Abstract
Heat shock protein 90 (HSP90), an essential component of several signal transduction systems, participates in the activation of endothelial nitric oxide synthase (eNOS) in cells. The objective of the current study was to determine if HSP90 and eNOS were functionally interdependent and colocalized in the cerebral circulation. The authors used isometric force recording, cyclic 3′5′-guanosine monophosphate (cGMP) radioimmunoassay (RIA), and immunogold electron microscopy (EM) to study canine basilar artery. They found that geldanamycin (0.1 to 10 μg/mL), a selective HSP90 inhibitor, caused concentration-dependent contractions in arterial rings (n = 6 dogs). Contractions to geldanamycin were unaffected by a cyclooxygenase inhibitor, indomethacin (10 μmol/L;P < 0.05, n = 6). Functional evidence for interaction between HSP90 and nitric oxide (NO)-mediated signaling included observations that the contractile effect of geldanamycin was the following: (1) endothelium-dependent, (2) abolished by Ng-nitro-l-arginine methylester (L-NAME; 0.3 mmol/L), and (3) nonadditive with the contractile effect of this NOS inhibitor (P < 0.01, n = 6 for each). Furthermore, RIA showed significant reduction in cGMP levels in arteries treated with geldanamycin (3 μg/mL;P < 0.02, n = 8), whereas immunogold EM demonstrated areas of colocalization of HSP90 and eNOS selectively in the cytoplasm of endothelial cells. The current findings suggest that in cerebral arteries, endothelial HSP90 plays an important role in modulation of basal NO-mediated signaling. This interaction may be particularly important in stress-induced up-regulation of HSP90 with subsequent alteration of vasomotor function.
The heat shock response originally observed in Drosophila melanogaster by Ritossa in 1962, refers to a rapid, stress-induced synthesis of a small set of proteins first identified in the 1970s and subsequently known as heat shock proteins (Parsell and Lindquist, 1993; Smith et al., 1998; Karunanithi et al., 1999). This response—whose triggers include thermal, chemical, and inflammatory stress—has been highly conserved throughout evolution. It has been proposed that induction of heat shock proteins is cytoprotective, as their roles include participation in normal protein folding and transport, abnormal protein degradation, and the prevention of protein aggregation (Parsell and Lindquist, 1993; Morimoto and Santoro, 1998; Smith et al., 1998; Karunanithi et al., 1999).
Members of the heat shock protein 90 (HSP90) family are present in the cytoplasm and nucleus of all eukaryotes and in the endoplasmic reticulum of higher eukaryotes (Parsell and Lindquist, 1993; Pratt, 1997, 1998; Scheibel and Buchner, 1998). Human HSP90, encoded by two genes, exists as a 180 kDa dimer and functions as a heterocomplex involving adenosine triphosphate (ATP), HSP70, and other smaller protein cochaperones including p23, p60 (Hop), and the immunophilin FKBP52 (Parsell and Lindquist, 1993; Pratt, 1997, 1998; Smith et al., 1998). Heat shock protein 90 is composed of multiple domains, the most highly conserved being the N-terminal domain (Parsell and Lindquist, 1993; Pratt, 1997; Scheibel and Buchner, 1998). This region has been studied extensively by x-ray crystallography (Prodromou et al., 1997; Stebbins et al., 1997), which has shown it to bind ATP at an ATPase active-site in a critical 15α-deep pocket (Grenert et al., 1997; Stebbins et al., 1997; Toft, 1998). It has now been demonstrated that geldanamycin, a benzoquinone ansamycin related to herbimycin A, selectively binds to the unconventional ATP-binding site in HSP90 and inhibits the function of this protein (Whitesell et al., 1994; Grenert et al., 1997; Stebbins et al., 1997; Toft, 1998), most likely by preventing its ATP-mediated change in conformation (Pratt, 1998; Smith et al., 1998). Given the high selectivity of its binding (Lawson et al., 1998; Smith et al., 1998; Toft, 1998), geldanamycin represents an important investigative tool that can be used to unmask the association between HSP90 and other proteins in a specific manner.
The association between HSP90 and endothelial nitric oxide synthase (eNOS) was first described by Garcia-Cardena et al. (1998). Findings from their study in cultured endothelial cells and rat aorta suggested that agonist-and shear-stress-stimulated production of nitric oxide (NO) was dependent upon a dynamic interaction between HSP90 and eNOS. Using geldanamycin, these authors reported attenuation of agonist-stimulated cyclic 3′5′-guanosine monophosphate (cGMP) production and acetylcholine-induced vasorelaxation (Garcia-Cardena et al., 1998). In the cerebral circulation, interaction between HSP90 and eNOS would have important implications for the regulation of cerebral blood flow under normal conditions and during hemodynamic or inflammatory stress, where up-regulation of HSP90 expression with subsequent alteration of vasomotor function may occur. In this light, the possibility of HSP90 as a specific pharmacologic target (Scheibel and Buchner, 1998) seems attractive from the perspective of potential control, albeit indirect, of NO production in vascular diseases including cerebral vasospasm, in which impaired NO-mediated signaling has been implicated (Chen et al., 1998). Therefore, to investigate this paradigm in cerebral arteries, the authors commenced a study with the aim of determining both if HSP90 modulated basal (unstimulated) versus agonist-stimulated NO-mediated signaling, and if it was colocalized with eNOS in canine basilar artery, a preparation that the authors have studied extensively in the past (Chen et al., 1997, 1998; Onoue et al., 1999). Based on the preliminary data in isolated canine basilar arteries (Khurana et al., 1999), the authors reported concentration-and endothelium-dependent contractions to geldanamycin that were insensitive to indomethacin but abolished by Ng-nitro-l-arginine methylester (L-NAME), findings that supported the notion that HSP90 could modulate basal vasomotor tone in isolated arteries through an interaction with eNOS. Viswanathan et al. (1999) subsequently postulated the involvement of HSP90 in flow-induced vasodilation of rat middle cerebral artery. In their experiment, geldanamycin, like L-NAME, significantly reduced the percentage change in the diameter of serotonin-contracted arteries in response to pulsatile flow, however no further characterization of this phenomenon was reported. In the current study, the authors used isometric force recording, cGMP radioimmunoassay (RIA), and immunogold electron microscopy (EM) to provide functional, biochemical, and morphologic evidence for an association between HSP90 and eNOS proteins in canine basilar artery.
MATERIALS AND METHODS
Animal tissue
Experiments were performed ex vivo using basilar arteries from mongrel dogs anesthetized with 25 mg/kg sodium pentothal administered intravenously. All procedures were performed in accordance with institutional guidelines.
Isometric force recording
Basilar artery rings (3 to 4 mm in length), with or without endothelium (Onoue et al., 1999), were connected to isometric force transducers and suspended in organ chambers filled with modified Krebs (control) solution as described previously (Chen et al., 1997; Onoue et al., 1999). Where specified, L-NAME (0.3 mmol/L) or indomethacin (10 μmol/L) was added to the control solution 15 or 30 minutes, respectively, before adding other compounds. Geldanamycin (3 μg/mL) was added 15 minutes before other compounds (Garcia-Cardena et al., 1998), except when used for acquiring concentration-response data (when it was added in a cumulative manner). Contractions are expressed as percentage of maximal contraction induced by KCl (50 mmol/L; 100%) and relaxations are expressed as percentage of maximal relaxation induced by papaverine (0.3 mmol/L; 100%). Median effective concentration (EC50) values were calculated as described elsewhere (Onoue et al., 1999).
cGMP assay
The RIA used to determine the tissue content of cGMP has been detailed previously (Onoue et al., 1999). Note that during 30-minute incubation in modified essential medium at 37°C, certain rings were treated in the last 2 minutes with geldanamycin (3 μg/mL) or L-NAME (0.3 mmol/L), whereas others (control) remained untreated; a 2-minute exposure time was chosen based on the average time to achieve maximal responses using these inhibitors. Subsequent steps were followed as previously described (Onoue et al., 1999).
Immunogold electron microscopy
Localization of eNOS and HSP90 proteins was examined in ultrathin sections of specimens on uncoated nickel grids through immunogold labeling (Bendayan, 1989). Specimens were fixed in 4% paraformaldehyde/0.2% glutaraldehyde and embedded in LR White resin. Thin cross-sections of arteries were mounted on 300-mesh nickel grids, preincubated in PBS-Tween 20 with 2% normal goat serum, and incubated in mouse monoclonal anti-eNOS (Transduction, San Diego, CA, U.S.A.) or anti-HSP90 (StressGen, Victoria, BC, Canada) antibody (for single-labeling) diluted 1:250. They were then rinsed in PBS-Tween 20, incubated in goat anti-mouse IgG-serum, conjugated to 5 nm colloidal gold, and rinsed again. After drying, grids were stained with uranyl acetate and lead citrate. Photomicrographs of labeled cells were obtained using a CM-10 transmission electron microscope. Control specimens were exposed to the same procedure after omission of primary antibody. The above protocol was also followed in sections used for double-labeling, except that each of the two cross-section faces was incubated separately with monoclonal anti-eNOS or anti-HSP90 antibody, with indirect conjugation to 5 nm (for HSP90) or 15 nm (for eNOS) colloidal gold.
Drugs
The authors used bradykinin, calcium ionophore A23187, geldanamycin, L-NAME, substance P, uridine triphosphate (UTP; all from Sigma, St. Louis, MO, U.S.A.), and diethylamine NONOate (DEANONOate; Cayman, Ann Arbor, MI, U.S.A.). All drugs were dissolved in distilled water (Onoue et al., 1999), except for A23187 and geldanamycin (dissolved in dimethyl sulfoxide). For geldanamycin (0.1 to 10 μg/mL), corresponding dimethyl sulfoxide volumes were 0.5 to 50 μL (or 0.002% to 0.2% of control solution volume). All drugs are expressed in final molar (mol/L) concentration in the bathing solution, except for geldanamycin (conventionally expressed in μg/mL (Whitesell et al., 1994; Stebbins et al., 1997; Garcia-Cardena et al., 1998; Lawson et al., 1998), where 0.1 to 10 μg/mL = 0.18 to 18 μmol/L).
Statistical analysis
Data are expressed as mean ± SD; n refers to the number of animals studied. Data were analyzed by Student's paired t-test or, for paired concentration-response curves, repeated measures analysis of variance (Onoue et al., 1999). Statistical significance was accepted at P < 0.05.
RESULTS
Functional study
Geldanamycin (0.1 to 10 μg/mL), a selective HSP90 inhibitor, caused concentration-and endothelium-dependent contractions of basilar artery rings (Fig. 1A). In rings with endothelium, the maximum contraction to geldanamycin was 28% ± 15%, whereas in rings without endothelium, the maximum contraction was 0% ± 3% (Fig. 1B). Differences between responses to geldanamycin in rings with and without endothelium were found to be statistically significant (P < 0.01; n = 6) and not attributable to the vehicle used (dimethyl sulfoxide; n = 6; data not shown). Peak contractile responses to geldanamycin (3 μg/mL) applied to rings with endothelium were observed within 90 to 120 seconds of exposure to this compound. In rings with endothelium, geldanamycin-induced contractions were not affected by the presence of a cyclooxygenase inhibitor, indomethacin, but were abolished in the presence of a NOS inhibitor, L-NAME. The maximum contraction to geldanamycin (0.1 to 10 μg/mL) in the absence and presence of indomethacin (10 μmol/L) was 28% ± 15% and 25% ± 9%, respectively (P > 0.05, n = 6); whereas in the absence and presence of L-NAME (0.3 mmol/L), the maximum contraction to geldanamycin was 28% ± 15% and 3% ± 6%, respectively (P < 0.01, n = 6). In rings without endothelium, relaxations to the NO donor DEANONOate (1 nmol/L to 10 μmol/L) were unaffected by the presence of geldanamycin (3 μg/mL;P > 0.05, n = 5; Table 1). In a separate set of experiments, contractions to geldanamycin (3 μg/mL) were found to be 19% ± 5%, and these were significantly different from contractions to L-NAME (0.3 mmol/L; 60% ± 23%;P < 0.01, n = 6). However, contractions to L-NAME were not significantly different from contractions to L-NAME plus geldanamycin (61% ± 14%;P > 0.05, n = 6; Fig. 2). In arteries contracted with UTP (10 μmol/L), the authors did not observe any difference in relaxations to the endothelium-dependent vasodilators bradykinin, substance P, or calcium ionophore A23187 in the absence or presence of geldanamycin (3 μg/mL;P > 0.05 and n = 6 for each;Table 1); nor did they observe any difference in contractions to UTP (0.1 μmol/L to 30 μmol/L) in the absence (maximum contraction and −log mol/L EC50 = 79% ± 20% and 5.4 ± 0.3, respectively) or presence (maximum contraction and −log mol/L EC50 = 90% ± 24% and 5.6 ± 0.3, respectively) of geldanamycin (3 μg/mL;P > 0.05, n = 6).
Effect of geldanamycin on relaxations to DEANONOate, bradykinin, substance P, and calcium ionophore A23187 in canine basilar artery
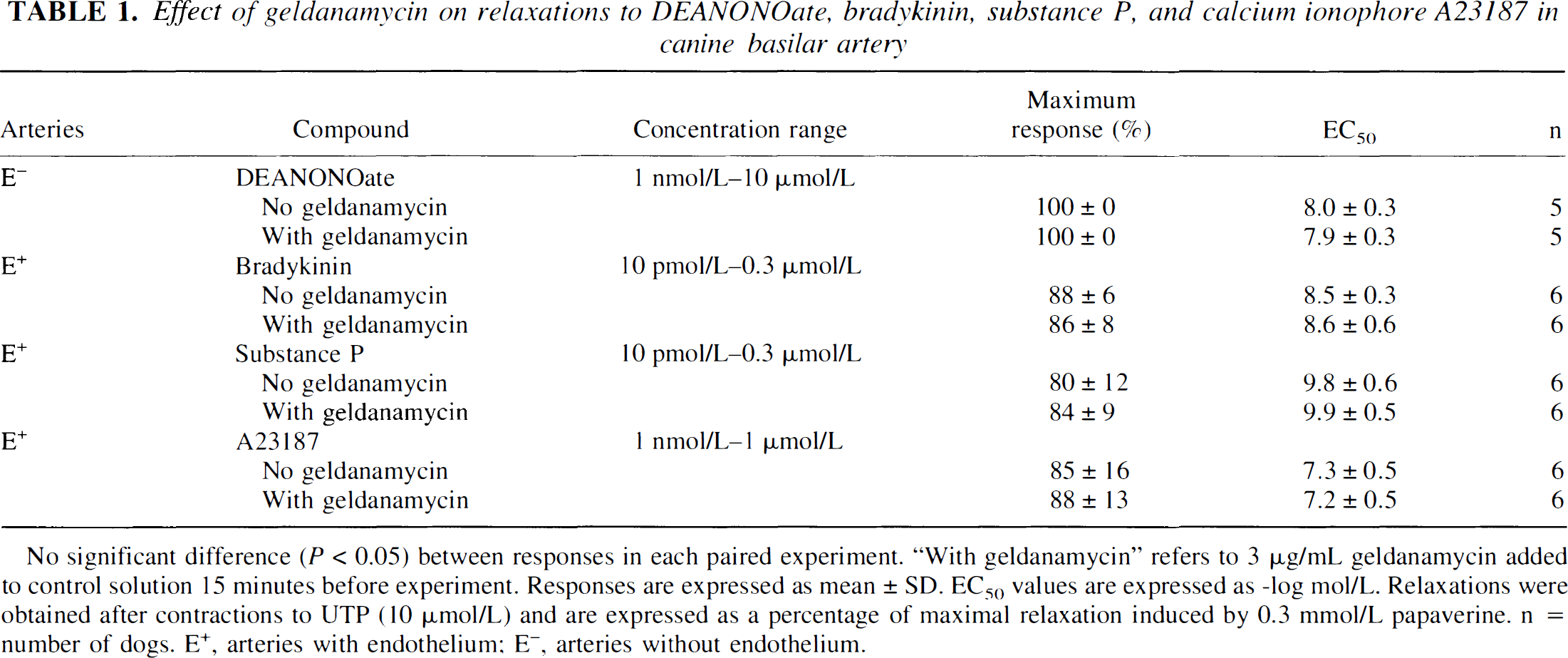
No significant difference (P < 0.05) between responses in each paired experiment. “With geldanamycin” refers to 3μg/mL geldanamycin added to control solution 15 minutes before experiment. Responses are expressed as mean ± SD. EC50 values are expressed as -log mol/L. Relaxations were obtained after contractions to UTP (10μmol/L) and are expressed as a percentage of maximal relaxation induced by 0.3 mmol/L papaverine. N = number of dogs. E+, arteries with endothelium; E−, arteries without endothelium.
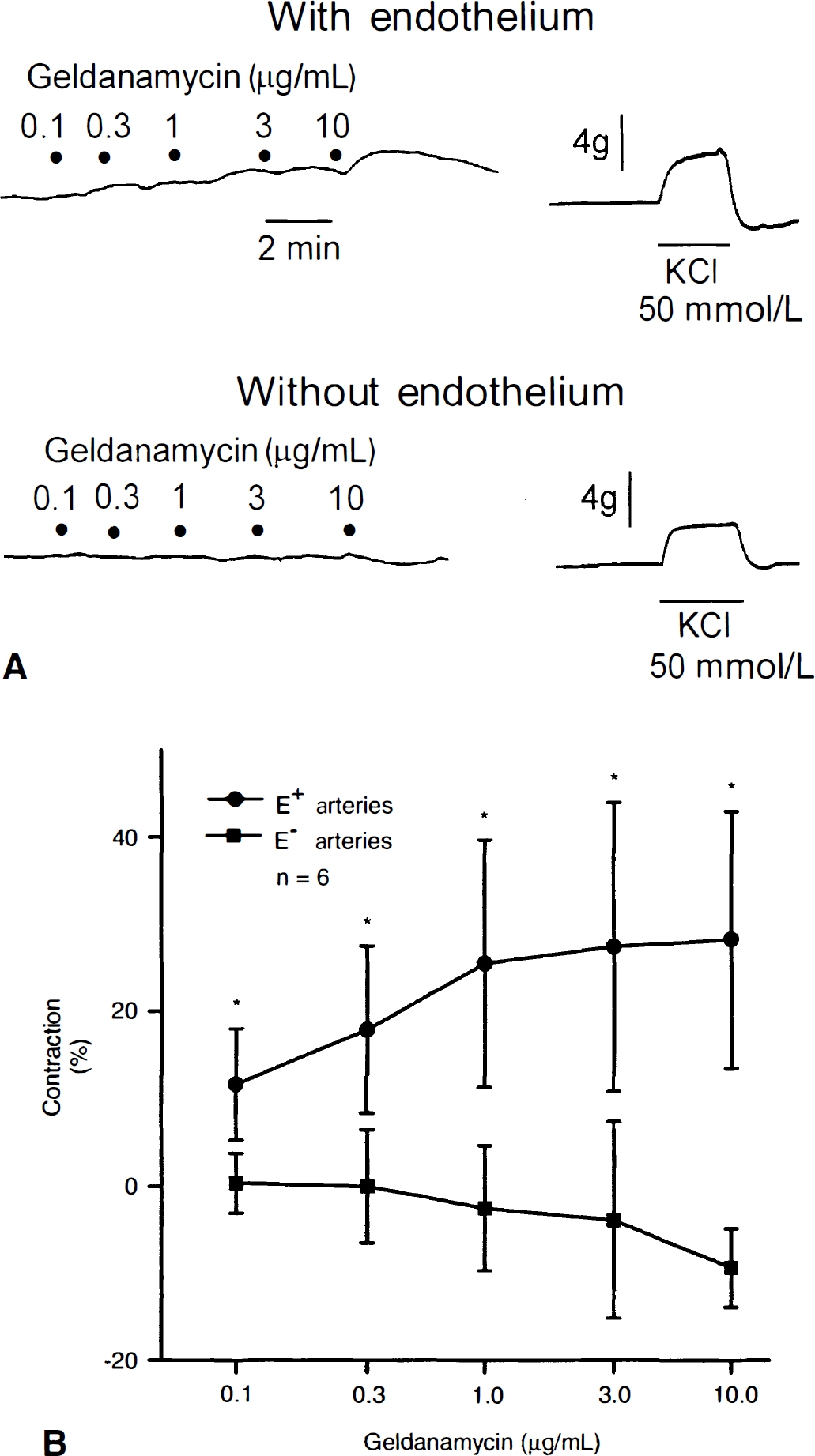
Contractions to geldanamycin in canine basilar artery.
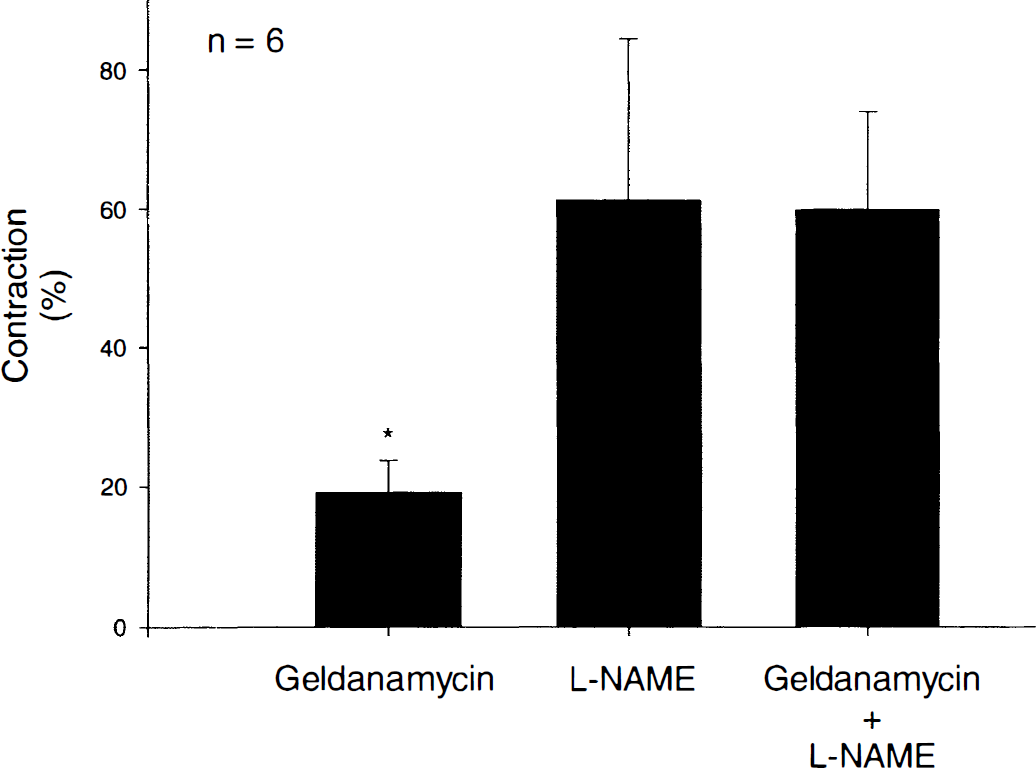
Functional response to geldanamycin and Ng-nitro-l-arginine methylester (L-NAME) in canine basilar artery. Contractions to geldanamycin (3 μg/mL) alone versus L-NAME (0.3 mmol/L) alone versus geldanamycin plus L-NAME in rings with endothelium. *Statistically significant difference (P < 0.01; n = 6) among data in the geldanamycin alone column compared with the other two columns. Data are shown as mean ± SD and expressed as percentage of maximal contraction induced by KCl (50 mmol/L; 100% = 4.5 ± 1.6 g).
Biochemical study
In control (untreated) rings, cGMP levels were 55 ± 61 pmol/mg protein. These were significantly different from cGMP levels in rings treated with geldanamycin (3 μg/mL; cGMP = 14 ± 14 pmol/mg protein;P < 0.02, n = 8) or L-NAME (0.3 mmol/L; cGMP = 14 ± 5 pmol/mg protein;P < 0.002, n = 8; Fig. 3). However, there were no differences in cGMP levels in rings treated with geldanamycin compared with L-NAME (P < 0.05, n = 8).
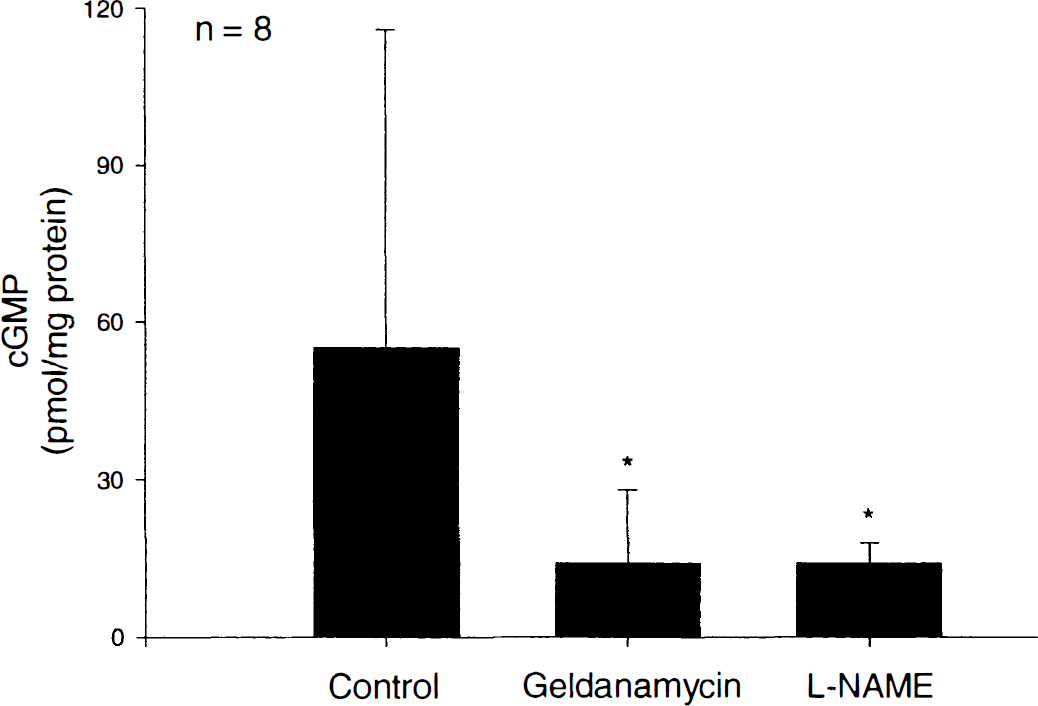
Biochemical response to geldanamycin and Ng-nitro-l-arginine methylester (L-NAME) in canine basilar artery. Cyclic 3′5′-guanosine monophosphate (cGMP) measurements in rings treated for 2 minutes with geldanamycin (3 μg/mL) or L-NAME (0.3 mmol/L) compared with control (untreated) rings. *Statistically significant differences between control and geldanamycin data (P < 0.02; n = 8), and between control and L-NAME data (P < 0.002; n = 8). Data are shown as mean ± SD.
Morphologic study
The authors detected eNOS selectively in the cytoplasm of canine basilar artery endothelial cells using EM with colloidal gold single-immunolabeling (Fig. 4A). Using this technique, HSP90 was detected in endothelial (Fig. 4B) and smooth muscle cells (data not shown). Double-immunolabeling methods demonstrated areas of colocalization of eNOS and HSP90 proteins selectively in the cytoplasm of endothelial cells (Fig. 4C). No immunogold labeling was detected in control single-labeling studies performed in the absence of primary antibody (data not shown). In control double-labeling studies, only HSP90 or eNOS immunogold labeling (in the distribution described above) was detected after omission of anti-eNOS or anti-HSP90 antibody, respectively; no immunogold labeling was detected (data not shown) after omission of both antibodies.
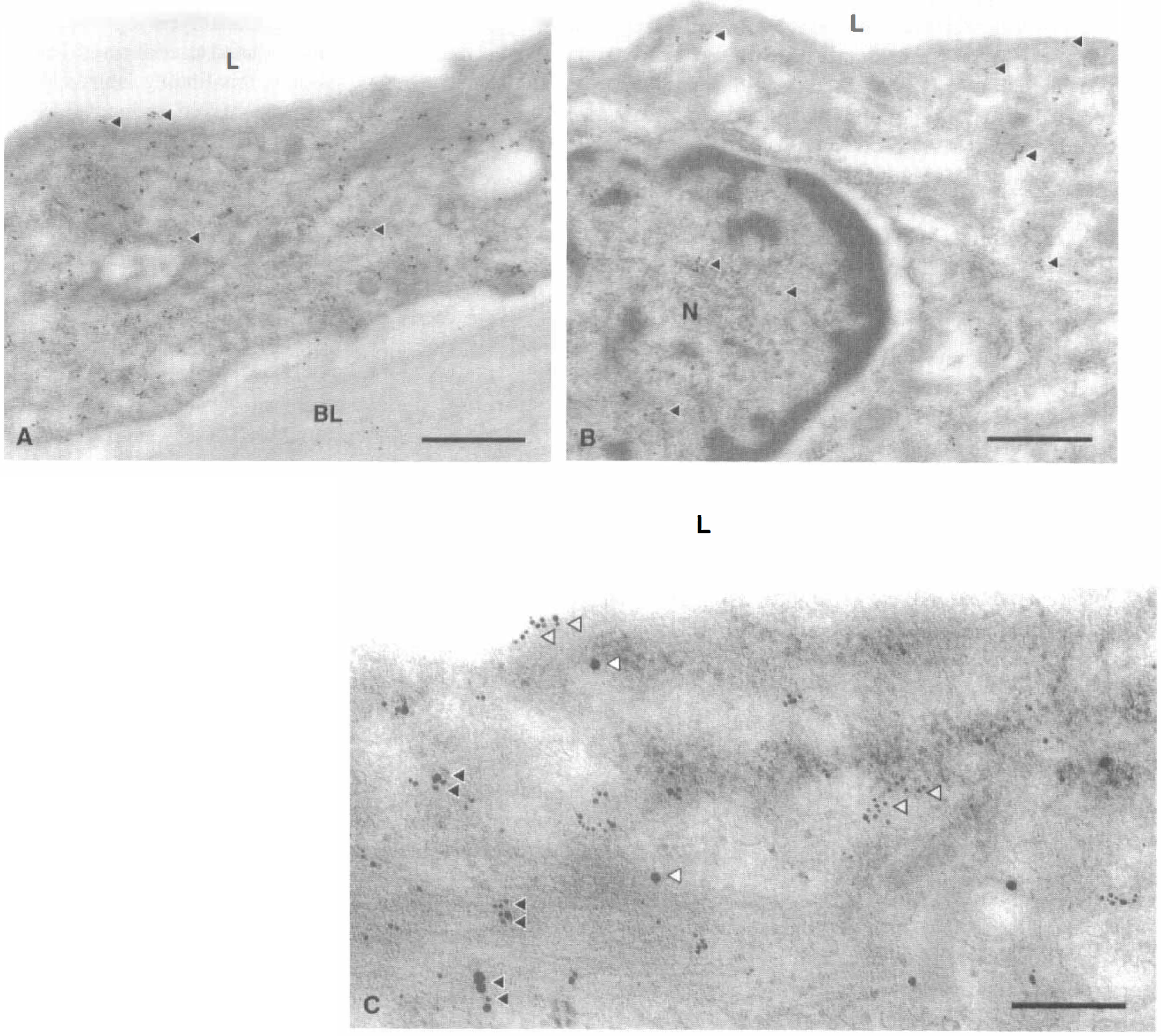
Immunogold electron microscopy in canine basilar artery.
DISCUSSION
Previous reports alluding to HSP90-eNOS interaction in isolated arteries (Garcia-Cardena et al., 1998; Viswanathan et al., 1999) suggest that HSP90 modulates the component of NO production stimulated by receptor-mediated agonists, shear stress, and pulsatile flow. However, the authors have no information regarding the effect of HSP90 on basal NO-mediated signaling in any arterial bed, nor any morphologic data regarding the subcellular distribution of HSP90 versus eNOS. In light of the pivotal role played by NO in regulation of the cerebral circulation and its contribution to the pathogenesis of cerebrovascular disease, the authors believed that obtaining such information would be a logical and important continuation of their efforts to characterize mechanisms involved in the local control of NO biosynthesis and release in cerebral arteries (Chen et al., 1997, 1998; Onoue et al., 1999). The novel findings of this study are that in cerebral arteries: the selective HSP90 inhibitor, geldanamycin, causes endothelium-dependent vasoconstriction; the vasoconstrictive effect of geldanamycin is acute and appears to be associated with inhibition of basal, but not agonist-stimulated, NO production; and there is evidence for colocalization of HSP90 and eNOS selectively in endothelial cell cytoplasm.
Functional evidence
Consistent with the role of HSP90 in facilitating eNOS function (Garcia-Cardena et al., 1998), the authors found that inhibition of HSP90 through geldanamycin was associated with concentration-and endothelium-dependent contraction. That this was not attributable to interaction of this inhibitor with cyclooxygenase or smooth muscle guanylate cyclase was suggested by the lack of effect of indomethacin on geldanamycin-induced contractions in arteries with endothelium, and the lack of effect of geldanamycin on relaxations to the NO donor DEANONOate in arteries without endothelium. Furthermore, it has been suggested that the high specificity of geldanamycin for HSP90 is because of the unconventional topology of the geldanamycin binding site in HSP90, a glycine-rich region situated in a deep cleft (Grenert et al., 1997; Stebbins et al., 1997). In fact, the only known protein with homology to HSP90 is bacterial topoisomerase II/DNA Gyrase B, similar only in the N-terminal ATP-binding fragment (Stebbins et al., 1997; Toft, 1998). In the current study, functional evidence for an association between geldanamycin-mediated inhibition of HSP90 function and reduced activity of eNOS included observations that the contractile effect of geldanamycin was (1) endothelium-dependent, (2) abolished by L-NAME, and (3) nonadditive with the contractile effect of this NOS inhibitor. The ability of geldanamycin to cause acute, L-NAME-sensitive contraction in unstimulated canine basilar artery suggests that HSP90 modulates basal NO-mediated signaling in this preparation. Interestingly, maximal geldanamycin-induced contractions represented only one-third of the magnitude of L-NAME-induced contractions, indicating that basal eNOS activity is only partially dependent upon its interaction with HSP90. Furthermore, in contrast with Garcia-Cardena et al. (1998), who reported geldanamycin-mediated impairment of relaxations to acetylcholine in rat aorta, the authors did not observe any effect of geldanamycin on agonist-stimulated relaxations, using receptor-mediated (bradykinin and substance P) and nonreceptor-mediated (A23187) endothelium-dependent relaxing substances. This disparity may be attributed to normal species, or regional vasomotor variation, or both, or to possible differences in the signal transduction mechanisms leading to activation of eNOS under basal versus agonist-stimulated conditions in cerebral arteries. Lastly, the authors did not detect any effect of geldanamycin on contractions to UTP. Although this indicates that inhibition of HSP90-mediated NO signaling by geldanamycin does not increase the sensitivity of arteries to the vasoconstrictor, UTP, it does not preclude the possibility of hyperresponsiveness to vasoconstrictors whose mechanism of action may depend on other HSP90-mediated protein–protein interactions.
Biochemical evidence
The authors found geldanamycin significantly reduced cGMP levels in unstimulated arterial rings. In light of the current functional data, this finding lends support to the possibility that geldanamycin reduces basal eNOS-mediated NO production, as cGMP is the principal second messenger for NO. Interestingly, the level of reduced cGMP formation caused by geldanamycin was not significantly different from that caused by L-NAME, despite the authors' finding that contractions to L-NAME in unstimulated rings were approximately three times greater than those to geldanamycin. This apparent inconsistency may be partially explained by the different conditions involved in these two sets of experiments: functional studies were performed using passively stretched rings suspended in aerated, Krebs-filled organ chambers, and biochemical studies were performed using unstretched rings incubated in modified essential medium. It is also possible that the sensitivity of the cGMP assay may not be high enough to detect any difference between the inhibitory effect of geldanamycin versus L-NAME on cGMP production.
Morphologic evidence
It should be noted that as a consequence of optimizing immunogold detection in the tissue sections, the authors were unable to precisely detect membrane-bound structures such as caveolae and the Golgi apparatus. Therefore, the authors' localization data were limited by their inability to distinguish between specific organelle associations (if any) for HSP90 and eNOS in canine basilar artery. In isolated endothelial cells, Prabhakar et al. (1998) have shown that eNOS resides in cell membrane caveolae under basal conditions, but can translocate between caveolae and cytosolic structures in response to agonist stimulation. To the authors' knowledge, there is no information regarding the possibility of HSP90-enrichment in caveolae. In the current study, immunogold EM study of canine basilar artery revealed localization of eNOS only in the endothelial cell cytoplasm and the presence of HSP90 in endothelial and smooth muscle cell cytoplasm (with some nucleoplasmic detection). These findings are consistent with other immunogold EM studies (Biggiogera et al., 1996; Shochina et al., 1997). Through double-labeling, the authors demonstrated for the first time discrete foci of eNOS–HSP90 colocalization in endothelial cytoplasm. Although, when taken alone, this finding is not definitive in terms of indicating a physical interaction between HSP90 and eNOS, in the context of the authors' functional and biochemical data it does lend support to this possibility. It should be noted that the current morphologic data pertain to the basilar artery, a relatively large-diameter cerebral artery. The authors have no information regarding the distribution of HSP90 in smaller cerebral arteries and in the cerebral microvasculature. Given that HSP90 is known to be a ubiquitous intracellular protein and that its presence has been demonstrated in the endothelium of peripheral arterioles (Shah et al., 1999), it seems likely that HSP90 is present in small brain arteries, however the extent of its interaction with eNOS in these vessels remains to be determined.
Implications of HSP90-mediated protein–protein interactions in the cerebral circulation
Heat shock protein 90 interacts dynamically with a wide variety of proteins—including steroid receptors, hepatitis B virus reverse transcriptase, and p53 tumor suppressor mutants—and plays an important role in protein kinase-mediated signal transduction (Pratt, 1998; Scheibel and Buchner, 1998). The work of Garcia-Cardena et al. (1998) suggests that the recruitment of this chaperone to cellular targets such as eNOS depends on the activation state of the cell. The current work suggests that in cerebral arteries HSP90–eNOS interaction can also occur under basal (or unstimulated) conditions. Taken together, these findings indicate that in the vasculature, the level of HSP90 expression may be closely related to the functional integrity of critical vasoregulatory enzymes such as eNOS. This has two major implications for the cerebral circulation. First, HSP90 may represent a novel molecular target for pharmacologic agents aimed at indirectly up-regulating, for example, NO production in vascular diseases such as cerebral vasospasm in which impaired NO signaling is known to occur (Chen et al., 1997, 1998; Onoue et al., 1999). Such an approach may be an alternative to restoring NO biosynthesis in the blood vessel wall through recombinant eNOS gene transfer to arteries (Chen et al., 1998). Second, the induction of HSP90 synthesis during hemodynamic, thermal, hypoxic, or inflammatory stress may be cytoprotective in the central nervous system by enhancing the ability of cells to transport, stabilize, and maintain the activity of normal proteins while participating in the degradation of abnormal proteins (Parsell and Lindquist, 1993; Morimoto and Santoro, 1998; Smith et al., 1998; Karunanithi et al., 1999). Interestingly, cytoprotection may also result from coinduction of HSP70 synthesis. Up-regulated expression of this chaperone, known to form part of the HSP90 chaperone supercomplex, is associated with inhibition of nuclear factor-kappa B (NF-κB), a molecular signal critical to inflammatory responses triggered by cytokines and mediated by the inducible isoform of NOS (iNOS;De Vera et al., 1996; Wong et al., 1997; Morimoto and Santoro, 1998).
In summary, the current study suggests that HSP90 and eNOS are functionally interdependent and colocalized in canine basilar artery. The current findings indicate that in cerebral arteries, endothelial HSP90 may play an important role in modulating the basal production of NO. This interaction may be particularly important in the setting of stress-or pharmacologically-induced up-regulation of HSP90 with subsequent alteration of vasomotor function.
Footnotes
Acknowledgements
The authors thank Drs. William Sessa and David Toft for their invaluable suggestions with regard to the content of this manuscript, and Ms. Janet Beckman for her assistance with its preparation.
