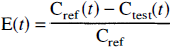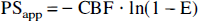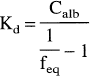Abstract
The calculated fraction of receptor ligands available for blood–brain barrier passage in vivo (favail) may differ from in vitro (feq) measurements. This study evaluates the protein–ligand interaction for iomazenil and flumazenil in rats by comparing feq and favail. Repeated measurements of blood–brain barrier permeability for two benzodiazepine antagonists were performed in 44 rats by the double-indicator technique. Cerebral blood flow was measured by intracarotid Xe-injection. The apparent permeability–surface product (PSapp) was measured while CBF or bolus composition was changed. Comparison of PSapp obtained in the absence and presence of 5% albumin in the injectate yielded favail, whereas feq was measured by equilibrium dialysis. Iomazenil and flumazenil favail was 62% and 82%, respectively, whereas feq was significantly lower, 42% and 61%. The PSapp for iomazenil and flumazenil increased significantly by 89% and 161% after relative CBF increases of 259% and 201%, respectively. The results demonstrate that application of feq in neuroreceptor studies underestimates the plasma input function to the brain. Model simulations render possible that the differences between favail and feq as well as the effect of CBF on PSapp can be caused by capillary heterogeneity.
In the last decade, radiopharmaceuticals applicable for in vivo emission tomography for the study of neurotransmitter systems have been developed. For quantification of receptor characteristics in the living human brain, different models have been used. Many ligands are bound to plasma proteins, but only the unbound ligand dissolved in plasma water is available for brain uptake. Therefore, the binding interaction between ligand and plasma protein in the brain capillary is critical for ligand uptake across the blood–brain barrier (BBB). The free fraction of the ligand, as measured by equilibrium dialysis or ultrafiltration (feq), has previously been assumed to represent the fraction available for BBB transport. However, it remains to be established whether the free fraction of ligands as measured in vitro corresponds to in vivo determinations. It has been shown in in vivo studies that the brain uptake of plasma protein–bound substances (Jones et al., 1988; Pardridge and Fierer, 1990) is higher than what could be expected from the in vitro measured feq. Similar findings have been reported in studies of the liver (Forker et al., 1985; Keiding, 1993) and of isolated adipocytes and cardiac myocytes (Sorrentino et al., 1989). This has been interpreted as an accelerated dissociation of ligand from the plasma protein in the capillary bed (Pardridge and Fierer, 1990). Notice that even if the ligand–protein dissociation is instantaneous and the brain uptake extremely high, the difference between in vitro and in vivo studies is not explained, since the plasma water ligand concentration will never exceed that of the equilibrium concentration. If, on the other hand, the dissociation of the plasma protein–ligand complex is so slow that this process becomes the rate-limiting step for brain uptake, a nonequilibrium condition may arise in the capillary (Robinson and Rapoport, 1986; Weisiger, 1985). In this condition, the mean capillary transit time (or CBF as a determinant of this) may play a role, whereas the difference between in vitro and in vivo studies remain unexplained. By contrast, the brain uptake then would become smaller than predicted, since the plasma water ligand concentration would drop below the equilibrium concentration.
Previous models addressing this problem (Pardridge and Fierer, 1990; Robinson and Rapoport, 1986; Weisiger, 1985; Weisiger et al., 1991), however, all assume that brain capillaries are homogeneous and arranged functionally in parallel so that they share the same inlet and outlet concentrations. As is shown later, violation of this assumption, here termed capillary heterogeneity, invalidates predictions from the Bohr-Kety-Renkin-Crone equation (Bass, 1983). This study addresses the relevance of the in vitro–determined free fraction of ligands for the benzodiazepine receptor system as well as the brain uptake dependency of changes in the capillary transit time. We determined the apparent in vivo fraction available for BBB transport (favail) in rats for two benzodiazepine receptor ligands, [125I]iomazenil and [3H]flumazenil, by comparing brain uptake in the presence and absence of 5% albumin. The influence of changes in mean capillary transit time on cerebral ligand uptake was evaluated by inducing changes in CBF.
METHODS
Materials and surgery
Forty-four male Wistar rats with a body weight between 320 and 370 g were studied. The experiments were approved by the local authorities. Anesthesia was initiated by 4% halothane. After tracheostomy had been performed, anesthesia was maintained with 0.7% halothane in a gas mixture of 70% N2 and 30% O2, The animals were artificially ventilated by a Harvard Miniature animal ventilator (Edenbridge, KY, U.S.A.) and were immobilized by intravenous injections of 40 mg/kg succinyl choline every hour. An adequate level of anesthesia was monitored continuously throughout the study by blood pressure and heart rate measurements that remained stable also under noxious stimuli. Both femoral arteries were cannulated for recording of blood pressure, heart rate and blood gases (pH, P
Skin and muscles of the scalp were deflected. On the right side of the neck, the carotid bifurcation was exposed, and the external carotid artery was ligated. All major extracerebral branches from the carotid artery, including the pterygopalatine artery, were ligated. These extensive ligations ensure that blood from the extracranial tissues draining into the sagittal sinus does not contribute to the outflow curves (Hertz and Bolwig, 1976). A polyethylene catheter was placed in the external carotid artery with the tip at the carotid bifurcation so that flow obstruction of the right internal carotid artery was avoided. The animals were heparinized by 1000 IE intravenously, and all wounds were infiltrated with lidocaine 1%. After 30 minutes' rest, a hole was drilled in the skull, and a blunt sawn-off 18-gauge needle was placed in the confluence sinus for cerebral venous blood sampling. Blood gases were analyzed by means of an ABL 30 (Radiometer, Copenhagen, Denmark).
Measurement of cerebral blood flow
Cerebral blood flow was measured twice to establish baseline values. Subsequently, CBF measurements were performed at least once every hour or at P
Blood–Brain barrier measurements
Between 4 and 10 BBB measurements were performed in each animal, and at least once it was checked that significant extracranial contamination did not take place. In extracranial tissues, [36Cl] and [24Na] have an extraction of about 50%, whereas the extraction in brain tissue is only 1% to 2% (Hertz and Bolwig, 1976). The macromolecules [3H]dextran (Amersham, UK) and [99mTc]-DTPA (Diethylenetriamine pentaacetic acid, Isotope Pharmacy, Copenhagen, Denmark) have negligible extractions during a single capillary passage, both in extracranial tissues and in the brain. Possible extracerebral contamination was evaluated in this study by comparing the extraction of [36CI] or [24Na] relative to [3H]dextran or [99mTc]-DTPA (Hertz and Bolwig, 1976) with BBB permeability studies.
To determine BBB permeability, a 20-μL bolus containing a BBB-impermeable reference substance, either [36Cl] (Amersham, UK) or [24Na] (Risø, Roskilde, Denmark), and a test substance ([125I]iomazenil (Paul Sherrer Institute, Villigen, Switzerland) or [3H]flumazenil (Dupont, NEN Research Products, Boston, MA, U.S.A.), was rapidly injected into the carotid artery just after blood sampling from the cerebral venous sinus was started. Twelve cerebral venous blood samples, approximately one sample per second, were collected from the needle placed in the confluence sinus. The blood was allowed to drip into the vials so that suction from the venous sinus was avoided. For normalization of outflow curves, an aliquot of injectate was mixed with saline and analyzed with the blood samples. All samples were bleached by H2O2 to decrease quenching.
To evaluate bolus mixing with blood during the capillary passage, the size of the saline bolus was altered between 20 and 120 μL.
Since iomazenil and flumazenil are rapidly metabolized in plasma by esterase, either plasma esterase–inactivated (NaF-heparin) plasma or 5% albumin had to be used in the preparation of the injectate. Since the effect on the BBB of repeated NaF exposures is unknown, a 5% albumin solution was preferred. In eight animals it was, however, ensured that brain uptake was similar, whether a bolus with NaF-treated plasma or 5% albumin was used.
In BBB experiments where CBF changes had been induced, a 5% albumin bolus was used.
Counting the samples
Samples with [24Na] and [125I]iomazenil were counted in a γ-counter (Cobra 5002/5003, Canberra Packard, Meriden, CT, U.S.A.) with subsequent spillover and decay correction. When [36CI] was used as a reference, 10 mL of scintillation fluid (Ultima Gold, Packard Instrument, Groningen, Holland) was added to the vial, which then was counted in a γ-counter (Packard 1900 CA, Canberra Packard) with subsequent spillover and quench correction.
Calculation of blood–brain barrier permeability
The brain extraction at time t [E(t)] was calculated from
The permeability–surface product (PSapp) was calculated according to the Bohr-Kety-Renkin-Crone equation:
This equation was derived for a single cylindrical capillary within which a fluid of uniform composition flows at a constant velocity.
Calculation of favail
If it is assumed that the brain capillary bed is homogeneous and the ligand is not protein bound, PSapp (Eq. 2) equals PS defined as the sum of individual capillary PS values. For protein-bound ligands, PSapp equals PS · favail (Pardridge, 1983), thus
Measurement of feq
Measurements of feq were performed in vitro by equilibrium dialysis using a Kontron Diapack model 4000 with a cellulose membrane (Sigma, St. Louis, MO, U.S.A.), which retains proteins with a molecular weight larger than 12,000. The molecular weight of iomazenil and flumazenil is 409 and 305, respectively. To evaluate the effect of binding proteins other than albumin, [125I]iomazenil was added to NaF-heparinized plasma samples from eight Wistar male rats and to eight samples of 5% human albumin, whereas [3H]flumazenil was added to eight samples of human albumin. A mixture of plasma and 5% human albumin (1.5 mL) was dialyzed at 37°C for 4 hours when [125I]iomazenil was added and for 24 hours when [3H]flumazenil was added, against an equal volume of isotonic phosphate buffer (pH 7.4). Subsequently, 50 μL was taken from both compartments. Two milliliters of Pico-Flour (Packard Instrument) were added to vials with flumazenil and counted in a Hewlett Packard 1900 CA β-counter with subsequent quench correction. Vials containing iomazenil were counted in a Cobra 5002/5003 γ-counter. The feq was calculated as the cpm ratio between buffer and plasma.
Assuming tracer concentrations of the ligand, the dissociation constant, Kd, was estimated for both ligands according to Eq. 4, where Calb is the albumin concentration in the sample (725 μmol/L).
The calculated Kd values were used for the computer simulations as presented in the appendix.
Simulation models
To evaluate the potential influence of nonequilibrium conditions, unstirred water layer effects and capillary heterogeneity simulation models were applied, and the results were compared with the observed data. These simulations are described in the appendix.
Statistics
Results are presented as median values with quartiles in brackets. The Wilcoxon paired nonparametric test was used for comparison of CBF, extraction, and PSapp/feq. The Mann-Whitney test was used to compare the in vivo measured available fraction for BBB transport and in vitro measured free fractions. A significance level of 0.05 was chosen.
RESULTS
Iomazenil
There was no difference in feq for iomazenil as measured in plasma compared with measurements in 5% human albumin where feq was 42%.
An example of a venous outflow curve for [125I]iomazenil with its corresponding extraction curve is shown in Fig. 1 No difference between peak and the total extraction was found. Values for CBF, extraction, and PSapp for the paired observations are presented in Table 1. In the presence of 5% albumin, brain extraction and PSapp decreased significantly by 29% (20% to 39%) and 38% (27% to 51%), respectively, compared with the saline bolus. In hypercapnia, P
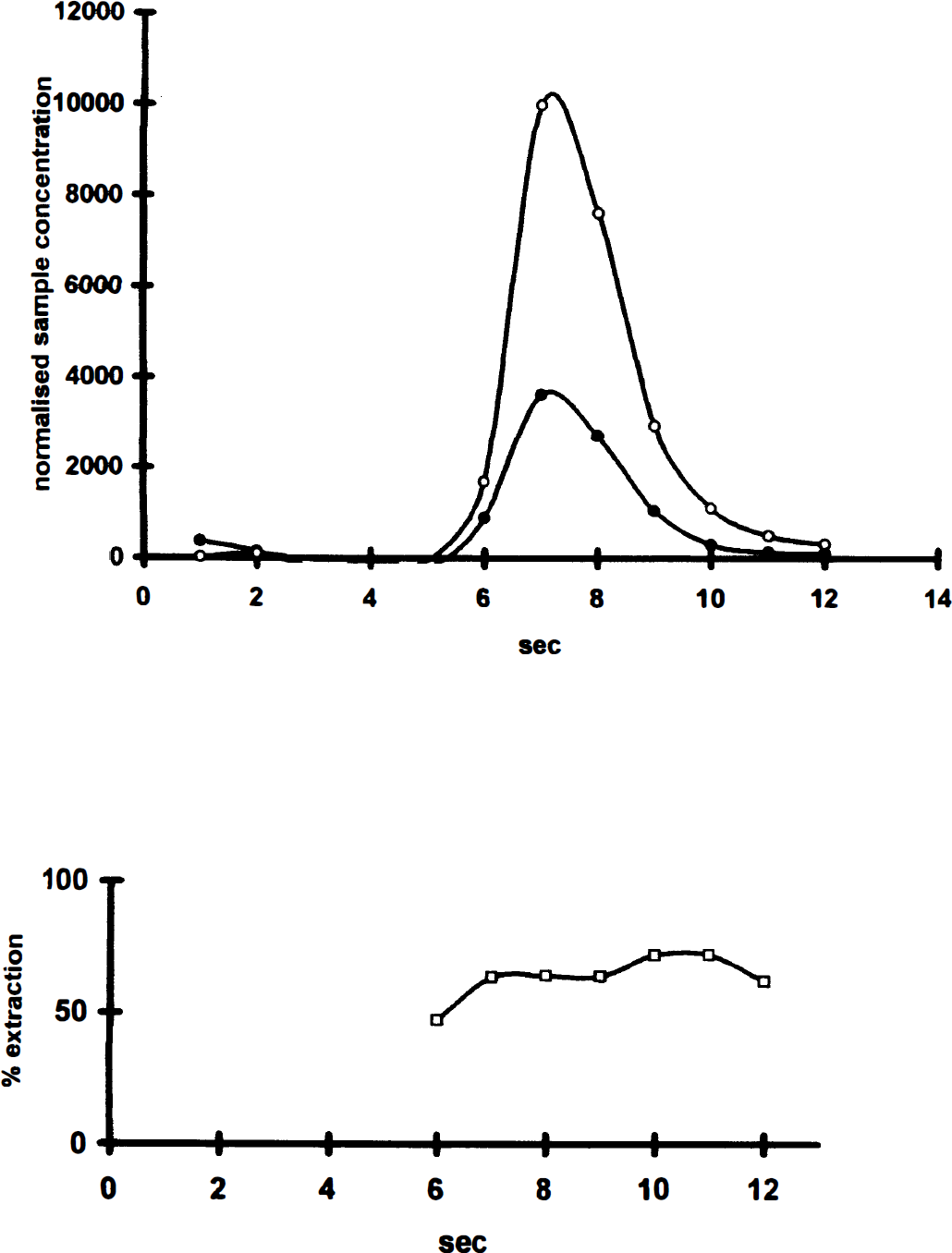
Example of venous outflow curve showing the time-activity curves for [125I]iomazenil (•) compared with the intravascular reference [36Cl] (○). The bottom panel shows the extraction in percentage of reference substance (□). Notice that even at late time points, back-diffusion seems negligible.
Values for CBF, extraction, and PSapp for [125I]iomazenil and [3H]flumazenil
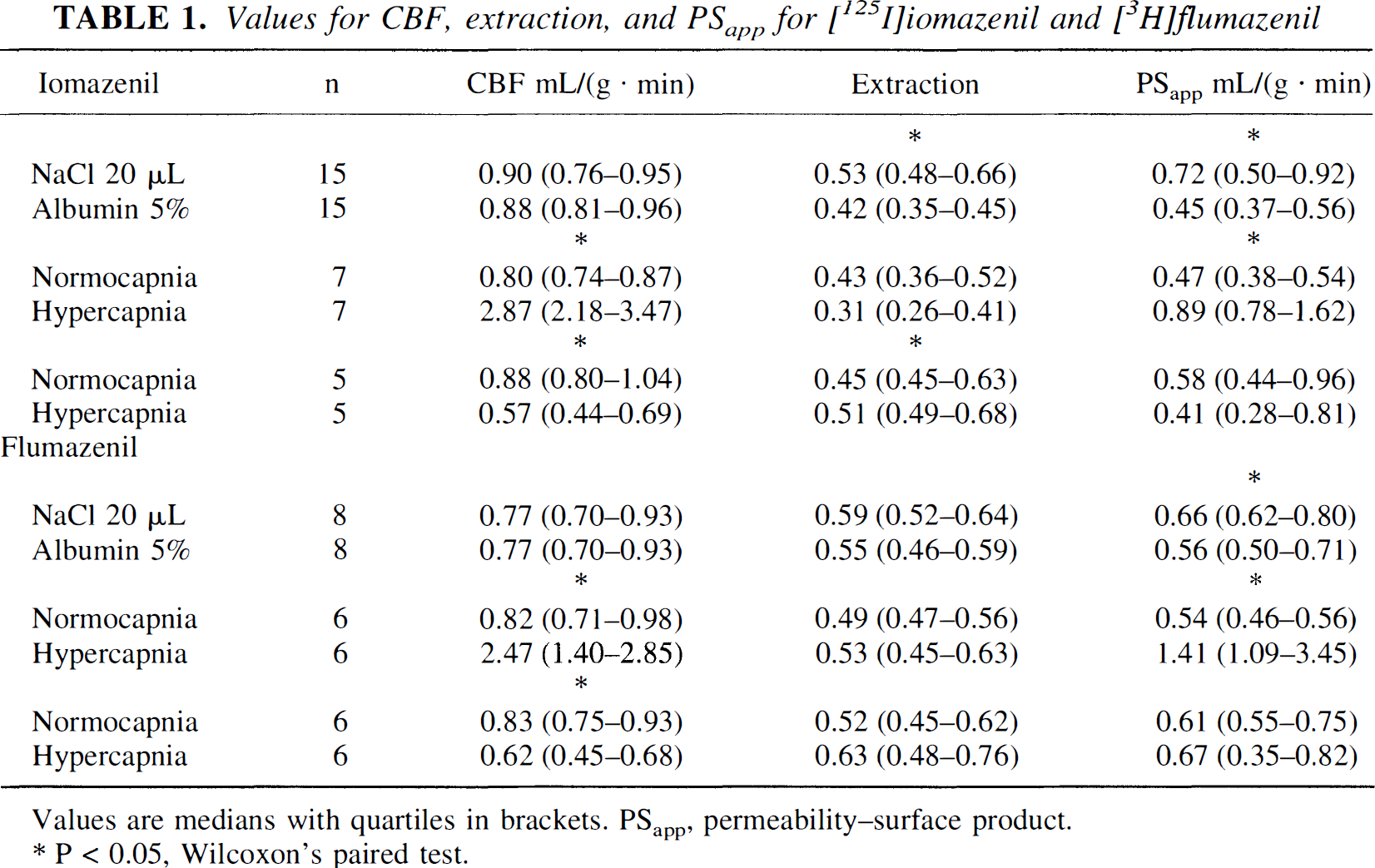
Values are medians with quartiles in brackets. PSapp, permeability–surface product. * P < 0.05, Wilcoxon's paired test.
Flumazenil
For flumazenil, feq was 61% in 5% human albumin. Brain extraction and PSapp were unaltered, whether a 20-or 120-μL bolus was used (n = 7, Mann-Whitney test). No difference between peak and the total extraction was found. Values for CBF, extraction, and PSapp are presented in Table 1. At the presence of 5% albumin in the bolus injectate, PSapp decreased significantly by 17% (11% to 22%) compared with a saline bolus, whereas the extraction did not change significantly. In hypercapnia, P
Protein binding
With saline bolus, extraction and PSapp were similar for the two ligands, whereas PSapp was significantly higher for flumazenil than for iomazenil when 5% albumin was present in the bolus.
For iomazenil, feq was 42% (40%, 44%) and favail was 62% (49%, 73%), which is a 32% larger favail than feq. For flumazenil, favail was 27% higher than feq. The feq was 61% (59%, 62%), and the favail was 84% (78%, 89%).
As evaluated from Eq. 4, Kd was 525 μmol/L for iomazenil and 1135 μmol/L for flumazenil.
Simulations
Table 2 shows the results of best fit simulations and the experimental observations. With the nonequilibrium model simulations, some of the observed effect of changing CBF could be explained when a plasma proteinligand dissociation constant (koff) of 0.05 seconds−1 was applied. For the heterogeneity model simulations, normal, log(normal), and exponential distributions of capillary lengths were applied to the data for iomazenil. All distributions produced effects in the observed direction (favail > feq, and PSapp increased with CBF), but only a declining exponential distribution provided a good accordance between simulated and observed results. When the same distribution then was applied to the flumazenil data, a good accordance with the observed data was found (Table 2).
Resulting PSapp from simulations with the binding disequilibrium model and the capillary heterogeneity model, as compared to measured PSapp values
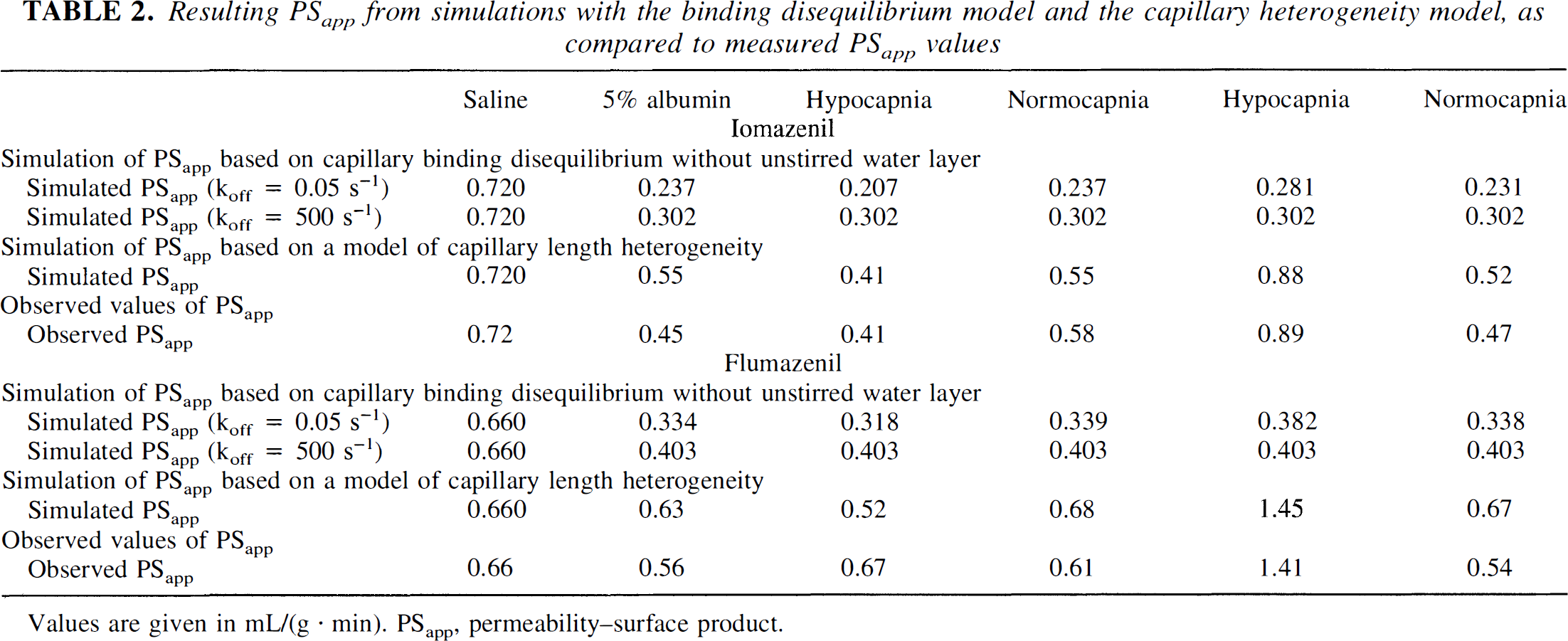
Values are given in mL/(g · min). PSapp, permeability–surface product.
DISCUSSION
Validation of cerebral blood flow-related changes
For both ligands, PSapp increased significantly with CBF (Fig. 2). This is in accordance with the study of Holthoff et al. (1991), where K1 for [11C]flumazenil increased with visual activation. Similar phenomena also have been observed previously for flow tracers (Andersen et al., 1988), glucose, and amino acids (Hertz and Paulson, 1982; Chen J-L et al., 1994).
The double-indicator technique yields global values for brain extraction, whereas the CBF technique measurements represents CBF values weighted toward gray matter, which theoretically would lead to a slight overestimation of PS. Since the rat brain contains relatively small amounts of white matter, this difference is less crucial. Further, in the calculation of favail, CBF cancels out. Theoretically, blood flow may increase during the intracarotid injection, but it has been demonstrated that bolus sizes up to 100 μL do not alter CBF (Hertz et al., 1984).
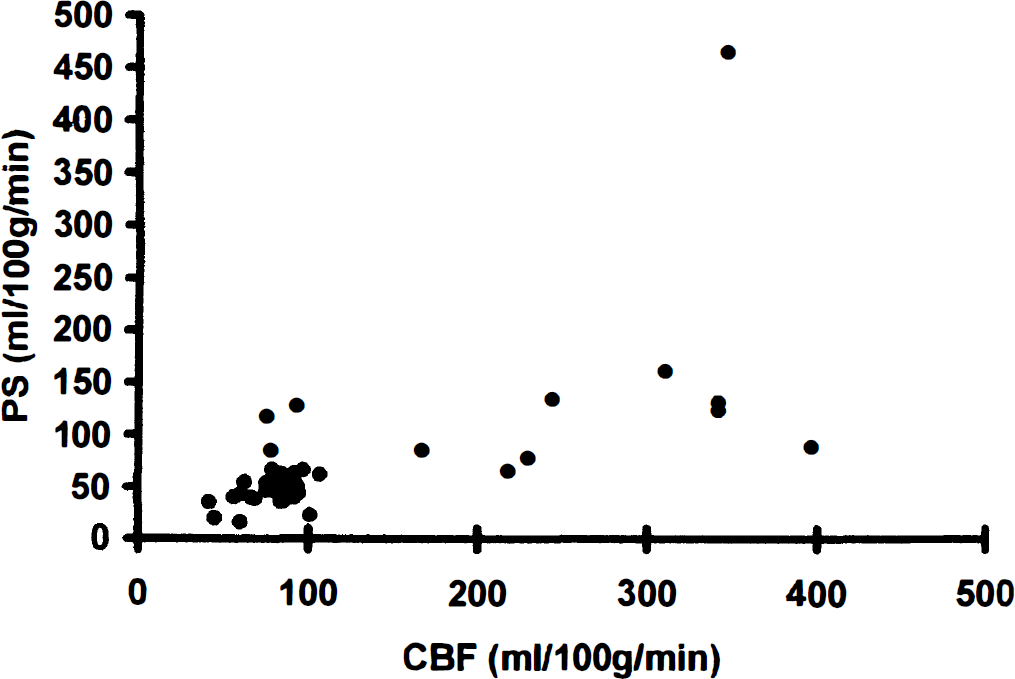
The apparent permeability surface product at various CBF values illustrated for [3H]flumazenil (•).
Most authors have interpreted the flow-dependent increase in brain uptake as capillary recruitment, that is, that the brain under certain circumstances is able to recruit unperfused capillaries to increase the surface area available for diffusion (Kuschinsky and Paulson, 1992). Less frequently, it has been considered that capillary heterogeneity may account for this phenomenon (Knudsen et al., 1991).
Validation of in vitro and in vivo protein–ligand interaction
The in vitro free fraction of iomazenil and flumazenil is comparable with previously published values of 29% to 46% (Gandelman et al., 1994) and 50% to 60% (Brogden and Goa, 1991), respectively.
The addition of albumin to the injectate is expected to reduce PSapp. Albumin addition, however, lead to a smaller decrease in PSapp than expected from Eq. 3. Theoretically, the difference between feq and favail may arise from blood contamination of the albumin-free bolus, since then PSprotein-absent would be underestimated (Eq. 3). The difference between feq and favail is, according to Eq. 4, only explained if protein contamination of the saline bolus increases plasma albumin concentration from zero to 247 μmol/L in the iomazenil study and 490 μmol/L in the flumazenil study. This seems highly unlikely, since with the brain uptake index technique, the upper bound for bolus mixing with blood has been estimated to be about 5% (Pardridge et al., 1985).
Our observation is in line with previous studies in the brain where the presence of albumin apparently enhances uptake of protein-bound substances such as tryptophan (Pardridge and Fierer, 1990), hormones, (Pardridge and Landaw, 1985) and drugs (Jones et al., 1988) by 4 to 25 times. Generally, the degree of enhancement increases with extraction and protein binding.
Explanations for unexpected behavior
To explain our findings (i.e., both the difference between feq and favail and the increase in PSapp with CBF), the underlying assumptions for the hypothesis that favail = feq needs to be examined. These are (1) only free ligand in the plasma water can be taken up from by the brain, (2) plasma protein–ligand equilibrium is present at any position along the capillary, (3) cross-sectional capillary concentration gradients are not present, and (4) the brain can be modeled as a set of identical capillaries (no capillary heterogeneity).
Violation of the first assumption often has been considered responsible when PSapp increased as albumin is added. Pardridge and Fierer (1990) interpreted it as being caused by in vitro and in vivo differences in the protein–ligand dissociation/association. Others have proposed that enhancement is caused by either the existence of an albumin receptor that facilitates ligand uptake from the bound state, by conformational changes of albumin near the endothelial cell surface, or by membrane near pH changes. The hypotheses of facilitation (Keiding et al., 1993) are conceptually similar, since they all assume facilitation of ligand uptake from the bound form. Since our changes in pH were minor and the transit time of albumin is short, facilitation seems unlikely,
Violation of assumption 2 can occur if the dissociation of ligand from plasma protein to plasma water compared with the capillary transit time is slow and the brain uptake is high (Robinson and Rapoport 1986, Weisiger et al., 1991). During the passage of the capillary, the brain uptake would decrease the concentration of free ligand in the capillary plasma water from the equilibrium free fraction. Because of the slow release of bound ligand into the plasma water, this decrease is not compensated. In the current study, we chose the approach of Weisiger et al. (1991), which provides an extension of the Renkin-Crone equation where protein–ligand dissociation is taken into account. Robinson and Rapoport (1986) also have elaborated a similar extension, but whereas their approach is confined to three limiting cases, Weisiger (1991) provides a more general solution.
With increasing CBF, the free ligand concentration would decrease less during the passage of the capillary. That is, binding disequilibrium may explain an increase in PSapp found with increasing CBF. As can be seen from Table 2, PSapp did change in the observed direction, but the model predicted much smaller changes than those observed. Thus, binding disequilibrium does not account completely for the observed PSapp–CBF relation.
Violation of assumption 3 could occur if an unstirred water layer close to the endothelial membrane limited the uptake (Weisiger et al., 1991). However, including an unstirred water layer in the model did not explain our data.
It is a violation of assumption 4 if the single capillary model does not apply for whole organs with a diversity of capillaries with different sizes, flow, and permeabilities (Hertz and Paulson, 1982; Knudsen et al., 1991). Capillary heterogeneity may arise when capillaries are different with respect to surface area (length or diameter), permeability, or flow. In our simulations of capillary heterogeneity, for simplicity, we chose to vary only one parameter, namely, the capillary length. It was found that a left skewed distribution with many short capillaries was able to produce both the observed differences between feq and favail as well as the CBF dependence for PSapp for iomazenil. Application of the same capillary length distribution for flumazenil also showed reasonable agreement with observed data. Thus, heterogeneity of capillaries was the only single, unified explanation for our finding of both the difference between favail and feq and the increase in PSapp with CBF.
Although mathematically simple, it may be difficult to envision how capillary heterogeneity is able to produce these effects. What we observe with the double-indicator method is the venous average of the individual capillaries' outlet. For simplicity, let us assume that the brain exists of two capillaries: one that does not take ligand up, and another with a certain permeability. Since the first capillary's contribution to the outlet is a fixed fraction, any changes inflicted on the system tend to be dampened. For example, in the permeable capillary, E changes in the opposite direction of CBF (Eq. 2). In the averaged outlet, an increase in CBF is not be sufficiently counteracted by a concomitant decrease in the extraction, since only the permeable capillary's extraction is changeable. Similarly, in the permeable capillary, the extraction decreases after addition of protein (Eq. 3), whereas the extraction in the other capillary remains zero, resulting in a smaller decrease in the extraction than predicted. Notice that we only varied one of the factors contributing to capillary heterogeneity, namely, the capillary lengths. Therefore, an anatomical correlate of the specific distribution is not needed. A less asymmetric distribution may account sufficiently for the observed data if the diameter of the capillaries, permeability, or flow rates also varies. Although firm evidence for capillary recruitment is missing (Kuschinsky and Paulson, 1992), this may theoretically explain the CBF effect on PSapp. Furthermore, contamination of the bolus injectate and nonequilibrium conditions also could play a role. It is impossible to quantitate the relative influence of each of these factors on the basis of our data.
We conclude that no other single explanation than capillary heterogeneity can account completely for both of the unpredicted results, and capillary heterogeneity warrants further consideration in this kind of study. Future studies should aim at elucidating the importance of capillary heterogeneity for substrates with different properties.
Implications for receptor studies
The separation of ligand–receptor binding effects from tracer delivery–flow effects is an important consideration in receptor studies with emission tomography. In the compartmental models frequently used for the analysis of receptor binding, the BBB transport constant K1 that also is dependent on CBF, capillary permeability, and protein binding is determined. Errors in the estimation of K1 will be propagated to the pharmacologic parameters for receptor binding. The arterial input function should be corrected for protein binding, but in the presence of facilitation, application of the in vitro–measured free fraction underestimates the true input function. Varying protein binding of radioligands may introduce interindividual variability in the estimates of ligand distribution volumes, as previously pointed out by Logan et al. (1994), who also noticed that the distribution volume of raclopride reduced as CBF decreases.
In tissue reference models, a reference region void of receptors is used to estimate nonspecific ligand binding, and the distribution volume of the target area is determined relative to the reference region. For this method, underestimation of favail does not hamper the measurement, although the CBF dependence of uptake potentially may lead to differences between the region of interest and the reference region. This consideration may be particularly relevant in receptor activation studies.
In neuroreceptor studies where the ligand is infused to obtain a steady-state condition, eventual changes in PSapp caused by regional CBF variations between the studies will cancel out. The free fraction of unlabeled ligand then may be calculated as the total blood concentration times feq if heterogeneity alone causes the observed difference between feq and favail. By contrast, if enhancement takes place, the free fraction is underestimated, whereby the affinity of the studied ligand is overestimated.
Footnotes
APPENDIX
Abbreviations used
Acknowledgments
The authors thank Dr. P. A. Shubiger and The Paul Sherrer Institute (Villigen, Switzerland) for providing [125I]-labeled iomazenil. The authors also thank technicians Kent Pedersen and Karin Stahr for their assistance, and Professor Ludvik Bass for helpful suggestions. The computer simulation program (ESPV5-1) was provided by Dr. R. A. Weisiger, Liver Center, University of California, San Francisco.