Abstract
The effect of systemic administration of human recombinant interleukin-1 receptor antagonist (rhIL-1ra) on behavioral outcome and histopathologic damage after lateral fluid-percussion brain injury of moderate severity was evaluated. In study 1, brain-injured Sprague Dawley rats received timed subcutaneous injections beginning 15 minutes after injury of either 100 mg/kg rhIL-1ra (high dose, total dose = 1900 mg/kg), 10 mg/kg rhIL-1ra (low dose, total dose = 190 mg/kg), or vehicle over 7 days. No effect of low-dose rhIL-1ra was observed in study 1. High-dose rhIL-1ra significantly attenuated posttraumatic neuronal loss in the injured hippocampal CA3 region (P < 0.05), dentate hilus (P < 0.05), and cortex (P < 0.05) but impaired recovery of motor function at 7 days after trauma (P < 0.05). In study 2, rats were pretrained to learn a visuospatial task in a Morris water maze, subjected to fluid-percussion brain injury or sham treatment, and randomly assigned to receive multiple subcutaneous injections at timed intervals of 100 mg/kg rhIL-1ra (total dose = 900 mg/kg) or vehicle over 42 hours, followed by continuous infusion of a lower concentration of rhIL-1ra (20 mg/kg/day, total dose = 100 mg/kg), or vehicle for 5 days using subcutaneously implanted osmotic minipumps. Postinjury administration of rhIL-1ra significantly attenuated cognitive deficits compared with vehicle-treated animals at 42 hours (P < 0.05) but did not affect motor function at 48 hours, 1 week, and 2 weeks. These results suggest that inhibitors of cytokine pathways may be therapeutically useful for the treatment of brain trauma.
Traumatic brain injury (TBI) is known to cause severe neurologic dysfunction in humans and animals. Posttraumatic functional deficits have been associated with secondary or delayed damage, which develops hours to days after the initial trauma. Several studies suggest that this secondary injury may be mediated, in part, by the expression and release of specific cytokines, including the interleukins such as interleukin-1β (IL-1β), interferons, and tumor necrosis factor-α (Rothwell et al., 1993).
Although specific cellular sources of IL-1β in the CNS are unknown, activated microglia, neurons, and astrocytes have been implicated. In addition, systemic IL-1β is transported across the blood—brain barrier by a saturable transport system (Gutierrez et al., 1994). Interleukins such as IL-1β have been reported to play a role in neurodegeneration after TEl or ischemia (Rothwell, 1998). An exacerbation of ischemic brain damage has been observed after localized striatal injections of IL-1β in the rat (Stroemer and Rothwell, 1998). Patients with severe head injury have been shown to have elevated CSF levels of IL-1β (Young et al., 1988), and experimental models of brain injury also are associated with increased levels of IL-1β throughout the injured cortex in the acute posttraumatic period (hours to 2 days) (Taupin et al., 1993). Moreover, Northern blot analyses have revealed significantly increased gene expression (messenger ribonucleic acid [mRNA]) for IL-1β in injured hippocampus and cortex as early as 1 hour after fluid-percussion (FP) brain injury in the rat, which returned to undetectable levels by 24 hours (Fan et al., 1995).
Interleukin-1β has been linked to the development of neuritic β-amyloid plaques in Alzheimer's disease (Sheng et al., 1996), and increased expression of IL-1 has been correlated with elevated levels of β-amyloid precursor protein (βAPP) after human head injury, suggesting a possible role for interleukins in Alzheimer's disease—like neurodegenerative changes associated with brain trauma (Griffin et al., 1994). Interleukin-1β has been reported to stimulate astroglial proliferation and gliosis in the injured brain (Scripter et al., 1997) and has been implicated in the activation of inducible nitric oxide synthase (Lee et al., 1995; Rothwell and Relton, 1993), leading to accumulation of potentially pathologic levels of nitric oxide.
An endogenously occurring antagonist of interleukin-1 (IL-1) receptors has been isolated from the conditioned medium of cultured human monocytes (Hannum et al., 1990). Recombinant human IL-1 receptor antagonist (rhIL-1ra) is a 17-kd polypeptide, which is highly homologous to IL-1α and IL-1β and competitively binds to IL-1 type-I receptors (Hannum et al., 1990). Intracerebroventricular administration of rhIL-1ra has been reported to reduce infarct volume in a variety of models of cerebral ischemia (Garcia et al., 1995; Relton et al., 1996). Recently, the cortical lesion induced by FP brain injury has been shown to be reduced by repetitive postinjury intracerebroventricular infusions of rhIL-1ra (Toulmond and Rothwell, 1995), whereas DeKosky and colleagues report that cells transfected to express rhIL-1ra, when transplanted into the brain, can attenuate trauma-induced increases in nerve growth factor (NGF) levels (DeKoskly et al., 1996). Like IL-1β, rhIL-1ra crosses the blood—brain barrier through a transport system, although only 0.3% to 0.6% of a systemically administered dose per gram of brain tissue enters into the eNS (Gutierrez et al., 1994). Pretreatment or posttreatment with zinc protoporhyrin, an IL-1 receptor antagonist and hemeoxygenase inhibitor, also was reported to reduce total size and area of infarct after middle cerebral artery occlusion (Kadoya et al., 1995; Yamasaki et al., 1995), whereas infarct volume and striatal neuronal loss after experimental hypoxia—ischemia have been shown to be reduced by subcutaneous injection of rhIL-1ra in the neonatal rat (Martin et al., 1994). The current study is the first examination of the effects of systemically administered rhIL-1ra on neurobehavioral and histologic outcome after experimental TBI in the rat.
METHODS
Fluid-percussion injury
A two-part study was conducted using adult male Sprague-Dawley rats (n = 102, 348 to 422 g). Rats were anesthetized with sodium pentobarbital (60 mg/kg) and mounted in a stereotaxic frame. A midline incision was made in the scalp, followed by a craniotomy (5-mm diameter) centered between lambda and bregma. A modified Luer-Lok cap was secured with dental cement into the craniotomy, over the intact dura. Lateral FP injury of the left hemisphere was induced, as initially described (McIntosh et al., 1989). Briefly, a calibrated saline-filled device was attached to the rat by the Luer-Lok cap, and a 25-millisecond pulse of saline was delivered under pressure to the intact dura to create a brain injury of moderate severity (study 1: 2.5 ± 0.1 atm, n = 45; study 2: 2.5 ± 0.1 atm, n = 27). Animals were monitored closely for respiratory distress during the first 15 minutes after injury and allowed to recover on a heating pad. Sham (uninjured) rats were subjected to anesthesia and surgery without brain injury (study 1, n = 16; study 2, n = 14). Control and experimental groups were behaviorally tested, injected, and killed at equivalent times.
Drug administration
Study 1. Both rhIL-1ra (Amgen, Inc., Thousand Oaks, CA, U.S.A.) and vehicle (lO mmol/L citrate, pH 6.5, 140 mmol/L NaC, 0.05 mol/L ethylenediamine tetraacetic acid) were stored at −20°C, then warmed to room temperature at the time of injection. In study I, brain-injured animals were randomly selected to simultaneously receive an intravenous bolus and subcutaneous injection of vehicle, low-dose rhIL-1ra (10 mg/kg), or high-dose rhIL-1ra (100 mg/kg) at 15 minutes after injury. Doses were based on previously published studies (Martin et al., 1994; Relton et al., 1996). This was followed by repeated subcutaneous injections at 4, 8, 12, 20, 24, 32, and 44 hours and every 12 hours until day 7 after injury. Sham animals received either vehicle or high-dose rhIL-1ra using an identical administration paradigm.
Study 2. Brain-injured and sham animals received a similar simultaneous intravenous and subcutaneous bolus of either vehicle or high-dose rhIL-1ra at 15 minutes after injury, followed by subcutaneous injections at 4, 8, 16, 20, 24, 28, and 40 hours. Although the exact timing of postinjury drug administration differed slightly between studies 1 and 2, the total dose of rhIL-1ra (900 mg/kg) and volume of vehicle (9 mL/kg) up to 48 hours after injury was identical in the two studies. Since repeated administration of high-dose rhIL-1ra between days 2 and 7 after injury in study 1 appeared to slightly retard the recovery of motor function, we decided to use the low dose (10 mg/kg/injection) between days 2 and 7 in study 2. However, in study 2, we also avoided the potential stress of the twice-daily injection schedule of study I by administering the low-dose rhIL-1ra and vehicle through a continuous infusion miniosmotic pump (Alza, Inc., Wilmington, DE, U.S.A.). To implant these pumps, animals were reanesthesized (sodium pentobarbital, 60 mg/kg intraperitoneally) at 48 hours after injury, and two Alzet pumps (1001D) were implanted subcutaneously above the haunches of the rat. On day 7 after injury, animals were reanesthetized (sodium pentobarbital, 60 mg/kg intraperitoneally), the pumps removed, and the contents analyzed to determine the amount of drug each animal received. The total dose of rhIL-1ra (100 mg/kg) and the total volume of vehicle (10 mL/kg) received by brain-injured animals between days 2 and 7 after injury in study 2 were identical to those received by their study 1 counterparts in the same postinjury period.
Physiologic monitoring
In a separate group of rats maintained under sodium pentobarbital anesthesia (n = 8 brain-injured, n = 3 shams), temporal muscle temperature, core body temperature, mean arterial blood pressure, and heart rate were monitored after brain injury/sham surgery and intravenous treatment with rhIL-1ra (100 mg/kg, n = 4) or vehicle (n = 4). Parameters were measured for 15 minutes before FP brain injury for baseline measurements, then every 15 minutes for 2 hours after injury and drug/vehicle administration. Core body temperature was recorded by insertion of a plastic coated thermocouple into the rectum, whereas temporal muscle temperature was monitored through placement of a temperature probe between the skull and the temporal muscle. Mean arterial blood pressure and heart rate were recorded with a Cardiomax II (Columbus Instruments, Columbus, OH, U.S.A.) through a catheter placed in the femoral artery.
Behavioral testing: motor function
Animals in study 1 were evaluated for neurologic motor function at 2 and 7 days after injury, whereas animals in study 2 were tested for motor function using identical tests at 2, 7, and 14 days after brain injury. Motor function tests were conducted by a trained investigator who was blinded to the treatment regimen and injury status of the animals and consisted of forelimb flexion, hindlimb flexion, lateral pulsion, and angle board tests. A score from 0 to 4 was given on the performance for each task (where 0 = afunctional and 4 = normal, uninjured performance). The forelimb flexion test consisted of rapid introduction of a flat surface to the rat when suspended by the tail where the position of each of the forelimbs is recorded. The hindlimb flexion test examined the backward extension of each of the hindlimbs when the animal's tail was lifted and the forelimbs remained on a hard surface. Lateral pulsion evaluated the animal's ability to resist hand pressure when moved in either direction. The angle board test evaluated the rat's ability to stand on an inclined plane in a right, left, and vertical position compared with a baseline performance (4 = above or equal to baseline; 3 = 2.5° below baseline; 2 = 5° below baseline; 1 = 7.5° below baseline; 0 = 10° or more below baseline). The angle board scores were averaged across the three positions and added to those from other tests to achieve a maximum composite score for motor function of 28 points.
Behavioral testing: cognitive function
Performance of cognitive function was evaluated in study 2 using a visuospatial memory Morris water maze paradigm described previously (Smith et al., 1991). One day before and on the day of injury, all animals were pretrained in the Morris water maze, a 1-m diameter tank filled with water at 18°C. Animals were trained over a series of 20 trials (10 trials/day) to find a submerged platform after being placed at several different entry points in the Morris water maze, using visual cues located outside the maze. Latency from the starting point to the hidden platform was recorded for each trial to determine the learning ability of each rat. At 42 hours after sham treatment (n = 14) or FP injury and treatment with either rhIL-1ra (n = 13) or vehicle (n = 14), the platform was removed from the maze, and animals were evaluated for their ability to recall the platform location. The swim patterns were video recorded, and a computer-generated numerical score was calculated where more weight was assigned for time spent in regions closest to the former platform location (Smith et al., 1991). The swim distance and average swim speeds also were calculated for each animal.
Histologic evaluation
Animals were overanesthetized with sodium pentobarbital (200 mg/kg) and intracardially perfused with a solution of 100 mL of heparin/saline followed by approximately 300 mL of 10% buffered formalin (Sigma, St. Louis, MO, U.S.A.). Animals from study 1 (n = 15, vehicle treated; n = 16, 10 mg/kg rhIL-1ra treated; n = 14, 100 mg/kg rhIL-1ra treated; n = 16, sham) were perfused at 8 days after injury. Brains were removed immediately and postfixed in formalin for 24 hours.
After fixation, brains were dehydrated, embedded in paraffin, cut coronally in 5-μm-thick sections, and stained with cresyl violet. Using a Leitz microscope, viable neuronal counts were performed by a trained observer blinded to the treatment status of the animals. Regional areas were measured using a 100- mm2 eyepiece corresponding to 0.25 mm on a linear calibrated scale using a 20× objective. Cell counts were expressed as the number of cells per given area. Brain regions were chosen in accordance with previous data indicating specific areas of cell loss in the traumatically injured brain (Hicks et al., 1996). Neurons were defined based on the presence of an intensely stained nucleolus, unstained nucleus, and the presence of a cell body. Neuronal counts in the following 10 brain regions were obtained: two areas of the parietal cortex (areas 1 and 2, corresponding to the site of maximal injury and the periinjury “penumbra” region, respectively, at −3.3 mm bregma), temporal cortex, retrosplenial agranular cortex, CA3 region of the hippocampus, hilus of the hippocampal dentate gyrus and medial geniculate nucleus (all at −4.8 mm bregma), lateral septal nucleus (at 0.2 mm anterior to bregma), ventral posterolateral thalamic nucleus (at −3.3 mm bregma), and substantia nigra (at −6.04 mm bregma).
Statistical analysis
All neuroscore data were statistically examined using Kruskal-Wallis analysis of variance (ANOVA) followed by Mann-Whitney U tests for comparisons between groups. Analysis of memory data was performed using ANOVA and post hoc Bonferroni tests. Analysis of histologic data was performed using one-way ANOVA followed by the Newman-Keuls test for comparison between groups. Analysis of physiologic (temperature, blood pressure) data was performed using a one-way ANOY A followed by post hoc Bonferroni tests.
RESULTS
Study 1
Neurologic motor function. In study 1, the composite neuroscores of sham (uninjured) rats treated with rhIL-1ra (high dose) were not statistically different from uninjured animals treated with vehicle at any time point evaluated. Compared with uninjured animals, vehicle-treated, brain-injured animals exhibited significant motor deficits at 48 hours (P < 0.05, Fig. 1A) and 1 week (P < 0.05, Fig. 1B) after injury, although some recovery was noted in the brain-injured animals over time. Treatment of brain-injured animals with low-dose rhIL-1ra (10 mg/kg/dose) resulted in a slight, but nonsignificant improvement in motor function at 48 hours and 1 week compared with vehicle treatment (Figs. 1A and 1B). Motor function in brain-injured rats treated with high-dose rhIL-1ra (100 mg/kg/dose) was not different at 48 hours (Fig. 1A), but the recovery noted in vehicle-treated brain-injured animals was not observed at 1 week in brain-injured animals treated with high-dose rhIL-1ra (Fig. 1B).
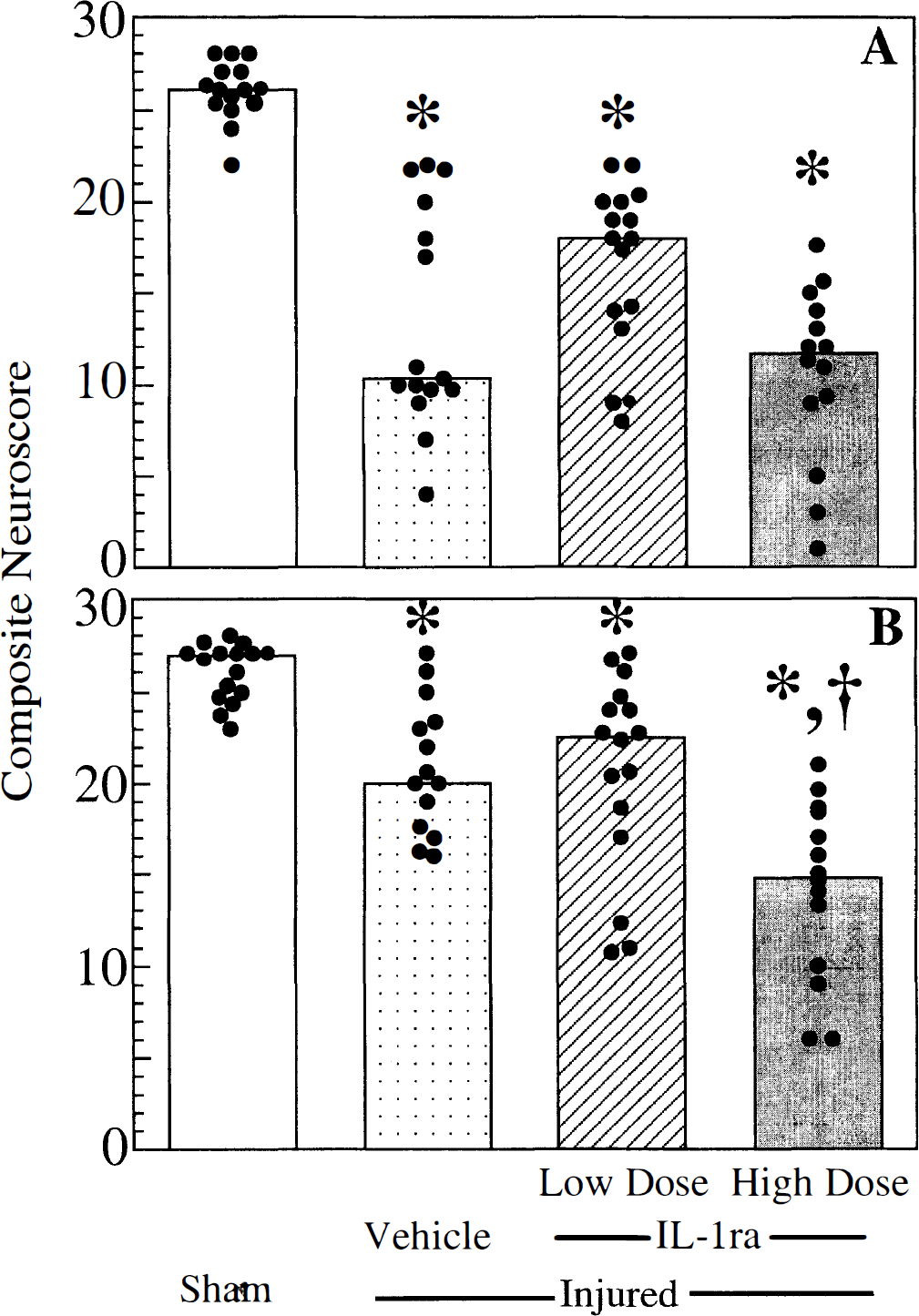
Composite neuroscore examination at 48 hours
Study 2
Cognitive function. Lateral FP brain injury induced significant cognitive deficits at 42 hours in vehicle-treated animals compared with uninjured animals treated with either vehicle or rhIL-1ra (P < 0.05, Fig. 2). Postinjury administration of rhIL-1ra significantly improved the cognitive ability of brain-injured animals compared with vehicle-treated injured animals (P < 0.05). Notably, the spatial memory scores of brain-injured animals receiving rhIL-1ra were not significantly different from sham-injured animals.
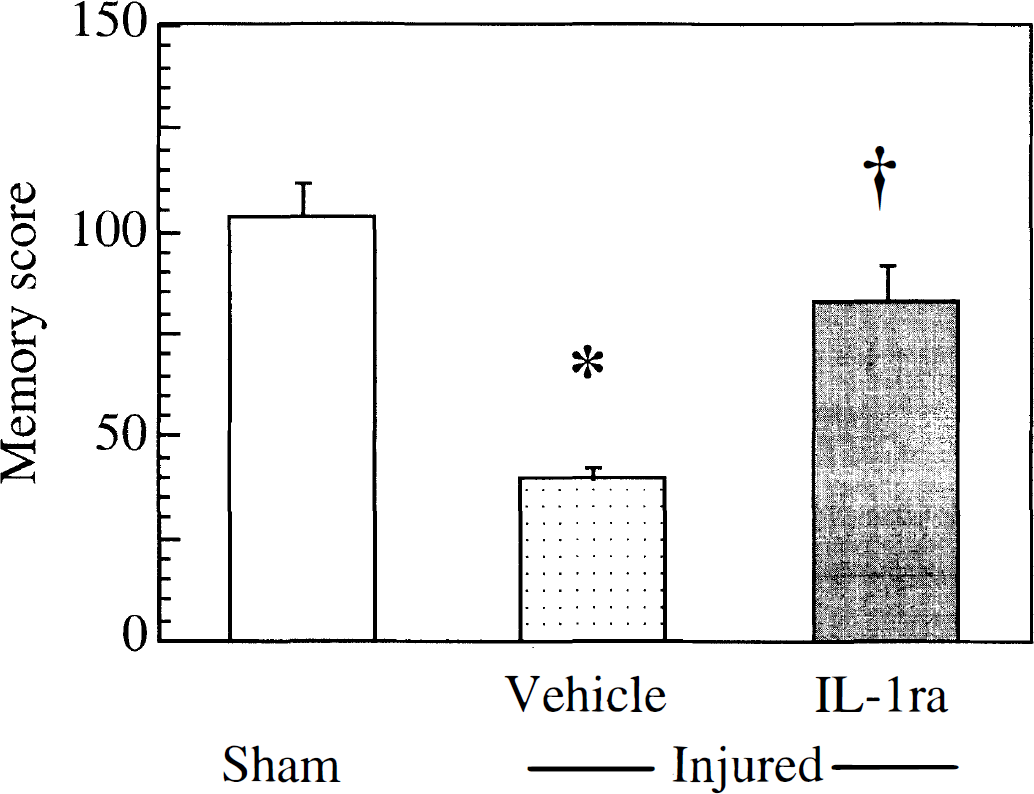
Memory score of animals tested in the Morris water maze at 42 hours in study 2. Graph indicates mean ± SD of the data. *, a significant difference in mean from sham (uninjured), animals; †, a significant difference from injured, vehicle-treated animals (P < 0.05).
Neurologic motor function. Neurologic motor function of sham-injured rats treated with rhIL-1ra (900 mg/kg) was not statistically different from that of sham-injured animals treated with vehicle at any time point evaluated. Lateral FP brain injury resulted in significant motor deficits at 48 hours (P < 0.05, Fig. 3A), 1 week (P < 0.05, Fig. 3B), and 2 weeks (P < 0.05, data not shown) in both vehicle- and rhIL-1ra—treated rats. Treatment of brain-injured rats with rhIL-1ra had no beneficial or detrimental effect on posttraumatic motor function over the 2-week evaluation period.
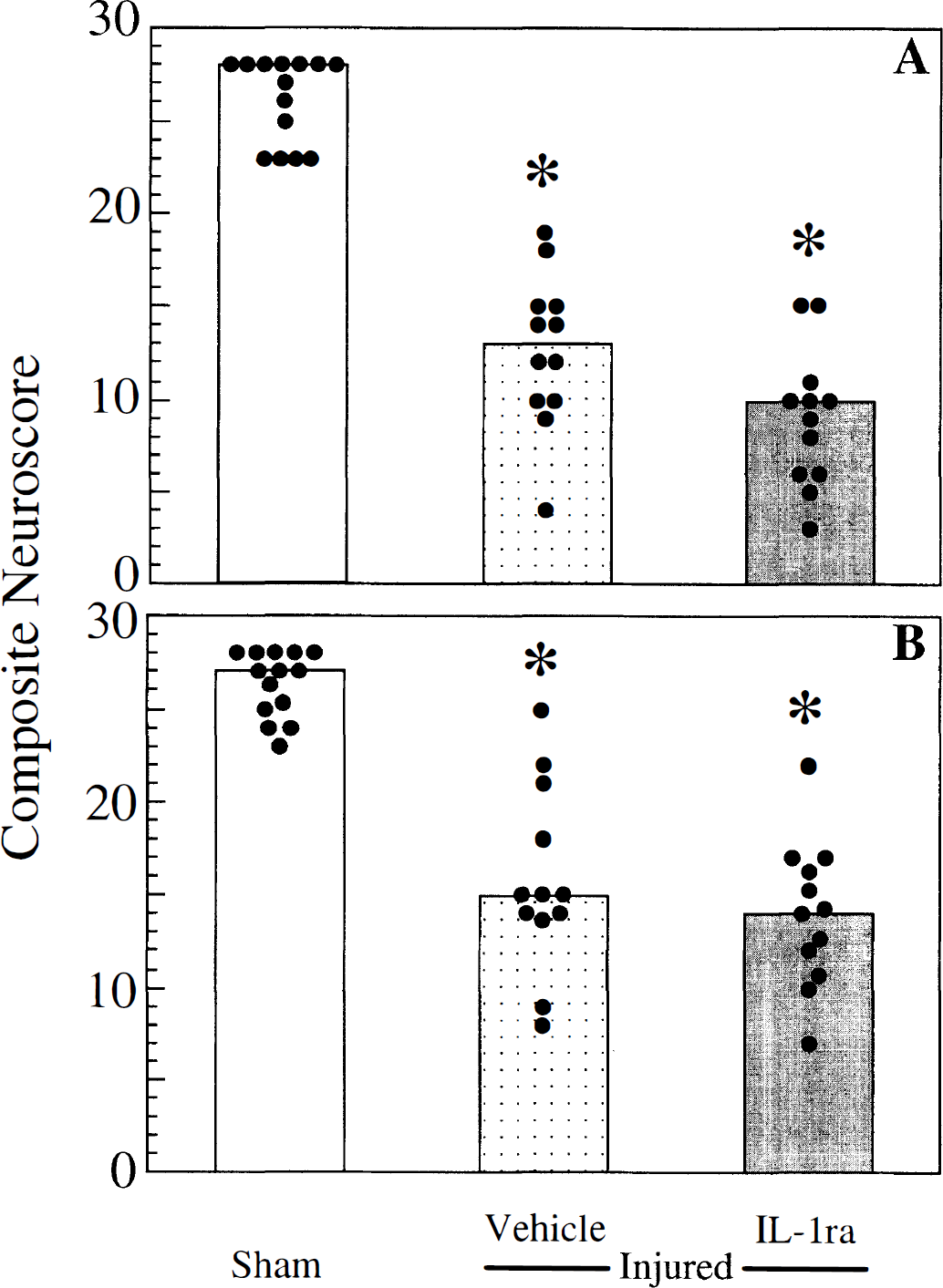
Composite neuroscore at 48 hours
Histologic analysis. Quantitation of neuronal cell loss in the injured cortex demonstrated that high-dose rhIL-1ra was effective in attenuating neuronal loss in area 2 of the parietal cortex (Fig. 4A) and the temporal cortex (Fig. 4B) (P < 0.05 compared with injured, vehicle-treated rats for both regions). Although lateral FP brain injury resulted in neuronal loss in area 1 of the ipsilateral parietal cortex and the medial geniculate nucleus and ventroposterior lateral thalamus of the ipsilateral thalamus, neither low- nor high-dose rhIL-1ra was neuroprotective in these regions (Table 1). Neuronal loss was not observed in the ipsilateral lateral septal nucleus, substantia nigra, or retrosplenial agranular cortex after brain injury (Table 1). Treatment with high-dose rhIL-1ra after brain injury did not exacerbate neuronal loss in any region examined.
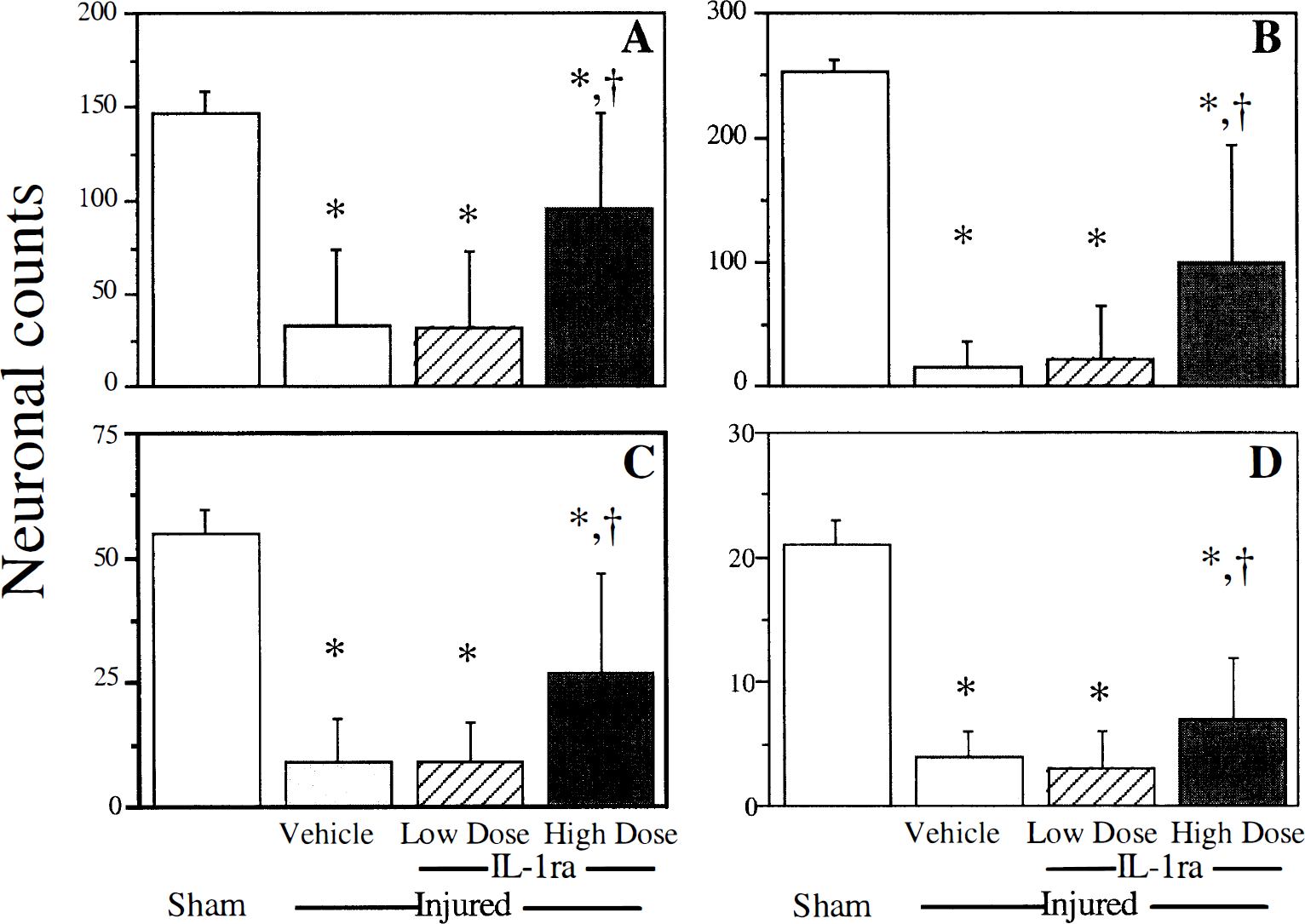
Quantitative neuronal counts in area 2 of the injured parietal cortex
Regional neuronal counts at one week after lateral fluid-percussion brain injury
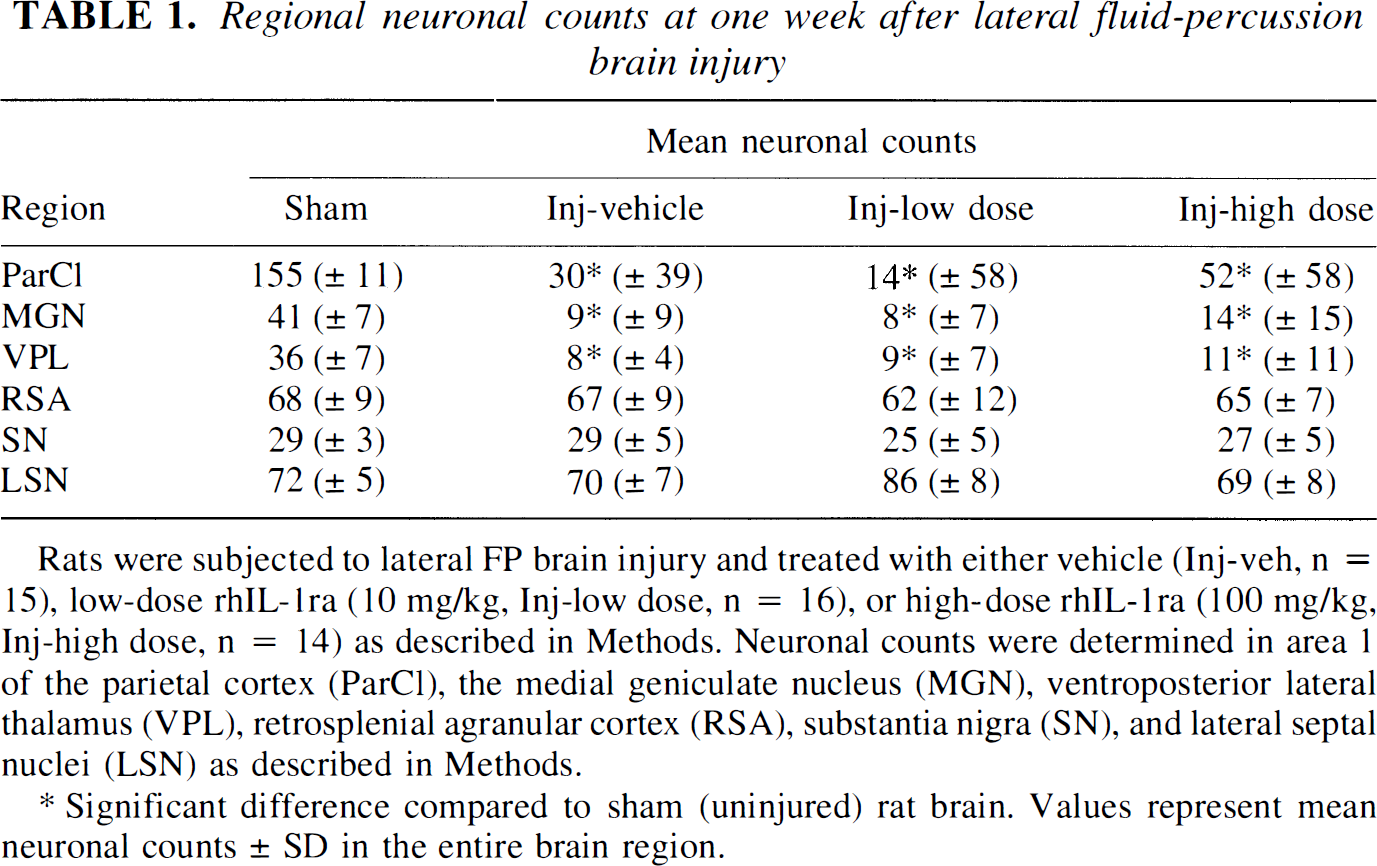
Rats were subjected to lateral FP brain injury and treated with either vehicle (Inj-veh, n = 15), low-dose rhIL-1ra (10 mg/kg, Inj-low dose, n = 16), or high-dose rhIL-1ra (100 mg/kg, Inj-high dose, n = 14) as described in Methods. Neuronal counts were determined in area 1 of the parietal cortex (ParCl), the medial geniculate nucleus (MGN), ventroposterior lateral thalamus (VPL), retrosplenial agranular cortex (RSA), substantia nigra (SN), and lateral septal nuclei (LSN) as described in Methods.
Significant difference compared to sham (uninjured) rat brain. Values represent mean neuronal counts ± SD in the entire brain region.
Compared with uninjured rats, brain-injured rats treated with vehicle demonstrated marked neuronal loss in area CA3 (Fig. 4C) and the dentate hilus (Fig. 4D) of the ipsilateral hippocampus at 1 week after injury. Quantitative analysis (manual cell counting) revealed significant sparing of both CA3 and dentate hilar neurons in brain-injured rats treated with high-dose rhIL-1ra (P < 0.05 compared with brain-injured, vehicle-treated rats) but not in those treated with low-dose rhTL-1ra (Figs. 4C and 4D).
Drug and physiologic monitoring. Analysis of the contents of the osmotic pumps from study 2 after removal from the animals revealed concentrations of drug remaining in each pump. Animals treated with rhIL-1ra received an average of 45.58 ± 4.14 mg of rhTL-1ra over a 5-day period. Concentrations of rhTL-1ra delivered to sham (uninjured) animals (22.93 mg/kg/day) were identical to those administered to injured animals (22.73 mg/kg/day). Lateral FP brain injury did not affect mean arterial pressure, heart rate, or core body and brain temperature over the 2-hour postinjury study period (Table 2). No differences were observed in temporal muscle temperature, core body temperature, mean arterial pressure, or heart rate of brain-injured animals treated with 100 mg/kg rhIL-1ra or vehicle (Table 2).
Physiological parameters after lateral fluid-percussion brain injury
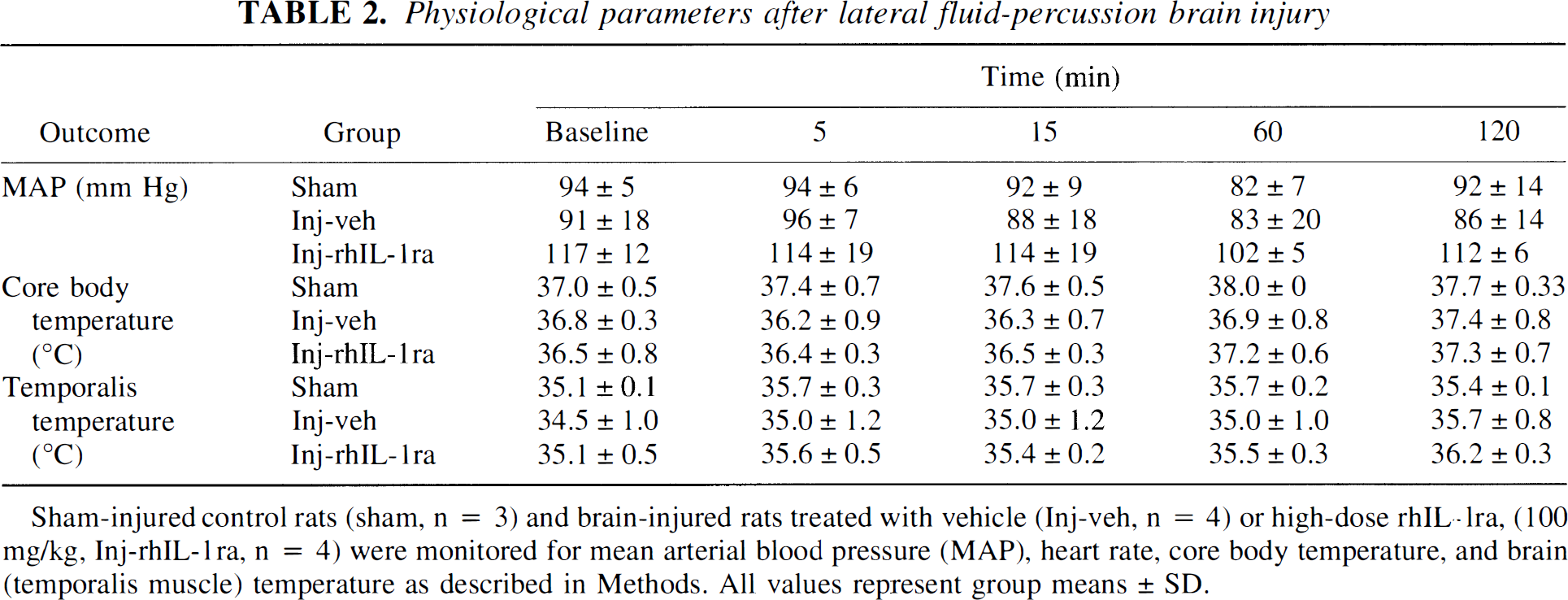
Sham-injured control rats (sham, n = 3) and brain-injured rats treated with vehicle (Inj-veh, n = 4) or high-dose rhIL-1ra, (100 mg/g, Inj-rhIL-1ra, n = 4) were monitored for mean arterial blood pressure (MAP), heart rate, core body temperature, and brain (temporalis muscle) temperature as described in Methods. All values represent group means ± SD.
DISCUSSION
This study demonstrates that systemic postinjury administration of high-dose rhIL-1ra can significantly attenuate cognitive dysfunction and regional neuronal loss associated with experimental brain trauma in rats. The improvement in memory function observed in this study may be related to the ability of rhIL-1ra to inhibit neuronal damage in brain regions known to be essential for the retention of a spatial memory task in the rat (Smith et al., 1991). Cognitive deficits after experimental brain injury have been associated with neuronal loss in the hippocampus, and treatments that prevent cell loss in the CA3 pyramidal layer of the hippocampus may concurrently attenuate spatial memory dysfunction (Hicks et al., 1994; Smith et al., 1993).
Trauma-induced memory deficits have been shown to correlate with the loss of hilar neurons in rats (Hicks et al., 1993), and this is the first report of an attenuation of trauma-induced hippocampal hilar cell loss by a pharmacologic agent. Hilar cells of the dentate gyrus are selectively vulnerable in models of TBI and are thought to be intimately involved in memory function (Hicks et al., 1993; Lowenstein et al., 1992). The ability of IL-1β to inhibit long-term potentiation in vitro (Bellinger et al., 1993) has been related to its ability to induce the generation of reactive oxygen species, which are known to inhibit long -term potentiation (Pellmar et al., 1991) and to be expressed after TEI (Shohami et al., 1997). Murray and Lynch (1998) have observed a relation between the upregulation of IL-1β in the dentate gyrus and impaired long-term potentiation, which was associated with the induction of reactive oxygen species. The neuroprotective effects in the dentate gyrus of rhIL-1ra in the current study may, therefore, be related to the cognitive improvement observed in the rhIL-1ra-treated animals. Although the protection of cells against trauma-induced injury demonstrates the therapeutic potential of rhIL-1ra treatment, the relation between posttraumatic behavioral dysfunction and neuronal cell loss is not straightforward. Treatment with trophic factors such as NGF or moderate hypothermia have been previously shown to be efficacious in attenuating cognitive deficits induced by TBI without any apparent protective effects on neurons in the injured hippocampus (Bramlett et al., 1997; Sinson et al., 1995). The current study emphasizes the importance of incorporating multiple behavioral and histologic measures to conduct a thorough evaluation of pharmacologic efficacy of novel experimental compounds.
In study 1, vehicle-treated brain-injured animals recovered somewhat with respect to the neurologic motor scores during the first week after injury. Although brain-injured animals treated with high-dose rhIL-1ra did not demonstrate this improvement, we do not believe that these data imply that this dose of rhIL-1ra had a detrimental effect on motor function. In study 2, the magnitude of the recovery of vehicle-treated animals was not as great as in study 1, and the comparison between rhIL-1ra-treated and vehicle-treated animals showed no difference in motor function scores. Previous research has shown that rhIL-1ra improves motor function after middle cerebral artery occlusion (Garcia et al., 1995). The composite scores for motor function obtained in study 2 at 7 days after injury in rhIL-1ra—treated animals were virtually identical with the scores of rhIL-1ra—treated animals in study 1, we believe that these data indicate no beneficial or deleterious effect of rhIL-1ra on posttraumatic motor function.
Our histologic observations regarding the attenuation of neuronal cell loss in vulnerable areas of the cortex support those of Toulmond and Rothwell (1995), who previously reported that central (intracerebroventricular) administration of rhIL-1ra significantly reduces cortical neuronal cell loss in the brain after FP TBI. Administration of rhIL-1ra also has been shown to be neuroprotective after ischemic and excitotoxic damage (Betz et al., 1995; Garcia et al., 1995; Loddick and Rothwell, 1996; Relton et al., 1996), and attenuation of the ischemic inflammatory response has been reported in mouse brain using an adenoviral vector to induce overexpression of rhIL-1ra (Yang et al., 1995). In addition to prevention of cortical cell loss, our observations indicate that rhIL-1ra is efficacious in attenuating both cognitive dysfunction and the marked hippocampal cell loss associated with TBI.
Since the amount of rhIL-1ra administered daily to the injured animals represents a potentially large colloid protein load (almost 2 g/kg over 1 week), this colloid load may have had hemodynamic or local effects on edema (which were not assessed in the current study). Early studies by Albright et al. (1984) suggest the possibility of important effects of colloid administration after brain injury, whereas more recently, Belayev et al. (1998) demonstrated a beneficial effect of albumin (3 g/kg) on functional and histologic outcome after lateral FP brain injury in rats. Although no significant hemodynamic changes were observed during the first few hours after brain injury and rhIL-1ra administration, hemodynamic effects may have been observed with a larger number of animals or at later postinjury time points. However, the short plasma and tissue half-life of rhIL-1ra (approximately 30 minutes) suggests that prolonged hemodynamic effects would be unlikely.
The mechanisms underlying the apparent neurotoxic effects of IL-1 are largely unknown. Inhibition of N-methyl-
In contrast to its putative role as a pathologic mediator of cell damage and death after CNS injury, IL-1 also may have protective effects in the setting of CNS injury. Upregulation of IL-1β in astrocytes in fetal mice subjected to brain lesions has been associated with a protective reconstruction of a glial limitans, which may be involved with healing and repair (Scripter et al., 1997). Interleukin-1β also has been shown to increase levels of NGF twofold to fourfold in brain regions known to have numerous IL-1β receptors, including the cerebral cortex and hippocampus (Carman-Krzan et al., 1991). Transplantation of fibroblasts transfected with a adenoviral vector expressing rhIL-1ra into traumatically injured cortex was found to decrease the upregulation of NGF after cortical stab wound– and weight drop—induced trauma (DeKosky et al., 1996). Since NGF may play a protective role in the traumatically injured brain, the trophic and potentially beneficial actions of endogenous NGF could be compromised after administration of rhIL-1ra. These data suggest that although acute treatment of rhIL-1ra has beneficial effects, chronic administration of this drug must be considered with caution because of the potential for prolonged inhibition of NGF in the CNS. Our study is the first to reveal comprehensive behavioral and histologic effects of systemic administration of rhIL-1ra in a rodent model of head injury. In view of the apparent neuroprotective effects of rhIL-1ra, this drug may have therapeutic potential in treatment of clinical brain injury.
Footnotes
Acknowledgments
The authors thank Seamus Ferandez, Kelli McDermott, and Joshua Leitner for invaluable technical assistance in surgical preparations and behavioral testing, Jeanne Marks for preparation of the manuscript, and Desireé Davis for housekeeping.
