Abstract
For much of the twentieth century migraine and cluster headache have been considered as vascular headaches whose pathophysiology was determined by changes in cranial vascular diameter. To examine nociceptive neural influences on the cranial circulation, the authors studied healthy volunteers' responses to injection of the pain-producing compound capsaicin in terms of the caliber of the internal carotid artery. The study was conducted using magnetic resonance angiographic techniques. Injection of capsaicin into the skin innervated by the ophthalmic (first) division of the trigeminal nerve elicited 40% ± 27% (mean ± SD) increase in vascular cross-sectional area in the right (ipsilateral) internal carotid artery when compared with the mean baseline (P < 0.001). Injection of capsaicin into the skin of the chin to stimulate the mandibular (third) division of the trigeminal nerve and into the leg led to a similar pain perception and failed to produce any significant change in vessel caliber. The data suggest that there is a highly functionally organized, somatotopically congruent trigeminal innervation of the cranial vessels, with a potent vasodilator effect of the ophthalmic division on the large intracranial vessels. The data are consistent with the notion that pain drives changes in vessel caliber in migraine and cluster headache, not vice versa. These conditions therefore should be regarded as primary neurovascular headaches not as vascular headaches.
For many years there has been argument over whether there is a functional innervation of the cranial vessels, particularly the cerebral vessels (Heistad and Marcus, 1978; Purves, 1978). Cerebral blood flow is clearly influenced by local neuronal activity, vasoneuronal coupling, which is at the heart of functional neuroimaging using blood flow surrogates (Frackowiak and Friston, 1994). In recent years the propensity for the neural innervation of the cerebral circulation to influence blood flow has become of interest as the trigeminovascular system has emerged as pivotal in understanding primary headaches, such as migraine and cluster headache (Goadsby, 2001).
The trigemino-cerebrovascular system consists of trigeminal neurons, whose cell bodies are located in the trigeminal ganglion, that innervate the cerebral vessels (Uddman et al., 1985). When activated this system causes vasodilatation in experimental animals (Goadsby and Duckworth, 1987). In terms of headache pathophysiology, these vessels are important because the large extra-parenchymal vessels are pain producing in humans (Feindel et al., 1960; McNaughton, 1938; Penfield and McNaughton, 1940). In comparison, brain substance is largely not pain producing (Wolff, 1963), with the exception of perturbations of the rostral brainstem (Raskin et al., 1987). The trigeminal innervation is unique in the cerebral circulation in providing sensory input to the brain covering, dura mater, and for its vascular supply. The trigeminal fibers pivotal to the cerebrovascular system are predominantly found in the first (ophthalmic) division of the trigeminal nerve (Cushing, 1904). They have widely ramifying axons that may innervate several vessels ipsilaterally (Mayberg et al., 1984), involving both cerebral (middle cerebral artery) and extracerebral (middle meningeal artery) vessels (O'Connor and van der Kooy, 1986). The trigeminovascular system has been demonstrated in experimental animals to have a role in settings such as severe hypertension or seizures (Sakas et al., 1989).
Clinical conditions, such as cluster headache and migraine, are widely described as vascular headaches, even though from a pathophysiologic viewpoint the concept of a vascular headache implies primary abnormalities in vessel behavior. For both conditions there is considerable clinical evidence to suggest that they are primarily driven from the brain, and are thus more correctly regarded as neurovascular headaches (May and Goadsby, 1999). We have observed dilatation of the intracranial arteries in nitroglycerin-induced cluster headache (May et al., 1998a), spontaneous cluster headache (May et al., 2000), and migraine (Bahra et al., 2001), which involve the internal carotid artery (May et al., 1999b). Using magnetic resonance angiography (MRA) in experimentally induced pain, the authors aimed to determine whether vasodilation of the internal carotid arteries is specific to certain headache syndromes, or simply secondary to trigeminal activation. The authors also were interested in whether trigeminal transmitted pain and peripheral (somatic) pain have different effects on the vascular behavior of the internal carotid artery. The chemogenic algesic agent capsaicin is the pungent ingredient of red peppers, which selectively acts on C-fibers. The authors chose it for a rapid short-lasting pain sensation, while avoiding possible tactile components of the pain stimulus. Craniovascular pain is transmitted predominantly through the ophthalmic division of the trigeminal nerve, the other divisions being involved to a lesser extent (McNaughton, 1966). In accordance with the authors' previous work using the same experimental pain stimulus in functional imaging, a small amount of capsaicin was administered subcutaneously to the forehead to evoke a painful sensation in the first division of the trigeminal nerve (May et al., 1998b). As a control, the same amount of capsaicin was injected subcutaneously into the third division of the trigeminal nerve (lateral chin) and lower limb in the same subjects.
MATERIALS AND METHODS
Subjects
Nine healthy volunteer subjects (men, aged 25 to 37 years, mean = 31 years) were studied during an acute pain state evoked by injecting capsaicin subcutaneously into 3 different locations on 3 different days. The participants were all right-handed and were free from cerebrovascular disorders. The Ethics Committee of the National Hospital for Neurology and Neurosurgery approved the study. All participants gave informed consent.
Design
All volunteers were scanned three times before the injection. In 6 volunteers, 0.05 mL of a sterile 0.1% solution of capsaicin was injected subcutaneously on the right side of the following:
the forehead, 2 cm above the middle of the eyebrow;
the lateral chin, 1 cm inferior to and 2 cm lateral to the angle of the mouth; and
the lateral calf, 10 cm below the fibula and 3 cm lateral from the tibia.
The remaining three volunteers received a capsaicin injection into the forehead only. The injections were performed on three different days and were given in a random order. Pain was described as being analogous to a bee-sting. It reached the maximum intensity immediately after injection in all volunteers on each occasion. During the acute pain state, three MR angiograms were performed.
Magnetic resonance angiography
An electrocardiogram-gated three-dimensional multislab MRI inflow angiogram (MRA) with a repetition time (TR) of 39 milliseconds, 32 mm slab thickness, an effective slice thickness of 1 mm, and a flip angle of 25° was performed (Potchen et al., 1993), using a Siemens MAGNETOM Vision scanner operating at 2T. In the six volunteers receiving all three injections (that is, forehead, chin, and leg), a pilot scan was performed for each patient and each session, and the transverse section chosen each time was at a level just above the internal carotid syphon. In addition, in three volunteers receiving the injection into the forehead, the MRA was performed using the transverse section through the extracranial part of the internal artery just distal to the carotid bifurcation. In all subjects, three consecutive MR-angiograms were performed at rest, and again three during the acute pain state in each session. The entire three-dimensional data set was processed using maximum intensity projections. Using the integrated software (Siemens calculation tool) regions of interest were drawn around the vessels identified as the internal or external carotid arteries on either side.
Data analysis
The absolute value for vessel cross-sectional area (cm2), bulk blood flow (cm3 /s), and mean and peak velocity (cm/sec) were available. The changes in cross-sectional area in the vessels between rest and pain are expressed as a mean percentage change ± one standard deviation. The primary endpoint was vessel cross-sectional area, because this is directly measured by MRA, and the authors wanted to determine if there were caliber changes, based on observations of primary headache syndromes (Bahra et al., 2001; May et al., 1999a, 1999b). The authors compared the cross-sectional area of the carotid artery from the immediate preinjection control period to the effect of capsaicin across all sites using a repeated measures analysis of variance (SPSS version 9). Each volunteer contributed three repetitions at each of three sites and two treatment periods per site (control and after capsaicin). The site by treatment interaction then was compared post hoc using a paired t-test. The authors assessed the outcome at P < 0.05 with Bonferroni correction.
RESULTS
Intracranial part of the internal carotid artery (n = 6)
The internal carotid artery was identified in the neck using anatomic markers (Fig. 1). Resting internal carotid cross-sectional area was 0.20 ± 0.03 cm2 on the day of the forehead study, 0.23 ± 0.02 cm2 on the day on the chin study, and 0.25 ± 0.04 cm2 on the day of the leg study. The overall analysis was performed for the cross-sectional area data as the directly measured primary end point. There was no significant effect of the repetitions (F1,5 = 1.6, P = 0.27), indicating a stable baseline for the measurements. The treatment effect was significant (F1,5 = 14.7, P = 0.012), indicating there was an effect of the capsaicin, while the treatment by site interaction also was significant (F1,5 = 7.3, P = 0.043), indicating that one or more sites were impacting on the outcome. The individual sites were then tested.
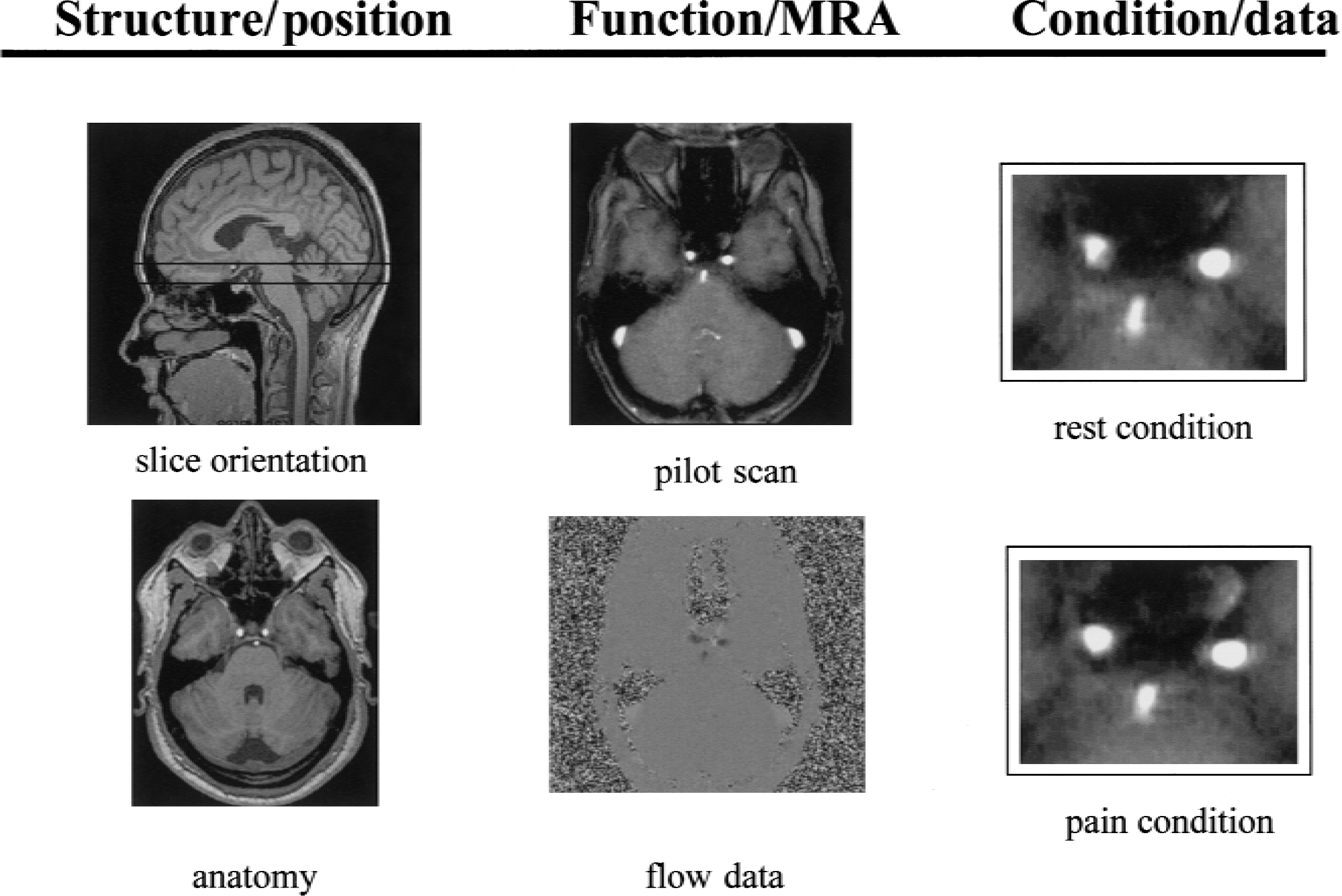
Illustration of the slice orientation for the study and the relevant local anatomy of the carotid artery (anatomy). An example of a pilot scan and the flow data are presented. The effect of capsaicin is illustrated in the final panel contrasting the rest and pain states. MRA, magnetic resonance angiography.
Experimental pain: forehead.
Capsaicin injection into the right forehead resulted in a significant mean 40% ± 27% increase in vascular cross-sectional area in the right (ipsilateral) internal carotid artery when compared with the mean baseline (P = 0.012). Similarly, a 15% ± 8% in the left (contralateral) internal carotid artery cross-sectional area was observed (P = 0.04;Fig. 2).
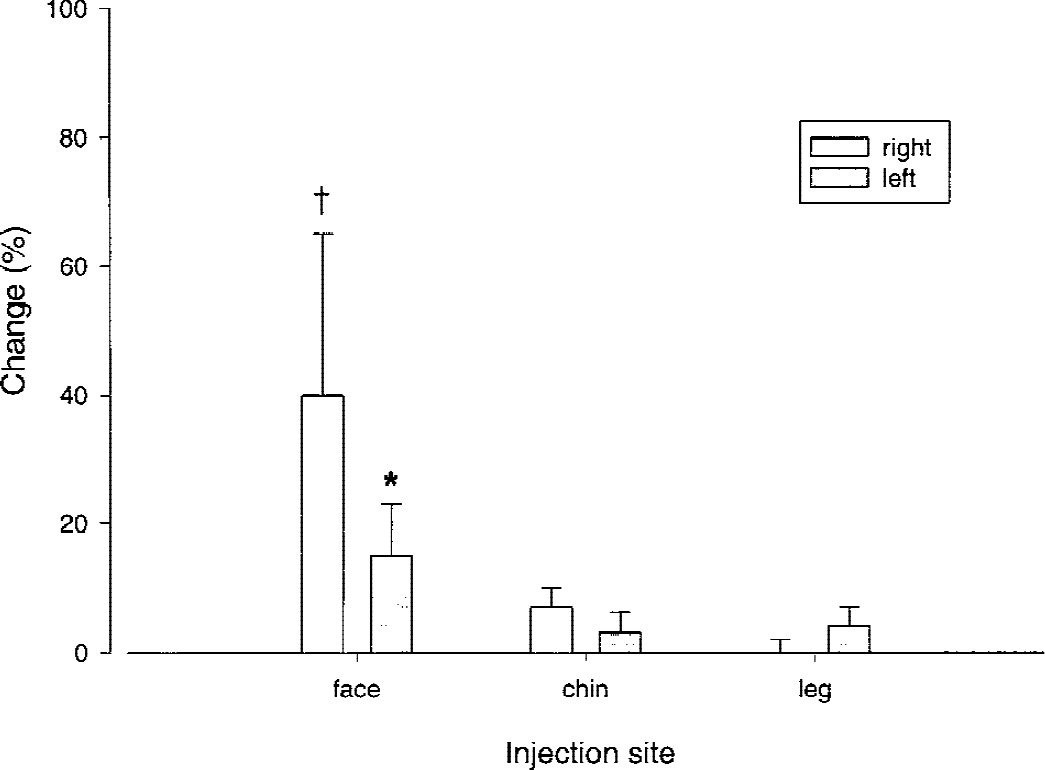
Bar graph of percentage change (ordinate) in internal carotid cross-sectional area (CSA) after painful stimulation of various body areas with subcutaneous capsaicin. Injections were made on the right side of the body in the distribution of the first (face) and third (chin) division of the trigeminal nerve and into the skin of the right calf. There was significant increase in internal carotid CSA ipsilateral (†P = 0.012) and contralateral (* P = 0.04) to the forehead (ophthalmic division of trigeminal), but there was no increase in carotid CSA on the contralateral side or when injections were made in the chin or leg.
Experimental pain: lateral chin.
Capsaicin injection into the lateral chin resulted in a 7% ± 4% increase (change from mean) in vascular cross-sectional area in the right (ipsilateral) internal carotid artery (P = 0.52) and 3% ± 3% in the left (contralateral) internal carotid artery.
Experimental pain: lateral calf.
Capsaicin injection into the lateral calf prompted no change in vascular cross-sectional area in the right (ipsilateral) internal carotid artery (−0.1% ± 0.9%;P = 0.63) and a 4% ± 3% increase (change from mean) in vascular cross-sectional area in the left (contralateral) internal carotid artery.
Extracranial part of the internal carotid artery (n = 3)
Experimental pain: forehead.
Capsaicin injection into the forehead resulted in a 5% increase (change from mean) in vascular cross-sectional area in the right (ipsilateral) internal carotid artery and 2% in the left (contralateral) internal carotid artery, when measuring the extracranial part of the internal carotid artery.
DISCUSSION
This study demonstrates an increase in internal carotid cross-sectional area with painful cutaneous stimulation of skin innervated by the ophthalmic (first) division of the trigeminal nerve in the absence of any active primary headache syndrome. In the same volunteers no changes were observed in internal carotid cross-sectional area after painful stimulation of the skin of the mandibular (third) division, or painful stimulation of the leg. These data demonstrate a somatotopically specific, reflex neurogenic dilatation of the internal carotid artery in response to pain. The data provide further physiologic evidence for an innervation of the human cerebral vessels that is functionally and anatomically specific. Moreover, the anatomy and physiology of this response (May and Goadsby, 1999) underpins the clinical phenotype of the primary neurovascular headaches, such as migraine and cluster headache.
It is a challenge to conduct human cerebrovascular studies. Magnetic resonance angiography offers the opportunity to do repeated studies in the same volunteer. Magnetic resonance angiography is suited to study changes in vessel anatomy, such as dilatation, whereas Doppler measurements are ideal for measurement of blood velocity (Thomsen et al., 1995). The primary end point of the study, change in cross-sectional area of the internal carotid artery, was selected to provide detailed information about vascular diameter, having observed dilatation of the internal carotid in experimental headache (May et al., 1998b), cluster headache (May et al., 1999b), and migraine (Bahra et al., 2001). Volunteers were randomized in the order of the site that was injected, because on any one day it could not be guaranteed that the effect of the capsaicin had worn off to provide an adequate baseline. Observations on bulk flow can be calculated from MRA but have limited value. First, the carotid is a large conducting vessel whose effects may be negated by downstream arteriolar change. Second, it is known from positron emission tomography (PET) studies of facial (May et al., 1998b) and other pains (Derbyshire and Jones, 1998) that cerebral blood changes are different in distinct brain regions. However, the authors' primary end point of changes in cross-sectional area of the internal carotid artery is reliably and appropriately measured by MRA. The authors' inferences are limited to this local dilatation, but what seems clear is that this dilatation is completely independent of headache type and represents a true neurogenic vasodilation.
The trigeminal innervation of the cerebral circulation is provided by branches of the first (ophthalmic) division of the nerve (Mayberg et al., 1981, 1984). This innervation is well documented in humans (Cushing, 1904; Feindel et al., 1960; McNaughton, 1938, 1966) and has been implicated in headache either in terms of the vessels (Willis, 1664) or dura mater (Penfield and McNaughton, 1940) for many years. It has been shown in humans (Tran-Dinh et al., 1992) and experimental animals (Goadsby and Duckworth, 1987; Lang and Zimmer, 1974) that stimulation of the trigeminal ganglion increases brain blood flow. Stimulation of pain-producing structures, such as the superior sagittal sinus in the cat, produces greater increases in brain blood flow than does trigeminal ganglion stimulation (Lambert et al., 1988). Similarly, using the combination laser–Doppler flowmetry applied to the cat cortex and direct measurements of common carotid blood flow, it can be shown that stimulation of the superior sagittal sinus elicits a much more brain-selective change in flow than does trigeminal ganglion stimulation (Goadsby et al., 1996). In the current study, the authors show that injection of the pain-producing substance capsaicin into skin innervated by the first (ophthalmic) division of the trigeminal nerve in humans elicits a selective dilatation of the internal carotid artery that involves its intracranial but not its extracranial portion. Furthermore, third division trigeminal injection has no such effect. It is likely that the dilatation observed relates to the fact that the ophthalmic division provides the largest part of the innervation of the intracranial circulation (Mayberg et al., 1981; O'Connor and van der Kooy, 1986), certainly above the tentorium cerebelli. The data provide physiologic evidence for a collateral innervation of the forehead and the large intracranial vessels by the same branches of the ophthalmic branch of the trigeminal nerve.
In humans, thermocoagulation (Onofrio, 1975; Sweet and Wepsic, 1974) or injection of alcohol (Oka, 1950) into the trigeminal ganglion (VG) is accompanied by flushing of the skin in the distribution of the appropriate division or divisions of the nerve. Consistent with this flush, increases in skin temperature and capillary pulsation have been observed after thermocoagulation of the VG in humans for tic douloureux (Drummond et al., 1983). Corresponding with this flush, substance P and calcitonin gene-related peptide (CGRP) levels are increased in the ipsilateral external jugular vein of both humans and cats (Goadsby et al., 1988). Perhaps the best evidence for activation of the trigeminovascular system in headache is the cranial release of CGRP in both acute migraine attacks (Gallai et al., 1995; Goadsby et al., 1990), between migraine attacks (Ashina et al., 2000b), and in attacks of cluster headache (Fanciullacci et al., 1995; Goadsby and Edvinsson, 1994). These data suggest a pathophysiologic class of headache that could be described as primary neurovascular headaches.
Indeed, the recent exciting report that some patients with phenotypic chronic tension-type headache, whose pain is throbbing, have elevated CGRP levels (Ashina et al., 2000a) suggests a marker with physiologic specificity exceeding the current clinical classification system (Headache Classification Committee of The International Headache Society, 1988). In this context, the current results support the broad significance of the ophthalmic (first) division of trigeminal innervation in terms of the cerebral circulation. It is key that carotid dilatation is not simply caused by pain, or it would be seen with equally painful stimuli on the chin or the leg. Rather the anatomic site of pain is pivotal, indeed it determines the nature of the physiologic response. Furthermore, the dilator response is not a privilege of primary neurovascular headaches, because the volunteers had no headache syndrome. Taken together the data suggest that neurovascular activation in the trigeminal system is a function of its afferent role in any pain and is highly potent and somatotopically organized. Pain signals in the ophthalmic division can generate vascular change de novo without a superimposed primary headache.
In summary, painful stimulation of skin innervated by the ophthalmic (first) division of the trigeminal nerve, and not painful stimulation of skin in the mandibular (third) division or skin of the leg, can elicit dilatation of the internal carotid artery. These data are consistent with observations in experimental animals of a highly functionally organized somatotopically congruent arrangement of the trigeminovascular system. The data are consistent with the notion that vascular change in primary headaches is likely to be on the basis of neural activation, as neurovascular headache syndromes. Understanding the physiology of the neural innervation of the cranial circulation, particularly understanding its pharmacology, will facilitate neurally based strategies for the management of primary headache syndromes, such as migraine and cluster headache.
