Abstract
Factors regulating cerebral tissue P
An understanding of the factors that influence cerebral tissue P
Chronic acclimation to hypoxia has been shown to increase microvascular density in brain (Boero et al., 1998; Diemer and Henn, 1965; Harik et al., 1995, 1996; LaManna et al., 1992, 1994; Miller and Hale, 1970; Opitz, 1951). Oxygen carrying capacity also increases, whereas the oxyhemoglobin dissociation curve in rats may or may not exhibit a right shift with altitude (Baumann et al., 1971; LaManna et al., 1992; Monge and Leon-Velarde, 1991; Turek et al., 1972). In addition, the sensitivity of cerebral blood flow to CO2 is thought to increase (Severinghaus et al., 1966). These changes should result in increases in cerebral PtO2, unless there are homeostatic mechanisms that operate to maintain a relatively static level of PtO2 in awake animals.
The authors chronically adapted rats to one half an atmosphere of barometric pressure for 27 days and measured PtO2 when animals were breathing normobaric gases (nonanesthetized and spontaneously ventilating).
MATERIALS AND METHODS
Crystals of lithium phthalocyanine (LiPc) were used as the oxygen sensitive material (Rolett et al., 2000). The LiPc crystals were individually calibrated (Fig. 1). These data show that the calibration was linear over at least 0 to 156 mm Hg. Crystals of approximately 50 × 500 μm (0.05 mg) were implanted using a 25G needle into the frontal cortex under ketamine/xylazine anesthesia. Electron paramagnetic resonance oximetry was performed using a low frequency (1.18 GHz, L-band) spectrometer. For measurements, awake animals were placed in a plexiglas restraining tube, and the head was positioned below the EPR surface-loop resonator. The P
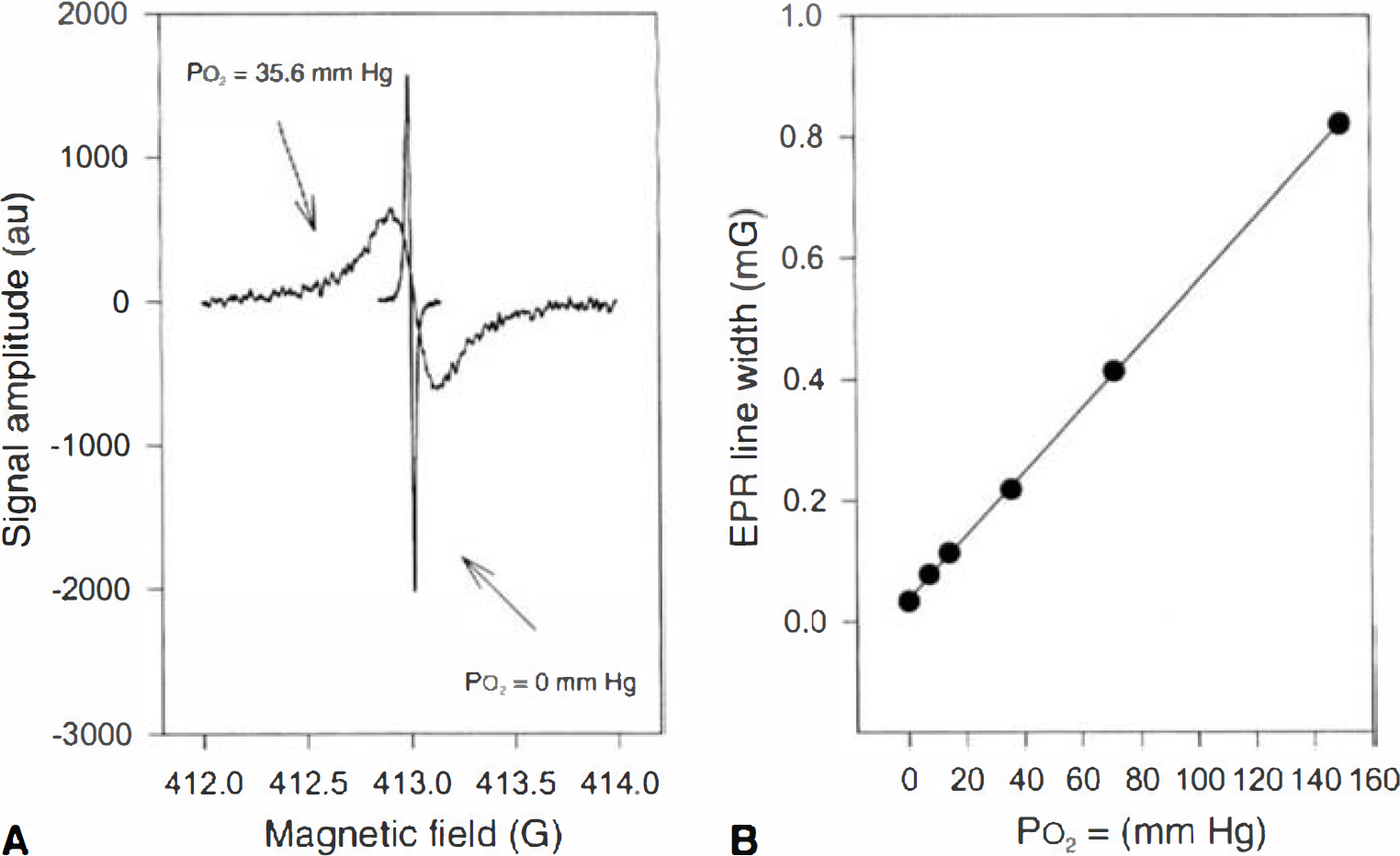
Calibration curve of lithium phthalocyanine (LiPc) for P
Acclimation was performed in 40-gallon drums fitted to a vacuum line to reduce pressure to 370 ± 2.1 mm Hg (range 367 to 372, mean ± SD), corresponding to an altitude of approximately 6000 m. Control subjects were placed in a drum at normobaric pressures. Ambient temperatures varied from 22.2°C to 24.4°C. Animals were housed to a maximum of 6 per drum.
The LiPc was implanted 7 days before acclimation. Each rat underwent the P
Analysis of the data on the time course of PtO2 was performed using a general linearized model, followed by a Mann—Whitney U test (P < 0.05 was significant).
RESULTS
The LiPc was in close proximity to the surrounding tissue with no evidence of edema, red blood cell accumulation (vascular damage), or lymphocyte infiltration (Fig. 2). This was consistent with a previous report on the histologic appearance of LiPc implants in cortical tissue (Rolett et al., 2000).
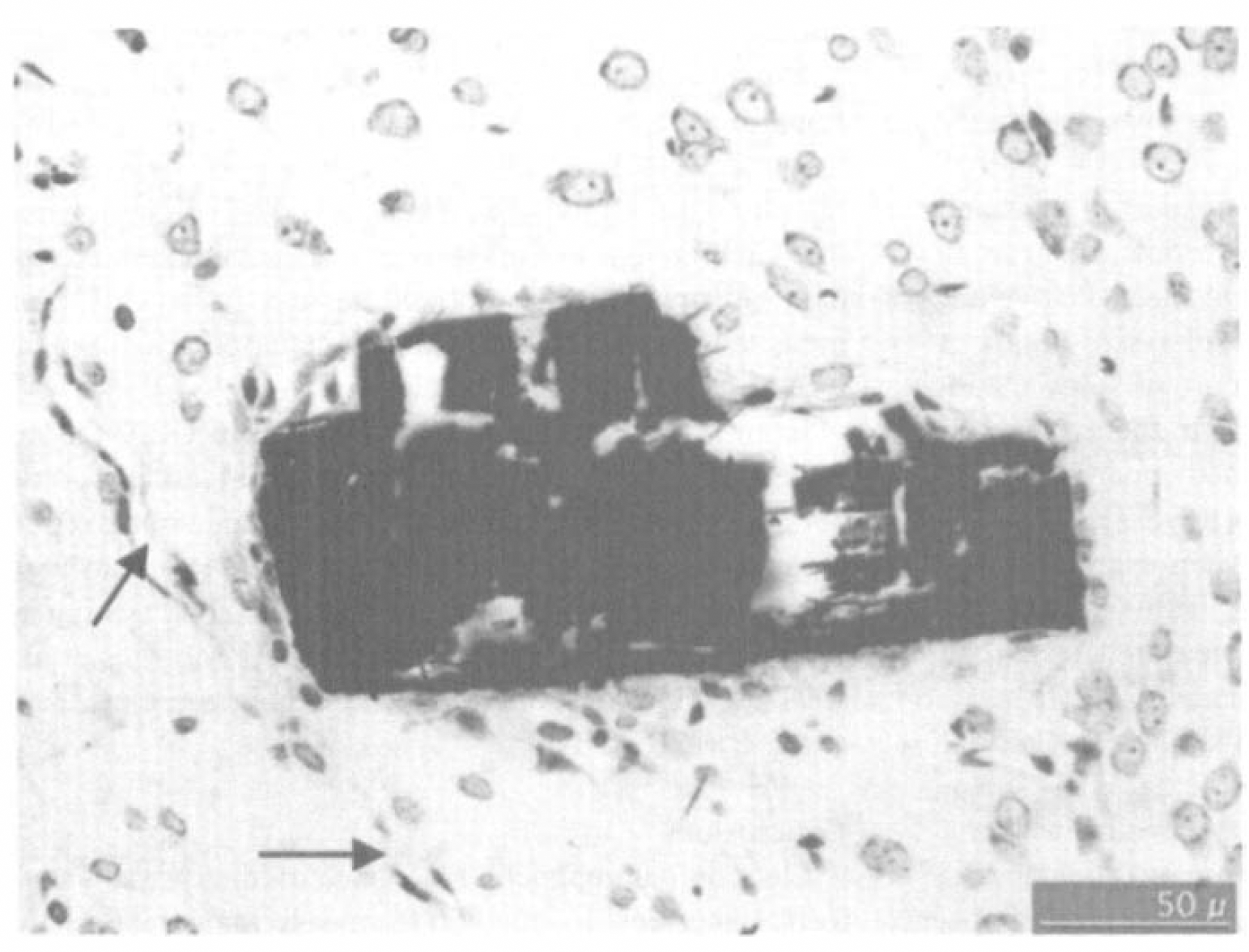
Histologic appearance of the electron paramagnetic resonance (EPR) oximetry material (lithium phthalocyanine [LiPC]) in the rat cortex. Dark area is the crystal of LiPc shown in a histologic section stained with cresyl violet. Viable tissue is in close approximation to the crystal. There is no evidence of swelling, red blood cells, or local vascular damage. Arrows indicate a small blood vessel in proximity to the implant. The section was taken 7 days after implanting the material, fixed in phosphate buffered formalin, and embedded in paraffin.
Figure 3 shows the time course of changes in cerebral PtO2, measured in awake, restrained animals, at normobaric pressures. The PtO2 before acclimation was not significantly different (27.1 ± 7.5 vs. 26.0 ± 15, n = 6, mean ± SD in the acclimated and normobaric groups). The PtO2 in the acclimated versus control groups were significantly different by day 4 (the first EPR measurement during acclimation), with values of 49 ± 11 mm Hg versus 24 ± 6 mm Hg, respectively. The PtO2 in acclimated animals increased rapidly in the first 7 days, then stabilized for the duration of the study. The average PtO2 in the acclimated group (from 7 days on) was 62 mm Hg versus 26 mm Hg in the control groups, an increase of 238%.
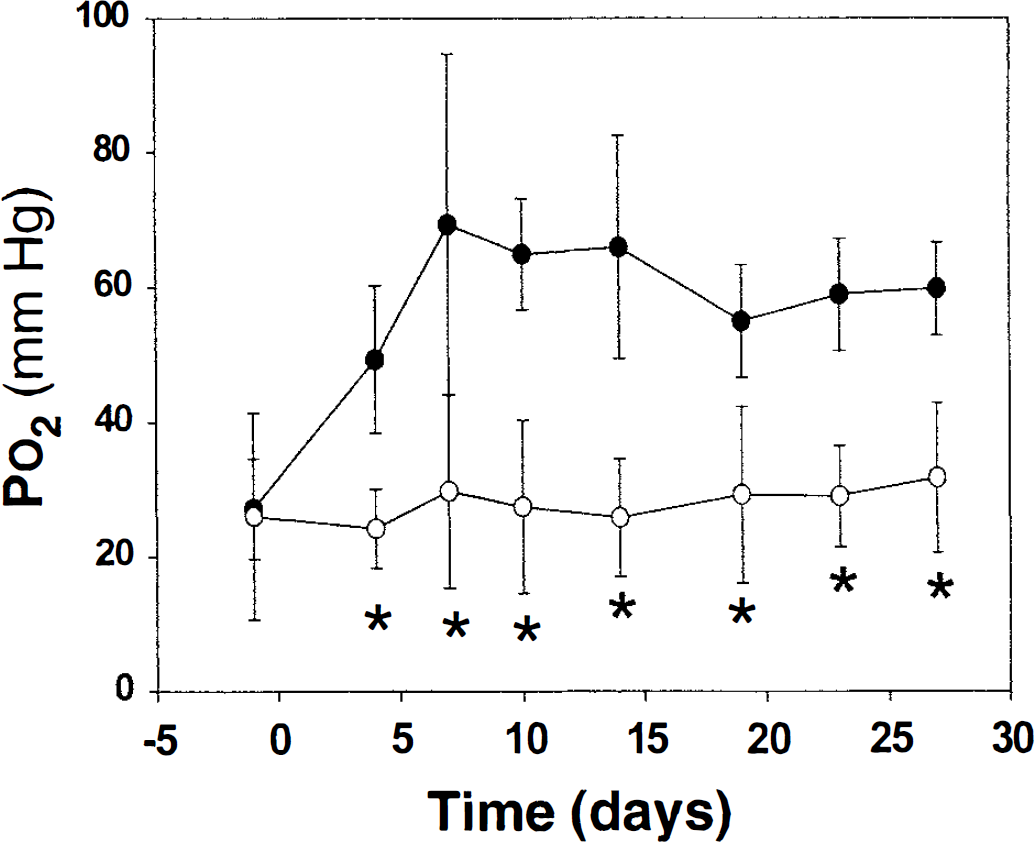
The PtO2 in brains of animals being acclimated to chronic hypoxia. Cortical PtO2 was measured when both groups were breathing normoxic FiO2 at normobaric pressure. Closed circles represent rats living at one-half atmospheric pressures. Open circles designate rats living at normobaric pressures.
DISCUSSION
Electron paramagnetic resonance oximetry is a relatively new method for monitoring cerebral PtO2 (Liu et al., 1993; Rolett et al., 2000). Although trauma can occur during implant, the damage is minimal and would be similar to that which would occur when using a micro-electrode of similar size to the needle. However, because the measurements were obtained more than 5 days after the implant, there was time for the tissue to recover (Hoopes et al., 1997; Rolett et al., 2000). This feature, and the capability to measure PtO2 in awake animals, makes EPR an attractive alternative.
Because the implant is large, with respect to cells and capillary units, the PtO2 will likely reflect an average value and may be more comparable to mean P
Studies with animal models show that there is vascular remodeling in response to chronic hypoxia. Early histologic studies show increased capillary density in neural tissue after chronic exposure to hypoxia (Diemer and Henn, 1965; Opitz, 1951). More recent studies of microvessel density confirm that acclimation increases density and segment length in studies on rats, mice, and cats (Boero et al., 1998; Cervos-Navarro et al., 1991; Harik et al., 1995; LaManna et al., 1992; Mironov et al., 1994).
Interestingly, the time course of the increase in vascularity does not match the changes in the regulation of cerebral oxygenation observed in this study. Rats acclimated to a similar pressure did not have an increased microvessel density after 4 days of chronic hypoxia (Harik et al., 1996). However, in the current study, there was a significant increase in PtO2 after 4 days and the maximum level was reached by 7 days.
Elevated PtO2 values would be expected if perfusion was higher than was required for oxygen utilization. A possible cause of an elevation in cerebral blood flow (CBF) would be a relative hypoventilation on returning to normobaric high oxygen environment, which would result in an increase in Pa
However, Lamanna et al. (1992) measured CBF and found it to be similar to the normoxic controls, and similar to or less than in rats acclimated to hypoxia and measured during hypoxia. Their flow data do not indicate that CBF is high in the brains of rats acclimated to hypoxia but breathing normobaric gasses. These CBF data indicate that rats do not show the increased sensitivity to CO2 that has been reported in humans.
Even if there was a marked increase in CBF, the reported changes in PtO2 indicate that this variable could not account for the magnitude of the change observed in the current study. In rats anesthetized with sodium pentobarbital, exposed to inspired values of P
Conclusions
Electron paramagnetic resonance oximetry was an effective and sensitive method for measuring cerebral PtO2 over a period of weeks in awake animals. These data show that the model of chronic hypoxia exhibits a large elevation in cerebral PtO2. The model would allow for studying factors that influence PtO2, such as the relative impact of changes in vessel density, oxygen carrying capacity, and CBF. A host of questions could be asked with respect to chronic adaptation to hypoxia: What are the values for PtO2 at the acclimated oxygen levels? Would chronic exposure to high oxygen result in a relative hypoxia on returning to normal oxygen? What oxygen levels would cause the acclimation observed in this study? Could they be achieved by duty cycles such as those encountered by flight crews? Also, Does the change in PtO2 indicate a time course of angiogenesis that may relate to the onset of high altitude cerebral edema?
Footnotes
Acknowledgments
The authors thank Dr. J. Hoopes and the neurosurgery laboratory at Dartmouth for the processing and photography of our histologic samples. The authors also thank Drs. J. Leiter and L.C. Ou for the acclimation chambers.
