Abstract
The authors studied, by in vivo phosphorus magnetic resonance spectroscopy (31P-MRS), the occipital lobes of 19 patients with mitochondrial cytopathies to clarify the functional relation between energy metabolism and concentration of cytosolic free magnesium. All patients displayed defective mitochondrial respiration with low phosphocreatine concentration [PCr] and high inorganic phosphate concentration [Pi] and [ADP]. Cytosolic free [Mg2+] and the readily available free energy (defined as the actual free energy released by the exoergonic reaction of ATP hydrolysis, i.e., ΔGATPhyd) were abnormally low in all patients. Nine patients were treated with coenzyme Q10 (CoQ), which improved the efficiency of the respiratory chain, as shown by an increased [PCr], decreased [Pi] and [ADP], and increased availability of free energy (more negative value of ΔGATPhyd). Treatment with CoQ also increased cytosolic free [Mg2+] in all treated patients. The authors findings demonstrate low brain free [Mg2+] in our patients and indicate that it resulted from failure of the respiratory chain. Free Mg2+ contributes to the absolute value of ΔGATPhyd. The results also are consistent with the view that cytosolic [Mg2+] is regulated in the intact brain cell to equilibrate, at least in part, any changes in rapidly available free energy.
Intracellular total and free magnesium concentration ([Mg2+]) has been assessed in patients with some neurologic disorders (Ramadan et al., 1989; Taylor et al., 1991; Yasui and Ota, 1992; Yasui et al., 1992; Stelmiasiak et al., 1995; Welch and Ramadan, 1995; Lodi et al., 1997) and has been found to be decreased in multiple sclerosis (Yasui and Ota, 1992; Stelmiasiak et al., 1995), amyotrophic lateral sclerosis (Yasui et al., 1992), and migraine (Ramadan et al., 1989; Welch and Ramadan, 1995; Lodi et al., 1997). Therefore, deranged magnesium homeostasis could be involved in the pathogenesis of these diseases (Yasui and Ota, 1992; Yasui et al., 1992; Welch and Ramadan, 1995). The findings of low [Mg2+] in migraine also has led to treatment of patients with magnesium, although with controversial results (Mauskop et al., 1996; Peikert et al., 1996; Pfaffenrath et al., 1996).
A defective energy metabolism in both brain (Welch et al., 1989; Barbiroli et al., 1992; Montagna et al., 1994; Montagna et al., 1997) and skeletal muscles (Barbiroli et al., 1992; Montagna et al., 1994; Lodi et al., 1997; Montagna et al., 1997) also has been documented in some of the migraine headaches associated with low intracellular [Mg2+] (Lodi et al., 1997; Welch and Ramadan 1995). The latter findings raise the question of whether low magnesium concentration or defective mitochondrial energy production represents the primary causative factor in pathogenesis.
From a biochemical point of view, magnesium is a critical cofactor for several enzyme reactions involved in the pathways of energy transductions and is known to strongly influence the actual amount of energy released by the exergonic reaction of ATP hydrolysis (Veech et al., 1979; Masuda et al., 1990). Hence, magnesium functionally is closely related to the cell bioenergetics and ion transport systems.
To clarify the functional relations existing between magnesium ions and tissue bioenergetics, we assessed by in vivo phosphorus magnetic resonance spectroscopy (31P-MRS) the mitochondrial functioning and the cytosolic free Mg2+ concentration in the occipital lobes of 19 patients with mitochondrial cytopathies resulting from known enzyme or mitochondrial DNA (mtDNA) defects. To further investigate the functional relations between changes in intracellular free [Mg2+] and tissue bioenergetics, we took advantage of the fact that treatment with coenzyme Q10 (CoQ) increases the efficiency of mitochondrial respiration both in vitro (Lenaz et al., 1997) and in vivo (Bendahan et al. 1992; Barbiroli et al., 1997). Therefore, we performed 31P-MRS in nine of our patients before and after treatment with CoQ.
SUBJECTS AND METHODS
Patients, treatment, and controls
The patient group (Table 1) consisted of 19 subjects (9 women and 10 men) with a mean age of 43 ± 19 (SD) years (13 to 75 years) with mitochondrial cytopathies. Four patients had chronic progressive external ophthalmoplegia (CPEO), isolated or associated with mild myopathic involvement of the limbs; 10 had familial Leber's hereditary optic neuropathy with different mtDNA point mutations, as specified in Table 1; 2 had neurogenic atrophy, ataxia, retinitis pigmentosa syndrome; I had a mitochondrial encephalomyopathy with mtDNA deletion; 1, a myoclonic epilepsy with ragged red fibers (MERRF); and 1, a mitochondrial encephalomyopathy with lactic acidosis and stroke-like episodes (MELAS) syndrome. Three of CPEO patients had deficit of cytochrome oxidase on muscle biopsy. Ragged red fibers, the hallmark of mitochondrial alterations on muscle biopsy, were present in all CPEO patients, in one case associated with mtDNA deletions, and in patients with MERRF, MELAS, and mitochondrial encephalomyopathy.
Phosphorus MR spectroscopy data of brains (occipital lobes) from 19 patients with mitochondrial cytopathies caused by known enzyme/mtDNA defects
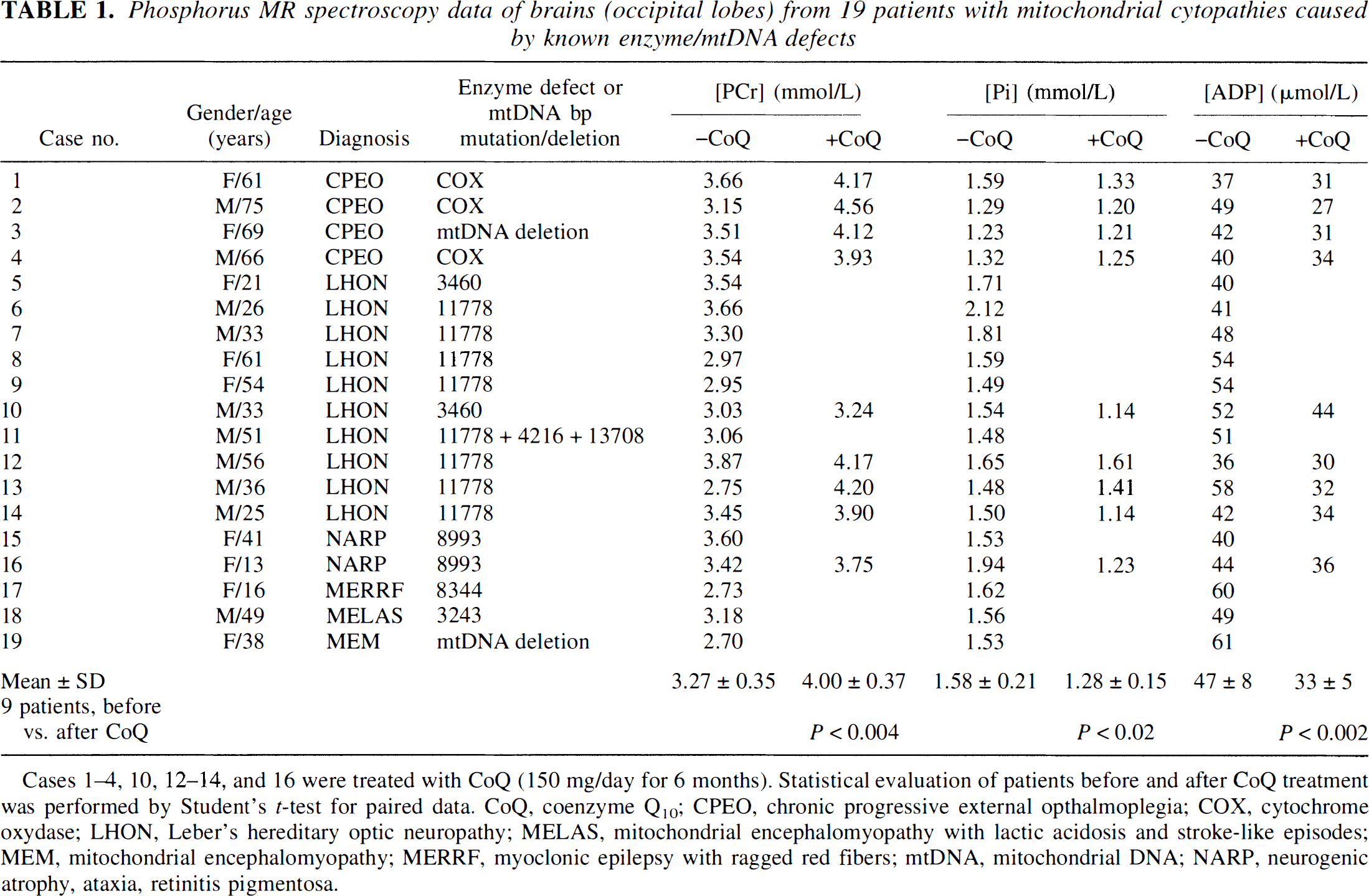
Cases 1–4, 10, 12–14, and 16 were treated with CoQ (150 mg/day for 6 months). Statistical evaluation of patients before and after CoQ treatment was performed by Student's t–test for paired data. CoQ, coenzyme Q10; CPEO, chronic progressive external opthalmoplegia; COX, cytochrome oxydase; LHON, Leber's hereditary optic neuropathy; MELAS, mitochondrial encephalomyopathy with lactic acidosis and stroke-like episodes; MEM, mitochondrial encephalomyopathy; MERRF, myoclonic epilepsy with ragged red fibers; mtDNA, mitochondrial DNA; NARP, neurogenic atrophy, ataxia, retinitis pigmentosa.
Treatment with oral CoQ (150 mg/day) was given for 6 months. The MRS scans were performed just before starting therapy and 6 months later.
Thirty age-matched healthy control subjects (11 women and 19 men) with a mean age of 38 ± 16 years (SD) (16 to 60 years) who were free from any neurologic disorders, including migraine, volunteered for the study. Informed written consent was obtained from patients and control subjects. The MRS scans on healthy controls, untreated patients, and treated patients before and after CoQ were performed randomly.
Phosphorus magnetic resonance spectroscopy
The 31P-MRS was performed by means of a GE 1.5-T Signa System (General Electric, Milwaukee, WI, U.S.A.) with a spectroscopy accessory as already described (Barbiroli et al., 1995). All spectroscopic measurements were performed according to the quantification and quality assessment protocols defined by the EEC Concerted Research Project on “Tissue Characterisation by MRS and MRI,” COMAC-BME II.1.3 (EEC Concerted Research Project, 1995).
Occipital lobes were examined by the depth resolved surface coil spectroscopy localization technique, which, in our experimental conditions, localizes the signals mainly from the brain cortex. Patients and controls were asked to lie at rest with closed eyes. The surface coil was placed directly on the skull in the occipital region and precisely positioned by imaging the brain.
Cytosolic pH and free Mg2+ concentration
Cytosolic pH was calculated from the chemical shift of inorganic phosphate (Pi) from phosphocreatine (PCr), and free [Mg2+] was assessed by a semiempirical equation that correlates the chemical shift of β-ATP resonance from PCr to the free [Mg2+], taking into account most cytosolic phosphate compounds binding Mg2+, cytosolic pH, ionic strength, and temperature (Iotti et al., 1996). The chemical shift was carefully determined from the centroid of the PCr peak to the centroid of the phosphate and β-ATP peaks.
The ΔG of ATP hydrolysis (ΔGATPhyd), defined as the actual free energy released by the reaction of ATP hydrolysis in the intact cell, was calculated by the equation
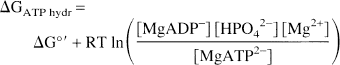
in which the brackets denote molar concentrations; R is the gas constant (8.315 J/mol); T, the absolute temperature (310°K); and ΔG°' is the free energy change of the reaction of ATP hydrolysis in standard conditions, that is, molar concentration of reactant and products at 25°C and pH 7. [MgADP−], [HPO42–], and [MgATP2–] denote the prevalent species at pH 7, but calculation of ΔGATPhyd did take into account all species of ATP, ADP, and Pi bound and unbound to magnesium.
Data analysis
Spectra were analyzed blindly without knowing the subject's status (whether a treated or untreated patient or healthy subject). Brain metabolite concentrations were calculated assuming a cytosolic [ATP] of 3.0 mmol/L in patients and healthy subjects (Bottomley and Hardy, 1989), reasoning that if [ATP] were lower than controls in these patients, [PCr] would be even lower and [ADP] even higher. Brain mitochondrial functionality was assessed by calculating [ADP] from the creatine kinase equilibrium (Chance et al., 1986), assuming creatine kinase to be a near-equilibrium reaction (Veech et al., 1979) in our patients. Statistical significance was determined using Student's t–test for both paired and unpaired data. Significant difference was assumed with P < 0.05. We also assumed two SD from the mean value of healthy controls to be the lower and upper reference limits for all metabolite contents and variables.
RESULTS
Figure 1 shows representative 31P-MR spectra of occipital lobes from a patient with Leber's hereditary optic neuropathy (case 6, spectrum A) compared with a sex- and age-matched control (spectrum B). A low intensity of PCr resonance can be seen in the patient's spectrum accompanied by a small increase in Pi resonance. Calculated [ADP] was abnormally high, and cytosolic free [Mg2+] was abnormally low. Cytosolic pH was within normal range.
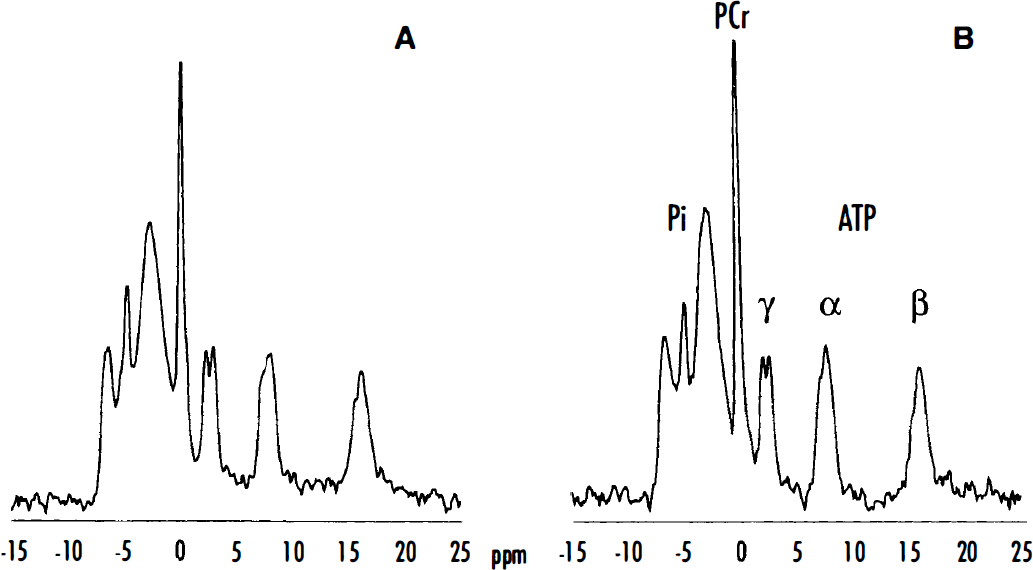
Typical phosphorus magnetic resonance (31P-MR) spectra of occipital lobes from a patient with Leber's hereditary optic neuropathy (LHON) (case 6, spectrum
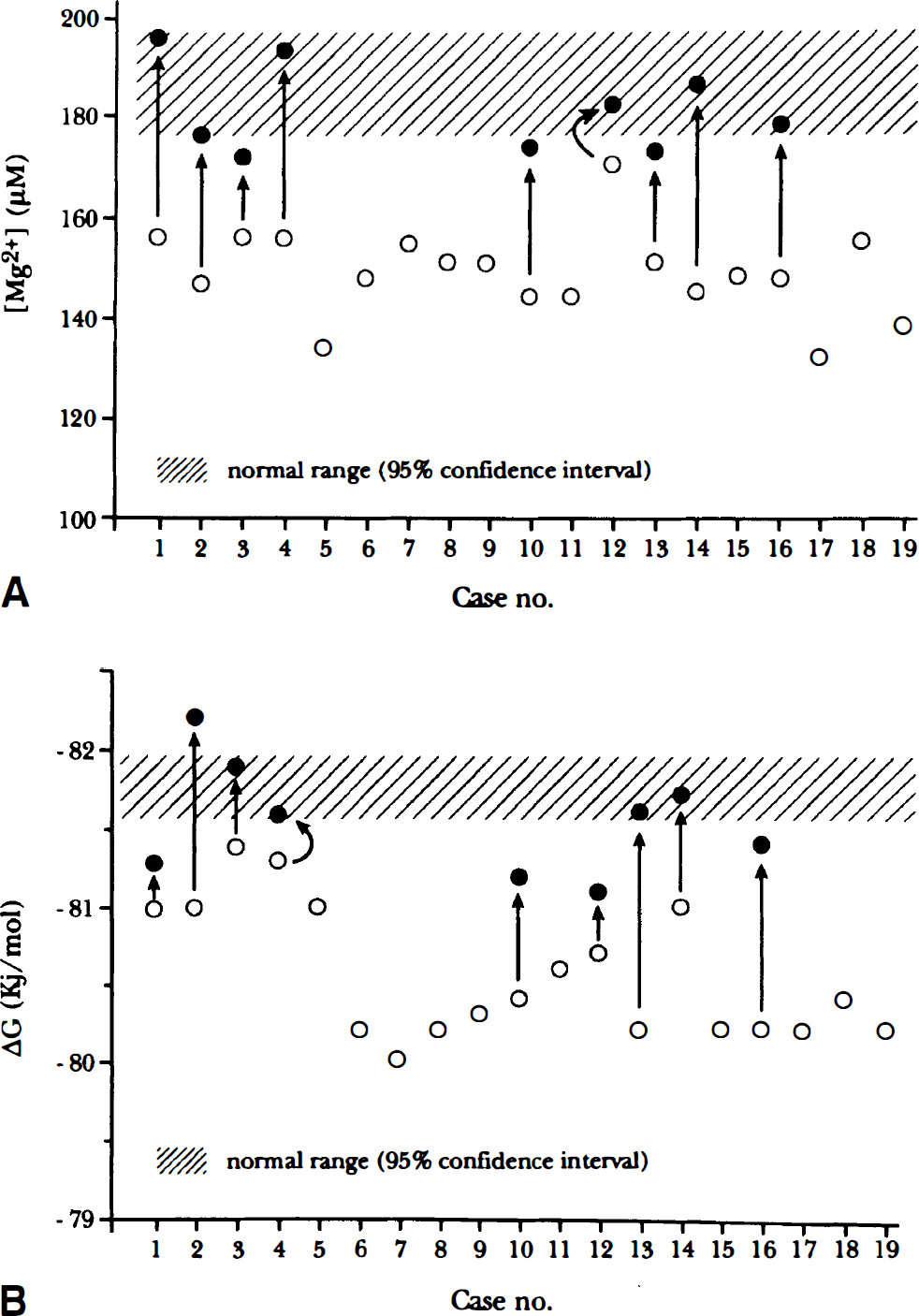
These MRS findings were representative of the entire patient group, as shown in Table 1. The mean [PCr] (3.27 ± 0.35, SD) was remarkably (P < 0.001) lower than the mean control value (4.39 ± 0.27, SD), and the average concentration of brain cytosolic Pi (1.58 ± 0.21, SD) was significantly (P < 0.001) higher than the mean of the control group (1.28 ± 0.12, SD). Intracellular pH was normal in all patients (data not reported). Calculated from the creatine kinase reaction (Chance et al., 1986), [ADP] was more than two SD higher than the mean control value (29 ± 2, SD) in all patients (Table 1). The average of cytosolic free [Mg2+] was remarkably (P < 0.001) lower (149 µmol/L ± 9, mean ± SD) than the mean control value (185 µmol/L ± 27, mean ± SD). The average ΔG value of cytosolic ATP hydrolysis in the brains of our patients (–80.6 kJ/mol ± 0.4, mean ± SD) also was significantly (P < 0.001) higher than the healthy controls (–81.8 kJ/mol ± 0.5, mean ± SD).
Nine patients (cases 1 to 4, 10, 12 to 14, and 16) were treated with CoQ for 6 months; the results are reported in Table 1 and Fig. 2. Treatment with CoQ significantly (P < 0.004) increased [PCr], leading to normal values of PCr content in the brains of all patients except for two (cases 10 and 16). The CoQ treatment also significantly (P < 0.02) decreased the brain [Pi] and significantly (P < 0.002) improved calculated [ADP]. Treatment with CoQ also led to a significant (P < 0.001) increase in cytosolic free [Mg2+] in the brains of all patients (Fig. 2), the mean value in these nine patients being 153 µmol/L ± 9 (SD) and 182 µmol/L ± 12 (SD) before and after treatment, respectively. The ΔG of ATP hydrolysis also was significantly (P < 0.002) decreased after treatment, showing an average value of −81.5 kJ/mol compared with the mean value of −80.8 kJ/mol found before therapy.
Cytosolic free Mg2+ concentration (
DISCUSSION
The brains of all of our patients showed a 31P-MRS pattern with low [PCr] and high [Pi], indicating defective mitochondrial respiration (Eleff et al., 1990; Barbiroli et al., 1993). The [ADP] calculated from the creatine kinase equilibrium (Chance et al., 1986) was increased in all patients. Adenosine diphosphate is a major regulator of oxidative phosphorylation and a sensitive index of mitochondrial functionality and high cytosolic free [ADP], indicating that brain cells are less able to handle any further energy demand because of the malfunctioning mutated mitochondria, thus reflecting defective metabolic conditions. All of our patients displayed a remarkably low value of free energy released by the reaction of ATP hydrolysis (high value of ΔGATPhyd), another index of mitochondrial functionality, since it takes into consideration the concentration of both high- and low-energy phosphates. All of our patients also displayed significantly low intracellular free [Mg2+], well below the 95% confidence interval in the healthy population. Since the mtDNA mutations and enzyme defects found in our patients (Table 1) are known to be primarily responsible for defective mitochondrial respiration in their brains, our results strongly suggest that the low cytosolic free [Mg2+] found in these patients (Fig. 2) was secondary to failure of the respiratory chain. We emphasize that we measured the free aliquot of cytosolic magnesium, which reflects the total cytosolic content as it is in equilibrium with all bound forms.
From a bioenergetic point of view, magnesium ions contribute to the absolute value of ΔG of ATP hydrolysis. As in most enzymatic reactions involving ATP the true substrate is MgATP2–, the actual amount of energy released by this highly exoergonic reaction is that for MgATP2–. Thus, the ΔGATPhyd can vary as a function of free [Mg2+]. As a consequence, in the presence of even a small increase in [Pi] and [ADP] with constant free [Mg2+], the value of ΔGATPhyd increases (i.e., the released free energy decreases), whereas a concomitant decrease of free [Mg2+] tends to limit any increase in ΔGATPhyd. In this light, the brains of our patients in the presence of normal free [Mg2+] (i.e., 185 µmol/L, as in controls) would have displayed an average ΔGATPhyd of −79.8 kJ/mol rather than −80.6 kJ/mol as found, corresponding to a further remarkable decrease of rapidly available free energy in the brain cells. Our interpretation of low free Mg2+ content in the brains of these patients is that when the cell's ability to respond to metabolic stress is impaired because of defective mitochondrial respiration, the intracellular [Mg2+] is down-regulated and the rapidly available free energy in the cell (i.e., the ΔG of ATP hydrolysis) is, at least in part, re-equilibrated.
Treatment of nine patients with CoQ resulted in a clear-cut improvement of the mitochondrial respiration efficiency in the brain cells, as shown by increased content of the PCr energy buffer, decreased [ADP], and decreased ΔGATPhyd in all cases. These results are consistent with Lenaz's current hypothesis, based on in vitro experiments, that increased CoQ concentration in the inner mitochondrial membrane increases the efficiency of oxidative phosphorylation, and that decreased respiratory rate from partial deficiency of electron transport chain enzymes, as in our patients, can be compensated by CoQ excess (Lenaz et al., 1994; Lenaz et al., 1997). Our current findings also are consistent with recent in vivo results (Bendahan et al., 1992; Barbiroli et al., 1997; Cortelli et al., 1997) obtained from patients with mitochondrial cytopathies disease showing a beneficial effect of quinol treatment on both the efficiency of mitochondrial respiration and patients' clinical status.
The increased efficiency of oxidative phosphorylation after treatment with CoQ was accompanied by an increased cytosolic free [Mg2+] in all patients, again showing that the changes in free [Mg2+] in the brain of our patients were secondary to the improved efficiency of mitochondrial energy production. Despite results showing that magnesium ions can modulate in vitro the activity of ATP synthase in isolated rat heart mitochondria (Rodriguez-Zavala et al., 1998), our current in vivo findings are consistent with the hypothesis that brain cells regulate their cytosolic free [Mg2+] as a function of the energy charge of the cell, and maintain constant (as far as possible) the energy released by the exergonic reaction of ATP hydrolysis, that is, the cell's free energy rapidly available to respond to sudden metabolic stresses.
Because of its large size relative to K+ and other ions, it is believed that Mg2+ does not cross plasma membranes through channels or proteins pores, and little information is available on the reactions catalyzing the distribution of free [Mg2+] across plasma membranes and their regulation in the brain. Therefore, it is not possible to answer the question of whether a fast short-term or a slow long-term process regulates the cytosolic free [Mg2+], and hence, the value of ΔGATPhyd. To answer this question, it is necessary to study the changes of cytosolic free [Mg2+] in other excitable tissues such as the skeletal muscles, where it is possible to bring about rapid changes to the value of the phosphorylation potential and the energy charge of the cell just by exercising.
