Abstract
The glucose concentration in the culture medium may affect the energy metabolism of cultured cells. The oxidative metabolism of glucose in astrocytes might also be affected because the glucose concentration (25 mmol/L) of many culture media is higher than the physiological levels (∼3 mmol/L). In the present study, we assessed the effects of reducing the glucose concentration in the culture medium on the oxidative metabolism of glucose in cultured rat astroglia by measuring the oxidation rates of L-[U-14C]lactate or D-[U-14C]glucose to 14CO2. The effects of D-aspartate and elevated extracellular K+ levels on oxidative and glycolytic metabolism in astroglia were also investigated. The rates of [14C]lactate and [14C]glucose oxidation in astroglia cultured in a medium containing 2 mmol/L of glucose (astroglia2) were approximately twofold of those in astroglia cultured in a medium containing 22 mmol/L of glucose (astroglia22). D-Aspartate (500 μmol/L) significantly increased [14C]lactate oxidation by 156% in astroglia22 and by 83% in astroglia2. D-[U-14C]glucose oxidation in astroglia22 and astroglia2 was also increased by 94% and 76%, respectively. In contrast, an elevated extracellular K+ concentration (7.4 mmol/L) did not affect glucose and lactate oxidation, although it increased 2-deoxy-D-[1-14C]glucose phosphorylation. Astroglia grown in the physiological glucose concentration are more dependent on the oxidative metabolism of glucose than that in high-glucose concentration. Glucose concentration in culture medium has a strong influence on astrocytic oxidative capacity in vitro.
Introduction
The energy demand arising in astrocytes during neuronal excitation has been hypothesized to be supplied by glycolysis, and the lactate produced and released from the astrocytes is thought to be shuttled to neurons to support neuronal oxidative metabolism (Magistretti et al, 1999). However, reports from other laboratories disagree with this concept (Hertz et al, 1998b; Chih and Roberts, 2003; Dienel and Cruz, 2004; Hertz, 2004). Based on the hypothesis of lactate shuttling between neurons and astrocytes, neurons have been suggested to have a larger capacity for oxidizing lactate and glucose to CO2 than astrocytes (McKenna et al, 2001; Itoh et al, 2003). Other reports have shown that astrocytes and neurons possess equivalent rates of lactate oxidation (Vicario et al, 1993; Peng et al, 1994).
Regarding brain energy metabolism in vivo, care should be taken when evaluating results obtained from cultured cells in vitro because the culture condition may influence the outcome. The different results obtained by laboratories might be ascribed to the glucose concentration in the culture medium (Hertz et al, 1998a). The glucose concentration of extracellular fluid in rat brain is generally accepted to be approximately 0.5 to 3.3 mmol/L (McNay and Gold, 1999). Many in vitro studies, however, have been performed using cells that have been exposed to concentrations of glucose higher than physiological concentrations for several weeks; for example, a commercially available culture medium (containing 25 mmol/L of glucose) is commonly used as culture media. Thus, the energy metabolism of astroglia cultured in a medium containing a physiological glucose concentration (∼ 3 mmol/L) has rarely been assessed.
In this study, we evaluated the differences in oxidative metabolism between astroglia cultured in a commercially available medium (with a final glucose concentration of 22 mmol/L) and those cultured in a medium containing a lower glucose concentration (2 mmol/L). We measured the rates of the oxidation of L-[U-14C]lactate ([14C]lactate) to 14CO2 to evaluate the lactate oxidation of astroglia. The oxidation of glucose that enters the tricarboxylic acid (TCA) cycle and is oxidized to CO2 was assessed by measuring the rates of oxidation of D-[U-14C]glucose ([14C]glucose) to 14CO2. The values obtained in astroglia were also compared with those obtained in neurons.
In addition to the basal condition, it should be investigated whether increased energy demand driven by Na+,K+-ATPase is fueled by the oxidative metabolism of glucose. Na+,K+-ATPase plays an important role in maintaining the Na+ and K+ gradients of the cell membrane during physiological activation (Clarke and Sokoloff, 1999). Increased intracellular Na+ and/or extracellular K+ are known to stimulate Na+,K+-ATPase. Thus, we also investigated the effects of the glucose concentrations in culture media on the oxidative metabolism of astroglia in response to conditions that stimulate Na+,K+-ATPase, resulting in an increased energy demand.
Materials and methods
Animals
Timed-pregnant Sprague–Dawley rats were purchased from Japan SLC, Inc. (Hamamatsu, Japan). All animal procedures were performed in accordance with The Animal Experimentation Guidelines of Keio University School of Medicine and were approved by the Laboratory Animal Care and Use Committee of Keio University.
Chemicals
Chemicals and materials were obtained from the following sources: 2-deoxy-D-[1-14C]glucose ([14C]dGlc) (specific activity, 54.0 mCi/mmol; 1 Ci = 37 GBq), D-[U-14C]glucose (specific activity, 303 mCi/mmol), [14C]lactate (specific activity, 131 mCi/mmol), Insta-Fluor Plus and hyamine hydroxide 10-X from Perkin-Elmer Life Sciences (Boston, MA, USA); Dulbecco's modified Eagle medium with or without glucose, penicillin, and streptomycin from Life Technologies (Grand Island, NY, USA); defined fetal bovine serum from HyClone Laboratories (Logan, UT, USA); trypsin-EDTA from Boehringer Mannheim (Indianapolis, IN, USA); Amplex Red Glucose/Glucose Oxidase Assay Kit from Molecular Probe (Eugene, OR, USA); and all other chemicals from Sigma (St Louis, MO, USA).
Preparation of Cells
Primary astroglial cultures were prepared from the cerebral cortex of rat pups 24 to 48 h after birth (Takahashi et al, 1995). The dissociated cells from the frontoparietal cortices (2.5 × 105 cells/mL) were plated (l5 mL/flask) in uncoated 75-cm2 culture flasks (Sumitomo Bakelite, Tokyo, Japan) and cultured in a high-glucose medium (final concentration, 22 mmol/L of glucose) comprised of Dulbecco's modified Eagle medium with 10% (v/v) fetal bovine serum, penicillin (100 U/mL), and streptomycin (100 μg/mL) at 37°C in humidified air containing 7% CO2 (day 0). The culture medium was changed twice a week until the cultures reached confluence. On day 10, the adherent cells were treated with trypsin-EDTA solution, suspended in fresh high-glucose medium, and placed in uncoated 24-well culture plates or in 25-cm2 culture flasks (Nalge Nunc., Rochester, NY, USA). From the day after subculturing, some of the cells were cultured in a low-glucose medium (final concentration, 2 mmol/L of glucose) comprised of Dulbecco's modified Eagle medium without glucose, but supplemented with D-glucose, fetal bovine serum, penicillin, and streptomycin. The culture medium was changed twice a week, and the cells were used once they reached confluence (days 28 to 32).
The primary neuron culture was prepared from the striata of fetal rats on embryonic day 16, as described previously (Takahashi et al, 1995). The mechanically dissociated cells were placed (0.4 mL/well) in 24-well culture plates or 25-cm2 culture flasks coated with poly-L-lysine (5 μg/mL). Cells were cultured in high-glucose (final concentration, 22 mmol/L) Dulbecco's modified Eagle medium at 37°C in humidified air containing 7% CO2. Cytosine arabinoside (10 μmol/L) was added 72 h later to induce the mitotic arrest of the astroglia. Assays were performed using cultures that were 7 to 10 days old. The nutrient medium remained untouched until the experiments were initiated.
Measurement of Intracellular Glycogen Content
The glycogen content of the astroglia was determined using a modification of the previously described method (Sorg and Magistretti, 1991). After washing the cells three times with ice-cold Dulbecco's phosphate-buffered saline (PBS), which almost completely eliminated the glucose from the nutrient medium, 0.4 mL of 20 mmol/L HCl was added to each well and the cells were sonicated. Two 100-μL aliquot samples of cell lysate were removed from each well, and, after adding 200 μL of acetate buffer (0.1 mol/L, pH 4.8) containing 0.5 mg/mL amyloglucosidase to the first sample and 200 μL of acetate buffer to the second, both samples were incubated at room temperature for 30 mins. The glucose content of both samples was then measured using a kit (Amplex Red Glucose/Glucose Oxidase Assay Kit), and the difference between the first and second samples was considered to be the cellular glycogen content expressed as glucosyl residues.
Assay for 2-Deoxy-D-[1-14C]Glucose Phosphorylation
The rates of glucose phosphorylation in neurons and astroglia were evaluated using the [14C]dGlc method (Sokoloff et al, 1977; Takahashi et al, 1995). The culture medium was replaced by 0.4 mL of Dulbecco's balanced salt solution (DBSS) containing 110 mmol/L NaCl, 5.4 mmol/L KCl, 1.8 mmol/L CaCl2, 0.8 mmol/L MgSO4, 0.9 mmol/L NaH2PO4, and 44 mmol/L NaHCO3 with 2 mmol/L glucose and 10 μL of [14C]dGlc (3.7 MBq/mL); incubation was continued for 30 mins at 37°C, 7% CO2. At the end of the incubation period, the DBSS was replaced by a fresh reaction mixture lacking [14C]dGlc; incubation was continued at 37°C, 7% CO2 for 5 mins to allow the efflux of residual [14C]dGlc from the cells. The cell carpets were washed quickly three times with ice-cold PBS and digested in 0.2 mL of 0.1 mol/L NaOH at room temperature. The cell digests were then assayed for protein content using the bicinchoninic acid method (Smith et al, 1985), and the 14C count was measured using a liquid scintillation counter (Tri-Carb 3100 TR; Perkin-Elmer Life Sciences, Boston, MA, USA).
Assay for Lactate Production
Lactate release into the medium was determined in the sister culture of the astroglia used in the [14C]dGlc assays using a slight modification of the previously described method (Itoh et al, 2003). The cells were incubated with DBSS containing 2 mmol/L glucose for 30 mins at 37°C, 7% CO2. Next, 40 μL of the supernatant solutions were added to 200 μL of 0.6 mol/L glycine buffer (pH 9.2) containing 0.5 mol/L hydrazine sulfate and 0.1 mol/L NAD+. After equilibration for 5 mins at room temperature, 25 μL (1 units/ μl) of lactic dehydrogenase was added and NAD+ reduction to NADH was determined by measuring the absorbance at a wavelength of 340 nm (OD340) using a Bio-Rad Benchmark Microplate Reader, until the completion of the reaction. The lactate contents of the samples were calculated from the changes in absorbance and the specific absorbance of NADH at 340 nm. The lactate concentration was normalized to the cellular protein content.
Measurement of the Rate of L-[U-14C]Lactate and D-[U-14C]Glucose Oxidation to 14CO2
The rate of [14C]lactate and [14C]glucose oxidation to 14CO2 was measured using a modification of a previously described method (Itoh et al, 2003). After cells cultured in 25-cm2 culture flasks were washed twice with PBS, 5 mL of DBSS containing 2 mmol/L of lactate and 2.5 μL of [14C]lactate (3.7 MBq/mL) or 2 mmol/L of glucose and 10 μL of [14C]glucose (3.7 MBq/ml) was added to the flasks. The culture flasks were then capped with rubber stoppers containing a center well and incubated at 37°C for 60 mins. The 14CO2 produced was trapped in the center well containing 150 μL of hyamine hydroxide 10-X. The reactions were terminated by the injection of 250 μL of 60% perchloric acid through the rubber stopper, and the flasks were kept at 4°C overnight to trap the 14CO2. The center wells were transferred to 20 mL glass scintillation counter vials, and 500 μL of ethanol and 10 mL of Insta-Fluor Plus were added. The 14C contents of the vials were then evaluated using a liquid scintillation counter. Waniewski and Martin (2004) reported that substantial 14C counts were obtained from a flask without cells. Therefore, the 14C counts obtained from a flask without cells in which the reaction had been stopped at 60 mins were regarded as the background values and were subtracted in our studies. The cell carpets left in the incubation flasks after the removal of the reaction mixtures were digested with 5 mL of 0.1 mol/L NaOH, and their protein contents were determined.
Statistical Analyses
Statistical comparisons among the values obtained for each group were carried out using grouped t-tests or a one-way analysis of variance (ANOVA), followed by Dunnett's or Tukey's test for multiple comparisons. A P-value of < 0.05 was considered statistically significant.
Results
Characterization of Cell Cultures
The secondary cultures of both astroglia cultured in the presence of 2 mmol/L of glucose (astroglia2) or 22 mmol/L of glucose (astroglia22) appeared to be pure; no cells without GFAP were observed. Furthermore, no morphological differences were observed between astroglia2 and astroglia22 (data not shown).
Glucose Phosphorylation, Storage, and Conversion to Lactate by Astroglia Grown in Low or High Glucose
First, we examined the glycogen content of astroglia22 and astroglia2. The glycogen content in astroglia22 was approximately 250% of that in astroglia2 (Table 1). Next, we compared the rate of [14C]dGlc phosphorylation in astroglia2 with that in astroglia22. Although [14C]dGlc phosphorylation in neurons was 87% of that in astroglia, no significant differences were observed among astroglia22, astroglia2, and neurons (Table 1). Finally, the lactate concentration of the assay solution was measured under the same conditions as the [14C]dGlc phosphorylation assay. The amount of lactate released by astroglia22 was 49% larger than that released by astroglia2 (Table 1). The lactate released in the assay solution of the neurons was 35% of that released by astroglia2 (Table 1).
Glucose phosphorylation, storage, and conversion to lactate by astroglia grown in high or low glucose

Values are the mean ± s.d. of number (n) of experiments carried out under the same conditions. ND, not determined.
Not significant between neurons, astroglia cultured in low-glucose medium, and astroglia cultured in high-glucose medium (ANOVA).
P < 0.01 versus astroglia cultured in high-glucose medium (ANOVA followed by Tukey's multiple-comparisons test).
P < 0.01 versus astroglia cultured in low-glucose medium (ANOVA followed by Tukey's multiple-comparisons test).
P < 0.05 versus astroglia cultured in high-glucose medium (ANOVA followed by Tukey's multiple-comparisons test).
P < 0.01 versus astroglia cultured in low-glucose medium (grouped t-test).
Rates of L-[U-14C]Lactate Oxidation to 14CO2 in Astroglia22 and Astroglia2
The rates of [14C]lactate oxidation were expressed as the amount of metabolized lactate based on the obtained 14C count. However, these values were minimal rates because the intracellular specific activity of [14C]glucose and [14C]lactate metabolites might be lowered by dilution with the amino-acid pools. Astroglia2 had a higher rate of [14C]lactate oxidation to 14CO2 than astroglia22 (Figure 1). This 147% increase was statistically significant (P < 0.01). In neurons, the rate of [14C]lactate oxidation to 14CO2 was even higher, approximately four times that in astroglia22 and almost twofold that in astroglia2 (Figure 1).
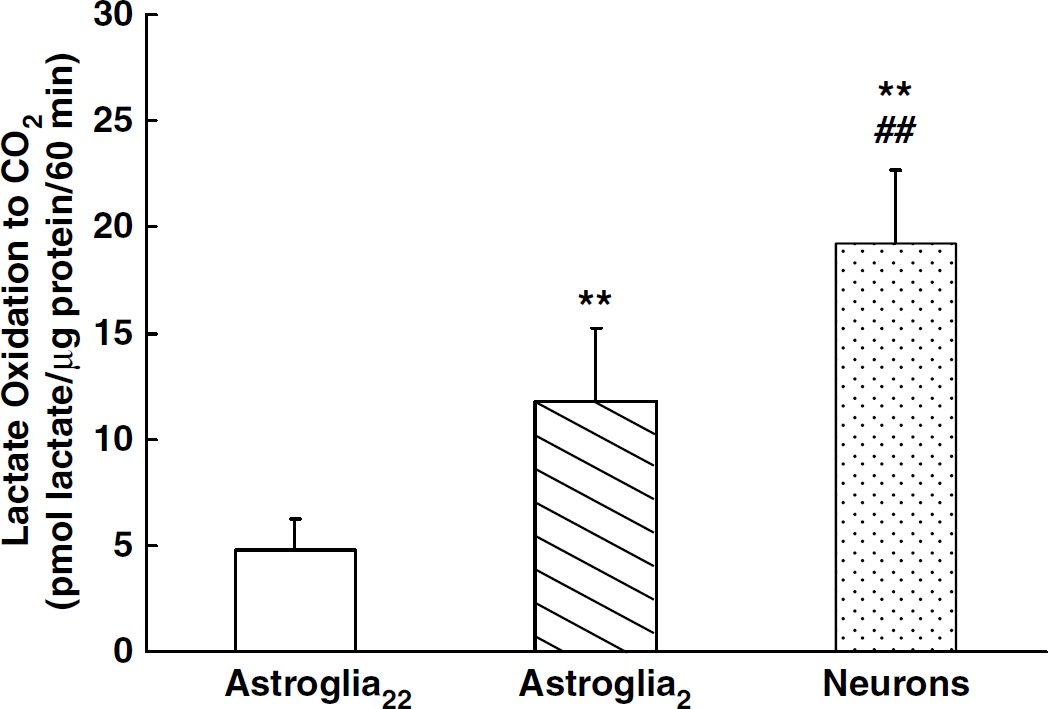
Rate of [14C]lactate oxidation to 14CO2 in neurons (dotted column) and astroglia cultured in the presence of 22 mmol/L (open column) or 2 mmol/L of glucose (hatched column). Column heights and error bars represent the mean and s.d. of oxidized [14C]lactate for five to seven experiments from at least two different batches. **P < 0.01 with respect to astroglia2, ##P < 0.01 with respect to astroglia2 (ANOVA followed by Tukey's multiple-comparisons test).
Rates of D-[U-14C]Glucose Oxidation to 14CO2 in Astroglia22 and Astroglia2
The oxidation of [14C]glucose by astroglia2 was also examined to enable a comparison with that by astroglia22 (Figure 2). The results were similar to those for [14C]lactate oxidation, indicating a difference in oxidative metabolism in these two populations. Astroglia2 were found to have a significantly higher rate of [14C]glucose oxidation than astroglia22 (Figure 2). Neurons showed an even higher capacity for glucose oxidation.
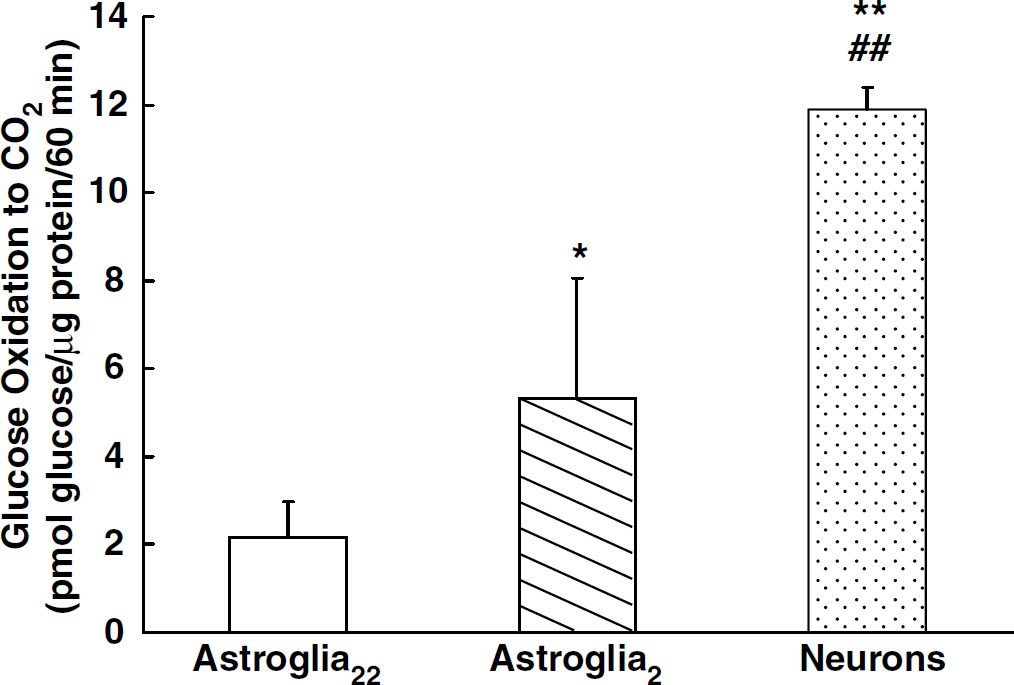
Rate of [14C]glucose oxidation to 14CO2 in neurons (dotted column) and astroglia cultured in the presence of 22 mmol/L (open column) or 2 mmol/L of glucose (hatched column). Column heights and error bars represent the mean and s.d. of oxidized [14C]glucose for four to six experiments from at least two different batches. **P < 0.01, *P < 0.05 with respect to astroglia55, ##P < 0.01 with respect to astroglia2 (ANOVA followed by Tukey's multiple-comparisons test).
Effects of D-Aspartate on Lactate Oxidation in Astroglia
D-Aspartate elicits increases in intracellular Na+ concentration, leading to the stimulation of Na+,K+-ATPase. We first confirmed that both 500 μmol/L of D-aspartate and 500 μmol/L of L-glutamate increased [14C]dGlc phosphorylation (Table 2). Then, we evaluated the effects of 500 μmol/L of D-aspartate on astroglial oxidative metabolism. Statistically significant increases in lactate oxidation were observed in the presence of D-aspartate both in astroglia22 and astroglia2 (Figure 3A). D-Aspartate also led to an increase in [14C]glucose oxidation in astroglia (Figure 3B). The [14C]glucose oxidation (pmol glucose/μg protein per 60 min) in astroglia22 increased from 2.59 ± 0.48 to 5.02 ± 0.80, while that in astroglia2 increased from 5.40 ± 0.78 to 9.49 ± 1.06.
[14C]dGlc phosphorylation in astroglia cultured in the presence of a high (22 mmol/L) or low (2 mmol/L) glucose concentration and L-glutamate or D-aspartate stimulation
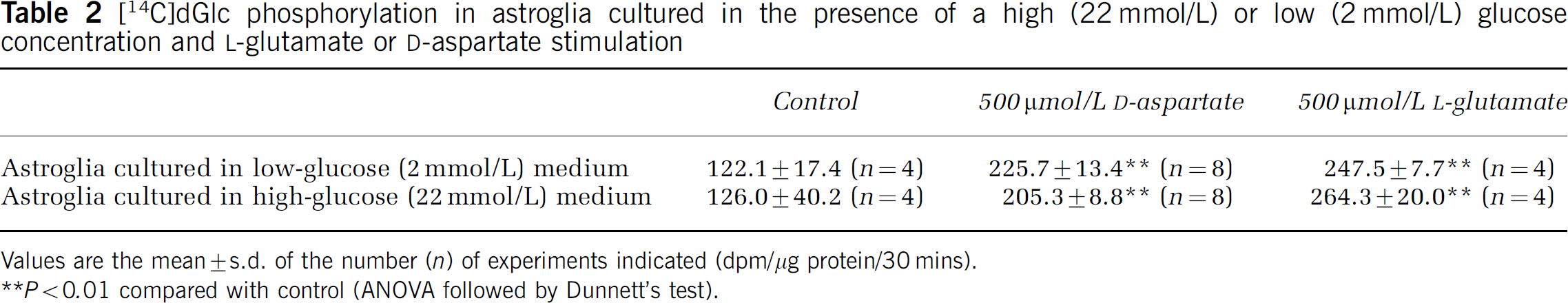
Values are the mean ± s.d. of the number (n) of experiments indicated (dpm/μg protein/30 mins).
P < 0.01 compared with control (AN0VA followed by Dunnett's test).
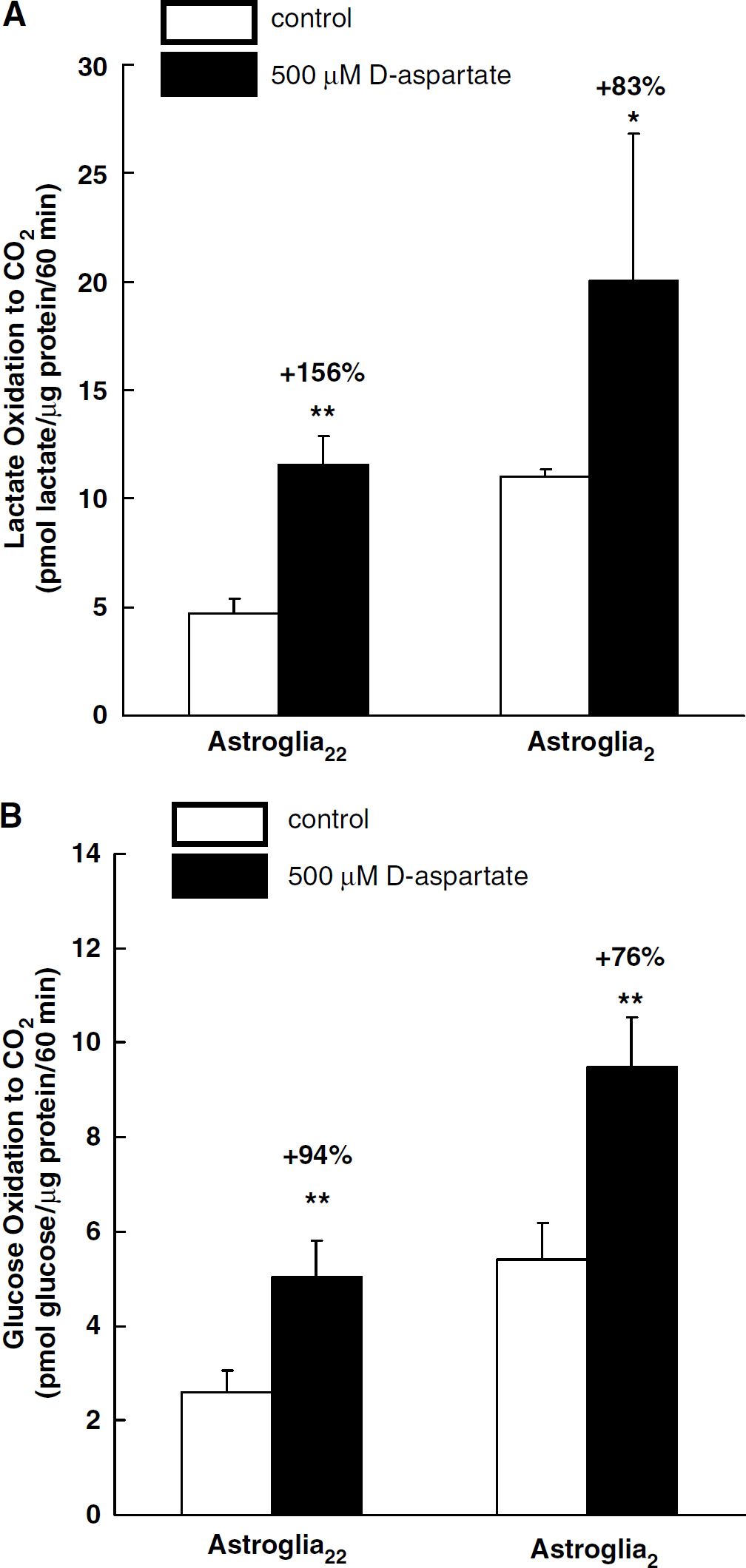
Effects of 500 μmol/L of D-aspartate on astroglial [14C]lactate oxidation (
Effects of High Extracellular K+ Concentration ([K+]o) on Oxidative Metabolism in Astroglia
The effect of an elevated [K+]o level on oxidative metabolism in astroglia was investigated. An elevated [K+]o within a physiological range (∼ 20 mmol/L) increased glucose phosphorylation in astroglia by increasing the activity of Na+,K+-ATPase (Brookes and Yarowsky, 1985), although an elevated [K+]o at levels far beyond the normal range did not (Takahashi et al, 1995). Extracellular K+ concentration was increased from 5.4 to 7.4 mmol/L, since glucose utilization has been reported to peak at 8 mmol/L [K+]o (Pentreath and Kai-Kai, 1982). The rate of [14C]dGlc phosphorylation increased significantly with K+ stimulation in both astroglia22 and astroglia2 (Table 3), while an elevated [K+]o had no effect on [14C]lactate or [14C]glucose oxidation in both astroglia2 and astroglia22 (Figures 4A and 4B).
[14C]dGlc phosphorylation in astroglia cultured in the presence of a high (22 mmol/L) or low (2 mmol/L) glucose concentration and [K+]o stimulation
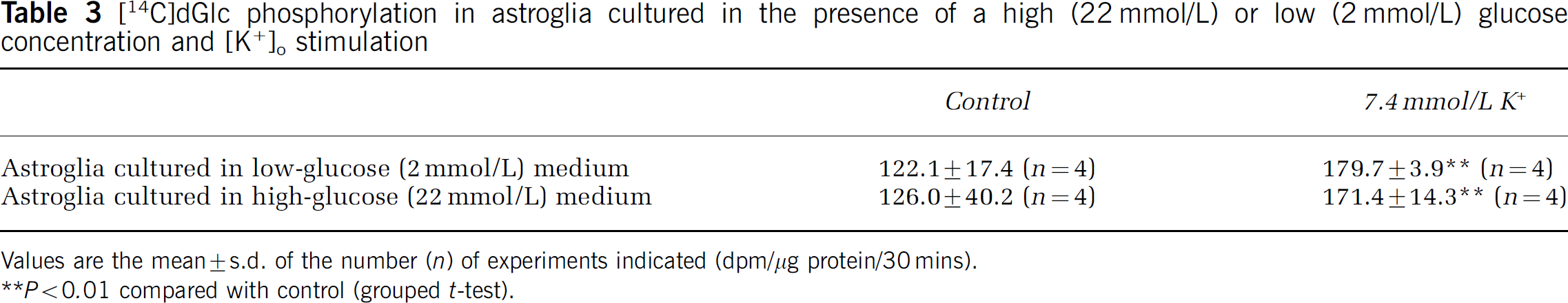
Values are the mean ± s.d. of the number (n) of experiments indicated (dpm/μg protein/30 mins).
P < 0.01 compared with control (grouped t-test).
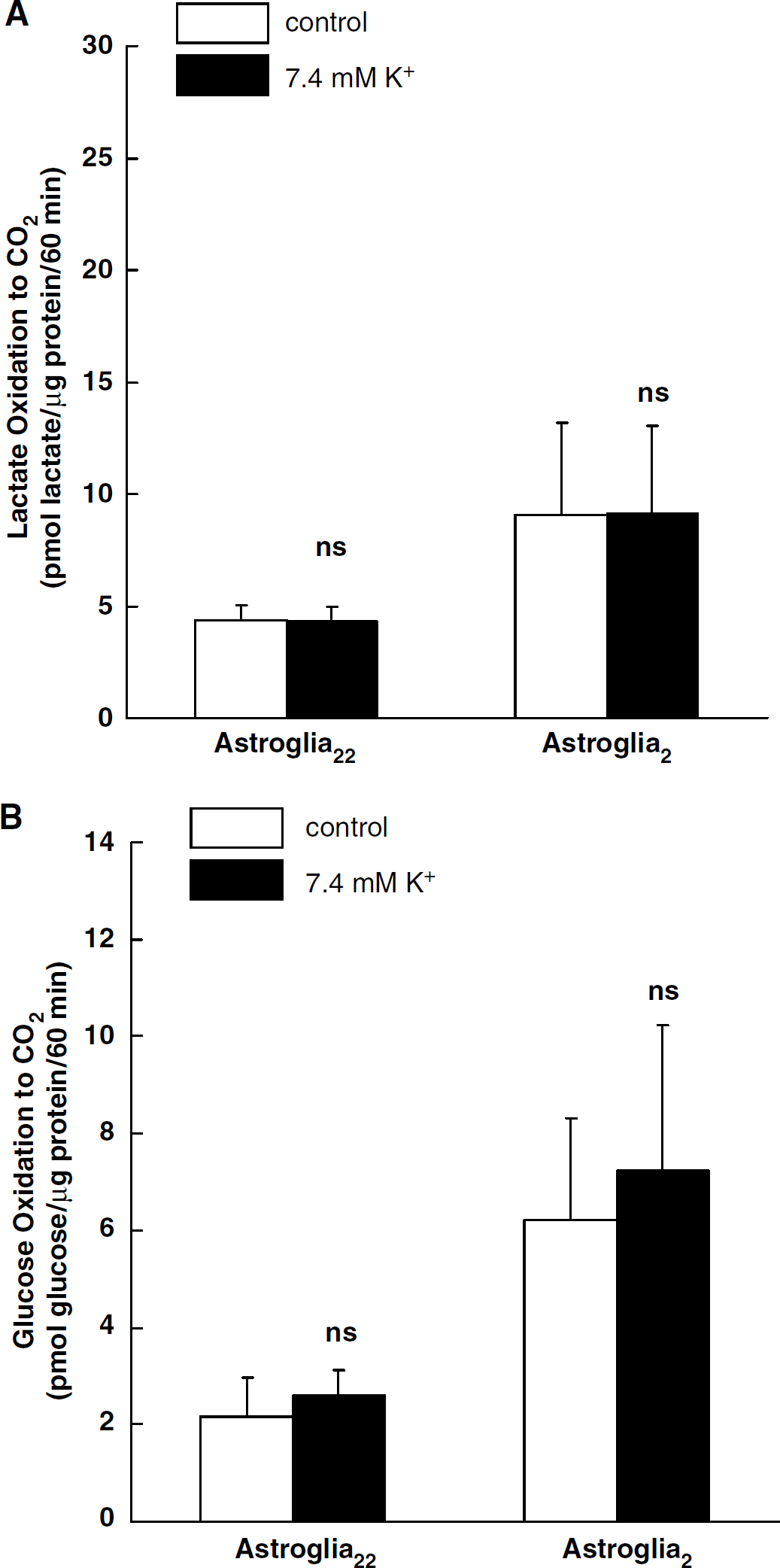
Effects of a high [K+]o level on [14C]lactate oxidation (
Discussion
Previous studies have emphasized the oxidative capability of neurons and the glycolytic capacity of astrocytes (Abe et al, 2002; Takahashi et al, 2002), but few have investigated the effects of culture conditions on astroglial metabolism at rest and during metabolic challenge. In the present study, cultured striatal neurons had glucose phosphorylation rates similar to those of cortical astroglia that were grown in either 2 or 22 mmol/L glucose, but glucose and lactate oxidation rates were higher and lactate release was lower in astroglia grown in 2 mmol/L glucose compared with astroglia grown in 22 mmol/L glucose. The astroglia grown in high glucose had higher glycogen levels, greater lactate release, and much lower glucose and lactate oxidation rates compared with those grown in 2 mmol/L glucose; in both groups, the rates of lactate oxidation were approximately twice those of glucose oxidation when both substrates were 2 mmol/L. During exposure to D-aspartate, both groups increased their rates of glucose phosphorylation and oxidation of lactate or glucose. Increased levels of extracellular K+ stimulated glucose phosphorylation in both groups of astroglia, but did not show differential effects on oxidative metabolism. Thus, astroglia grown in either culture medium were capable of oxidizing glucose and lactate at increased rates when activated, but the growth in high glucose markedly suppressed the oxidative capabilities of the cultured astroglia.
In our study, increases in intracellular Na+ associated with D-aspartate stimulated glucose and lactate oxidation. This finding is consistent with a recent report that monensin, a Na+ ionophore that facilitates Na+ entry into cells, increased [14C]glucose oxidation to 14CO2 (Peng et al, 2001). However, L-glutamate, which is released from neurons, is taken up by astrocytes as an oxidizable substrate (Hertz et al, 1998b). Thus, L-glutamate oxidation may meet some fractions of the increased energy demand during neuronal activation. Nevertheless, as the energy demand increases in the presence of glucose restriction, astroglial lactate oxidation may increase.
Our findings of increased [14C]dGlc phosphorylation by elevated [K+]o were consistent with the previous report that showed increase of [14C]dGlc phosphorylation when [K+]o is raised to or above 12 mmol/L in mouse cultured astrocyte (Peng et al, 1996). However, in the other study employing rat astroglia, 28 and 56 mmol/L of [K+]o were reported to cause no increase in [14C]dGlc phosphorylation (Takahashi et al, 1995). The response of [K+]o increase far beyond the physiological level may differ between cultured rat astrocytes and mouse astrocytes. In contrast to the effects of an increase in intracellular Na+, neither lactate nor glucose oxidation was affected by an increase in [K+]o, although an elevation in [K+]o did indeed cause [14C]dGlc phosphorylation to increase. Previously, 50 mmol/L [K+]o has been reported not to stimulate lactate oxidation (Peng et al, 1994).
The reason why cultivation in media with lower glucose concentration makes astroglial metabolism more oxidative was not determined in the present study. Cellular oxidative metabolism is thought to be regulated by pyruvate dehydrogenase (PDH) complex, which catalyzes in the decarboxylation of pyruvate to form acetyl CoA (Wieland, 1983). The ATP/ADP ratio could be higher in astroglia cultured with high glucose because of ATP synthesis by continuous glycogen degradation, and a high ATP/ADP ratio downregulates the activity of PDH (Clarke and Sokoloff, 1999). The difference in malate-aspartate shuttle capability, the exchange rate between TCA cycle keto acid and amino acids, and the size of the amino-acid metabolic trapping pools may affect the oxidative metabolism of neurons and the groups of astrocyte grown under two conditions. In TCA cycle, labeled α-ketoglutarate is converted to [14C]glutamate by transaminase exchange reactions. Furthermore, it is known that malate-aspartate shuttle contributes to mixing and dilution of labeled carbon crossing the mitochondrial membrane within cellular amino-acid pools (Dienel and Cruz, 2004). The larger glycogen degradation in astroglia grown in high glucose may increase the amount of produced NADH via glycolysis, accelerating malate–aspartate shuttle. It may lead to the dilution of labeled substrate within the amino-acid pool of cytoplasm.
Increased lactate production during neuronal excitation (Prichard et al, 1991) is thought by some authors to be taken up mainly by neurons (Magistretti et al, 1999). However, our results suggested that astrocytes may also have the capacity to use lactate. In addition, the contribution of lactate transport through gap junctions between astroglia remains to be resolved (Dienel and Cruz, 2003). However, neurons do indeed use glucose in vivo and in vitro (Itoh et al, 2004). Both neurons and astrocytes use glucose and may produce and use lactate.
