Abstract
The effectiveness of 6-[18F]fluoro-L-m-tyrosine (6FMT) to evaluate dopamine presynaptic integrity was compared to that of 6-[18F]fluoro-L-dopa (6FDOPA) in vivo by positron emission tomography (PET). Six normal and six 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) -lesioned monkeys received 6FDOPA and 6FMT PET scans on separate occasions with identical scanning protocols. Four measures, the rate of uptake of tracer into striatum using either the arterial input function (Ki) or the activity in the occipital cortex as the input function (Kc), the rate of loss of striatal radioactivity (kloss), and an index of “effective turnover” of dopamine (kloss/Ki), were obtained for both tracers during extended PET studies. 6-[18F]Fluoro-L-m-tyrosine was as effective as 6FDOPA in separating normals from MPTP-lesioned subjects on the basis of the uptake rate constants Ki and Kc. However, in contrast to 6FDOPA, it was not possible to differentiate the normal from the lesioned animal using kloss or kloss/Ki for 6FMT. Thus, FMT appears to be a reasonable, highly specific tracer for studying the activity of aromatic dopa decarboxylase enzyme as an index of presynaptic integrity. However, if one is interested in investigating further the metabolic pathway and obtaining an in vivo estimate of the effective turnover of dopamine (after pharmacologic manipulation, for example), 6FDOPA remains the tracer of choice.
6-[18F]Fluoro-L-dopa (6FDOPA) has been, for over a decade, one of the most widely used tracers to assess in vivo presynaptic dopaminergic (DA) integrity. The accumulation of 18F activity in brain, and especially in the striatum, has been shown to reflect the combined effects of the decarboxylation of 6FDOPA by aromatic amino acid decarboxylase (AAAD) into 6-[18F]fluoro-dopa-mine (FDA) and its subsequent vesicular storage and metabolism in DA neurons and terminals (Firnau et al., 1987). The cerebral metabolism of 6FDOPA compares well with the metabolism of the natural compound (Cumming et al., 1987; Endres et al., 1997; Firnau et al., 1987; Melega et al., 1991), i.e., decarboxylation processes, affinity for the vesicular monoamine transporter, and susceptibility to the catabolic enzymes monoamine oxidase (MAO) and catechol O-methyltransferase (COMT). The striatal 6FDOPA positron emission tomographic (PET) data are strongly correlated with the number of nigral DA neurons and the concentrations of endogenous DA and metabolites in both brain and cerebrospinal fluid (Pate et al., 1993; Snow et al., 1993). Thus, 6FDOPA appears to be an ideal compound for the in vivo evaluation of central DA function. Unfortunately, 6FDOPA, like L-dopa itself, is subject to peripheral metabolism by AAAD and COMT. The product of the methylation process, the large neutral amino acid 3-O-methyl-[18F]fluoro-L-dopa (3OMFDOPA) readily crosses the blood-brain barrier thereby complicating the interpretation of the data. Modeling approaches to 6FDOPA kinetics in the brain must therefore take into account the existence of a second input function for 3OMFDOPA with its own transport, clearance, and tissue distribution (Dhawan et al., 1997; Doudet et al., 1991; Wahl et al., 1994) to estimate 6FDOPA decarboxylation and clearance rates. Peripheral decarboxylation is usually inhibited in clinical 6FDOPA PET studies by pretreatment with the decarboxylase inhibitor carbidopa, and more recently, the peripheral inhibition of COMT has been achieved safely and effectively in selected groups of normal subjects and patients with Parkinson's disease (Hartvig et al., 1992; Ishikawa et al., 1996; Laihinen et al., 1992; Sawle et al., 1994) and in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) animal model of parkinsonism (Doudet et al., 1997a). However, timely administration of multiple enzyme inhibitors complicates the logistics of 6FDOPA PET studies.
Recently, several reports have advocated the use of fluorinated analogs of m-tyrosine to evaluate DA integrity (DeJesus and Mukherjee, 1988; Melega et al., 1989b; Nahmias et al., 1995). L-m-Tyrosine, an amino acid that is an excellent substrate for AAAD but not for COMT, has been labeled with 18F in either the four or six position of the benzene ring (DeJesus et al., 1997; Namavari et al., 1993). Although both forms appear suitable for the visualization of DA terminals by PET, 6-[18F]fluoro-L-m-tyrosine (6FMT) kinetics in brain and plasma make it the more relevant tracer for PET studies (Barrio et al., 1996; Nahmias et al., 1995; DeJesus et al., 1997). The polar metabolites of 6FMT in plasma, 6-[18F]fluoro-m-tyramine (FMA), 6-[18F]fluoro-hydroxyphenylacetic acid (FPAC), and sulfated conjugates, do not cross the blood-brain barrier, thus greatly simplifying the metabolic profile of the tracer and the modeling of cerebral kinetics. However, recent publications suggest that the metabolic pathway of 6FMT-FMA differs significantly from that of L-dopa-DA and 6FDOPA-FDA. Endres et al. (1997) recently showed that FMA has a low affinity for the DA transporter and the monoamine vesicular transporter. Thus, unprotected FMA, a good substrate for MAO, one of the main catabolic enzymes in the DA metabolic pathway, is rapidly metabolized into FPAC. Accordingly, FPAC is responsible for most of the striatal radioactivity 60 minutes after 6FMT injection (Endres et al., 1997; Firnau et al., 1991; Jordan et al., 1997), as opposed to FDA after 6FDOPA injection. FPAC is not a substrate for COMT, the other catabolic enzyme in the DA metabolic pathway, and appears to have a very slow clearance from the striatum.
On the one hand, the simplification in peripheral metabolism makes 6FMT an attractive choice for PET studies of the DA presynaptic system. On the other hand, the divergence between the metabolic pathways of L-dopa-DA and 6FMT raises concerns as to the usefulness of 6FMT for studies of DA “turnover” and catabolism. Thus, we compared the information provided by 6FMT and 6FDOPA, in the same monkeys, both normal and with a lesion of the DA nigrostriatal pathway, using the same scanning procedure and analysis techniques. A good correlation between 6FMT and 6FDOPA uptake during routine PET studies (up to 2 hours) was already reported in a few normal non-human primates (DeJesus et al., 1997; Barrio et al., 1996). We have recently described a simple method, based on an extension of the graphical analysis of Patlak and Blasberg (1985) to measure the rate of reversibility of 6FDOPA trapping, or rate of loss of radioactivity out of the striatum, kloss, during extended PET studies (3 hours or more) (Holden et al., 1997). The rate constant kloss is a measure of the frequency of turnover of the trapped tracer component and its inverse represents the mean dwell time of that component, but it does not completely characterize the long-term trapping of the tracer because that is also determined by the accumulation rate constant Ki. Thus, the ratio kloss/Ki was used as an in vivo index of the “effective turnover” of DA. However, this effective turnover, estimated in vivo through PET measurement of the kinetics of a fluorinated analog of L-dopa, differs from the traditional concept of neurotransmitter turnover. A tracer of exogenous L-dopa such as 6FDOPA cannot trace DA turnover in the conventional sense. Under normal conditions, DA is synthesized in nerve terminals from the decarboxylation of L-dopa endogenously produced from circulating tyrosine through the action of tyrosine hydroxylase. Exogenously administered L-dopa (or 6FDOPA) can bypass this hydroxylation step. Thus, L-dopa analogs can only test the relative strengths of the multiple relevant processes (uptake, decarboxylation, storage, and elimination). Although these processes may not act on endogenous and exogenous L-dopa at exactly the same rates, our turnover index provides information about the variation of DA storage capacity between brain regions, subjects, and disease states. A change in the ratio kloss/Ki then reflects an active physiologic process, and an increase in turnover appears as an increase in the ratio kloss/Ki. We have already demonstrated in monkeys treated with the neurotoxin MPTP (Doudet et al., 1998) and in animals receiving the central and peripheral COMT inhibitor tolcapone (Doudet et al., 1997b) that, for 6FDOPA, this effective turnover is a useful index of the turnover of DA made from exogenous 6FDOPA and correlates well with traditional neurochemical measures of DA turnover (i.e., concentration of metabolites relative to concentration of DA).
One of the goals of this study is to determine whether 6FMT shares these properties and can be used to provide in vivo a meaningful index of the effective turnover of striatal DA. To this aim, we compared the capabilities of 6FMT and 6FDOPA to separate disease from normality not only by their uptake rate constant (decarboxylation and storage processes) but also by the rate of reversibility of tracer trapping and an in vivo index of effective DA turnover.
MATERIALS AND METHODS
Tracer synthesis
6-[18F]Fluoro-L-dopa was synthesized as previously described (Namavari et al., 1992). 6-[18F]Fluoro-L-m-tyrosine synthesis was as follows: [18F]F2 gas was prepared as described by Roberts et al. (1995). L-m-Tyrosine (36.2 mg; 200 μmol) was placed in a glass reaction vessel (2 × 10 cm) and dissolved in 12 mL of 1:1 trifluoroacetic acid and glacial acetic acid (DeJesus et al., 1995). 18F-Acetyl hypofluorite was bubbled into the mixture at a flow rate of 120 mL/min. The product was transferred to a rotary evaporator and the mixture of trifluoroacetic acid and glacial acetic acid was removed. The crude product was dissolved in potassium dihydrogen phosphate buffer (0.06 mol/L, 2.5 mL) and purified by high-performance liquid chromatography (HPLC) using an Alltech Econosil C-18 column (10 μm, 250 × 25 mm) (Alltech Associates, Deerfield, IL, U.S.A.) with an eluant of 4.5% tetrahydrofuran and 0.15% trifluoroacetic acid in water at a flow rate of 10 mL/min. The product was detected with a UV detector set at 254 nm and a radioactivity detector. Two fractions eluting at 32 and 38 minutes were collected and 19F nuclear magnetic resonance (NMR) spectra were recorded. The fractions were determined to be the 2-fluoro and 6-fluoro isomers. The 6FMT sample for injection was collected from the HPLC, evaporated to dryness, and reconstituted with saline. The 19F NMR on this fraction showed less than 5% of the 2-fluoro isomer to be present. The specific activity of both 6FDOPA and 6FMT were 250 to 500 mCi/mmol.
Tomographic imaging
Twelve monkeys (Macaca mulata and Macaca fascicularis) were included in the comparison. Half the animals were normal controls and half the animals had been MPTP-lesioned several months or years before the PET studies according to already reported procedures. Each animal received, on separate occasions within a 4-month period (2.23 ± 1.7 months), two PET scans, one with 6FDOPA and one with 6FMT in random order. Before the PET studies, the monkey was pretreated with ketamine (10 mg/kg intramuscularly) and atropine (0.05 mg/kg intramuscularly) followed by anesthesia with sodium pentobarbital (10 mg/kg intravenously). The monkey was intubated, and intravenous and intraarterial catheters were inserted for radioligand administration and blood sampling. For the remainder of the PET study, the animal was lightly maintained on an isoflurane-oxygen mixture. All procedures were approved by the Committee on Animal Care of the University of British Columbia.
The monkey was placed prone in a stereotaxic frame and coronal slices of the brain were acquired with a Siemens ECAT 953B-31 scanner (CTI/Siemens, Knoxville, TN, U.S.A.) with an in-plane spatial resolution of 6 mm full width at half maximum, 4.5- to 5-mm slice thickness full width at half maximum, and a 3.4-mm slice interval.
For both the 6FDOPA and 6FMT studies, most animals received the peripheral decarboxylase inhibitor carbidopa (5 mg/kg intraperitoneally) 40 to 100 minutes before tracer injection. Because 6FMT is very sensitive to amino acid decarboxylation, the timing of carbidopa administration was kept constant for each animal, e.g., if carbidopa was administered 80 minutes before injection in the first study, it was given 80 minutes before tracer injection in the second study. The animal was injected intravenously with 3 to 5 mCi (adjusted to 10 mL with saline and injected for 1 minute with a Harvard injection pump) of 6FDOPA (4.9 ± 0.43 mCi) or 6FMT (4.7 ± 0.8 mCi). For both scans, the PET protocol consisted of a sequence of six 0.5-minute, two 1-minute, and one 5-minute scans followed by a succession of 10-minute scans for a total duration of 200 minutes. The two-dimensional acquisition was interrupted between 120 and 140 minutes to allow the acquisition of a three-dimensional scan to improve the identification of brain structures.
Analysis of peripheral metabolism
Arterial blood samples were drawn at multiple time points during the study to obtain the plasma input function. The main metabolite analysis was performed on plasma samples obtained at 2, 5, 10, 15, 30, 60, 90, 120, 150, 180, and 200 minutes using an alumina extraction method with anion/cation exchange columns, as previously described (McLellan et al., 1991). With this method, the cation and anion columns capture the positively and negatively charged metabolites, i.e., for 6FDOPA: FDA, 6-fluoro-3-methoxytyramine, 6-fluoro-homovanillic acid (FHVA), 6-fluoro-dihydroxyphenylacetic acid (FDOPAC), and sulfated conjugates; for 6FMT: FMA, FPAC, and sulfated conjugates. 6-[18F]Fluoro-L-dopa and its main metabolite 3OMFDOPA, both not retained by the columns, are then separated by alumina extraction. 6-[18F]Fluoro-L-m-tyrosine is also not retained by the columns and is contained in the supernatant. Thus, plasma radioactivity can be separated into 3 main groups for 6FDOPA (6FDOPA, 3OMFDOPA, and other metabolites) and into 2 groups for 6FMT (6FMT and metabolites). The plasma metabolite data were expressed as the percentage of total plasma activity.
In addition, for 6FMT, plasma samples from two animals at 5, 15, 30, 60, 120, 150, and 180 minutes after injection were subjected to both the anion/cation exchange and an HPLC system in parallel to (1) compare our findings with already published metabolic profiles and (2) verify the accuracy of our plasma metabolite results with the exchange columns as compared with the HPLC system. The procedures and the system used for HPLC analysis of 6FMT are the same as those described for HPLC analysis of 6FDOPA (Chan et al., 1992).
Data analysis
For each monkey, circular regions of interest (ROI) were placed over the left and right striatum (caudate and putamen) on four consecutive slices (ROI: 37 pixels; pixel size: 4 mm2). Twelve ROI (9 pixels each) were positioned over an area of nonspecific 18F accumulation in the occipital cortex in two consecutive slices. Time-activity curves were obtained for each ROI and averaged for left striatum, right striatum, and occipital cortex. Among the MPTP-treated monkeys, four animals had received intravenous or subcutaneous injections of the toxin, inducing bilateral lesions of the DA nigrostriatal pathways. The other two animals had received unilateral intracarotid injections leading mainly to asymmetrical striatal lesions (i.e., severe DA depletion on the MPTP-injected side and mild depletion on the uninjected side). Although a paired t test showed there was no significant difference between the left and right striatal values in the normal and bilaterally lesioned animals, we used data from the right striatum alone from the normal and bilaterally lesioned animals. However, we elected to report both the left and right striata of the asymmetrically lesioned monkeys in the MPTP group to show the effects of both a severe lesion (right) and a minimal exposure to neurotoxin (left). The use of both the left and right striatal data of the unilaterally lesioned animal in the MPTP did not significantly change the statistical significance of the results as compared with using only the right side of the unilaterally lesioned monkey in the statistical comparison. Thus, for all the analyses of the cerebral data, the normal group shown is composed of six striatal values and the MPTP group of eight striatal values.
The uptake rate constant Ki was calculated for each study by conventional graphical analysis of the data acquired between 30 and 120 minutes using the metabolite-corrected arterial plasma as the input function (Patlak and Blasberg, 1985; Martin et al., 1989). The first-order rate constant kloss that expresses the reversibility of tracer (6FDOPA or 6FMT) trapping, or loss of radioactivity out of the striatum, was calculated using an extended graphical analysis of the full time courses acquired between 30 and 200 minutes (Patlak and Blasberg, 1985; Holden et al., 1997). Details of the fitting method and its theoretical basis were described previously (Holden et al., 1997). The ratio kloss/Ki was calculated as the index of effective DA turnover (Doudet et al., 1997b, 1998). The influx rate constant Kc using the occipital activity as the input function was calculated from 30 to 120 minutes after injection (Brooks et al., 1990).
For each tracer, the normal and MPTP-treated group were compared using unpaired Student's t tests, with Welch's correlation for unequal variance when appropriate. To compare the 6FDOPA and the 6FMT data to one another, we used both a parametric method, Pearson's correlation, and a nonparametric method, Spearman's rank order correlation.
Postmortem data
Postmortem results have been obtained on five of the animals included in the PET study, including two normal controls and three MPTP-treated monkeys. Among the lesioned animals, monkey DI had received right unilateral intracarotid injections of MPTP and displayed a severe symptomatology in the contralateral body side with loss of balance, rigidity, and no use at all of the contralateral arm and a mild symptomatology (lack of strength and fine motoricity) in the ipsilateral arm. The other two monkeys (RA and JI) had received subcutaneous MPTP injections. RA was initially severely affected bilaterally with pronounced hypokinesia and bradykinesia, rigidity, and loss of balance and coordination, but after recovery from the acute effects of MPTP, the monkey was able to care for himself, although he remained very slow and unsteady for all the months before death. After a low dose of MPTP, JI became very severely affected, and although he could slowly move by himself around his cage, he could not independently feed himself adequately; he was maintained for a few weeks with intravenous fluids and a liquid diet. Levodopa therapy was attempted early on but proved unsuccessful (providing short improvement of the akinesia but severe dyskinesia) and was not continued. When it was determined that there would be no further behavioral recovery, the animal was scanned with 6FDOPA and 6FMT within a few days and killed.
The monkeys were killed by a pentobarbital overdose. After rapid removal of the brain, fresh samples of the striatum were dissected and stored at −70°C until assay. The concentrations of DA and its metabolites, HVA, DOPAC, and 3-methoxytyramine were determined by HPLC using a previously described technique (Pate et al., 1993). The tissue concentrations of catecholamines were expressed as micrograms per milligram of protein. Two measurements were considered from the postmortem data: the striatal concentration of DA and the ratio of DA metabolites (HVA + DOPAC + 3-methoxytyramine)/DA because this ratio has often been used as an index of DA turnover. The postmortem data are given here for illustration but because of the small number of subjects, the comparisons of the 6FDOPA and 6FMT PET index of effective turnover, kloss/Ki, and the postmortem levels of DA and the DA metabolites/DA ratio are to be interpreted with caution.
RESULTS
The plasma metabolic profiles for 6FMT and 6FDOPA after pretreatment with carbidopa are shown in Fig. 1. The ratio of 6FMT to its metabolites remained high during the course of the study, whereas this ratio decreased dramatically within 20 minutes of 6FDOPA injection. In the two animals in which plasma metabolite analysis was performed using HPLC and the anion/cation exchange system, the fractions of plasma 6FMT obtained by the two methods were comparable during the course of the 3-hour study: there was less than 9% (mean ± SD, 5% ± 2%) difference between the fractions of plasma 6FMT measured by the two methods. An example of HPLC data from one animal after 6FMT is shown in Fig. 2.
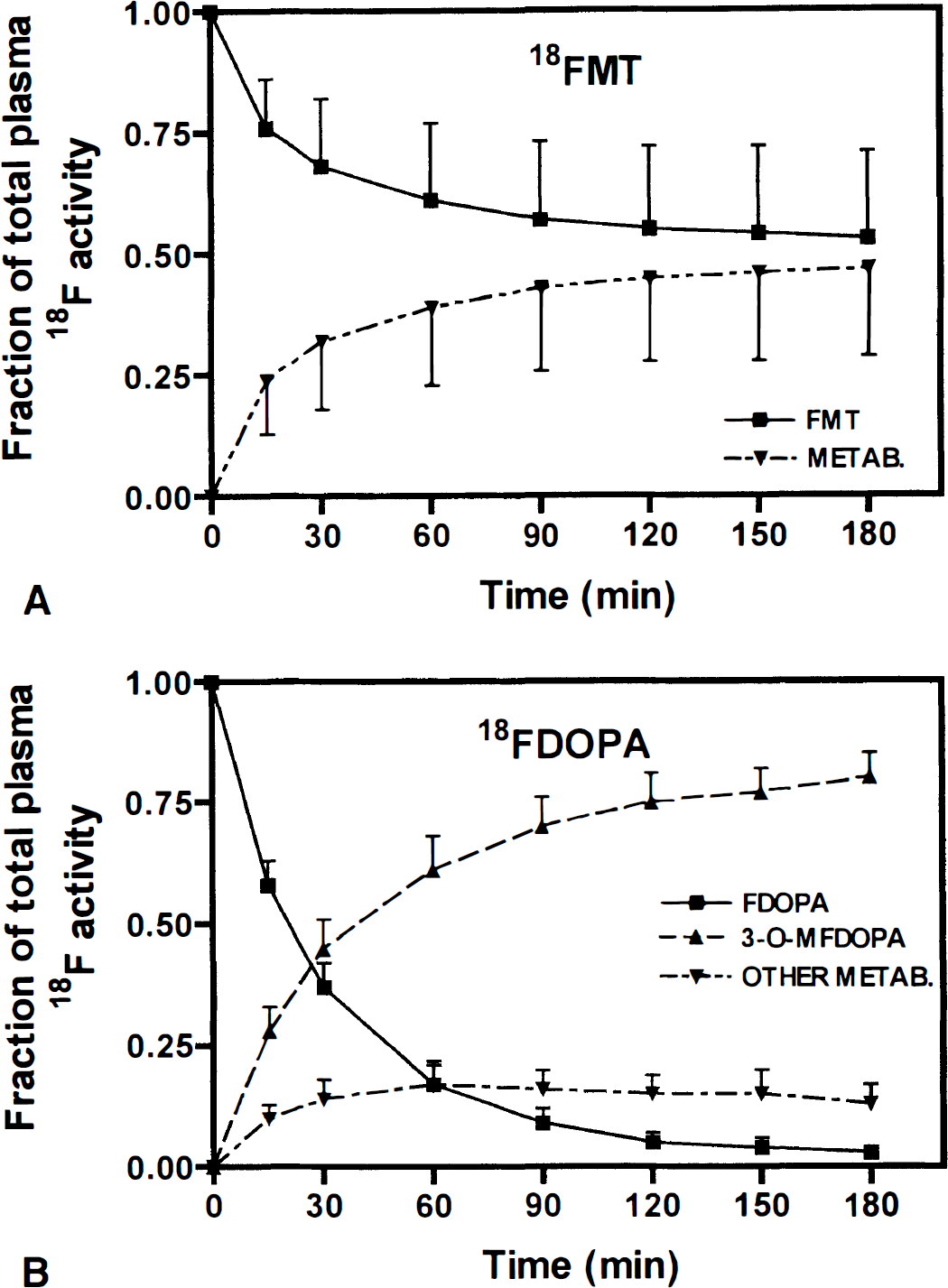
Fractions of the total plasma 18F activity of unchanged fluoro-L-m-tyrosine (FMT)
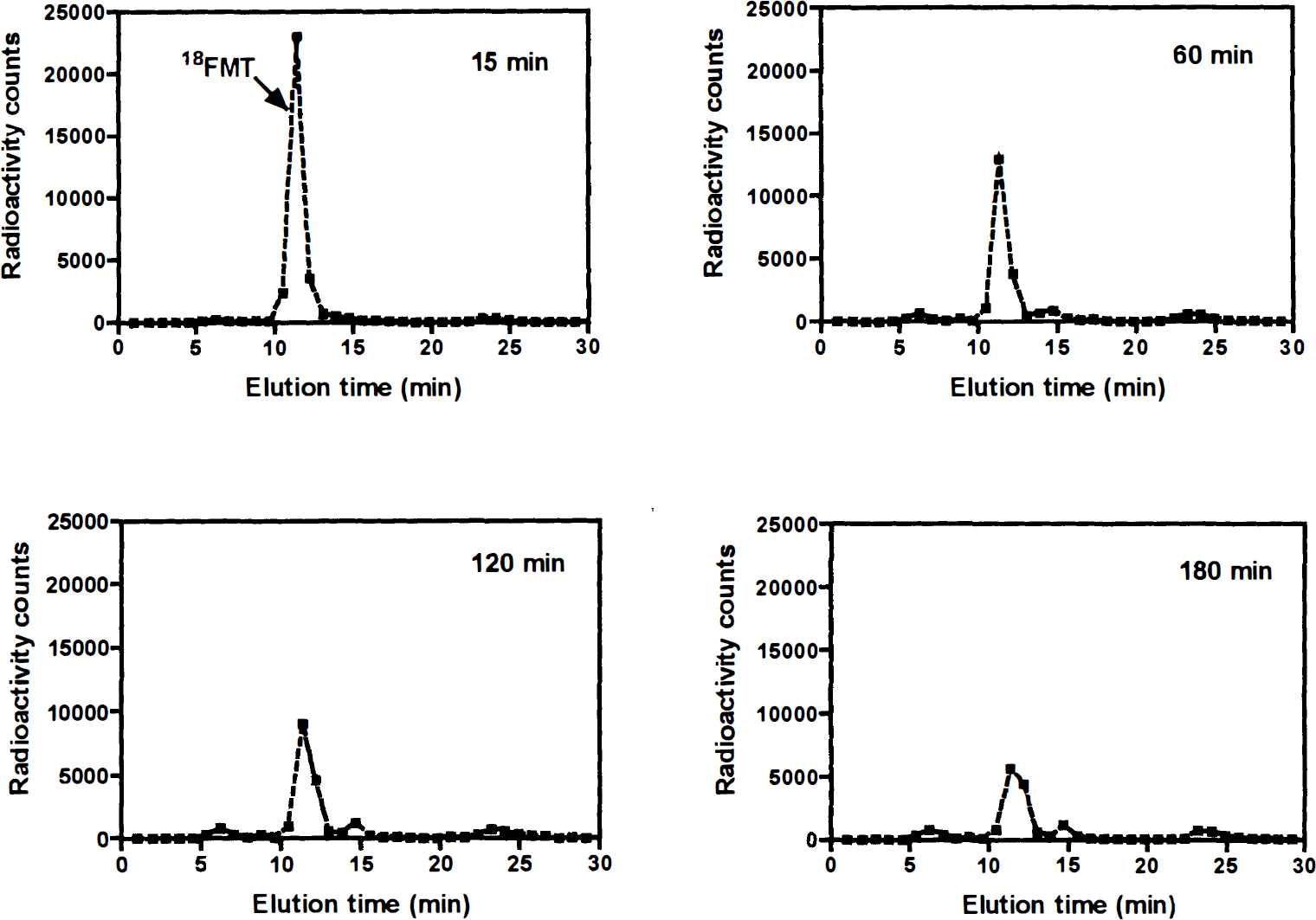
High-performance liquid chromatograms of arterial plasma samples at different time points after 18FMT injection showing unchanged FMT and metabolites using a triple coincidence detector. Fluoro-L-m-tyrosine was eluted at approximately 11.5 minutes and was identified with intact 18FMT.
The brain data are summarized in Table 1 and Figs. 3, 4 and 5. For both 6FMT and 6FDOPA, the uptake rate Ki and the influx rate constant Kc were significantly decreased in the MPTP-treated monkeys compared with normal (unpaired t tests: 6FDOPA Ki, P = 0.0003; 6FMT Ki, P = 0.0005; 6FDOPA Kc, P = 0.0024; 6FMT Kc, P = 0.0067). The kloss and the DA turnover were significantly increased in the MPTP-lesioned striata compared with normal after 6FDOPA injection (unpaired t tests, respectively, P = 0.0025 and P = 0.005) but there was no significant difference in either measurement after 6FMT injection (unpaired t tests, respectively, P = 0.97 and P = 0.24). Fig. 3 shows the spread of the data for the 6FDOPA and 6FMT scans. Fig. 4 shows the 6FDOPA and 6FMT data in the same unilateral MPTP-treated animal, on top (panels A and C) using the routine Patlak analysis assuming kloss = 0, and on the bottom (panels B and D) using our extended fitting procedure.
Striatal kinetic data
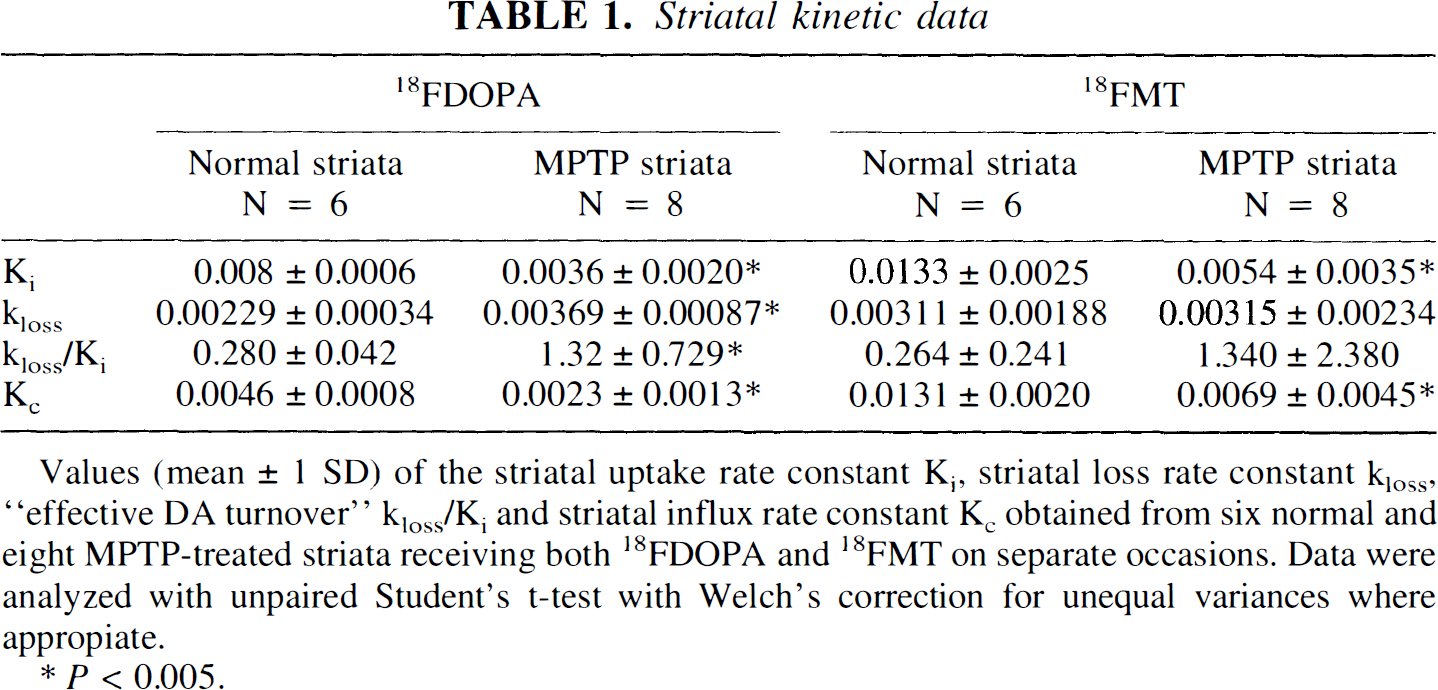
Values (mean ± 1 SD) of the striatal uptake rate constant Ki, striatal loss rate constant Kloss, “effective DA turnover” kloss/Ki and striatal influx rate constant Kc obtained from six normal and eight MPTP-treated striata receiving both 18FDOPA and 18FMT on separate occasions. Data were analyzed with unpaired Student's t-test with Welch's correction for unequal variances where appropiate.
p<0.005.
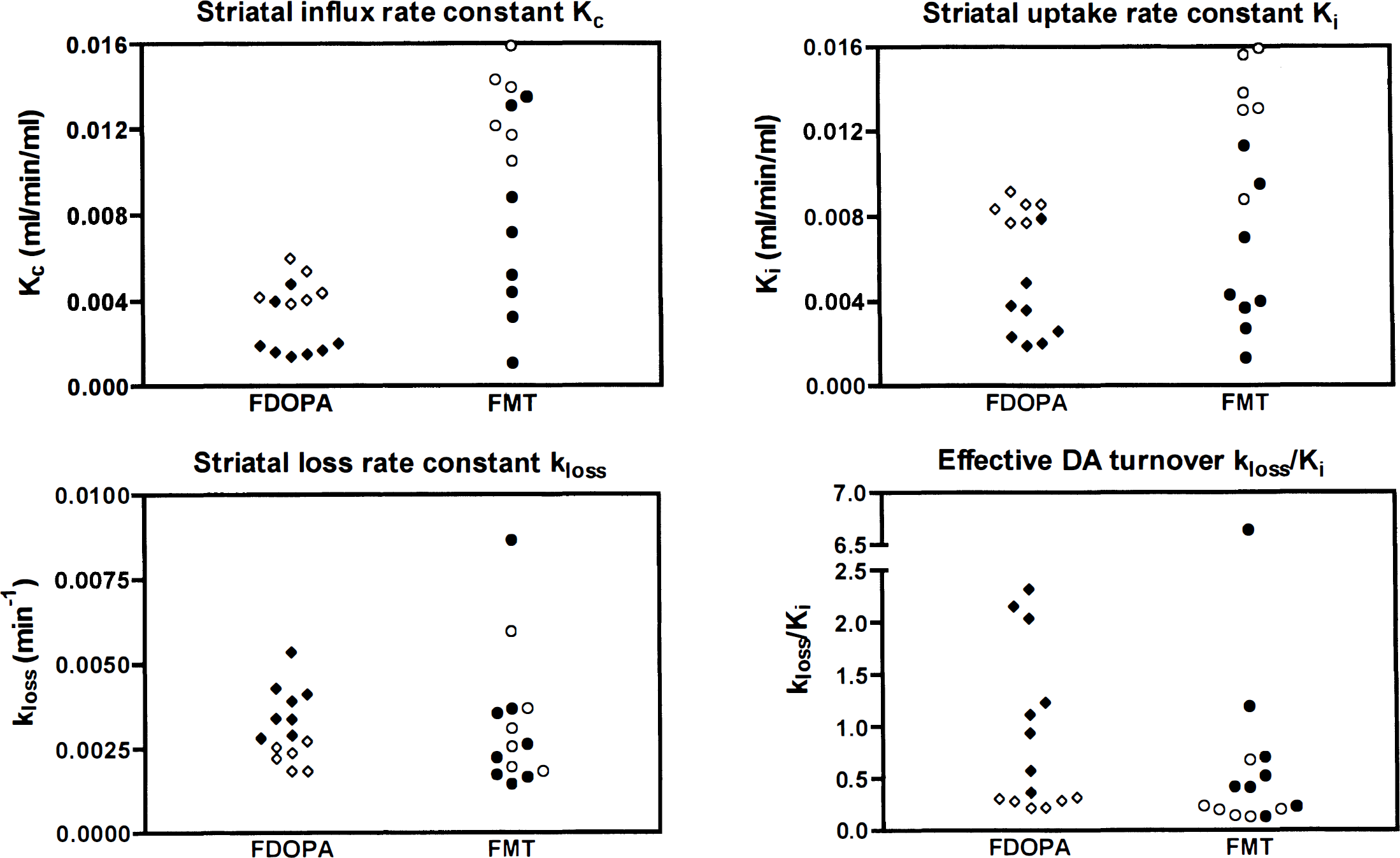
Comparison of the 18FDOPA and 18FMT scans by the spread of striatal influx rate constant Kc, striatal uptake rate constant Ki, striatal loss rate constant kloss, and index of effective DA turnover kloss/Ki in six normal striata (◊ for FDOPA and ○ for FMT) and eight MPTP-exposed striata (⧫ for FDOPA and • for FMT) obtained with both tracers on separate occasions.
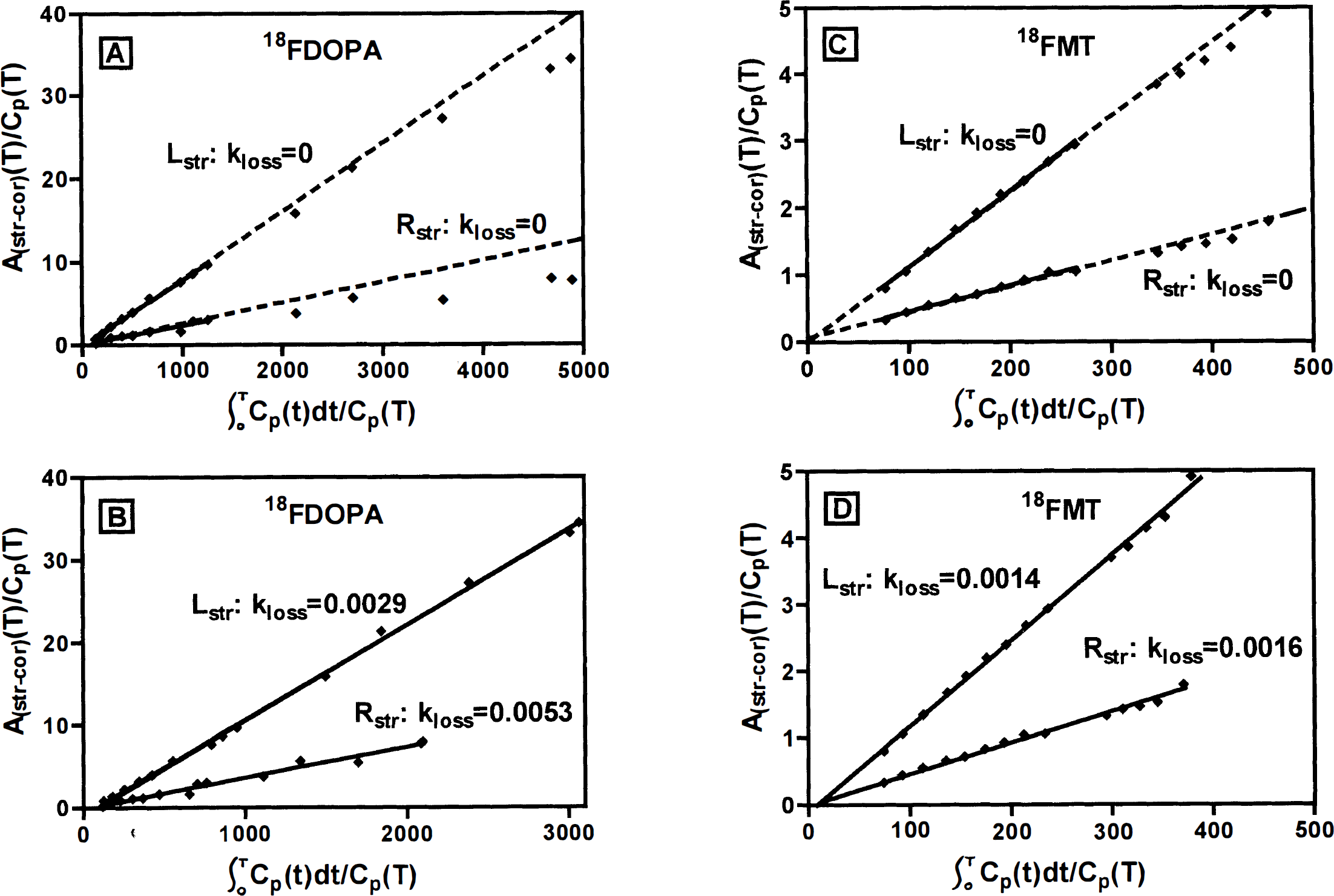
Analysis of brain data of 6FDOPA and 6FMT obtained from a single unilateral 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) -treated monkey using the Patlak analysis assuming kloss = 0 (
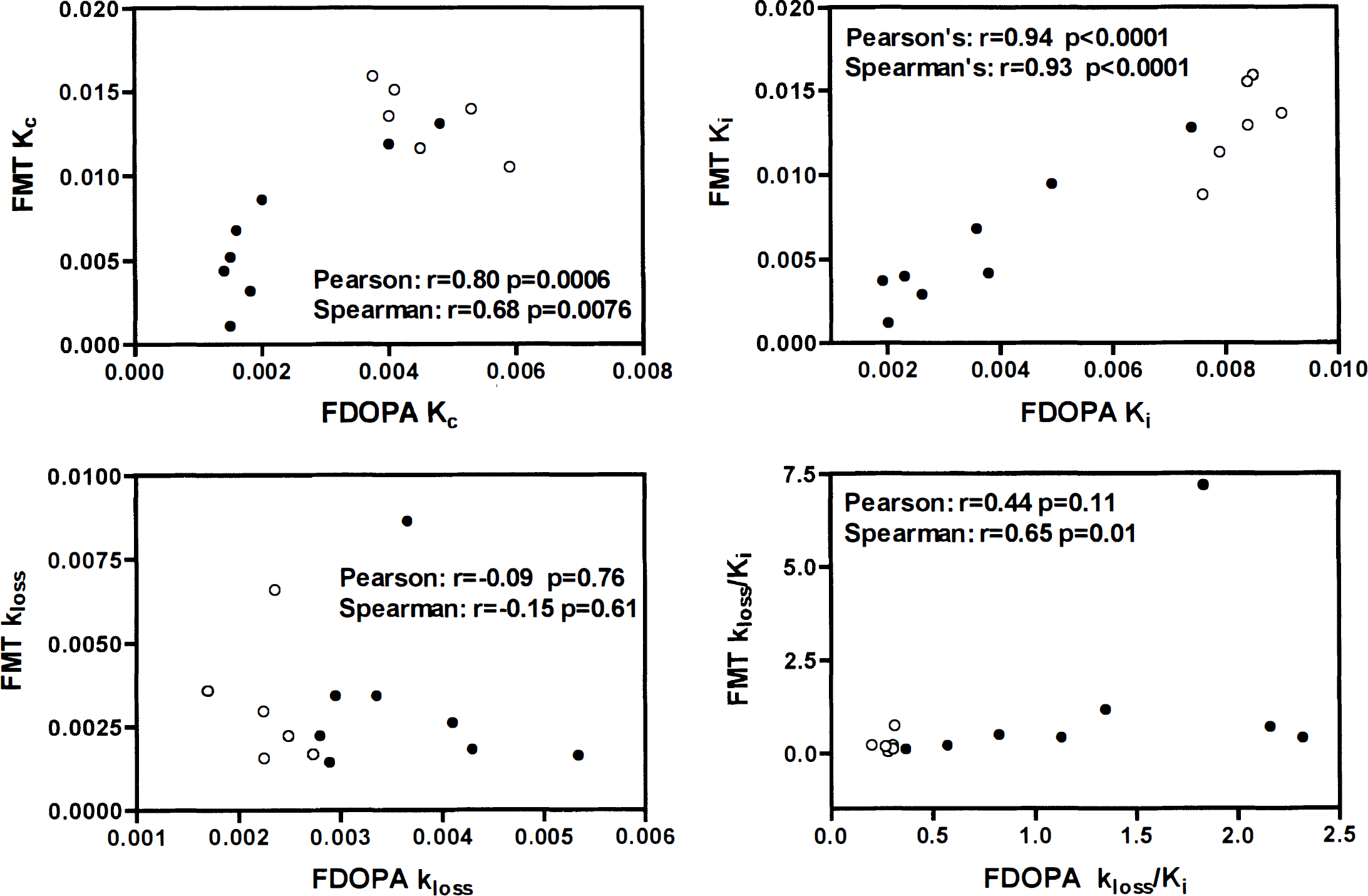
Influx rate constant Kc, uptake rate constant Ki, loss rate constant kloss, and effective turnover kloss/Ki in six normal (○) and eight MPTP-treated striata (•) with 6FDOPA and 6FMT. Each graph shows the correlation coefficient r and the P value obtained from both the Pearson's linear correlation and the Spearman's rank order correlation.
There were also significant correlations (Pearson's and Spearman's) between the overall 6FDOPA and 6FMT Ki (P < 0.0001) and Kc (P < 0.008) but no significant correlation between the 6FDOPA and 6FMT kloss (P > 0.6). There was no significant correlation between the 6FDOPA and 6FMT turnover index kloss/Ki with Pearson's correlation (P = 0.11) but a significant correlation using Spearman's correlation (P = 0.01), suggesting that if a correlation exists between the two sets of data, it is not linear. Fig. 5 shows the distribution of the 6FDOPA and 6FMT data and the r and P values associated with each analysis.
Compared with normals, the postmortem concentrations of DA and its metabolites in the MPTP-exposed striata were markedly reduced (respectively, 78% and 69%). The ratio DA metabolites/DA, which is often used as an index of DA turnover, was increased in two severely lesioned striata. There was a significant correlation between the postmortem DA turnover index (DA metabolites/DA) and the FDOPA effective turnover (r = 0.85, P = 0.031). There was no correlation with the 6FMT effective turnover index (r = −0.22, P = 0.68). However, these correlations are shown for information only and must be viewed with caution as we had postmortem data on only a very small number of animals with values at opposite ends of the range (i.e., normal or severely lesioned).
DISCUSSION
For years 6FDOPA has been used as the primary tracer of DA presynaptic integrity. But recently, L-m-tyrosine analogs, and in particular 6FMT, have been suggested as alternatives for 6FDOPA (DeJesus and Mukherjee, 1988). Both tracers are substrates for central decarboxylation processes in the DA terminals; both their decarboxylation products, FMA and FDA, are substrates for catabolism by MAO. In addition, 6FDOPA is a good substrate for peripheral and central COMT, and the peripheral production of significant concentrations of 3OMFDOPA, an inert amino acid that can cross the blood-brain barrier, represents a serious impediment to the accurate quantification of 6FDOPA kinetics in the brain. In this study, in an effort to evaluate the respective advantages of 6FDOPA and 6FMT, we scanned two groups of monkeys, normal and age-matched animals with lesions of the DA nigrostriatal pathway, using both tracers and compared them over a wide range of measurements.
Analysis of 6FMT plasma metabolites gave results that are consistent with a previous report by Nahmias et al. (1995) in human subjects receiving carbidopa and by Jordan et al. (1997) in monkeys with incomplete AAAD inhibition with benserazide but differ from that of DeJesus et al. (1997) in monkeys without carbidopa pretreatment. This discrepancy is likely because of the lack of peripheral AAAD inhibition. Indeed, the metabolic profile of the one animal that did not receive carbidopa was similar to that shown in DeJesus et al. (1997) (i.e., about 20% 6FMT at 60 minutes and 10% at 120 minutes). Peripherally, 6FMT presents the advantage of a simpler metabolic profile, with charged peripheral metabolites that do not cross the blood-brain barrier, reduced metabolism, and a single input function to the brain. The lack of peripheral O-methylation also allows for a substantially higher input function to the brain for an equivalent injected dose, better counting statistics in both plasma and brain, and with a lower nonspecific background, increased contrast in the tomographic images.
This increased contrast between specific and nonspecific accumulation allows for better identification of small structures: for example, in our animals, it was possible to identify visually in the images an area of accumulation in the brainstem at the level of the substantia nigra and ventral tegmental area. Although this was not the purpose of our study and we did not report the data, this fact deserves mention as it suggests that, in 6FMT scans in human subjects, these areas, which contain the DA cells bodies of origin of the nigrostriatal and mesocortical pathways, as well as their projection areas could be identified and studied. In addition, the uptake rate and influx rate constants for both tracers are tightly correlated and separate normal from diseased state equally efficiently. However, as can be seen in Fig. 3, there appears to be somewhat more variance in the 6FMT data. This may be caused in part by the increased variability in the input function (either metabolite-corrected plasma activity or cortical activity) produced by the exquisite sensitivity of 6FMT to peripheral decarboxylation and the time of carbidopa administration before 6FMT injection. More extensive test-retest reproducibility studies need to be performed to address this issue in greater detail. Nevertheless, for routine clinical studies, 6FMT appears to be a very attractive tracer. In addition, although 6FMT in this study was produced using the direct radiofluorination of L-m-tyrosine, which required HPLC separation of product isomers, a more region-selective synthesis using a stannylated precursor, similar to that for 6FDOPA, can now be used (Namavari et al., 1993) that would simplify the production of 6FMT for routine studies.
However, if one is interested in investigating further the function of the DA system in vivo, 6FMT may not be as good a tracer as 6FDOPA. In this study, it was not possible to measure meaningfully the rate of reversibility of 6FMT trapping. Similarly, a meaningful index of effective DA turnover as evaluated through analysis of the effective turnover of a fluorinated analog could not be obtained. In our small sample of subjects, 6FDOPA was able to separate significantly the normal controls from the MPTP-treated striata on the basis of the rate of loss of striatal radioactivity and the index of effective turnover whereas nonsignificant results were obtained with 6FMT. In support, although we only had a very small sample of postmortem data, and thus should consider the results of the correlations with caution, it is interesting to note that the index of postmortem turnover (DA metabolites/DA) correlated with the index of effective turnover of 6FDOPA but not that of 6FMT.
Like DA and FDA, FMA is a substrate for MAO metabolism but has a 15-fold lower affinity for the vesicular DA transporter and the synaptic DA transporter (Endres et al., 1997). Thus, FMA is not protected in vesicles from MAO oxidation, which is consistent with the finding that most of the striatal radioactivity in the primate brain is caused by FPAC, the FMA decarboxylation product (Jordan et al., 1997; Wong et al., 1987). Neither FMA nor FPAC is a substrate for COMT. In a previous study of the effect of COMT inhibition, we showed that in the absence of COMT catabolism, the clearance of striatal radioactivity (presumably through FDA metabolites after action of MAO and COMT) is significantly reduced (Doudet et al., 1997a, 1997b). Thus, one can hypothesize that in the absence of COMT catabolism, the clearance of FPAC is significantly slower than that of the metabolites of DA and its fluorinated analog (DOPAC and FDOPAC and ultimately HVA and FHVA).
It has been hypothesized that an increase in DA turnover is one of the compensatory mechanisms used by the DA nigrostriatal system in response to degeneration of nigral neurons. Microdialysis and postmortem studies suggest an increased turnover in rodents exposed to the neurotoxins MPTP and 6-hydroxydopamine. In a much larger group of rhesus and cynomolgus monkeys than used here, we observed in vivo an increase in DA effective turnover in both asymptomatic and symptomatic MPTP-treated animals compared with controls (Doudet et al., 1998). A similar observation was made by Barrio et al. (1990) using kinetic modeling of 6FDOPA PET data in rhesus monkeys. These authors suggested that DA or FDA of exogenous origin was stored in a slow turnover pool in the nigrostriatal DA terminals and that the lack of endogenous DA after nigrostriatal degeneration led to an increase in the use of this pool. We recently showed a selective effect of a central pharmacologic manipulation (COMT inhibition) on the effective turnover of DA in MPTP-treated monkeys but not in normal animals at a clinically relevant dose (Doudet et al., 1997a). Thus, the index of effective DA turnover could prove an attractive index to evaluate the effects of age, injury, or pharmacologic manipulations on the DA system in vivo. The lack of sensitivity of 6FMT is an impediment for its use as a marker of DA effective turnover.
In conclusion, for short, routine clinical PET studies, 6FMT is a good, highly specific tracer for studying the activity of the aromatic acid decarboxylase enzyme and may give a useful measure of DA presynaptic integrity. However, in studies aimed at assessing the complete turnover in normal and patient populations in vivo, 6FDOPA remains the tracer of choice.
Footnotes
Acknowledgements
The authors thank Merck, Sharp and Dohme, Canada for their gift of the carbidopa. The authors also thank the staff of the UBC/TRIUMF PET program for their assistance and contribution to this work, especially Dr. M. J. Adam and Ms. Salma Jivan for tracer synthesis, and Ms. Carolyn English and Ms. Teresa Dobko for data acquisition and processing. The authors also thank Ms. Edith Kim Singh for the postmortem HPLC assays. Special thanks are due to Dr. J. Love and Mr. Mike Boyd and the personnel of the UBC Animal Care Facilities.
