Abstract
To detect endogenous nitric oxide (NO) produced in a rat bacterial meningitis model, the authors applied an electron paramagnetic resonance (EPR) NO-trapping technique. Iron complex with N,N-diethyldithiocarbamate were used as a trapping agent. Experimental meningitis was induced by a mixture of lipopolysaccharide and interferon-γ. Sequential changes of NO formation under meningitis were observed in rat brain tissue by using X-band (9 GHz) EPR spectroscopy, and endogenous NO was detected in the head of a living rat with a 700-MHz EPR system. Inducible NO synthase mRNA expression in the brain tissues also was proven by using a reverse transcriptase-polymerase chain reaction technique.
It is recognized that bioradicals such as nitric oxide (NO) and reactive oxygen radicals play essential and critical roles in many physiologic and pathologic situations, Electron paramagnetic resonance (EPR) spectroscopy has provided unique information on the biochemical reactions of bioradicals in vitro because it can specifically and selectively detect free radicals, It has been claimed that the study on bioradicals should be shifted from in vitro isolated tissue or cell systems to an in vivo situation because the former system is readily affected by atmospheric and unphysiologic oxygen molecule and various constituents of growth media, which may result in the accidental generation of bioradicals (Halliwell, 1996). In vivo EPR spectroscopy using low-frequency microwave (around 1 GHz) is a promising technique for assessing bioradical metabolism and distribution in the bodies of living animals, although the technique is less sensitive than a conventional X-band (approximately 9 GHz) EPR system (Swartz and Walczak, 1996). We have been studying in vivo EPR measurements of endogenously produced excess NO, which is a crucial factor in pathologic situations (Fujii et al., 1998; Suzuki et al., 1998; Yoshimura et al., 1996).
Excessive NO synthesis is involved in various phases during bacterial meningitis (Boje, 1996; Kim and Täuber, 1996; Koedel et al., 1995; Kornelisse et al., 1996; Lieb et al., 1998; Pfister et al., 1992; Tunkel and Scheld, 1993). An elevated concentration of nitrites, a stable metabolite of NO, has been noted in the CSF of both humans (Kornelisse et al., 1996) and rodents (Koedel et al., 1995) during bacterial meningitis. Administering NO synthase (NOS) inhibitors prevents pathophysiologic alterations under bacterial meningitis (Boje, 1996; Koedel et al., 1995). The inducible form of NOS (iNOS) is expressed in response to immunologic stimuli, and after induction it is active for a period of 4 to 24 hours without calcium stimulation and synthesizes NO in cytotoxic concentrations (Dawson et al., 1995). However, NO in living animals or iNOS mRNA during meningitis has never been measured directly. We, therefore, examined NO formation in the rat brain under experimental bacterial meningitis by using both X-band and 700-MHz EPR spectroscopy, and iNOS mRNA expression by a reverse transcriptase-polymerase chain reaction (RT-PCR).
Experimental bacterial meningitis can be induced in animals by intracisternal inoculation of endotoxin (lipopolysaccharide [LPS)), and inflammatory cytokines such as interferon-γ (IFN-γ) and tumor necrosis factor (Boje, 1996; Koedel et al., 1995; Moncada et al., 1991; Tunkel and Scheld, 1993). Accordingly, we used a mixture (5 μL sterile saline solution) of Escherichia coli LPS (500 μg, serotype 055:85, Sigma, St. Louis, MO, U.S.A.) and recombinant rat IFN-γ (5000 U, Biosource, CA, U.S.A.) as an inducer. Female Sprague-Dawley rats weighing approximately 200 g were stereotaxically administered the mixture into the right lateral ventricle. A vehicle (sterile saline of equal volume) was administered to the control group (n = 3). After the inoculation of LPS plus IFN-γ, animals exhibited tufted fur, lethargy, and somnolence in contrast to the alert and active vehicle (sterile saline)-treated animals that exhibited typical rodent behaviors (e.g., cage exploration and grooming).
To detect NO formation in the brain, we used an iron complex with N,N-diethyldithiocarbamate (Fe-DETC)—composed of N,N-diethyldithiocarbamic acid, sodium salt trihydrate solution (200 mg/mL), and an Fe-citrate mixture (40 mg/mL of FeSO4 · 7H2O plus 200 mg/mL Na citrate) (Wako, Japan)—as an NO trapping reagent (Mülsch et al, 1992; Ohnishi, 1998). The Fe-DETC complex reacts with NO to form a stable NO complex (NO-Fe-DETC) and shows a characteristic EPR signal. The DETC solution (2 mL/kg) and Fe-citrate mixture (0.5 mL/kg) were injected intraperitoneally and subcutaneously, respectively, 30 minutes before NO measurement. The animals were decapitated under deep anesthesia with sodium pentobarbital. The right cerebral hemisphere was immediately excised, transferred into an EPR quartz tube (outer diameter 5 mm), and immediately frozen in liquid nitrogen.
The EPR spectra were recorded at a liquid nitrogen temperature by using an X-band spectrometer (TE-200, JEOL, Tokyo, Japan). The sequential changes in NO formation in the brain tissues after the inoculation were followed by the NO-Fe-DETC signal, where the height is proportional to the amount of NO generated in the brain (Lai and Komarov, 1994; Mülsch et al., 1994; Suzuki et al., 1998; Tominaga et al., 1994). The EPR spectra were recorded at 0, 3, 6, 8, 10, 12, and 24 hours (n = 4) after the intraventricular injection of LPS and IFN-γ. The NO-Fe-DETC signal intensified 6 hours after the inoculation, reached a maximum at 8 hours, then weakened after 24 hours (Fig. 1). Signal heights of the NO-Fe-DETC complex are presented as mean ± SD (n = 4). This time course of NO formation is consistent with that observed in a rat's brain during sepsis (Suzuki et al., 1998) and that of iNOS mRNA expression found in a murine primary glial culture after incubation with LPS and IFN-γ (Fujimura et al., 1997).
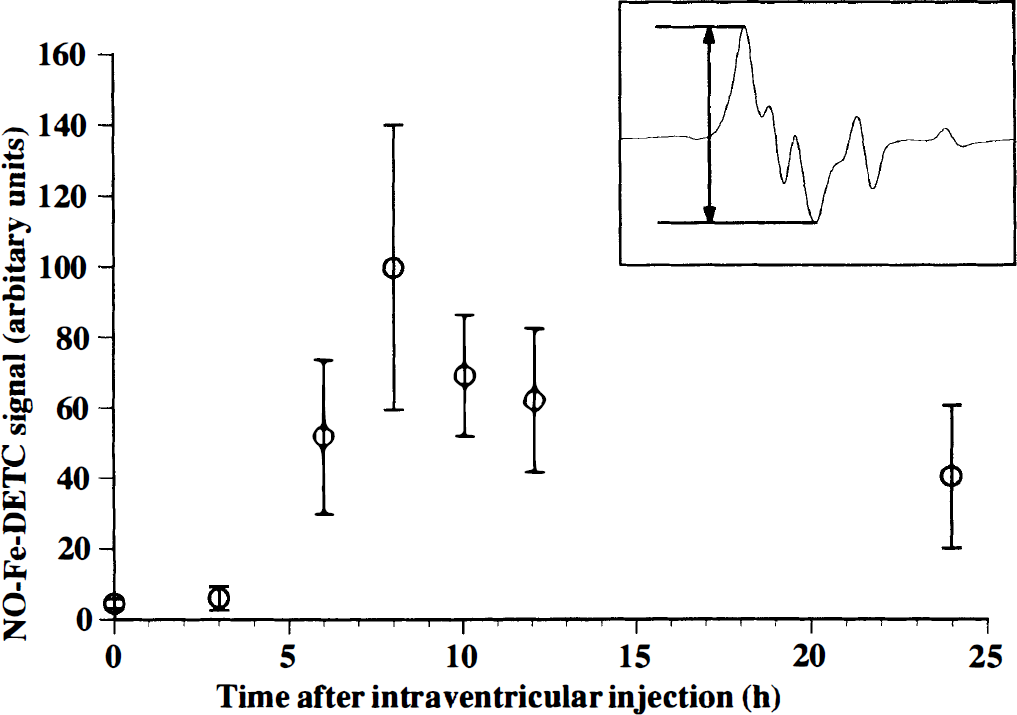
Sequential changes of nitric oxide—iron—N,N-diethyldithiocarbamate (NO-Fe-DETC) signal height in the rat brain tissues during experimental meningitis, which was induced by the intraventricular injection of lipopolysaccharide (LPS) plus interferon (IFN)-γ. X-band electron paramagnetic resonance (EPR) spectra were recorded at 0, 3, 6, 8, 10, 12, and 24 hours after the intraventricular injection of LPS plus IFN-γ (n = 4, mean ± SO). The NO-trapping reagent was injected 30 minutes before taking measurements. The signal was not observed in the brain tissue of the sham-operated group (n = 3) and vehicle-treated control group (n = 3). The inset shows an example of X-band EPR spectrum of NO-Fe-DETC complex at liquid nitrogen temperature. The signal is superimposed on g⊥ component of Cu-DETC signal (Suzuki et al., 1997). The instrument settings were as follows: field scan, 20 mT; sweep time, 4 minutes; time constant, 0.3 seconds; modulation amplitude, 0.63 mT; modulation frequency, 100 kHz; microwave power, 10 mW.
For the inhibition experiment, 100 mg/kg of NG-monomethyl-l-arginine (NMMA, Sigma), an NOS inhibitor, was injected intraperitoneally into the animals 7 hours after the intraventricular injection of the mixture defined above and 30 minutes before the administration of the NO-trapping reagent. When NMMA was injected intraperitoneally, the NO-Fe-DETC signal was not observed, indicating that the signal detected had originated from the endogenously synthesized NO.
Next, we tried to detect NO in the head of a living rat under the experimental conditions that produced the maximum NO signal in the X-band EPR study described earlier. In vivo EPR spectra were measured with a 700-MHz EPR system constructed at our laboratory (Yoshimura et al., 1996), and composed of the following: power supplies; a personal computer; a main electromagnet (air-core, water-cooled, two-coil Helmholtz-designed) equipped with a pair of field gradient coils and field scan coils; and a 700-MHz microwave EPR unit that consisted of a two-gap loop-gap resonator (41 mm in diameter; 10 mm in axial length) and modulation coils. The rat's head was held in the resonator, set by thermostat at 37°C, and located between a pair of gradient coils that were attached to the pole faces of an electromagnet. The NO-trapping reagent and NMMA were injected the same manner as in the X-band measurement.
Eight hours after the intraventricular inoculation of the mixture of LPS and IFN-γ (30 minutes after the injection of Fe-DETC), a weak triplet signal was initially observed. The height of the triplet signal (giso = 2.040, aN = 1.28 mT) increased, peaked 60 to 90 minutes after the injection of Fe-DETC (Fig. 2A–1), then decreased. The line shape of this signal was identical to that observed in the rat's brain under sepsis (Suzuki et al., 1998). The NO-Fe-DETC signal was eliminated by the treatment of NMMA (Fig. 2A–2). No signal was observed in the control group (data not shown). These results demonstrated that the NO-Fe-DETC signal detected in the head of a living rat originated from NOS induced by experimental meningitis.
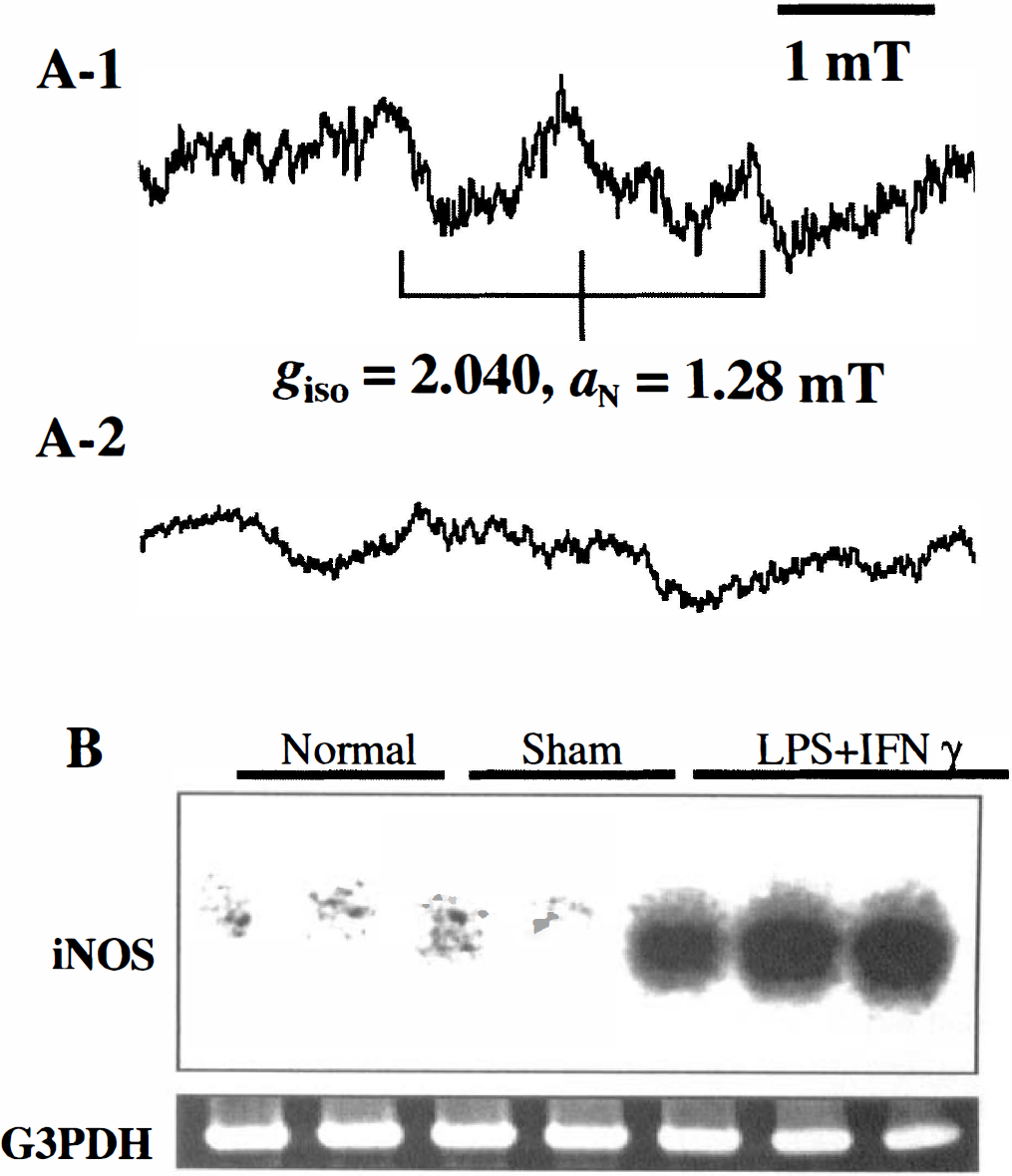
(A) In vivo 700-MHz EPR spectra of the NO-Fe-DETC complex detected in the head region of living rat during experimental meningitis, which was induced by the intraventricular injection of LPS plus IFN-γ. (
In general, iNOS gene expression during bacterial meningitis has been verified with difficulty, probably because the enzyme can be induced in extremely small amounts in living systems, or it is too fragile to be investigated by immunohistochemical methods. It was recently reported that in the brain homogenates of rats with meningitis that was caused by group B streptococci, the enzyme activity of iNOS increased 18 hours after the infection, and the expression of the iNOS could be verified by Western blot analysis (Lieb et al., 1998).
Thus, we tried to identify iNOS mRNA expression in the rat brain tissue during experimental meningitis at 6 hours after inoculation. Induction of iNOS mRNA was investigated with an RT-PCR combined with Southern blot analysis by using a specific cDNA probe for mouse iNOS, as described previously (Doi et al., 1996). Briefly, total RNA was extracted from the brain tissue by using Trizol (Gibco BRL, Grand Island, NY, U.S.A.), and 3 μg of the RNA was used for each RT-PCR. Similarly, mRNA for glyceraldehyde-3-phosphate dehydrogenase was examined as a standard mRNA expressed in the tissue. Southern hybridization was performed using an iNOS cDNA probe radiolabeled with [α-32P]-dCTP by the random primer technique.
It was possible to detect iNOS mRNA successfully 6 hours after the inoculation of LPS plus IFN-γ, although this mRNA was absent in both the normal control and the sham-operated group (Fig. 2B). The results of RT-PCR analysis in the current study definitely show that iNOS is induced during experimental meningitis.
It was demonstrated in the current study that NO is generated in vivo by iNOS in the brain during experimental bacterial meningitis. This is the first report on the EPR detection of endogenously synthesized NO in the head of a living animal during experimental meningitis. In vivo 700-MHz EPR spectroscopy with an NO-trapping technique is a potent tool for detecting NO that is produced excessively in the pathologic brain of living animals. This procedure should contribute to the assessment of pathophysiologic conditions associated with the overproduction of NO.
Footnotes
Acknowledgment
The authors thank Dr. H. Yokoyama, Institute for Life Support Technology, Yamagata Technopolis Foundation, for his comments and valuable assistance with in vivo electron paramagnetic resonance instrumentation.
