Abstract
The authors, and others, have recently reported that intracerebral administration of glial cell line-derived neurotrophic factor (GDNF) or osteogenic protein-1 protects against ischemia-induced injury in the cerebral cortex of adult rats. Because these trophic factors are highly expressed in the fetal, but not adult, kidney cortex, the possibility that transplantation of fetal kidney tissue could serve as a cellular reservoir for such molecules and protect against ischemic injury in cerebral cortex was examined. Fetal kidneys obtained from rat embryos at gestational day 16, and adult kidney cortex, were dissected and cut into small pieces. Adult male Sprague-Dawley rats were anesthetized with chloral hydrate and placed in a stereotactic apparatus. Kidney tissues were transplanted into three cortical areas adjacent to the right middle cerebral artery (MCA). Thirty minutes after grafting, the right MCA was transiently ligated for 90 minutes. Twenty-four hours after the onset of reperfusion, animals were evaluated behaviorally. It was found that the stroke animals that received adult kidney transplantation developed motor imbalance. However, animals that received fetal kidney grafts showed significant behavioral improvement. Animals were later sacrificed and brains were removed for triphenyltetrazolium chloride staining, Pax-2 immunostaining, and GDNF mRNA expression. It was noted that transplantation of fetal kidney but not adult kidney tissue greatly reduced the volume of infarction in the cerebral cortex. Fetal kidney grafts showed Pax-2 immunoreactivity and GDNF mRNA in the host cerebral cortex. In contrast, GDNF mRNA expression was not found in the adult kidney grafts. Taken together, our data indicate that fetal kidney transplantation reduces ischemia/reperfusion-induced cortical infarction and behavioral deficits in adult rats, and that such tissue grafts could serve as an unique cellular reservoir for trophic factor application to the brain.
Cerebral ischemia and reperfusion can induce irreversible damage to the brain. Several different classes of drugs have been proposed for pre- and postischemic treatment, but none have demonstrated clear-cut efficacy in clinical trials. Recent studies have indicated that expression of neurotrophic factors is associated with brain injury (Hoffer and Olson, 1997; Liberatore et al., 1997). Systemic administration of kainate or pilocarpine induces glial cell line-derived neurotrophic factor (GDNF) (Humpel et al., 1994; Schmidt-Kastner et al., 1994) and GDNF receptor-alpha (GFR-al) expression (Reeben et al., 1998). Similarly, GDNF mRNA expression in the striatum was increased in animals treated with 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine for 7 days (Tang et al., 1998) or within 6 hours after mechanical injury to adult mouse striatum (Liberatore et al., 1997). In complementary fashion, pretreatment with these neurotrophic factors often decreases the effect of insults. Pretreatment with GDNF reduces 6-OHDA (Hoffer et al., 1994; Bowenkamp et al., 1995), 1-methyl-4-phenyl-1,2,3,6- tetrahydropyridine (Tomac et al., 1995), or axotomy-induced neuronal damage in the nigrostriatal dopaminergic pathway (Beck et al., 1995). GDNF also diminishes damage produced by kainate-induced tonic-clonic convulsions (Martin et al., 1995). It is thus possible that these trophic factors are endogenous neuroprotective agents and that they are upregulated after brain insults to help rescue neuronal circuitry.
Recent studies have shown that hypoxia or brain ischemia induces trophic factor expression just as chemical insults do. GDNF, a protein in the TGF-β superfamily, is upregulated after cerebral ischemia/reperfusion induced by a MCA occlusion (Abe and Hayashi, 1997). We and others have recently reported that intracerebral administration or topical application of GDNF decreases ischemia-induced brain infarction (Wang et al., 1997; Kitagawa et al., 1998b). GDNF decreases MCA occlusion-induced brain edema, the density of TUNEL (+) neurons in the ischemic cortex (Abe et al., 1997; Kitagawa et al., 1998a, b ), and immunoreactivity of caspases-1 and 3 (Kitagawa et al., 1998b). We also found that ischemia and reperfusion-induced nitric oxide release is attenuated by pretreatment with GDNF (Wang et al., 1997). Taken together, these data suggest that GDNF may reduce ischemic insults by attenuating apoptosis and/or necrosis. We and others recently have also reported that osteogenic protein-1, another member of the TGF-β superfamily, is neuroprotective (Lin et al., 1999). Thus, it is possible that GDNF, OP-1, and other TGF-β superfamily proteins are endogenous neuroprotective agents in stroke.
In the absence of perturbations, little GDNF is expressed in adult neuronal tissue (Strömberg et al., 1993). GDNF, however, can be found in fetal neural and non-neuronal tissue. The outer mesenchyme of developing metanephric kidney contains a particularly strong GDNF mRNA signal, which peaks at a gestational age of 16 to 21 days (Nosrat et al., 1996). GDNF family α-receptors, such as GFRα1, are also highly expressed and coex-pressed with Ret mRNA in the developing kidney (Yu et al., 1998). GDNF and GFRα1 in fetal kidney are critically involved in the development of the renal cortex. It has been found that mice which are deficient in GDNF have no kidneys (Moore et al., 1996; Pichel et al., 1996). Most of the animals that lacked GFRα1 also had complete bilateral renal agenesis and ureteral deficits (Cacalano et al., 1998). Other trophic factors in the TGF-β superfamily, such as OP-1 (Dudley and Robertson, 1997), neurturin (Widenfalk et al., 1997), and TGF-β itself (Choi et al., 1997; Basile and Hammerman, 1998) are all highly expressed in the fetal kidney similar to GDNF.
Because these fetal kidney-containing trophic factors have all been shown to be neuroprotective, and because they may synergistically interact with other trophic factors in the brain, we examined the possibility that grafting of fetal kidney may elicit neuroprotection during ischemia. As adult kidney manifests much lower expression levels of these factors, such tissue was utilized for control grafts, in addition to standard vehicle controls.
METHODS
Animals
A total of 32 adult male Sprague-Dawley rats (body weight, >300 g) were used for kidney transplantation. The animals were divided into three groups for behavioral and infarction assays: 1) Fetal kidney transplantation group, which received fetal kidney grafts (n = 7). 2) Adult kidney transplantation group, which received grafts from adult kidney tissue (n = 8). 3) Vehicle controls, which received intracerebral Hank's balanced salt solution injection (30 μL, n = 8). The other nine rats, six grafted with fetal tissue and three grafted with adult kidney tissue, were used for GDNF in situ hybridation and Pax-2 immunostaining.
Transplantation
Fetuses, at the gestational day of 16 to 17, were removed from halothane-anesthetized pregnant rats by cesarean section. The kidneys were dissected, and the ureter and renal pelvis were removed. Kidneys were also obtained from adult rats. These animals were anesthetized with chloral hydrate. About 1 mm3 of cortical tissue from the adult kidney was dissected. Both adult and fetal kidney tissues were transferred into cold Hank's balanced salt solution after three washes with cold saline. Kidney tissues were cut into a slurry before transplantation.
Each recipient rat received one fetal kidney or 9 to 12 μL of adult kidney tissue for transplantation, which was further divided into three parts. The recipient rats were anesthetized with chloral hydrate (400 mg/kg, intraperitoneally). The kidney slurry, each about 3 to 4 μl, was transplanted through a glass micropipette into three cortical areas adjacent to the right MCA, 2.0 to 3.0 mm below the dura (Fig. 1). The approximate coordinates for these sites were: 1.0 to 2.0 mm anterior to the bregma and 3.5 to 4.0 mm lateral to the midline; 0.5 to 1.5 mm posterior to the bregma and 4.0 to 4.5 mm lateral to the midline;, and 4.0 to 4.5 mm posterior to the bregma and 5.5 to 6.0 mm lateral to the midline. The distance between transplant and major branches of MCA was approximately 1.0 to 1.5 mm. The speed of transplantation was 1 μL/min. The needle was retained in place for 5 minutes after each transplantation.
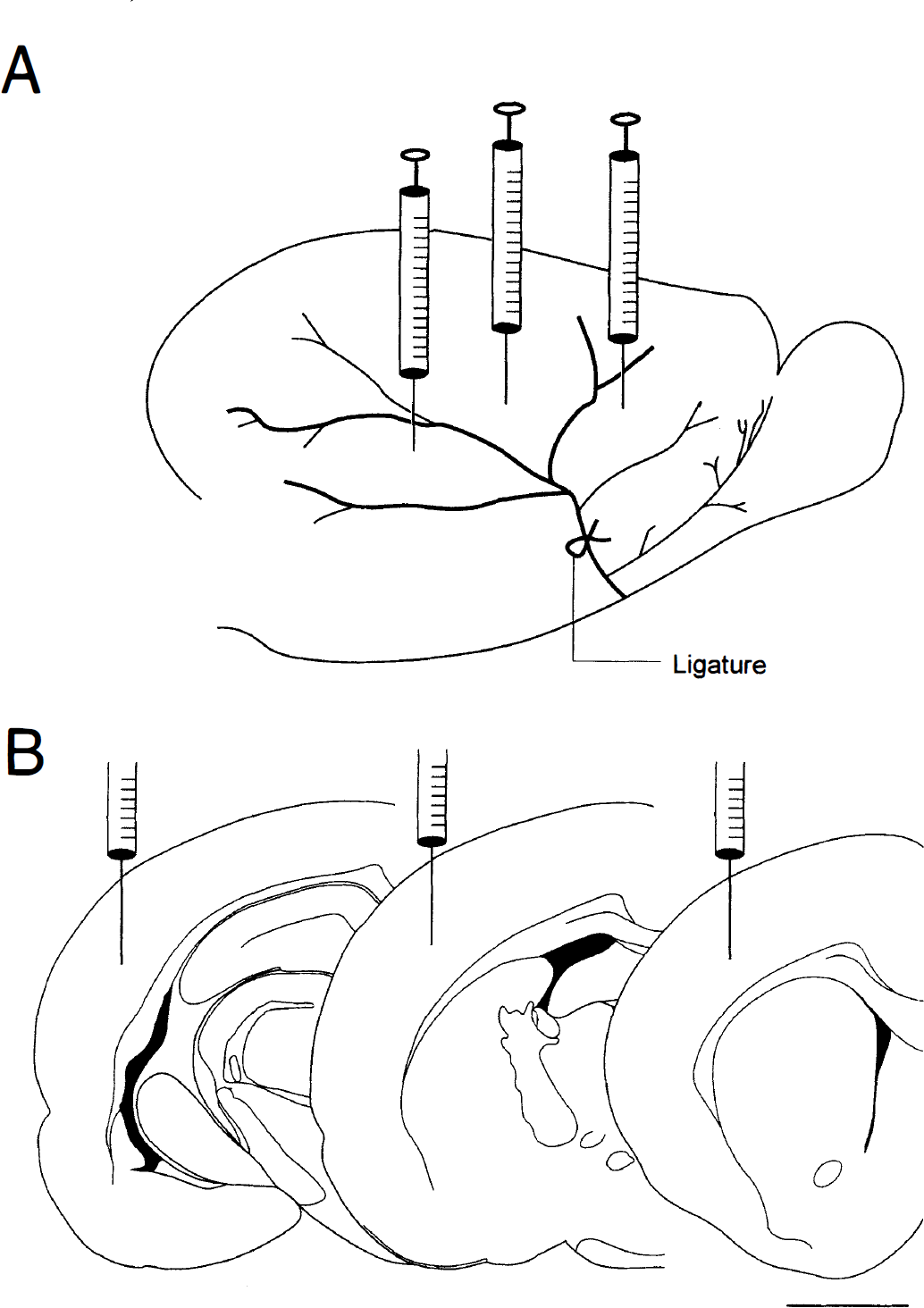
Schematic diagrams of the sites of kidney grafts. MCA was ligated distally between the zygomatic arch and the first bifurcation of MCA above it.
MCA ligation
Thirty minutes after grafting, the anesthetized animals (chloral hydrate, 400 mg/kg, intraperitoneally) were subjected to cerebral ischemia. The ligation of the right MCA and bilateral common carotid arteries was performed using methods previously described (Chen et al., 1986). The bilateral common carotid arteries were identified and isolated through a ventral midline cervical incision. The common carotid arteries were ligated with nontraumatic arterial clips. A craniotomy of about 4 mm2 was made in the right squamous bone. The right MCA was ligated with a 10-O suture for 90 minutes. The craniotomy was then covered with a piece of gelfoam. Sutures were removed 90 minutes later because a 90-minute ligation induces maximal infarctions in rats of this age (Du et al., 1996). After recovery from the anesthesia, the animals were returned to their home cages for a 24-hour reperfusion of the ischemic brain area.
Body temperature was monitored with a thermistor probe and maintained at 37°C with a heating pad during anesthesia. After recovery from the anesthesia, body temperature was further maintained at 37°C using a heat lamp.
Behavioral measurements
Two behavioral assays were carried out after the 24-hour reperfusion. In a qualitative postural reflex test modified from the Bederson behavioral assay (Bederson et al., 1986), animals were classified into two groups according to their neurologic deficits. (1) Behaviorally impaired: Rats keep their left forelimb to the breast and extend the right forelimb straight or twist the upper half of their body when suspended 30 cm above the table or show decreased resistance to lateral push. (2) Behaviorally normal: Rats extend both forelimbs straight and none of the observable deficits described in (1) are present.
The stroke behavior was also quantitatively analyzed using a body swing test (Borlongan et al., 1998). Rats were examined for lateral movements/turning when their bodies were suspended 30 cm above the testing table by lifting their tails. The frequency of initial turning of head or upper body contralateral to the ischemic side was counted in 20 consecutive trials.
Triphenyltetrazolium chloride staining
One day after reperfusion, animals were killed and perfused intracardially with saline. The brain tissue was then removed, immersed in cold saline for 5 minutes, and sliced into 2.0-mm thick sections. The brain slices were incubated in a 2% triphenyltetrazolium chloride (Sigma) dissolved in normal saline for 30 minutes at 37°C and then transferred into a 5% formaldehyde solution for fixation. The area of infarction in each slice was measured using a digital scanner and the Image Tools program (University of Texas Health Sciences Center in San Antonio) as described previously (Wang et al., 1997; Lin et al., 1999). The volume of infarction in each animal was obtained from the product of average slice thickness (2 mm) and sum of infarcted areas in all brain slices.
Pax-2 immunostaining and GDNF in situ hybridizations
Nine rats receiving fetal kidney tissue transplantation were perfused intracardially with 4% paraformaldehyde (100 mL/ min) in 0.1 mol/L phosphate buffer (PB), pH 7.3. Brains were then removed from the skull, postfixed from 2 to 15 hours at 4°C, rinsed with PB, and sequentially transferred to 10%, 12%, and 15% sucrose solutions. They were then frozen on dry ice and sectioned on a cryostat to obtain coronal sections of 20 μm in thickness.
Pax −2 immunocytochemistry. Serial sections were first incubated in a blocking solution (PB supplemented with 4% bovine serum albumin and 0.3% Triton X-100) for 1 hour and then incubated with either blocking solution (control) or a well-characterized rabbit polyclonal anti-Pax 2 antibody raised against amino acids 188 to 385 (Babco, Richmond, CA, U.S.A.). Primary antibody was used at a final concentration of 5 to 20 μg/mL for 48 to 72 hours at 4°C. After rinsing 3×10 minutes in PB, sections were processed with an alkaline phosphatase ABC kit (Vector, Burlingame, CA, U.S.A.). Material was incubated for 30 minutes at room temperature in a 1:200 dilution of biotinylated anti-rabbit IgG. After rinsing with PB, sections were incubated with avidin-biotinylated alkaline phosphatase for 30 minutes to 1 hour at room temperature. Samples were rinsed and the alkaline phosphatase reaction was developed with nitroblue tetrazolium and 5-bromo-4-chloro-3-indolyl phosphate (Life Technologies, Gaithersburg, MD, U.S.A.). Controls consisted of omission of the primary antibody.
GDNF in situ hybridization. In situ hybridization was performed as previously described (Morales and Bloom, 1997). After prehybridization, sections were hybridized at 55°C for 16 hours with [35S]-labeled sense or antisense single-stranded RNA probes (corresponding to GDNF nucleotides 256 to 936 [Gen Bank Accession No. D 88264]) at 107 cpm/mL. Sections were treated with RNase A at 4 μg/mL at 37°C for 1 hour, washed in 1 × standard saline citrate, 50% formamide at 55°C for 1 hour, and in 0.1 × standard saline citrate at 68°C for 30 minutes. Material was dehydrated and exposed on photographic film for 3 days.
Statistics
Data were expressed using mean and standard deviation. One way ANOVA and post hoc Newman-Keuls test were used for statistical analysis.
RESULTS
Behavioral measurements
Previous studies have shown that MCA ligation and reperfusion induce neurologic deficits in rats. We also found that 7 of 8 stroke rats pretreated with vehicle developed postural imbalance as measured by the postural reflex test. Rats pretreated with fetal kidney tissue did not develop this behavioral abnormality. The incidence of postural imbalance in the fetal kidney transplant group was significantly lower than that in the vehicle control group (P < 0.05, Fisher Exact test). In the body swing test, we found that the frequency of swinging the body or head contralateral to the ischemic side was significantly higher in the stroke rats grafted with adult kidney tissue or injected with vehicle than in those pretreated with fetal kidney (Fig. 2, P < 0.05, F = 13.0, df = 33, One way ANOVA + Newman-Keuls' test). There was no difference between the stroke rats that received fetal kidney grafts and nonlesioned rats in the swing test (Fig. 2).
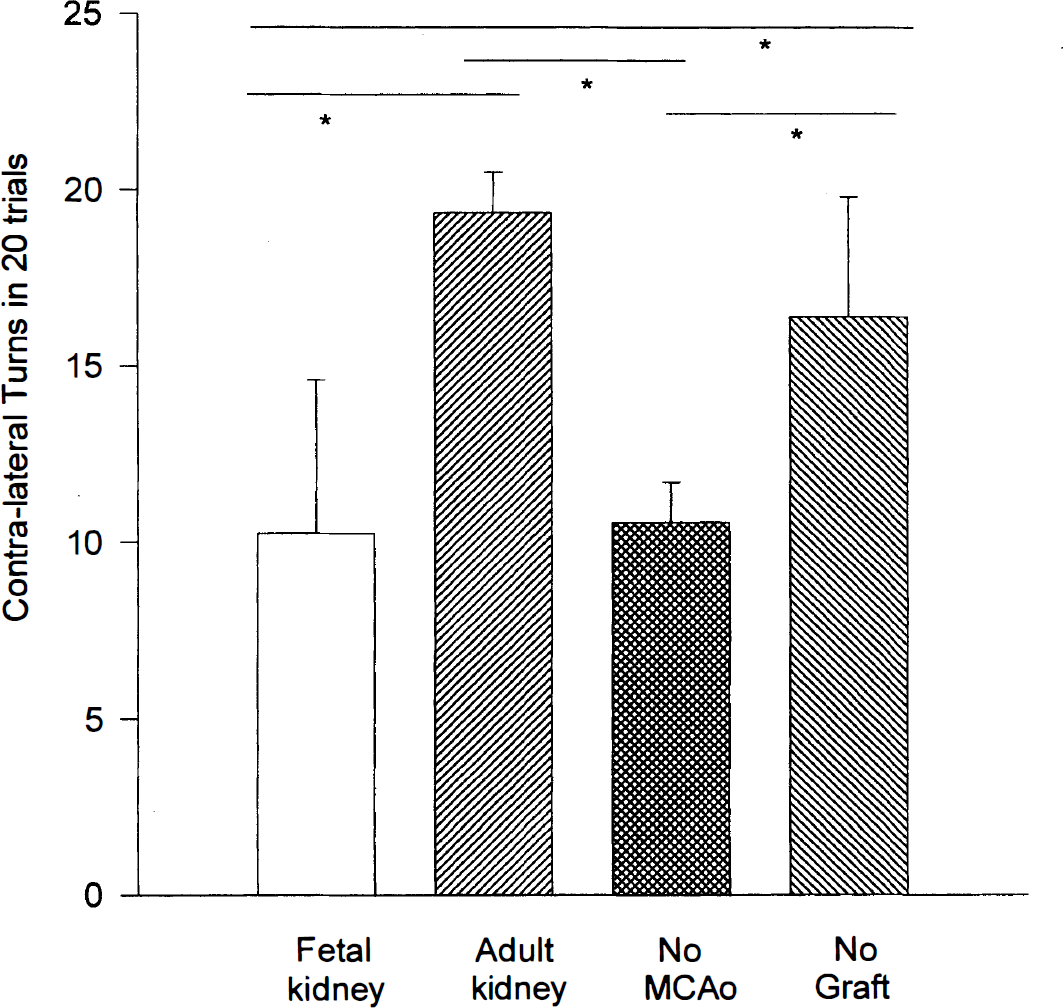
Motor imbalance is improved by fetal kidney tissue transplantation in stroke rats. In 20 consecutive trials, the frequency of swinging the upper body to the side contralateral to the lesion was significantly reduced in the stroke animals grafted with fetal kidney, compared to those pretreated with adult kidney cortex or vehicle (*P < 0.05, F = 13.0, df = 33, one-way ANOVA + Newman-Keuls' test). There is no difference between the lesioned rats that receive fetal grafts and nonlesioned animals (No MCAo).
Brain infarction
We and others have previously reported that ligation of the MCA for 90 minutes and reperfusion for 24 hours cause cortical infarction in rats. In the present study, we found that MCA ligation and reperfusion resulted in clear-cut infarction of the cortex in all the vehicle control animals studied (n = 8, Fig. 3). The volume (99.1 ± 44.2 mm3, mean ± SD) of infarction in vehicle control rats was not different from those in stroke rats pretreated with PB, or without any pretreatment, reported in our earlier studies (Wang et al., 1997).
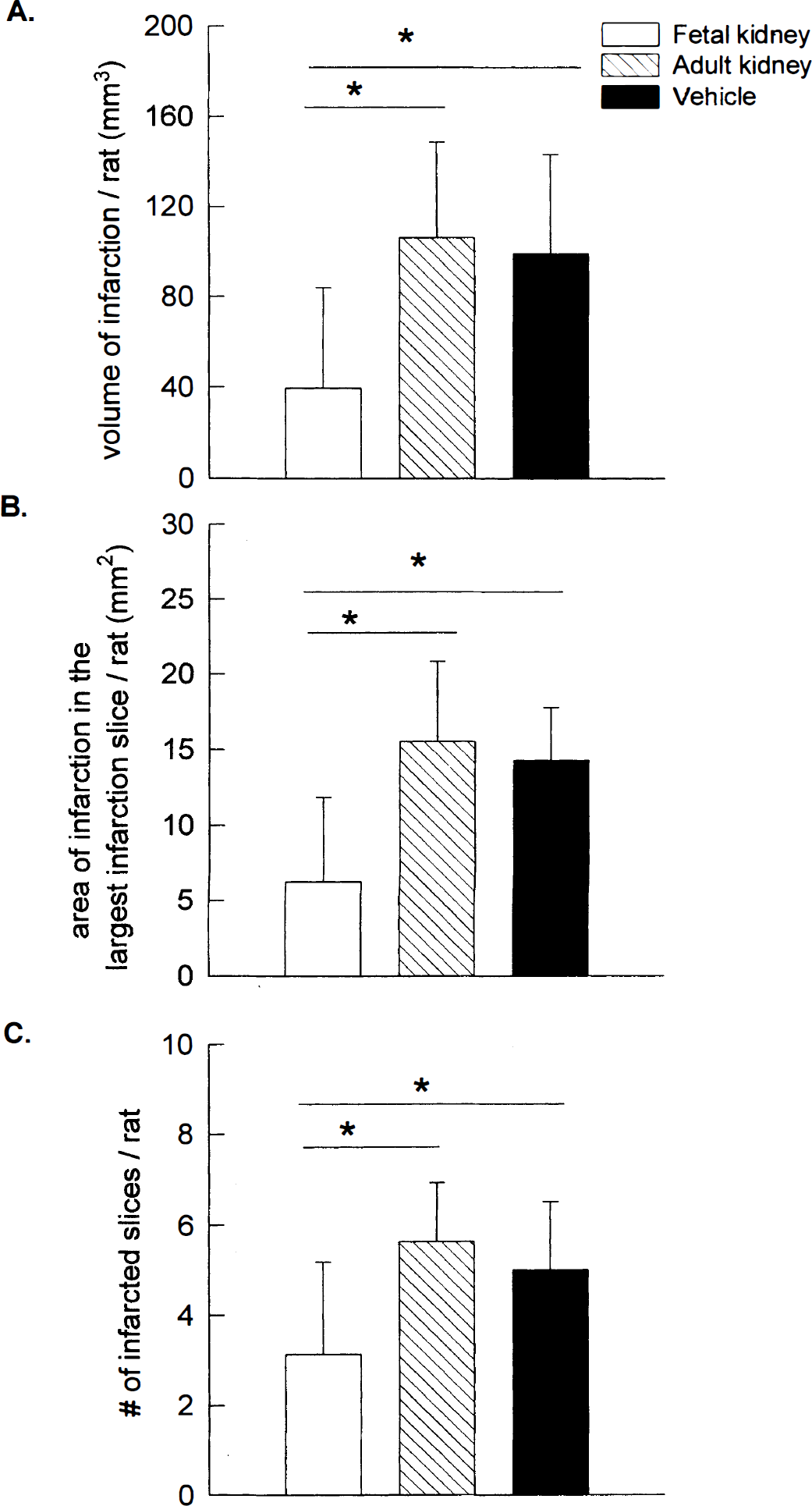
Fetal kidney transplants attenuate cortical infarction induced by middle cerebral arterial (MCA) ligation.
Eight rats were transplanted with adult kidney tissue 30 minutes before MCA ligation. The volume (106.4 ± 42.4 mm3) of infarction was not different from vehicle control animals (Fig. 3). In contrast, the seven rats that received fetal kidney tissue transplants showed significantly smaller infarction volumes (39.5 ± 44.5 mm3) than vehicle control (99.1 ± 44.2 mm3) and adult kidney graft (106.4 ± 42.4 mm3) groups (Fig. 3, P < 0.05, F = 5.15, df = 22, one-way ANOVA + Newman-Keuls' test). The area of the largest infarction in a slice from each rat was also significantly diminished, from 15.6 ± 5.3 mm2 to 6.3 ±5.6 mm2, after the fetal kidney transplantation (Fig. 3, P < 0.05, F = 7.89, df = 22, one-way ANOVA + Newman-Keuls' test). Furthermore, the number of infarcted slices in each rat was significantly reduced (Fig. 3), from 5.0 ± 1.5 slices/rat in vehicle-treated rats to 3.1 ± 2.0 slices/rat in the fetal kidney transplant rats (P < 0.05, F = 4.64, df = 22, one-way ANOVA + Newman-Keuls' test). Taken together, these data suggest that fetal kidney grafts diminish not only the volume but also the extent of infarction in the ischemic brain.
Pax-2 immunoreactivity of fetal kidney transplants in the rat neocortex
We found that Pax-2 immunoreactivity was confined to the fetal kidney transplant (Fig. 4C). The average area of Pax-2 (+) staining was 0.42 mm2. No migration of grafted renal cells into adjacent cortical areas was seen. Sections where primary antibody was omitted showed no immunoreactivity of the fetal kidney transplant (Fig. 4D).
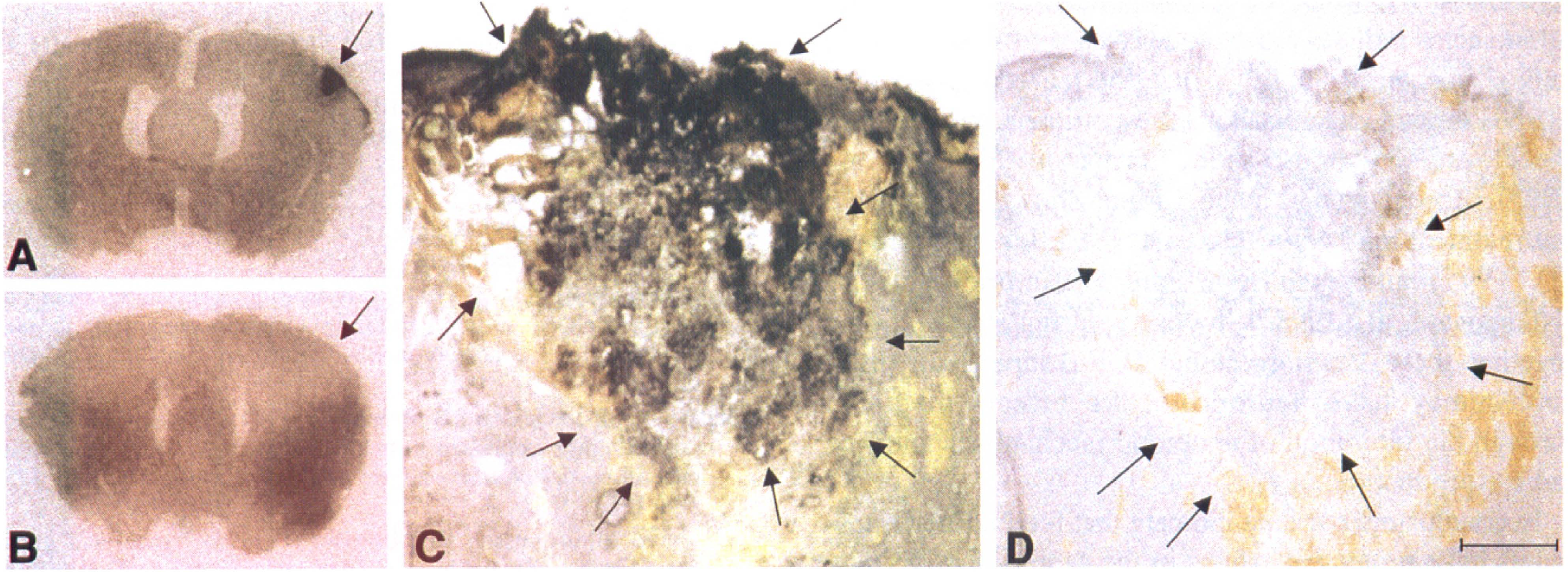
GDNF mRNA and Pax-2 immunoreactivity are present in the fetal kidney transplants in the rat neocortex.
GDNF expression in the kidney grafts
Our unpublished observations have indicated that MCA ligation alone can induce upregulation of GDNF mRNA in the ischemic cortex. To eliminate confounds from the ischemic brain tissue, GDNF mRNA expression was studied in fetal and adult kidney tissue transplanted to the cortex of 6 nonischemic rats. We found that GDNF mRNA was expressed and restricted to the fetal transplant (Fig. 4A, arrow). In contrast, animals (n = 3) receiving adult kidney grafts did not show any GDNF mRNA expression in the transplants (Fig. 4B).
DISCUSSION
Previous studies have demonstrated that fetal cortical grafts can survive in the infarcted brain (Grabowski et al., 1994; Tillotson et al., 1995; Jansen et al., 1997; Mattsson et al., 1997). However, the extent of innervation and behavioral recovery is controversial (Grabowski et al., 1995, 1996; Sorenson et al., 1996) in the stroke animals. In the present study, we found that pretreatment with fetal kidney grafts in the cerebral cortex diminishes ischemia-induced behavioral deficits. It has been reported that the fetal kidney contains high levels of trophic factors and no neurons, as compared to fetal cortex. The fetal kidney graft-induced behavioral restoration data suggest that trophic factors, rather than fetal neurons, are critical for neuroprotection in brain ischemia.
The decrease in infarction after fetal kidney transplantation is probably not attributable to kidney cells in the ischemic zone. Pax-2 immunostaining indicated that kidney tissue was limited to the graft site. The average volume of Pax-2 (+) staining was 0.7 mm3 (0.4 mm2 × 0.6 mm × 3 sites). In contrast, the average reduction of infarction volume was 59.6 mm3 in animals receiving fetal kidney transplants. Furthermore, marked infarction was also found in the animals with adult kidney grafts. Previous studies have indicated that Pax-2 immunoreactivity is only present in the embryonic kidney which contains GDNF/GFRα1 moieties. The expression of Pax-2 in the fetal kidney may be regulated by GDNF mechanisms. Kidneys of E 12.5 GFRα1 knock-out embryos do not express Pax-2 (Cacalano et al., 1998). We also found that GDNF mRNA is expressed only in the fetal kidney transplants. No GDNF mRNA activity was found in the adult kidney graft. We thus postulate that the decreased infarction shown by triphenyltetrazolium chloride staining is not caused by the physical presence of renal cells but that diffusion of substances, possibly GDNF from the fetal kidney, underlies the neuroprotection seen here.
A recent study has indicated that fetal kidney has GDNF-like activity on midbrain dopamine neurons. Co-grafting of fetal kidney with ventral mesencephalic grafts in the 6-OHDA lesioned striatum increases outgrowth of DA fibers from these transplants (Granholm et al., 1998), also suggesting that fetal kidney tissue, like GDNF, has trophic activity for dopaminergic transplants.
Previous experiments have demonstrated that fetal kidney contains not only GDNF but also OP-1, neurturin, and TGF-β (Choi et al., 1997; Dudley and Robertson, 1997; Widenfalk et al., 1997; Basile and Hammerman, 1998). We, and others, have previously reported that these trophic factors are also neuroprotective. GDNF, given on the day of MCA ligation, or OP-1, given 24 hours before ischemia, decreases ischemia/reperfusion-induced brain infarction (Lin et al., 1999; Wang et al., 1997). The decrease in cortical infarction and restoration of behavioral deficits after fetal kidney grafting could represent a summation or interaction of several of these trophic factors. Moreover, previous studies have indicated that GDNF-induced responses are potentiated by OP-1 (Bengtsson et al., 1998) or TGF-β (Krieglstein et al., 1998). It is thus possible that fetal kidney transplants may provide a natural “cocktail” of various trophic factors, which synergistically interact. Future experiments combining fetal kidney grafts and intracerebral injection of blocking antibodies will be needed to dissect out the contribution of the various TGF-β family members (Messer et al., 1998).
Previous studies have indicated that proteins in the TGF-β superfamily are activated during degeneration and regeneration in adult tissues. The expression of TGF-β 1 iriRNA is increased in regenerating renal tubules after acute ischemic injury (Basile et al., 1996). Similarly, TGF-β 1 transcript expression is enhanced in hippocampus after transient forebrain ischemia (Klempt et al., 1992). In vivo and in vitro studies have demonstrated that TGF-β 1 reduces global ischemia- or nitric oxide-induced damage in hippocampal CA1 neurons (Henrich-Noack et al., 1996). GDNF, a trophic factor of TGF-β superfamily, has been shown to protect dopaminergic neurons from damage induced by neurotoxins that elevate intracellular free radicals and produce damage to mitochondrial respiratory enzymes (Kearns and Gash, 1995; Cheng et al., 1998). We and others have reported that ligation of the MCA or local application of excitatory amino acids induces nitric oxide release from the cortical cells (Lin et al., 1996; Liu et al., 1997). Pretreatment with GDNF protects against both the cortical infarction and the elevation in nitric oxide formation (Wang et al., 1997). Recent studies have further shown that GDNF pretreatment markedly reduces TUNEL labeling (Abe et al., 1997) and caspase-1 and 3 immunoreactivity (Kitagawa et al., 1998a) in cortex during stroke. Taken together, these data suggest that TGF-β superfamily molecules may have neuroprotective effects mediated through the inhibition of apoptotic mechanisms and free radical production.
In conclusion, our data indicate that fetal kidney transplants have protective effects in the central nervous system and can reduce ischemia-induced injury in the adult cerebral cortex. The precise mechanisms for this action and determination of the involvement of specific trophic factors require further study. However, fetal kidney may act as a unique cellular reservoir for administration of multiple trophic factors to the injured brains.
