Abstract
Expression of tissue plasminogen activator (tPA) substantially determines endothelial-dependent fibrinolysis. We used a blood-brain barrier (BBB) model to analyze regulation of brain capillary endothelial tPA and its inhibitor, plasminogen activator inhibitor-1 (PAI-1). This model consists of coculture of murine astrocytes with bovine brain capillary endothelial cells grown as capillary-like structures (CS); after 1 week, astrocytes become extensively associated with CS, and the BBB-associated enzyme γ-glutamyl transpeptidase is present. We measured tPA and PAI-1 mRNA and tPA activity in this model. Reverse transcription-polymerase chain reaction (RT-PCR) studies showed similar tPA and PAI-1 mRNA levels after 1 day mono-culture (endothelial cells only) versus astrocyte-endothelial coculture preparations. After 7 days (i.e., when elements of the BBB are present), astrocyte-endothelial cocultures (compared with endothelial mono-cultures) showed a 50.7% ± 27.1% (mean ± SD) reduction in tPA mRNA (P < 0.03) and a 183.3% ± 86.9% increase in PAI-1 mRNA expression (P < 0.02). Moreover, 7-day cocultures demonstrated reduced tPA activity compared with mono-cultures (14.6 ± 2.9 IU/mL versus 30.2 ± 7.7 IU/mL, P < 0.01); 1-day cocultures and mono-cultures had similar tPA activity. These findings demonstrate that astrocytes regulate brain capillary endothelial expression of tPA when elements of the BBB phenotype are present in this model. These data suggest an important role for astrocytes in the regulation of brain capillary endothelial fibrinolysis.
Endothelial cells play a critical role in the regulation of the fibrinolytic system by synthesis and secretion of tissue plasminogen activator (tPA) and its inhibitor, plasminogen activator inhibitor-1 (PAI-1). Tissue plasminogen activator proteolytically activates plasminogen to form plasmin, a protease critically involved in fibrinolysis (Collen and Lijnen, 1995). Plasminogen activator inhibitor-1 rapidly forms Table 1:1 complexes with tPA and functions as the most important plasma inhibitor of tPA (Wiman, 1995). Expression of tPA has been demonstrated in the brain microvasculature (Levin and del Zoppo, 1994; Zlokovic et al., 1995) and in cultured brain microvascular endothelium (Saksela et al., 1987; Kollros et al., 1994; Shatos et al., 1995); these findings suggest a role for brain endothelium in the regulation of fibrinolysis. Moreover, fibrinolytic drugs used in stroke models reduce infarct size in both thromboembolic and non-thrombotic occlusion of intracranial vessels (Zivin et al., 1985; del Zoppo et al., 1986). The importance of brain expression of tPA has been further emphasized by the demonstration that exogenous tPA can improve outcome in acute ischemic stroke (NINDS rt-PA Stroke Study Group, 1995). Nevertheless, little is known about the mechanisms regulating endothelial fibrinolysis within the brain.
Brain capillary endothelial cells are unique in that they demonstrate features of the blood-brain barrier (BBB), including tight junctions, increased electrical resistance, lack of pinocytic vesicles, and expression of highly selective transport systems to deliver essential nutrients across the BBB (e.g., GLUT-1) (Pardridge, 1984, 1991). The induction and maintenance of the endothelial BBB phenotype is thought to be largely under the regulatory control of perivascular astrocytes (DeBault and Cancilla, 1980; Stewart and Wiley, 1981; Tao-Cheng et al., 1987; Dehouck et al., 1990; Raub et al., 1992), whose processes ensheathe capillaries (Goldstein, 1988; Risau and Wolburg, 1990). Astrocytes also play a regulatory role in other endothelial functions, including upregulating endothelial expression of low-density lipoprotein receptor (Dehouck et al., 1994) and Na-K-Cl cotransporter (Sun et al., 1995). Moreover, endothelial cells expressing the BBB phenotype exhibit enhanced expression of the GLUT-1 glucose transporter (Hayashi et al., 1997) and downregulation of MECA-32 (Hallman et al., 1995) and thrombomodulin (Tran et al., 1996). Several lines of investigation show limited expression of tPA by brain microvascular endothelium (Levin and del Zoppo, 1994; Shatos et al., 1995, Grau et al., 1997). These observations suggest brain-specific tPA expression and led us to hypothesize that astrocytes regulate brain capillary endothelial expression of genes important in the fibrinolytic pathway.
MATERIALS AND METHODS
Cell culture
We isolated bovine brain capillary endothelial cells by modification of techniques of Carson and Haudenschild (1986). After transportation at 4°C from a local meat processing company, bovine brains were rinsed in a medium containing Dulbecco's modified Eagle's medium (DMEM), 1% bovine albumin serum, 100 U/mL penicillin, 100 mg/mL amphotericin B, and 2 mmol/L
Neonatal mouse astrocytes were isolated according to the methods of McCarthy and DeVellis (1980), and experiments were performed within institutional guidelines. Briefly, cerebral hemispheres were removed from 1- to 2-day-old Swiss-Webster pups, cleaned of meninges and choroid plexus, and serially sieved through meshes of 230 and 140 µm. The filtrate was centrifuged at 200g for 5 minutes at room temperatures and resuspended in DMEM supplemented with 10% fetal bovine serum (Hyclone Labs, Logan, UT, U.S.A.), 100 U/mL penicillin, 100 mg/mL streptomycin, and 2 mmol/L
Immunocytochemistry
Methanol-fixed frozen slides were rehydrated with phosphate-buffered saline (PBS, pH 7.4), incubated with 0.3% H2O2 for 5 minutes, and washed with PBS. After a 15-minute incubation with 5% normal goat serum, the slides were incubated for 1 hour at room temperature with rabbit anti-bovine GFAP antibody (Dako, Carpinteria, CA, U.S.A.) or rabbit antihuman vWF antibody (Dako) at a dilution of 1:200 and 1:100, respectively. Control slides were incubated with PBS in place of the primary antibody. Slides were then incubated with bio-tinylated goat anti-rabbit immunoglobulin (Vector, Burlingame, CA, U.S.A.) at 1:200 dilution for 30 minutes, avidin-biotin peroxidase complex (Vector) for 15 minutes, and amino-ethyl carbazole for 10 minutes. Slides were counterstained in Mayer's hematoxylin and mounted in glycerol.
Blood-brain barrier model
We prepared our BBB model as previously described (Minakawa et al., 1991; Tran et al., 1996). To prepare capillary-like structures (CS) we first coated two 2- ***× 2-cm chamber Lab-Tek glass slides with 1% gelatin (Sigma) and then added 4 × 104 endothelial cells/chamber in 1.0 mL DMEM with 2.5% equine serum. After incubation for 24 hours, the cells were washed with cold PBS, then 0.4 mL of a collagen solution (pH 7.4) containing 80% type 1 collagen (Vitrogen, Celtrix Lab, Palo Alto, CA, U.S.A.), 10% 10 × minimum essential medium (Gibco, Gaithersburg, MD, U.S.A.), and 10% 0.1 mol/L NaOH was added to the subconfluent monolayer and excess Vitrogen solution was then aspirated. The slides were incubated for 10 minutes at 37°C and culture medium (2.5% equine serum-supplemented DMEM) was then added to the slides. Endothelial cells elongated and formed CS within 24 hours (Fig. 1). We established astrocyte-endothelial or liver-endothelial cocultures 3 days after the addition of the second collagen layer. Before the addition of astrocytes (4 × 104 cells/chamber) or liver cells (4 × 104 cells/chamber) to the capillary preparations, serum-supplemented media from astrocyte or liver preparations were removed, the cells were treated with trypsin-EDTA, and resuspended in endothelial culture medium. One and seven days after the addition of astrocytes, the cultures were fixed with 80% ethanol for 10 minutes and stained for GFAP to demonstrate association with CS. After 1 day of coculture, only 35% of the astrocytes were associated with CS. After 7 days, nearly all of the astrocytes were associated with CS, and their processes frequently enveloped the entire CS (Fig. 2). Thus, astrocyte-endothelial cell cocultures exhibited morphologic features of the BBB after 7 days of coculture. We performed histochemical staining for the presence of γ-glutamyl transpeptidase (GGTP), a putative marker of the BBB, in coculture and mono-culture preparations (Tran et al., 1996). At the end of 1 day in coculture GGTP activity was not detectable. However, after 7 days extensive GGTP activity was detectable along the length of CS. γ-Glutamyl transpeptidase activity was neither detectable when CS were cultured without astrocytes nor in astrocyte mono-cultures. Additional work has described tight junctions in this model (Minakawa et al., 1991).
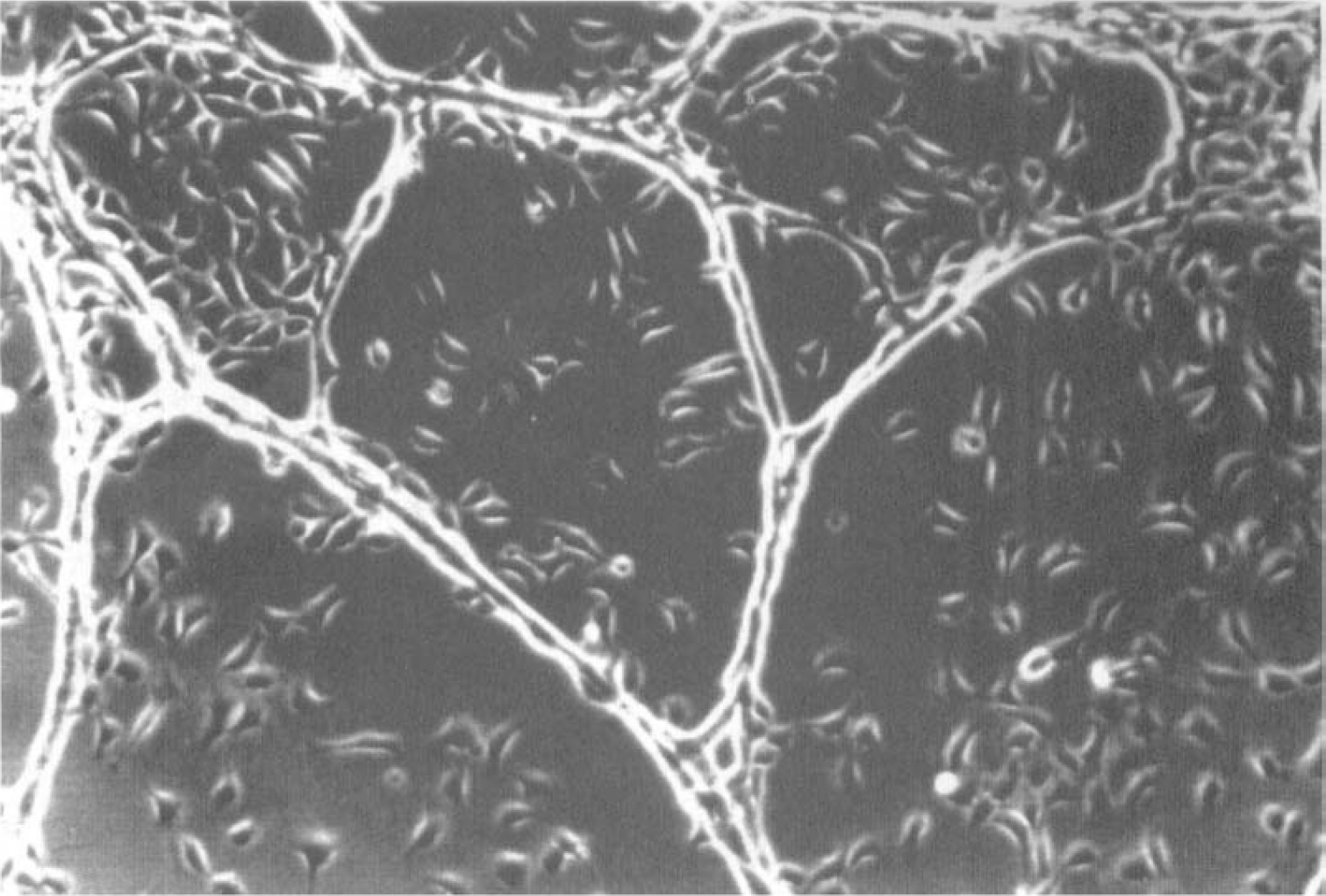
Phase contrast photomicrograph of capillary-like structures. Original magnification ×25.
Glial fibrillary acidic protein (GFAP) immunocytochemistry. Immunocytochemistry for GFAP, a marker for astrocytes, showed astrocytes associated with capillary-like structures at 7 days. Original magnification ×125.
We monitored and photographed (10 × magnification) cultures with an Olympus CK-2 phase-contrast microscope. The extent of CS formation for both mono-cultures and cocultures was determined by computer-assisted image analysis of photomicrographs using a Quantimet 970 Image Analysis System and the Quips software package (Cambridge Instruments Limited, Cambridge, U.K.). Photomicrographs from CS preparations were digitized using a video camera and stored as a 512-x 512-pixel matrix. The image was displayed as a combination of a gray image and a binary overlay, representing the detection region. An Amend (Cambridge Instruments Limited) computer algorithm was used to measure CS length. Digitized images of CS photomicrographs were calibrated against a digitized image of a 10-cm ruler. Quantifications were performed in duplicate.
Cells from coculture and mono-culture preparations were treated with trypsin and counted. Cytopreps (Shannon Inc., Pittsburgh, PA, U.S.A.) were prepared from coculture and mono-culture preparations. Slides were fixed in 80% ethanol and stained for vWF and GFAP. The number of endothelial cells in culture was derived from the cell count and the percentage of cells showing immunoreactivity for vWF. The relative composition of endothelial cells and astrocytes showed only minor differences for 1 versus 7 days coculture (Tran et al., 1996).
Conditioned-media experiments
We performed conditioned-media experiments according to the method of Maxwell et al. (1987). Briefly, astrocyte-conditioned media were prepared by first aspirating the growth media from these cultures, washing with PBS, and replacing media with 5% equine serum-supplemented media. Twenty-four and forty-eight hours later the astrocyte-conditioned media (ACM-24 and ACM-48, respectively) were collected, centrifuged to remove cellular debris, and stored at -80°C until use. Bovine brain endothelial cells were plated in 24-well tissue culture plates (40,000 cells/well) in 1 mL 5% equine serum-supplemented media. At confluence the cells were incubated for 24 hours with ACM-24, ACM-48, or nonconditioned 5% equine serum-supplemented media.
Plasminogen activator assay
We assayed cultured media from mono-culture and coculture preparations for total plasminogen activator activity by amidolytic assay (American Diagnostica, Greenwich, CT, U.S.A.). Amiloride (0.5 mmol/L, Sigma) was used to inhibit urokinase plasminogen activator (uPA) and to allow for the determination of net tPA activity (Vassalli and Belin, 1987).
Polymerase chain reaction
Because our culture preparations contained a relatively small number of cells, we used reverse transcription-polymerase chain reaction (RT-PCR) to measure the abundance of tPA mRNA transcripts. Total RNA was isolated with the Glassmax DNA Spin Cartridge Isolation System (Gibco BRL). Total RNA from each preparation was resuspended in 40 µL of diethyl pyrocarbonate-treated water. cDNA was synthesized from equal volumes of total RNA in a total volume of 20 µL. RNA was incubated in 5 µL of diethyl pyrocarbonate-treated water at 65°C for 3 minutes and quickly placed on ice. The RNA was then added to the transcription solution: 1.5 µmol/L oligo dT primers, 50 mmol/L Tris-HCl, 75 mmol/L KCl, 3 mmol/L MgCl2, 0.5 mmol/L dNTP, 1 U/µL RNAse inhibitor, and 13.3 U/µL avian myoblastosis virus reverse transcriptase. The reaction was carried out at 42°C for 1 hour and terminated at 52°C for 40 minutes. The cDNA was stored at -20°C until use.
Polymerase chain reaction primers (National Biosciences Inc., Plymouth, MN, U.S.A.) for bovine tPA (forward primer, BTPA-1: 5′ AAGGTTGCAGAAGAAGATGG 3′; and reverse primer, BTPA-2: 5′ GTGAGGCGGGTACCTCTCCTGGAA 3′) and PAI-1 (forward primer, BPAI-A: 5′ GCCTCCTAC-CAGCCCCAGTCT 3′; and reverse primer, BPAI-B: 5′ AATTCCAGGATGTCGTAGTAACGG 3′) were chosen from coding regions of the mRNA (Mimuro et al., 1989; Prendergast et al., 1990; Ravn et al., 1995; Rickles et al., 1988). To elucidate the contribution of endothelial cell transcription, we chose primers that were specific and selective for bovine endothelial tPA and PAI-1 mRNA (Fig. 3). Primers for tPA (BTPA-1 and BTPA-2) and PAI-1 (BPAI-A and BPAI-B) share 100% homology with sequences for bovine tPA and PAI-1 mRNA, respectively; these primers have limited homology with sequences for murine tPA (40% and 54%) and PAI-1 mRNA (57% and 75%, respectively) (Mimuro et al., 1989; Prendergast et al., 1990; Ravn et al., 1995; Rickles et al., 1988). β-Actin primers (Stratagene, LaJolla, CA; forward primer, 5′ TGAC-GGGGTCACCCACACTGTGCCCATCTA 3′; reverse primer, 5′ CTAGAAGCATTTGCGGTGGACGATGGAGGG 3′) were used to amplify β-actin mRNA as a housekeeping gene control. The PCR mixture contained 0.2 to 1.0 µg cDNA, 10 mmol/L Tris-HCl, pH 8.3, 50 mmol/L KCl, 0.1 mmol/L dNTP, 1.0 mmol/L MgCl2, 1.0 U Taq polymerase, and 0.5 µmol/L forward and reverse primers. Amplification was carried out in a DNA Thermal Cycler (Perkin-Elmer Corp., Norwalk, CT, U.S.A.): initial denaturation at 94°C, then each cycle consisted of 1 minute denaturation at 94°C, 1 minute annealing at 54°C, and 2 minutes extension at 72°C for 30 cycles. All RT-PCR assays were performed within the linear range of amplification. The PCR products were visualized by electrophoresis on a 2% agarose gel and stained with ethidium bromide. Negatives were prepared with a Polaroid camera (Polaroid Corp., Cambridge, MA, U.S.A.) and scanned by optical densitometry (Hoefer Instruments, San Francisco, CA). Optical densitometric (OD) measurements of tPA and PAI-1 levels were normalized to either endothelial cell counts cells (i.e., dividing mRNA concentration by cell count), or β-actin levels (i.e., dividing by β-actin OD measurements). Subsequent calculations of relative mRNA used the mean values of the control group (endothelial mono-culture or unstimulated endothelial monolayers) as 100%.
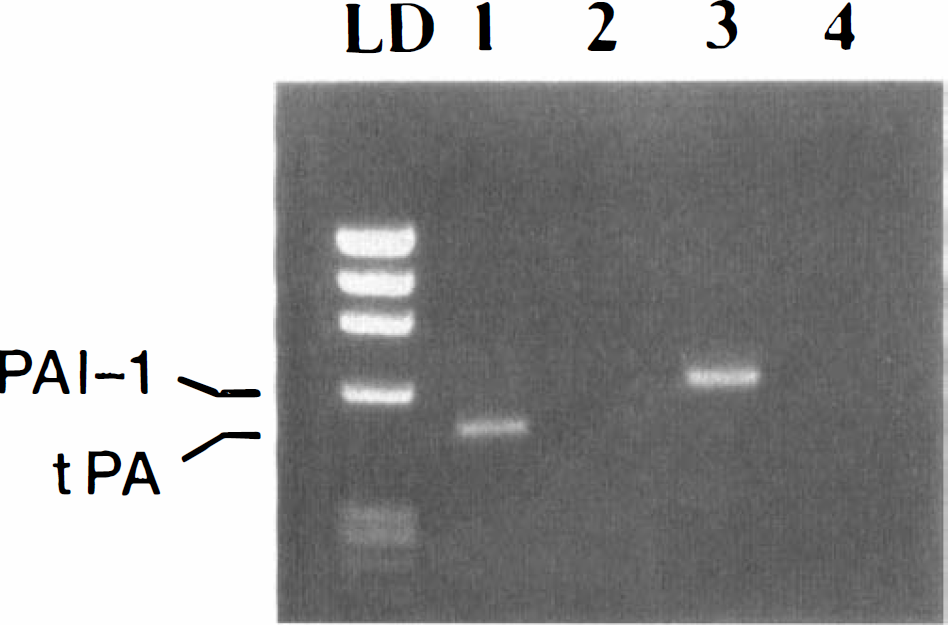
Polymerase chain reaction analysis of tissue plasminogen activator (tPA) mRNA expression. Reverse transcription-polymerase chain reaction showed amplification of tPA (lane 1) and plasminogen activator inhibitor-1 (PAI-1) (lane 3) from endothelial cells, and the lack of amplification of either tPA (lane 2) or PAI-1 (lane 4) from astrocytes. LD = kb marker.
We validated our RT-PCR results by comparing results of same samples (n = 27) analyzed by quantitative-competitive PCR (QC-PCR). The QC-PCR tubes contained all amplification reagents, a constant amount of target cDNA from each preparation, and serial dilutions of known concentrations of a competitor tPA cDNA template. The competitor tPA cDNA template was generated by RT-PCR (described above), using forward primer BTPA-3: 5′ AAGGTTGCAGAAGAAGATGGGA AGCACAACCACTGCA 3′ and reverse primer BTPA-2: 5′ GTGAGGCGGGTACCTCTCCTGGAA 3′. Both the wild-type tPA template (479 bp) and competitor tPA template (332 bp) contain primer binding sites for primers BTPA-1 and BTPA-2. The QC-PCR reaction mixture was coamplified, as described above. The target and competitor cDNA were separated by electrophoresis on a 2% agarose gel and visualized by ethidium bromide staining and UV transillumination. The OD readings of the target cDNA were multiplied by 332/479 to correct for differences in molecular weight. The OD measurements of the target and competitor cDNA were plotted as a function of competitor template concentration. The concentration of target cDNA was derived from the point at which the OD of the target and competitor cDNA were equal. The QC-PCR analyses of mRNA concentrations were normalized to β-actin levels. There was a high correlation between results from standard densitometric analyses of RT-PCR products and results by QC-PCR (r = 0.91, P < 0.001).
Statistical analysis
All data are expressed as means ± SD. For the conditioned-media experiments, statistical analysis was performed on pooled data from three independent experiments. Statistical comparisons between groups were performed using unpaired Student's t tests and Pearson's correlation coefficients. Differences were considered significant for P < 0.05.
RESULTS
We examined astrocyte effects on endothelial tPA transcription in our BBB model. There was no significant difference by RT-PCR in levels of tPA transcripts for astrocyte-endothelial cocultures versus endothelial mono-cultures after 1 day (Fig. 4). After 7 days, however, astrocyte-endothelial cocultures showed a 50.7% ± 27.1% (mean ± SD) reduction in tPA mRNA (P < 0.03) compared with endothelial mono-cultures. This reduction thus occurred when elements of the BBB were demonstrable in our model, i.e., when astrocytes extensively associated with CS and GGTP were detectable.
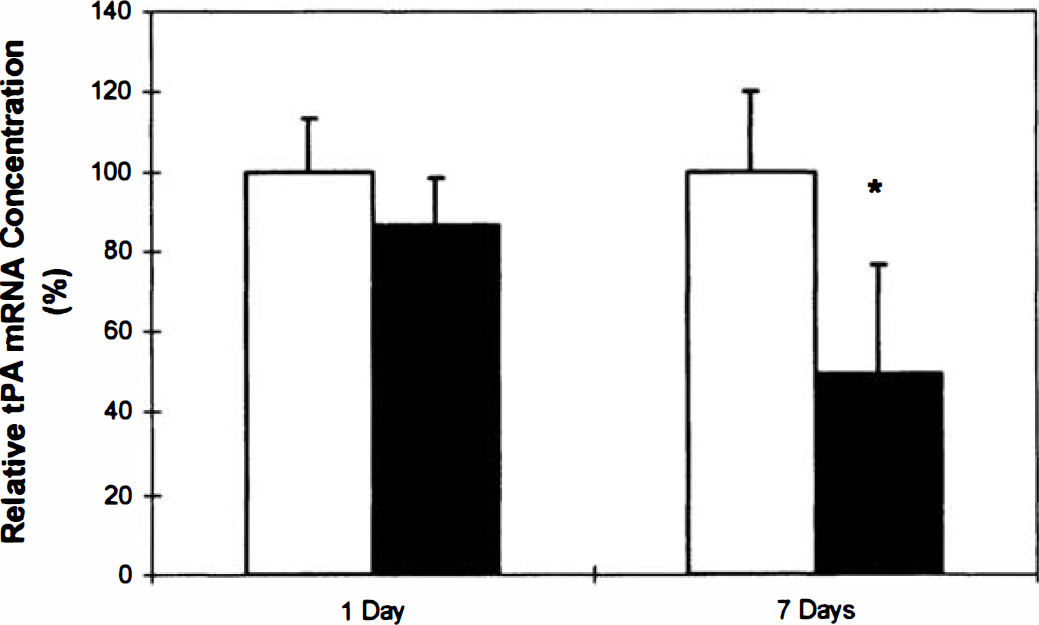
Densitometric analyses of polymerase chain reaction products. Reverse transcription-polymerase chain reaction analysis showed significant decrease in tissue plasminogen activator (tPA) mRNA levels in seven day astrocyte-endothelial cocultures compared with endothelial monocultures(* P < 0.03). Data are presented as mean ± SD. Data are from one experiment (performed in quadruplicate) and representative of three independent experiments. No significant differences were observed in tPA in 1-day cultures. (□) monoculture. (▪) co-culture.
To determine the specificity of the changes in tPA mRNA, we measured the levels of β-actin mRNA, a housekeeping gene. Astrocyte-endothelial cocultures showed no significant difference in β-actin mRNA compared with mono-cultures after 1 day (93.8% ± 5.4% of mono-cultures; P > 0.2) and 7 days (107.7% ± 20.8% of mono-cultures; P > 0.7). To investigate the cellular specificity of astrocyte regulation of endothelial tPA mRNA, we analyzed the expression of tPA mRNA in liver-endothelial cocultures. After 7 days, liver-endothelial cocultures exhibited no significant differences in levels of tPA mRNA (88.2% ± 30.0% of monocultures; P < 0.6).
We analyzed our data for a potential association between CS length and mRNA levels. There was no significant correlation between CS length and tPA mRNA concentration in mono-cultures (r = 0.41, P > 0.9) or cocultures (r = 0.43, P > 0.6). We then performed additional experiments using astrocyte-conditioned media in a nonangiogenic model to further assess a potential relationship between tPA mRNA regulation and angiogenesis. Bovine brain capillary endothelial cells were grown to confluence and incubated with media that had been conditioned by astrocytes for 24 (ACM-24) or 48 hours (ACM-48). Examination of tPA transcripts 24 hours after incubation with ACM-48 revealed a significant reduction (65.0% ± 15.0% of controls, P < 0.02) in endothelial expression of tPA mRNA as compared with endothelial cells incubated with nonconditioned media (Fig. 5). Thus, astrocyte-dependent tPA regulation was unrelated to the angiogenesis. Moreover, these studies suggested that this regulation was mediated in part by a soluble factor secreted by astrocytes.
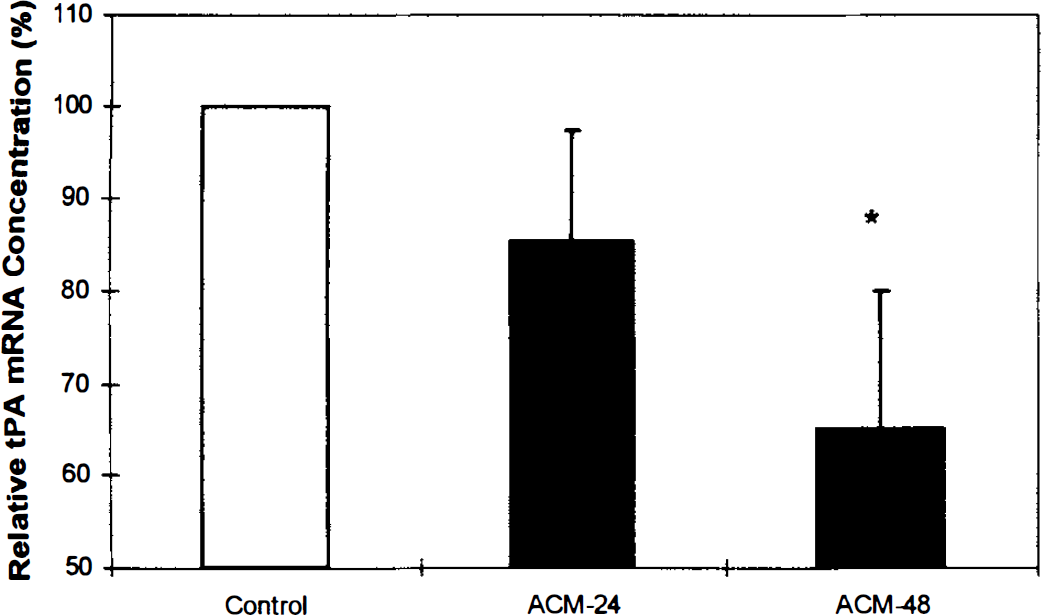
Tissue plasminogen activator (t-PA) mRNA expression in endothelial monolayer preparations. Reverse transcription polymerase chain reaction analysis of tPA transcripts after 24 hours incubation with media conditioned by astrocytes for 24 hours (ACM-24) and 48 hours (ACM-48) revealed reduction in endothelial expression of tPA mRNA as compared to endothelial cells incubated with nonconditioned media (* P < 0.02). Data represent three pooled experiments and are presented as mean ± SD.
To investigate the functional consequences of the downregulation of tPA mRNA, we examined tPA activity in culture media from our BBB model, i.e., from mono-cultures and cocultures of CS. There was no significant difference in tPA activity between monocultures and cocultures after 1 day of culture (2.7 ± 0.1 IU/mL versus 2.3 ± 0.6 IU/mL, P < 0.4). However, after 7 days of culture astrocyte-endothelial cocultures exhibited a substantial reduction in tPA activity (14.6 ± 2.9 IU/mL) as compared with endothelial mono-cultures (30.2 ± 7.7 IU/mL, P < 0.01) (Fig. 6). Concurrently, endothelial PAI-1 mRNA expression was significantly enhanced in coculture 183.3% ± 86.9% (P < 0.02) compared with mono-culture preparations (Fig. 7). These findings suggested that astrocytes produced reduced tPA activity by downregulation of endothelial tPA mRNA and increasing expression of the tPA inhibitor PAI-1.
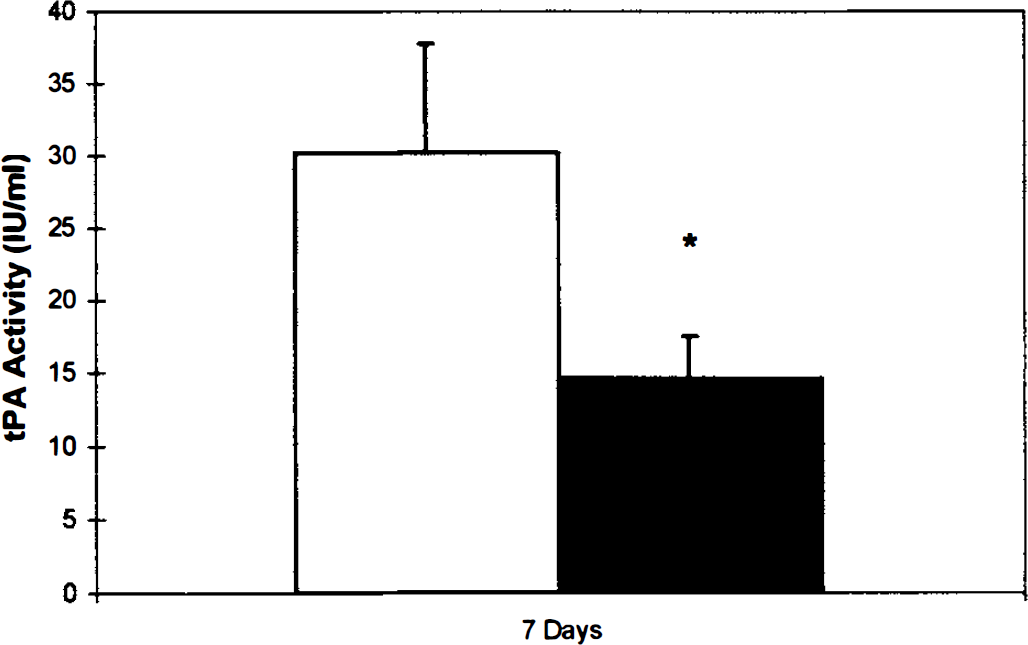
Tissue plasminogen activator (t-PA) activity. Assay of tPA activity in culture media from mono- and co-culture preparations showed a significant reduction in tPA activity in 7-day astrocyte-endothelial co-cultures compared with endothelial monocultures (* P < 0.01). Data are presented as mean ± SD. Data are from 1 experiment (performed in quadruplicate) and representative of 3 independent experiments. (□) monoculture. (▪) coculture.
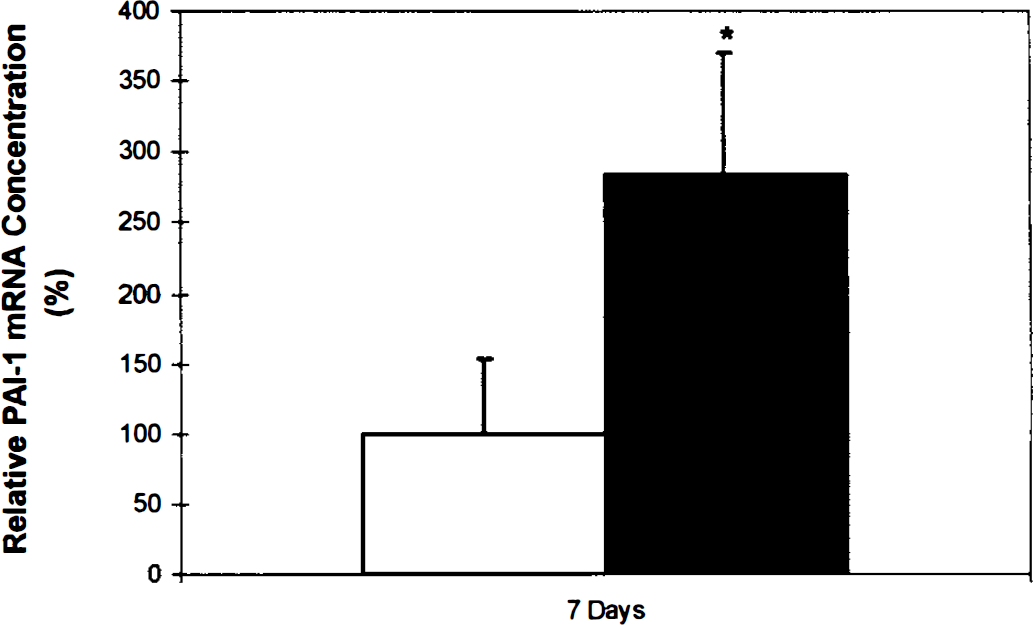
Plasminogen activator inhibitor-1 (PAI-1) mRNA expression. Reverse transcription-polymerase chain reaction analysis of PAI-1 transcripts showed significant increase in seven day astrocyte-endothelial cocultures compared with endothelial monocultures(* P < 0.02). Data are presented as mean ± SD. Data are from one experiment (performed in quadruplicate) and representative of three independent experiments. (□) monoculture. (▪) co-culture.
DISCUSSION
Studies of fibrinolysis and the BBB are confounded by potential contribution of both endothelial and glial plasminogen activators and plasminogen activator inhibitors (Kalderon et al., 1990). Because endothelial-derived fibrinolytic factors can directly contribute to circulating tPA and PAI-1 in plasma and participate in intravascular thrombolysis, we have chosen to concentrate on brain endothelial tPA and PAI-1 expression. Our BBB model involves direct coculture of endothelial cells with astrocytes, but the differing species (bovine and mouse, respectively) allow for the selective analysis of endothelial tPA and PAI-1 transcripts. Moreover, our use of later passage and dedifferentiated brain endothelial cells allows for determination of the relationship between induction of BBB features and regulation of tPA and PAI-1 expression.
We have shown that astrocytes negatively regulate brain capillary endothelial cell expression of tPA mRNA and tPA function in vitro. Direct astrocyte coculture with endothelial cell CS showed substantial downregulation of tPA mRNA and activity after 7 days of coculture, a time at which BBB characteristics are demonstrable in this model. Therefore, these results suggest that astrocytes at the BBB regulate tPA expression. Moreover, the increased endothelial PAI-1 mRNA after 7 days coculture suggests that the decreased tPA function is also caused by astrocyte-dependent enhanced expression of endothelial PAI-1.
Vascular endothelial expression of tPA in vitro is dependent on both the location of the vessel in question as well as its size (Levin et al., 1997). In primate brain, capillaries, representing 40% of all vessels less than 100 µm in diameter, account for only 3% of those vessels that are immunoreactive for tPA (Levin and del Zoppo, 1994). Moreover, bronchial capillaries are immunoreactive for tPA in the mouse, whereas pulmonary capillaries do not express tPA (Levin et al., 1997). These observations suggest organ-specific regulation of tPA expression. Our findings further suggest that the BBB, and specifically, BBB astrocytes, provide brain-specific regulation of tPA expression.
In vitro studies have shown that brain capillary endothelial cells have restricted expression of tPA, in comparison with systemic microvascular endothelial cells. Shatos et al. (1995) showed that human systemic, but not brain, microvascular endothelial cells secreted tPA in response to a-thrombin. Grau et al. (1997) demonstrated that systemic microvascular endothelial cells produced threefold higher levels of tPA compared with the brain microvascular endothelium. Our findings, consistent with these earlier studies, demonstrate distinct differences in tPA expression between capillary endothelial cells with and without the BBB phenotype.
Prior studies have shown a substantial relationship between plasminogen activators and angiogenesis in vitro (Yasunaga et al., 1989; van Hinsbergh et al., 1991). Endothelial cells can produce two molecular forms of plasminogen activators, uPA as well as tPA. Endothelial uPA is primarily involved with extracellular matrix degradation during cellular proliferation and angiogenesis (van Hinsbergh et al., 1991; Laterra et al., 1994; Rao et al., 1996). Astrocyte-derived uPA is known to induce endothelial cell differentiation into capillaries (Laterra et al., 1994; Rao et al., 1996). However, prior work has shown that tPA has no substantial role in angiogenesis of bovine capillary endothelial cells after 7 days in culture (Yasunaga et al., 1989). Our data are consistent with these latter findings, and indicate that astrocyte-dependent tPA regulation in vitro occurs independent of the angiogenic process.
Murine astrocytes express plasminogen activator activity (Kalderon et al., 1990). This activity is developmentally regulated, peaking at 8 to 14 days after birth and then declining twofold to threefold to a low level after postnatal day 16. We used primary culture astrocytes after postnatal day 16, which minimizes astrocyte plasminogen activity. Despite potential astrocytic contributions of tPA, our 7-day astrocyte-endothelial cocultures exhibited significantly decreased tPA activity compared with endothelial mono-cultures.
It is well established that astrocytes can modulate a variety of endothelial functions (DeBault and Cancilla, 1980; Dehouck et al., 1990, 1994; Murphy et al., 1994; O'Donnell et al., 1995; Raub et al., 1992; Stanimirovic et al., 1995; Stewart and Wiley, 1981; Sun et al., 1995; Takakura et al., 1991; Tao-Cheng et al., 1987; Tran et al., 1996). A number of these studies demonstrated that the effects can be induced by astrocyte-conditioned media, thus implicating a role for astrocyte-derived soluble factors in the regulation of endothelial function (Murphy et al., 1994; O'Donnell et al., 1995; Raub et al., 1992; Stanimirovic et al., 1995; Takakura et al., 1991). The astrocyte regulation of endothelial fibrinolysis may require direct contact between astrocytes and endothelial cells, or may be mediated by diffusable substances. Our data show significant regulation of tPA expression by endothelial cells incubated with media conditioned by astrocytes for 2 days, These findings suggest that an astrocyte-derived soluble factor is at least partially responsible for the regulation of endothelial tPA expression by astrocytes. Astrocytes can express cytokines known to transcriptionally regulate tPA and PAI-1, including interleukin-1β and tumor necrosis factor-α (Lieberman et al., 1989; Schleef et al., 1988; Sharif et al., 1993). Moreover, astrocytes express transforming growth factor-β (Constam et al., 1992), also known to regulate fibrinolysis (Saksela et al., 1987; Fujii et al., 1991). These factors may play a role in astrocyte-dependent regulation of endothelial tPA and PAI-1 transcription and, ultimately, fibrinolytic activity.
The importance of brain capillary expression of tPA in cerebrovascular disease has been emphasized in recent work analyzing tPA expression in two important stroke models, In a diabetic stroke model, reduced microvascular expression of tPA mRNA and protein was associated with substantially increased infarct size after middle cerebral artery occlusion (Kittaka et al., 1996). Moreover, in a rat model using nicotine infusion to mimic smoking, reduced microvascular tPA protein was also associated with increased infarct size after occlusion of the middle cerebral artery (Wang et al., 1996). These observations suggest a link between brain capillary tPA expression and outcome after large artery occlusion in the brain.
Our findings add to the emerging picture of the astrocyte as a key mediator for hemostasis within the central nervous system. Tissue factor, the primary generator of the coagulation cascade, is abundantly expressed by astrocytes (Eddleston et al., 1993). Astrocytes can also express tPA (Kalderon et al., 1990; Tranque et al., 1992), PAI-1 (Tranque et al., 1992), thrombomodulin (Pindon et al., 1997), and the tPA inhibitor protease nexin-1 (Scott et al., 1983; Gloor et al., 1986; Choi et al., 1990; Festoff et al., 1996). Moreover, astrocytes regulate endothelial thrombomodulin expression (Tran et al., 1996), as well as endothelial tPA and PAI-1 expression (as described herein). Thus, astrocytes critically participate in hemostasis by both expression and regulation of multiple hemostasis factors. The abundance of astrocyte tissue factor, as well as negative regulation by astrocytes of endothelial thrombomodulin and tP A, suggests a prothrombotic milieu within the brain microvasculature. We speculate that such a prothrombotic milieu provides protection from neonatal intracranial hemorrhage. This potential evolutionary advantage could become counter-productive with aging and development of stroke risk factors, predisposing to brain infarction.
In conclusion, astrocytes negatively regulate brain capillary endothelial expression of tPA mRNA and function. This regulation is associated with development of features of the BBB in vitro. These findings support an important role for the BBB in the regulation of hemostasis by the brain. Modulation of astrocyte-dependent tPA regulation may lead to new strategies for the treatment of ischemic stroke.
