Abstract
Considerable structural plasticity is possible in the damaged neocortex and connected brain areas, and the potential for significant functional recovery remains even during the chronic phases of the recovery process. In this article, the authors review the literature on use-dependent morphologic events, focusing on the direct interaction of behavioral experience and structural changes associated with plasticity and degeneration. Experience-associated neural changes have the potential to either hinder or enhance functional recovery; therefore, issues concerning the nature, timing, and intensity of behavior-based intervention strategies are addressed.
The potential influence of behavioral experience on neural plasticity and recovery of function in the adult mammalian brain has received increased attention in recent years. Behavior typically changes when the brain is injured, which means that behavioral experience often is altered quite dramatically. Indeed, there is evidence that compared with the intact brain, the damaged brain may be particularly sensitive to self-imposed behavioral outcome and experiential interventions (Fig. 1).
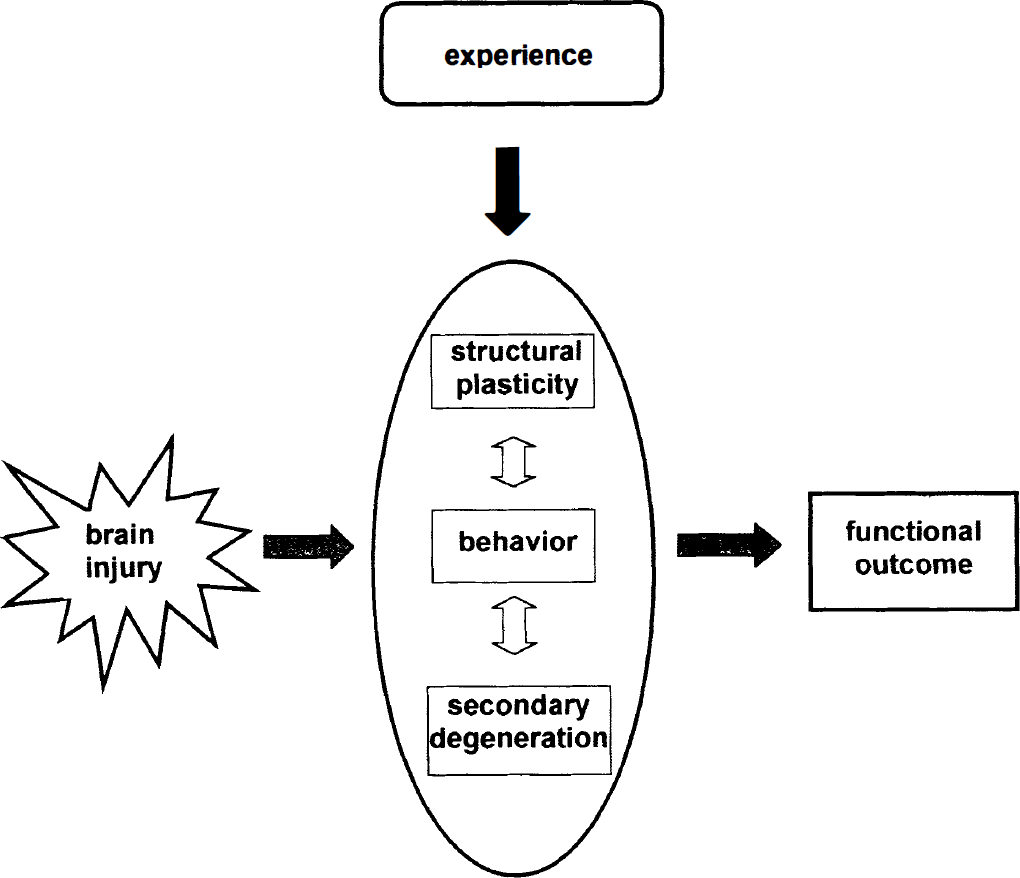
Schematic diagram of the interrelations between posttraumatic neural events and behavior. Experience can change these relations, thereby impacting functional outcome.
Adequate behavioral and anatomic assessment methods are crucial for teasing apart the separate and interactive contributions of experience-dependent and experience-independent events (Schallert et al., 2000a; Kolb, 1995; Jones et al., 1994, 1996). Use-dependent anatomic events are under-appreciated but are fundamental to understanding recovery of function after ischemic and other brain injury. The authors reviewed the literature on behaviorally driven morphologic events in the neocortex, particularly studies that have implications for establishing optimal behavior-based rehabilitative training.
ACTIVITY-DEPENDENT NEURAL EVENTS AFTER BRAIN INJURY
Greenough and colleagues (Ivanco and Greenough, 2000; Bury et al., 2000) have repeatedly demonstrated that enriched environments and motor learning in adult animals are associated with growth of dendrites, and increases in dendritic spines and synaptogenesis. These morphologic changes are also found after brain injury, suggesting that experience and plasticity after brain injury may share common processes. Indeed, Kolb et al. (Kolb and Gibb, 1991; Kolb, 1995; Kolb et al., 1998c) showed that recovery after brain injury was invariably predicted by dendritic growth in remaining tissue.
Rutledge et al. (1974; Rutledge, 1978) examined the structural effects of loss and chronic reinstatement of neural activity after undercutting the grey matter in the suprasylvian gyrus in cats. The surgical procedure, which deafferented the neocortex without detectable interruption of blood flow or intrinsic cortical circuitry, drastically reduced dendritic and axonal branches, spines, and synapses in the pyramidal neurons of undercut tissue. The interesting finding was that low level electrical stimulation produced substantial reinnervation, as indicated by protection against, for example, the loss of dendritic spines in apical dendrites. These experiments illustrated that structural events after brain injury can depend on activity.
Therefore, it was reasonable to expect that other forms of activity, such as novel sensorimotor behavior required for deficit compensation, might induce structural changes in the injured brain, or that deficit-related sensorimotor disuse might limit the potential extent of structural change. Focal unilateral cortical injury may severely limit use of one upper extremity while leaving function of the other intact. Taub and his colleagues (1993) argued that the potential for recovery of function is retarded in part because the patients have learned not to use the affected extremity; that is, it is more difficult and fatiguing to use the impaired arm, whereas learned compensatory use of the nonaffected arm, and postural adjustments, provide adequate compensatory function. Forcing the patients to use the affected arm by constraining the nonimpaired arm leads to dramatic improvement in function and expansion of the motor map (Liepert et al., 1998, 2000).
In animal models, unilateral damage to the forelimb area of the sensorimotor cortex leads to preferential use of the forelimb ipsilateral to the damage for novel movements, especially in the first few weeks (Jones and Schallert, 1992, 1994; Schallert et al. 2000a). The impaired forelimb is used for some tasks, but if it is constrained further by a plaster of paris cast, dendritic arborization of pyramidal cells in the surrounding cortical tissue is reduced (Fig. 2) and restoration of function is retarded.
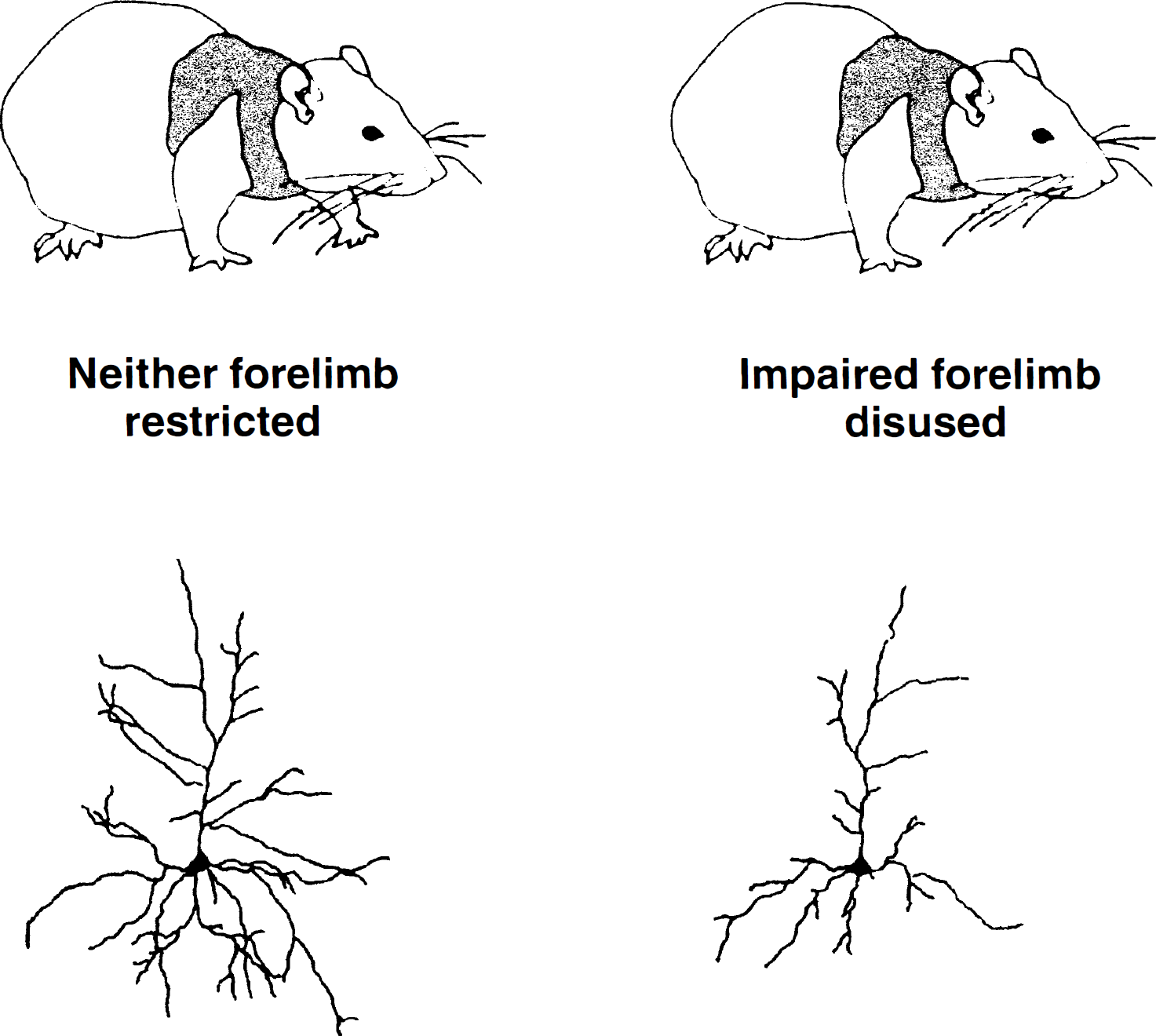
Dendritic trees of neurons in the perilesion area of rats that were not casted after injury and rats that rested the impaired forelimb because it was restrained inside a plaster cast. Note the paucity of dendritic branches in animals that rested the impaired forelimb.
Under use of the affected forelimb may be promoted not only because it becomes more difficult and tiresome to use it, but also because there is reorganization in the intact cortex that permits improved function in the nonimpaired forelimb or in bimanual coordination. Jones and colleagues (1992; 1994; Schallert and Jones, 1993; Jones et al., 1996; Bury et al., 2000; Kolb, 1995) found that there was a time-and use-dependent increase in dendritic arborization in Layer V pyramidal neurons of the intact sensorimotor cortex if the injury is severe enough and the neural events are optimal. Dendritic arborization was preceded by astrocytic hypertrophy and followed by arbor pruning and marked synaptogenesis, including multiple synaptic boutons and perforated synapses of the kind associated with long-term potentiation.
Motor skills learning and acrobatic training enhanced these structural changes (Jones, 1999; Jones et al., 1999). In contrast, immobilization of the corresponding unaffected forelimb during the period of structural change (but not afterwards) completely prevented dendritic arborization (Fig. 3) (Jones and Schallert, 1994). Not only were the structural events use-dependent, but they (at least the dendritic events) appeared to be injury-dependent as well (Jones and Schallert, 1994; Bury et al., 2000; Schallert et al., 1997, 2000b). Thus, forced use of the forelimb in sham operated animals did not produce dendritic arborization in the corresponding cortex, indicating that degenerative events associated with denervation may be particularly permissive in promoting plasticity (Fig. 3). Forced use of one limb does induce astrocyte proliferation and fibroblast growth factor-2 (FGF-2) expression in Layer V astrocytes in the corresponding sensorimotor cortex (Fig. 4), and not in nonforelimb area regions (Bury et al., 2000).
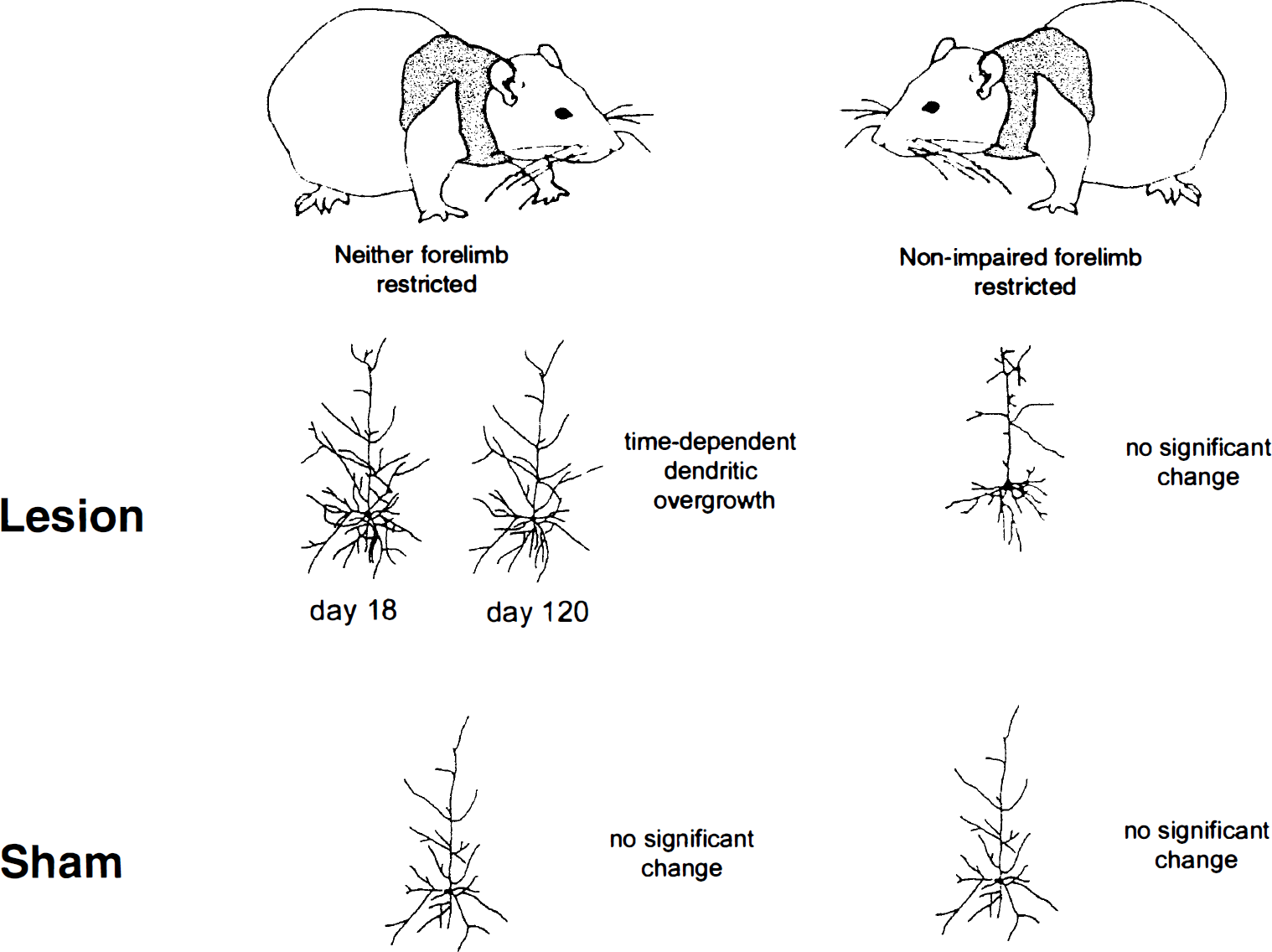
Injury-dependent dendritic growth occurs in the homotopic cortex after unilateral cortical injury. This overgrowth is maximal at 18 days postinjury and is blocked by restriction of use of the nonimpaired forelimb.
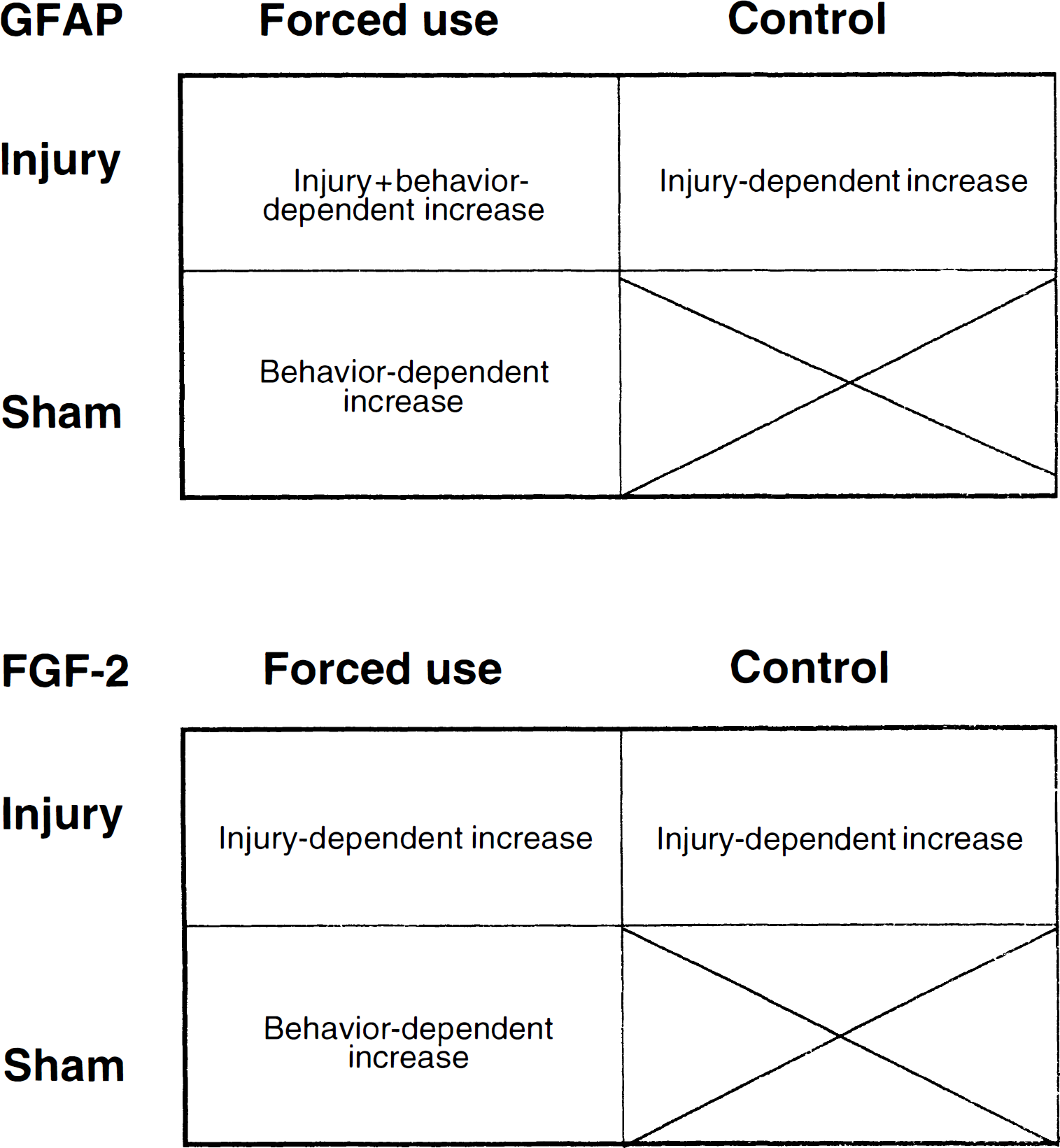
Summary of the injury-dependent, behavior-dependent, and injury + behavior-dependent effects on glial fibrillary acidic protein (GFAP) immunoreactivity and FGF-2 immunoreactivity after corpus callosum transection.
Growth-related synaptophysin (Stroemer et al., 1998) and GAP-43 expression (Kawamata et al., 1998) associated with enhanced functional outcome also have been found in the contralateral cortex after neocortical infarction, and these events were enhanced by amphetamine and FGF-2, respectively. The beneficial effects of amphetamine on recovery of motor function after stroke and other cortical injury (Feeney et al. 1982; Stroemer et al., 1998; Crisostomo et al., 1988; Walker-Batson et al., 1992, 1995; Goldstein et al., 1991; Boyeson, 1992; Hurwitz et al., 1989) appear to depend on task-specific practice and noradrenergic innervation of the homotopic, but not the injured, neocortex (Goldstein, 2000; Schmanke and Barth, 1997). As with undrugged conditions (Schallert and Jones, 1993; Schallert et al., 1997), the improved function observed with sympathomimetic agents may well depend importantly on use-related plasticity, such as dendritic arborization and changes in spine morphology and density (Robinson and Kolb, 1999a, 1999b) in both the injured and the intact hemisphere (Cramer et al., 1997; Cao et al., 1998; Silvestrini et al., 1998; Chollet et al., 1991; Netz et al., 1997). Notably, however, not all compensatory motor strategies or noncompensatory recovery of function depends on practice (Schallert and Whishaw, 1984; Whishaw, 2000; Soblosky et al.,1997; Schallert et al., 2000a; Schmanke and Barth, 1997).
The question of what injury-related neural events might set up the potential for use-dependent remodeling was addressed in a series of elegant studies by Jones and her collaborators (Bury et al., 2000). Severing a subset of afferent projections to the motor cortex through transection of the rostral aspect of the corpus callosum (simulating damage to the cortex in the opposite hemisphere) caused transient decrease in axonal volume, increased expression of the astrocytic marker glial fibrillary acidic protein-immunoreactivity (GFAP-IR) and other indices of axonal degeneration. Forcing preferential use of one forelimb led to further increases in Layer V GFAP-IR in the sensorimotor cortex corresponding to the used forelimb (Fig. 4). FGF-2 was increased by callosal section, but was not further increased by behavioral demand. Behavioral demand alone increased FGF-2 expression, suggesting that FGF-2 may have been maximally upregulated in astrocytes with either denervation or forced forelimb use (Fig. 4).
Reactive astrocytes associated with increased production of neurotrophic factors, cytokines, and early response genes have been suggested to promote growth and restructuring of remaining neurons after brain damage (Eddleston and Mucke, 1993; Reilly and Kumari, 1996; Kawamata et al., 1996, 1997; Bury et al., 2000), as well as to improve function in intact animals exposed to complex environments and enriched learning experiences (Sirevaag and Greenough, 1991; Matsuni and Leon, 1993; Gomez-Pinilla et al., 1998; Gomez-Pinilla and Cotman, 1992; Rowntree and Kolb, 1997).
Callosal sectioning followed by forced use of one forelimb also leads to an increase in dendritic arborization in the Layer V pyramidal cells in the cortical region associated with that forelimb (Bury et al., 1998). In contrast, no net change in arborization was found with callosal section alone, which produces no asymmetry in limb use (Bury et al., 1998). Taken together, these studies suggest that: (a) brain injury activates trophic factors or other mechanisms that can provide a fertile milieu for substantial neuronal remodeling and enhanced function, but (b) the neuronal remodeling and enhanced motor function can depend on experiential demand.
INTERVENTION STRATEGIES
A number of studies suggest that early chemical intervention after brain injury can enhance functional outcome. In addition to amphetamine, which has been useful in clinical trials, these include, but are not limited to, growth factors (Koketsu et al., 1994; Kawamata et al., 1996, 1997, 1998; Kolb, 1985), bone morphogenetic proteins (Chen et al., 2000; Ren et al., 2000; Schallert et al., 2000a), N-methyl-d-aspartate (NMDA) receptor antagonists (Foster et al., 1988; Barth et al., 1990; Dirnagl et al., 1990; Bielenberg and Beck, 1991; Dietrich et al., 1995; Bullock and Fujisawa, 1992), cholinergic and GABAergic antagonists (Barth, 1999;Schallert and Hernandez, 1998), and antioxidants (Barth et al., 1998). Despite a number of promising preclinical results, NMDA receptor antagonists did not prove efficacious in human studies (Lee et al., 1999). At the present time, the only efficacious drug for poststroke outcome has been d-amphetamine. There is another promising drug, however, namely nicotine. Like amphetamine, nicotine is a psychomotor stimulant. Chronic administration of both amphetamine and nicotine stimulate dendritic growth in prefrontal cortex (Brown et al., 2000; Robinson and Kolb, 1999a, 1999b), and it has been shown recently that two weeks of nicotine treatment after cortical or hippocampal injury facilitates recovery of performance in cognitive tasks (Brown et al., 2000).
LeVere (1980) was among the first to argue that use of compensatory strategies after brain injury prevents true recovery of preinjury abilities, and postulated that functional recovery might be enhanced if early, postinjury measures are taken to minimize compensation. As noted above, Taub and colleagues (1993) have investigated compensatory limb use in both humans and monkeys. If one upper extremity is disabled by a stroke, humans and monkeys become adept at relying on the nonimpaired upper extremity. It is possible that failed attempts to use the impaired limb early after injury discourage future use of that limb. In other words, continued lack of use of an impaired upper extremity may stem not from an actual inability to use the limb, but from habitual non-use in favor of the unimpaired limb.
Consistent with this hypothesis, it has been demonstrated in primates and humans that restricted use of the ipsilateral (nonimpaired) upper extremity, even years after brain injury, can increase the ability of an individual to effectively use the impaired limb, an effect that is associated with reorganization of the motor representation maps (Ogden and Franz, 1917; Ostendorf and Wolf, 1981; Wolf et al., 1989; Liepert et al., 1998, 2000; Kopp et al., 1999). Nudo and colleagues (1996) have provided evidence that targeted training of an impaired hand can greatly improve functional outcome. After a microstroke injury to the hand representation of the primate cortex, the investigators placed a flexible mitten on the unaffected hand. This mitten allowed the monkey to use the hand for gross motor functions, such as walking or grasping the bars of the cage, but forced the animal to use the affected hand for tasks involving fine motor dexterity, such as removing food pellets from conically shaped containers. This posttraumatic training improved functional outcome in the impaired hand relative to untrained animals. Nudo found that the motor abilities of the untrained animals continued to deteriorate after the injury, presumably because of disuse. In contrast, the trained animals did not worsen after the injury. It appears that the training acted largely to stabilize the motor performance rather than actually improve it. Kolb et al. (2000) have found a similar result in rats. Rats with motor cortex lesions were trained for 1 hour per day to reach for food with their impaired limb. After three months of training the animals showed little improvement, whereas the performance of the untrained animals deteriorated.
COMBINED PHARMACOLOGIC AND REHABILITATIVE STRATEGIES
Although compelling evidence indicates that rehabilitative therapy is useful when combined with pharmacologic treatment, there are surprisingly few studies that take potential advantage of this idea, except amphetamine experiments (Hovda and Feeney, 1984; Crisostomo et al., 1988; Kline et al., 1994; Walker-Batson et al., 1995; Schmanke and Barth, 1997). Postlesion training is also important for attaining maximum efficacy of striatal transplants of embryonic tissue after 6-hydroxydopamine (6-OHDA) injection. If postlesion motor training is combined with grafts of embryonic tissue after striatal injury, the combination of training + graft is much more effective at promoting recovery than a graft alone (Mayer et al., 1992; Brasted et al., 1999). These data are interesting in light of recent work showing that in 6-OHDA-treated animals, forcing early (but not late) use of the forelimb corresponding to the degenerating nigrostriatal dopamine system dramatically improves functional outcome and protects against the loss of dopamine (Tillerson et al., 1998; Schallert et al., 2000b). Results of these studies appeal to common sense because they suggest that, for optimal motor recovery to occur, an animal sometimes must make adaptive movements to actively engage the restorative potential of the chemical treatment being administered and to protect against ongoing degenerative events. It is possible that providing exogenous neurotrophic factors that enhance axonal (Kawamata et al., 1996, 1997) or dendritic growth (Ren et al., 2000; Schallert et al., 2000a; Kawamata et al., 1998) might provide more favorable conditions for concomitant rehabilitative training.
ENVIRONMENTAL COMPLEXITY MANIPULATIONS
Effects of exposure to an enriched environment (EE) on the brain and subsequent behavior have been extensively studied. Hebb (1949) was the first to discover that rats living amid stimulating surroundings had better problem solving skills than rats living in standard laboratory cages. It appears that he brought several laboratory rat pups home as pets for his children. When he later returned them to the laboratory, he noticed that the pet rats demonstrated better problem-solving skills than the laboratory rats. This led Hebb to postulate that the enriched environment the rats had been exposed to in his home afforded them the ability to make use of new experiences. Hebb's hypothesis gained support in the 1950's from work by Rosenzweig and colleagues (1962), who had found that training rats on various problem solving tasks caused an increase in levels of the enzyme acetylcholinesterase (AchE) in the brain (Krech et al., 1956). Because training animals was time consuming, the experimenters placed rats in an environment that would make them solve problems, a sort of self-training facility. This environment typically comprised 10 to 12 rats housed communally in an enclosure that contained a number of novel objects, changed daily. Like training, enriched housing also elevated AchE (Krech et al., 1960) and increased the weight of the neocortex (Rosenzweig et al., 1962; Rosenzweig and Bennett, 1996).
Enriched environment produces effects on the brain far beyond increasing the weight of certain cortical regions and increasing AchE. The thickness of various cortical areas, in particular the visual cortex, increases by 5% to 7% (Diamond et al., 1964). Much of the research investigating the effects of EE on the cortex focuses on the visual cortex, and includes findings of increased higher-order cortical dendritic branching (Greenough and Volkmar, 1973), more dendritic spines (Globus et al., 1973), and larger synapses (Mollgaard et al., 1971), although not all neural populations within the visual cortex demonstrate all of these changes (Juraska et al., 1980). Furthermore, there is evidence to indicate the presence of active synapse formation in rats exposed to EE (Greenough et al., 1985), and electron microscopic evidence for increased numbers of synapses per neuron (Turner and Greenough, 1985). In addition to structural changes within the visual cortex, metabolic changes have been inferred from findings of larger capillary volume and more densely packed capillaries (Black et al., 1987), and more mitochondria per neuron (Sirevaag and Greenough, 1987). It has also been shown that the visual cortex of animals exposed to EE contains an increased number of oligodendrocytes (Diamond et al., 1966; Szeligo and LeBlond, 1977). Although visual cortex has received much attention, other brain regions, including the hippocampus (Diamond et al., 1976), cerebellum (Greenough et al., 1986), superior colliculus (Fuchs et al., 1990), and sensorimotor cortex (Kolb et al., 1998c) undergo morphologic and ultrastructural changes in response to EE.
Given the extensive changes that occur in the brain after exposure to EE, it is not surprising that rats exposed to it show enhanced behavioral capabilities, just as Hebb's original pet rats did. Enriched environment rats show enhanced performance on the radial arm maze (Juraska et al., 1984), are better at using visual cues to escape from the Morris water maze (Mohammed et al., 1990; Falkenberg et al., 1992; Park et al., 1992; Paylor et al., 1992), are better at visual discrimination (Bors and Forrin, 1996), and reach criterion in a delayed alternation task faster than standard-housed controls (Mogenson, 1991). There is also evidence that EE reduces anxiety in rats, enabling them to explore novel objects longer and more frequently than rats housed in an impoverished environment (Widman and Rosellini, 1989), and reduces activity in an open field situation (Bors and Forrin, 1996). There is also evidence that EE reduces feeding neophobia (Bors and Forrin, 1996), and reduces the amount of defensive behavior displayed when confronted with a predator (Klein et al., 1994).
The effects of EE on the brain appear dependent on whether animals are housed together in the apparatus or individually. Neither rats housed collectively in standard laboratory housing or rats housed singly in EE show the dendritic changes characteristic of animals group-housed in EE, suggesting that the effects of EE are either due to a combination of social interaction and a complex environment or to the manner in which individuals interact with the complex environment (Rosenzweig, 1971). In an elegant experiment, Rosenzweig and Bennett (1972) showed that the effects of social housing in EE are because social housing serves to increase the activity of each rat, and that a single animal can show impressive cerebral changes after housing in EE, if that single animal is induced to be active in the enriched housing. The experimenters placed single rats in EE during the dark cycle, or under the influence of methamphetamine. Needless to say, these measures greatly increased the activity of the single rats, and vastly increased dendritic changes to the level of socially housed EE rats.
Age at onset of EE also influences the outcome of this manipulation on the adult brain. Although EE increases dendritic growth in occipital cortex of middle-aged rats, just as it does in young rats (Green et al., 1983), subsequent analyses suggest that the nature of dendritic changes differs among age groups. Kolb and colleagues (1998 c) characterized the dendritic changes that occur in response to EE in various age groups. In comparison with age-matched controls, rats placed into EE as young adults or in senescence show an increase in dendritic length and spine density, whereas animals placed in EE as juveniles show an increase in dendritic length, but a decrease in spine density. The latter analysis, however, was conducted on neurons in the sensorimotor cortex, whereas the age-dependent effects on dendritic morphology in the occipital cortex has not been explored.
A vast amount of literature documents the recovery-enhancing effects of EE housing after brain injury. This is perhaps not surprising, because EE has repeatedly been shown to enhance dendritic growth and synapse formation, as well as increase glial activity, all events have been linked to positive outcome after neurotrauma. Most studies concerning the effects of EE on functional recovery have used postoperative EE housing, and examined its effects on performance in the Morris water maze. Enriched environment attenuates water maze deficits in rats after fimbria-fornix lesions (van Rijzingen et al., 1997), hemidecortication (Whishaw et al., 1984), occipital lesions (Rose et al., 1993), neocortical damage (Kolb and Gibb, 1991), hippocampal damage (Galani et al., 1997), and in mice after septal lesion (Engellenner et al., 1982). Postoperative EE housing also improves performance on the Hebb-Williams maze (Galani et al., 1997) in rats. Ohlsson and Johansson (1995) found that postoperative housing in EE enhanced motor recovery after cerebral ischemia. Preoperative enrichment has been less extensively studied, but has been shown to reduce initial deficit and speed recovery of locomotion after bilateral sensorimotor cortex lesions (Held et al., 1985; Gentile et al., 1987). Thus, both pre-and postoperative enrichment appear to enhance performance on spatial tasks after a wide variety of central nervous system injuries.
Why EE enhances recovery of function is a matter of some debate. It has been speculated that preoperative enrichment functions to increase the level of organization of task-important brain regions, thus allowing them to better maintain function after damage (Held et al., 1985). It has also been postulated that EE offers plenty of opportunity for physical conditioning, which may help subsequent recovery of locomotor function after injury. Sheer physical exercise in the absence of enrichment, however, does not produce impressive effects on postinjury recovery of locomotor function (Gentile et al., 1987).
One critical issue regarding environment manipulations is the age of the animal and the time that treatment is initiated. In a series of studies on the effects of enrichment on functional recovery, the authors have shown (for example, Kolb et al., unpublished data) that rats with cortical injury in infancy show very large facilitation of recovery of cognitive and motor behaviors, and enhanced dendritic growth, but only if the enrichment begins at weaning. If enriched housing is delayed until adulthood, the experience is essentially without benefit. One explanation for this result is that the brain is particularly plastic after an injury, and it will change whether or not there is any type of special experience. On the other hand, if a treatment is delayed, the brain may have “used up” its capacity for further change and the treatment is essentially without benefit. It is not clear whether similar time constraints are also operating after injury in adulthood, but it is certainly an important issue.
Although most studies of EE look at the postoperative effects of treatment, there is a considerable literature on the preoperative effects of experience too (Shulkin, 1989). Overall, the results are inconclusive and appear to vary depending upon the details of the location of the injury and the circumstances surrounding its occurrence (Kolb, 1989). Nonetheless, it does appear that under certain circumstances preoperative experience can influence recovery. More recently, the authors found evidence that exposure to complex environments before cortical injury alters the response of the brain to the injury (Kolb and Rowntree, unpublished data). For example, if brains are analyzed for FGF-2 reactivity after a focal cortical injury, there is a 50% increase in response in preoperatively enriched animals versus untreated animals. Similarly, the authors also have shown that if pregnant rats are placed in complex environments for several hours a day throughout their pregnancy, the recovery from perinatal cortical injury in the offspring is enhanced (Gibb and Kolb, 2000). It appears that the experience alters the developing brain in some way that affects the later outcome from injury, although the mechanism of action is still not known.
Rose and colleagues (1987, 1993) have repeatedly pointed out that most studies investigating the effects of EE on recovery of function after injury have used a paradigm in which rats are injured, and then postoperatively housed in EE. Therefore, because rats are not required to learn the behavioral task until after EE, performance of that task cannot signify recovery of function because a preoperative baseline was not established. Instead, these researchers propose that the effect of EE is to enhance compensation after injury, and not recovery. Kolb and Gibb (1991) have brought attention to another potential limitation of EE. They made large frontal cortex lesions and examined morphology of cortical dendrites. They found that EE increased dendritic branching in the visual cortex of lesioned and sham-operated animals, and in the parietal cortex of sham-operates, but not injured animals. Enriched environment did not enhance dendritic branching in the parietal cortex of lesioned animals above and beyond that caused by the lesion. In other words, having responded to the cortical injury, parietal neurons were unable to further respond to EE. These researchers postulate, therefore, that lesion-induced dendritic plasticity may preclude any changes caused by EE.
In summary, the effects of EE on the brains of intact animals are impressive and have been repeatedly documented. The effects of EE on the damaged brain and subsequent behavioral deficits are also well documented, and the protective effect of EE on behavior has been amply demonstrated (Johansson, 2000). However, the mechanisms by which EE produces its effects on behavior have yet to be elucidated.
TIMING AND INTENSITY OF INTERVENTION STRATEGIES
It has been suggested that injury sensitizes or prepares remaining neural tissue for drastic changes that allow it to compensate for injury (Schallert and Jones, 1993; Ivanco and Greenough, 2000). Indeed, the immediate postinjury milieu is exquisitely malleable, and considerable structural plasticity is possible. However, it is apparent that during the early postinjury period, depending on the lesion site, functional recovery is susceptible to irreparable disruption by aggressive or inappropriate measures. For example, treatment with diazepam during the early postoperative period (before day 4) disrupts recovery of sensory function after injury to certain cortical regions (Hernandez et al., 1988; Schallert and Hernandez, 1998; Jones and Schallert, 1992), although chronic exposure to diazepam after recovery has already occurred does not affect sensory function (Schallert et al., 1986, 1998).
Like drug therapies, physical therapy measures instituted early after injury must be used with discretion. In contrast to the beneficial effects of forcing overuse of the affected limb after 6-OHDA degeneration (Tillerson et al., 1998; Schallert et al., 2000b), intense rehabilitation measures after cortical injury can be detrimental. Physical interventions targeted at the forelimb impaired by injury to the forelimb representation area of the rat sensorimotor cortex (FL-SMC) have been found detrimental to surviving neural tissue and subsequent functional recovery (Kozlowski et al., 1996). Immediately after focal ablation of the FL-SMC, the nonimpaired forelimb was immobilized in a plaster-of-Paris cast, and remained immobilized for 2 weeks, forcing the animal to rely exclusively on the impaired forelimb. It was anticipated that this early, targeted therapy would prevent animals from learning to rely on the nonimpaired forelimb, which is normally what happens after FL-SMC injury (Jones and Schallert, 1992, 1994), thus enhancing use of the impaired forelimb. Rather than ameliorating functional deficits, however, this manipulation drastically worsened the outcome. Furthermore, the initial lesion was greatly enlarged (use-dependent exaggeration of cortical injury), compared with lesioned animals that were not forced to use the impaired forelimb (Fig. 5) (Kozlowski et al., 1996).
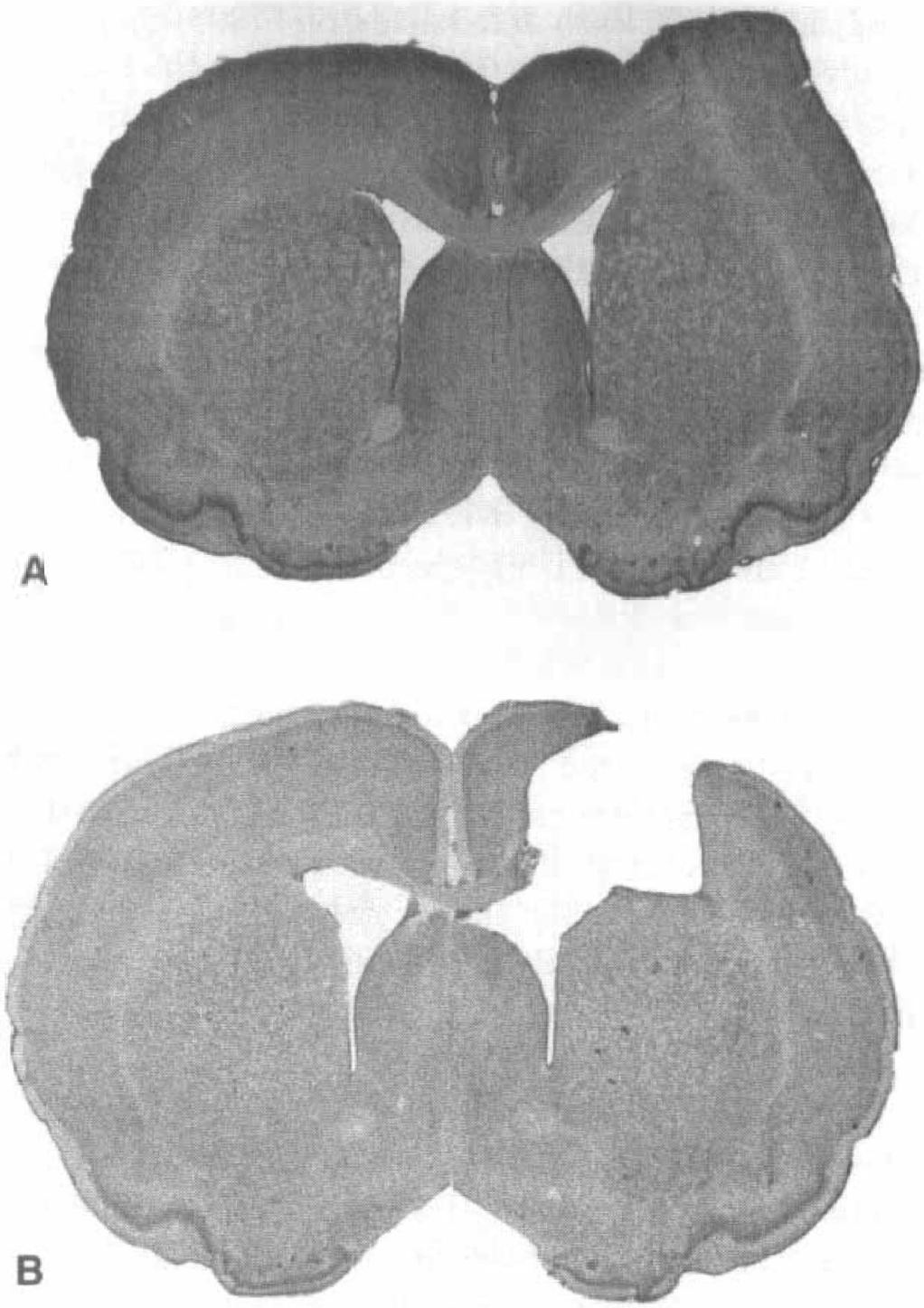
Use-dependent exaggeration of cortical injury
This overuse-dependent exaggeration of injury may involve glutamatergic activity because administering the glutamate antagonist (MK-801) during the intense motor experience reversed the effect and even enhanced outcome (Humm et al, 1999). If the ischemic injury is confined to the cortex, forced early use of the impaired forelimb after middle cerebral artery occlusion also can have an adverse outcome (Bland et al., 2000). Risedal et al. (1999) found that infarct size was exaggerated in rats with middle cerebral artery occlusion if they were exposed to a battery of various motor tests. However, functional deficits were not detected, perhaps because the extra motor training in the tests canceled the effects of the larger infarct.
If forced overuse is not begun until the second postinjury week, however, the injury does not expand (Humm et al., 1998; Risedal et al., 1999), although minor exaggeration of limb use deficits was found (Humm et al., 1998). Therefore, mild physical therapy measures begun several days or a week after injury seem less detrimental. For example, Colbourne et al. (1998) waited four days to begin mild behavioral training in gerbils with hypoxic–ischemic injury, and found no exaggeration of anatomic or functional deficits. Also, complex environments generally have a positive or neutral effect on motor outcome and do not worsen function on tests of motor noncompensation that are not confounded by practice or repeated testing.
Other research suggests that mild behavioral training early after central nervous system damage does not worsen functional outcome or increase the extent of the initial injury, but actually enhances motor recovery. For example, in the spinally-injured rat, mild treadmill or swimming activity for 20 minutes per day, beginning 4 days postinjury, did not exaggerate lesion size and lessened hindlimb sensory deficits (Hutchinson et al., 1999). Jones and colleagues (1999) began acrobatic training in cortically injured rats two days after the lesion. The training was performed in a gradual manner beginning with light training on the easier acrobatic tasks in the battery of tasks eventually given. Not only did the training not exaggerate injury, it increased the number of synapses formed per Layer V neuron in the homotopic cortex and enhanced functional recovery.
In summary, it is apparent that early, intense rehabilitative measures that target an impaired forelimb can be detrimental to anatomic and behavioral recovery. It seems that, in the rat, the third postinjury week may represent a “safer” time period in which to begin aggressive behavioral interventions. In contrast, mild or gradual motor rehabilitation can begin in the acute phase after brain injury leading to ameliorating chronic deficits.
EFFECTS OF POSTINJURY TESTING CONDITIONS ON FUNCTIONAL OUTCOME
Animal studies that assess functional outcome rely on the validity of the behavioral tests used to measure recovery. The necessity of sensitive behavioral measures specific to the deficits produced by the brain damage under study has been addressed elsewhere (Schallert et al., 2000a). Here, the authors discuss the effects that the postinjury environment may exact on functional and neural outcome.
It has been suggested that functional recovery is actually a “fragile” state that can be easily disrupted by environmental manipulation (Schallert, 1989). For example, hemidecorticated rats had apparently recovered from deficits in the orienting response as long as they were tested in the home cage. If, however, the home cage was left partially open during a trial, the response asymmetry reappeared (Schallert and Whishaw, 1984). Kolb and colleagues (1977) found that rats that had apparently recovered from the aphagia, adipsia, and sensorimotor impairment characteristic of bilateral orbital frontal cortical lesions would not eat, drink, or orient to sensory stimulation if they were tested in an apparatus that was larger than the home cage or not enclosed by four walls and a ceiling. The floor of the testing environment is also important. Schallert and Whishaw (1978) found that hypothalamically lesioned animals raised preoperatively in cages with a grid floor demonstrated less impaired feeding and movement initiation when tested on a grid surface than when tested on a surface covered with wood shavings. The reverse is also true, namely, that animals raised on wood shavings showed less behavioral impairment when tested on wood shavings, in comparison with their performance when tested on a grid floor (Schallert, 1989).
Other characteristics of the postinjury environment may influence outcome measures. Recent evidence from the authors' laboratory suggests that group housing may attenuate motor deficits that result from use-dependent exaggeration of brain injury. Previous studies investigating functional outcome after forced reliance on the impaired forelimb found pronounced deficits on a variety of behavioral tests (Kozlowski et al., 1996; Humm et al., 1998, 1999), and made use of singly housed subjects. Recently, it was found that group-housed animals that were forced to rely on the impaired forelimb after cortical injury showed characteristic exaggeration of the lesion, but did not have the exaggerated behavioral deficits previously observed (Leasure et al., 2000). Previous research indicates that social housing (without the other components of enriched environment) can enhance recovery of function after central nervous system injury. For example, social housing produced an enhancement of recovery from dorsal hippocampal lesions that was intermediate between that produced by EE and that observed in individually-housed rats (Einon et al., 1980). Furthermore, after medial preoptic area lesions, male rats that were socially housed after injury were able to copulate successfully, whereas rats that were housed alone showed a much lower copulatory success rate (Meisel, 1982). Thus, there is evidence that social housing can enhance recovery of cognitive function and sexual function; it may be that social housing also enhances recovery of motor function. Given the many situations that would arise in social housing that would encourage movement, it is not difficult to imagine that this sort of environment might provide brain-injured rats with the impetus to move, thereby using the impaired limb in an adaptive fashion and promoting “self-rehabilitation”. This finding may be particularly relevant to preclinical stroke studies in which motor function is the outcome measure, because housing condition may mask or enhance the effects of the drug that is administered.
Another facet of the postinjury environment that may be pertinent to stroke studies is repeated anesthetization by inhalable anesthetics. It has long been known that some anesthetic agents are neuroprotective when administered during ischemia or spinal trauma (Cole et al., 1989; Salzman et al., 1990, 1993; Warner et al., 1995; Miura et al., 1998). The authors recently have demonstrated that repeated postinjury anesthetization with halothane blocks use-dependent exaggeration of brain injury (Gotts et al., 2000). Because the route of administration of potentially neuroprotective agents after ischemia is frequently intracisternal injection, repeated anesthetization is required. Alternatively, if imaging techniques are used to visualize the extent of infarct, anesthetization is necessary. Researchers must be aware that intervention effects could be confounded by otherwise innocuous or transient pre-or postinjury experimental conditions (Schallert, 1989, 1982; Bruce-Keller et al., 1999).
COULD NEURAL STEM CELLS REPLACE NEURONS LOST TO DISEASE OR INJURY?
Neural stem cells exist in the adult mammalian brain and are capable of producing a constant supply of new neurons and glia (Gage, 2000). Once thought to be curiosity limited only to rodents, neurogenesis has been found in higher primates (Gould et al., 1999; Kornack and Rakic, 1999) and humans (Ericksson et al., 1998).
One region of the adult brain that contains a population of neural stem cells is the subependymal zone (SEZ), a thin layer of cells that surrounds the lateral ventricle. The main target of precursor cells generated in the SEZ appears to be the olfactory bulb (Altman, 1969; Luskin, 1993; Kishi, 1987). Recently, however, a primate study has been reported linking cells generated in the SEZ to targets in striate cortex and other regions involved in learning and memory (Gould et al., 1999). Therefore, it is possible that the function of adult neurogenesis is to provide cells to existing brain structures and circuits that require new ones, either because of natural turnover or learning on the part of the animal.
It is also possible that neurons generated in the adult brain migrate to areas of injury or neurodegeneration in an attempt to replace dead neurons or maintain functional circuitry. In support of this idea, there is evidence to indicate that the SEZ responds to injury by increasing its proliferative capacity (Willis et al., 1976; Weinstein et al., 1996; Szele and Chesselet, 1996), FGF-2 expression (Schallert et al., 2000b), and metabolic activity (Valla et al., 1999) (Fig. 6).
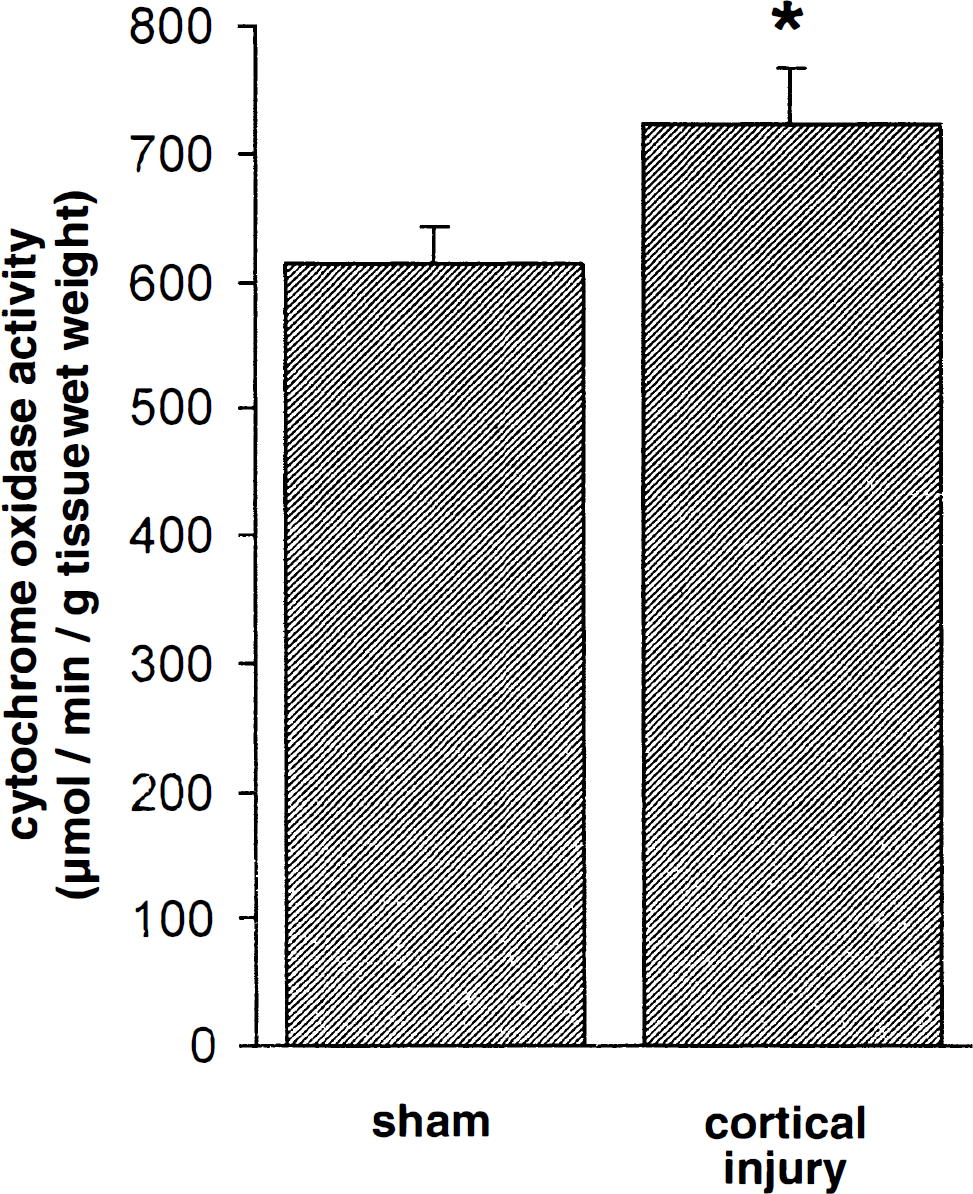
Injury to the cortex increases metabolic activity in the subependymal zone. * P < 0.02 SEZ of lesioned animals significantly more metabolically active than that of shams.
Recently, Magavi and colleagues (2000) provided evidence that neural progenitor cells replace layer VI corticothalamic projection neurons lost because of induced apoptotic degeneration in mice. Replacement cells appeared to originate from the cortex itself and progenitors within the SEZ, and expressed markers for migrating immature and mature neurons. These results suggest that endogenous precursor cells may serve as a reservoir of neurons that can respond to neurodegeneration.
That the SEZ may replace neurons lost to neurodegeneration is supported by another observation, namely, that hyposmia is an ubiquitous consequence of neurodegenerative disease, including Parkinsons disease (Ansari and Johnson, 1975; Doty et al., 1988, 1989), Alzheimer's disease (Serby, 1986; Doty et al., 1987), Korsakoffs syndrome (Gregson et al., 1981; Jones et al., 1975, 1978), Downs syndrome (Warner et al., 1988; Hemdal et al., 1993; Zucco and Negrin, 1994), and Huntingtons disease (Moberg et al., 1987). This fact is interesting if one considers that the known function of the SEZ is to provide cells for the olfactory bulb. What if the SEZ also provides cells or trophic support for areas of the brain that are undergoing persistent and continuous neurodegeneration? It is possible that newly born cells from the SEZ are being re-routed from the rostral migratory stream, and migrating instead to the area of degeneration. This would explain why hyposmia is so common a symptom of neurodegenerative disease. In Parkinson's disease, the olfactory deficit is one of the earliest symptoms to appear. It is at least conceivable that the SEZ may be partially co-opted to respond to ongoing nigrostriatal degeneration even before motor symptoms are detectable. If the SEZ functions to promote plasticity or replenish cells lost to neurodegenerative disease, it could also do so when cells are lost in a more rapid fashion, such as traumatic brain injury or ischemia.
There are two lines of evidence suggesting that the SEZ can stimulate the production of new cells after injury. First, in the course of studying rats with restricted lesions of the midline frontal region in infancy, the authors discovered that if lesions were made at approximately 10 days of age, there was a massive upregulation of SEZ activity (Kolb et al., 1998a). These cells migrated to the site of lesion, differentiated into neurons and glia, and formed some, but not all, of the expected connections of this region. To demonstrate that this tissue was functional, the authors either removed the regrown region in adulthood (Dallison and Kolb, unpublished data) or blocked the SEZ activity by pretreating animals at E13 with the mitotic marker bromodeoxyuridine (BrdU) (Kolb et al., unpublished data). Second, in vitro studies have shown that cells taken from the subventricular zone can be stimulated by epidermal growth factor (EGF) or FGF-2 to give rise to neurons (Reynolds and Weiss, 1992). Detection of the mRNA for the EGF receptor has led to the hypothesis that an EGF-responsive precursor cell may play a role in forebrain neurogenesis in vivo. This idea has been supported by studies showing that infusion of EGF into the mouse ventricle increases the population of dividing SEZ cells up to 17-fold (Craig et al., 1996). Craig et al. also reported that 7 weeks after 6 days of EGF infusion there is a subpopulation of new immunohistochemically-identified neurons and glia. A logical extrapolation of the Craig study was to infuse EGF into the lateral ventricles of normal rats. Like Craig et al. (1996) study in mice, the authors found that 7 days of EGF infusion into the lateral ventricle of normal brain increases SEZ size dramatically and produces cells that express nestin (Kolb et al., 1998b).
Expression of nestin is significant because mammalian neuronal precursor cells can be identified by the specific expression of the intermediate filament, nestin (Frederiksen and McKay, 1988). The nestin-positive cells were found especially in the SEZ and along the rostral migratory pathway to the olfactory bulb. Next, the authors made bilateral medial frontal lesions and then infused EGF into the ventricle for 7 days. When the animals were killed 2 weeks later the lesion cavity was filled with cells (Kolb et al., 1998b). Immunohistochemical analyses of these cells found BrdU-positive cells that were nestin-or GFAP- (an astrocytic marker) positive, but there were no neurons. Although the authors do not know where these cells originated, the SEZ was enlarged even more than in the normal EGF-stimulated brain, implying that the SEZ was the source. In another series of animals, the authors made hemidecortications and administered EGF (Fig. 7). Again, there was a massive proliferation of undifferentiated cells that partially filled the cavity. Histologic analyses showed that the striatum on the injured side was markedly enlarged and filled with undifferentiated cells that the authors presume were migrating to the brain surface. In a final series of animals, the authors varied survival time after frontal lesions or hemidecortications. Animals killed 1 week after injury showed a massive proliferation of cells, but the number of surviving cells declined over time so that by 1 month at least 50% of the cells are gone. Thus, there are two problems to solve: cell differentiation and cell survival. They may, of course, be related problems. The key point, however, is that it does appear that the SEZ may be a source of cells that can contribute to functional recovery.
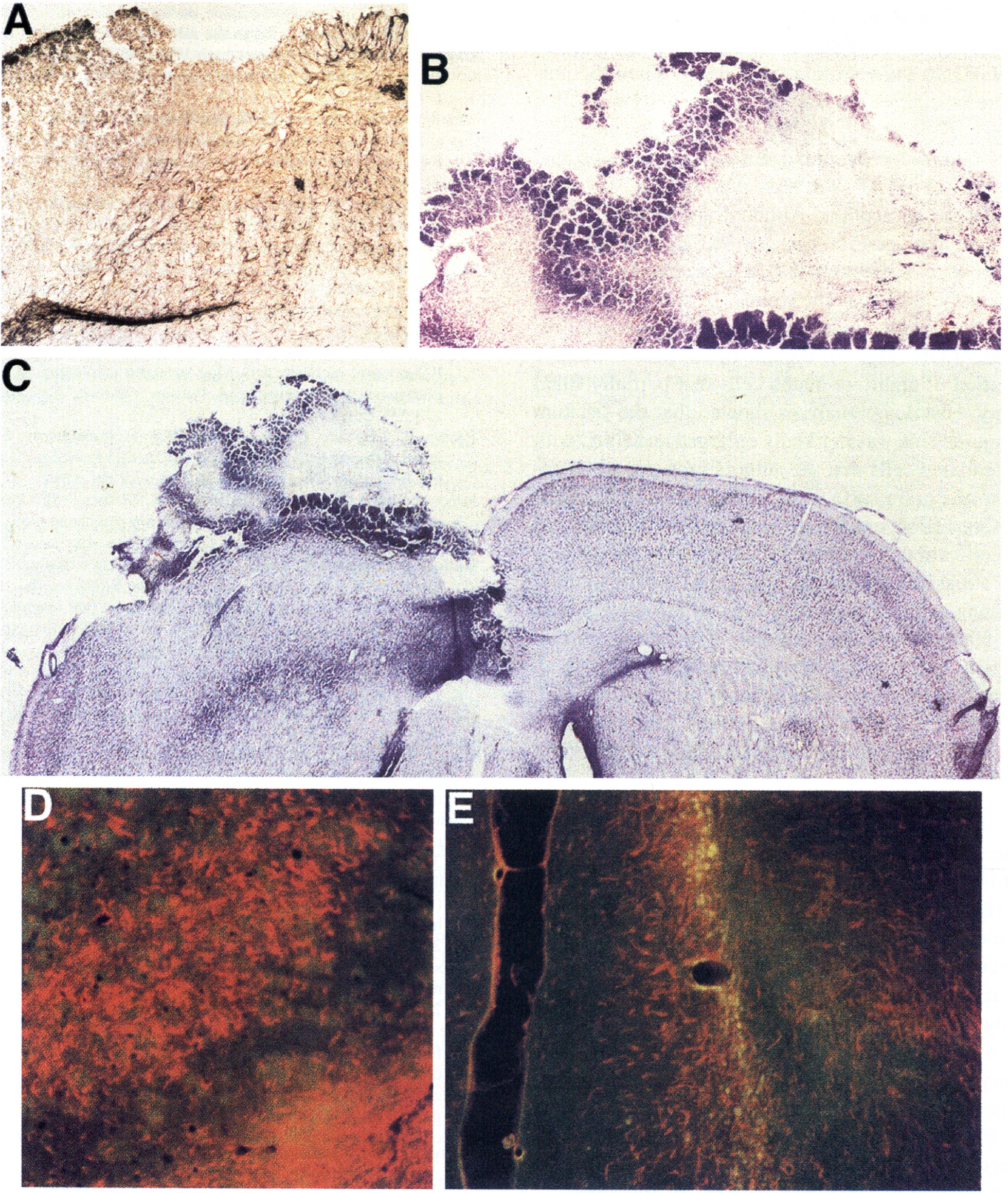
Photomicrographs of the EGF-stimulated cell generation in a rat with a hemidecortication and a postoperative survival of two weeks. New cell growth in the lesion hemisphere is clearly visible on top of the striatum.
CONCLUSION
There is ample evidence to support the idea that the injured brain is fertile ground for structural plasticity to occur. However, behavior is often necessary for certain neural events to be initiated and can dramatically shape the structural changes and the degree and rate of recovery. It may not be sufficient simply to administer therapeutic agents such as drugs or stem cell transplants after trauma to the central nervous system. Rather, it may be necessary to combine such therapeutic agents with physical interventions to actively engage plasticity mechanisms. Finally, the timing and magnitude of therapy, combined or otherwise, is an important consideration when attempting to maximize recovery of function.
