Abstract
This study presents a quantitative comparison of the time courses and regional distribution of both constitutive HSC73 and inducible HSP72 mRNA expression and their respective encoded proteins between young (3-week-old) and adult (3-month-old) gerbil hippocampus after transient global ischemia. The constitutive expression of HSC73 mRNA and protein in the hippocampus of the young sham-operated gerbils was significantly higher than in the adults. The HSC73 mRNA expression after ischemia in the CA1 layer of young gerbils was greater than in adult gerbils. HSC73 immunoreactivity was not significantly changed after ischemia—reperfusion in adult hippocampus, whereas it decreased in young gerbils. Ischemia—reperfusion led to induction of HSP72 mRNA expression throughout the hippocampus of both young and adult gerbils. HSP72 mRNA induction was more intense and sustained in the CA1 subfield of young gerbils; this was associated with a marked induction of HSP72 proteins and neuronal survival. The transient expression of HSP72 mRNA in the CA1 layer of adult gerbils was not associated with a subsequent synthesis of HSP72 protein but was linked to neuronal loss. Expression of HSP72 mRNA was shifted to an earlier period of reflow in CA3 and dentate gyrus (DG) subfields of young animals. These findings suggest that the induction of both HSP72 mRNA and proteins in the CA1 pyramidal neurons of young gerbils, as well as the higher constitutive expression of HSC73, may partially contribute to higher neuronal resistance of young animals to transient cerebral ischemia.
Heat shock proteins (HSPs) are highly conserved proteins that are induced in cells when exposed to elevated temperature or other types of cellular stresses (Massa et al., 1996; Koistinaho and Hökfelt, 1997). Acting as molecular chaperones is a major function of HSPs (Hendrick and Hartl, 1993). Heat shock proteins have been shown to regulate protein maturation (i.e., protein folding, unfolding, and translocation across membranes) (Hightower, 1991; Nelson et al., 1992; Brodsky, 1996), thereby exerting a protective function by binding to denatured proteins produced under stressful conditions. Some members of the HSP multigene family are constitutively expressed, whereas others are induced under stressful conditions. Constitutive or inducible members, or both, of the 70 kDa HSP family appear to have multiple roles and have been implicated in protein synthesis and degradation as well as in transport to the endoplasmic reticulum, mitochondria, and nucleus (Chappel et al., 1986; Beckman et al., 1990; Palleros et al., 1991; Terlecky et al., 1992; Nelson et al., 1992). Under normal physiologic conditions, HSP72, unlike the cognate HSC73, is not expressed in the mammalian brain. However, HSP72 can be induced after a variety of stresses such as cerebral ischemia, seizures, and hyperthermia (for review, see Massa et al., 1996). Under such stressful conditions HSC73 can also be overexpressed in the brain(Kawagoe et al., 1992; Aoki et al., 1993c) and, it has been suggested, may interact with HSP72 to protect against ischemic neuronal injury (Abe et al., 1991, Hightower, 1991, Abe et al., 1993).
The expression of HSC73 or HSP72 mRNA, or both, and their respective proteins after focal and global ischemia has been extensively studied (Nowak and Jacewick, 1994; Massa et al., 1996; Planas et al., 1997). However, the precise functions of HSC73 and HSP72 in the postischemic brain remains unknown. Pioneering studies on in vivo cerebral HSP72 induction have proposed that HSP72 may function as a putative marker of neuronal injury because its expression was seen in the brain regions that are particularly vulnerable to ischemic injury (Vass et al., 1988). In vitro studies have shown that HSP72 performs a protective role for cells subjected to stress (Amin et al., 1996; Papadopoulos et al., 1996; Sato et al., 1996; Xu and Giffard, 1997). Furthermore, in vivo experiments have shown that ischemic tolerance induced by sublethal ischemic preconditioning involved the induction of HSP72 (Kitagawa et al., 1990; Kirino et al., 1991; Aoki et al., 1993a, b ). Recent work has suggested that gene transfer therapy with defective herpes simplex virus vectors overexpressing hsp72 improved neuronal survival in focal cerebral ischemia (Yenari et al., 1998).
Some investigators have reported that young gerbils are more resistant than adult animals to global cerebral ischemia induced by carotid occlusion (Levine and Sohn, 1969; Payan and Conard, 1977; Matsuyama et al., 1983; Kusumoto et al., 1995). However, the mechanism responsible for such ischemic resistance has not been fully understood. Currently proposed hypotheses attempting to define the mechanism include the following: a more highly developed network of collateral blood vessels between the vertebrobasilar and the carotid circulation in young gerbils (Matsuyama et al., 1983); differences in neuronal functions of neurotransmitters, (Cvejic et al., 1990; Cvejic et al., 1992) or cerebral metabolic rates, or both, between young and adult animals (Nehlig et al., 1988); a lack of extracellular glutamate release after ischemia (Cherici et al., 1991).
The goal of the present study was to investigate HSP72 and HSC73 expression at the transcriptional (mRNA expression) and translational (protein immunoreactivity) levels after transient cerebral ischemia in young and adult gerbils. The collected data were compared with findings regarding neuronal survival to identify the putative protective roles, if any, of HSP72 proteins and HSC73 proteins.
MATERIALS AND METHODS
Animal experiments
Experiments were performed on male adult (3-month-old) and young (3-week-old) Mongolian gerbils obtained from Tumblebrook Farm (West Brookfield, MA). Transient forebrain ischemia was induced by a 15-minute bilateral carotid artery occlusion under anesthesia with 1.5% halothane in 70% N2O-30% O2, in accordance with the protocol approved by the Animal Care and Use Committee, National Institute of Neurological Disorders and Stroke. Restoration of blood circulation was then induced at various times (1, 3, 8, 24, and 48 hours; n = 4 to 8 animals per group). Sham-operated groups of animals were also subjected to anesthesia and to all surgical procedures except clamping the carotid arteries. During the entire experimental period, the body (rectal) and brain (temporal muscle) temperatures were continuously monitored and maintained at 37°C to 37.5°C with a thermostatically regulated heating lamp and pad. Furthermore, the severity of the ischemic insult was assessed by measuring the cerebral blood flow (CBF) using laser-Doppler (LaserFlo, BPM 403A; TSI, St. Paul, MN, U.S.A.) on separate groups of young and adult gerbils. Animals were killed by transcardiac perfusion under deep pentobarbital anesthesia. Isotonic saline solution was perfused through the left ventricle to wash out the blood. The brain was then fixed with 4% (wt/vol) paraformaldehyde solution. Brains were removed, postfixed for 30 minutes in the same fixative, and transferred overnight in 20% sucrose in PBS. Brains were frozen in isopentane and stored at −70°C until coronal sections (20 μm-thick) were cut in a cryostat at −28°C, mounted on charged slides (Probe on Plus, Fisher Scientific, Pittsburgh, PA), and stored at −70°C.
In situ hybridization experiments
The in situ hybridization experiments for detection of either HSP72 or HSC73 mRNA were performed on frozen brain sections as described previously (Sairanen et al., 1997). They were fixed for 5 minutes in 4% paraformaldehyde in PBS, rinsed twice with PBS, and acetylated with 0.25% acetic anhydride in 0.1 mol/L triethanolamine 0.9% NaCl for 10 minutes. The sections were then dehydrated through graded ethanol and chloroform, air-dried, and incubated at 37°C for 24 hours with 106 cpm/slide of the labeled probe/hybridization buffer mix. The hybridization buffer was composed of the following: 80 mmol/L Tris-HCl/4 mmol/L EDTA, 50% formamide, 10% dextran, 0.1 mol/L dithiothreitol, 0.6 mol/L NaCl, 0.1% tetrasodium pyrophosphate, 0.2% sodium dodecyl sulfate, 0.2 mg/mL heparin sulfate. Two different probes were used that consisted of previously characterized synthetic oligonucleotides specific for either the inducible or constitutive mRNA of the rodent hsp70 family (Miller et al., 1991). The two probes were 3′ - end labeled with [35S] thio-dATP using terminal deoxynucleotide transferase (Promega, Madison, WI, U.S.A.). After hybridization, the sections were rinsed in 1× SSC (150 mmol/L sodium chloride/15 mmol/L sodium citrate, pH 7.4) at 40°C, then rinsed 4 times for 15 minutes each in 2× SSC/50% formamide, and finally rinsed twice for 30 minutes in 1× SSC. After dehydration, the hybridized radioactivity was visualized after 3 to 4 days of exposure using Amersham Hyperfilm MAX film (Amersham, Piscataway, NJ, U.S.A.). Sections were subsequently coated with liquid photoemulsion (Kodak NBT2), and developed after exposure for 4 weeks. Sections were counterstained with hematoxylin. To minimize variability among the experiments, brain sections from each condition (various postischemic times in adult and young) were consistently hybridized at the same time to ensure that the sections were exposed to the same amount of radioactivity. The specific activity of the labeled probe was adjusted for each experiment to expose the sections to a similar amount of labeled probe. Finally, the exposure time of the slides to the photoemulsion was always the same. The quantitative analysis was further standardized by choosing a similar zone for overall measurements (i.e., the CA1b region). Regional intensity of in situ hybridization was quantitated on sections using a video camera and microcomputer-based analysis system (MetaMorph, West Chester, PA, U.S.A.). The appropriate threshold window was chosen, and the selected window was converted to a binary image. The total number of threshold silver grains in the outlined area was counted, and the total area of the outlining regions was determined using the spatial calibrations loaded in the MetaMorph application. Data are expressed as the mean ± SEM of silver grains/μm2 of hippocampal area.
Immunochemistry experiments
Double immunofluorescent staining was carried out using a murine anti-72 kDa HSP monoclonal antibody (code RPN 1197, Amersham) specific for the inducible form of HSP72 and a rat anti-HSC73 monoclonal antibody (SPA-815; StressGen, Victoria, BC, Canada) specific for the constitutive form of HSP70. This immunostaining was performed on 20-μm-thick coronal sections of the brain adjacent to those used for in situ hybridization (all brain sections were immunostained simultaneously). They were first rehydrated in PBS for 20 minutes and blocked for 20 minutes with Triton solution (0.2% Triton X100 in PBS containing 4% normal goat serum). Sections were incubated for 24 hours at 4°C with the first primary antibody (anti-HSP72; 1:100 in Triton solution) followed by washes in PBS. They were next incubated for 6 hours at 4°C with the second primary antibody (anti-HSC73; 1:100 in Triton solution), washed, and incubated with 1:200 dilution of secondary fluorescent antibodies (FITC goat anti-mouse IgG and TRITC goat anti-rat IgG antibodies). After washing, sections were coverslipped using fluorescent mounting medium, and were observed under a fluorescent microscope (Zeiss, Oberkochen, Germany). The measurement of fluorescent intensity in different hippocampal areas was performed using the MetaMorph analysis system. Data were expressed as total gray optical density/total count ratio.
Histologic experiments
Histologic examination was carried out on animals subjected to 15-minute ischemia followed by either 48 hours or 7 days of recirculation. After fixation with 4% paraformaldehyde in PBS by transcardiac perfusion, brains were removed, postfixed in the same fixative for 1 week, dehydrated in ethanol, and embedded in paraffin. The 5-μm-thick coronal sections were then deparaffinized, rehydrated, and stained with Cresyl Echt violet.
Neuronal damage was evaluated in CA1 subfield (same as used for Metamorph applications) by counting surviving neurons in the right and left hemispheres of a 0.36-mm length representative region under a light microscope at ×160 magnitude. The average value of both right and left hippocampi was calculated for each animal. Experimental groups consisted of 4 to 7 animals.
Statistical analysis
Statistical significance was analyzed using analysis of variance (ANOVA) followed by post-hoc Newman—Keuls test.
RESULTS
Physiologic parameters
Induction of bilateral carotid artery occlusion led to an almost complete cessation of CBF after 15 minutes of ischemia. The extent of CBF reduction was similar between young and adult gerbils. In all animals recirculation was associated with an initial recovery of CBF followed by a postischemic hypoperfusion after 30 minutes of reflow.
HSC73 mRNA expression in young and adult gerbils
A gradual and intense increase in HSC73 mRNA expression was detected in the CA1 hippocampal subfield of young gerbils after ischemia (Fig. 1A). The relative intensity of mRNA expression in CA1 region was 5- and 7.6-fold higher in ischemic brain than in sham-operated brains after 24 and 48 hours of recirculation, respectively. The relative intensity of HSC73 mRNA expression in the CA1 region of adult gerbils was stable for the first 24 hours of reflow but it increased significantly at 48 hours of reflow (6.8-fold vs sham-operated). A comparison of the HSC73 mRNA expression in CA1 of young and adult gerbils showed that the constitutive expression of this transcript was 2-fold higher in the young animals. The difference between the two groups was enhanced as the reperfusion time increased, indicating the ability of CA1 neurons in young gerbils to induce an earlier and more intense HSC73 mRNA response.
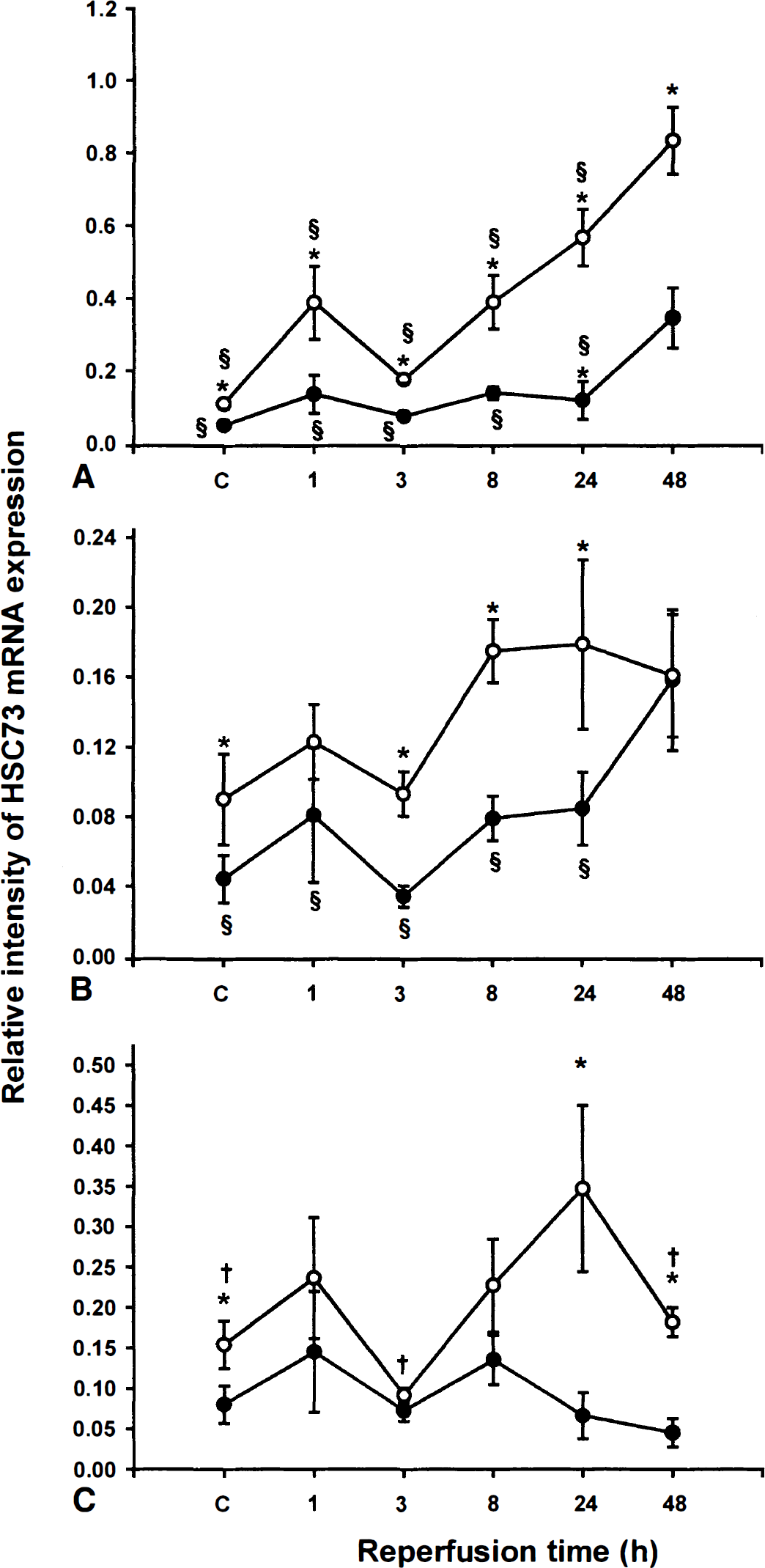
Relative intensity of HSC73 mRNA expression. The quantitative analysis of HSC73 mRNA expression was assessed in CA1
In the CA3 subfield of young gerbils, the relative intensity of HSC73 mRNA expression increased after 8 hours of reflow, but no significant differences were observed between the various reperfusion times (Fig. 1B). The time course of HSC73 mRNA expression in CA3 subfield of adult gerbils was similar to what was observed in CAL The relative stability of early expression was followed by a significant increase after 48 hours of reflow. As in CA1, the constitutive HSC73 mRNA expression was significantly higher in the CA3 region of young gerbils. Young gerbils also exhibited significantly higher HSC73 mRNA expression after 8 and 24 hours of reflow. Interestingly, the intensity of the expression after 48 hours of recirculation was similar between the two groups.
A significant increase in the relative intensity of HSC73 mRNA expression was seen in DG of young animals only after 24 hours of reflow (2.2-fold versus sham-operated). No significant changes were observed throughout all reperfusion periods in DG of adult gerbils (Fig. 1C). In sham-operated animals, as well as in animals subjected to ischemia with reperfusion for 24 and 48 hours, the constitutive expression of HSC73 mRNA was significantly greater in young than adults animals.
HSP72 mRNA expression in young and adult gerbils
The time course of HSP72 mRNA expression in the CA1 subfield of young gerbils was similar to that observed for HSC73 mRNA (Fig. 2A). In addition to a rapid increase as early as 1 hour after reflow (3.7-fold vs sham-operated), a significant induction was found after 24 (6.7-fold) and 48 hours (8.2-fold) in comparison with that found in sham-operated gerbils. Unlike the observations of HSC73 mRNA expression in the adult brain, data showed a significant induction of HSP72 mRNA from 3 hours (5.5-fold) up to 24 hours of reflow (4.5-fold), followed by a decrease in the intensity of HSP72 mRNA expression after 48 hours. Comparing the hybridization signal intensities in the CA1 subfield of young and adult gerbils indicated a significantly greater induction in young gerbils after 1 hour of reflow (3-fold vs. adult). Slightly decreasing expression in adult and increasing expression in young, up to 48 hours of reflow, further indicated significant differences in responsiveness between these two groups.
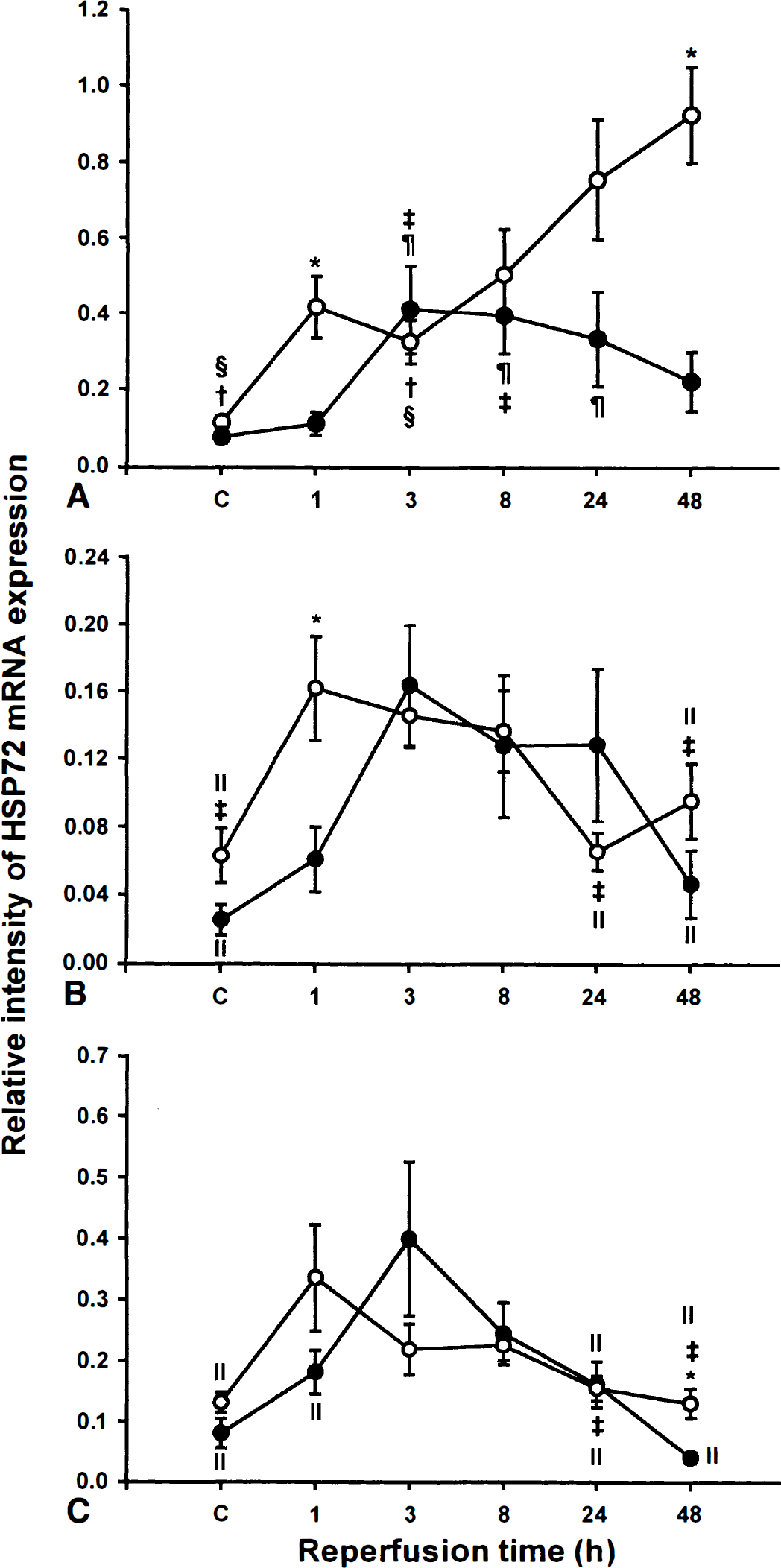
Relative intensity of HSP72 mRNA expression. The quantitative analysis of HSP72 mRNA expression was assessed in CA1
The time course of HSP72 mRNA expression in the CA3 subfield revealed an initial induction followed by a decline in mRNA expression in both young and adult animals (Fig. 2B). However, the time-response curve in young gerbils was shifted left to earlier times of reflow, and the expression peaked at 1 hour (2.6-fold higher vs sham-operated) of reperfusion; in adult animals, the expression peaked at 3 hours of reflow (6.5-fold higher vs sham-operated). The only significant difference between these biphasic responses in young and adult animals was observed after 1 hour of reflow.
Quantitative analysis of HSP72 mRNA expression in DG of young animals revealed an earlier induction of the transcription (2.6-fold vs sham-operated, P < 0.005; peaked at 1 hour of reflow) than that observed in adult (5-fold higher vs sham-operated, P < 0.05; peaked at 3 hours of reflow) (Fig. 2C). The only significant difference obtained between young and adult animals was observed after 48 hours of reflow.
Representative photomicrographs of HSP72 mRNA hybridization after ischemia in hippocampi of young and adult gerbils are shown in Fig. 3. Specific regional increases in levels of HSP72 were observed at earlier time points and persisted for longer periods of time in young gerbils (Fig. 3). More specifically, the intensity of staining in the hippocampus of young animals was greater than in adult counterparts. The intensity of staining in CA1 regions from all experimental conditions was relatively greater than was observed in either CA3 or DG regions.
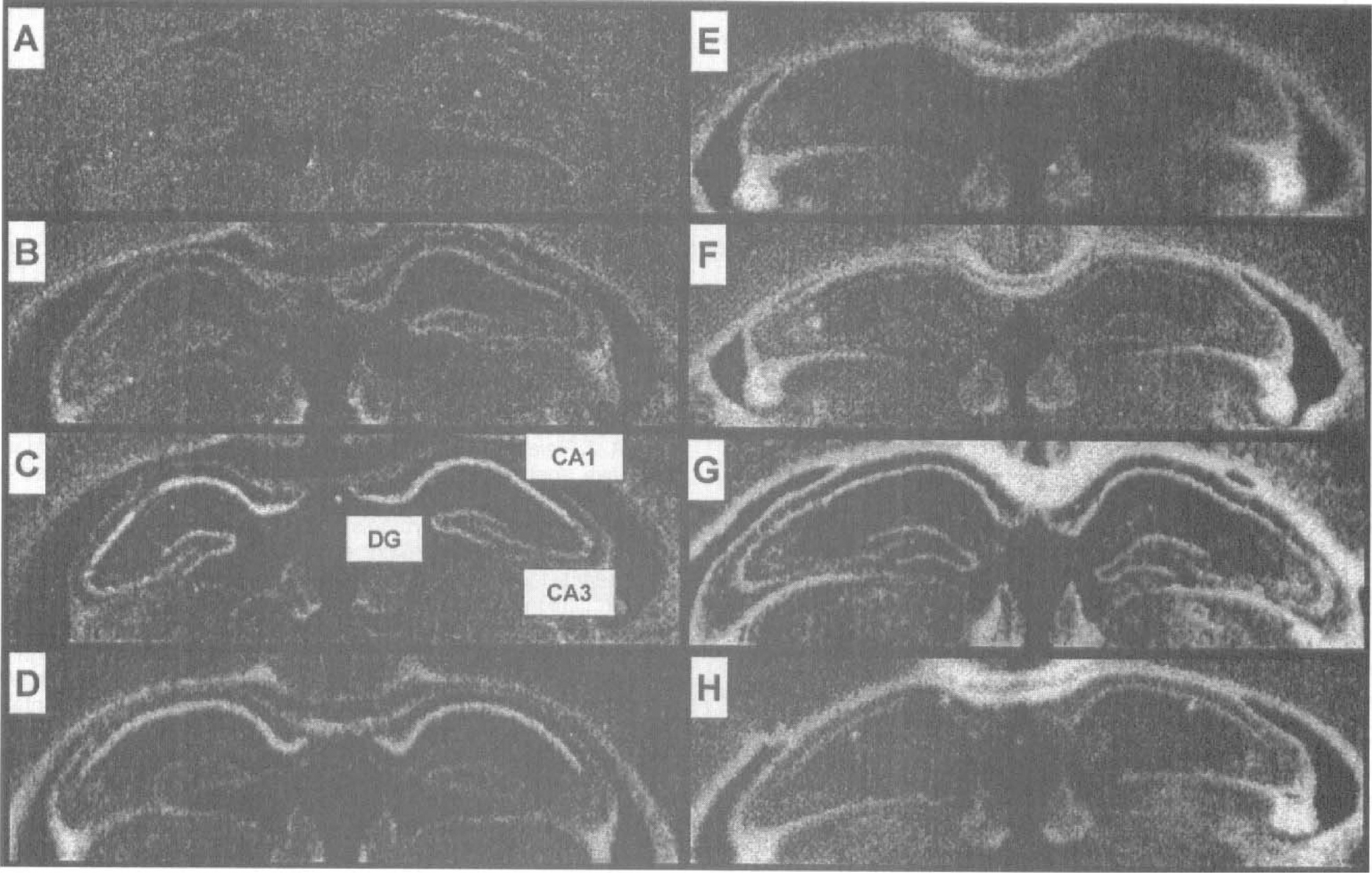
Representative photomicrographs of HSP72 mRNA expression in hippocampus of young and adult gerbils. Images are autoradiograms obtained after in situ hybridization using a 35S-labeled oligonucleotide probe specific for HSP72. They were obtained from coronal brain sections of young (left) and adult (right) gerbils that were sham-operated
HSC73 immunoreactivity
The quantitative analysis of HSC73 immunoreactivity expressed as total gray optical density/total count ratio is shown in CA1, CA3, and DG hippocampal subfields of young and adult gerbils (Fig. 4). Constitutive expression of HSC73 protein was approximately twofold higher in CA1 and DG subfields of young sham-operated gerbils than in adult animals. After 15 minutes of ischemia and 48 hours of reflow, a slight, but significant, induction of HSC73 immunoreactivity was observed in CA1 of adult gerbils, whereas in CA1 of young gerbils, significant decreases were observed at 24 hours and 48 hours of reflow (Fig. 4A). The level of HSC73 protein in CA3 and DG subfields tended to increase after 48 hours of reflow of adult gerbils; however, these changes were not significant. Transient global ischemia did not induce significant variations in HSC73 immunoreactivity in DG and CA3 hippocampal neurons of young gerbils.
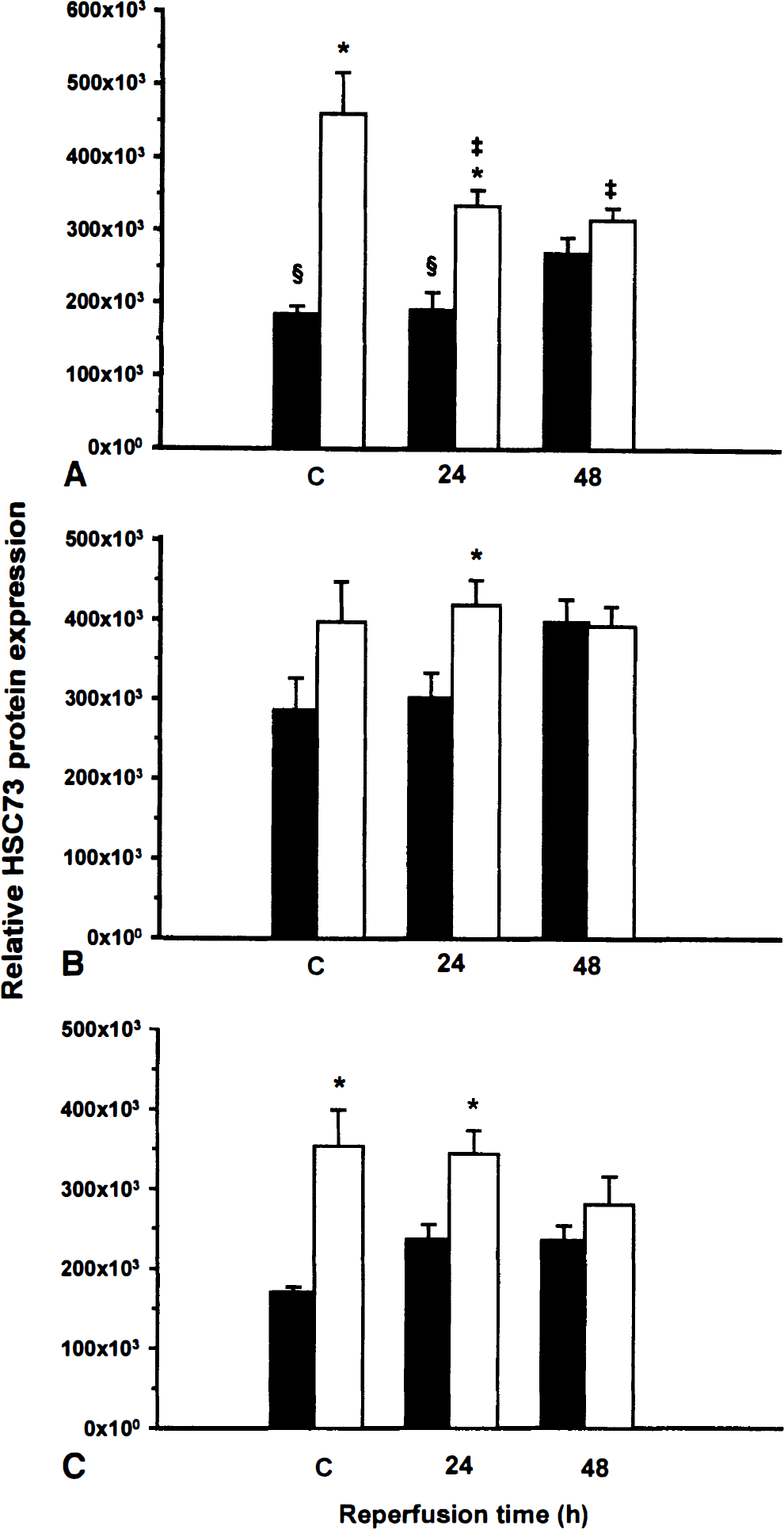
Relative HSC73 protein expression. The quantitative analysis of HSC73 protein expression was assessed in CA1
HSP72 protein induction
Figure 5 shows quantitative analysis of HSP72 immunoreactivity expressed as total gray optical density/total count ratio in CA1, CA3, and DG hippocampal subfields of young and adult gerbils. Ischemia led to a significant induction of HSP72 expression in CA1 and CA3 subfields of young gerbils. This response was characterized by the presence of many fluorescent pyramidal neurons in those two areas (Figs. 6A and 6B, respectively). Quantitative analysis of the response in young gerbils indicated that the induction was more marked in CA1 (+150%; P < 0.05) than in CA3 (+90%; P < 0.05) subfield after 48 hours of reflow. Compared with sham-operated animals, adult gerbils exhibited only a 66% increase in HSP72 expression in CA1 subfield after 48 hours of reflow, whereas a 180% increase in the fluorescent signal was observed in the CA3 subfield.
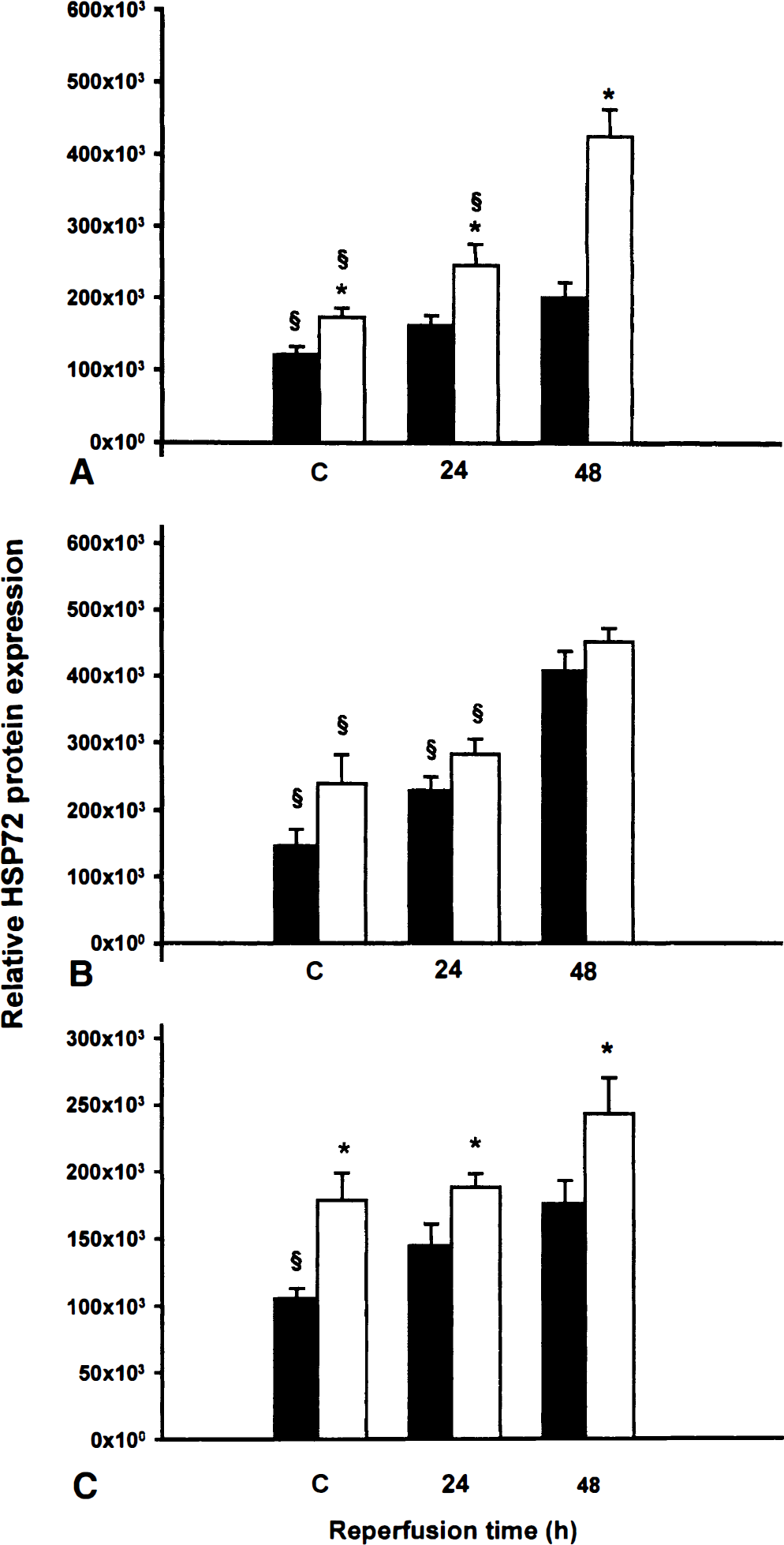
Relative HSP72 protein induction. The quantitative analysis of HSP72 protein induction was assessed in CA1
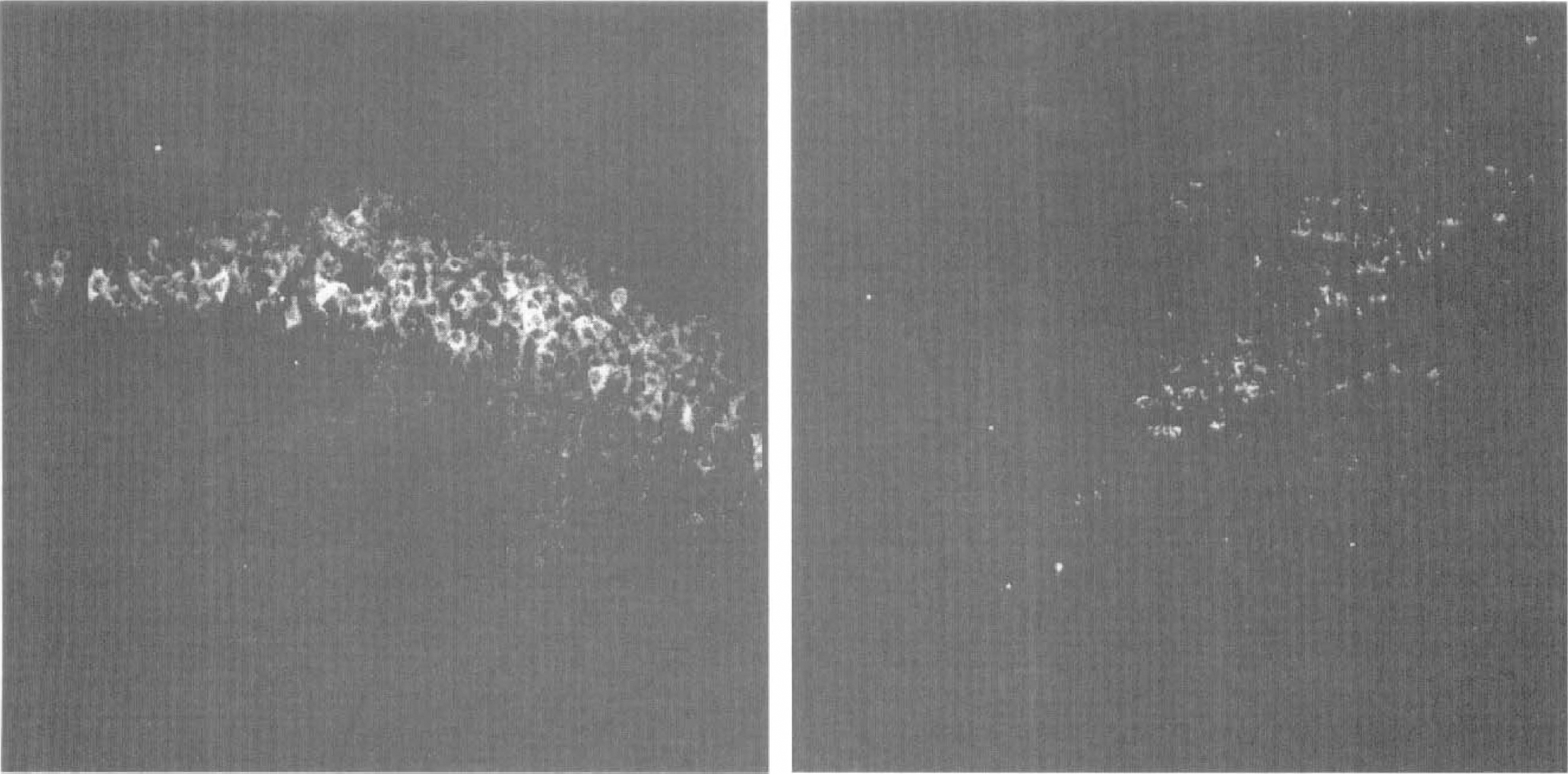
Representative photomicrographs of HSP72 immunostaining in CA1 subfield of young and adult gerbils. HSP72 immunostaining was examined in CA1 subfield of young (left) and adult (right) gerbils subjected to 15-minute ischemia followed by 48 hours of reperfusion. Note the absence of HSP72 positive neurons in adults and the intense immunostaining in young CA1 neurons.
The findings of this study show a weak postischemic HSP72 immunostaining in DG of both young and adult gerbils. In young gerbils, there were no significant differences between experimental and sham-operated animals; however, HSP72 immunostaining significantly increased in adult gerbils after 48 hours of reflow. Nevertheless, in sham-operated and all experimental conditions, the level of HSP72 immunostaining was significantly higher in young than in adult animals.
Neuronal damage
No significant differences were observed between the neuronal densities of sham-operated young and adult gerbils (i.e., 294 ± 33 and 277 ± 26 neurons/mm, respectively) (Table 1). The average neuronal densities after 15 minutes of ischemia, followed by 48 hours or 7 days of recirculation in the CA1 subfield of young animals, were not significantly different from those of sham-operated controls. However, adult gerbils subjected to the same period of ischemia/reperfusion had extensive neuronal damage in the CA1 subfield. Neuronal density was significantly lower than in sham-operated and respective young animals.
Effect of ischemia/reperfusion on hippocampal CA1 neuronal survival in young and adult animals

Animals were subjected to ischemia/reperfusion as described in Materials and Methods section. All values represent number of neurons per 1-mm length of CA1 pyramidal cell layer and are expressed as mean ± SEM; n = number of animals/group.
P <0.05 versus sham-operated group;
P <0.05 versus respective young gerbil group.
DISCUSSION
This study presents a quantitative and comparative analysis of the time course of expression and regional distribution of constitutive HSC73 and inducible HSP72 mRNA expression and immunoreactivity in the hippocampi of young and adult gerbil hippocampi after transient global ischemia.
The data show that in adult gerbils, a 15-minute ischemia-induced HSP72 mRNA expression in all hippocampal subfields was associated with a weak expression of HSP72 immunoreactivity in hippocampal CA1 neurons but a marked induction in CA3 and DG neurons. The ischemia-induced cell death of CA1 neurons—apparent 48 hours after the insult—agrees with previous studies (for review, see Massa et al., 1996). For example, in earlier studies, 5-minute ischemia produced a marked induced HSP72 mRNA expression but only minimal HSP72 induction in CA1 neurons, whereas intensive HSP72 protein synthesis was observed in CA3 neurons and DG cells, demonstrating the induction of HSP72 immunoreactivity in cell populations that are expected to survive (Vass et al., 1988; Nowak, 1991). In contrast, short durations of ischemia (2 to 3 minutes) that do not damage CA1 neurons induce HSP72 protein and mRNA (Abe et al., 1991; Kato et al., 1993).
Most important, this study showed that a 15-minute transient cerebral ischemia resulted in extensive neuronal death of CA1 pyramidal neurons of adult gerbils, whereas these cells were remarkably preserved in young gerbils subjected to identical insult. These observations indicate that younger animals exhibit a greater tolerance for cerebral ischemia agreeing with previous reports that indicated that immature gerbils were more resistant to global ischemia than adult gerbils (Levine and Sohn, 1969; Payan and Conard, 1977; Matsuyama et al., 1983, Kusumoto et al., 1995). For example, Kusumoto et al. (1995) showed that 15 minutes of ischemia caused significant neuronal death in the CA1 layer of 3-week-old, but not 2-week-old, gerbils. In addition, no neuronal abnormalities in the CA1 subfield were detected after a 20-minute ischemic episode induced by bilateral carotid occlusion of young gerbils (Soriano et al., 1994).
The mechanism underlying apparent tolerance of young animals to ischemia remains unclear. It has been suggested that a better-developed network of collateral blood vessels between the vertebrobasilar and carotid circulations in young animals may be the basis for their resistance to cerebral ischemia (Matsuyama et al., 1983). However, recent work showing that bilateral carotid occlusion produces the same magnitude of forebrain ischemia in 2-week-old animals as in adults contradicts that hypothesis (Kusumoto et al., 1995). Other reports suggest that immature brains could possess a greater tissue resistance to ischemia or hypoxia because of a lower metabolic rate (Nehlig et al., 1988). Another possibility involves decreased levels of ischemia-induced release of neurotransmitters in young brains (Cherici et al., 1991; Cvejic et al., 1990).
Neuroprotection by HSP72 or HSC73 proteins, or both, can be viewed as possible factors contributing to ischemic resistance of young animals. In vitro experiments have already shown the protective role of HSP72 in cell survival under various stressful conditions (Amin et al, 1996; Papadopoulos et al., 1996; Sato et al., 1996; Xu and Giffard, 1997). Furthermore, gene therapy with hsp72 in rat models of stroke and epilepsy are neuroprotective (Yenari et al., 1998). The current data showed that 15-minute ischemia led to a more pronounced induction of HSP72 mRNA associated with the subsequent induction of the encoded proteins in the CA1 subfield of young gerbils than in adult gerbils. Thus, ischemic resistance of young CA1 neurons might be attributed to the ability of young CA1 neurons to synthesize HSP72 proteins.
Most information available on histopathology and HSP72 expression in models of brain hypoxia-ischemia has been obtained using rat neonates (Dwyer et al., 1989; Ferriero et al., 1990; Blumenfeld et al., 1992; Munell et al., 1994; Kobayashi and Welsh, 1995; Gilby et al., 1997; Towfighi et al., 1997). The data presented here are more readily compared with findings concerning HSP72 mRNA and protein expression in young (7 to 30 days) and adult gerbils after 20-minute ischemia as reported by Soriano et al. (1994). In their article, the HSP72 mRNA expression was studied only after 3 hours of reflow and the HSP72 protein synthesis after 24 hours of reflow. As the current data confirm, a strong induction of HSP72 mRNA in DG and a moderate induction in CA1 was observed in 3-week-old gerbils after 3 hours of reflow. In addition, age-dependent differences in induction of HSP72 mRNA were observed, although in the current data, they were detected at earlier and longer times of reflow (1, 24, and 48 hours) in all hippocampal subfields. Furthermore, Soriano et al. (1994) observed intensive immunostaining of HSP72 in DG and only a few faintly positive cells in CA1 after 24 hours of reflow. At this time of reflow, we were unable to detect significant levels of induction of immunoreactive neurons in either young or adult gerbils. However, after 48 hours of reflow, significantly greater immunoreactivity for HSP72 was observed in all hippocampal subfields of both young and adult gerbils. In addition, significantly higher immunoreactivity was detected in young gerbils. These observations indicate a possible correlation between induction of HSP72 and neuronal injury, which contradicts the finding reported by Soriano et al. (1994) and may be resulting from the incorporation of additional time periods in the present study.
In summary, because the HSP72 mRNA expression slightly decreased in adult gerbils and increased in young gerbils after 48 hours, the resulting significant difference indicates that the so-called vulnerable CA1 neurons of young gerbils still had transcriptional activity, whereas those of adult gerbils had already lost such activity.
The possible relation between the survival of neurons and their ability to synthesize HSP72 remains a perplexing issue. It can be argued that most neurons surviving ischemic insult synthesize HSP72 proteins, whereas lethally injured neurons do not synthesize HSP72 proteins even though they may synthesize HSP72 mRNA. The possibility that this is an epi-phenomenon remains to be clarified.
For instance, the findings of Gaspary et al. (1995) are consistent with the hypothesis that HSP72 immunoreactive-neurons must be alive. The authors showed that the number of HSP72 immunostained neurons in the cortical layer is more than 10-fold greater than the number of acid fuchsin stained (dead or dying) neurons. However, Gilby et al. (1997) reported that both dying and surviving neurons in young rats subjected to hypoxia—ischemia are HSP72 immunoreactive. In addition, major differences between rat and gerbil models of global ischemia have been reported. Although both gerbil and rat CA1 pyramidal neurons synthesize HSP72 mRNA after ischemic insults sufficient enough to produce delayed neuronal death, HSP72 protein synthesis is only induced in CA1 neurons of the rat (Kawagoe et al., 1993; Massa et al., 1996).
The current data, which show a shift in HSP72 mRNA induction in the hippocampus to earlier period of reflow in young gerbils, may indicate its important role in the ischemic resistance observed in young animals. Supporting this possibility, Aoki et al. (1993a, b ) reported an acceleration of the induction of HSP72 mRNA in gerbil hippocampal neurons made tolerant to ischemia by preconditioning with short (2-minute) ischemia.
Mechanisms other than HSP72 mRNA and protein may be involved in the induction of ischemic tolerance. In contrast to HSP72 mRNA, HSC73 mRNA was abundantly present in hippocampal neurons of sham-operated animals. A higher constitutive expression of HSC73 mRNA and HSC73 immunoreactivity was observed in the hippocampi of young gerbils rather than adult gerbils. HSC73 expression in hippocampal neurons was also up-regulated by a brief duration of ischemia that induced ischemic tolerance (Aoki et al., 1993b). Although some studies show that constitutive levels of HSC73 mRNA are increased after ischemia (Kawagoe et al., 1992; Abe et al. 1993), we observed only a slight increase in its expression in adult animals after 48 hours of recirculation (in CA1 and CA3 regions). However, a strong induction of HSC73 mRNA was observed in young gerbils after ischemia, and it was associated with a significant decrease in HSC73 protein synthesis only in the CA1 subfield; no significant changes in HSC73 protein were observed in CA3 and DG subfields. Harrub and Nowak (1998) also reported that HSC70 immunoreactivity was decreased in CA1 neurons in gerbil hippocampus subjected to ischemia/reperfusion. Decreased HSC70 expression may be caused by its involvement in protein-protein interactions, resulting in a masking of the epitope recognized by the monoclonal antibody used in the current study. It has also been suggested that HSC70 involvement in claritin metabolism may be altered during ischemia-induced dysfunction of membrane-associated vesicle transport system in CA1 cells (Kawagoe et al., 1992). The precise mechanism responsible for this observation remains to be clarified.
It is known that transient cerebral ischemia is associated with a prolonged severe inhibition of protein synthesis in CA1 pyramidal neurons (Cooper et al., 1977; Thilmann et al., 1986; Nowak, 1990; Widmann, et al., 1991). Impairment of protein synthesis during the initial hours of reperfusion may be a critical factor related to enhanced vulnerability of CA1 neurons. Therefore, CA1 neurons in young gerbils may have a greater capacity to preserve or recover protein synthesis, allowing earlier HSP72 protein expression, which is essential for the restoration of normal ribosome assembly and promotion of the synthesis of new ribosomes (Lindquist, 1986). The higher level of the constitutive protein HSC73 expression may also participate in the recovery of HSP72 translation, thereby accelerating recovery of protein synthesis and preventing loss of CA1 neurons.
In contrast to the delayed detection of HSP72 immunoreactivity observed in the CA3 and DG of both young and adult animals reported here, a considerably shorter (6-hour) recovery time of protein synthesis was observed in these hippocampal regions of gerbils after 5-minute ischemia (Widmann et al., 1991). Therefore, it is possible that the reported results might have been caused by three factors: (1) an earlier synthesis of HSP72 protein, (2) a failure of selective monoclonal antibody to adequately detect the mature (posttranslationally modified) form of HSP72 in CA3 and DG, and (3) the sequestration (and inaccessibility) of HSP72 protein in the nucleolus after its induction (Harrub and Nowak, 1998).
In conclusion, these findings suggest that the induction of both HSP72 mRNA and proteins in the CA1 pyramidal neurons of young gerbils, as well as the higher constitutive expression of HSC73, may partially contribute to the higher neuronal resistance of young animals to transient cerebral ischemia.
