the corpus callosum on the ischemic side after 2 weeks of recirculation. As compared with the lateral region, the medial region demonstrated a persistent phosphorylation of CREB and preservation of myelin basic protein, indicating that CREB phosphorylation is closely associated with maintenance of myelination after ischemia.
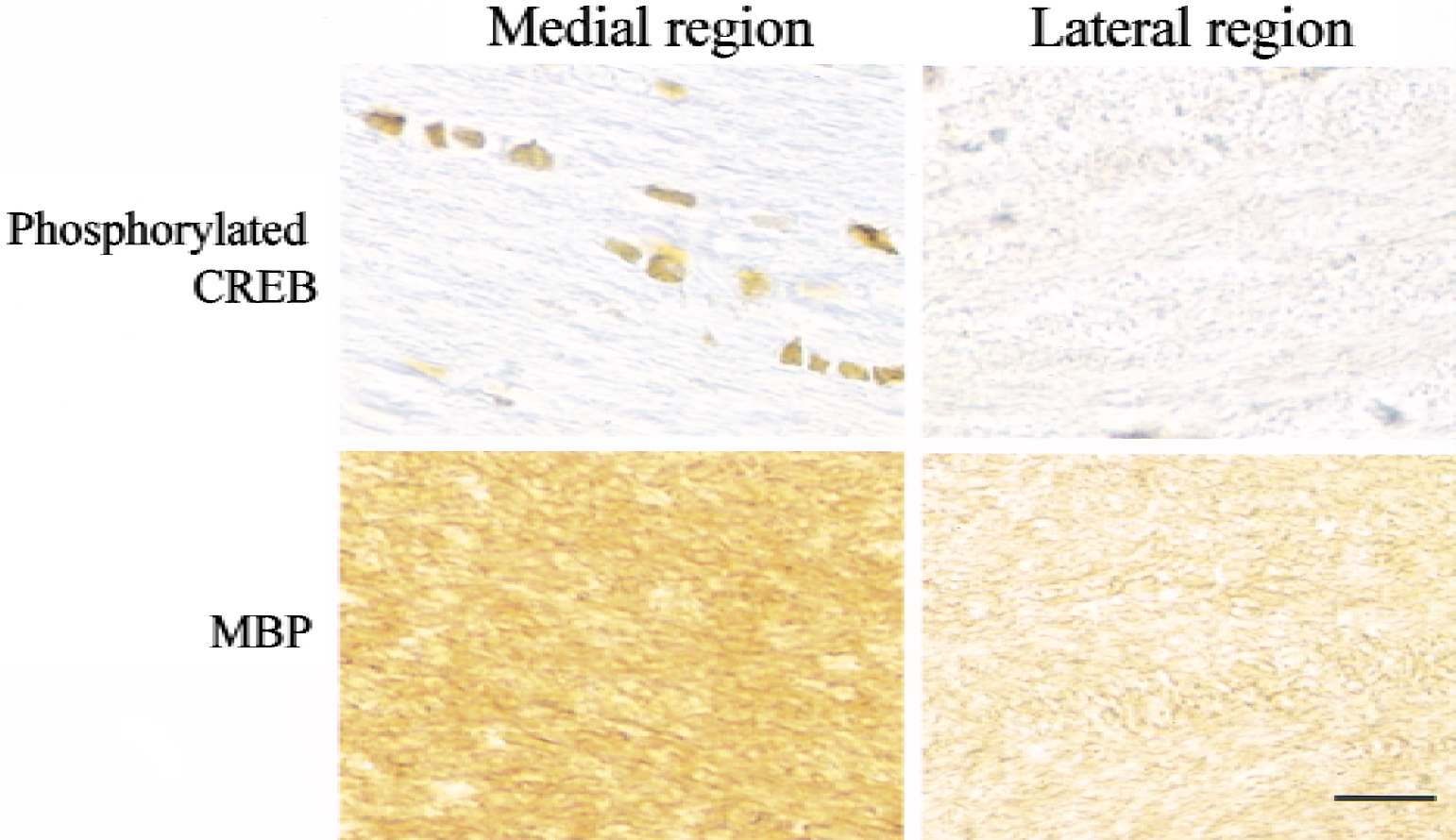
Representative photomicrographs obtained with anti-phosphorylated cyclic adenosine monophosphate response element binding protein (CREB) antibody and anti-myelin basic protein (MBP) antibody in the medial and lateral regions of the corpus callosum on the ischemic side after 2 weeks of recirculation. Scale bar = 20 μm.
DISCUSSION
The major finding in the current study is that phosphorylation of CREB was significantly and continuously activated in OLGs in the medial region of the corpus callosum after temporary occlusion of MCA, where normal morphology of OLGs, intact myelination, and expression of bcl-2 were maintained and TUNEL staining was negative. By contrast, the lateral region of the corpus callosum revealed only transient activation of CREB phosphorylation and was followed by the disappearance of bcl-2 expression accompanied by significant demyelination and positive TUNEL staining. These findings suggest that persistent CREB phosphorylation is closely associated with OLG survival and maintenance of myelination in the corpus callosum during postischemic recirculation. Because damage to myelinated fibers in the white matter, such as leukoaraiosis, is now considered to be one of the major causes of various pathologic signs and symptoms in ischemic stroke (Leys et al., 1999), the survival of OLGs and maintenance of myelination in the corpus callosum in the current study seem to be important findings. The finding of activated CREB phosphorylation in OLGs in this study is consistent with a recent report that briefly describes an increase in phosphorylated CREB-positive OLGs in the subcortical white matter after cerebral ischemia (Irving et al., 2000). Double-immunostaining with anti-APC antibody and anti-phosphorylated CREB antibody confirmed that CREB phosphorylation in the corpus callosum took place exclusively in the nuclei of OLGs. The APC tumor suppressor gene is localized to the cell body of OLGs (Bhat et al., 1996), and the antibodies against APC protein have been used as a useful OLG marker (Bhat et al., 1996; Liu et al., 2001).
The medial region of the corpus callosum was exposed to a moderate (58.4%) reduction in CBF, whereas the lateral region was subjected to a severe (91.9%) decrease in CBF. Cerebral blood flow in both regions was restored to the control level after the release of the MCAO, suggesting that the level of CBF during the 90-minute ischemic period may have largely determined the subsequent metabolic consequences and the fate of the OLGs. If CBF is reduced by less than 60% of the control value in the current ischemia model, OLGs may be able to sustain their normal morphology and intact myelination with enhanced CREB phosphorylation. In fact, the animals with grade 1 neurologic deficits during MCAO showed persistent CREB phosphorylation even in the lateral region of the corpus callosum, without any definite ischemic damage to OLGs.
This study revealed that OLGs in the corpus callosum of mature rats constitutively express CREB protein under normal conditions, but that it is barely phosphorylated. Once OLGs were exposed to temporary ischemia followed by recirculation, however, the CREB in the OLGs was rapidly phosphorylated. Signaling cascades converging on CREB phosphoryation include PKA, MAP kinases, calcium/calmodulin-dependent protein kinases (CaMKs), PKC, and AKT/PKB (Andrisani, 1999; De Cesare et al., 1999; Du and Montminy, 1998). Protein kinase A has been found to up-regulate CREB phosphorylation in OLGs at all developmental stages, whereas PKC and MAPKs have been shown to lose their capacity for CREB phosphorylation in mature OLGs (Sato-Bigbee et al., 1999; Wegner, 2000), suggesting that signaling pathways mediated through PKA, not PKC nor MAPKs, may have been involved in the activation of CREB phosphorylation in the current study.
CREB phosphorylation has been studied extensively in neurons and found to play an important role in neuronal development, synaptic plasticity, memory function, regeneration, and survival in response to various stresses (Andrisani, 1999; Finkbeiner et al., 1997; Finkbeiner, 2000; Impey et al., 1999; Tao et al., 1998; Walton and Dragunow, 2000). CREB mediates neuronal responses to various neurotrophins, including brain-derived neurotrophic factor (BDNF) and nerve growth factor, which regulate neuronal survival, differentiation, and synaptic function (Finkbeiner et al., 1997). Conversely, CREB itself controls BDNF transcription (Tao et al., 1998). Riccio et al. (1999) revealed that nerve growth factor–dependent neuronal survival is dependent on the expression of the antiapoptotic protein bcl-2, and that bcl-2 expression is mediated by phosphorylation of CREB. CREB also has been implicated in the mediation of BDNF-dependent neuronal survival through expression of bcl-2 (Bonni et al., 1999). In contrast, the role of CREB phosphorylation in OLGs has not been widely investigated, although CREB has been implicated as an important mediator of myelination as well as of differentiation and cell process outgrowth in OLGs (Sato-Bigbee and DeVries, 1996; Wegner, 2000). In the current study, persistent CREB phosphorylation was closely associated with expression of bcl-2 and normally maintained myelination, suggesting that CREB phosphorylation may play a crucial role in the survival and normal function of OLGs in response to ischemic stress. Although proliferation of OLG precursor cells is stimulated by neurotrophin-3 through CREB phosphorylation (Johnson et al., 2000), the effects of neurotrophic factors on mature OLGs remains unknown. Expression of BDNF has been shown to be up-regulated in the cerebral regions outside the damaged area after focal ischemia (Kokaia et al., 1998). Accumulation of BDNF immunoreactivity also has been observed in varicose and nonvaricose fibers in the corpus callosum ipsilateral to the MCAO (Kokaia et al., 1998). A high level of the mRNA for the high-affinity BDNF receptor, TrkB, has been found in cultured OLGs (Condorelli et al., 1995). Accordingly, BDNF may be one of the factors responsible for CREB phosphorylation in OLGs and their survival in the current ischemia model.
Irving et al. (1997) have reported that OLGs respond rapidly to an ischemic challenge by increasing immunoreactivity for protein tau, which can be induced by free radical-mediated mechanisms. The same mechanisms are considered to operate in the marked toxic effects of glutamate on OLGs (Oka et al., 1993). In line with these findings, OLGs appear to be vulnerable to a severe ischemic insult, as shown in the lateral region of the corpus callosum in the current study. Expression of bcl-2 in OLGs is associated with rescue from tumor necrosis factor-α–induced death of OLGs (Burgmaier et al., 2000) and prevents apoptosis by inhibiting caspase-3 activation (Soane et al., 1999). Moreover, Shibata et al. (2000) also have reported that caspases are involved in the ischemia-induced cell death of OLGs. CREB phosphorylation in OLGs may be protective against ischemic injury by up-regulating bcl-2 expression, although other target genes of CREB also may have contributed to the survival of the OLGs in the current study. A unilateral hypoxia-ischemia model in the immature rat brain showed an induction of immediate early gene proteins including c-Fos and JunB in the OLGs (Dragunow et al., 1994). The promotors of c-Fos and JunB contain the CRE motif, suggesting that CREB phosphorylation may be responsible for expression of these proteins. The signaling cascade downstream of CREB phosphorylation in OLGs in the white matter in cerebral ischemia should be investigated in a future study, because the results may identify a target for stroke therapy.
Footnotes
Acknowledgments:
The authors are grateful to Dr. Hiroaki Asou and Jin Nakahara for their valuable suggestions.
