Abstract
GPIIb/IIIa antagonists are expected to have a beneficial effect on acute cerebral infarction, however, the occurrence of intracranial hemorrhage has not been as widely investigated. A rabbit focal thrombotic occlusion model of the middle cerebral artery was established by creating a photochemical reaction between green light and Rose Bengal. Hemorrhagic transformation was common in the area of cerebral infarction. Using this model, the effect of a GPIIb/IIIa antagonist, ME3277 (low dose, (L); 0.15 mg/kg + 0.125 mg/kg·h, middle dose, (M); 0.3 mg/kg + 0.25 mg/kg·h and high dose, (H); 0.6 mg/kg + 0.5 mg/kg·h), aspirin (20 mg/kg) and sodium ozagrel (thromboxane A2 synthase inhibitor, 1 mg/kg + 2 mg/kg·h) were evaluated. Drugs were intravenously administrated 30 minutes after the photochemical reaction for 24 hours. Aspirin inhibited the ex vivo platelet aggregation induced by arachidonic acid and collagen but not by adenosine diphosphate (ADP), while sodium ozagrel only inhibited the arachidonic acid-induced aggregation. ME3277 dose-dependently inhibited the platelet aggregation induced by all the inducers (approximately 60% in L, 80% in M, and 90% in H). At 24 hours of middle cerebral artery (MCA) occlusion, infarct volume was significantly reduced by aspirin and each dose of ME3277. These agents improved neurologic deficits, with ME3277 being more potent than aspirin. Sodium ozagrel did not alter the infarct volume nor neurologic deficits. No drug was found to worsen hemorrhage volume despite increasing bleeding time (2–3 fold) in the skin. In this model, the occluded artery was spontaneously recanalized and re-thrombosed frequently. One mechanism by which antiplatelet agents reduced infarct volume was inhibition of rethrombosis of the MCA. These results suggest that treatment with a GPIIb/IIIa antagonist is a useful intervention for acute cerebral infarction prolonging dose bleeding time to 3 times the basal value.
Keywords
Platelet activation occurs in the acute cerebral infarction (Koudstaal et al., 1993; Kooten et al., 1997; Grau et al., 1998) and a recent clinical trial of antiplatelet therapy revealed advantages of aspirin (CAST Collaborative Group, 1997; International Stroke Trial Collaborative Group, 1997). Secondary prevention studies predicted that more potent antiplatelet agents, such as ticlopidine and clopidogrel, have better efficacy than aspirin (Antiplatelet Trialists' Collaboration, 1994; CAPRIE Steering Committee, 1996); although the fact that they could not be administered intravenously has restricted their efficacy against acute ischemic stroke. In addition to having limited antiplatelet activity, aspirin reduces prostaglandin I2 production in endothelium. This unfavorable action was not shown by selective thromboxane A2 synthase inhibitors such as sodium ozagrel, whereas the protective effect on cerebral infarction has not been confirmed. Attempts to inhibit platelet aggregation more strongly resulted in the development of agents that block fibrinogen binding to its receptor, GPIIb/IIIa, on activated platelets. The binding of fibrinogen to GPIIb/IIIa is considered the final common pathway of platelet aggregation (Phillips et al., 1991; Pytela et al., 1986). Therefore, the blockade of GPIIb/IIIa is expected to be the sole effective antiplatelet agent compared to classical antiplatelet agents. The above facts lead to the question of whether a GPIIb/IIIa antagonist would have a beneficial effect on the acute cerebral infarction.
In the course of antiplatelet therapy, care should be taken to minimize bleeding, particularly to avoid an intracranial hemorrhage (mainly hemorrhagic infarction), which is the most lethal side effect of drug therapy for cerebral infarction. Clinical and preclinical studies have indicated that intracranial hemorrhage was induced by treatment with thrombolytic agents as well as anticoagulants (International Stroke Trial Collaborative Group, 1997; The NINDS t-PA Stroke Study Group, 1997; TOAST Investigators, 1998), while the risks associated with using antiplatelet agents remain controversial.
Against this background, the authors established a rabbit model of cerebral infarction by thrombotic occlusion of the middle cerebral artery (MCA). The rabbit was chosen because it is often used for models of intracranial hemorrhage (Lyden et al., 1987; Slivka & Pulsinelli, 1987; Yenari et al., 1997). The pathogenic process of arterial occlusion in humans is different from that in experimental animals. In most patients, the cerebral artery is spontaneously recanalized in the early phase of cerebral infarction (Alexandrov et al., 1994). This event is frequently followed by rethrombosis, which was successfully prevented by a GPIIb/IIIa antagonist (Wallace et al., 1997). However, the other models of focal ischemia, such as nylon thread insertion and mechanical compression of the MCA, do not satisfy these conditions (Tamura et al., 1981; Longa et al., 1989). In contrast, thrombotic occlusion of the MCA mimics recanalization followed by rethrombosis in guinea pigs and primates (Kaku et al., 1998; Kawano et al., 1998). The effect of a nonpeptide GPIIb/IIIa antagonist, ME3277 (sodium hydrogen [4-[(4,5,6,7-tetrahydrothieno [3, 2-c] pyridin-2-yl) carbonylamino] acetyl-o-phenylene] dioxydiacetate) in a guinea pig model of cerebral infarction has been reported previously; however, in that study, the drugs were injected before thrombotic occlusion (Kawano et al., 1999). In the current study, the authors investigated the effects of ME3277, sodium ozagrel, and aspirin on cerebral damage and intracranial hemorrhage.
MATERIALS AND METHODS
ME3277 (sodium hydrogen [4-[4,5,6,7-tetrahydrothieno [3, 2-c] pyridin-2-yl) carbonylamino] acetyl-o-phenylene] dioxydiacetate) was obtained from Meiji Seika Kaisha Co., Tokyo, Japan. Sodium ozagrel was purchased from Ono Pharmaceutical Co., Osaka, Japan. Aspirin and sodium arachidonic acid were purchased from Sigma Co., WA, U.S.A. Adenosine diphosphate and collagen were donated by MC Medical Co., Tokyo, Japan. Rose Bengal was obtained from Wako Chemical, Co., Osaka, Japan.
The experimental protocol was approved by the local committee on ethics of animal experimentation, and extra care was taken to avoid animal suffering. Male Japanese white rabbits (12 animals in each group) weighing 2.0–3.0 kg were anesthetized with 1–2% isoflurane in a 70% N2O and 30% O2 mixture using a face mask. Arterial blood pressure and heart rate were monitored continuously with a catheter inserted into the femoral artery. Arterial blood gases were analyzed with a blood gas analyzer (model 860; Ciba-corning, MA, U.S.A.). A catheter was inserted into the femoral vein and the tip was subcutaneously advanced to the dorsal side. The catheter tip was connected to an infusion system (Lomir Biomedical, Quebec, Canada), which allows stable drug infusion under free moving. Animal body temperature was maintained at 38°C with a heating pad (K-module K-20; American Pharmaseal, CA, U.S.A.).
Photothrombotic occlusion of the middle cerebral artery
After a left temporal incision, the eyeball was removed. An 8-mm-diameter oval window was opened in the orbital bone using a dental drill (model PAL-7; Morita, Tokyo). The MCA stem was observed under an operation microscope without cutting the dura matter. The authors regularly sketched the vessel's anatomic construction, including the MCA, through the cranial window. Photoirradiation with green light (wavelength, 520–620 nm) was conducted using a xenon lamp (model L-4887, Hamamatsu Photonics, Hamamatsu, Japan) with a heat absorbing filter and a green filter. The irradiation was directed by a 3-mm-diameter optic fiber mounted on a micromanipulator. The head of the optic fiber was placed on the MCA segment proximal to the olfactory tract. The blood flow velocity in the MCA was measured with a pulse Doppler flow probe (PVD-20, Crystal Biotech) which mounted to another manipulator. The Doppler flow probe was placed on the dura matter just above the MCA positioned on the distal to the irradiated segment, and its angle was adjusted to obtain maximal blood flow. When a stable baseline blood flow was established, Rose Bengal (20 mg/kg body weight for 3 minutes) was administered. The green light was irradiated for 20 minutes with an irradiation dose of 0.636 W/cm2. The photochemical reaction between Rose Bengal and transluminal light irradiation leads to endothelial injury followed by platelet adhesion, aggregation, and a formation of occlusive platelet rich thrombus at the irradiated site (Watson et al., 1985; Matsuno et al., 1991; Saniabadi et al., 1995). All drugs were injected through the catheter 30 minutes after the photochemical reaction for 24 hours. ME3277 was administered at three different doses (low dose, 0.15 mg/kg bolus followed by 0.125 mg/kg·h; middle dose, 0.3 mg/kg bolus followed by 0.25 mg/kg·h; and high dose, 0.6 mg/kg bolus followed by 0.5 mg/kg·h). A clinical report on a GPIIb/IIIa antagonist for ischemic heart disease described that 80% inhibition of adenosine diphosphate-induced platelet aggregation was effective and did not result in major bleeding (Tcheng, 1997). Thus, the authors set up three doses of ME3277 based on an inhibition of ADP-induced platelet aggregation of 60, 80 and 90%. Aspirin was bolusly administered at 20 mg/kg followed by an infusion of vehicle. Sodium ozagrel was bolusly administered at 1 mg/kg, then at 2 mg/kg/h, the dose inhibiting by more than 90% the thromboxane A2 synthetase activity (Zheng et al., 1995).
Ex vivo platelet aggregation in platelet rich plasma
At the point immediately before, 2 hours after, and 24 hours after the photochemical reaction, blood samples were collected from the catheter using a syringe containing 1:9 sodium citrate (final concentration, 0.38%). Platelet rich plasma was obtained by centrifugation of the blood samples at 150 × g for 10 minutes. Platelet poor plasma was obtained by recentrifugation of the supernatant at 2,000 × g for 10 minutes. Platelet counts were made with a cell counter (MEK-6158, Nihon Kohden, Japan) and adjusted to 300,000/μL with platelet poor plasma. Platelet aggregation was measured according to the method of Born (Born, 1962). The maximal aggregation response was measured and expressed as an inhibitory ratio of the basal value. Inducers of platelet aggregation and the final concentrations in glass cuvettes were as follows: ADP, 100 μmol/L; collagen, 2 μg/mL; arachidonic acid, 0.25 mmol/L. The light transmission for the various inducers was recorded for 7 minutes after a 1 minute preincubation period.
Measurement of bleeding time
Bleeding time was measured at just before, 2 hours after, and 24 hours after the photochemical reaction as described previously (Ivy et al., 1941). Bleeding time was assessed with a disposable standard device (Simplate R, Organon Teknika, Durham, NC, U.S.A.), has a single retractable blade and makes a 5-mm-long and 1-mm-deep skin wound. A manometer cuff was inflated to 40 mm Hg and an incision was made on the left thigh.
Measuring of blood flow of the middle cerebral artery
The blood flow in the MCA was monitored for 2 hours after the photochemical reaction. At the end of the observation period, the surgical area was sterilized with kanamycin (Meiji Seika; Tokyo). Animals were allowed to recover from anesthesia after the surgical area had been closed. Twenty-four hours later, the Doppler flow probe was repositioned on the same segment of the MCA referring to the sketch of vessel anatomical construction.
The following parameters were measured for the evaluation of MCA stem blood flow. (1) occlusion time, the time taken from injection of Rose Bengal for thrombotic occlusion to occur in the MCA; (2) total reflow time, a percentage of the amount of time when the vessel was open from the start of drug infusion until 2 hours after the photochemical reaction; (3) 24 hours patency, the status of MCA blood flow 24 hours after the photochemical reaction and expressed as occlusion, perfusion, or cyclic flow reductions. Cyclic flow reductions (CFRs) were repetitive cycles of reduction and restoration of MCA blood flow.
Neurologic function
Neurologic function of each animal was evaluated 24 hours after the photochemical reaction. In the wryneck test, the animal was placed in a cage and the torsion of the neck was observed. Behavior was scored as follows: 0, normal; 1, twist of the neck. In the righting reflex test, the animal was placed on its back and scored as follows: 0, righted within 1 second; 1, righted within 5 seconds; 2, did not right within 5 seconds. In the dysfunction of paws, the fore paw, or hind paw, was pulled toward the body. The time to re-extend the paw was scored as follows: 0, achieved within 1 second; 1, achieved within 5 seconds; 2, not achieved within 5 seconds. In the postural reflex test, the animal was pushed in the contralateral direction and scored as follows: 0, normal; 1, reduced resistance to lateral push; 2, fell down on the contralateral side. Each score was summed and represented as a neurologic score (0 to 9). The highest score (10) was given when the animal died within 24 hours.
Determination of infarct volume and hemorrhage volume
After the evaluation of other variables at 24 hours, animals were anesthesized with pentobarbital (30 mg/kg, IV). The brain was carefully isolated and cut into eight coronal slices of 1.5-mm thickness using a microslicer (DTK-3000W, D. S. K., Kyoto, Japan). A photograph of the brain slices was taken to measure hemorrhage volume. Then, the brain slices to be used for measuring infarct volume were stained with 1 % triphenyltetrazolium chloride at 37°C and fixed for 24 hours in a formalinphosphate buffer. In each slice, the areas of infarction were measured by computerized image analysis using the public domain NIH image program (developed at the U.S. National Institutes of Health and available on the Internet by anonymous FTP from zippy.nimh.nih.gov). Infarct volume was calculated by integrating the infarct area in each of the eight sections. The proportion to whole brain volume was expressed as a percentage of infarct volume. The proportions of the hemorrhage volume to infarct volume and to the whole brain volume were expressed as a percentage of hemorrhage volume.
Statistical analysis
All data are expressed as mean ± SD. Student's unpaired t-test, or Wilcoxon's test, was used for comparisons between two groups. Dunnett's or Steel's post hoc test was used for comparisons among more than three groups. One-way analysis of variance was used to compare baseline physiologic variables. Fisher's exact test was used for the analysis of mortality rate. The correlation coefficient was obtained with Spearman's or Pearson's test. A P value < 0.05 was considered to be a significant difference.
RESULTS
The physiologic variables did not significantly differ among the six groups at the basal level (Table 1), nor over the 2 hour observation period (data not shown). In a preliminary study of rabbits, sham operation (Rose Bengal injection without green light irradiation) did not result in MCA occlusion (data not shown), whereas Rose Bengal injection with green light irradiation did produce thrombotic occlusion of the MCA followed by cerebral infarction and neurologic deficits. In the current study, four animals given vehicle and one animal each that received a high dose of ME3277 and sodium ozagrel died within 24 hours of the MCA occlusion. The mortality rate was not significantly different among the groups. All of the animals recovered from anesthesia at 2 hours after the operation, including the animals that died later. Therefore, these animals are thought to have died 4 to 24 hours after MCA occlusion. Among the dead animals, brain slices of two of the vehicle-treated rabbits could not be stained with triphenyltetrazolium chloride.
Physiologic variables before middle cerebral artery occlusion
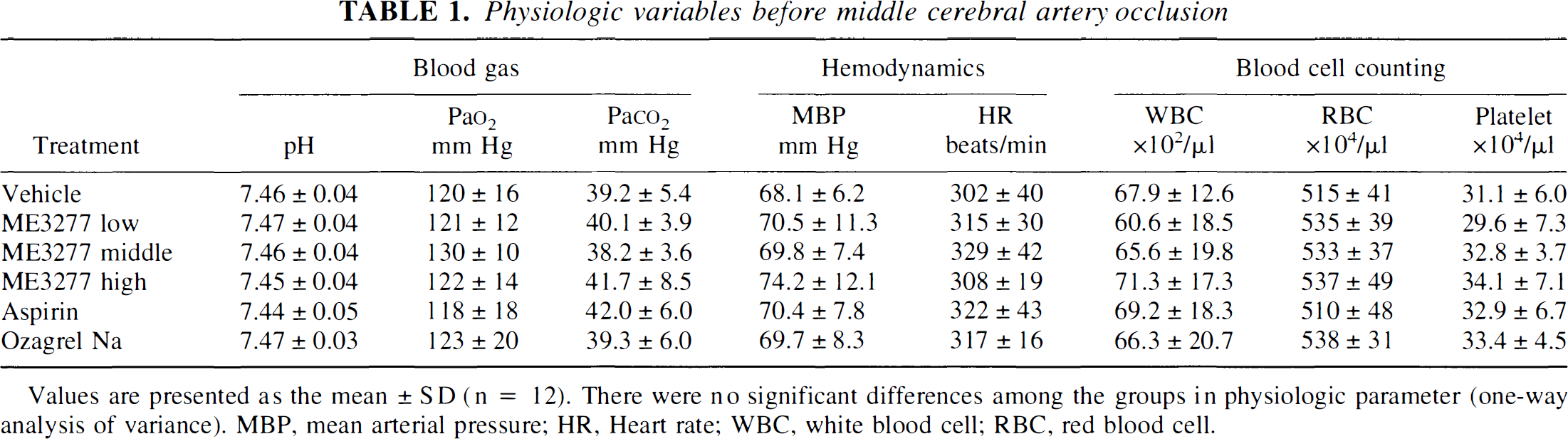
Values are presented as the mean ± SD (n = 12). There were no significant differences among the groups in physiologic parameter (one-way analysis of variance). MBP, mean arterial pressure; HR, Heart rate; WBC, white blood cell; RBC, red blood cell.
Effects on ex vivo platelet aggregation
The inhibition of ex vivo platelet aggregation by antiplatelet agents at 2 hours and 24 hours is shown in Table 2. Aspirin selectively inhibited the platelet aggregation with arachidonic acid (P < 0.01) and partially inhibited the collagen-induced aggregation by 46% (P < 0.05) at 2 hours, and by 65% (P < 0.01) at 24 hours. Sodium ozagrel tended to inhibit the collagen-induced platelet aggregation and significantly inhibited arachidonic acid-induced aggregation (P < 0.05). ADP-induced platelet aggregation was resistant to aspirin and to sodium ozagrel. In contrast to aspirin or sodium ozagrel, ME3277 inhibited ex vivo platelet aggregation significantly(P < 0.01), independent of the inducers. Inhibitory ratios at low, middle, and high doses were approximately 60, 80, and 90%, respectively. The inhibition of platelet aggregation was maintained through the 24-hour period in each group.
Effect of antiplatelet agents on inhibition of ex vivo platelet aggregation and prolongation of bleeding time
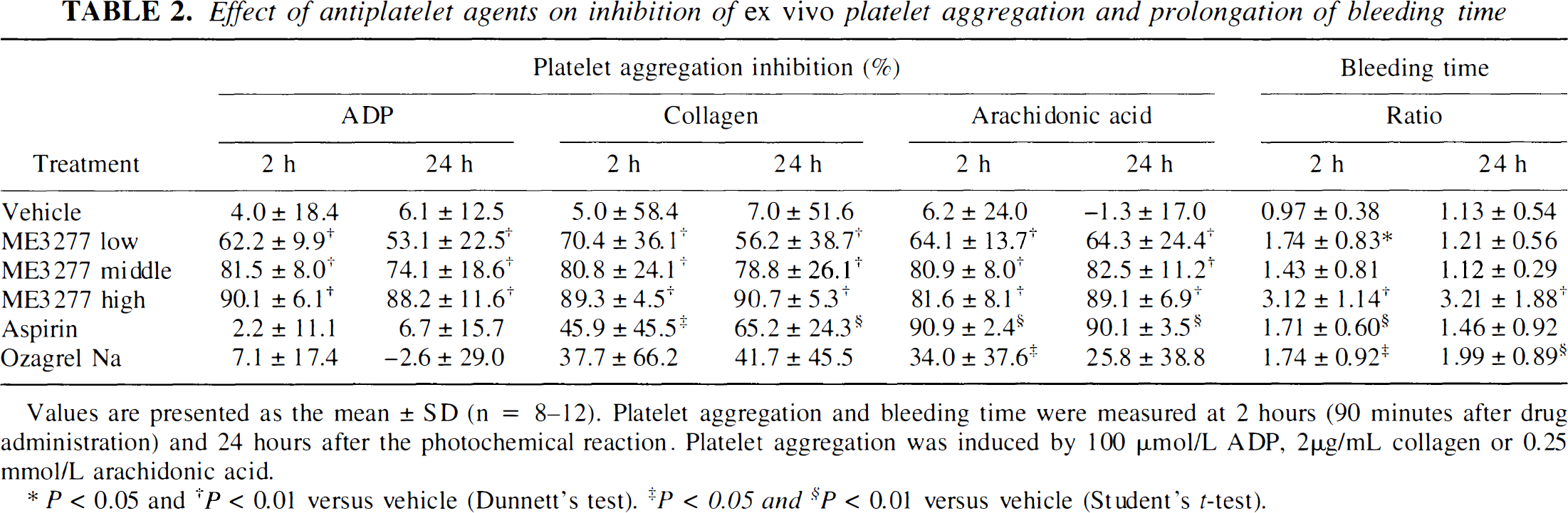
Values are presented as the mean ± SD (n = 8–12). Platelet aggregation and bleeding time were measured at 2 hours (90 minutes after drug administration) and 24 hours after the photochemical reaction. Platelet aggregation was induced by 100 μmol/L ADP, 2μg/mL collagen or 0.25 mmol/L arachidonic acid.
P < 0.05
P < 0.01 versus vehicle (Dunnett's test).
P < 0.05 and
P < 0.01 versus vehicle (Student's t-test).
Effect on bleeding time
The bleeding time of the left thigh in the vehicle group was 3.9 ± 0.4 minutes before the photochemical reaction and was not affected over the 24 hour period. The ratios of bleeding time prolongation are shown in Table 2. Administration of a low dose of ME3277, aspirin, or sodium ozagrel prolonged bleeding time at 2 or 24 hours after the photochemical reaction but by less than 2-fold. A high dose of ME3277 significantly (P < 0.01) prolonged the bleeding time by about 3-fold at both 2 hours and 24 hours.
Blood flow in the middle cerebral artery
Approximately 4 minutes after the photochemical reaction, the MCA blood flow decreased to zero, and the lumen of the middle cerebral artery was occluded by a platelet rich thrombus of decreasing arterial diameter. A schematic illustration and the parameters of MCA blood flow are summarized in Fig. 1 and Table 3, respectively. The lack of a significant difference in occlusion time among the groups revealed that the degree of damage to the MCA was the same. The MCA in most of the vehicle-treated animals had remained occluded for 2 hours after the photochemical reaction. Seventy-five percent of vehicle-treated animals exhibited occlusion even at 24 hours. Administration of ME3277 increased the total reflow time in a dose-dependent manner. A high dose of ME3277 significantly increased the total reflow time (31.7% vs. 3.8% in vehicle; P < 0.05) and had a tendency to improve the 24 hours patency. Sodium ozagrel significantly improved the 24 hours patency but did not improve total reflow time. Aspirin affected none of these variables.

Schematic presentation of the patency of the middle crebral artery (MCA) stem after photochemical reaction. In each column, open and closed areas represent the presence or absence of blood flow, respectively. Striped column at 24 hours represents cyclic flow reduction of the MCA. *Represents death at 24 hours. Data for individual rabbits are presented in each group:
Parameters for blood flow in the middle cerebral artery stem

Values are presented as the mean ± SD (n = 8–12). There were no significant differences among any groups in occlusion time (one-way analysis of variance). Values are significatnly different from the vehicle group, *P < 0.05 (Dunnett's test)t, P < 0.05 (Wilcoxon's test). MBF, basal mean blood flow; OCC, occlusion; CFRs, cyclic flow reductions; PER, perfusion.
Effects on neurologic deficit
The effects of antiplatelets on neurologic function are shown in Figure 2. The animals in the vehicle group exhibited neurologic deficits in most of the parameters. The median value (first and third quartile value) of neurologic score was 9 (7–10). ME3277 significantly decreased the neurologic score to 3.5 (3–5.5) at a low dose, 4.5 (3–5.5) at a middle dose, and 3.5 (1.5–5.5) at a high dose, respectively. Aspirin also decreased the neurologic score to 6.5 (3–7.5), although the improvement was weaker than ME3277. Ozagrel tended to decrease the neurologic score to 6 (2.5–8.5).
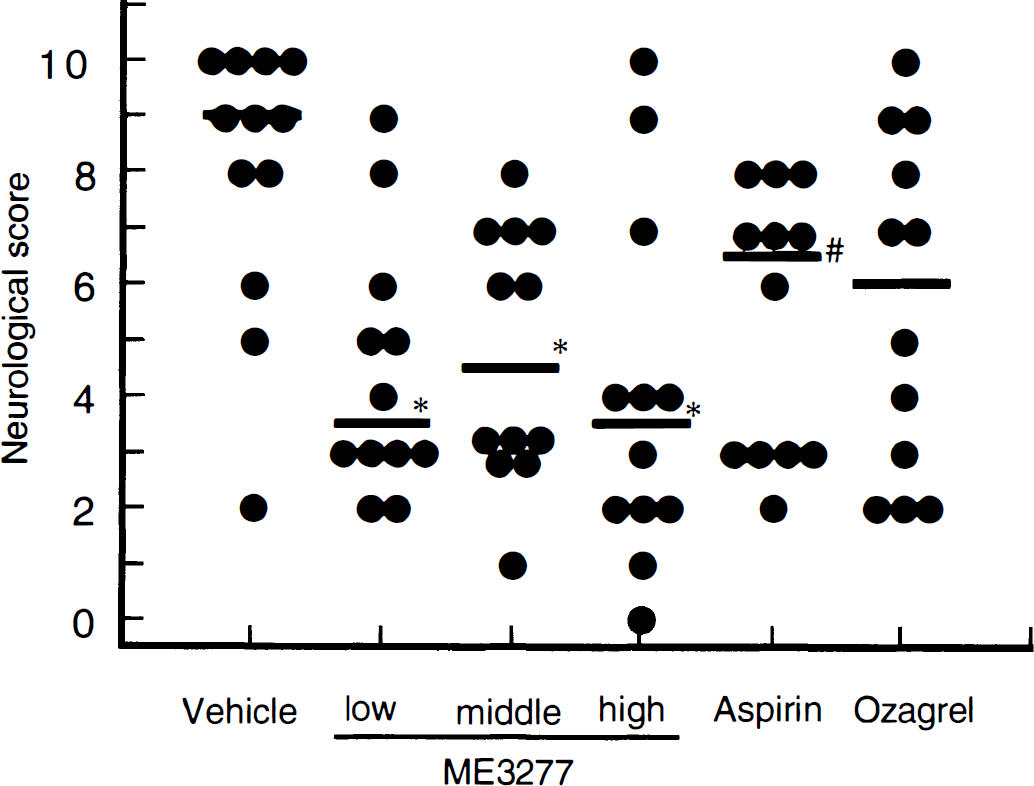
Effect of antiplatelet agents on neurologic deficit. Each point and horizontal bar represents individual score and median value of neurologic score, respectively. The neurologic score was graded for 0 to 10. Higher score represents severe deficit and score 10 means death. *P < 0.05 (Steel's test), #P < 0.05 (Wilcoxon's test) versus vehicle.
Effects on infarct volume
Triphenyltetrazolium chloride staining of brain sections revealed that the brain lesions in the vehicle group consistently expanded to the cortical and subcortical area (Fig. 3). The total infarct volume in the vehicle group at 24 hours was 865.9 ± 286.7 mm3 and 23.6% of the whole brain. The animals who died within 24 hours showed a slightly larger infarct volume (27.0%, n = 4) than vehicle-treated survivors (23.5%, n = 8). ME3277 decreased the infarct volume of total and cerebral cortex area to 13.1% and 4.7% at a low dose, 16.4% and 7.1% at a middle dose, and 12.9% and 5.2% at a high dose, respectively (Fig. 4). Administration of aspirin (20 mg/kg) significantly improved infarct volume to 15.6% and 6.3% in the total and cerebral cortex. Sodium ozagrel tended to reduce the infarct volume.
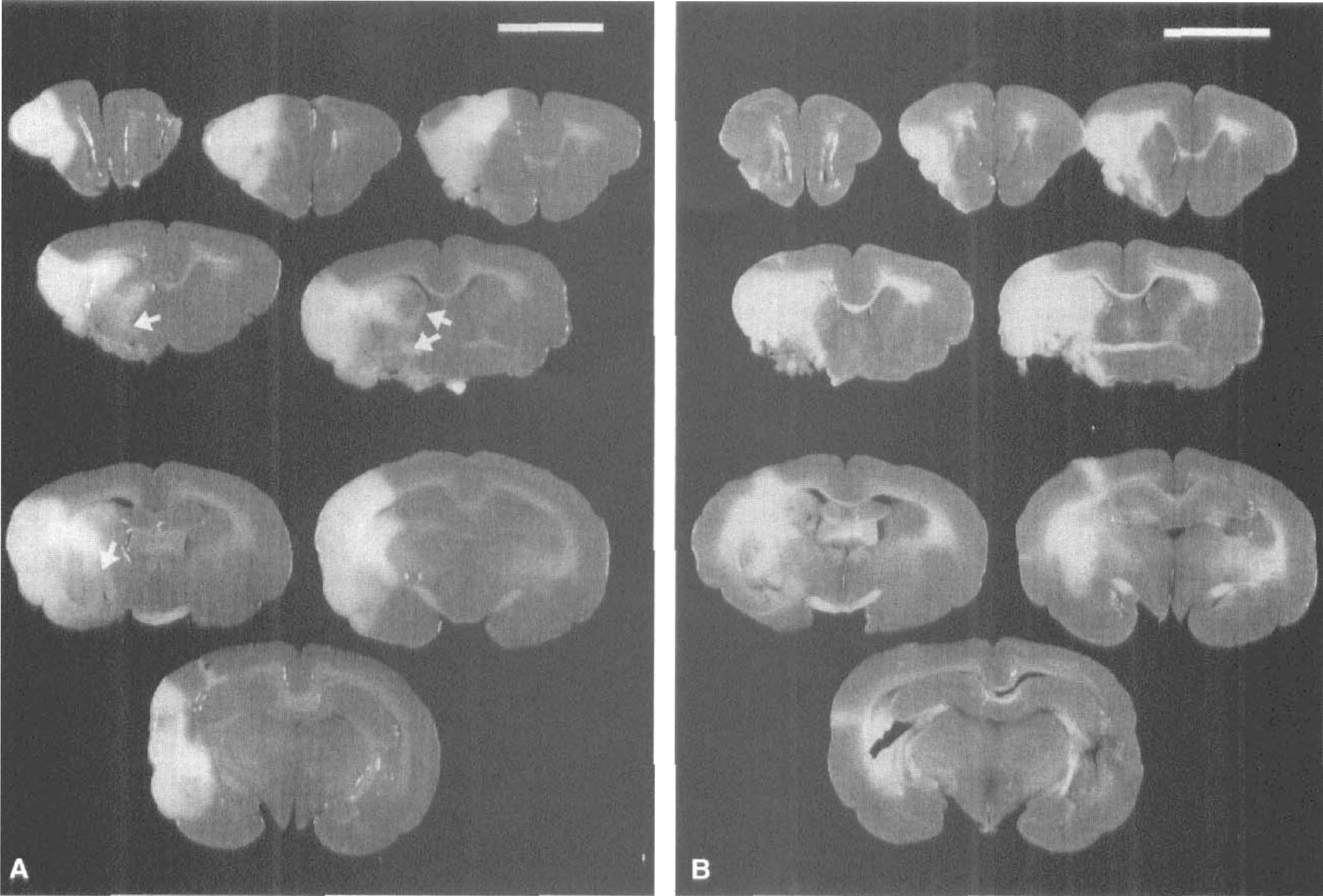
Effect of ME3277 on the brain damage at 24 hours. Coronal brain sections stained with triphenyltetrazolium chloride show the extent of hemispheric infarction in each group;
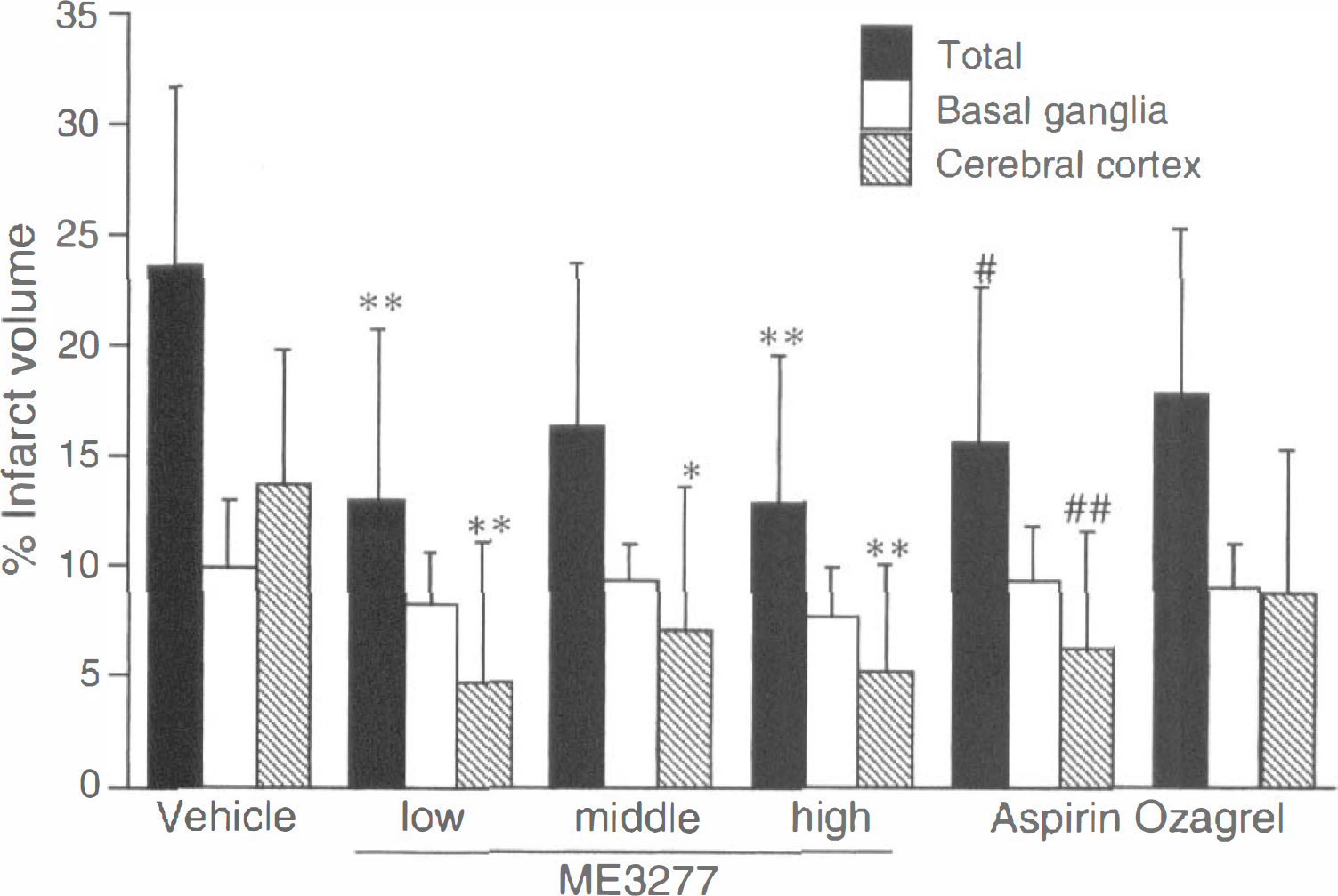
Effect of antiplatelet agents on infarct volume. Each column represents mean ± SD (n = 10–12). *P < 0.05, **P < 0.01, and P < 0.001 versus vehicle (Dunnett's test); #P < 0.05 and #P < 0.01 versus vehicle (Student's t-test).
Animals that showed thrombotic occlusion or CFRs at 24 hours had a significantly higher infarct volume than those which showed perfusion (Fig. 6). In animals that showed occlusion at 24 hours (n = 24), the infarct volume of those that received antiplatelet agents tended to be less than in those given vehicle (data not shown). A correlation was found between total infarct volume and neurologic deficit (r = 0.636; n = 68; P < 0.0001).
Effects on hemorrhage volume
Hemorrhagic infarction (intraparenchymal hemorrhage) in brain slices was recognized as dark brown spots located within the infarct area (Fig. 3A). Most of the animals (69 out of 70) showed the hemorrhagic infarction which varied from 2.4 mm3 to 90.1 mm3 in area and existed both in the cerebral cortex and the basal ganglia. The mean hemorrhage volume in the vehicle group was 36.8 mm3. The hemorrhage volumes normalized to infarct volume and brain volume were 4.6% and 1.0%, respectively. In dead animals, the hemorrhage volume normalized to infarct volume (5.2%, n = 4) was slightly larger than the vehicle-treated survivors (4.2%, n = 8). Treatment with ME3277 or aspirin significantly decreased the hemorrhagic volume, while the hemorrhage volume normalized to infarct volume did not differ between groups (Fig. 5A). The extent of hemorrhagic infarction was significantly correlated to the total infarct volume (r = 0.456; n = 68; P < 0.001; Fig. 5B). Animals who showed CFRs had a significantly higher hemorrhage volume than those who showed occlusion or perfusion (Fig. 6). The hemorrhage volume did not correlate with the bleeding time (r = 0.137; Fig. 7).
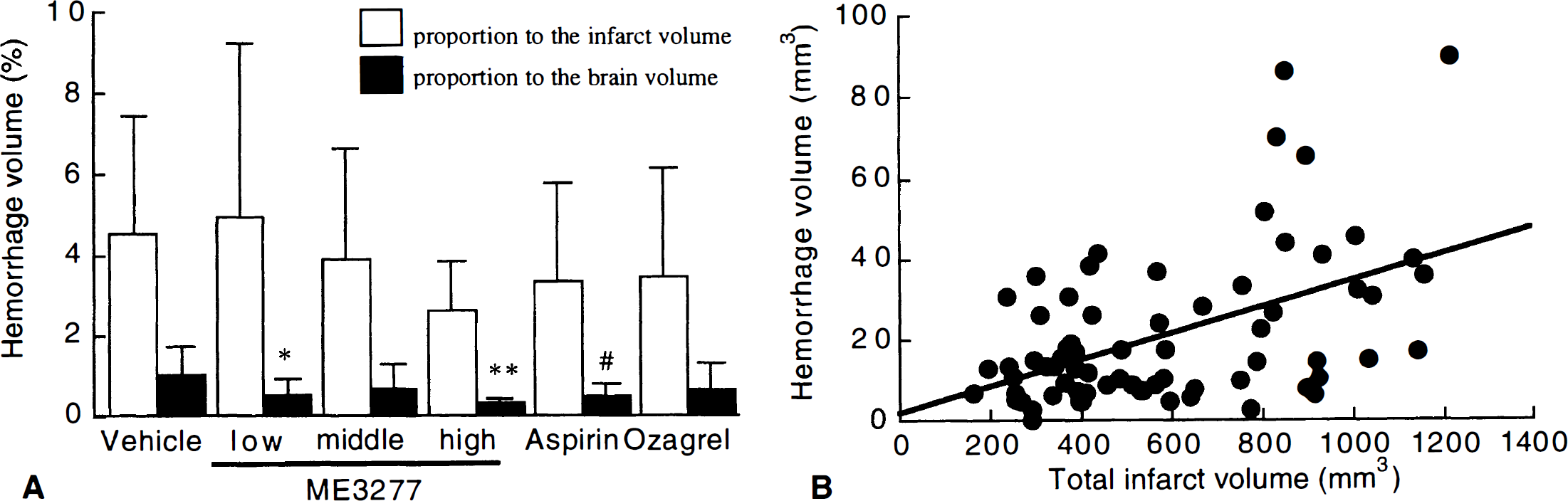
Effect of antiplatelet agents on hemorrhage volume.
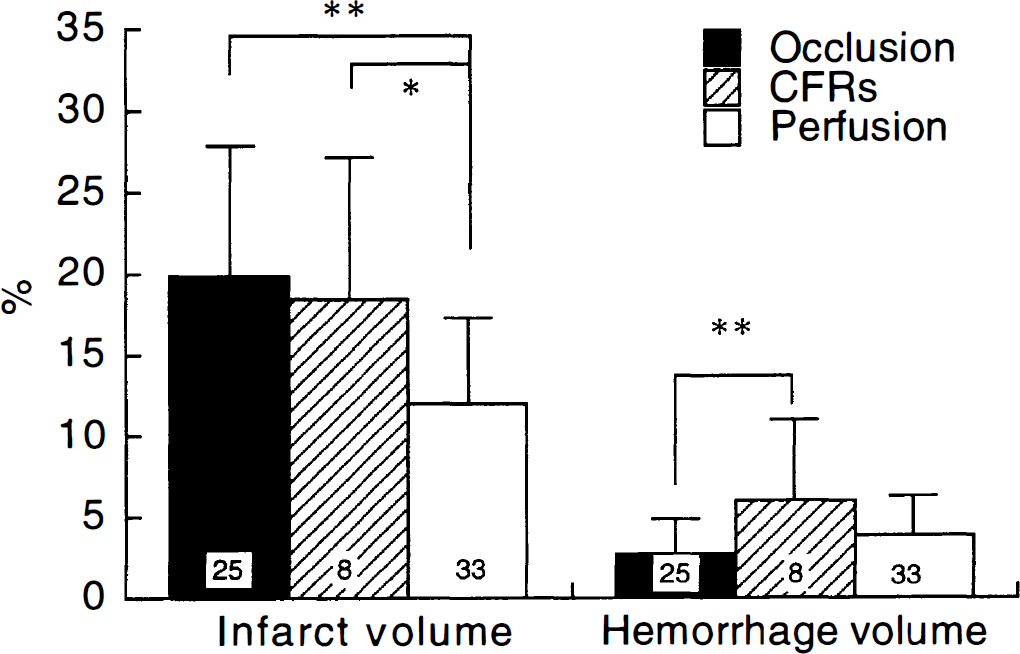
Analysis of total infarct volume and hemorrhage volume by the patency of middle cerebral artery blood flow at 24 hours after photochemical reaction. Each column represents mean ± SD. *P < 0.05 and **P < 0.01 versus vehicle (Tukey–Kramer's test).
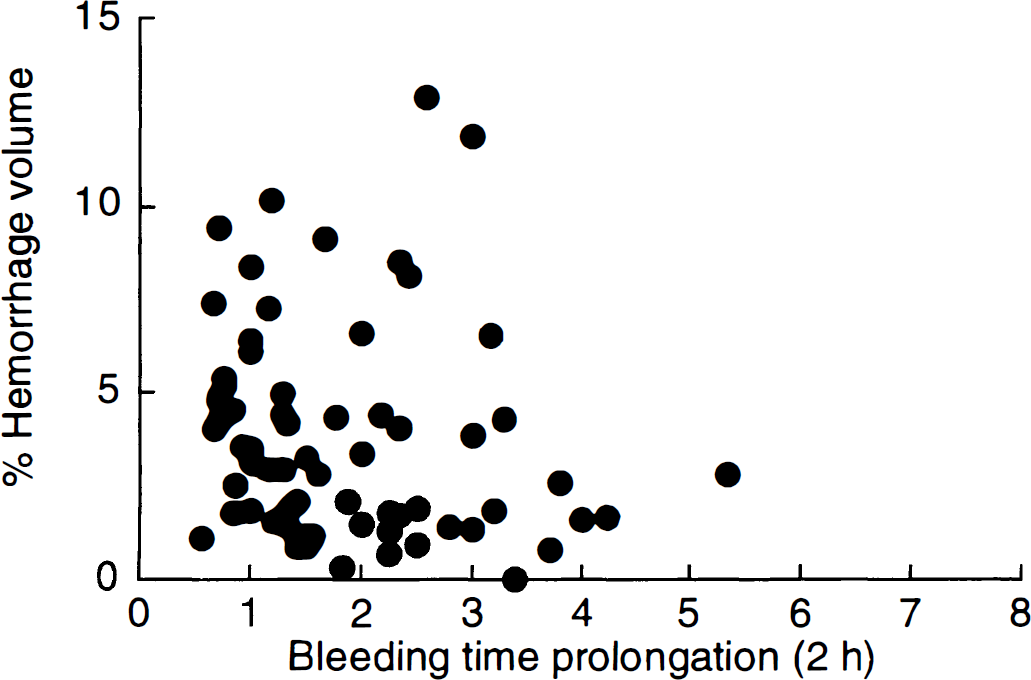
Correlation between bleeding time prolongation and hemorrhage volume (proportion to infarct volume). There is no significant correlation between the factors (r= 0.137).
DISCUSSION
ME3277 or aspirin reduced the infarct volume even when injected 30 minutes after the photochemical reaction. The reduction of infarct volume did not correspond to the increasing dose of ME3277. Therefore, about a 50% reduction of infarct volume seems to be the maximal effect of antiplatelet agents in the present conditions. Infarct volume reflected the improvement in neurologic deficits as reviewed previously (Hunter et al., 1998). Some animals died within 24 hours of arterial occlusion in this model. In the preliminary studies, most rabbits died within 48 hours after MCA occlusion, although the infarct volume already reached the maximal extent at 24 hours after MCA occlusion. Therefore, we used this condition and a 24-hour observation period. We think this condition is relatively similar to the clinical situation because patients with severe cerebral infarction would die in an acute phase. We could not determine any definite reason for death in the current study. The observed change in the hemorrhagic transformation did not explain the cause of death in this model. Therefore, the expansion of infarct volume and subsequent brain swelling are considered most likely to have led to the deaths.
Although antiplatelet agents prolonged the bleeding time, this did not simply reflect the risk of hemorrhagic infarction in the current study. Remarkably, the highest dose of ME3277 did not increase hemorrhagic infarction despite the fact that it prolonged bleeding time 3-fold. This is partly explained by the hypothesis that brain hemostasis is more likely to be dependent on an intrinsic procoagulant than on platelet aggregation (MacDonald et al., 1994). However, since young and healthy animals were used in the current study, caution should be taken in extrapolating the results to elderly humans who have risk factors such as hypertension and atherosclerosis.
Middle cerebral artery stem patency was usually measured in stroke patients with the transcranial Doppler or by angiography because stem blood flow was expected to be a clinical outcome (Ringelstein et al., 1992; Toni et al., 1996). The improvement in MCA stem patency within a few hours after thrombotic occlusion is likely the result of a mechanism for reducing infarct volume and subsequent paralysis in animal models. In contrast to the mechanical occlusion model of the MCA, the thrombotic occlusion model does not allow for a precise control of the ischemic period. However, it mimics the human pathophysiologic status including thrombus formation and spontaneous recanalization. We previously reported that CFRs were observed followed by recanalization in a thrombotic occlusion model of the MCA in guinea pig (Kawano et al., 1998). Similarly, CFRs of the MCA were frequently observed in the current model. The animals with perfusion or CFRs at 24 hours had smaller infarct volumes than those who showed occlusion. In contrast, hemorrhage volume was more severe in the animals with CFRs at 24 hours than in those with occlusion or perfusion. This indicates that recanalization of the occluded artery is necessary to reduce infarct volume, but subsequent reocclusion-reperfusion contributed to the development of hemorrhagic infarction. A GPIIb/IIIa antagonist did not increase hemorrhage volume possibly because of the prevention of reocclusion of recanalized artery. Therefore, the inhibition of CFRs by antiplatelet agents is an ideal intervention to reduce cerebral infarction without affecting hemorrhagic infarction.
In the current study, the MCA stem of most animals given vehicle did not recanalize within 2 hours and 24 hours after the photochemical reaction. Notwithstanding, the preliminary experiments showed spontaneous recanalization of the MCA blood flow followed by CFRs within 6 hours in conscious animals (data not shown). The presence of hemorrhagic infarction in the animals in which the MCA was occluded at 24 hours also supported recanalization because hemorrhagic infarction mainly results from recanalization of the artery responsible. Therefore, we believe that the MCA stem was usually recanalized in the present conditions regardless of whether the rethrombosis was dependent on the drug treatment. ME3277 dose-dependently prolonged the total reflow time within 2 hours and this was reflected in the MCA patency at 24 hours. Because GPIIb/IIIa antagonists themselves do not have thrombolytic activity, ME3277 is thought to inhibit the rethrombosis occurring within the first few hours of thrombotic occlusion. In spite of the improved 24 hours patency, sodium ozagrel did not reduce the infarct volume or total reflow time. Weak antiplatelet activity of sodium ozagrel could hinder the recovery of blood flow early after thrombotic occlusion. The improvement of 24 hours patency with sodium ozagrel is possibly caused by vasorelaxation (Komatsu et al., 1986). The inefficiency of aspirin to improve MCA stem patency under the present conditions could be caused by a weak inhibitory efficacy to platelet aggregation in the damaged region where platelet activation was remarkable. For example, aspirin resistant platelet aggregation such as shear-induced platelet aggregation is predominant in these regions (Barstad et al., 1996).
In addition to maintaining the blood flow of the MCA stem, the recovery in the peripheral blood flow by antiplatelet agents might also contribute to reduced cerebral infarction. Several reports showed that the peripheral blood flow does not necessarily reflect the stem blood flow of the MCA in ischemia-reperfusion models. In these models, peripheral arterial blood flow was prevented by occlusive thrombus consisting of leukocyte, platelet, and fibrin (del Zoppo et al., 1991; Connolly et al., 1997). By improving the peripheral circulation, another GPIIb/IIIa antagonist reduced cerebral infarction in a mouse model of focal ischemia (Choudhri et al., 1998). In the current study, even among the animals in which the MCA remained occluded at 24 hours, antiplatelet agents tended to reduce infarct volume. Therefore, the improvement of peripheral blood flow could also contribute to the reduction of infarct volume by ME3277. Despite no beneficial effect on the MCA blood flow, aspirin reduced infarct volume by a similar extent to ME3277. We can not deny the possibility that other mechanisms independent of antithrombotic effect might participate in the observed effect of aspirin. There are several reports about the antioxidant properties of aspirin and radical scavenge activity of its metabolic derivative salicylic acid (Kuhn et al., 1995). Oberle et al. (1998) demonstrated that aspirin increased ferritin synthesis in endothelial cells. The elevated ferritin is thought to play a broader role in the cellular defense against oxidative injury. It was also reported that ischemic tolerance was observed in neuronal cells from aspirin pretreated rat in vivo (Riepe et al., 1997). The effect of lowering body temperature by aspirin is plausible because our preliminary study and other studies did not indicate any change of body temperature by aspirin when the body temperature was maintained with a heat pad (Bednar, 1999). In general, aspirin at a moderate dose is known to lower the elevated body temperature but not affect normal temperature. The above results indicate that extensive studies will be needed to examine the precise mechanisms of aspirin on reducing cerebral infarction.
Finally, the clinical relevance of this model for acute ischemic stroke in humans may have some limitations as mentioned below. In this study, thrombus was induced by the photochemical reaction which was initiated by endothelial injury of the MCA; this was followed by platelet adhesion and aggregation. Although it is recognized that animal models could not completely reflect the characteristic of human acute ischemic stroke, this type of MCA occlusion is more similar to the human pathophysiology than the previous models. Consistent with human ischemic stroke, MCA occlusion in the present model was prevented by antiplatelet agents and recanalized by spontaneously or thrombolytic agents (Kawano et al., 1998; Umemura et al, 1993, 1995). These did not occur in the previous models induced by mechanical MCA compression or nylon thread insertion. Furthermore, arterial thrombosis in humans is mainly induced by a rapture of atherosclerotic plaque which induces platelet adhesion and aggregation. In general, the atherosclerotic plaque is thought to evolve together with arterial stenosis for more than 10 years, therefore allowing development of collateral circulation (Bornstein et al., 1989). Thus, if thrombotic MCA occlusion occurs, collateral blood flow may play a key role in the development of subsequent cerebral infarction. In our model, the MCA is occluded by thrombus without generation of collateral circulation. Therefore, we think that collateral circulatory formation between our model and human pathophysiology varies greatly.
In conclusion, ME3277, as well as aspirin, reduced cerebral infarction in thrombotic occlusion of the rabbit MCA when given by 30 minutes after the occlusion. ME3277 more potently improved the neurologic functions than did aspirin. These antiplatelet agents did not deteriorate hemorrhagic infarction at doses that prolonged bleeding time by 3-fold. Therefore, antiplatelet therapy with a GPIIb/IIIa antagonist could be useful for the control of acute cerebral infarction.
