Abstract
Striatal coadministration of interleukin-1β (IL-1β) with α-amino-3-hydroxy-5-methyl-4-isoxazolepropionate (S-AMPA) in rats results in widespread cortical cell death not caused by either treatment alone. This cortical damage was unaffected by cortical infusion of the AMPA-receptor antagonist NBQX. Cortical infusion of an NMDA-receptor antagonist D-AP5 significantly inhibited (57%;
The cytokine interleukin-1 (IL-1) has diverse actions in the central nervous system (Rothwell and Hopkins, 1995) and has been implicated in acute and chronic neurodegeneration (Rothwell, 1999). Injection or overexpression of human recombinant interleukin-1 receptor antagonist (hrIL-1ra) significantly inhibits neuronal damage induced in the rat by focal cerebral ischemia, traumatic brain injury, or excitotoxin administration (Rothwell, 1999)). Studies to date suggest that the β form of IL-1 (IL-1β) is the primary mediator of acute brain damage (Rothwell, 1999).
Although IL-1β alone is not toxic to healthy neurones, administration of exogenous IL-1β exacerbates neuronal damage induced by focal (Loddick and Rothwell, 1996) or global cerebral ischemia (Yamasaki et al., 1995). In focal ischemia, the effects of IL-1ra and IL-1β are mediated through actions in the striatum that influence cortical damage (Stroemer and Rothwell, 1997, 1998). The authors have demonstrated similar site-specific effects of IL-1β in excitotoxic damage. IL-1β, coinjected with the excitatory amino acid agonist α-amino-3-hydroxy-5-methyl-4-isoxazolepropionate (S-AMPA) in the rat striatum, has no significant effect on the local striatal injury but produces widespread neuronal death in the ipsilateral cortex (Lawrence et al., 1998).
The authors' recent data indicate that IL-1β actions in the striatum involve the hypothalamus, which suggests the involvement of a polysynaptic pathway from the striatum to the cortex (Allan et al., 2000). Cortical damage resulting from striatal coinjection of S-AMPA and IL-1β is reduced by systemic administration of MK-801, which suggests the involvement of glutamatergic pathways (Lawrence et al., 1998). Therefore, the objective of the current study was to test further the hypothesis that IL-1β acting in the striatum causes secondary cortical damage through activation of glutamatergic pathways, and to identify the receptors involved in the distant damage in the cortex.
MATERIALS AND METHODS
S-AMPA,
Male Sprague-Dawley rats (250 to 350 g; Charles River, Margate, U.K.) were anesthetized with halothane (2.5% in 100% O2) and placed in a stereotaxic frame. Stereotaxic injections were performed as described previously (Lawrence et al., 1998) into the appropriate area (coordinates relative to bregma (mm): anterior-posterior (AP) +0.7, medial lateral (ML) −2.7, dorsal-ventral (DV) −5.5 for the striatum; AP +0.7, ML −4.8, DV −1.8 for the cortex). Striatal injections were performed directly with a Hamilton syringe (Hamilton, Carnforth, U.K.), whereas cortical infusions were through a 26 g needle, connected by polyethylene tubing to a Hamilton syringe in an infusion pump. The latter method was used for cortical infusions as preliminary studies indicated that, because of less reflux of the injected solution, more reproducible lesions were obtained in the cortex when an infusion pump was used, whereas no obvious difference in the striatal lesions between the two methods was found. All drug administrations were performed for 2 minutes at a rate of 0.5 μL/min with the needle left in place for an additional 5 minutes to allow diffusion of the injected solution.
Animals were killed 24 or 48 hours after infusion of methanoglutamate or S-AMPA treatment, respectively. Brains were removed and snap frozen in isopentane on dry ice. Neuronal loss throughout the entire lesion was determined from coronal cryostat sections (Lawrence et al., 1998) or, when there was excessive edema (S-AMPA and hrIL-1β coinjections), indirectly from brain maps onto which the area of damage was drawn (to compensate for any swelling). All doses of drugs used were based on preliminary studies (data not shown) or on published work using similar reagents (Massieu and Tapia, 1994).
Data are expressed as mean ± SD of the mean for the number of animals given. Significant differences in lesion volume between groups of animals were assessed either by unpaired
RESULTS
Damage produced by direct infusion of the selective NMDA-receptor agonist methanoglutamate (7.5 nmol) in either the striatum or cortex was significantly reduced (87% to 95%) by coadministration of D-AP5 (Table 1). However, no change in lesion size was observed when D-AP5 was infused together with S-AMPA (Table 1). NBQX, infused directly into the striatum with S-AMPA, also produced a marked reduction (98%) in the lesion volume (Table 1). This shows that the agonists and antagonists used in the current study were indeed selective for the appropriate receptor subtype.
Effect of glutamate receptor antagonists on methanoglutamate, S-AMPA, or S-AMPA plus hrIL-1β induced cell death

(A) Effect of cortical D-AP5 (25 nmol) treatment on the lesion volume (L
The volume of striatal damage resulting from intrastriatal coinjection of S-AMPA and hrIL-1β was no different between rats treated with vehicle, D-AP5, or NBQX in the cortex (Table 1). In contrast, neuronal damage in the cortex produced by striatal injection of S-AMPA and hrIL-1β was not affected by infusion of NBQX in the cortex, whereas D-AP5 significantly reduced (57%) cortical cell death (Table 1, Figs. 1 and 2). In most cases (n = 9 of 13) D-AP5 completely abolished the neuronal damage in the parietal cortex, although in some animals there was only partial inhibition, and a small amount of neuronal loss was still observed in this region (representative examples of each are illustrated in Fig. 2).
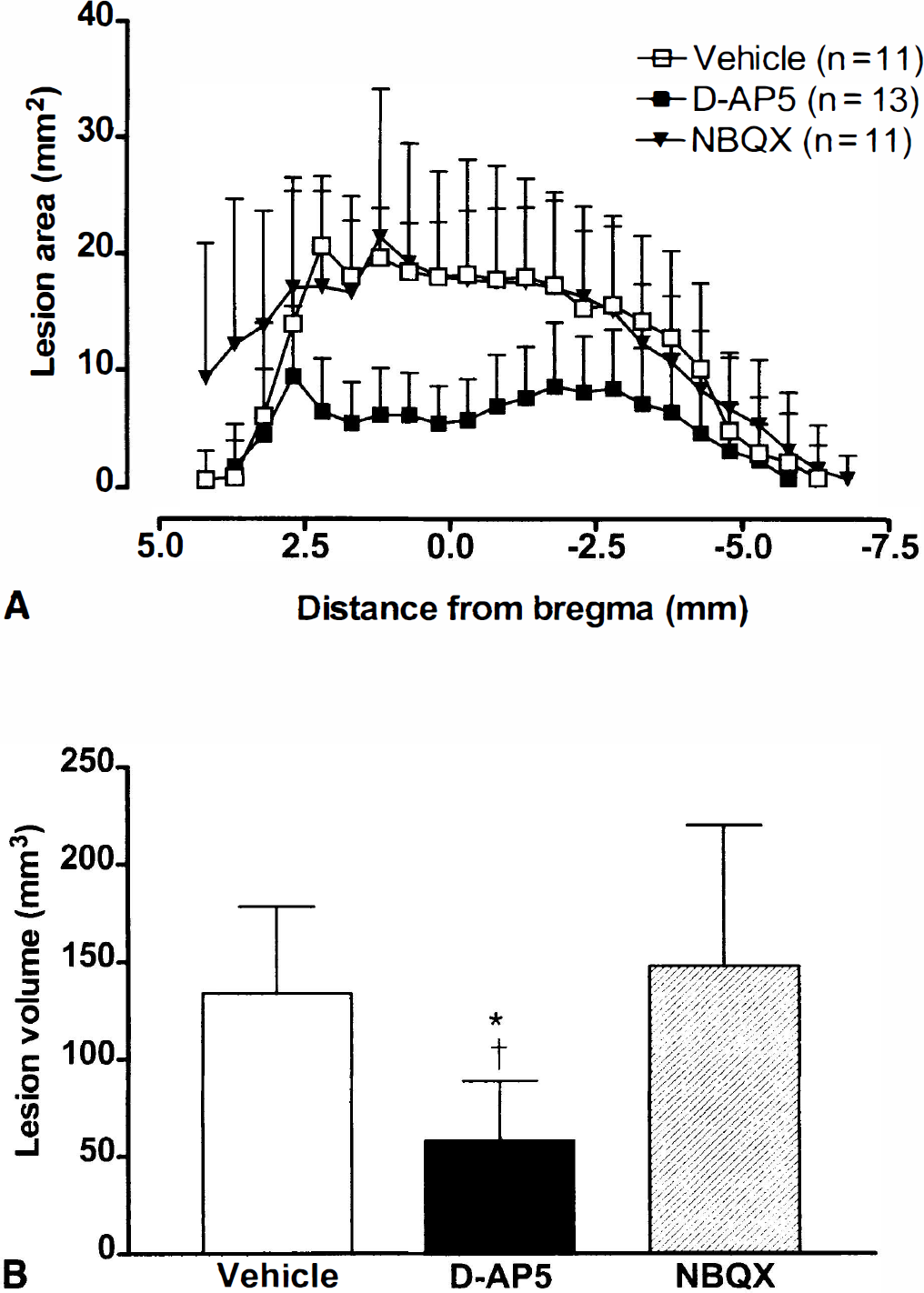
Graphs showing the cortical damage induced by intrastriatal administration of S-AMPA (7.5 nmol) and hrIL-1β (10 ng) immediately followed by vehicle, D-AP5 (25 nmol), or NBQX (25 nmol) infusion in the cortex.

Coronal brain sections (20 μm; taken at the site of injection, 0.7 mm anterior of bregma) stained with cresyl violet to demonstrate the effect of cortical infusion of D-AP5 (25 nmol) on the neuronal damage after striatal coinjection of S-AMPA (7.5 nmol) and hrIL-1β (10 ng).
The current study shows that the extensive cortical cell death caused by S-AMPA and hrIL-1β in the striatum is inhibited by direct cortical infusion of an NMDA antagonist, but not by cortical infusion of a selective AMPA antagonist. This suggests that the remote death in the cortex is mediated through a glutamatergic pathway and activation of NMDA receptors in the cortex.
Intrastriatal coinjection of hrIL-1β with S-AMPA resulted in extensive cortical cell loss throughout the ipsilateral hemisphere, particularly in the frontal and parietal cortex. In the most severe cases (n = 5 of 11) this extrastriatal lesion extended over the entire ipsilateral cortex, involving most cortical regions through a rostrocaudal distance of >7 mm. Infusion of D-AP5 in the parietal cortex region, ipsilateral to the site of the S-AMPA and hrIL-1β injection, produced a significant reduction in the mean lesion size. Although some animals (n = 7 of 13) in this group showed cell loss in the amygdala and piriform and entorhinal cortex, with no damage in other cortical regions, this damage appeared to be unaffected by D-AP5 (data not shown). Damage in the amygdala and piriform and entorhinal cortex, but not the parietal cortex, is frequently observed in animals treated with S-AMPA alone, but does not appear different from that produced by striatal AMPA and hrIL-1β injection (Allan and Rothwell, unpublished observations).
DISCUSSION
Brain damage in sites “remote” from the site of excitotoxic injection has been documented previously (Ciani et al., 1994; Wozniak et al., 1994), and has been ascribed to the activation of “seizure-related pathways” (Ben-Ari, 1985). The observed effects of D-AP5 reported in the current study are consistent with studies reporting that NMDA antagonists reduce the “remote” cell death caused by excitotoxin administration in a number of brain regions, but have no effect on the local damage (Facchinetti et al., 1992; Ciani et al., 1994; Wozniak et al., 1994). Both local and distal toxicity caused by hippocampal injection of kainic acid is inhibited by peripheral administration of the noncompetitive antagonist MK-801, with only approximately 50% of treated animals showing complete prevention of all the damage in “remote” areas (Lees, 1992). A similar finding was made in the current study, in which the damage produced in the parietal cortex by striatal injection of S-AMPA and hrIL-1β was completely prevented in 9 of the 13 animals treated with D-AP5 in the cortex, with the remaining others displaying only partial inhibition (Fig. 2).
The mechanism by which striatal coinjection of S-AMPA and hrIL-1β produces cortical neuronal death is not known. As there are no described direct striato-cortical neuronal connections, IL-1β actions are probably not mediated by a direct neuronal pathway. It is also unlikely that IL-1β has a retrograde action (Laduron, 1995) because direct injection of hrIL-1β into the cortex, even at high doses, fails to modify the neuronal damage resulting from infusion of excitotoxins in these areas (Lawrence et al., 1998). The striatum signals to the cortex indirectly through the globus pallidus and thalamus (Parent and Hazrati, 1995) or the hypothalamus (Saper, 1985; Risold et al., 1997), and the observed effects of IL-1β may due in part to excessive release of glutamate in the ipsilateral cortex, mediated through activation of these anterograde pathways. Indeed, the authors have demonstrated increased expression of IL-1β in the hypothalamus after striatal coinjection of S-AMPA and hrIL-1β, and have shown that direct administration of IL-1ra into the lateral hypothalamus reduces the resultant cortical death (Allan et al., 2000). This suggests that IL-1β can act in this region to somehow affect cortical cell viability.
A recent report has demonstrated that IL-1β can prolong seizure activity, and the authors propose that IL-1β can enhance glutamatergic neurotransmission (Vezzani et al., 1999), although the mechanisms by which it does so are not discussed. Similar effects could explain the actions of IL-1β on the pathways being driven by intrastriatal administration of S-AMPA in the current study. However, IL-1β may not directly activate glutamatergic neurones but rather produce an indirect effect by modifying GABAergic transmission to cause disinhibition of an excitatory pathway. Indeed, a number of reports have shown effects of IL-1β on inhibitory systems (Yu and Shinnick-Gallagher, 1994; Miller et al., 1991).
The results presented here, together with the authors' recent observations (Allan et al., 2000), lead to the propososition that IL-1β activates polysynaptic pathways leading from the striatum to the cortex through the hypothalamus and possibly other regions. When accompanied by AMPA and/or kainate receptor activation in the striatum, IL-1β produces extensive cell death throughout the ipsilateral cortex that is dependent on the activation of NMDA receptors in this region. These findings may be of direct relevance to diverse neurodegenerative conditions associated with increased expression of brain IL-1β including stroke, traumatic brain injury, and Alzheimer's disease.
Footnotes
Acknowledgements
The authors thank Ralph Davies and Helen Anforth for their invaluable technical assistance.
