Abstract
Thrombospondins 1 and 2 (TSP-1/2) belong to a family of extracellular glycoproteins with angiostatic and synaptogenic properties. Although TSP-1/2 have been postulated to drive the resolution of postischemic angiogenesis, their role in synaptic and functional recovery is unknown. We investigated whether TSP-1/2 are necessary for synaptic and motor recovery after stroke. Focal ischemia was induced in 8- to 12-week-old wild-type (WT) and TSP-1/2 knockout (KO) mice by unilateral occlusion of the distal middle cerebral artery and the common carotid artery (CCA). Thrombospondins 1 and 2 increased after stroke, with both TSP-1 and TSP-2 colocalizing mostly to astrocytes. Wild-type and TSP-1/2 KO mice were compared in angiogenesis, synaptic density, axonal sprouting, infarct size, and functional recovery at different time points after stroke. Using the tongue protrusion test of motor function, we observed that TSP-1/2 KO mice exhibited significant deficit in their ability to recover function (
Introduction
New or recurrent stroke is a devastating disease affecting 700,000 people in North America alone every year (Thom et al, 2006). After a stroke, clinical deficits, such as motor paralysis, weakness, sensory numbness, and incoordination, are often observed in the upper and lower extremities contralateral to the injury. While the impaired limb could be permanently affected, significant recovery takes place during the first several weeks after the injury and lasts for at least 6 months, with the ultimate outcome, mainly a function of initial severity (Duncan, Lai and Keighley, 2000). Although part of the recovery process most likely involves resolution of pathophysiologic events caused by ischemic brain injury, many studies have provided substantial evidence that regenerative processes contribute toward the restitution of function (Nudo, 2007). Such processes include dendritic arborization among neurons spared by ischemic injury, the reorganization of existing synaptic structures, and regeneration of synapses.
Thrombospondins (TSPs) are large extracellular matrix proteins that mediate cell-cell and cell-matrix interactions by communicating with an array of membrane receptors, other extracellular matrix proteins, and cytokines (Adams and Lawler, 2004). There are 5 known TSPs, each encoded by a separate gene. Thrombospondin-1 and 2 have been shown to be involved in platelet aggregation, inflammation, and angiogenesis (Adams and Lawler, 2004). After ischemic injury in rats, TSP-1 and TSP-2 are upregulated in the affected area, and this robust expression was postulated to drive the resolution of postischemic angiogenesis through their antiangiogenic properties (Lin et al, 2003). Recently, however, TSP-1 and TSP-2 have been found to promote synaptogenesis
Materials and methods
Surgical Methods
Experimental protocols were approved by the Stanford University Administrative Panel on Laboratory Animal Care. Eight to 12–week-old FVB wild-type (WT) and FVB mice with homozygous deletion of the
RNA Extraction and qPCR Analysis
After permanent unilateral occlusion of the dMCA and the CCA, WT FVB mice were killed at days 1, 7, 14, and 28 after stroke (
Primers for quantitative real-time PCR (qPCR) was obtained from PrimerBank (Wang and Seed, 2003) and synthesized (Invitrogen, Carlsbad, CA, USA). They were screened for the generation of a single amplification product using gel electrophoresis, for PCR efficiency using standard curve analysis, and for the absence of primer—dimer formation using melt-curve analysis (Halstenberg et al, 2005). The primers for
Quantitative polymerase chain reactions were run in the Mx3000P QPCR System (Stratagene, La Jolla, CA, USA) using SYBR Green chemistry (Brilliant SYBR Green QPCR Master Mix, Stratagene). All reactions were performed in triplicate using the following conditions: 10 mins at 95°C; then 40 cycles of 30 secs at 95°C, 1 min at 60°C, and 30 secs at 72°C. This was followed by melting curve analysis using a temperature range from 55°C to 95°C.
Mx3000P software was used to calculate the threshold cycle for each reaction, and the average threshold cycle (
Tissue Preparation
On days 1, 7, 14 (WT,
Immunohistochemistry
Free-floating slices of 25 μm thick were washed three times in PBS, and blocked in PBS buffer containing 3% donkey serum (Jackson ImmunoResearch, West Grove, PA, USA) and 0.1% Triton X-100 (Sigma, St Louis, MO, USA) for 1 h. For synaptic staining, primary antibody solution was added consisting of mouse anti-synaptophysin (1:500, Sigma), rabbit anti-PSD-95 (1:250, Zymed Laboratories, South San Francisco, CA, USA), and anti-glial fibrillary acidic protein (GFAP) (1:500, Advanced Immunochemical Inc., Long Beach, CA, USA). Slices were incubated in primary antibody for three nights at 4°C, washed three times in PBS and 0.6% Triton X-100, and incubated with Cy3-conjugated donkey anti-mouse (1:500), FITC-conjugated donkey anti-rabbit (1:500), and Cy5 donkey anti-guinea pig (1:500) (Jackson ImmunoResearch, West Grove, PA, USA). After incubation in secondary antibodies for 3 h, sections were washed, incubated with 4′-6-diamidino-2-phenylindole for 5 mins, and mounted onto slides. To localize TSP expression, sections were stained with either a mouse monoclonal TSP-1 antibody (sc-59887, 1:200; Santa Cruz Biotechnology, Santa Cruz, CA, USA) or a mouse monoclonal TSP-2 antibody (sc-12313, 1:200; Santa Cruz Biotechnology). To verify TSP-2 antibody specificity, TSP-2 expression was also examined in a GENESAT transgenic mouse line that expresses green fluorescent protein under the TSP-2 promoter (Gong et al, 2003). Colocalization experiments for TSP expression were carried out with S100 (1:200, Novus Biologicals, Littleton, CO, USA) and GFAP (1:200, Advanced Immunochemical, Inc.).
Confocal Analysis of Synapse Number
Images of immunostained brains were collected on a Leica LSM 510 Meta confocal microscope. Optical sections were line-averaged and collected at 0.28 μm intervals from the penumbral and homotopic contralateral cortices. Gain thresholds and amplitude offsets were kept constant between imaging of the penumbral and homotopic contralateral areas for each section. Stacks of 10 optical sections were quantified for synapse number by projecting a series of 5 optical sections, and counting the number of synapses in each projection volume. Synaptic puncta were automatically counted using the Image J puncta analyzer program in TSP-1/2 KO mice (
Blood Vessel Density Quantification
Four weeks after unilateral occlusion of the dMCA and the CCA, WT (
Biotinylated Dextran Amine Tracing of Axonal Sprouting
Four weeks after unilateral occlusion of the dMCA and the CCA, WT (
Behavioral Assay
To compare the rate of functional recovery after unilateral occlusion of the dMCA and the CCA between WT (
Statistical Analysis
Data analysis was performed with the statistic software package PRISM (GraphPad, San Diego, CA, USA). All means are presented ± s.e.m. All statistical analyses were performed with the use of analysis of variance followed by
Results
Thrombospondin-1/2 are Upregulated in the Ischemic Penumbra
Previous studies have shown that TSP-1/2 are upregulated after stroke in rats (Lin et al, 2003). To verify whether a similar upregulation was present in our mouse stroke model, we investigated the levels of TSP gene expression with qPCR. Polymerase chain reaction amplification of TSP-1 or TSP-2 each produced a single cDNA fragment when visualized with ethidium bromide on agarose gel and qPCR melt-curve analysis resulted in a single melting temperature, thus confirming the specificity of the primer pairs used. Compared with the expression level of sham-operated animals (
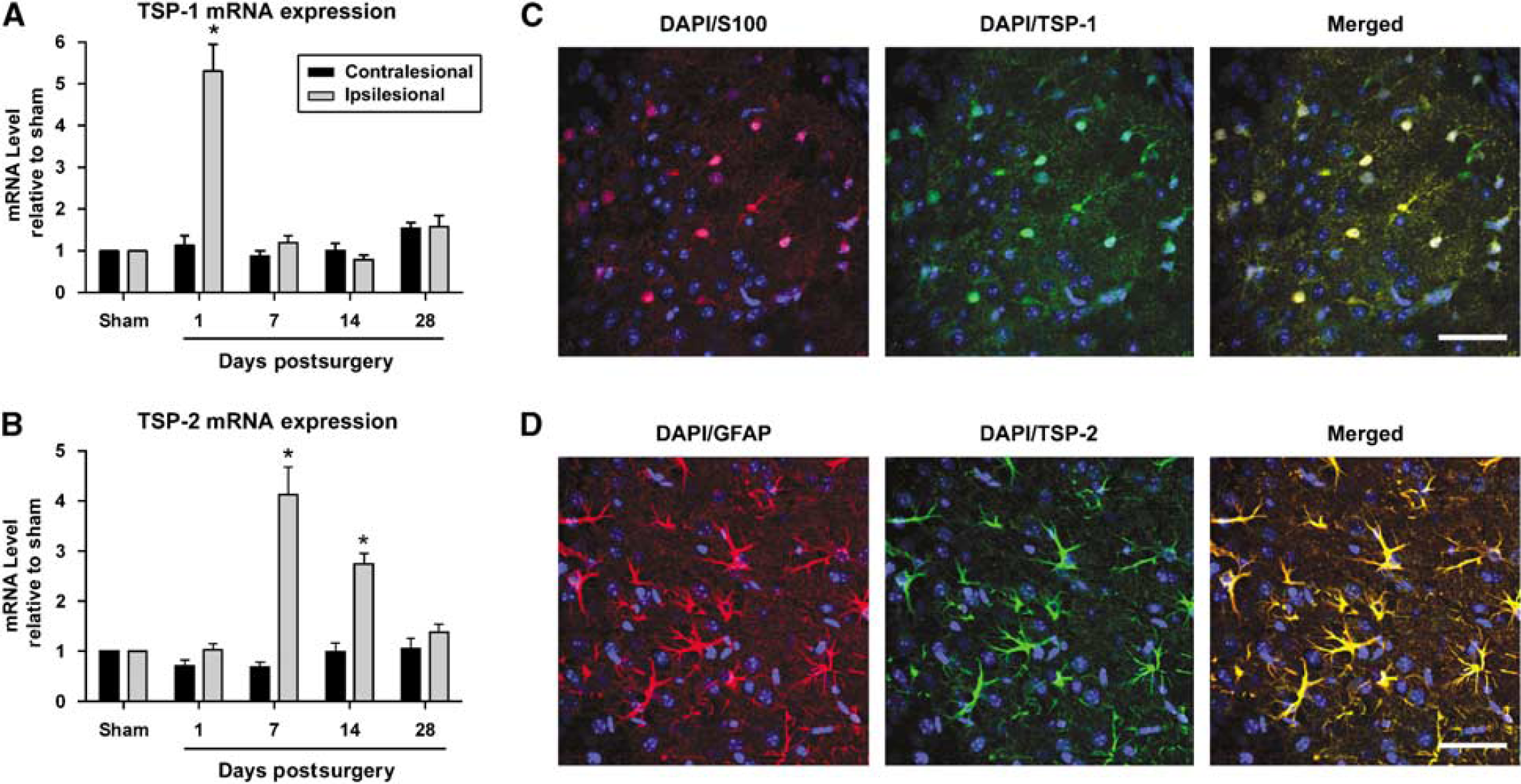
Thrombospondin-1 and 2 mRNA and protein expression are increased after stroke. (
Loss of Thrombospondin-1/2 does not Affect Blood Vessel Density after Cerebral Ischemia
Given the TSP's role as angiostatic factors, we investigated whether microvascular density after unilateral occlusion of the dMCA and the CCA would be affected by the loss of TSP-1/2. We compared microvascular densities at day 28 between TSP-1/2 KO and WT mice. The ratio of blood vessel density in the ischemic penumbra relative to that in the contralateral cortex in TSP-1/2 KO mice did not differ significantly from that found in WT mice. The average normalized blood vessel density was found to be 0.8149 ± 0.06 for WT and 0.8357 ± 0.03 for TSP-1/2 KO (
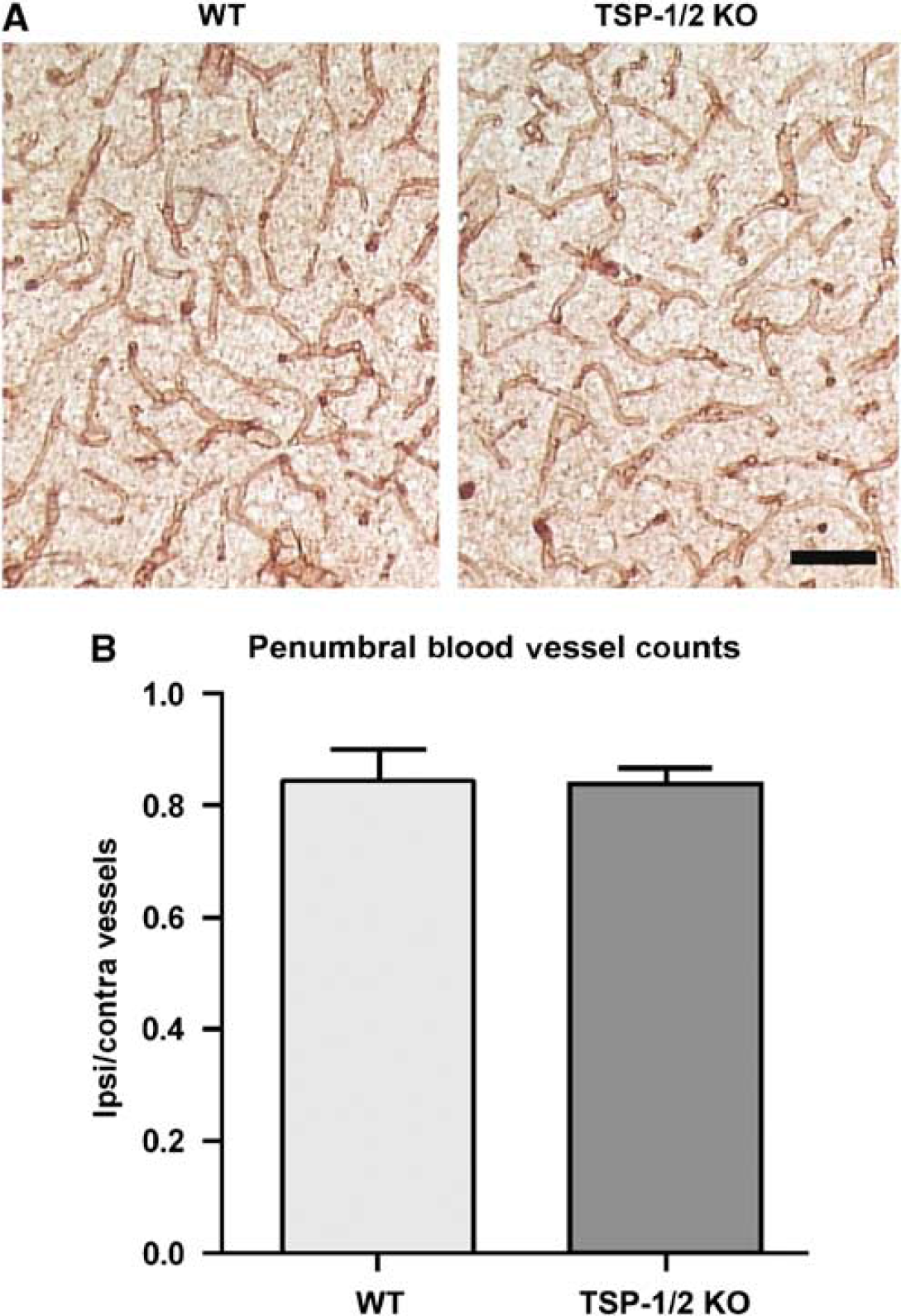
Thrombospondin-1/2 KO mice do not show deficit in blood vessel density after stroke. (
Synaptic Density Recovers Gradually in the Ischemic Penumbra after Stroke
To determine whether there are synaptic changes in WT mice following our model of stroke, synaptic density in the ischemic penumbra was examined at 1, 7, 14, 21, and 28 days after stroke in WT mice. Regions with interspersed GFAP-positive reactive astrocytes bordering a GFAP-negative core were identified as ischemic penumbra. Synaptic puncta, defined by a colocalization of presynaptic synaptophysin and postsynaptic PSD-95, were quantified. The ratio of synapses in the ischemic penumbra and the corresponding nonischemic contralateral cortex was calculated to correct for any endogenous differences. We defined this ratio as the synaptic density in the ischemic penumbra. After unilateral occlusion of the dMCA and the CCA, the synaptic density at day 7 was 68%, which is significantly lower than baseline at nearly 100% (
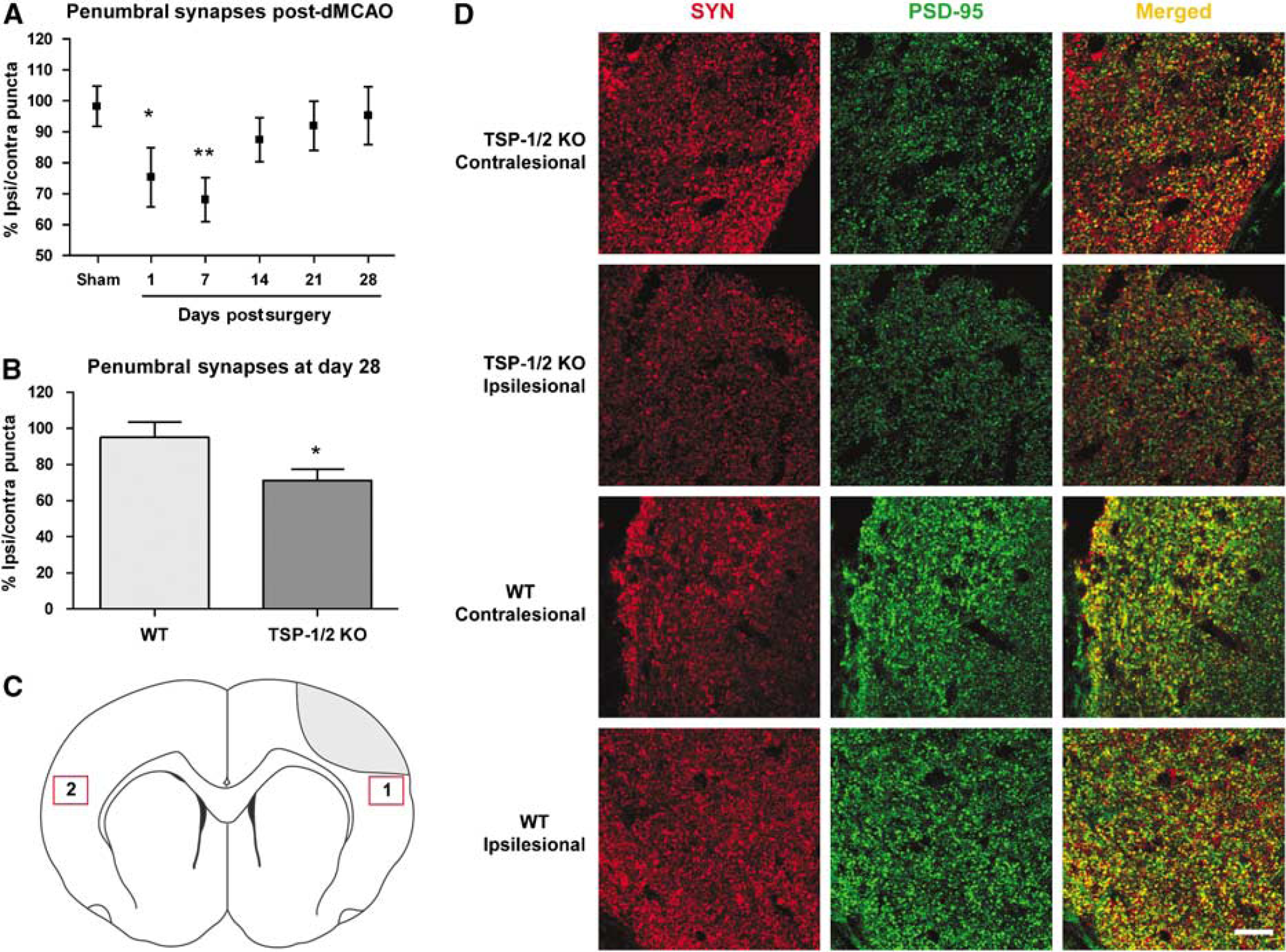
Thrombospondin-1/2 KO mice show impaired synaptic recovery after stroke. (
Loss of Thrombospondin-1/2 Impairs Synaptic Recovery after Stroke
Because TSP-1/2 have been shown to mediate synaptogenesis during development, we investigated the effect of a TSP-1/2 deficiency on the recovery of synapses after stroke. We quantified synapses in the ischemic penumbra and compared synaptic density in the ischemic penumbra between WT and TSP-1/2 KO mice 4 weeks after stroke. Because TSP-1/2 KO mice had been shown to have fewer synapses compared with WT mice (Christopherson et al, 2005), we corrected for this difference by defining a normalized synaptic density as the ratio of synaptic density in the ischemic penumbra to that in a homotopic area of the contralesional cortex. In TSP-1/2 KO mice, synaptic density in the ischemic penumbra was significantly reduced compared with that in WT mice at day 28 after stroke. Wild-type mice had a mean of 95.2 ± 8.2% of synapses whereas TSP-1/2 KO mice had a mean of 71.1 ± 6.2% (
Loss of Thrombospondin-1/2 Impairs Axonal Sprouting after Stroke
It had been previously shown that following stroke, there is an increase in axonal sprouting from the contralesional cortex toward the ischemic penumbra and striatum on the other side of the brain (Carmichael and Chesselet, 2002). To determine whether TSP-1/2 KO mice exhibited a deficit in axonal sprouting, we compared the density of axonal projections from the contralesional cortex found in the ischemic penumbra at 5 weeks after stroke. We injected BDA, an anterograde axonal tracer, into a homotopic area in the contralesional cortex and quantified the density of BDA-labeled axons in the ipsilesional ischemic penumbra and striatum. Although the infarct produced by this model did not extend into the striatum, we wanted to examine axonal sprouting in the striatum because a large portion of contralateral input projects in both the ipsilesional penumbra and striatum (Carmichael and Chesselet, 2002). To control for the efficiency of BDA uptake, we normalized the density of BDA-labeled axons in the ipsilesional penumbra and striatum to that of decussating axons in the corpus callosum (Figure 4C). This was defined as the axonal sprouting density. Compared with WT mice (
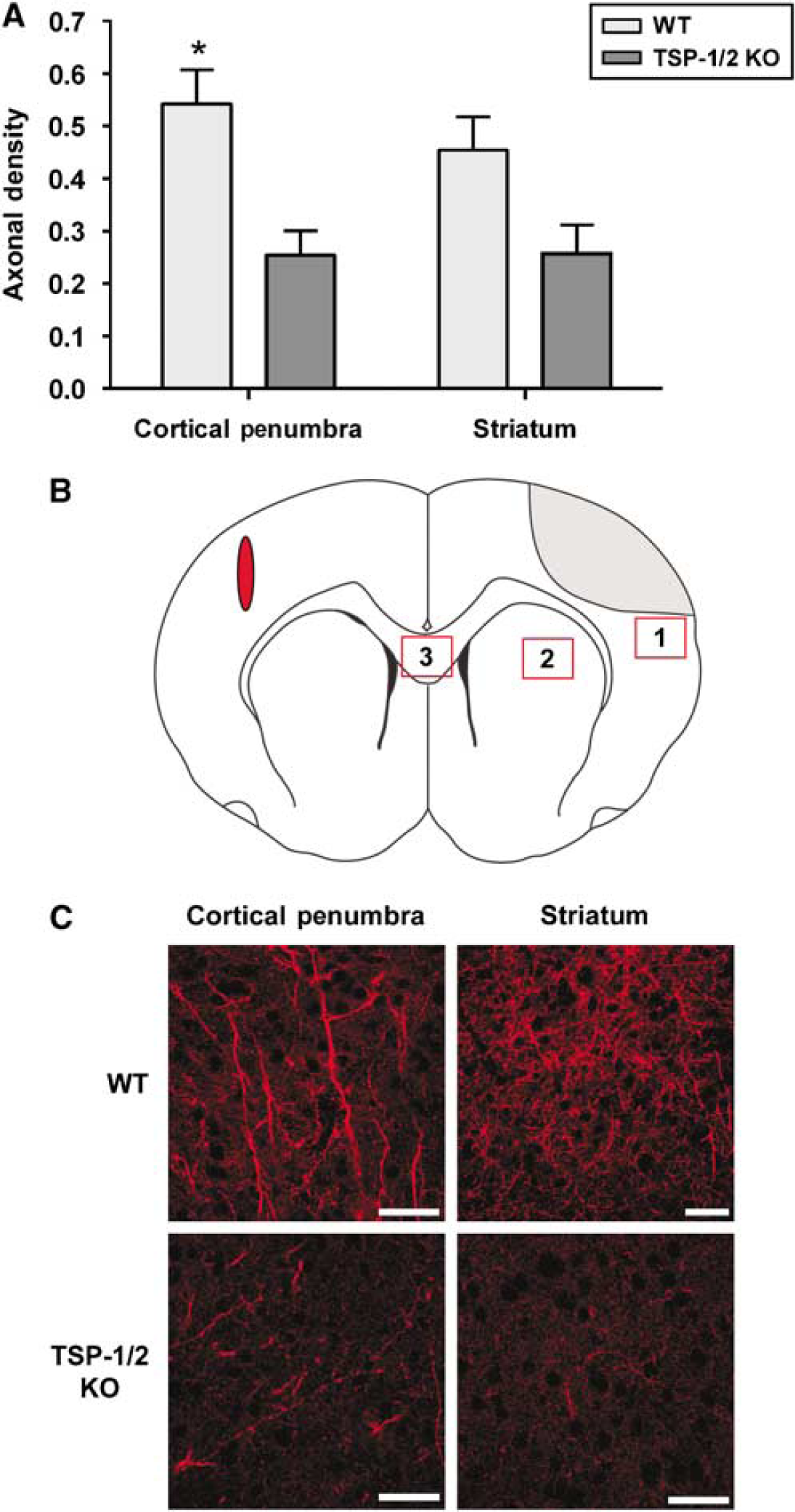
Thrombospondin-1/2 KO mice show impaired axonal sprouting from the contralesional cortex toward the ischemic penumbra after stroke. (
Loss of Thrombospondin-1/2 Impairs Behavioral Recovery after Stroke
Previous studies have postulated that functional recovery may involve a gradual regeneration and reorganization of synaptic structures (Ito et al, 2006; Nudo, 2007). To determine whether TSP-1/2 play a role in functional recovery, we compared performance in various behavioral tests between WT (
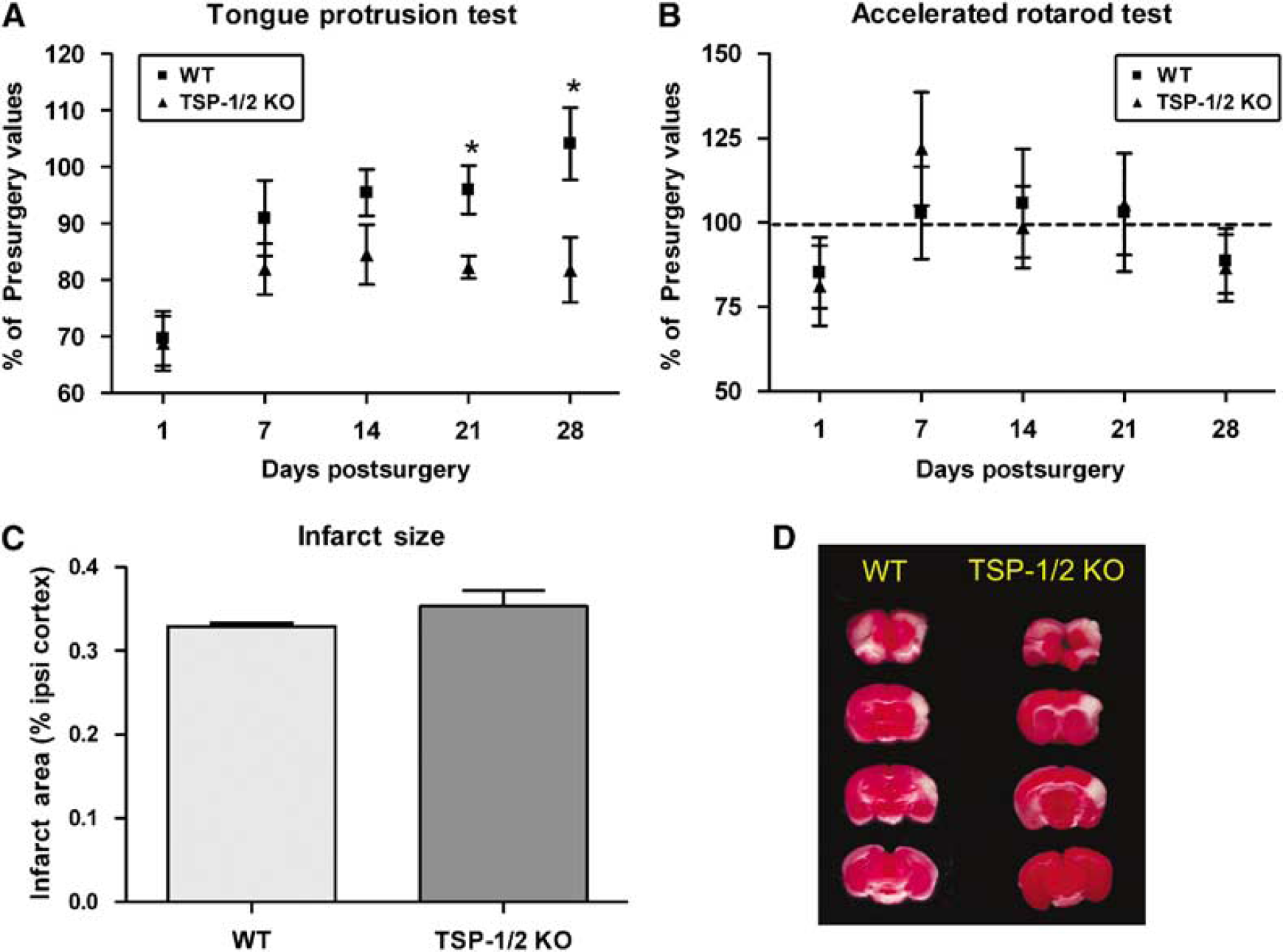
Thrombospondin-1/2 KO mice exhibit deficits in functional recovery after stroke in the tongue protrusion test. Performance on the tongue protrusion test (
It is possible that the differences in behavioral recovery could be attributed to differences in the severity of tissue infarction after unilateral occlusion of the dMCA and the CCA. To investigate this possibility, we compared the infarct volumes 2 days after unilateral occlusion of the dMCA and the CCA in both groups of mice. No differences were found in infarct volume between WT mice (
Discussion
In the present study, we showed that the rate of synaptic recovery and axonal sprouting was significantly decreased in the ischemic penumbra of TSP-1/2-deficient mice, and this deficiency in structural plasticity correlated with the recovery of tongue protrusion, a test of behavioral recovery. This correspondence in synaptic reorganization and axonal sprouting with behavioral recovery suggests that there exists a TSP-1/2-mediated process of structural plasticity, which is required for postischemic motor recovery. Although other studies have shown the role of TSP in wound healing (Agah et al, 2002), nerve outgrowth (Hoffman and O'Shea, 1999), and synaptogenesis during central nervous system development (Christopherson et al, 2005), this is the first study to show that the loss of TSP impairs motor recovery after focal cerebral ischemia in the adult animal. Consistent with the observations of Christopherson et al (2005) during development, we did not observe a difference between WT and TSP-1/2 KO mice in the levels of two synaptic proteins, synaptophysin and PSD-95, by western blots 4 weeks after stroke (data not shown). This supports the hypothesis that TSPs are needed for inducing and maintaining synaptic alignment and adherence of pre and postsynaptic specializations during synaptogenesis rather than enhancing the expression of synaptic proteins.
Cortical injury that occurs after stroke has been found to impair the execution of muscular contraction and affect fine manipulative abilities, such as lingual coordination. In the weeks and months after injury, a gradual recovery of some motor abilities is observed and the extent of this process is determined by both the severity of the injury and the endogenous plasticity that facilitates the process (Nudo, 2007). To assess the extent of functional recovery in a focal model of ischemia in mice, we used an extensive battery of behavioral tasks sensitive to motor disturbance after ischemia. To facilitate the quantification of synapses in the ischemic penumbra, we used a focal model of ischemia that results in a distinct border between ischemic and nonischemic regions. This strategy also prevents the complication of contralesional hypoxia, which would affect axonal sprouting from the contralesional cortex. The drawback of such a strategy is that the infarct area produced by experimental stroke is relatively small, which may fail to result in observable behavioral deficits. In our model, the dMCA and the CCA were permanently occluded. It is possible that reperfusion injury may play a role in causing more observable behavioral deficits. Indeed, in three of the four behavioral tests we used, including the accelerating rotarod test, the corner test, and the cylinder test, we could not detect (as a group) a deficit in performance at either 1 day or longer after stroke. However, using the tongue protrusion test, we observed a sustained deficit lasting several weeks. We attribute the success of the tongue protrusion test to a relatively small lesion necessary to induce a deficit in tongue function, as compared with the larger lesion required to affect gross motor function in the rotarod, corner, and cylinder tests. In an ischemia model of rat, the tongue protrusion test has been shown to correlate with infarct volume and neurologic scores (Gulyaeva et al, 2003). In addition, lingual discoordination such as tongue palsy and dysarthria has often been observed following stroke in human patients (Daniels, Brailey and Foundas, 1999). Importantly, we were able to show that TSP-1 and TSP-2 KO mice exhibited a delayed rate of recovery relative to WT mice, suggesting that TSP-1/2 may mediate molecular processes required for the recovery of motor function.
Spontaneous functional recovery in stroke patients has been associated with the remapping of cortical function and the redistribution of neuronal activity (Nudo, 2007). This cortical plasticity is attributable to various mechanisms, such as enhanced dendritic growth, synaptogenesis, axonal sprouting, and the increased production of growth factors. Here we show that the loss of TSP-1/2 results in a significant impairment of synaptogenesis and axonal sprouting in the ischemic penumbra after stroke. During development, it has been shown that TSP-1/2 are secreted by astrocytes and the absence of TSP-1/2 results in the reduction of synapses in the brain (Christopherson et al, 2005). After ischemia, TSP-1/2 protein expression has been shown to colocalize with markers for astrocytes and endothelial cells (Lin et al, 2003). We found that TSPs are, in part, secreted by astrocytes after stroke, as evident by the colocalization of TSP-1 and TSP-2 with S100 and GFAP, respectively, and thus may play a role in regenerative processes. The temporal pattern of TSP-1/2 mRNA upregulation after ischemia also correlates with the gradual pattern of nerve growth-promoting gene expression seen after stroke, which stabilizes around week 4 (Carmichael et al, 2005). In our model, TSP-1 mRNA expression increased at day 1 after stroke, whereas TSP-2 mRNA expression increased at weeks 1 and 2. This pattern of gene expression also correlates with the temporal profile of synaptic recovery that we observed, namely a decrease in synaptic density at days 1 and 7 after ischemia, followed by gradual recovery over the next several weeks. This result is consistent with electron microscope synaptic profiling performed after stroke in gerbil brains (Ito et al, 2006).
In addition, TSP expression has been observed to dramatically increase along the path of regenerating axons after optic nerve crush in the goldfish (Hoffman and O'Shea, 1999). Thrombospondin has been shown to promote neurite outgrowth (Osterhout, Frazier and Higgins, 1992) and may help stabilize the extracellular matrix by interacting with other neurite-promoting factors, such as laminin and fibronectin (Lahav, Lawler and Gimbrone, 1984; Mumby, Raugi and Bornstein, 1984). In cerebral ischemia, the axonal sprouting response from the contralesional cortex to the ischemic penumbra and ipsilesional striatum is characterized by spontaneous, synchronized waves of neuronal discharges in the peri-infarct and contralateral cortical areas and by the upregulation of a number of axonal growth-promoting genes (Carmichael, 2003, 2005). Because we have shown that TSP-1/2 KO animals are deficient in axonal sprouting and have a reduction in synapses, it is conceivable that one of the mechanisms contributing to impaired synaptic density is a reduction of axonal sprouting from contralesional inputs. Thrombospondin-1/2 may interact with matrix proteins and facilitate axonal sprouting, which may eventually result in synaptic and behavioral recovery.
It is well known that angiogenesis often takes place in the ischemic penumbra after cerebral ischemia (Hayashi et al, 2003), and the number of new vessels and thus the extent of angiogenesis are correlated with longer survival in patients with stroke (Krupinski et al, 1994). Both TSP-1 and TSP-2 have been shown to exert antiangiogenic activities (Streit et al, 1999a, 1999b), and their upregulation after ischemia in rats has been postulated to regulate angiogenesis (Lin et al, 2003). The present study shows that there was no significant difference in blood vessel density between WT and TSP-1/2 KO mice at day 28 after ischemia. However, it is still possible that TSP-1/2, by serving as angiostatic factors, may affect the initial rate of postischemic angiogenesis but may not affect the final extent of angiogenic regrowth. Nevertheless, in a skin excision wound model of injury, it was found that while TSP-2 knockout mice showed an increased vascular density, neither TSP-1 knockout nor TSP-1 and TSP-2 double knockout mice showed a difference in vessel density compared with WT mice at days 7 and 14 after injury (Agah et al, 2002). Our finding that there was no significant difference in blood vessel density at 4 weeks after cerebral ischemic injury parallels the findings in the skin excision wound model. Moreover, there has been no direct experimental evidence supporting the idea that angiogenesis and its sequelae lessen tissue damage (Hayashi et al, 2003). This finding lends further credence to the importance of neural plasticity in mediating postischemic motor recovery.
Many agents that induce cortical plasticity have been shown to promote functional recovery after ischemia. For example, rats subjected to permanent ischemia and treated with
Footnotes
Acknowledgements
We thank Kevin Choo, Jason Shen, Angeline Jocson, Vaughan Tuohy, Philip Chui, and David Kunis for technical help; Beth Hoyte for figure preparation; and Dr Bruce Schaar for critical reading of the manuscript.
