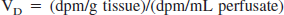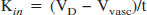Abstract
Brain capillary endothelial cells (BCECs) express transferrin receptors. The uptake of a potential drug vector (OX26, or anti—transferrin receptor antibody IgG2a) conjugated to polyethyleneglycol-coated liposomes by BCECs was studied using in situ perfusion in 18-day-old rats in which the uptake of OX26 is almost twice as high as in the adult rat. Using radio-labeling, the uptake of OX26 by BCECs after 15-minute perfusion was approximately 16 times higher than that of nonimmune IgG2a (Ni-IgG2a). OX26 and OX26-conjugated liposomes selectively distributed to BCECs, leaving choroid plexus epithelium, neurons, and glia unlabeled. Ni-IgG2a and unconjugated liposomes did not reveal any labeling of BCECs. The labeling of BCECs by OX26 was profoundly higher than that of transferrin. Perfusion with albumin for 15 minutes did not reveal any labeling of neurons or glia, thus confirming the integrity of the blood—brain barrier. The failure to label neurons and glia shows that OX26 and OX26-conjugated liposomes did not pass through BCECs. The expression of transferrin receptors by endothelial cells selective to the brain qualifies OX26 as a candidate for blood-to-endothelium transport. A specifically designed formulation of liposomes may allow for their degradation within BCECs, leading to subsequent transport of liposomal cargo further into the brain.
Brain capillary endothelial cells form the BBB, which effectively restricts the entry of molecules larger than 400 to 600 d from the circulation (Abbott and Romero, 1996;Begley and Brightman, 2003). This size limitation affects more than 98% of the drug candidates that emerge from high-throughput-screening drug programs (Pardridge, 1998), indicating the need to generate drug-delivery systems that will surpass the restraints of the BBB. One current principle of drug transport involves receptor-mediated targeting via intravenous injection of peptides that target receptors expressed on the luminal side of BCECs. To enable drug transport through the BBB, peptides are initially conjugated directly to drug molecules of interest, or indirectly via an entrapment of drugs in cargo carriers like liposomes that subsequently are conjugated to the peptide (Pardridge, 1998).
Peptide BCEC-receptors that have been targeted for drug-delivery purposes include the receptors for transferrin, insulin, epidermal growth factor, and melano-transferrin (Demeule et al., 2002;Moroo et al., 2003; c.f. Pardridge, 2001). Transferrin is of particular interest because the expression of the transferrin receptor in capillaries throughout the body is restricted to BCECs (Jefferies et al., 1984). Moreover, the profound transferrin receptor expression throughout the CNS during normal (Moos, 1996) and pathologic conditions (Hua et al., 2003;Wu et al., 2003) makes targeting with transferrin attractive. The major caveat when using drug conjugation to endogenous peptides for targeting receptors expressed by BCECs is the competition from endogenous peptides in the circulation. This has lead to an experimental paradigm shift towards using molecular antibodies that specifically target BCEC-receptor molecules. A monoclonal IgG2a antibody raised against the transferrin receptor protein (OX26) has been used to target BCECs in the rat alone (Friden et al., 1991;Pardridge et al., 1991), both directly via conjugation with putative drug molecules (Wu and Pardridge, 1996, 1999) and indirectly via conjugation to PEG-coated liposomes (Shi et al., 2001;Zhang et al., 2003a). Pardridge (1998) claimed that targeting the BCECs’ transferrin receptor would enable transcytosis of OX26 and its linker compounds through the BBB. A recent study, however, casts doubt on this notion. Using a brain capillary depletion technique, sampling of cisternal CSF, and immunohistochemical mapping of OX26 in the brain subsequent to intravenous injection, Moos and Morgan (2001) concluded that OX26 indeed accumulated in BCECs because of the presence of transferrin receptors, but that OX26 accumulation deeper within the brain was too small to justify the conclusion that receptor-mediated transcytosis of OX26 occurred in BCECs.
The rodent BCEC expression of transferrin receptor is much higher in the third postnatal week than in adulthood because of a physiologically upregulated iron transport into the brain at this developmental age (Taylor and Morgan, 1990). Reflecting this notion, intravenous injection of OX26 in 15-day-old rats leads to BCEC uptake of 3.9% OX26 of the injected dose per gram brain, which is significantly higher than the 0.5% OX26 of the injected dose per gram brain observed in the adult rat (Moos and Morgan, 2001). One advantage of using in situ perfusion method is that the composition of perfusion fluid and test compounds can be controlled with precision (Bickel et al., 2001). Moreover, because OX26 may compete with endogenous transferrin for binding to BCECs’ transferrin receptors (Ueda et al., 1993), we attempted to study OX26 uptake in the brain using the in situ perfusion model developed by Roberts et al. (1993) to determine the uptake of OX26 and OX26-conjugated liposomes at the developing stage of the 18-day-old (P18) rat. Although unable to show transport through the BBB, an observed blood-to-endothelium transport of OX26 and OX26-conjugated liposomes may be of significance for designing drug carriers that will enable a diphasic drug transport from blood to BCECs, and from BCECs further into the brain.
MATERIALS AND METHODS
Materials
Isotope 125I was obtained from Amersham International (Amersham, U.K.) as Na[125I]. Purified monoclonal mouse anti—rat transferrin receptor IgG2a (clone OX26) and Ni-IgG2a were purchased from Serotec (Oxford, U.K.). Monoclonal antibodies raised against human transferrin and human albumin were purchased from Biogenesis (Dorset, U.K.) and (Sigma-Aldrich Denmark, DK) respectively. SATA, cholesterol, human transferrin, human albumin, bovine serum albumin, medium M199, tribromoethanol, sodium bicarbonate, paraformaldehyde, borate (H3BO4), and IodoGen (1,3,4,6-tetrachloro-3α,6α-diphenylglycouril) were purchased from Sigma. Dipalmitoylphosphatidylcholin was bought from Lipoid (Ludwigshafen, Germany), and distearoylphosphatidylcholin (DSPE)-PEG2000 (DSPE-PEG) and Rhodamine-PE from Avanti Polar Lipids, Inc. (Alabaster, AL, U.S.A.). The DSPE-PEG-malemide was custom-synthesized by Shearwater (Huntsville, AL, U.S.A.). Protein concentrations were determined with the Bio-Rad RC DC protein assay kit (Bio-Rad, Hemel Hempstead, U.K.). Rabbit anti-laminin, fluorescein-5-isothiocyanate—conjugated goat anti-rabbit, and biotinylated rabbit anti—mouse IgG were from Dako (Dako, Glostrup, D.K.). Alexa 488 conjugated rabbit anti—mouse IgG came from Molecular Probes (Eugene, OR, U.S.A.). Horseradish peroxidase-conjugated streptavidin—biotin complex was from Vectastain (Vector Laboratories, Burlingame, CA, U.S.A.). TSA Indirect (NEN, Boston, MA, U.S.A.) was used for tyramide amplification. DePeX was purchased from British Drug House (Poole, U.K.).
Experimental procedures
Uptake studies were performed using P18 male rats (n = 3 per time point; mean ± SD body weight, 32.0 g ± 2.30 g; mean ± SD brain weight, 1.41 g ± 0.09 g), which were either perfused in situ according to Roberts et al. (1993) or injected intravenously. For in situ brain perfusion, the rats were deeply anesthetized with an intraperitoneal injection of 0.2 mL Avertin per 10 g animal (1.87 g tribromoethanol dissolved in 2 mL 99% ethanol added to 98 mL distilled water and heated until completely dissolved). The chest was opened surgically, and the rats were perfused transcardially with 5 mL/min for 2, 5, or 15 minutes with medium M199 to which distilled water and 2 mg/mL bovine serum albumin were added. The pH was adjusted to 7.4 with sodium bicarbonate. The M199 solution was kept at 37°C by placing it in a heating bath and oxygenated continuously with bubbling gaseous 100% connected to O2 plastic tubing at a pressure of 1 atm. For the intravenous injection, rats were briefly anesthetized with 0.5% Avertin (0.07 mL intraperitoneally per 10 g animal) and injected with a 30-G needle into a lateral tail-vein. These rats were allowed to live from 5 to 120 minutes after the intravenous injection. Uninjected P18 male rats (n = 4) and adult male Wistar rats weighing approximately 180 g (n = 4) were examined for the distribution of transferrin receptors in BCECs.
The protein uptake was studied after in situ perfusion with the M199 solution to which OX26, Ni-IgG2a, or human transferrin was added to yield final concentrations of 1.25 μg/mL OX26, 1.25 μg/mL Ni-IgG2a, and 0.9 mg/mL human transferrin in the perfusion solutions, respectively. Human transferrin was used to trace the BCEC rat transferrin receptor because it can be detected by immunohistochemistry. The affinity constant of human transferrin relative to rat transferrin for the rat transferrin receptor is 1.04 (i.e., slightly higher than that of endogenous rat transferrin) (Lim et al., 1987). To evaluate the morphologic integrity of the BBB against macromolecules during in situ perfusion, rats were perfused for 15 minutes with 0.1 mg/mL human albumin.
For the studies of the OX26–liposome complex uptake, liposomes were prepared as described below and were either conjugated to OX26 or examined unconjugated. They were then added to 250 mL of the M199 solution to reach final concentrations of 1.06 μg OX26 per 0.01 μmol phospholipids per milliliter perfusate (OX26–liposomes) or 0.08 μmol phospholipids per milliliter perfusate (unconjugated liposomes) and perfused for 15 minutes. For intravenous studies, either 198 μg OX26 per 2.01 μmol phospholipids (OX26–liposomes) or 5.97 μmol phospholipids (unconjugated liposomes) dissolved in PBS (pH 7.4) was injected into the tail vein of anesthetized rats, which were allowed to survive for 2 hours. Experiments were terminated by transcardial perfusion of the deeply anesthetized rats using 4% paraformaldehyde in 0.1-mol/L PBS (pH 7.4).
Quantification of OX26 uptake
The OX26 uptake by BCECs in the in situ perfusion model was quantified by using Ni-IgG2a as a nonspecific uptake marker. OX26 and Ni-IgG2a were iodinated with 125I using IodoGen (Fraker and Speck, 1978). One microgram IodoGen was dissolved in chloroform (10 μg/mL) and dried to the bottom of a plastic tube at room temperature in a container kept free of oxygen by a continuous application of gaseous 100% N2. Another tube was used to mix 100 to 250 μg protein with 100 μL 0.2-mol/L borate buffer (pH 8.0), which was prepared by adding 0.2-mol/L H3BO4 with 0.16-mol/L NaCl, NaI (2atoms per molecule of protein), and 250 Ci 125I. The protein solution was transferred to the IodoGen-containing tube and mixed gently for 1 minute. The solution was then transferred to a gel to separate the protein-containing fraction from the non—protein-bound iodine. The presence of labeled protein was verified by counting samples to which 10% trichloroacetic acid was added. The concentration of labeled protein in the M199 solution was calculated, based on a 95% recovery of protein. The proteins were added separately to the M199 solution and perfused at 2, 5 and 15 minutes in concentrations of 0.3 to 0.6 μg/mL. The perfusion was terminated by flushing the rats for 60 seconds with 0.1-mol/L PBS (pH 7.4) at 37°C to empty vessels of radioactive content.
The presence of [125I]OX26 in the brain parenchymal fraction was evaluated after separating brain capillaries from the remaining brain tissue (Moos and Morgan, 2001;Triguero et al., 1990). In brief, brains were homogenized by four strokes in a dounce homogenizer in 3.5 mL of ice-cold buffer (10-mmol/L HEPES, 141-mmol/L NaCl, 4-mmol/L KCl, 2.8-mmol/L CaCl2, 1-mmol/L MgSO4, 1-mmol/L NaH2PO4, and 10-mmol/L glucose; pH 7.4). The homogenate was mixed with 3.5 mL ice-cold 26% dextran (molecular weight, 60.000; Sigma) and homogenized by another four strokes. The dextran-containing solution was centrifuged at 2,830 g for 30 minutes in a Swing Bucket Centrifuge (Beckman) at 4°C. The resulting supernatant and pellet were separated manually and counted for radioactivity.
The specificity of the separation was evaluated with an enzymatic assay for alkaline phosphatase (EC 3.1.3.1) that is only expressed in BCECs in the brain (Moos and Morgan, 2001). In brief, 0.1-mL suspensions of pellets and supernatants isolated by the capillary-depletion technique were added to a buffer consisting of 50-mmol/L MgCl2, 5-mmol/L CaCl2, 100-mmol/L KCl, 5-mmol/L p-nitrophenyl phosphate, and 100-mmol/L Tris (pH 9.0) in a 0.9-mL volume for 20 minutes at 37°C. The reaction was stopped by adding 0.2 mL 5-mol/L NaOH. Insoluble material was removed by spinning for 10 minutes at 3,000 g. Absorbance was determined at 420 nm and activity converted to nanomoles per minute per milligram protein using the activity of purified alkaline phosphatase (Sigma P-7640) in dilutions of 0.1 to 20 μg/mL to draw a standard curve of linear activity.
Preparation of the OX26–liposome conjugate
Thiolation of OX26.
Sulfhydryl groups were introduced to OX26 with SATA according to the protocol developed by Duncan et al. (1983). In brief, OX26 was thiolated with SATA dissolved in N,N,-dimethylformamide (1.2 mg/mL) in a molar ratio of SATA/OX26 of 8:1. The solution was incubated for 45 minutes at room temperature. Next, OX26 was concentrated and free SATA removed by Vivaspin (Vivascience AG, Hannover, Germany) centrifugation with a 50-kd cutoff. The derived OX26 monoclonal antibody was stored at −20°C.
Liposomes.
Dipalmitoylphosphatidylcholin (80 μmol), DSPE-PEG2000 (6.4 μ mol), cholesterol (43 μmol), and Rhodamine-PE (0.12 μmol) were dissolved in 1 mL chloroform. For conjugating of OX26 to liposomes, 3.2 μmol of a maleimide-derivate linker lipid (DSPE-PEG-maleimide) containing DSPE on one end and a sulfhydryl-reactive maleimide at the other was added to the lipid mixture together with 3.2 μmol DSPE-PE. A lipid film was prepared by vacuum evaporation, dried, and rehydrated at 50°C in a 3-mL solution consisting of 10-mmol/L HEPES, 136-mmol/L NaCl, and 1-mmol/L EDTA (pH 7.4) to yield a final lipid concentration of 40 mmol/L. Liposomes were sized by 10-fold extrusion through two stacked 100-nm polycarbonate membranes at room temperature using a high-pressure extruder. The phospholipid phosphor concentration of each liposome preparation was determined with a phosphate assay that involves perchloric acid—mediated destruction of the phospholipids to anorganic phosphate (Rouser et al., 1970). The size and distribution of the particles were determined by dynamic laser light scattering using a Nicomp model 370 submicron particle analyzer (Nicomp, Santa Barbara, CA, U.S.A.).
Coupling OX26 to liposomes.
The malemide-grafted liposomes were immediately conjugated to thiolated OX26 according to a sulfhydrylmalemide coupling method (Duncan et al. 1983). The sulfhydryl groups were reactivated by deacetylating the acetylthioacetate—antibody complex for 1 hour by adding 100 μL of freshly prepared 0.5-mol/L hydroxylamine HCl, 0.5-mol/L HEPES, and 25-mmol/L EDTA (pH 7.0) per milliliter protein solution. Deacetylated OX26 and maleimide-grafted liposomes were incubated at room temperature for 2 hours and thereafter at 4°C overnight. Unconjugated antibody was separated from the liposomes by ultracentrifugation. The liposome—protein solution was ultracentrifuged twice at 240,000 g for 1 hour, removing the supernatant in between. Immunoliposomes were characterized by determining the concentration of phospholipids (Rousser et al., 1970), protein concentration, and particle size. The protein concentration was determined with the Bio Rad RC DC protein assay kit. The particle size was determined by dynamic laser light scattering. The OX26–immunoliposomes and PEG—liposomes had a particle size of 166 nm and 141 nm, respectively. The increase in particle size after OX26 had been conjugated to the liposomes is due to the fact that OX26 attaches to the distal end of the PEG—liposome.
Morphologic analyses
Diaminobenzidine immunohistochemistry.
The brains were dehydrated in 30% sucrose and cut into serial, coronal 30-μm sections on a cryostat. The cryostat sections were quenched for endogenous peroxidase activity by incubating them in 1% H2O2 in PBS for 15 minutes. They were then incubated in normal swine serum to block nonspecific binding by the antibodies and subjected to immunohistochemistry. The sections were incubated overnight at 4°C with one of the following primary antibodies: (1) OX26 (MAS 262b) diluted 1:100, and (2) monoclonal anti—human transferrin diluted 1:100 and human albumin 1:1,000, both of which specifically detect the human proteins, leaving the respective rat proteins unlabeled (Moos and Morgan, 1998). These antibodies and injected OX26 and Ni-IgG2a were detected with biotinylated rabbit anti—mouse IgG diluted 1:500 for 30 minutes at room temperature, followed by incubation with horseradish peroxidase—conjugated streptavidin—biotin complex diluted according to the manufacturer's recommendations. Adjacent sections were additionally subjected to tyramide amplification diluted 1:100 for 5 minutes followed by horseradish peroxidase—conjugated streptavidin—biotin complex for 30 minuets. Finally, the sections were developed in diaminobenzidine and 0.01% H2O2 in 0.05-mol/L Tris buffer (pH 7.6) for 10 minutes. The cryostat sections were mounted on slides using 0.5% gelatin in distilled water and dried. The sections were coverslipped with DePeX.
Immunofluorescence histochemistry.
Sections of rats, perfused or injected with OX26, were processed with biotinylated rabbit anti—mouse IgG diluted 1:300 for 30 minutes at room temperature followed by incubation with Texas Red—conjugated streptavidin (for 30 minutes at room temperature) and tyramide amplification as described earlier. The sections were double-stained with an antilaminin antibody that detects the basal membrane of BCECs. The sections were incubated with rabbit antilaminin (Dako) diluted 1:100 overnight followed by fluorescein-5-isothiocyanate—conjugated goat anti—rabbit IgG diluted 1:50 for 30 minutes at room temperature (Dako). To detect OX26 and laminin simultaneously, the fluorescence of Texas Red and fluorescein-5-isothiocyanate was examined in a Confocal Laser Scanning Microscope (Zeiss LSM 510 Meta, Germany). Sections of rats perfused or injected with OX26-conjugated liposomes were processed for OX26 using Alexa 488–conjugated rabbit anti—mouse IgG diluted 1:300 for 30 minutes at room temperature. Simultaneous detection of OX26 and liposomes were examined via the fluorescence of Alexa 488 and Rhodamine in a fluorescence microscope (Zeiss Axiophot, Germany).
Calculations
The radioactive samples were precipitated with 10% trichloroacetic acid (TCA) and counted in a Cobra auto γ-scintillation counter (Packard, Albertville, MN, U.S.A.) for their content of protein-bound 125I. Uptake of OX26 and Ni-IgG2a was calculated as the percentage of the dose used for the in situ perfused brain. The brain uptake of OX26 and Ni-IgG2a was further analyzed in a simple linear two-compartment model, and the volumes of distribution (VD) for OX26 and Ni-IgG2a were calculated as the amount of trichloroacetic acid—precipitable radioactivity for the total brain homogenate, the pellet (BCECs), and supernatant (brain parenchyma) using the following equation (Tiguero et al., 1990):
The unidirectional transfer coefficient Kin (in milliliters per gram per minute) was estimated using the following equation (Chen et al., 2002):
where VD is the volume of distribution, Vvasc is the brain capillary volume, and t is the net perfusion time. Flushing the brains with 0.1-mol/L PBS after perfusing them with the proteins permits the exclusion of Vvasc from the equation (Chen et al., 2002); therefore, the Kin was measured as (VD)/t. Statistical evaluation of the data was performed by analysis of variance. When differences were detected (P < 0.05), means were tested using the Student's Newman-Keuls test for differences between individual means.
RESULTS
Immunohistochemical distribution of transferrin receptors in BCECs
Transferrin receptor immunoreactivity was observed in BCECs of brains from both P18 and adult rats (Fig. 1). The immunoreactive BCECs were observed throughout the CNS, with the exception of those situated within circumventricular organs. The transferrin receptor immunoreactivity in BCECs was more pronounced in P18 rats, although still discernable in both gray and white matter regions of adult rats (Fig. 1).
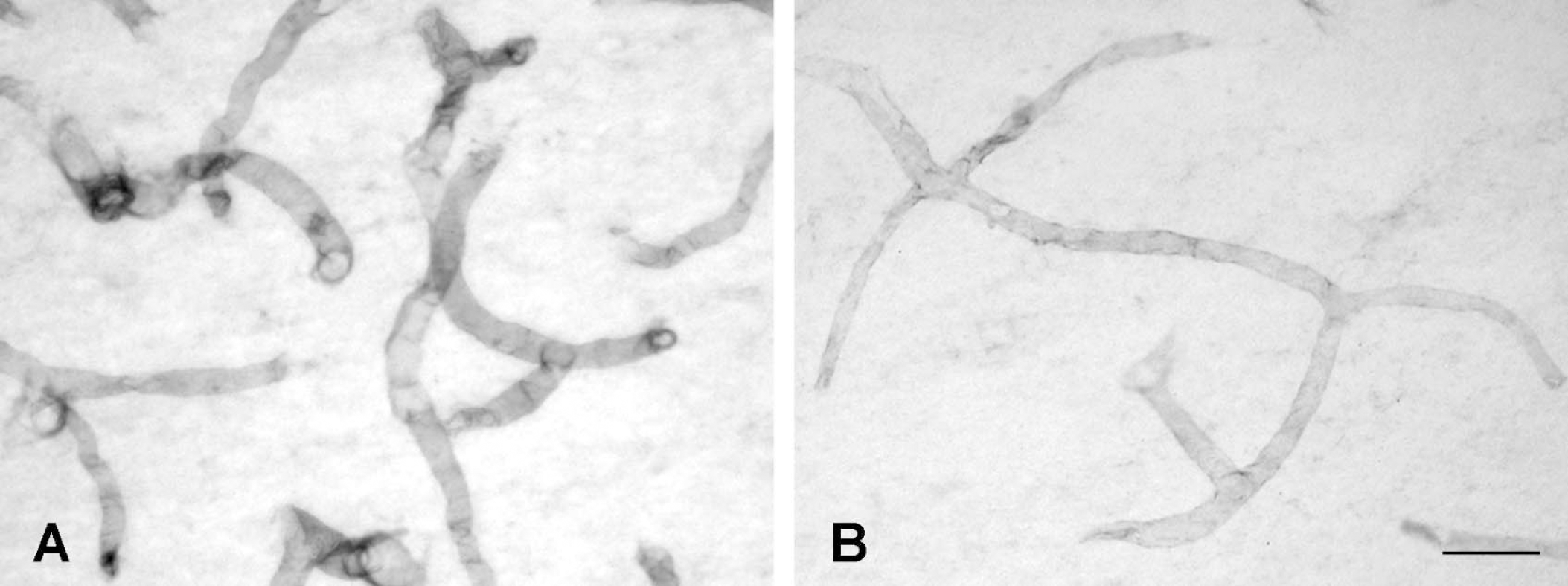
Transferrin receptor expression in BCECs of the striatum as verified by immunohistochemical analysis. Transferrin receptor expression is higher in BCECs of the P18 rat (
Quantification of OX26 uptake
The total uptake of [125I]OX26 increased from 22 μL/g brain at 5 minutes to a significantly higher level of 65 μL/g brain after 15 minutes (Fig. 2), indicating that a prolongation of the perfusion time increased the uptake of OX26. By 15 minutes, the uptake of [125I]OX26 was also significantly higher than that of [125I]Ni-IgG2a at 15 minutes (Fig. 2).
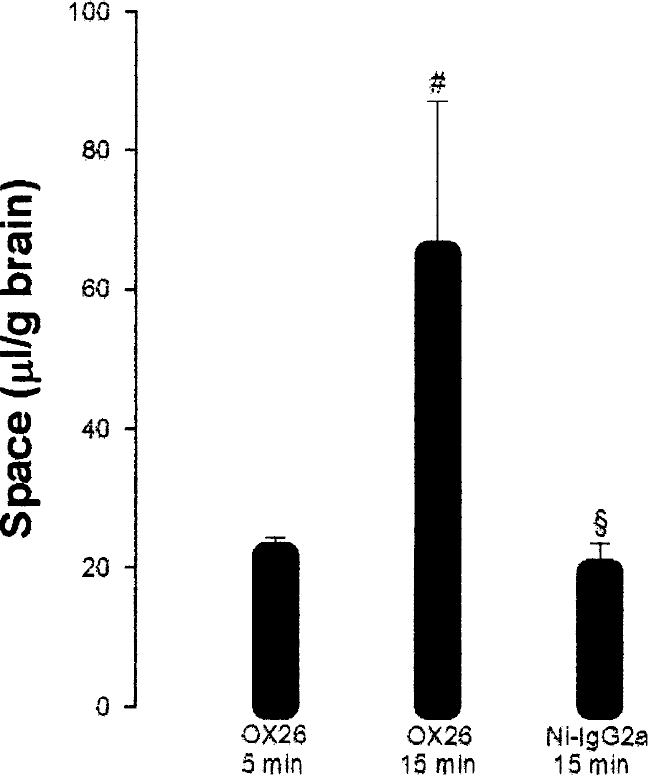
Uptake of OX26 and Ni-IgG2a in the P18 brain using in situ brain perfusion expressed as space (microliters per gram brain). #By 15 minutes, the accumulation of OX26 in the P18 brain is significantly higher than by 5 minutes. § The uptake of OX26 by 15 minutes is also higher than of Ni-IgG2a.
The capillary-depletion technique provided two different brain fractions (i.e., pellet and supernatant) containing the vascular and remaining brain fractions, respectively. In the vascular fraction, the VD value of [125I]OX26 was 18.4 μL/g after 5 minutes of perfusion, which increased significantly to 56.1 μL/g after 15 minutes. The fraction of [125I]OX26 in the supernatant was significantly smaller than in the pellet, but increased significantly from between 5 and 15 minutes. However, the fraction of radioactivity found in the supernatant and the total brain remained constant between 14% and 16.3% (Table 1).
Uptake of OX26 expressed as VD(μL/g brain) in fractions of pellet and supernatant using the brain capillary depletion experiment
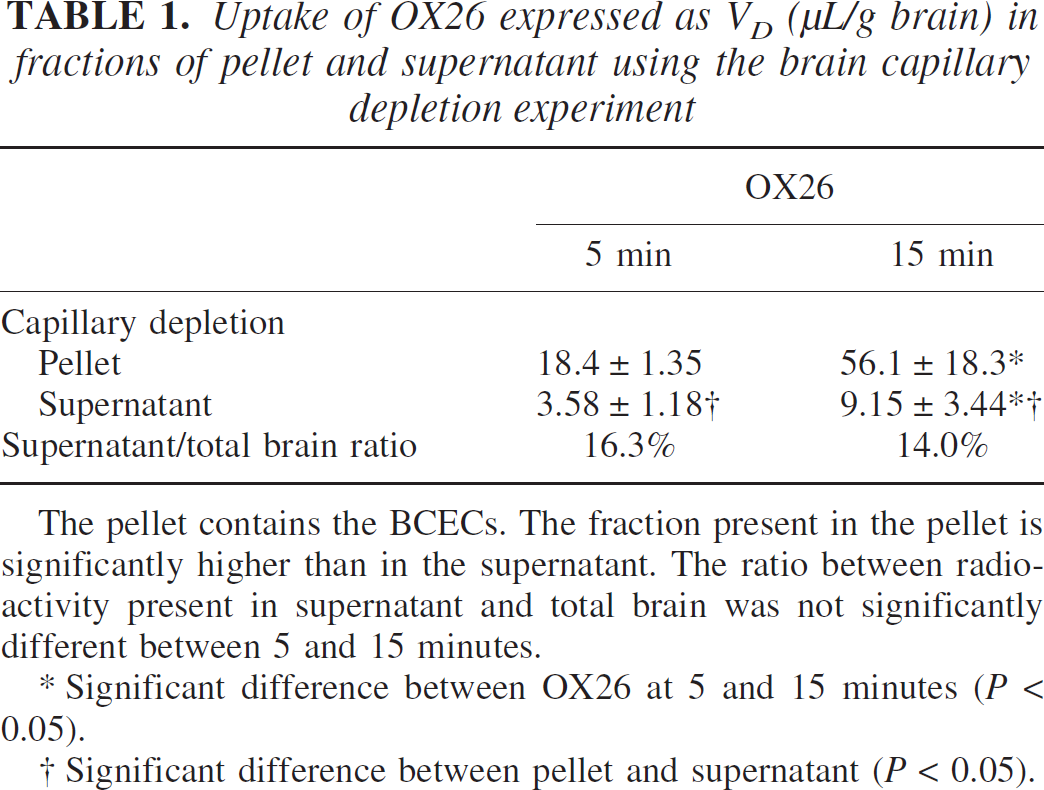
The pellet contains the BCECs. The fraction present in the pellet is significantly higher than in the supernatant. The ratio between radio-activity present in supernatant and total brain was not significantly different between 5 and 15 minutes.
Significant difference between OX26 at 5 and 15 minutes (P < 0.05).
Significant difference between pellet and supernatant (P < 0.05).
The BCECs selectively express the enzyme alkaline phosphatase in the brain, which indicates that the activity of this enzyme can be used to determine the contamination of the supernatant with BCECs that fail to precipitate during the dextran density centrifugation used in the capillary depletion technique (Triguero et al., 1990). Analyses of the pellet and supernatants for the BCEC-specific enzyme alkaline phosphatase revealed that the activity in the pellet and supernatant fractions corresponded to 10.5 ± 1.87 and 1.98 ± 0.28 μg/mL of alkaline phosphatase activity, respectively. Hence, 15.8% ± 0.13% of the alkaline phosphate activity was found in the supernatant fraction.
The Kin value describes the rate of protein uptake by the brain. The rate of OX26 uptake by the brain was found to be the same after 5 and 15 minutes of perfusion (Table 2). The Kin for OX26 was higher as compared with Ni-IgG2a at 15 minutes (Table 2).
The rate of uptake of OX26 and Ni-IgG2a expressed as the transfer constant Kin (mL · kg−1 · min−1)

Significant difference between OX26 at 5 or 15 minutes and Ni-IgG2a at 15 minutes (P < 0.05).
Morphologic analysis of protein uptake by BCECs
OX26.
Like the endogenous transferrin receptor, OX26 perfused in situ consistently labeled BCECs throughout the CNS, with the exception of those of circumventricular organs (Fig. 3). BCECs were labeled at all examined time points with the same intensity. However, when examined at high magnification, OX26 depicted an immunoreaction product that was confined to small, granular elements distributed diffusely within the cytoplasm of BCECs examined only at 15 minutes (Figs. 3C and 3D). A careful examination of the brain sections failed to identify other labeled cells in the brain except for those of BCECs. In intravenously injected rats, OX26 labeled BCECs diffusely in the brain, which corresponds to the findings reported recently (Moos and Morgan, 2001). In contrast to the observations in the in situ perfused brain, the intravenous injection of OX26 revealed a labeling of the choroid plexus epithelial cells.
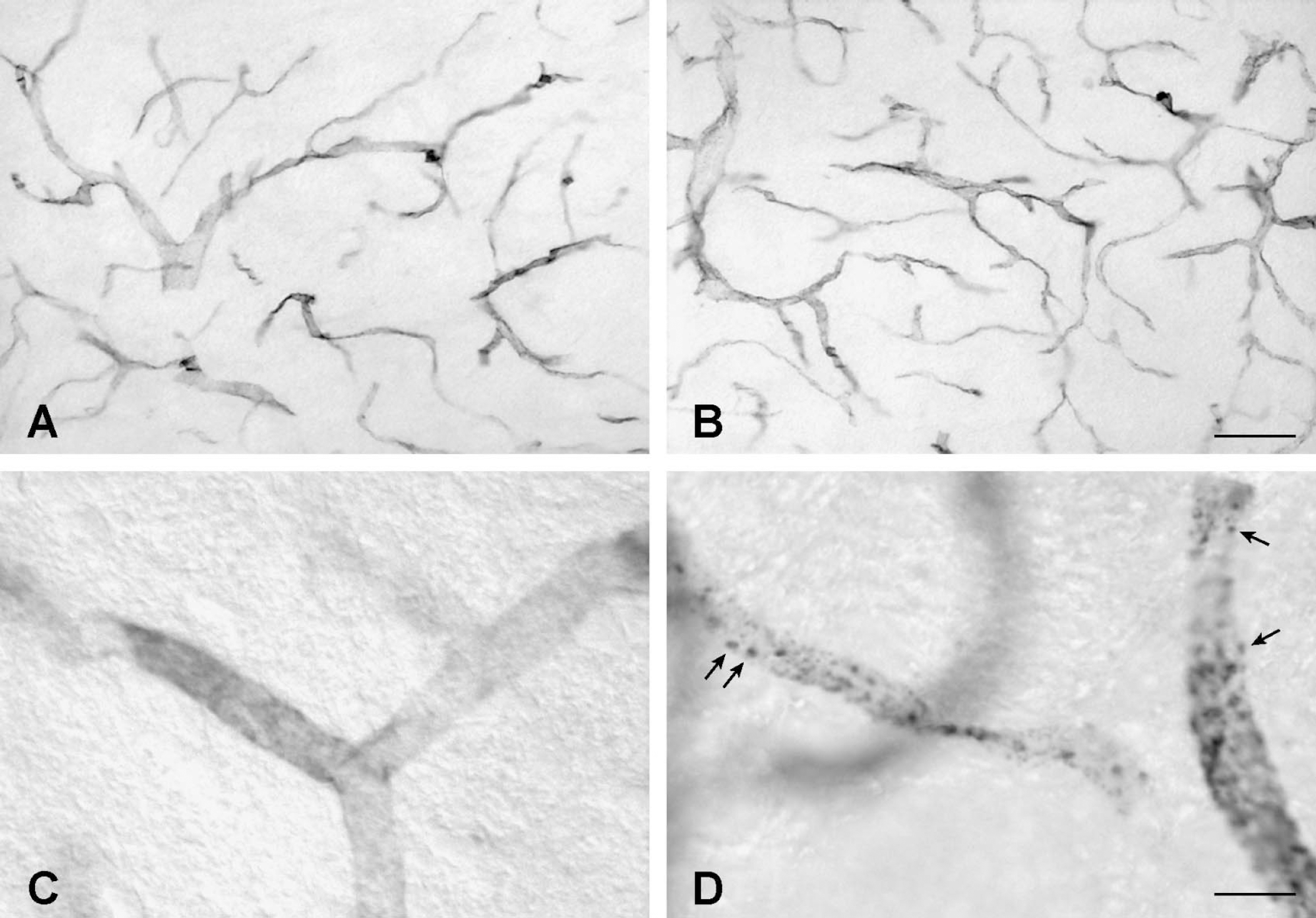
Distribution of OX26 in the P18 rat brain examined at 5 minutes (
Using double-labeling fluorescence, OX26 distributed differently from that of laminin (Fig. 4A–Fig. 4D). Confocal imaging made it apparent that the BCEC distribution of OX26 was different from that of laminin, suggesting that BCEC did not associate with the basal membrane as verified by the existence of a gap in fluorescence between the Texas Red—detecting OX26 and the fluores-cein-5-isothiocyanate—detecting laminin (Fig. 4E–Fig. 4H).
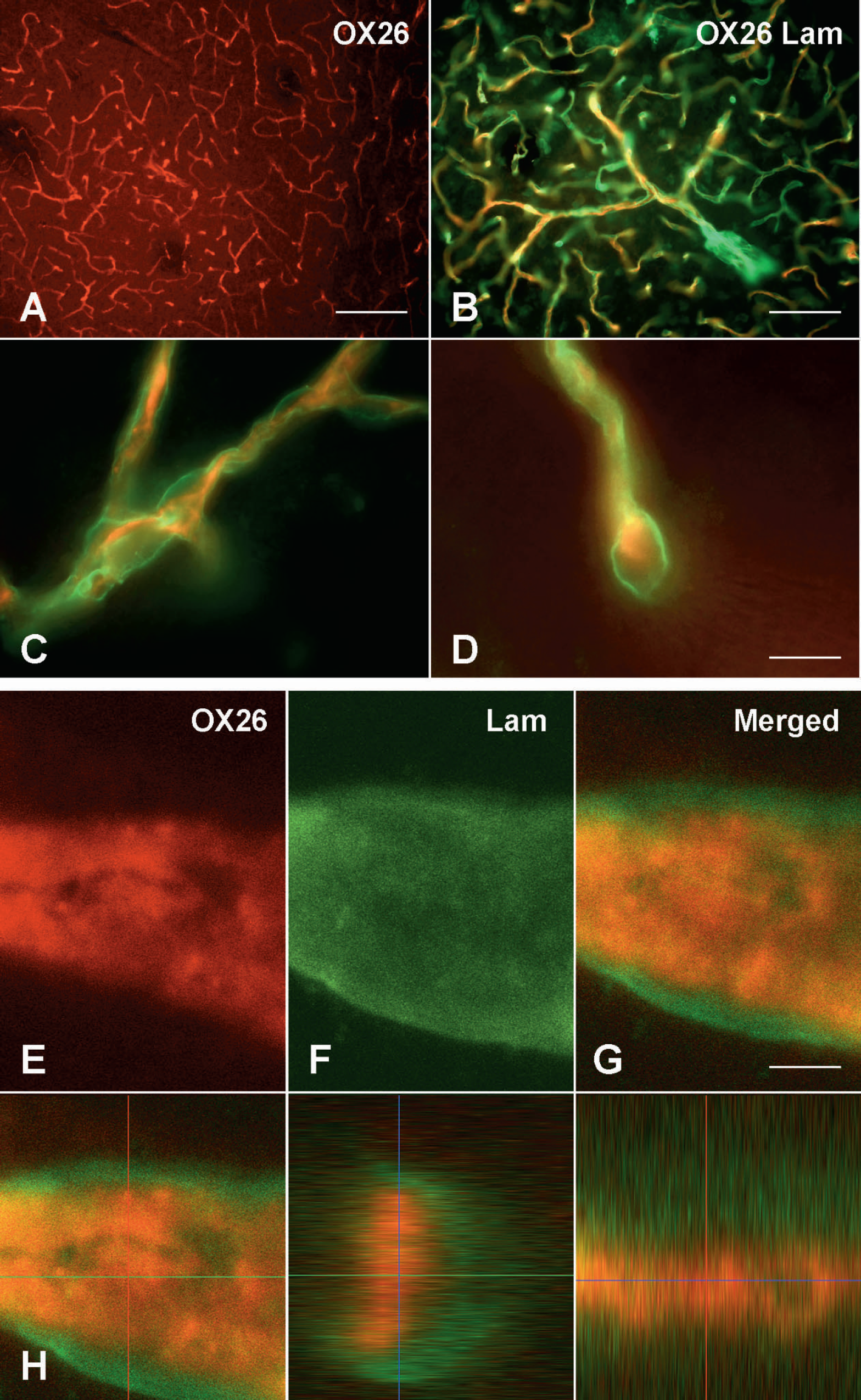
Sections from a P18 rat subjected to in situ perfusion with OX26 for 15 minutes and examined for OX26 (red) and laminin (green). (
Ni-IgG2a and human albumin.
Ni-IgG2a and human albumin were not present intracellularly in BCECs or perivascularly when examined up until 15 minutes after in situ brain perfusion (Figs. 5A and 5B). The absence from the perivascular core of human albumin, which was perfused in a concentration 80 times higher than that of OX26, confirmed the integrity of the BBB against macromolecules.
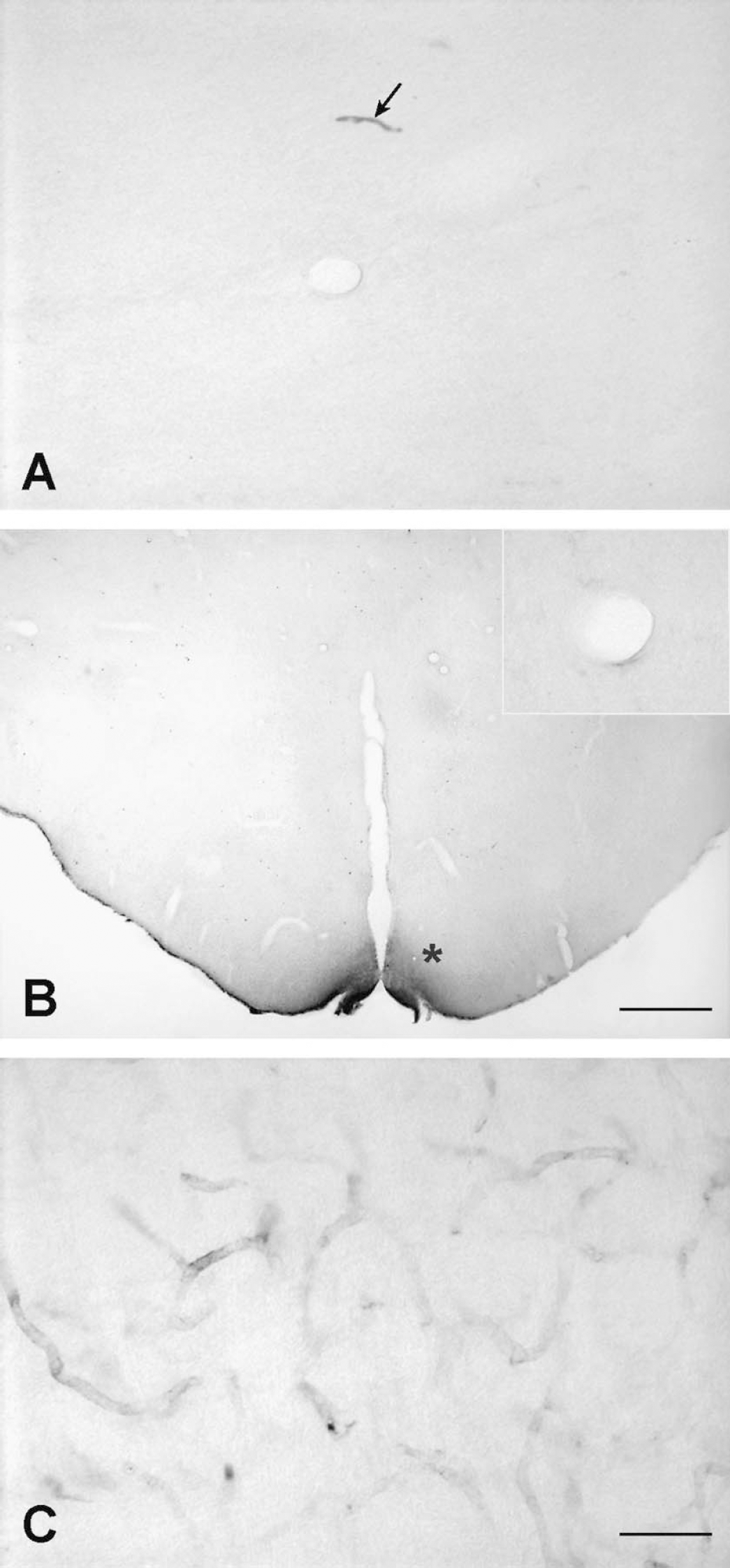
Distribution of Ni-IgG2a (
Human transferrin.
Similar to OX26, human transferrin labeled endothelial cells throughout the brain (Fig. 5C). In contrast to OX26, the labeling intensity obtained by the anti—human transferrin antibody was markedly lower, even though the amount of human transferrin was almost 20 times higher. Human transferrin was not found in any cell types other than BCECs in the brain.
Distribution of the OX26–liposome conjugate within BCECs
OX26 of the OX26–liposome conjugate detected by the Alexa 488–conjugated secondary antibody distributed to the brain in a pattern similar to that of unconjugated OX26. Fluorescence represented by Rhodamine-containing liposomes was confined to BCECs in a distributional pattern similar to that of OX26, leaving neurons and glia unlabeled (Figs. 6A and 6B). Examination of unconjugated OX26 detected by Alexa 488–conjugated anti—mouse IgG (Fig. 6C) revealed a complete absence of fluorescence when examined in the fluorescence filter used to detect Rhodamine (Fig. 6D), indicating that the fluorescence of the Rhodamine-containing liposome indeed colocalized with OX26. Brains of rats perfused in situ with unconjugated liposomes were devoid of any fluorescence that could be attributed to specific fluorescence representing liposomes (Fig. 6E). In brains of rats injected intravenously, BCECs were labeled with OX26 and liposomes in a pattern similar to that observed in in situ perfused brains.
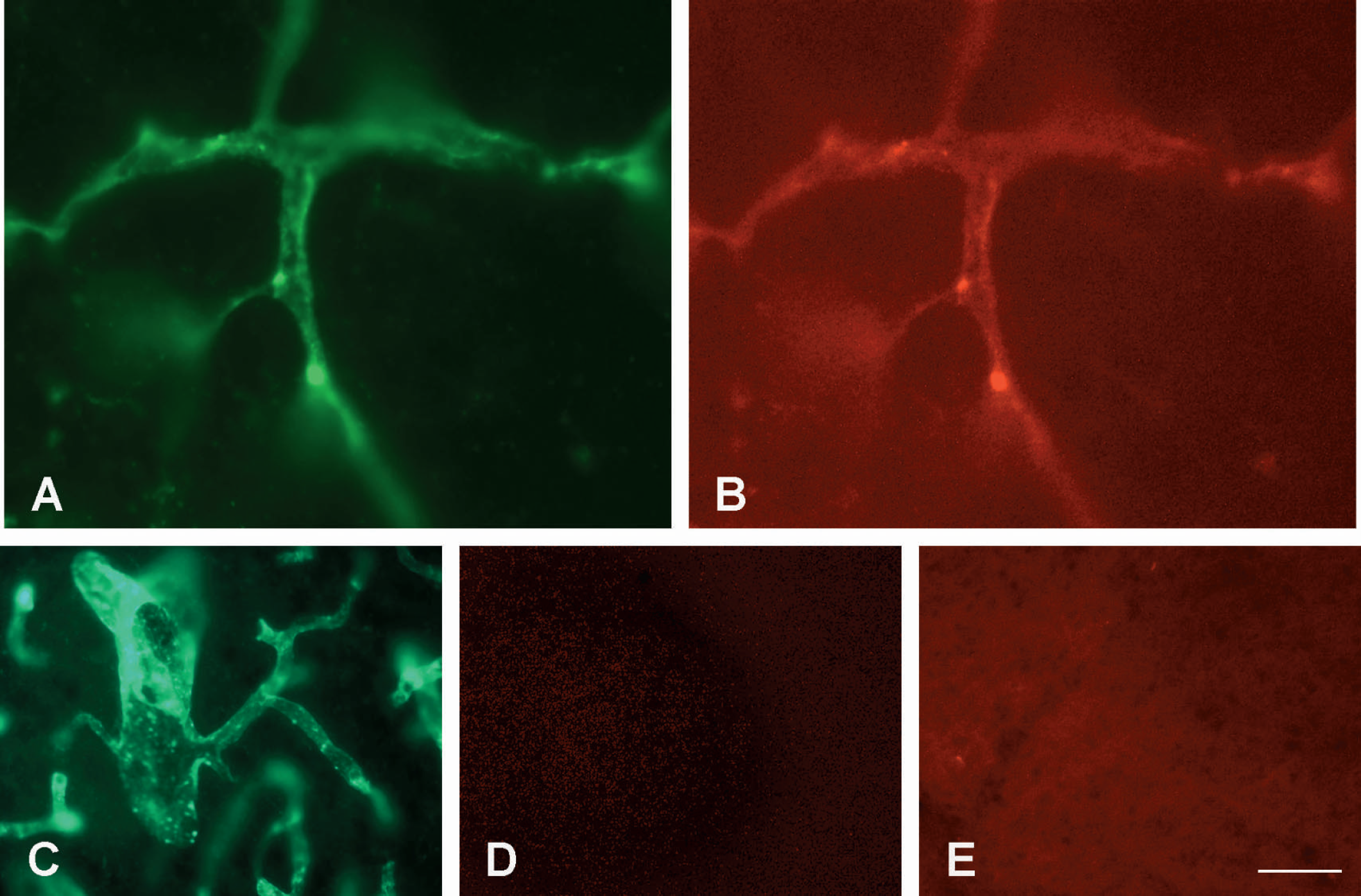
Uptake in BCECs after in situ brain perfusion with an OX26–liposome conjugate (
DISCUSSION
Perfusing the rats with radiolabeled OX26 and Ni-IgG2a revealed a significant difference in uptake by the brain, supporting the concept of facilitated uptake of OX26 in BCECs due to its attachment to transferrin receptors (Friden et al., 1991;Pardridge et al., 1991). The uptake of OX26 by the brain parenchyma fraction was significantly lower than the uptake by BCECs, suggesting that OX26 gets trapped and concentrated in the BCECs. The percentage of radiolabeled OX26 in the postcapillary fraction of the brain capillary depletion assay was not higher than in the corresponding fraction of the alkaline phosphatase assay. This indicates that the brain capillary depletion technique resulted in approximately 15% pollution of the postcapillary fraction due to artificial leakage of OX26 from the BCECs into the supernatant. Accordingly, the 84% to 86% fraction of the OX26 radioactivity in the brain was found in the BCEC fraction, which indicates that OX26 is not transported through the BBB. BCECs of the adult rat brain have a volume of approximately 1 μL (Pardridge, 2001), which corresponds to 0.56 μL/g brain as the brain of the adult rat weighs approximately 1.80 g (Moos and Morgan, 2001). The volume of distribution of OX26 in BCECs after 15 minutes of in situ brain perfusion was estimated to 56.1 μL/g brain (Fig. 2), which shows that OX26 taken up by the brain gets concentrated approximately 100 times within BCECs.
The absence of neuronal or glial labeling by OX26 or immunoliposomes also favors that OX26 accumulates within BCECs. This finding is in good accordance with that of a previous study on OX26 uptake in the intact rat brain that failed to detect OX26 injected intravenously beyond the BBB, except for in CSF and a few neurons situated periventricularly (Moos and Morgan, 2001). One explanation for this difference is that the presence of OX26 in CSF, as observed by Moos and Morgan (2001), probably relies on transport through the blood—CSF barrier and not on transport through the BBB, and that periventricular neurons take up OX26 in axons projecting to the ventricular system (Moos and Morgan, 2001;Moos, 2003). The present study found OX26 in circumventricular organs, indicating that OX26 passes the fenestrated endothelium, including that of the choroid plexus, although the latter was not labeled sufficiently to yield immunohistochemical detection.
Ni-IgG2a was used as a control for the BBB integrity and specificity of the brain uptake. The uptake of Ni-IgG2a by the brain was significantly lower than that of OX26 at 15 minutes. Performing in situ perfusion for longer than 15 minutes may lead to unacceptable hypoxic-mediated degeneration of the BBB, which prevents examinations for longer time intervals (Roberts et al., 1993). We did not observe unacceptable background staining at 15 minutes with antibodies for human albumin or Ni-IgG2a. In addition, the VD of Ni-IgG2a after 15-minute perfusion was comparable with VD of sucrose 10 minutes after in situ perfusion of the intact rat (Skarlatos et al., 1995).
OX26 uptake by BCECs
In vivo studies have revealed that the kinetics of the uptake of OX26 and transferrin by BCECs are similar, both increasing rapidly during the first 30 to 60 minutes after injection with little further change (Friden et al., 1991;Moos and Morgan, 2001;Taylor and Morgan, 1990). Together with these results, thevalues for Kin OX26 uptake after 5 and 15 minutes of in situ perfusion were similar and much higher than those observed after 120 minutes in vivo (Moos and Morgan, 2001), suggesting that OX26 continuously takes up by BCECs because the transferrin receptors are not yet saturated. The rats were perfused with OX26 without transferrin that competes with the BCEC transferrin receptor (Ueda et al., 1993). Thus, the difference in experimental approach probably also explains why the in is higher when taking the in situ approach.
The fate of OX26 bound to the BCEC transferrin receptor remains unresolved, but the present study and a previous study on intravenously injected rats (Moos and Morgan, 2001) suggest that OX26 remains bound to the receptor. The transferrin receptor and OX26 form a high-affinity antibody—antigen interaction that is not easily reversible (Morgan, 1996). This raises the question whether OX26 can be released from the receptor within endosomes to enter the postcapillary fraction of the brain. Once inside the BCECs, OX26 probably enters endosomes after their internalization by the transferrin receptor. Supporting this hypothesis, the cellular distribution becomes more punctate with increasing time, which was also the case in a previous study that detected transferrin in endosomes after in situ perfusion (Roberts et al., 1993). Possibly, the acidic pH of the endosomal interior could weaken the interaction between OX26 and the transferrin receptor, leading to the release of a fraction of OX26 followed by transport across the luminal surface by a transcytotic mechanism. However, there are many observations that contradict this hypothesis. In vivo studies failed to detect OX26 in the postcapillary compartment when the brain capillary depletion technique was used, or in neurons or glia subsequent to immunohistochemical mapping (Moos and Morgan, 2001). Likewise, the present study failed to detect OX26 except inside BCECs, even though transferrin was experimentally cleared from the perfusate to prevent interaction with the binding of OX26 to the BCEC transferrin receptor. Confocal microscopy failed to associate OX26 with laminin situated near the abluminal surface of BCECs. The presence of OX26 in the postvascular compartment did not show any quantitative evidence for transcytosis.
A marked difference in the intensity of the immunoreaction was observed between brains of rats perfused with OX26 and transferrin. The immunoreactivity of OX26 was much higher, even though the concentration of transferrin in the perfusion fluid was more than 700 times higher, albeit still within the physiologic range. This difference is undoubtedly due to differences in the chemical nature of transferrin and OX26's respective binding to the transferrin receptor.
Basic fibroblast growth factor conjugated to OX26 targets and protects neurons from ischemia, possibly via receptor-mediated transport through BCECs of bFGF—OX26 (Song et al., 2002). Interestingly, the cerebral endothelium increases its expression of transferrin receptors during ischemia (Omori et al., 2003), probably due to increased expression of hypoxia-inducing factor known to increase the expression of the transferrin receptor mRNA (Lok and Ponka, 1999). An increased transferrin receptor expression could provide enhanced uptake of bFGF—OX26 by BCECs, thereby promoting an effect of bFGF on the ischemic endothelium. An ischemic insult, however, also compromises the BBB integrity profoundly (Abbruscato and Davis, 1999;Plateel et al., 1997). Neurons express receptors for transferrin receptors (Moos, 1996). These facts support the notion that bFGF—OX26 provides its protective effect via transport through a compromised blood—brain interphase, with OX26 targeting directly to the neuronal surface and subsequently producing the bFGF-mediated neuroprotective effect.
BCEC-to-brain transport
Clearly the receptor-mediated targeting to BCECs qualifies as a method to transfer liposomes and their cargo into BCECs, whereas the capacity of liposomes to travel to the postvascular compartment of the brain is questionable (Jiang et al., 2002; the present study). Cerletti et al. (2000) studied BBB transport of liposomes with encapsulated anticancer drugs using immunoliposomes formulated with a similar design to those used in the present study. [125I]OX26 labeled liposomes in the postvascular compartment after 7 minutes of in situ perfusion detected by scintillation counting, giving rise to the idea of transcytosis of immunoliposomes (Cerletti et al., 2000). The conclusion of Cerletti et al. (2000), however, can be challenged. Control experiments were not made to ensure the presence of BCECs’ enzyme activity in the vascular fraction of the capillary purification technique. Moreover, no immunohistochemical proof was provided to show any uptake of OX26 in cells beyond the BBB.
Given that OX26-coated liposomes probably distribute selectively to endosomes, the challenge of allowing liposomes and their cargo to release into the brain interior relies on the establishment of mechanisms that will either allow the entire immunoliposome to transport through BCECs or allow the liposome to release its cargo to diffuse deeper into the brain. One so-far-unexploited approach for allowing the liposomal content to escape its encapsulation, and possibly transport deeper into the brain, would be to redesign liposomes making them fragile, once they are taken up by BCECs. Specifically designed liposomes can be made pH sensitive (Gerasimov et al., 1999;Mastrobattista et al., 2002a, b), which would theoretically allow for degradation and release once they enter the endosome, whose slightly acidic environment is advantageous (Morgan, 1996). Subsequent to their liposomal escape, the cargo might be transported into the brain via fusion of endosomal and abluminal plasma membranes.
Another approach towards transport through the BBB would be to transfect BCECs in vivo with plasmids encoding the relevant protein (Jiang et al., 2002). Jiang et al. (2003) recently showed that subsequent to perfusion of the entire rat brain with liposomes injected into the carotic artery, transfected BCECs were able to secrete glial-derived neurotrophic factor into the brain extracellular space. This mechanism can probably not explain the profound physiologic effect of introducing plasmid-encapsulated immunoliposomes coated with targeting antibodies to the diseased rodent or primate brain (Shi et al., 2000; Zhang et al., 2003a, b) because the molecules in charge, tyrosine hydroxylase and beta-galactosidase, are not likely to undergo secretion from transfected BCECs. The absence of OX26 transport through BCECs as observed in the present study and elsewhere (Moos and Morgan, 2001) indicates that plasmids are unlikely to enter the brain by transcytosis of plasmid-loaded immunoliposomes. Instead, alternative routes of entry should be pursued. The studies of Shi, Zhang, and coworkers revealed a profound labeling of the choroid plexus (Shi et al., 2000; Zhang et al., 2003a) using targeting to the transferrin receptor (Shi et al., 2000; Zhang et al., 2003b) or the insulin receptor (Zhang et al., 2003a). The beneficial effect of treating parkinsonian rats with tyrosine hydroxylase-encoding plasmids (Zhang et al., 2003b) may be due to immunoliposomes passing through the choroid plexus and accessing nigral or striatal neurons, many of them situated in limited distance from the ventricular or brain surfaces.
Apart from addressing transport of immunoliposomes through BCECs, drug delivery to the BCECs proper may also be a valid target. OX26-coated liposomes were used to circumvent the luminally localized p-glycoprotein efflux transporter in cancer cells (Huwyler et al., 2002). This approach may also be operative in preventing export of drugs from BCECs back to the circulation. Loading BCECs with relevant drugs, peptides, or genes may also prove suitable for inducing ischemic preconditioning that markedly reduces ischemic damage (Andjelkovic et al., 2003), or for inhibiting proliferation of endothelial cells in cerebral cancer (Marikovsky 2002; Mishima et al., 2001). The recent identification of transcytosis of the soluble form of melanotransferrin (p97) at the BBB (Demeule et al., 2002;Moroo et al., 2003) suggests that immunoliposome targeting to the BCEC p97 receptor rather than transferrin or insulin receptors may prove another valid principle for drug targeting to the brain.
In conclusion, this study showed that BCECs can be loaded with OX26 or OX26-conjugated liposomes, but failed to show their transport through the BBB. The selective BCEC expression of transferrin receptors in BCECs qualifies OX26 as a candidate vector for blood-to-endothelium transport. The possible mechanisms for endothelium-to-brain transport are far from resolved. Targeting drug-loaded liposomes to BCECs may be of significance for treating BCECs to inhibit drug efflux to the circulation, or for inhibiting cell proliferation in cancer growth. Targeting plasmid-loaded immunoliposomes may allow infected BCECs to continuously secrete their compound deeper into the brain.
Footnotes
Acknowledgments
The authors thank Grazyna Hahn, Susan Peters, and Louis van Bloois for their excellent technical assistance.