Abstract
Nitric oxide (NO) regulates basal CBF. In a number of animal models NO has been implicated in the mediation of the regional changes in CBF (rCBF) that accompany neuronal activation (vasoneuronal coupling). However, some results in animal models have failed to confirm this finding, and the validity of extrapolation to man from animal data is uncertain. To determine the contribution of NO to basal global CBF and activation-induced changes in rCBF, the authors have performed quantitative H215O positron emission tomography (PET) studies before and after administration of the non-isoform-specific NO synthase inhibitor, NG-monomethyl-
Vasoneuronal coupling describes the characteristic ability of the cerebral vasculature to match rapidly, and in a spatially restricted manner, regional CBF (rCBF) with neuronal activation. The mediator underlying this coupling process remains uncertain: it is not coupled to oxidative metabolism (Fox et al., 1984) and is not adequately explained by changes in levels of perivascular adenosine (Northington et al., 1992), tissue Po2 (Frahm et al., 1996) or pH (Leniger-Follert, 1984), potassium shifts, or perivascular neurotransmitter release (Iadecola, 1993). Recently it has been suggested that nitric oxide (NO) plays a role (Iadecola, 1993). Nitric oxide is a rapidly diffusible relaxant mediator, generated by enzymes constitutively expressed by the endothelium and neurons (Edelman and Gally, 1997). Studies using NO synthase inhibitors in both animals and man have demonstrated the importance of NO in maintaining basal CBF (Iadecola et al., 1994). A number of studies with NO synthase inhibitors in rodents have shown inhibition of the cortical rCBF increase in response to vibrissal stimulation (Dirnagl et al., 1993) but other studies have failed to replicate this finding (Wang et al., 1993). Recent work with NO synthase isoform knockout mice suggests that neuronally derived NO is the prime mediator of vasoneuronal coupling (Ma et al., 1996).
The human circulation has unique features, which include an extensive intracranial—extracranial collateralization and the need to supply a highly developed neocortex. This makes direct extrapolation of findings from animal studies difficult. Blood flow in peripheral vascular beds in man is NO-dependent (Vallance et al., 1989), and we have recently demonstrated that human internal and common carotid artery flow is reduced by NO synthase inhibition (White et al., 1998). To test the hypothesis that NO contributes to vasoneuronal coupling we have used the non-isoform-selective NO synthase inhibitor, NG-monomethyl-
METHODS
Subjects
Ten healthy male volunteers were studied, 37.2 ± 4.2 years of age and weighing 85.1 ± 8.5 kg (mean ± SD). Local Hospital Ethics Committee approval was obtained for the study and all subjects gave informed written consent. Permission to administer radioactive H215O was given by the Administration of Radioactive Substances Advisory Committee of the Department of Health, United Kingdom.
Chemicals
NG-Monomethyl-
Data acquisition and CBF measurement
Measurements of rCBF were performed using a CTI 953B PET camera (CTI/Siemens), with interplane septa retracted to acquire data in three-dimensional mode, and a low-energy window was used to measure scatter. Data were acquired simultaneously over 31 planes within the camera's 10.65-cm axial field of view. Backprojection and filtering with a Hanning filter (0.5 cycles/pixel) produced an image resolution of 8.5 × 8.5 × 4.3 mm full-width half-maximum (FWHM) after reconstruction. Images were displayed as a matrix of 128 × 128 × 31 voxels. Scans were acquired after a 20-second intravenous bolus of 11.5 mCi of oxygen-15-labeled water (H215O) in 3 mL of isotonic saline, given via an antecubital fossa vein. Time frames comprised a 30-second background frame, started approximately 5 seconds before the point of rise in head counts, followed by five frames of 7 seconds, two of 30 seconds, one of 90 seconds, and one of 180 seconds. Peak head counts occurred 30 to 40 seconds after head count rise. To enable quantitative rCBF analysis arterial whole blood 15O activity was continuously measured using an on-line detection system with blood withdrawn continuously through a radial artery cannula at a rate of 5 mL/min. After each rCBF measurement, 15 minutes was allowed to elapse to ensure decay of any residual 15O nuclide before the next scan. A total of six to eight dynamic CBF scans were performed for each subject.
Activation paradigm
Scans were performed under two different experimental conditions: a baseline rest condition and an activation condition during which a novel sequence of finger movements was learned by trial and error (Jenkins et al., 1994). The activation paradigm required subjects to learn sequences of keypresses eight moves long, previously programmed into a personal computer, using a keypad with four keys corresponding to the fingers of the right hand. The keypad was linked to an Amiga 2000 computer, which prompted a finger movement every 3 seconds and indicated by high and low tones whether a subject had selected the correct key in the sequence (Jenkins et al., 1994). It also monitored subject performance in terms of response time and number of errors. The preprogrammed sequences were the same for all 10 subjects.
Before the subjects were positioned within the scanner they were given an opportunity to familiarize themselves with the paradigm by learning a sequence of finger movements that was not repeated during the actual study. Subjects had either three or four activation scans interspersed with rest scans. To control for auditory input during the activation study the Amiga computer was set to generate random prompt and feedback tones during the rest condition.
The scans were performed in the following order: rest, activation, rest, activation, rest, activation. The first four scans were performed during an infusion of isotonic saline via the left antecubital fossa. Before the fifth scan 10 mg/kg L-NMMA was administered as a 30-second intravenous bolus and then a 10-mg·kg−1·h−1 infusion of L-NMMA was commenced via a left antecubital fossa vein. In previous studies of the administration of intravenous L-NMMA the maximal effect on systemic blood pressure and carotid artery volume flow has been found between 5 and 10 minutes after administration (White et al., 1998); therefore, the fifth scan was delayed until this time after the dose had elapsed. All subjects had six rCBF measurements, after which the effects of L-NMMA were reversed by giving a molar excess of
All scans were performed in a darkened room with the subject's eyes closed. Subjects were studied in a supine position, with head position maintained by a molded, cushioned head support. Axial positioning with a plane passing through the anterior and posterior clinoid processes was achieved by alignment of line markings drawn on the forehead and orbitomeatal lines with laser lines projected perpendicularly from the scanner gantry. Final axial head positioning to include the cerebral vertex in the field of view was performed after a short transmission scan. Before the emission data acquisition, a 20-minute transmission scan was performed to correct for tissue attenuation.
Data analysis
All calculations and image transformations were performed on a Sun SPARC 5 workstation (Sun Computers Inc.) using Analyze version 7.0 image display software (BRU, Mayo Foundation). Functional parametric images of CBF were generated essentially as described by Lammertsma et al. (1990). Integral images, formed from the summation of the counts collected in frames 2 through 10 (30 through 150 seconds, see above), were transformed to CBF using a lookup table. The lookup table was generated from the measured arterial input function with corrections for delay and dispersion during the on-line assay of radioactivity in blood. Delay and dispersion were estimated by fitting the total count rate from the scanner to a flow model with an exponential dispersion term (Lammertsma et al., 1990). Global CBF was determined from whole brain regions of interest excluding lateral ventricles, drawn on nine central transaxial planes of the CBF images. The observed estimates of global flow (see Results) were low relative to those obtained with classic methods. This may reflect a systematic bias arising from gray and white matter heterogeneity in the images, and also from the estimates of dispersion in the blood collection protocol (Lammertsma et al., 1990). It should be noted, however, that the present study was concerned with changes in regional flow and not just the absolute values.
Significant rCBF changes were localized using statistical parametric mapping software (SPM 95, Wellcome Department of Cognitive Neurology, London, U.K.), implemented in Matlab (Mathworks Inc.). Dynamic H215O scans for each individual were realigned on a voxel-by-voxel basis using an automated realignment program based on six-parameter rigid body transformations with a least-squares technique (Friston et al., 1995a). This generated an aligned set of images and a mean image (each of 31 planes) for each subject. For each subject, the mean image, having the most anatomic detail, was transformed into standard stereotactic space using linear, quadratic, and nonlinear three-dimensional deformation on a slice-by-slice basis (Friston et al., 1995a). The same transformations were then applied to each of the subjects' realigned images to allow intersubject averaging. Transformed scans consisted of 26 planes with voxel dimension 2 × 2 × 4 mm, corresponding to the atlas of Talairach and Tournoux (1988). Smoothing was performed using an isotropic Gaussian filter of 20 mm to increase signal-to-noise ratio and to allow for the variation in intersubject gyral anatomy. Relative differences in basal and activated rCBF before and after L-NMMA were localized after proportional scaling, which normalized mean global CBF. This procedure generated adjusted mean rCBF values for each of the conditions (rest and activation without L-NMMA; rest and activation with L-NMMA). The location of significant activity in different brain regions during performance of the motor task both before and after L-NMMA was determined by comparison of the activation with rest image using the Student's t statistic. This generated statistical parametric maps (SPM) which were subsequently converted to (SPM) maps for display. The threshold of the SPM maps was set at P < 0.01 (uncorrected) for display purposes to show areas of activation associated with each comparison in three orthogonal planes. Uncorrected thresholding was used to provide greater power to eliminate false negatives when searching for effects of L-NMMA on local activation changes. Corrected P values were applied to demonstrate the activation associated with the motor learning paradigm. In the comparison of activation with rest, significant results (P < 0.05) were displayed after corrections for multiple non-independent comparisons. The level of significance of these areas were characterized by their peak height, using estimations from the Theory of Gaussian Fields.
In two subjects an acquisition error led to the second rest scan before L-NMMA not being available for analysis. Therefore in all 10 subjects the first rest and activation scan and the post-L-NMMA rest and activation scan were analyzed; however the second rest and activation pair were only available for analysis in eight subjects. Results given in the text section are from all 10 pairs for comparisons when all 10 scans in the pair were available and for 8 pairs when comparisons included the second rest scan.
Changes in rCBF during the motor task were measured from the peak areas of activation using normalized values from the SPM matrix. Estimations of the smallest difference in the magnitude of the activation-induced rCBF change that could be detected at the 95% confidence limit were generated from a power calculation on the basis of the standard error of the contrasts in the SPM matrix and mean differences in corrected activation-induced rCBF changes before and after L-NMMA.
In addition a region-of-interest analysis was performed to compare rest before and after L-NMMA. This was performed by application of a standardized magnetic resonance imaging-based template, subdivided into regions of interest, and applied to the realigned and normalized CBF images. Regional CBF data thus generated were then proportionally scaled for changes in global CBF using the scaling factor generated within the SPM analysis, to generate a relative rCBF value. These were compared by paired t test.
Systemic hemodynamic variables
Mean arterial blood pressure was measured by an automated arm cuff (Dynamap) and pulse rate by a three-lead ECG rhythm strip. Three measures of arterial blood pressure and three 30-second rhythm strips were acquired before and after each dose or scan, and mean values are presented.
RESULTS
Global cerebral blood flow
Global CBF values during rest and activation conditions before and after L-NMMA are illustrated in Fig. 1. Infusion of L-NMMA resulted in a significant fall in global CBF: CBF fell from 33.3 ± 5.3 mL·100 g−1 ·min−1 during rest before L-NMMA, to 26.5 ± 7.7 mL·100 g−1·min−1 after L-NMMA (P = 0.001). Before L-NMMA administration there was no difference in global CBF during the rest and activation periods (33.3 ± 5.3 versus 35.3 ± 5.5 mL·100 g−1·min−1; P = 0.263). After L-NMMA administration, global CBF during activation was higher than that during the preceding rest period (31.4 ± 5.8 versus 26.5 ± 7.7 mL·100 g−1·min−1; P = 0.038).
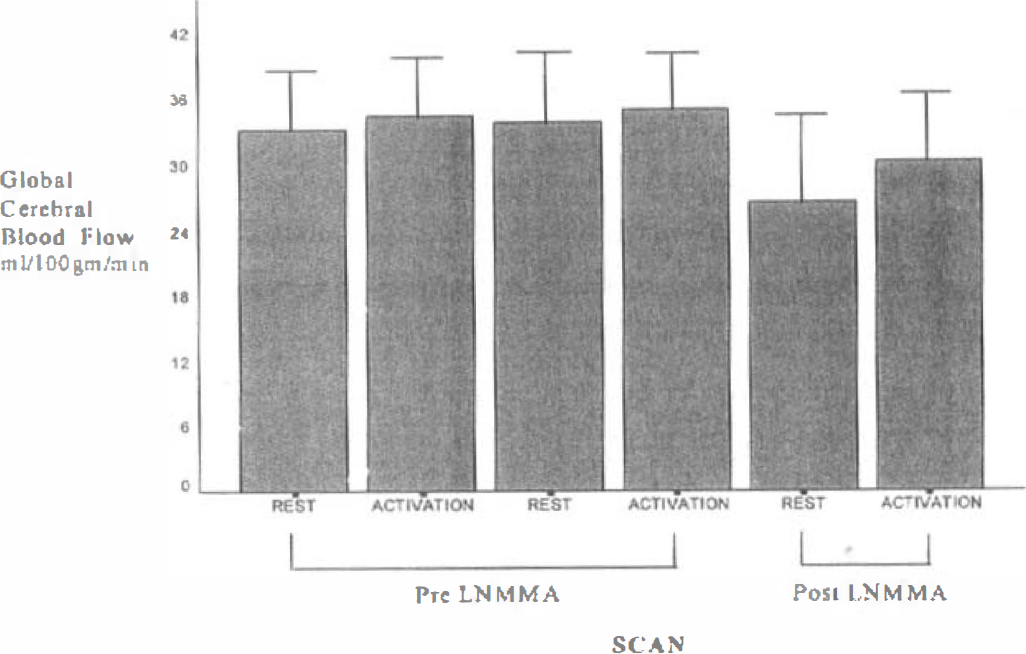
Global CBF values (mean ± SD) during rest and activation conditions before and after L-NMMA in eight subjects in whom complete data were available.
In those seven subjects who received
Activation-induced rCBF changes
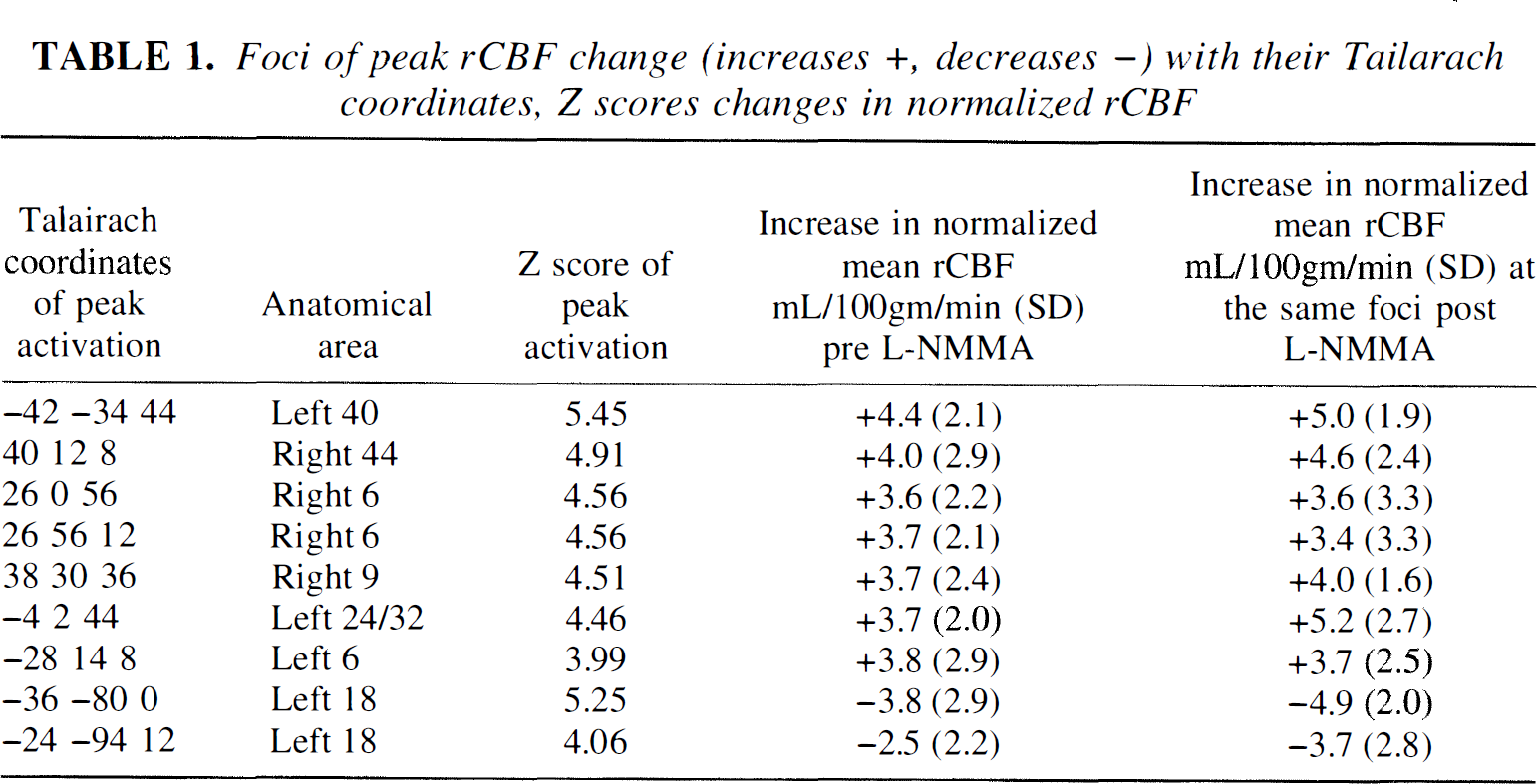
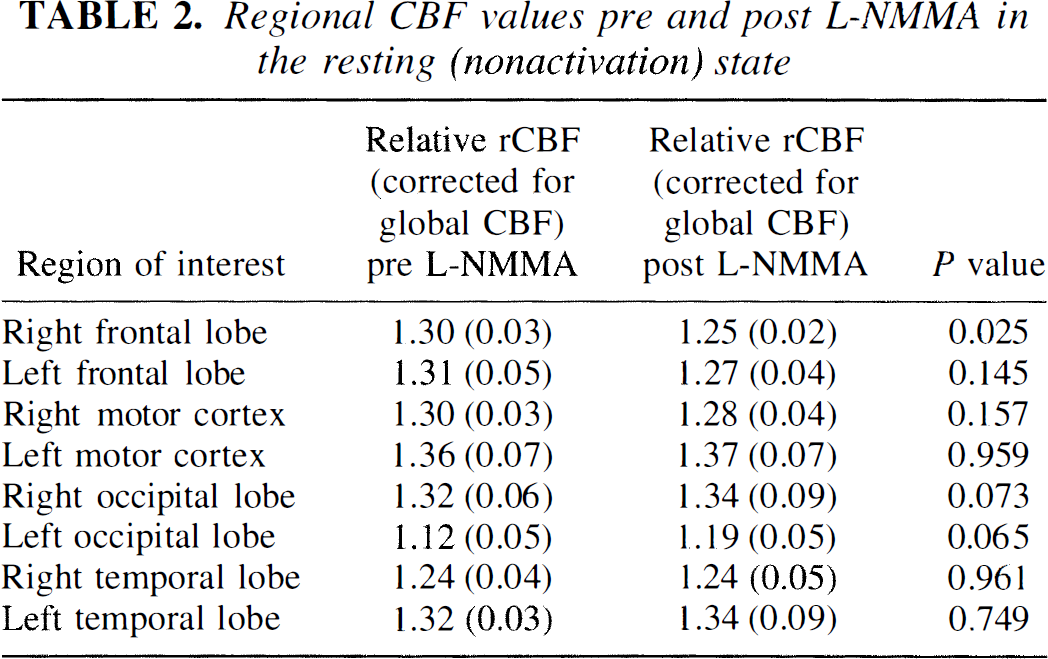
The areas in which there were significant rCBF changes after activation (P < 0.05, corrected) are listed in Table 1. No significant differences in rCBF were observed before and after L-NMMA comparing the rest condition using either the SPM analysis or the region-of-interest analysis, even at an uncorrected threshold of P < 0.01. Region-of-interest CBF relative to global CBF values are given in Table 2.
Foci of peak rCBF change (increases +, decreases –) with their Tailarach coordinates, Z scores changes in normalized rCBF
Regional CBF values pre and post L-NMMA in the resting (nonactivation) state
Increases Comparing activation versus rest before L-NMMA, the cortical areas of significantly increased blood flow were the left motor cortex, right prefrontal cortex (area 9), right and left premotor cortices (area 6), and left parietal area 40. No significant differences in activation versus rest were observed in rCBF after L-NMMA compared with before L-NMMA.
The increases or decreases in normalized rCBF after activation are given in Table 1. These are measured at the peak focus of activation. Notably, after L-NMMA, the rCBF values were consistently greater than the before L-NMMA values. Our study had 95% power at a P value of 0.05 uncorrected to detect a 5% reduction in the activation-induced rCBF increase in the left premotor cortex, and a 9% reduction in the activation-induced rCBF increase in the right premotor cortex.
Decreases A significant reduction in occipital lobe rCBF was observed during activation compared with rest before L-NMMA. Peak activation reduction in normalized rCBF was observed in left area 18. Again no difference in rCBF was observed comparing activation before L-NMMA with activation after L-NMMA.
The study had 95% power to detect a 15% reduction in the activation-induced rCBF decrease in left area 18 at a significance level of 0.05 (uncorrected).
Task performance During scanning no omissions in motor sequencing were made by the subjects. One subject managed to learn one of the sequences to the point of being able to reproduce it without errors during PET whereas a further subject managed to learn two sequences. Increased skill at performance took place across the sequences with a reduction in errors and decreased response times. No significant difference in response times (0.65 ± 0.17 and 0.65 ± 0.16 seconds) or total number of errors (21.5 ± 3.2 and 21.4 ± 4.8) was observed during task performance before and after L-NMMA, respectively.
Systemic hemodynamic response
After L-NMMA there was an increase in blood pressure: rest before L-NMMA, 84.0 ± 8.2 mm Hg versus rest after L-NMMA, 103.6 ± 12.3 mm Hg; P < 0.001. Blood pressure remained significantly elevated during activation after L-NMMA at 99.7 ± 12.6 mm Hg, but this was significantly less than the arterial blood pressure during rest after L-NMMA (P = 0.038).
After L-NMMA there was a reduction in pulse rate from 61.1 ± 9.0 beats/min (rest before L-NMMA) to 47.1 ± 7.2 beats/min (rest after L-NMMA) (P < 0.001). Pulse rate remained reduced during activation after L-NMMA at 49.1 ± 6.7 beats/min; this was not significantly different from the rest after L-NMMA pulse rate (P = 0.222).
In those 7 subjects who received
DISCUSSION
In this study inhibition of NO synthase with L-NMMA resulted in a 20% fall in global CBF. In those subjects who received
We found no reduction in the rCBF increase associated with a brain activation paradigm after L-NMMA administration. The sequence learning paradigm that we used in this study has been reported to result in a characteristic pattern of regional neuronal activation, with increased rCBF in the left motor, right and left premotor, and right prefrontal cortices, and reduced occipital cortex rCBF (Jenkins et al., 1994). Our results were consistent with this pattern of rCBF change. Systemic NO synthase inhibition did not alter the pattern or degree of this rCBF response. Statistical power calculations estimated that we should have been able to detect a 5% reduction in rCBF in premotor cortex activation after L-NMMA, implying that there was little likelihood of missing an effect of NO synthase inhibition on activation-induced rCBF. There was no objective change in levels of arousal or vigilance, which have been reported to fall after NO synthase inhibition (Dzoljic et al., 1997), as there was a similar frequency of errors and mean response rates recorded before and after L-NMMA.
The effect of NO synthase inhibition on vasoneuronal coupling has been examined in several animal models of regional neuronal activation. Superfusion of rat cerebral cortex with NO synthase inhibitors abolished the global, noncortical neuronal activation-dependent CBF increase observed after cerebellar fastigial stimulation in the rat (Iadecola et al., 1987), and the coordination of flow and metabolism coupling in a cat model of cortical spreading depression (Goadsby et al., 1992). The somatosensory rCBF increase after vibrissal stimulation in rats has been reported to be reduced (Dirnagl et al., 1993) or unaltered (Wang et al., 1993) after systemic or topically applied NO synthase inhibition. However, other similar experiments have failed to confirm this finding. Interpretation of the results of animal studies is made more difficult by the use of anesthetic agents. The NO synthase inhibitor, 7-nitroindazole, which has been suggested to have specificity for the neuronal isoform, has been reported to reduce both basal and stimulus-evoked flow increases without altering neuronal metabolism (Cholet et al., 1997). However, interpretation of these results is complicated by studies suggesting that 7-nitroindazole inhibits both endothelial and inducible isoforms of NO synthase as well as the neuronal isoform (Blandward and Moore, 1995). Studies in endothelial and neuronal NO synthase knockout mice implicate neuronally derived NO as the mediator of stimulus-evoked rCBF increases (Ma et al., 1996). However, interpretation of these results is complicated by the possibility of other compensatory pathways developing to subserve for any NO absence. Indeed one of the most striking features of NO synthase knockout mice is the normality of their cerebrovascular physiology (Irikura et al., 1995). The observation that the glutaminergic neurotransmitter system can increase rCBF via NO-dependent mechanisms provides for a potential link between regional neuronal activity and blood flow (Faraci and Bresse, 1993).
To ensure continuous sustained inhibition of NO synthase we followed the bolus of L-NMMA with a maintenance intravenous infusion. However, it is possible that there may have been a lesser degree of NO synthase inhibition during the post-L-NMMA activation paradigm compared with the preceding resting scan. The observation that global CBF values were higher during activation compared with rest after L-NMMA is consistent with this. Nevertheless, the resting CBF and systemic hemodynamic indices were still significantly altered at this time, suggesting that there was significant endothelial NO synthase inhibition.
A potential problem with the use of
Footnotes
Acknowledgments
The authors thank Glaxo Wellcome for providing L-NMMA, David Leonard for helping prepare the
