Abstract
Mammalian hibernation is a state of natural tolerance to severely decreased brain blood flow. As protein tyrosine phosphorylation is believed to be involved in the development of resistance to potentially cell-damaging insults, we used immunoblotting for the phosphotyrosine moiety to analyze extracts from various tissues of hibernating and nonhibernating ground squirrels. A single, hibernation-specific phosphoprotein was detected in the brain, but not in any other tissue tested. This protein, designated pp98 to reflect its apparent molecular weight, is distributed throughout the brain, and is associated with the cellular membrane fraction. The presence of the protein is tightly linked to the hibernation state; it is not present in contemporaneously assayed animals that are exposed to the same cold temperature as the hibernators, is present for the duration of a hibernation bout (tested from 1 to 14 days), and disappears within 1 hour of arousal from hibernation. The close association of pp98 with the hibernation state, its presence in cellular membranes, and the known properties of membrane phosphotyrosine proteins suggest that it may transduce a signal for adaptation to the limited availability of oxygen and glucose and low cellular temperature that characterizes hibernation in the ground squirrel.
The pathophysiology of progressive brain damage and neuronal death during stroke are multifactorial, interwoven, and complex (Hallenbeck and Frerichs, 1993). Experimental therapies that are aimed at counteracting one or another of the many mediators of neuronal damage and death often have worked in animal stroke models that are sensitive to small effects; however, they have tended to perform weakly in clinical trials of acute cerebral infarction (Dorman et al., 1996; Hallenbeck and Dutka, 1990). It is difficult to design a new therapeutic approach that accounts for the many factors that participate in progressive ischemic brain injury which may act as a constellation of minor causes. Natural states of tolerance to ischemia, in contrast, could guide brain researchers to effective treatment strategies accessible to logic and reason only in retrospect (Frerichs et al., 1994). Mammalian hibernation is a state of natural resistance to ischemia in that such animals exhibit tolerance to extremely low CBF and restricted availability of oxygen and glucose in vivo (Hochachka, 1986; Frerichs et al. 1994, 1995; Wang, 1988) and temperature-independent resistance of hippocampal slices to glucose and oxygen deprivation in vitro (Frerichs and Hallenbeck, 1997) in addition to their tolerance of deep hypothermia.
There is abundant evidence that induction and maintenance of the hibernating state involves specific cellular regulation rather than being a simple suspension of temperature control or “reversion to primitive poikilothermy”(Wang, 1988). Biochemical and molecular mechanisms of homeostatic regulation permit cells of hibernating animals to survive severe reduction in nutrient perfusion and oxygen availability under extreme hypothermic conditions. Phosphofructokinase and pyruvate kinase from the liver of the meadow jumping mouse, Zapus hudsonius, showed kinetic changes indicative of phosphorylation inactivation of these enzymes (Storey, 1987). Oxygen consumption for succinate-supported respiration of liver mitochondria has been shown to be depressed in hibernating as compared to active animals at identical incubation temperatures (Pehowich and Wang, 1984). Comparison of respiratory control ratios and oxidative phosphorylation rates of heart mitochondria from active and hibernating ground squirrels revealed significant inhibition of the latter during 37°C incubations that controlled for temperature effect (D'Alecy et al., 1990). Differential gene expression during hibernation has also been noted. Levels of liver α2-macroglobulin mRNA were found to be increased in hibernating relative to active ground squirrels (Srere et al., 1992).
There is a high likelihood that the biochemical and molecular mechanisms specific for hibernation are rigorously controlled by internal hormonal, neuronal, or other signals under the influence of circannual rhythms and external environmental changes. Some form of intracellular signaling may transduce such extracellular signals into appropriate cellular responses. Protein phosphorylation on the tyrosine residues, regulated dually by tyrosine kinases and phosphatases, plays an essential role in signal transduction pathways for a wide range of cellular processes including cell growth and differentiation (Fisher et al, 1991; Schlessinger and Ullrich, 1992). We examined whether modulation of protein tyrosine phosphorylation in brain was specific for the hibernating state.
MATERIALS AND METHODS
Experimental animals
The experimental protocol was reviewed and approved by the National Institute of Neurological Disorders and Stroke Animal Care and Use Committee. Thirteen-lined ground squirrels(Spermophilus tridecemlineatus), weighing 120 to 180 g, were captured in Illinois during August and September by a US Department of Agriculture-licensed trapper (TLC Research, Bartlett, IL) and shipped via air fleet to an animal housing facility on the National Institutes of Health campus (Bethesda, MD). Initially, the squirrels were treated for external and internal parasites for 5 to 6 weeks. They were housed in a holding room in individual shoe box cages with environmental conditions maintained at 22 to 24°C, 60% humidity, and a 12/12 h light/dark cycle. They were given free access to food and water. All the experiments were performed between late October and mid-December. Each animal was weighed weekly to determine its phase in the annual hibernation cycle; animals that showed a rapid increase in body weight to levels of 220 to 240 g were placed in a totally dark chamber kept at 5 to 6°C and 60% humidity, the hibernaculum, to facilitate hibernation. Animals were referred to as warm-adapted active when they displayed a low and stable body weight and did not show any signs of torpor or hibernation in the holding room. After being placed in the hibernaculum, most of the animals entered a state of deep torpor within 3 to 8 days. The animals were subjected to experiments as hibernators after 1 to 7 days of hibernation. Animals that did not show any sign of hibernation 7 to 14 days after being placed in the hibernaculum were referred to as cold-adapted active animals.
Western blotting
Animals were euthanized with sodium pentobarbital (100 mg/kg, intraperitoneally) and the brain, heart, lung, liver, kidney, and spleen were resected. Some brains were separated into the hippocampus, cerebral cortex, caudoputamen, midbrain-diencephalon, and cerebellum (Ohtsuki et al., 1995). Each specimen was homogenized in Laemmli's buffer consisting of 62.5 mmol/L Tris-HCl, pH 6.8, 2% sodium dodecyl sulfate (SDS), 10% glycerol, and 5% 2-mercaptoethanol. An equal amount of protein(5 μg) was separated on 4% to 20% SDS polyacrylamide gradient gels for general screening (Laemmli, 1970) and 7.5% SDS polyacrylamide gels for focused analysis of approximately 100-kd proteins and electrophoretically transferred to polyvinilidene difluoride membranes (Millipore, Bedford, MA) (Towbin et al., 1979). The membranes were probed with antiphosphotyrosine antibody, either clone py20 or py54 (Calbiochem/Oncogene Science, Cambridge, MA) and the antigen-antibody complex was visualized as described previously (Ohtsuki et al., 1996).
Preparative procedures
The membranous and cytosolic fractions of the brain were prepared as described previously (Ohtsuki et al., 1995) with the modification that phosphatase inhibitors were added. Each sample was homogenized by means of a Dounce's homogenizer (Wheaton, Millville, NJ) in 0.32 mol/L sucrose, 10 mmol/L Tris-HCl, pH 7.6, 1 mmol/L sodium orthovanadate, 30 mmol/L sodium pyrophosphate, 50 mmol/L NaF, 5 mmol/L ethylenediaminetetraacetic acid (EDTA), 1 mmol/L ethylene glycol-bis(b-aminoethyl ether) N,N, N′,N′-tetraacetic acid (EGTA), 1 mmol/L dithiothreitol (DTT), a protease inhibitor cocktail consisting of 0.18 trypsin inhibitor unit/L aprotinin, 1 mmol/L phenylmethylsulfonyl fluoride, and 0.5% leupeptin, and centrifuged at 900g for 10 minutes at 4°C to remove cell debris and nuclei. The resultant supernatant was centrifuged at 10,000g for 30 minutes at 4°C. The resultant supernatant and pellet were preserved as cytosolic fraction and membranous fraction, respectively. The membraneous fraction contained cellular membranes with some contamination by mitochondria as indicated by a mitochondrial cytochrome C oxidase assay. Each fraction was solubilized in Laemmli's buffer for Western blotting.
Protein purification procedures
Membranous fractions of the crude homogenates from 100 hibernating ground squirrel brains were prepared following previously described procedures (Ohtsuki et al., 1995) and subjected to preparative isoelectric focusing. The membranous fractions were homogenized in 2% 3-[(3-cholamidopropyl) dimethylammonio]-1-propane sulfonate (CHAPS), 10 mmol/L Tris-HCl, pH 7.6, 250 mmol/L NaCl, 5 mmol/L EDTA, 1 mmol/L EGTA, 1 mmol/L sodium orthovanadate, 30 mmol/L sodium pyrophosphate, 50 mmol/L NaF, 10% glycerol, 1 mmol/L DTT, the protease inhibitor cocktail, and incubated for 2 hours at 4°C, and then centrifuged at 100,000g for 24 hours at 4°C. The supernatant was dialyzed against 1% CHAPS, 6 mol/L urea, and 10% glycerol to remove salts. The sample was adjusted to yield 1% CHAPS, 6 mol/L urea, 4% pharmalyte(3–10, 2%: 4–6.5, 2%) (Pharmacia, Sweden), 10% glycerol, 1% DTT and clarified by 0.45μm-pored membrane filtration (Millipore). The sample was loaded into the Rotofor preparative isoelectric focusing cell (BioRad, Hercules, CA). Isoelectric focusing required 4 hours at 10 W constant power at 4°C. The initial conditions were 510 to 550 V and 28 to 29 mA. At equilibrium the values were 980 to 1000 V and 12 to 13 mA. Twenty fractions were collected, their pH values were measured, and aliquots were analyzed by Western blotting. The fraction that contained pp98 around isoelectric point 5.1–5.3 was then dialyzed against 62.5 mmol/L Tris-HCl, pH 6.8, 0.1% CHAPS, and 10% glycerol. The sample was prepared to contain 50 mg of protein dissolved in Laemmli's buffer.
For preparative denaturing polyacrylamide gel electrophoresis, a 12-cm high 6% acrylamide (30 acrylamide:0.8 bisacrylamide) resolving gel and a 1.2-cm high 2.67% acrylamide stacking gel were cast in the 37-mm ID Model 491 Prep Cell (BioRad). After a 5-minute incubation at 80°C, the samples were run on gel at 4°C at a constant power of 12 W for 16 hours (initial voltage, 119 V; final voltage, 174 V). The elution chamber outlet was pumped at 1 mL/min to a fraction collector and 3-mL fractions were collected. Fraction 1, the first fraction containing bromphenol blue marker dye, was collected after 6 hours of electrophoresis. Aliquots of the sample were subjected to Western blotting. Fraction 200, collected after 12 hours of electrophoresis, contained pp98 and was filtration-concentrated by Centricon(Amicon, Beverly, WA). The concentrated pp98 sample was electrophoresed on 6% SDS polyacrylamide gel, and the gel was stained with a copper stain kit (BioRad). The interest band was excised and subjected to amino acid sequence analysis at Harvard Microchem (Cambridge, MA) by collisionally activated dissociation on a Finnigan TSQ 7000 triple quadrupole mass spectrometer.
RESULTS
As shown in Fig. 1A, immunoblotting of brain extracts for the phosphotyrosine moiety revealed several bands. One of these proteins, which we designate pp98 to reflect its apparent molecular weight, was detected in hibernating squirrels but not in the nonhibernating animals. The same result was obtained using either of the two phosphotyrosine antibodies tested, py20 and py54 (see Methods). In addition, the specificity of the immunoreaction was verified by competition experiments: the signal was inhibited by addition of 10 mmol/L phosphotyrosine but not by 10 mmol/L phosphoserine or phosphothreonine.
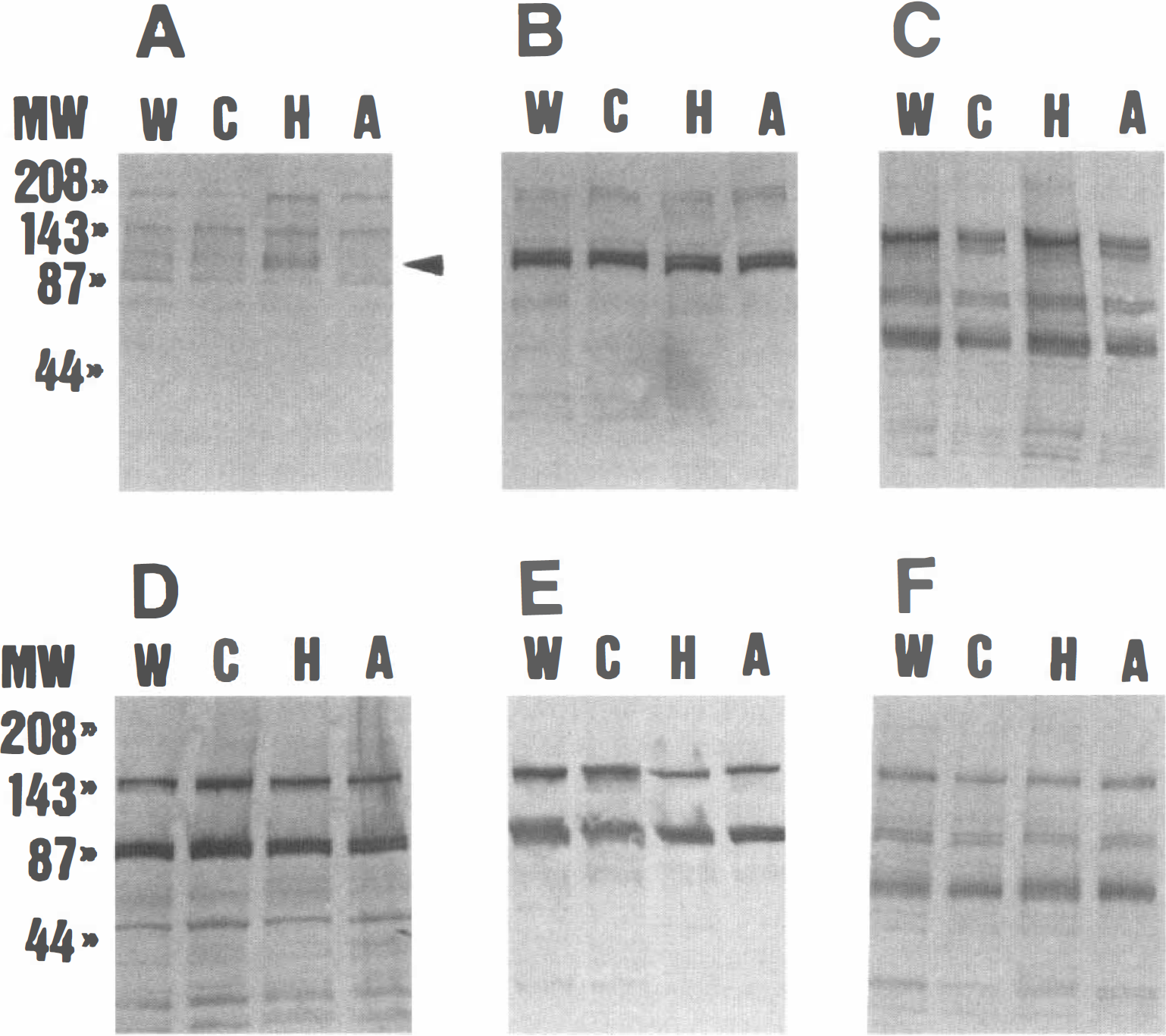
Protein tyrosine phosphorylation during hibernation in extracts of various tissues (
To determine if pp98 is expressed in other tissues, immunoblotting was performed for extracts from the heart, lung, liver, kidney, and spleen of hibernating and nonhibernating animals, the latter including warm-adapted active and cold-adapted active animals and those recently aroused from hibernation. In no case was pp98 detected (Fig. 1, B-F). In addition, no other band showed a significant difference between the hibernation and nonhibernation extracts.
The above results suggest that pp98 is specific to the hibernating brain. To determine if it might be localized to a specific brain region, the hippocampus, cerebral cortex, caudoputamen, midbrain-diencephalon, and cerebellum were separately isolated and immunoblotted. The protein could be detected in extracts from each of these regions, but was relatively more abundant in the caudoputamen and hippocampus and less abundant in the midbrain-diencephalon and cerebellum (Fig. 2A). The cellular localization of the protein was then determined by fractionating a brain extract into cytosolic and cellular membrane fractions. The pp98 was preferentially located in the cellular membrane fraction (Fig. 2B).
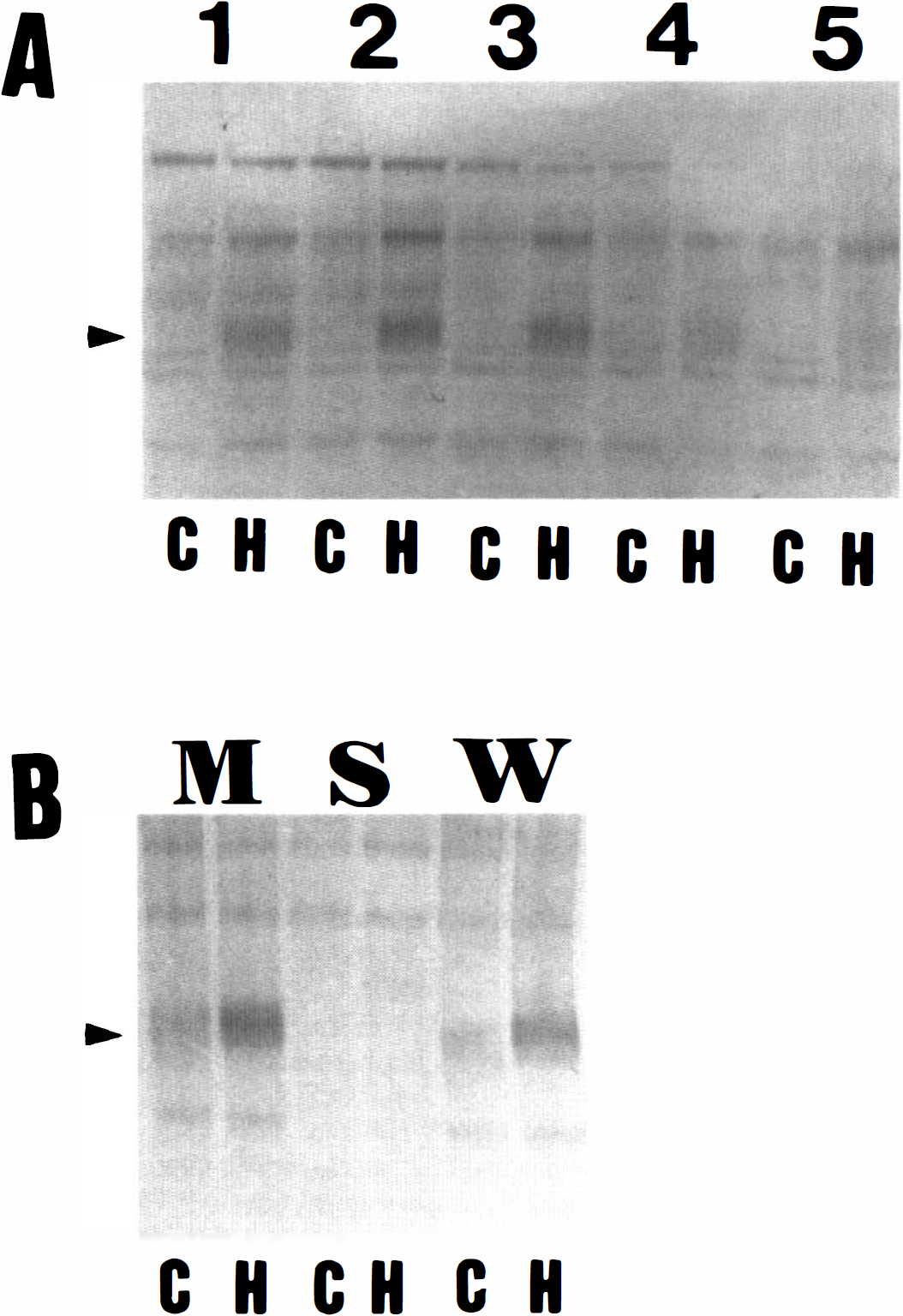
Localization of pp98.
The data in Fig. 1 indicate a hibernation-specific control of pp98 appearance, as the phosphoprotein is not detected in cold-adapted active contemporaries or in animals that have been recently aroused from hibernation. To probe further the relationship between pp98 and the hibernation state, brain extracts were analyzed from animals after 1, 3, 7, and 14 days of hibernation (Fig. 3A), and at various times after arousal from hibernation (Fig. 3B). The protein was detected at relatively equal levels throughout hibernation, but it had disappeared as early as 1 hour after arousal (the earliest time point studied). Hence, there is an extremely close correlation between the hibernation state and the presence of pp98.
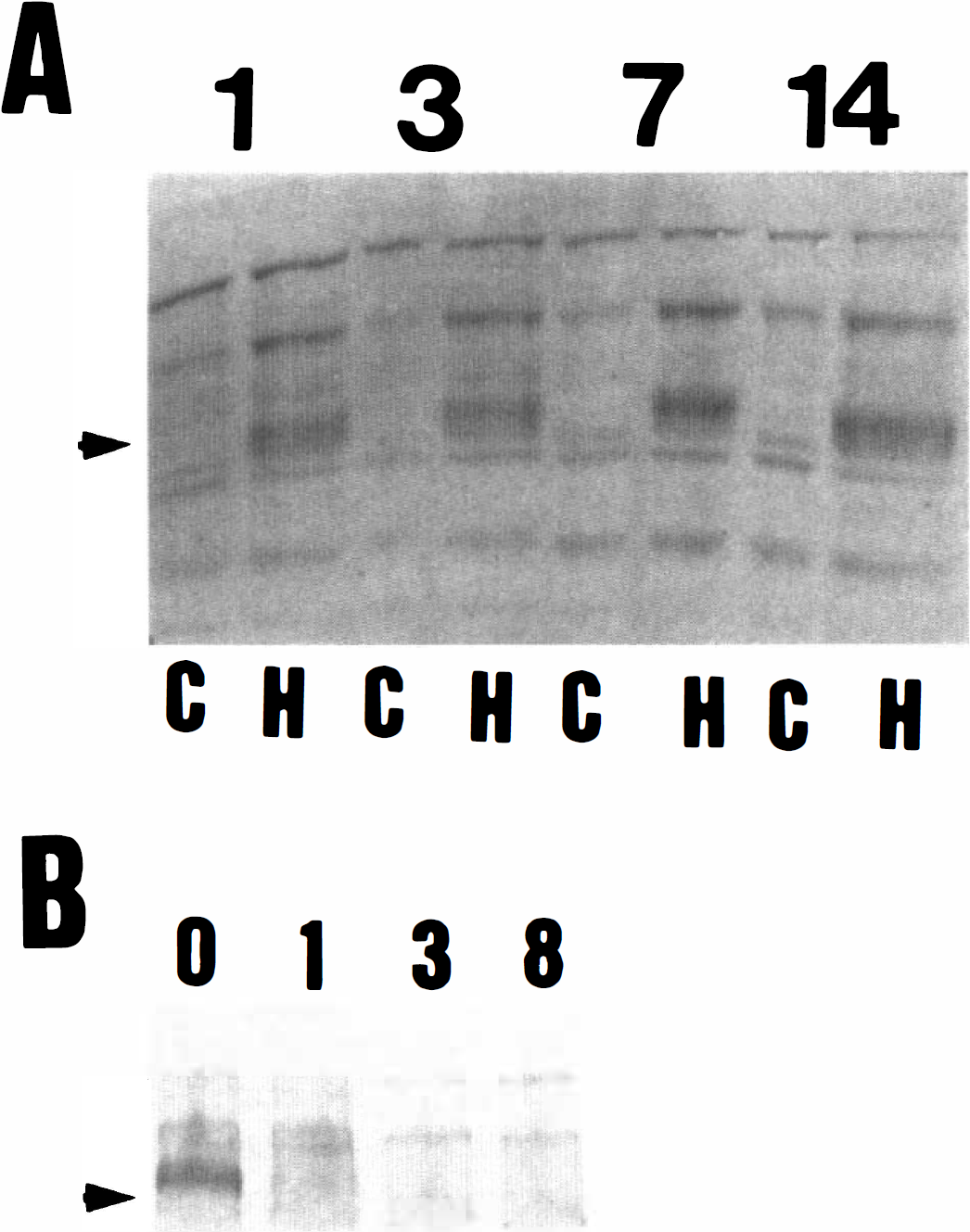
Tyrosine phosphorylation of pp98 in the brain during hibernation and arousal.
Identification of the sequence of pp98 would be a major step toward understanding its role in hibernation. However, thus far this goal has been frustrated by the extremely low abundance of the protein. When gels such as those shown in Fig. 1 were analyzed with either silver stain or Commassie brilliant blue, no band migrating in the region of pp98 was discerned. These gels contain 5 μg of protein, which is equivalent to about 0.0003% of a squirrel brain. A faint silver-staining band that could correspond to pp98 was visualized only when extracts from the membrane fraction of 10 hibernating squirrel brains, purified by immunoaffinity chromatography and containing phosphotyrosine protein that could be solubilized by CHAPS, was analyzed(data not shown). The protein was then purified from 100 hibernating squirrel brains by preparative isoelectric focusing electrophoresis with the Rotofor Cell and subsequent preparative SDS-polyacrylamide gel electrophoresis with the Model 491 Prep Cell. These procedures yielded an estimated 10 to 50 ng of pp98 based on copper staining of a preparative gel. However, microsequencing by collisionally activated dissociation on a Finnigan TSQ 7000 triple quadrupole mass spectrometer was unsuccessful, as the quantity of isolated protein was still beneath the threshold for sequence determination.
DISCUSSION
We have shown the presence of a hibernation-specific, sustained increase in tyrosine phosphorylation of a membraneous fraction 98-kd protein in various brain regions of the 13-lined ground squirrel. The tyrosyl phosphoprotein was not found in heart, lung, liver, kidney, or spleen, and it was not found in active animals or animals recently aroused from hibernation.
The mammalian brain is extremely vulnerable to cerebral hypoperfusion and reduced availability of glucose and oxygen, and is also susceptible to damage at low cellular temperatures (Hochachka, 1986). Transmembrane ion and electrical potentials cannot be sustained because of energy insufficiency and high membrane permeability for ions under these conditions; metabolic and membrane functions, therefore, become uncoupled, leading to cellular dysfunction and irreversible neuronal damage (Hochachka, 1986). The adjustments that characterize mammalian hibernation, however, confer a state of natural tolerance to cerebral hypoperfusion, to reduced availability of oxygen and glucose, and to hypothermia that permits animals to endure these stresses without any apparent neuronal death (Hochachka, 1986; Frerichs et al., 1994, 1995). An understanding of the molecular mechanisms that regulate hibernation could guide efforts to increase resistance to brain damage during brain ischemia. The membrane-associated 98-kd tyrosyl phosphoprotein found in the brain of hibernating ground squirrels is a candidate regulatory factor.
Both protein tyrosine kinases and phosphatases are highly expressed in the brain (Brugge et al., 1985; Fisher et al., 1991). Protein tyrosine phosphorylation participates in the regulation of glutamate receptor function (Bading and Greenberg, 1991; Wang and Salter, 1994), neuronal development, synaptic plasticity and long-term potentiation (Grant et al, 1992; O'Dell et al., 1991). After exposure to various sublethal stresses, activation of tyrosine kinases and an increase in tyrosine phosphorylation of membrane-associated proteins play a pivotal role in cellular resistance and survival against otherwise lethal insults and environmental stresses (Devary et al., 1992; Yao and Cooper, 1995; Hu and Wieloch, 1994; Lindvall et al., 1992; Merlio et al., 1993). By analogy, we suggest that stimulation of pp98 tyrosyl phosphorylation in hibernation may be involved in an intracellular signaling network that develops and maintains tolerance to stressful conditions in the brain. We are continuing our efforts to identify the protein in the belief that it constitutes an important clue to the regulation of hibernation.
Footnotes
Acknowledgements
The authors thank Mrs. Mary Crawford for secretarial assistance.
