Abstract
The authors performed investigations to functionally determine the route of efferent innervation in vivo responsible for cerebral vasodilation mediated by nitric oxide (NO). In anesthetized beagles, electrical stimulation of the pterygopalatine ganglion vasodilated ipsilateral cerebral arteries such as the middle cerebral and posterior communicating arteries. Intravenous injections of NG-nitro-L-arginine (L-NA) markedly inhibited the response to nerve stimulation, and the effect was reversed by L-arginine. Stimulation of the proximal portion of the greater superficial petrosal nerve, upstream of the pterygopalatine ganglion, also produced cerebral vasodilation, which was abolished by L-NA and restored by L-arginine. Treatment with hexamethonium abolished the response to stimulation of the petrosal nerve but did not affect the response to pterygopalatine ganglion stimulation. Destruction of the pterygopalatine ganglion by cauterization constricted the cerebral arteries. Postganglionic denervation abolished the vasodilation, lacrimation, and nasal secretion induced on the ipsilateral side by stimulation of the pterygopalatine ganglion and petrosal nerve. The vasodilator response was suppressed by L-NA but unaffected by atropine, whereas lacrimation and nasal secretion were abolished solely by atropine. It is concluded that postganglionic neurons from the pterygopalatine ganglion play crucial roles in cerebral vasodilation mediated by NO from the nerve, and preganglionic neurons, possibly from the superior salivatory nucleus through the greater superficial petrosal nerve, innervate the pterygopalatine ganglion. Tonic discharges from the vasomotor center participate significantly in the maintenance of cerebral vasodilation.
Keywords
Neurogenic control of CBF and vascular resistance has long been a matter requiring further investigation. Sympathetic vasoconstrictor has only slight influence on cerebral arteries and arterioles, if any (Heistad et al., 1978). Although cerebral vasodilation associated with stimulation of nonadrenergic, noncholinergic nerve has been reported (Toda, 1975), the mechanism of neurogenic vasodilation and the functional role of the vasodilator nerve have been determined only recently. Here we demonstrate for the first time that isolated dog and monkey cerebral arteries denuded of the endothelium respond to perivascular nerve stimulation by electrical pulses or nicotine with relaxations that are abolished by treatment with nitric oxide (NO) synthase inhibitors and restored by L-arginine (Toda and Okamura, 1990a, 1990b). Similar findings also have been reported in other mammals including the human, pig, cattle, cat, sheep, and rat (Toda, 1993; Lee and Sarwinski, 1991; Ayajiki et al., 1993; Ayajiki et al., 1994; Matthew and Wadsworth, 1997; Ignacio et al., 1997). Release NO and its oxidized compounds into the superfusate from superfused cerebral arterial strips by nerve stimulation and stimulationinduced increase in the content of cyclic GMP in the tissue are abolished by NO synthase inhibitors (Toda and Okamura, 1990a, 1991). Histochemical studies demonstrate the presence of perivascular nerve fibers and bundles containing NO synthase immunoreactivity or NADPH diaphorase (Yoshida et al., 1993; Nozaki et al., 1993). These findings led us to conclude that cerebral arteries are innervated by vasodilator nerves in which NO or its stable analog, such as S-nitrosocysteine, acts as a neurotransmitter; the nerve is called “nitroxidergic” or “nitrergic”.
Intracisternal injections of NG-nitro-L-arginine (L-NA) constricts basilar arteries in anesthetized dogs, and the effect is reversed by L-arginine (Toda et al., 1993a). Treatment with hexamethonium clearly reduced the L-NA-induced vasoconstriction, suggesting tonic discharges from the vasomotor center through the nitrergic vasodilator nerve innervating the basilar artery and possibly other cerebral arteries. Nicotine injected into the vertebral artery induces dilation of the basilar artery, which is abolished by treatment with L-NA (Toda et al., 1993a). Stimulation of parasympathetic nerves or ganglia elicits an increment in cerebral blood flow, which is depressed by treatment with NO synthase inhibitors (Morita-Tsuzuki et al., 1993; Iadecola et al., 1993, 1994). Therefore, it is possible that nitrergic nerves contribute to the control of cerebral arterial tone in vivo under resting conditions and when the nerve is stimulated. Although our study on denervation of the pterygopalatine ganglion suggests that the nitrergic nerve originates from this ganglion (Toda et al., 1993b), direct evidence for the route of vasodilator innervation is lacking. The detailed analysis can be performed, only when big mammals are used.
The current study was undertaken to determine whether electrical stimulation of the pterygopalatine ganglion induced cerebral vasodilation mediated by NO in anesthetized dogs and whether the ganglionic denervation altered the diameter of cerebral arteries, and to identify the preganglionic neurons involved and the nucleus in the brain stem possibly responsible for initiating efferent nerve action potentials. In addition, functional roles of neurons innervating the lacrimal and nasal glands were investigated.
MATERIALS AND METHODS
The Animal Care and Use Committee at Shiga University approved the use of beagles in this study. Beagles of both sexes (body weight 7 to 10 kg) were anesthetized with intravenous injections of pentobarbital (30 mg/kg), and stable anesthetic conditions were attained by additional injections as needed. Transfemoral vertebral angiography was performed with a digital subtraction angiography system (DFA-3-30, Hitachi Medical Co., Tokyo, Japan). Arterial blood pressure, heart rate, and body temperature were monitored. To make the right pterygopalatine ganglion microscopically visible so that a fine, bipolar, concentric stimulating electrode could be attached, the zygomatic arch and underlying muscles were excised to reach the fossa pterygopalatine. To reach a bifurcating portion of the facial and greater superficial petrosal nerves at a level of the sensory ganglion geniculi, a postauricular incision was made. The external auditory canal was cut and retracted anteriorly. The facial nerve was cut at the stylomastoid foramen and exposed along its course from this foramen to the bifurcating portion by removing the temporal bone with a cutting burr. The stimulating electrodes were stuck into the greater superficial petrosal nerve distal to the bifurcation. Bone wax was packed into the cavity, which was made by removing the temporal bone to fix the electrode and prevent electric leakage.
The pterygopalatine ganglion or the petrosal nerve was stimulated by electrical pulses of 1-millisecond duration at frequencies of 2, 5, and 10 Hz with a supramaximal intensity (0.3 to 0.6 mA, 5 to 10 times threshold) for 15 seconds; a 2-mL aliquot of iopamidol, a contrast medium, was injected by autoinjector (Angiomat 6000, Liebel-Flarsheim Co., OH, U.S.A.) into the right vertebral artery 5 seconds after the start of stimulation. In the control series, arterial diameters before and during stimulation at each frequency (rising from 2 to 10 Hz) at an interval of 10 minutes, and 5 minutes after termination of the last stimulation were measured by angiography. Effects of treatment for 20 minutes with phentolamine and atropine on the response to pterygopalatine ganglionic stimulation then were determined. Unless otherwise mentioned, studies were performed in the dogs treated with atropine and phentolamine. Modifications by L-NA, L-arginine, and hexamethonium were compared in the vasodilator response to stimulation of the pterygopalatine ganglion and the petrosal nerve. In the experimental series, the arterial diameter was measured 60 minutes after L-NA treatment, 30 minutes after L-arginine, and 20 minutes after hexamethonium. The right pterygopalatine ganglion was damaged by cauterization, and the left ganglion was left intact. Effects of L-NA on the left and right cerebral arteries were compared to determine the tonic innervation from the brain. For the study on denervation of efferent nerves from the pterygopalatine ganglion, only a stimulation frequency of 10 Hz was used.
The data obtained were stored in a digital data recorder (Hitachi Medical Co.), and the diameter was measured by an image analyzer at the selected two points (Fig. 1, inset) only in middle cerebral and posterior communicating arteries that were angiographically visible in all cases. In five dogs, time-dependent changes in the diameter of these arteries were tested by applying the contrast medium up to 12 times for 4 to 6 hours, and no significant change was observed; average changes of the right middle cerebral arterial diameter compared with that at the first application were + 1.5 ± 2.2%, +2.0 ± 2.7%, −1.3 ± 2.0%, and −1.6 ± 2.2% at the second, fifth, tenth, and twelfth applications, respectively, and those of the right posterior communicating arterial diameter were −1.3 ± 1.3%, +0.5 ± 3.8%, +2.1 ± 2.0%, and −1.4 ± 2.0%, respectively.
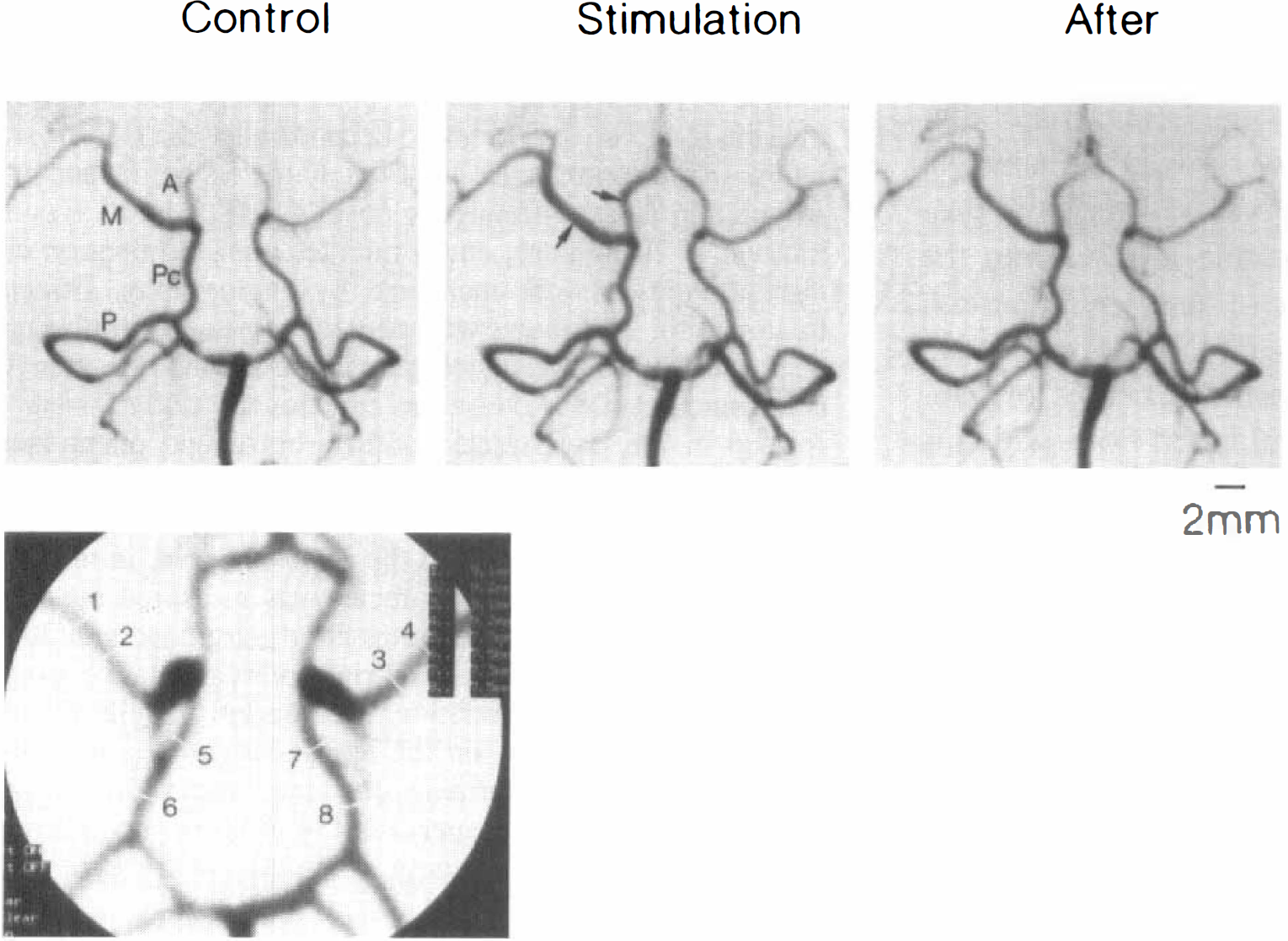
Typical angiographic recordings of the response to pterygopalatine ganglion stimulation (10 Hz for 15 seconds) of anterior (A), middle (M) and posterior (P) cerebral, and posterior communicating (Pc) arteries before (control), during, and 5 minutes after the stimulation in an anesthetized dog. Arrows in the middle recording indicate ipsilateral arteries clearly vasodilated by nerve stimulation. The inset at the lower left shows measurements of the diameter of left and right middle cerebral (1 to 4) and posterior communicating arteries (5 to 8); the numbers and diameters (measured with 10× magnification) of the upper right columns correspond to numbers in the angiographic recording.
The results shown in the text, tables, and figures are expressed as mean values ± SD. Statistical analyses were made using the paired and unpaired Student's t test or Tukey's test after one-way analysis of variance. Drugs used were L-NA (Peptide Institute, Minoh, Japan), L-arginine, hexamethonium bromide (Nacalai Tesque, Kyoto, Japan), atropine sulfate (Tanabe, Osaka, Japan), and phentolamine mesylate (Novartis, Takarazuka, Japan).
RESULTS
Electrical stimulation of pterygopalatine ganglion
In anesthetized dogs, the application of electrical stimulation to the right pterygopalatine ganglion induced vasodilation of ipsilateral middle cerebral and posterior communicating arteries. In the preliminary study, mean values of an increase in the diameter of these arteries by nerve stimulation (10 Hz for 15 seconds) relative to that before the stimulation were 21.1 ± 20.4% and 10.5 ± 4.2% (n = 7); absolute values of the diameter of middle cerebral and posterior communicating arteries were 0.801 ± 0.092 and 0.753 ± 0.062 mm, respectively. The diameters of ipsilateral and contralateral arteries before, during, and 5 minutes after the nerve stimulation at 10 Hz were compared; ratios of the diameters of right and left middle cerebral arteries were 1.03 ± 0.02, 1.21 ± 0.15 (P < 0.01, versus control and after cessation of the stimulation, Tukey's test) and 1.04 ± 0.02, respectively, and those with posterior communicating arteries were 1.01 ± 0.01, 1.11 ± 0.07 (P < 0.01, versus control and after stimulation, Tukey's test) and 1.01 ± 0.02, respectively.
Treatment with atropine (1 mg/kg intravenously, n = 7) plus phentolamine (1 mg/kg intravenously, n = 7) did not significantly change the diameters (with combined treatment, from 0.74 ± 0.05 to 0.75 ± 0.05 mm in the middle cerebral artery, and from 0.67 ± 0.11 to 0.68 ± 0.11 mm in the posterior communicating artery). Treatment with these antagonists lowered systemic blood pressure (mean blood pressure fall, 23.8 ± 12.4 mm Hg, P < 0.01, from 110 ± 6.6 mm Hg) and increased heart rate (heart rate increase, 11.0 ± 0.6 beats per minute [bpm], P < 0.01, from 144.8 ± 17.6 bpm). Vasodilator responses to nerve stimulation were unaffected by the treatment (20.7 ± 15.0% and 12.8 ± 3.4% in middle cerebral and posterior communicating arteries, respectively, versus 19.6 ± 19.2% and 11.3 ± 3.8%, respectively, n = 4). Therefore, in the remainder of this study, anesthetized dogs were treated with the α-adrenergic and muscarinic receptor blockers unless indicated otherwise.
Electrical stimulation (2, 5, and 10 Hz for 15 seconds) of the pterygopalatine ganglion elicited frequency-related dilation of ipsilateral cerebral arteries. Figure 1 illustrates angiographic recordings of the response to ganglionic stimulation (10 Hz) of cerebral arteries. Systemic blood pressure and heart rate were not altered. Vasodilation induced by the stimulation in the middle cerebral, posterior communicating, and posterior cerebral arteries was seen only on the ipsilateral side and disappeared by 5 minutes after termination of the stimulation. Mean values of the increased diameter of middle cerebral and posterior communicating arteries during stimulation at 2, 5, and 10 Hz are summarized in Fig. 2. Intravenous injections of L-NA (5 mg/kg) abolished or markedly inhibited the vasodilation by nerve stimulation, and L-arginine (500 mg/kg intravenously) restored the response.
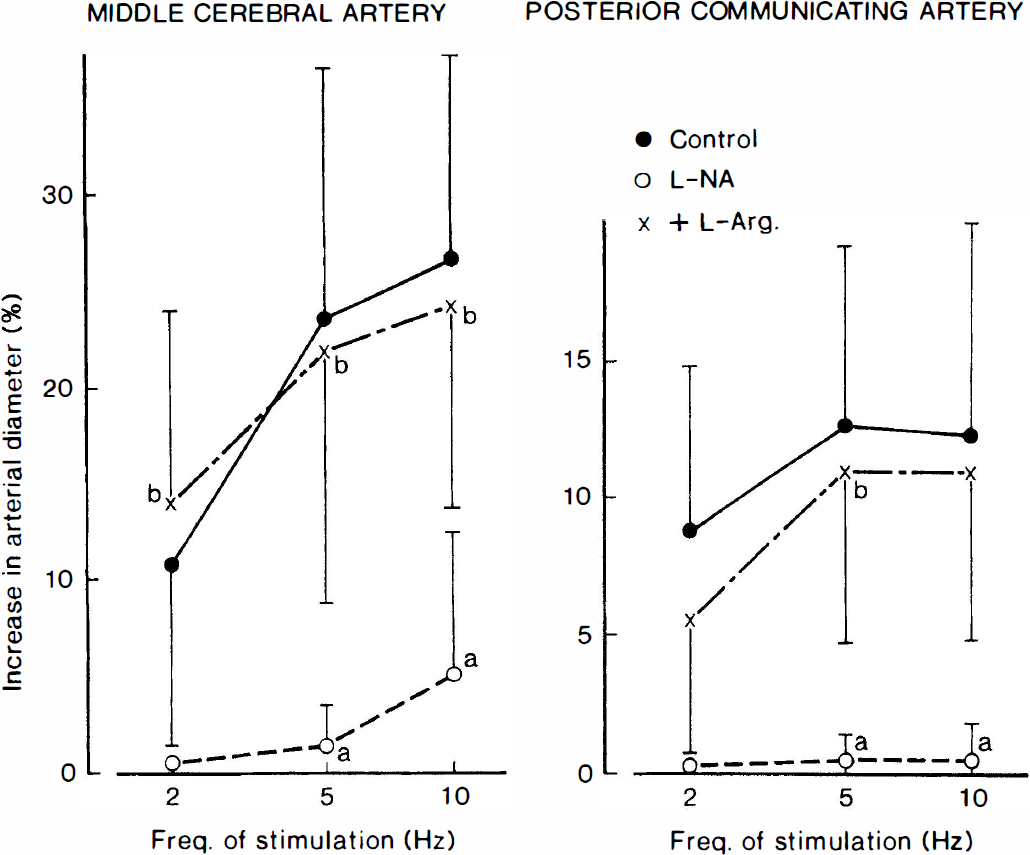
Frequency-vasodilator response curves for electrical stimulation of the pterygopalatine ganglion in ipsilateral middle cerebral and posterior communicating arteries of anesthetized dogs as affected by NG-nitro-L-arginine (L-NA; 5 mg/kg intravenously) and L-arginine (L-Arg; 500 mg/kg intravenously). The ordinate indicates the percent increase in the arterial diameter relative to that before the electrical stimulation; absolute values of the control diameter for middle cerebral and posterior communicating arteries were 0.772 ± 0.116 and 0.733 ± 0.053 mm, respectively. Significantly different from the control, aP < 0.05; significantly different from the value with L-NA, bP < 0.05 (Tukey's test). Seven dogs were used in each experiment. Vertical bars represent SD.
Cauterization of the pterygopalatine ganglion significantly decreased the diameter of ipsilateral middle cerebral and posterior communicating arteries (12.7 ± 2.2%, P < 0.001, n = 5, and 9.7 ± 1.6%, P < 0.001, n = 5, respectively).
Electrical stimulation of greater superficial petrosal nerve
Stimulation of the petrosal nerve (2 and 5 Hz) elicited a frequency-related increase in the diameter of arteries, but increasing the frequency to 10 Hz did not cause additional vasodilation (Fig. 3). The response was markedly depressed or abolished by treatment with L-NA (5 mg/kg intravenously), and L-arginine (500 mg/kg intravenously) restored the response. Typical responses are shown in Fig. 4. Vasodilation by nerve stimulation is seen only in the ipsilateral arteries. As shown in the lower figures, not only the middle cerebral and posterior communicating arteries, but also the anterior and posterior cerebral and basilar arteries, are clearly dilated. In two of six dogs, the basilar artery was dilated by 6% and 10%, respectively, by the nerve stimulation but was unaltered in the remaining four.
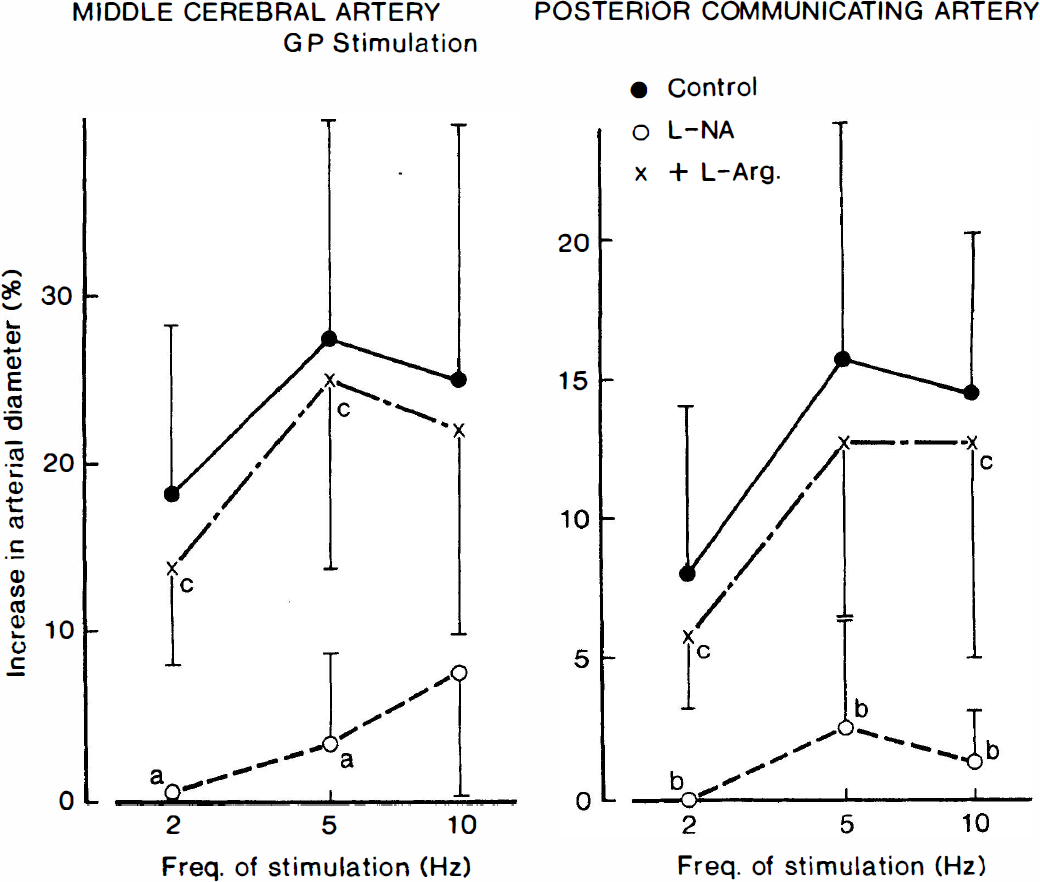
Frequency-vasodilator response curves for electric stimulation of the greater superficial petrosal nerve (GP) in ipsilateral middle cerebral and posterior communicating arteries of anesthetized dogs as affected by L-NA (5 mg/kg intravenously) and L-arginine (L-Arg; 500 mg/kg intravenously). The ordinate indicates the percent increase in the arterial diameter relative to that before the electric stimulation; absolute values of the control diameter for middle cerebral and posterior communicating arteries were 0.822 ± 0.046 and 0.737 ± 0.037 mm, respectively. Significantly different from the control, aP < 0.01, bP < 0.05; significantly different from the value with L-NA, cP <0.05 (Tukey's test). Seven dogs were used in each experiment. Vertical bars represent SD.
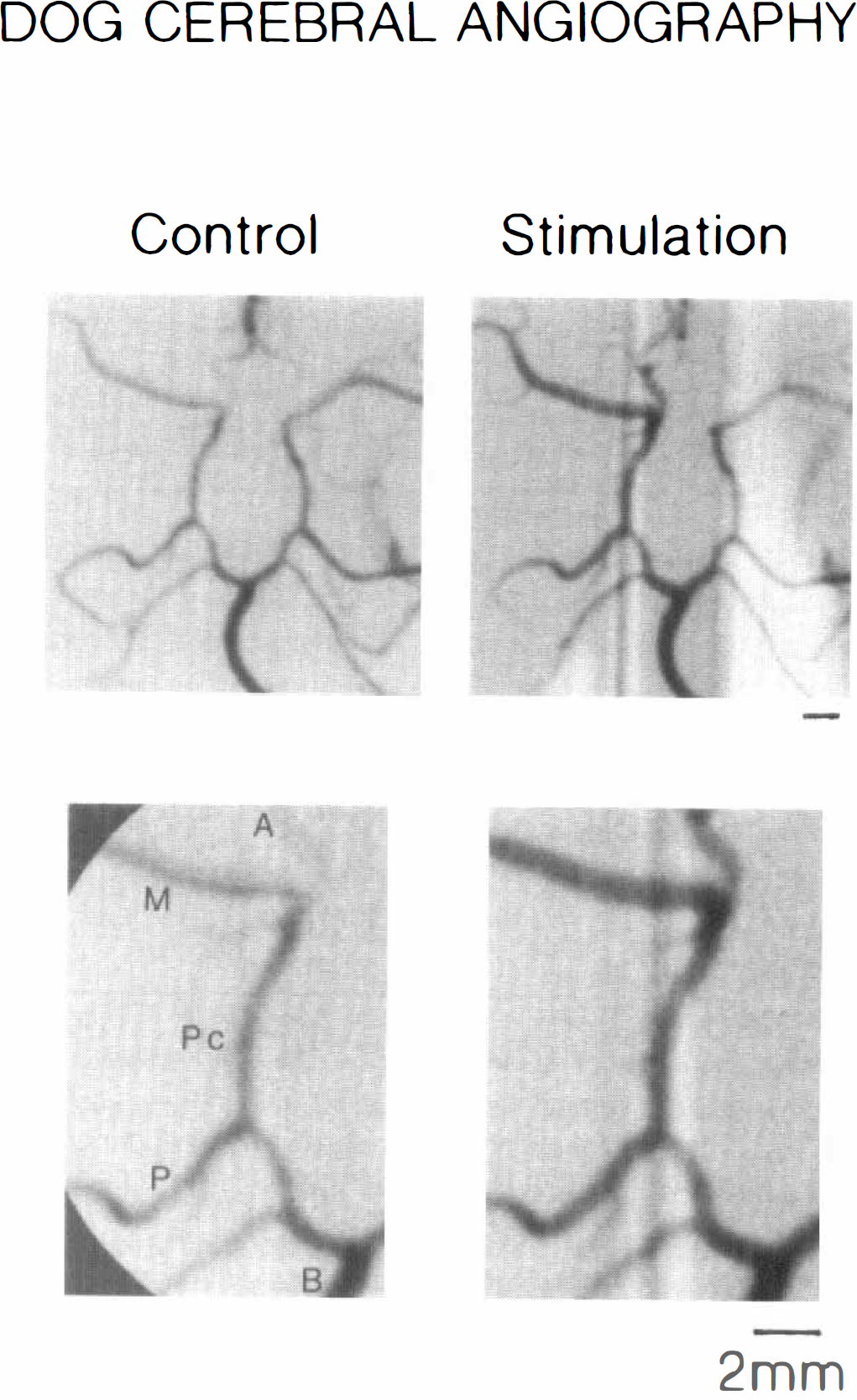
Angiographic recordings of the response to stimulation of the proximal petrosal nerve (10 Hz for 15 seconds) of anterior (A), middle (M) and posterior (P) cerebral, basilar (B), and posterior communicating (Pc) arteries before (control) and during the stimulation in an anesthetized dog. In the upper recordings, ipsilateral and contralateral arteries are compared; during the stimulation, only the ipsilateral arteries are dilated. The lower two photographs show a high magnification of the ipsilateral side.
Effects of L-NA and L-arginine
In nine dogs treated with atropine (1 mg/kg intravenously) and phentolamine (1 mg/kg intravenously), intravenous injections of L-NA produced vasoconstriction of the left and right cerebral arteries; average constrictions were 15.6 ± 9.6% and 14.8 ± 12.0% (P < 0.01), respectively, in middle cerebral arteries and were 15.3 ± 15.3% (P < 0.02) and 16.1 ± 14.1% (P < 0.01), respectively, in posterior communicating arteries. Intravenous administration of L-arginine restored the diameters (14.7 ± 8.7% and 14.2 ± 11.7% dilation, P < 0.01, from the constricted level in left and right middle cerebral arteries, respectively, and 15.1 ± 14.4% and 15.8 ± 15.3%, P < 0.02, in left and right posterior communicating arteries, respectively). In these dogs, systemic blood pressure (systolic, mean, and diastolic pressures, 120 ± 33.6, 96.6 ± 27.0, and 85.4 ± 18.9 mm Hg, respectively, n = 9) was raised by L-NA (135 ± 20.4, 115 ± 17.7 and 103 ± 18.0 mm Hg, respectively), and additional injections of L-arginine lowered the pressures (122 ± 16.2, 98.1 ± 17.4 and 91.2 ± 13.8 mm Hg). Paired analysis indicates that L-NA induced a significant increase (12.1 ± 8.7 mm Hg, P < 0.01) in mean blood pressure, whereas L-arginine caused a significant decrease (11.9 ± 11.1 mm Hg, P < 0.02) from the elevated level. Heart rate was significantly lowered by L-NA from 143.9 ± 17.6 bpm to 119.4 ± 20.2 bpm (P < 0.01, paired t test), whereas L-arginine caused significant increase in heart rate to 121.4 ± 19.2 bpm (P < 0.05, paired t test).
In five dogs in which the right pterygopalatine ganglion was damaged, effects of intravenous L-NA and L-arginine on the size of ipsilateral and contralateral arteries were investigated. The data are summarized in Fig. 5. The arteries once constricted by denervation did not significantly constrict further with L-NA (Fig. 5, left), whereas the innervated arteries clearly constricted with the NO synthase inhibitor (Fig. 5, right). Magnitudes of the constriction by L-NA of the intact arteries were almost identical to those seen in the arteries subjected to denervation and then treatment with L-NA (17% and 16% in right and left middle cerebral arteries, respectively, and 15% and 13% in right and left posterior communicating arteries, respectively). L-Arginine restored the arterial size of only the portion that had been depressed by L-NA.
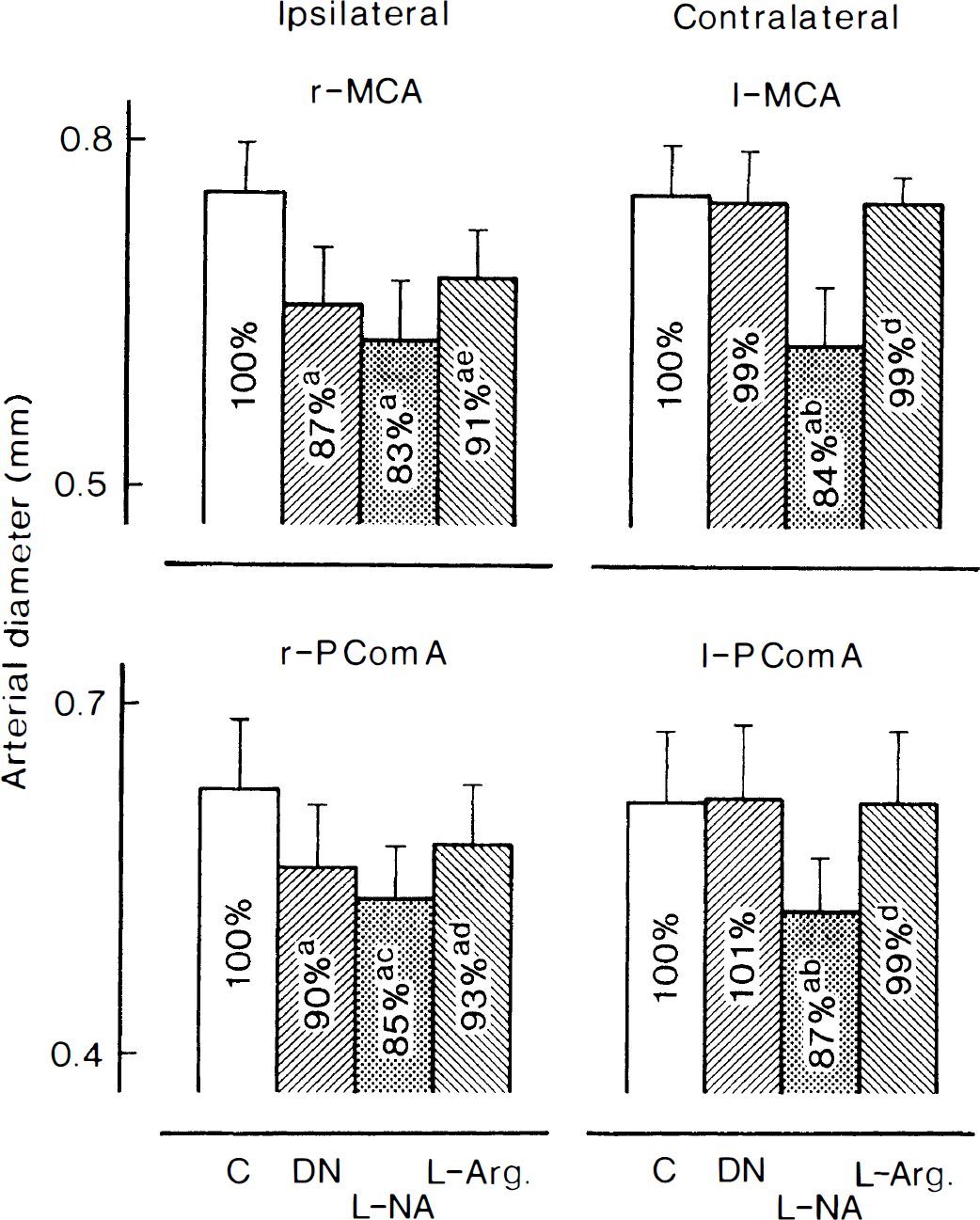
Effects of L-NA (5 mg/kg, intravenously) and L-arginine (L-Arg; 500 mg/kg, intravenously) on the diameter of denervated (left) and innervated (right) middle cerebral (r-MCA and l-MCA, upper panel) and posterior communicating arteries (lower panel) in anesthetized dogs. The ordinate represents the arterial diameter. Numbers in each column denote arterial diameters relative to those before denervation (for right arteries) or injection of L-NA (for left arteries). Significantly different from the control, aP < 0.01; significantly different from the value with denervation (DN), bP < 0.01; cP < 0.05; significantly different from the value with L-NA, dP < 0.01; eP < 0.05 (Tukey's test). Five dogs were used in each experiment. Vertical bars represent SD.
Modification by hexamethonium and denervation of the response to ganglionic stimulation
Vasodilator response of middle cerebral and posterior communicating arteries to electrical stimulation of the pterygopalatine ganglion was not influenced by intravenous hexamethonium (4 mg/kg intravenously) but abolished by denervation of the efferent nerves. In contrast, the response to petrosal nerve stimulation was abolished by the ganglionic blocking agent and also by denervation. The data are summarized in Table 1.
Vasodilator response to stimulation of the pterygopalatine ganglion and greater superficial petrosal nerve (10 Hz) of middle cerebral (MCA) and posterior communicating arteries (PcomA) before and after treatment with hexamethonium or denervation of efferent nerves from the pterygopalatine ganglion in anesthetized dogs
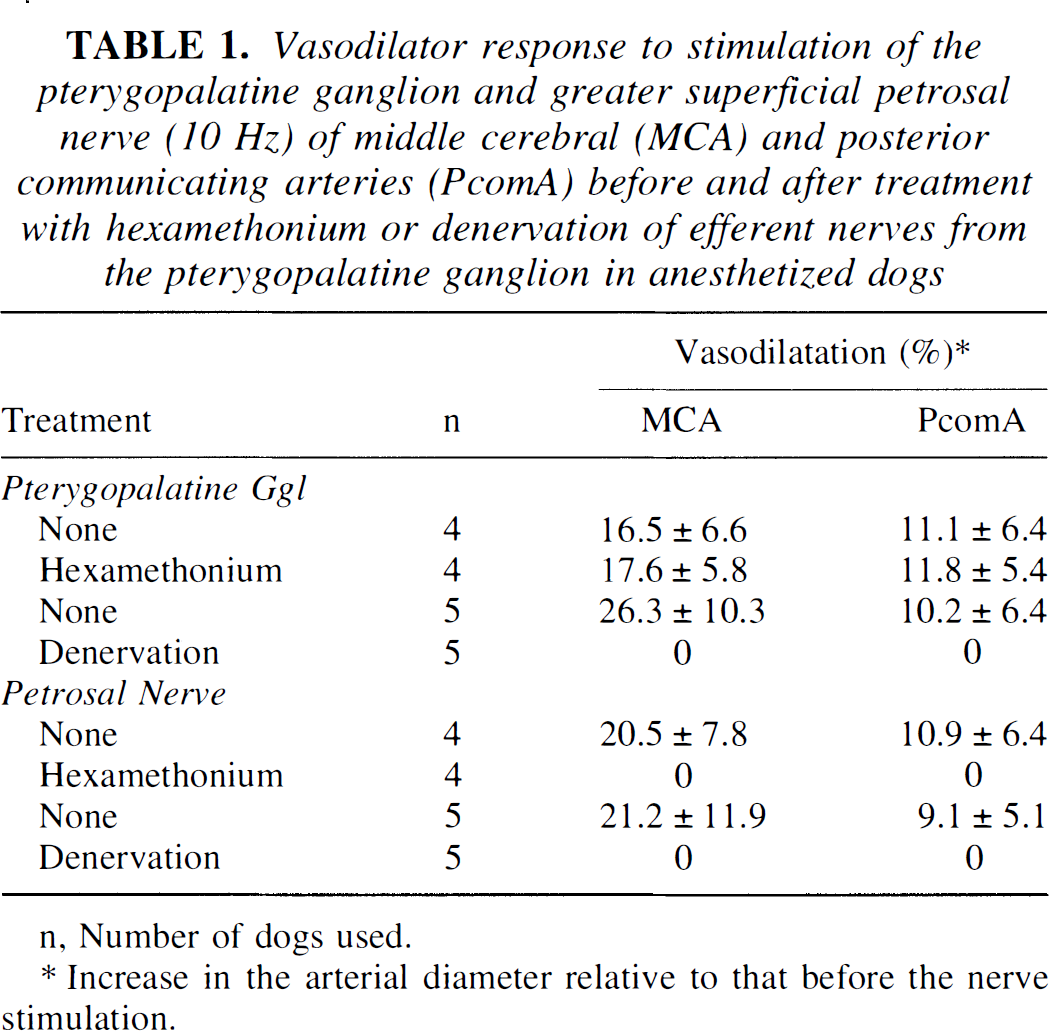
n, Number of dogs used.
Increase in the arterial diameter relative to that before the nerve stimulation.
Effects of ganglionic stimulation on lacrimation and nasal secretion
In anesthetized dogs not treated with atropine, electrical stimulation of either the pterygopalatine ganglion or petrosal nerve induced lacrimation and increased nasal secretion of the ipsilateral side, together with cerebral vasodilation. In this series of experiments, we investigate the involvement of efferent nerves from the pterygopalatine ganglion in these responses and pharmacologically analyze the mechanisms of action. When the nerves (numbered as 1 in Fig. 6, inset) were severed, the vasodilator response was abolished in 9 of 10 dogs and markedly inhibited in the remaining 1 (Table 2). Recordings of the response to nerve stimulation before and after denervation are illustrated in Fig. 6. In this case, electrical stimulation of the petrosal nerve caused the contrast medium to flow backward into the internal carotid artery, possibly by intense vasodilation. The stimulation-induced lacrimation also was abolished by excision of nerve 1, whereas a running nose caused by nerve stimulation was unaffected (Table 2). Sectioning of the nerves 1 and 2 abolished the response of nasal secretion. As described earlier, the vasodilator response was abolished by L-NA, whereas the response of lacrimal and nasal secretion was not influenced in the dogs tested. Atropine (1 mg/kg intravenously) did not affect the arterial dilation by nerve stimulation but abolished the increased secretory responses.
Modifications by denervation (#1 and #2, see the inset in Fig. 6 for cut positions) of vasodilatation of middle cerebral and posterior communicating arteries, lacrimation and nasal discharge caused by electrical stimulation (10 Hz) of either the pterygopalatine ganglion or the petrosal nerve in anesthetized dogs

n, number of dogs used; 'ppt stim.' and 'pet. stim.' indicate dogs in which the pterygopalatine ganglion and petrosal nerve, respectively, were stimulated.
In one dog in which the petrosal nerve was stimulated,
denervation markedly reduced the neurogenic vasodilatation and additional
denervation abolished the response.
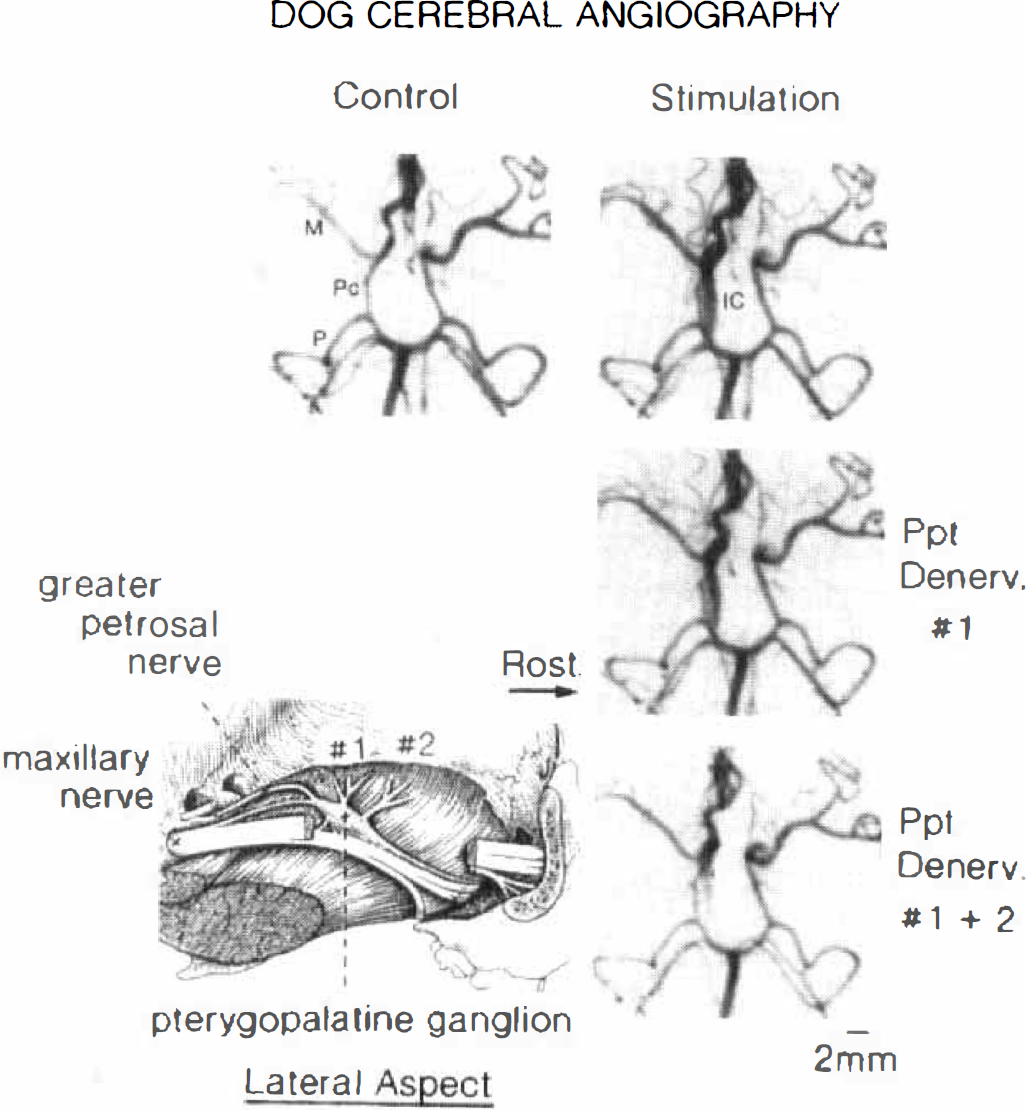
Modification by sectioning (Denerv) of postganglionic neurons from the pterygopalatine ganglion of vasodilator responses to stimulation of the proximal petrosal nerve (10 Hz) of middle (M) and posterior (P) cerebral, posterior communicating (Pc), and intracranial internal carotid (IC) arteries of an anesthetized dog. The inset indicates the lateral aspect (rostral side, right) of the pterygopalatine ganglion and neurons to the ganglion (greater superficial petrosal nerve) and those out of the ganglion (1 and 2). Recordings: top left, before nerve stimulation; top right, during nerve stimulation without nerve sectioning; middle right, during nerve stimulation after denervation of portion 1; during nerve stimulation after denervation of portions 1 and 2.
DISCUSSION
Electrical stimulation of the pterygopalatine ganglion in anesthetized dogs vasodilated the ipsilateral middle cerebral and posterior communicating arteries. Although treatment with atropine and phentolamine significantly lowered the systemic blood pressure, it did not alter the response to nerve stimulation, suggesting that adrenergic and cholinergic nerves are not involved in the neurogenic response and does not significantly modify the vasodilator nerve function. The frequency-vasodilator response curve for the ganglionic stimulation was shifted downward by treatment with L-NA and restored by the additional injection of L-arginine. These findings, together with those obtained from isolated cerebral arteries in response to transmural electrical stimulation (Toda and Okamura, 1996), led us to conclude that the vasodilation by nerve stimulation is mediated by NO synthesized from L-arginine by NO synthase in nerve terminals in vivo and that the nerve arises at least mainly from the ipsilateral pterygopalatine ganglion. It has been reported that stimulation of the pterygopalatine ganglion (Morita-Tsuzuki et al., 1993) in anesthetized rats increases cortical blood flow, and the response is suppressed by L-NA methylester. Although vasoactive intestinal peptideinduced cerebral vasodilation and existence of perivascular nerves containing vasoactive intestinal peptide immunoreactivity have been reported (Owman, 1988), evidence supporting the hypothesis that this peptide acts as a vasodilator neurotransmitter is lacking.
Greater petrosal nerve stimulation in dogs also produced evident vasodilation of the cerebral arteries in a frequency-related manner. The vasodilator response was suppressed by L-NA and restored by L-arginine, as seen in the response to pterygopalatine ganglion stimulation. The only difference was that the response to stimulation of the petrosal nerve was abolished by hexamethonium, whereas sphenopalatine ganglion stimulation was unaffected. The former response is likely to derive from preganglionic stimulation, but the latter is induced by activation of postganglionic neurons. Stimulation of the petrosal nerve does not change the CBF measured by microspheres in anesthetized cats (Busija and Heistad, 1981) and produces cerebral vasodilation that is suppressed by treatment with atropine (D'Alecy and Rose, 1977). However, our study strongly suggests that the induced vasodilation is not associated with stimulation of cholinergic nerves but is mediated by NO from nitrergic nerves. According to Nakai et al. (1993), stimulation by L-glutamate of the greater petrosal nerve cell group in the brain stem, a subgroup of the superior salivatory nucleus, decreases ipsilateral cerebrocortical vascular resistance in anesthetized rats; the response is not influenced by scopolamine but abolished by pentolinium and postganglionic denervation. The superior salivatory nucleus sends off parasympathetic preganglionic neurons through the greater superficial petrosal nerve and transmits information to pterygopalatine ganglion cells and its postganglionic neurons (Spencer et al., 1990). These findings support the hypothesis that the pterygopalatine ganglion contains nitrergic vasodilator neurons that innervate the cerebral vasculature and is innervated by cholinergic preganglionic neurons from the superior salivatory nucleus.
Despite the fact that the innervation of nitrergic vasodilator nerve has been demonstrated by functional and histologic studies (Yoshida et al., 1993; Toda et al., 1993a), basilar arterial dilation was slight and inconsistent in response to greater petrosal nerve stimulation compared with the response of the middle cerebral and posterior communicating arteries. This may result from supplies to the basilar artery of the vasodilator nerve from the pterygopalatine ganglion of both sides (but only unilateral nerve stimulation in this study) or from the other ganglia.
Intravenously injected L-NA vasoconstricted cerebral arteries, and the effect was reversed by intravenous L-arginine. Similar cerebroarterial constriction by NO synthase inhibitors applied intravenously or topically also was obtained in the mouse, rat, goat, pig, and monkey (Rosenblum et al., 1990; Faraci, 1990; Fernandez et al., 1993; Armstead et al., 1994; Okamura et al., 1995). Therefore, release of NO under resting conditions appears to contribute to maintaining the cerebroarterial tone dilated; however, the source of endogenous NO, either from the perivascular nerve or extraneuronal tissues, such as the endothelium, has not been determined. Destruction of the pterygopalatine ganglion elicited significant vasoconstriction, suggesting tonic vasodilator discharges that arise from the vasomotor center. This possibility also has been proposed in anesthetized dogs and monkeys in which basilar arterial constriction by intracisternal L-NA is appreciably reduced by treatment with hexamethonium (Toda et al., 1993a; Okamura et al., 1995). The vasoconstrictor effect of L-NA in denervated arteries was significantly less than that in the arteries of the innervated side (4% versus 16% in middle cerebral, and 5% versus 13% in posterior communicating arteries in Fig. 5); however, the magnitudes of vasoconstriction of both sides, compared with the diameter under control conditions (control before denervation for denervated side), did not differ (17% versus 16%, and 15% versus 13% in Fig. 5). These results suggest that once the NO-mediated neurogenic vasodilation is abolished by denervation, the extent of the constriction elicited by NO synthase blockade is a direct representation of the endothelial contribution to basal tone.
Parasympathetic nerve stimulation, either at the pterygopalatine ganglion or greater petrosal nerve, induced lacrimation and nasal discharge in the ipsilateral side as well as cerebral vasodilation. Treatment with L-NA abolished the vasodilator response but did not influence the other responses, whereas atropine abolished the stimulatory effect on lacrimal and nasal glands but was without effect on stimulation-induced vasodilation. We conclude that the stimulated secretion is mediated by acetylcholine from cholinergic nerves but not by neurogenic NO.
As mentioned earlier, electric stimulation of the pterygopalatine ganglion increases cortical blood flow in anesthetized rats (Morita-Tsuzuki et al., 1993). Unilateral denervation of the ganglion exacerbates the infarction caused by unilateral occlusion of the middle cerebral artery combined with bilateral common carotid occlusion (Kano et al., 1991). On the other hand, intracisternal injections of L-NA constricts the basilar artery by depressing NO synthesis, mainly in the nitrergic nerve (Toda et al., 1993a), although the vasoconstriction is less than that caused by intracisternal oxyhemoglobin (Toda et al., 1991), a NO scavenger, which is expected to be a key substance for the generation of cerebral vasospasm after subarachnoid hemorrhage (Macdonald and Weir, 1991). These findings indicate that neurogenic NO has an important role in cerebral circulation.
In summary, postganglionic nitrergic neurons from the pterygopalatine ganglion innervate the circulus arteriosus, including the middle cerebral and posterior communicating arteries, and the intracranial internal carotid artery in dogs; and preganglionic neurons innervating the pterygopalatine ganglion originate through the greater superficial petrosal nerve, possibly from the superior salivatory nucleus in the brain stem (Fig. 7). This nucleus appears to tonically send off nerve action potentials along the preganglionic and postganglionic neurons, resulting in cerebral vasodilation under resting conditions. Postganglionic cholinergic neurons are expected to innervate cerebral vasculature (Owman, 1988; Toda and Ayajiki, 1990) and lacrimal and nasal glands (Fig. 7); cholinergic neurogenic activations stimulate exocrine secretions.
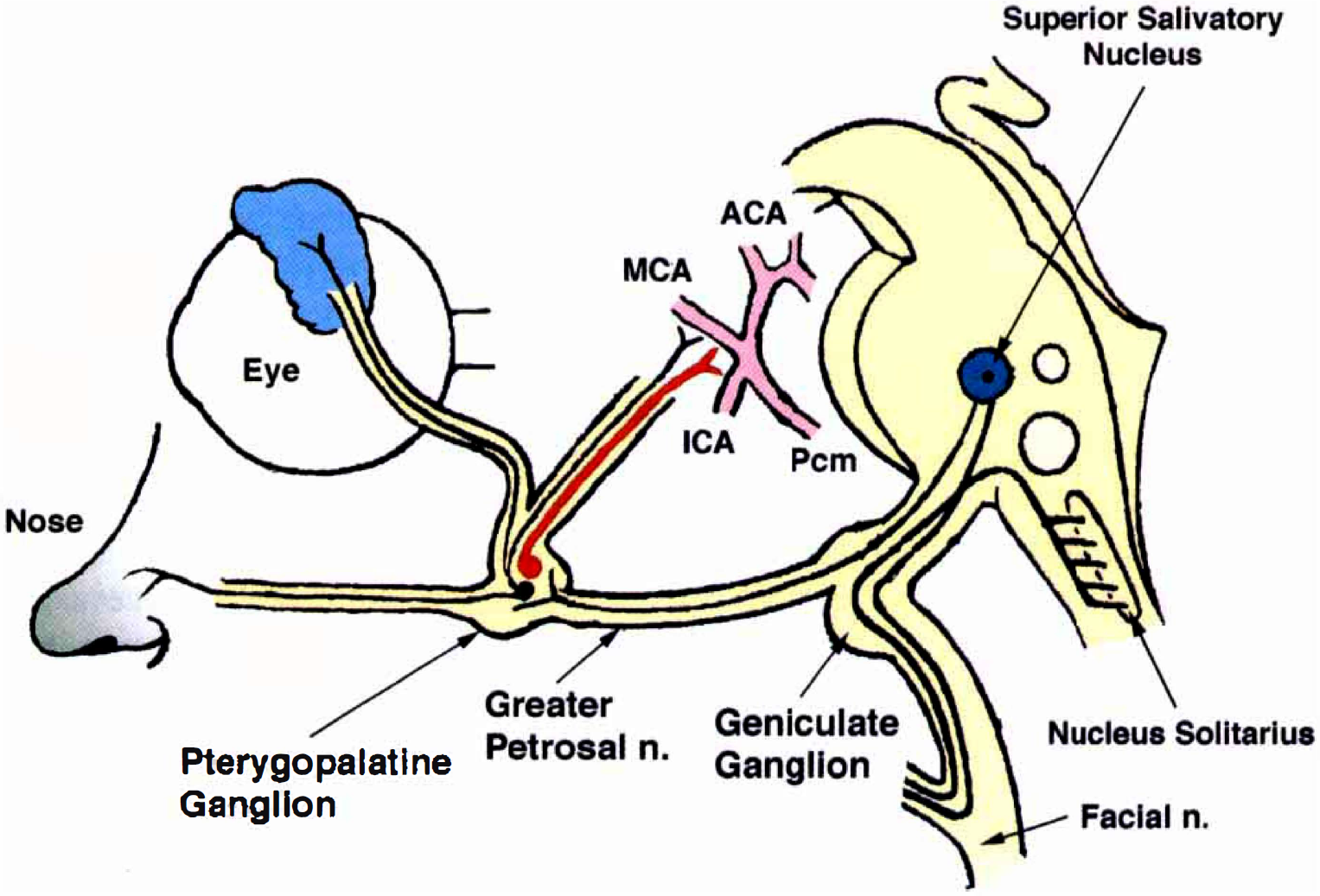
Summary of innervation of nitrergic (red line) and cholinergic nerves (black lines) originating from the superior salivatory nucleus through the geniculate ganglion, greater superficial petrosal nerve, and pterygopalatine ganglion. The nerves regulate functions of the cerebral arteries (anterior cerebral [ACA], middle cerebral [MCA], intracranial internal carotid [ICA], and posterior communicating [Pcm] artery) and those of the lacrimal and nasal glands.
