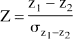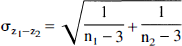Abstract
Covariance analyses of positron emission tomography (PET) data are used increasingly to elucidate the functional connectivity between brain regions during different cognitive tasks. Functional connectivity may be estimated by examining the covariance between regions over time or across subjects. In functional brain-mapping studies, across-subjects covariance matrices derived from within-task (nonsubtracted) and between-task (subtracted) data characterize different, complementary aspects of functional interactions. The authors study amygdala-orbitofrontal interactions during three task conditions (aversive olfaction, odor detection, and resting with eyes closed) to illustrate the strengths and limitations of across-subjects covariance analyses based on subtracted and nonsubtracted data. This example underscores the dynamic nature of connectivity between the amygdalae and orbitofrontal cortices and highlights the importance of including data from resting conditions in covariance analyses.
Current research attempts to localize neurocognitive functions within the framework of distributed network models of brain functioning (e.g., Mesulam, 1990). Definition of such networks occurs at both anatomic and functional levels. Characterization at the anatomic level relies on the pattern of afferent and efferent fibers that interconnect distinct neural regions (e.g., Selemon and Goldman-Rakic, 1988). The density of structural interconnections between regions remains relatively stable during adulthood, providing a constant constraint on the extent to which different components of the network interact. Characterization at the functional level focuses on how neural activity within one component of the network changes in relation to neural activity in other components of the network. When the activity in two regions displays a high degree of covariance, the regions may be defined operationally as “functionally connected” (Friston, 1994). Thus, the level of covariance or correlation between two regions arising over multiple measurements may be taken as an empirical estimate of their functional connectivity. When two regions show a high degree of functional connectivity they also may be referred to descriptively as “functionally coupled” (Horwitz, 1990). In describing two areas as functionally coupled, we imply that the two regions some how work together, influence each other, or are commonly influenced by another source, without indicating the source or causality of their functional connection.
Although the level of anatomic connectivity between brain regions remains relatively stable over time, the level of functional connectivity can vary dramatically depending on the specific computations that the network performs at any given instant. Because different computations require the interaction of different brain regions, two areas may be functionally coupled during one task, but not during another, despite having a constant level of anatomic connectivity. In this regard, when the activity in two regions becomes independent from one another, the regions may be described as “functionally uncoupled.”
Positron emission tomography quantifies activity in multiple brain regions simultaneously, thereby enabling the assessment of the functional couplings and uncouplings within distributed neurocognitive networks. Several related statistical techniques allow the extraction of information on the functional connectivity between brain regions from PET data. These techniques include correlational analyses (Clark et al., 1984; Horwitz et al., 1992b), partial correlations (Horwitz et al., 1984), scaled subprofile modeling (Moeller et al., 1987), and principal components analysis (Friston et al., 1993).* All of these methods rely heavily on the covariance of measured activity in different brain regions. In each method, the covariance between different regions provides estimates of the level of functional connectivity between regions during a given task.
Several methodologic issues emerge when applying covariance analyses to PET data. The first issue involves whether functional connectivity should be estimated over time or over subjects. Analysis of functional connectivity over time examines whether the activity in one area of the brain is covariant over time with activity in another brain region. The level of functional connectivity is defined operationally in this context as the temporal correlation between activity in different brain regions (Friston, 1994). This approach to studying functional connectivity with PET parallels the study of functional connectivity as applied in electrophysiologic studies in which spike trains of cells in separate regions are correlated over time. Examining functional connectivity over time is ideal in situations in which subjects receive large numbers of scans, and there is reason to believe that the true covariance structure across scans is relatively constant. For instance, such an approach may prove extremely useful in a paradigm in which subjects perform the same task during each scan, and the task remains identical across scans or only changes parametrically (say in terms of difficulty). However, this approach to examining functional connectivity may have minimal utility in cases in which different tasks are performed over time. If functional connectivity is task specific (i.e., different tasks are associated with different covariance patterns), less pertinent information can be derived by examining the covariance over time.
An alternative approach to estimating functional connectivity estimates connectivity over subjects. In this approach, functional connectivity is operationally defined as the extent to which activity in one area of the brain is covariant with activity in another area of the brain across subjects. It is the across-subjects approach that we principally are concerned with in this article. The across-subjects approach often is more suited than the over-time approach when examining data from brain-mapping studies that use a subtraction paradigm to determine which regions show increased activity during a stimulation condition relative to a control condition (Fox and Mintun, 1989). It appears particularly useful when researchers wish to explore how functional connectivity changes across tasks. A typical analysis of across-subjects functional connectivity for a brain-mapping study proceeds by taking the normalized (nonsubtracted) radioactive counts within the same regions of interest (ROIs) for the stimulation and control conditions and then entering these values into two separate covariance matrices (e.g., Horwitz et al., 1992a). These within-task matrices provide an overall estimate of how much covariance exists between different regions when performing a given task. The comparison of the patterns of covariance that emerge within the stimulation and control conditions provides information on the extent to which functional connectivity shifts across different tasks or conditions.
A major limitation of examining functional connectivity over subjects with within-task matrices derives from the multidetermined nature of nonsubtracted cerebral blood flow (CBF) values. A wide range of variables not associated with the cognitive or perceptual parameters of interest contribute to CBF levels. In fact, CBF within ROIs in control and stimulation conditions usually correlate substantially, even if the two conditions probe markedly different cognitive computations. For instance, between 50% to 80% of the variance of CBF in each of the individual ROIs presented in this article is shared across conditions. Therefore, factors unrelated to the specific task parameters of interest contribute a substantial portion of the variance in CBF, which directly influences the emergent covariance between different regions. Such factors include relatively static features of anatomy and vascular supply as well as components of the experimental and image-processing procedures that are shared across the stimulation and control conditions. For example, global confounding factors such as whole-brain differences in measured activity can spuriously inflate covariance if not controlled with procedures such as analysis of covariance (ANCOVA) or normalization to an equal level of mean activity per subject. Even when such global confounds are controlled, because covariance matrices derived from within-task data are multidetermined, partitioning the covariance arising from the specific cognitive processes under study from the covariance attributable to other methodologic, anatomic, or psychological variables remains difficult, if not impossible. The inclusion of multiple stimulation and control conditions sometimes can aid in isolating the factors contributing to the covariance, but the extent to which the observed covariance matrices reflect the parameters of interest often remains vague.
An alternative across-subjects approach examines whether the change in CBF (ΔCBF) that occurs between two conditions for a given ROI is covariant with the ΔCBF in another ROI. The change in CBF results from the subtraction of the mean CBF for an ROI in the control condition from the mean CBF for the same ROI in the stimulation condition. This parallels the basic subtraction approach commonly employed in functional brain-mapping studies. Anatomic, physiologic, and psychological determinants of CBF that remain constant across the two conditions are removed using this technique. As a result of this cancellation, matrices containing ΔCBF data more powerfully isolate the specific activity associated with the cognitive or perceptual parameters of interest. The resultant covariance matrix reflects the extent to which changes in activity that occur within in response to specific experimental manipulations are correlated across ROIs. A major limitation of this approach derives from the fact that it fails to capture real and robust functional connectivity in situations in which two regions respond differently to the specific factors that distinguish the stimulation and control condition. For instance, if the CBF in two regions is covariant within both the stimulation and control conditions, yet does not change between conditions in a similar or proportional manner, analyses using between-task ΔCBF will fail to capture the stable connectivity between the two regions. This occurs because between-task matrices do not provide information on overall functional connectivity, but rather indicate the extent to which activity in different regions change together in response to specific experimental manipulations. As an example, imagine an experiment in which subjects read aloud in a stimulation condition and read silently in a control condition. The specific parameter of interest that distinguishes the two conditions is speaking aloud. Two areas involved in reading may be correlated highly in both conditions, but if their activity is not modified proportionally by the addition of speaking aloud, they will show little between-task correlation. It would be wrong to conclude on these grounds that the two areas are not functionally connected during reading aloud. Rather, these areas appear highly functionally connected, but the connectivity is not attributable to the specific parameter of interest.
Although covariance matrices derived from both subtracted and nonsubtracted data can contribute toward understanding functional connectivity across subjects, the conceptual and methodologic strengths and weaknesses of these techniques rarely have been addressed directly. Most studies examining functional connectivity apply only one approach to estimating functional connectivity, and few studies report estimates of specific functional connectivity based on the between-task matrices. We demonstrate the use of across-subjects covariance matrices based on both subtracted and nonsubtracted PET data to elucidate more fully the functional connectivity between brain regions. Specifically, we illustrate: 1) the divergent yet complementary nature of covariance matrices derived from nonsubtracted and subtracted CBF data; 2) the dynamic nature of functional connectivity across different conditions; 3) the importance of comparing covariance in active conditions to covariance in a resting state; 4) the relevance of functional uncoupling for understanding neurocognitive network functions; and 5) several of the core methodologic weaknesses of these approaches.
METHODS
The data are derived from an H215O study of olfactory hedonics. The study is described in detail in Zald and Pardo (1997). In brief, 13 right-handed women (ages 19–49) were exposed to different olfactory conditions while undergoing PET imaging. Cerebral blood flow was estimated from normalized (1,000 counts) tissue radioactivity (after correction with measured two dimensional attenuation) using a ECAT 953B camera (Siemens, Knoxville, TN, U.S.A.) with septa retracted; a slow-bolus injection of H215O (814 MBq or 22 mCi initial dose was infused at a constant rate over 30 seconds [Silbersweig et al., 1993]), with a 90-second scan acquisition beginning on radiotracer arrival into the brain and a 10-minute interscan interval. Images were reconstructed using a three-dimensional reconstruction algorithm with a Hanning filter (0.5 cycles/pixel [Kinahan and Rogers, 1989]). Software developed by Minsoshima and colleagues (Minoshima et al., 1992, 1993, 1994) enabled automated normalization, coregistration, and nonlinear warping to the atlas of Talairach and Tournoux (1988).
The stimulation condition involved exposure to a highly aversive combination of sulfides, consisting of 25 ppm each of diethyl sulfide, ethanethiol, and methanethiol, delivered from a 1-L plastic bag with the outlet positioned approximately 15 cm from the nostrils. Gas release began on the start of radiotracer infusion and continued through the first 60 seconds of scan acquisition. The odor detection control condition was identical, except that no odors were presented during the scan period. In both conditions, subjects were instructed to “Close your eyes. Breathe through your nose, and see if you can smell anything.” Pixel-wise subtraction revealed peak activations in the region of the left amygdala (LAMG), the right amygdala (RAMG), and the left orbitofrontal cortex (LOFC; Brodmann area 47/11; Zald and Pardo, 1997). Regions of interest (4.5-mm radius spheres) were centered on the peak ΔCBF between sulfide and no odorant conditions. The Talairach coordinates (x, y, z) for these ROIs are as follows: LAMG (−34, −4, −11); RAMG (26, −1, −14); LOFC (−42, 35, −14). We placed an additional ROI on the right orbitofrontal cortex (ROFC; Brodmann area 11) at the Talairach coordinates of (28, 28, −18); this ROFC focus showed increased CBF in the aforementioned study (z-score = 3.1), but failed to reach conservative levels of statistical significance. Subsequent analysis (Zald and Pardo, unpublished observations) and a previous PET study using a different olfactory paradigm (Zatorre et al., 1992) suggest that this ROFC area may become activated in both the stimulation and the control conditions compared with the resting state. Within-task CBF values were calculated by taking the mean value of all pixels within each ROI. Between-task ΔCBF consisted of the subtraction of the mean pixel value for each ROI in the odor detection, control condition from the mean pixel value for the same ROI in the aversive odorant condition. Pearson product moment correlations were calculated to provide a common metric for displaying the relation between activity in the different ROIs. Each subject contributed one scan pair to these analyses.
The control condition, which involved attempting to detect odorants, does not provide information about the baseline resting correlations between the four ROIs. However, interpretation of the correlations observed in active conditions requires knowledge of the baseline covariance between regions. Using identical scan acquisition and image-processing methods, we examined the same four ROIs in 33 healthy subjects (mean age, 25 years; range, 18–59 years) scanned while resting with eyes closed (ECR) in a quiet, darkened room. Whenever subjects received more than one ECR scan (n = 24), the ECR scans were averaged before extraction of the ROIs.
To formally test whether the level of functional connectivity changed significantly across the different conditions, we performed Fisher's z-transformations on all correlations for the within-task matrices. Because z-transformed correlations are normally distributed, they allow direct comparison of the difference between two correlations using the following equation (Fisher, 1915, 1921; Zar, 1984):
where z1 = the z-transformed correlation in condition 1, z2 = the z-transformed correlation in condition 2, and
The resultant Z-value then may be examined using a standard table of critical values of the t distribution (with degrees of freedom = ∞). We do not report an analysis of this sort for direct comparison of the odor detection and aversive sulfide conditions because the statistical power of such tests are prohibitively poor with n = 13. Differences in correlations must be extremely large (approaching or exceeding r = 0.60) to reach significance in such a small sample, and none of the changes in correlations between the aversive sulfide and odor detection conditions exceed this level. However, far greater statistical power is available for testing changes in correlations relative to the normative ECR data set, and these are reported in the following section.
RESULTS
Fig. 1 displays the correlations between the four ROIs. The pattern of correlations varies depending on the task condition and the type of covariance matrix. Analysis of the matrix with between-task ΔCBF reveals a significant correlation between the LAMG and LOFC (r = 0.68, P < 0.01): the LOFC and LAMG increase their activity proportionately in response to aversive odorants relative to when attempting to detect an odor. In contrast, within-task analyses show only modest, nonsignificant correlations between the LOFC and LAMG activity during the stimulation and control conditions. Although a somewhat greater correlation arises between these structures in the aversive odorant condition than in the odor detection condition, the within-task data alone fail to demonstrate the significant relation between the LOFC and LAMG response to aversive olfactory stimulation.
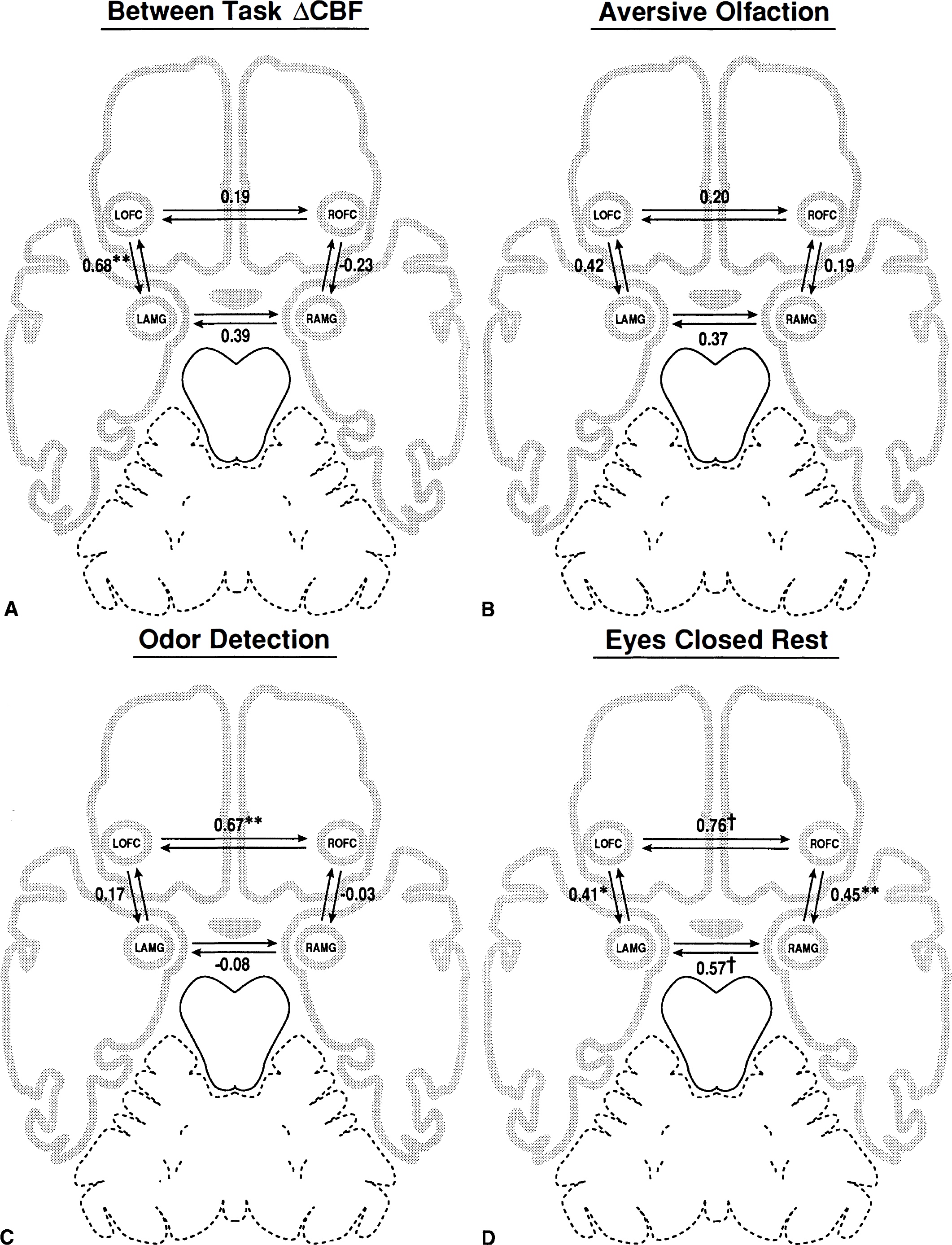
Correlations between the right and left orbitofrontal cortex (LOFC) and amygdalae derived from:
By comparison, the covariance matrices of the non-subtracted within-task data provide significant information on the functional couplings and couplings that occur during aversive olfactory stimulation, during odor detection, and during rest. For instance, the ROFC and LOFC exhibit a significant association in the odor detection condition (r = 0.67, P < 0.01). These regions also show highly correlated resting activity (r = 0.76, P < 0.001). However, the introduction of an aversive odorant causes a substantial decrease in functional connectivity between the two regions (r = 0.20, P > 0.10). The decrease in ROCF-LOFC functional connectivity in the aversive condition relative to the resting condition is statistically significant (t2-tailed(∞) = 2.16, P < 0.01). Thus, these regions appear functionally coupled at rest and when attempting to detect an odorant, but uncouple during perception of an aversive odorant.
The within-task matrices for the two amygdalae reveal a different pattern. The largest correlation between the RAMG and LAMG emerges in the ECR data (r = 0.57, P < 0.01). However, the two amygdalae become functionally uncoupled (r = −0.08, P > 0.10) in the odor detection condition. This represents a significant decrease in functional connectivity relative to ECR baseline levels (t2-tailed(∞) = 2.03, P < 0.05). The functional connectivity of the two amygdalae appears only moderately reduced in the aversive olfaction condition relative to the ECR condition, and this difference fails to reach statistical significance relative to ECR (t2-tailed(∞) = 1.18, P > 0.10).
Comparison of the nonsubtracted, within-task matrices also provides information on orbitofrontal cortex (OFC)-amygdala interactions. The ipsilateral amygdala and OFC show statistically significant associations at rest (right hemisphere, r = 0.45, P < 0.01; left hemisphere, r = 0.41, P < 0.05). These baseline, resting correlations provide a context for interpreting correlations between these structures in the stimulation and control conditions. In the left hemisphere, the amygdala-OFC correlation during aversive olfaction remains similar to that observed at rest, but decreases during odor detection. In the right hemisphere, the amygdala-OFC correlation never approaches the baseline level of association during the odorant conditions and falls close to zero during odor detection. This suggests that the RAMG and ROFC become functionally uncoupled during odor detection despite the significant association of these regions at rest. However, because the resting level of functional connectivity between the RAMG and ROFC is modest to begin with, formal comparison of the odor detection and ECR condition failed to reach statistical significance (t2-tailed(∞) = 1.27, P > 0.10).
DISCUSSION
The present analyses demonstrate the utility of examining functional connectivity across subjects with covariance matrices derived from both nonsubtracted, within-task data and subtracted, between-task data. Although it is becoming increasingly common to use nonsubtracted data to elucidate functional connectivity, using between-task ΔCBF matrix represents a novel approach. By canceling sources of variance unrelated to the experimental parameters of interest, between-task ΔCBF matrices allow a level of interpretational specificity that often is not available from nonsubtracted, within-task matrices. In the current example, the between-task ΔCBF matrix shows a commonality in how the LOFC and LAMG respond to aversive olfactory stimulation relative to an odor detection control task. This association remains hidden when using covariance analyses derived from the more multidetermined, within-task data. In contrast, the within-task matrices reveal a dramatic pattern of functional couplings and uncouplings during the odor detection, aversive olfaction, and ECR conditions. Analysis of between-task ΔCBF in isolation would not capture these changes in overall functional connectivity. This example demonstrates how matrices based on subtracted and nonsubtracted data provide different types of information, which together elucidate issues of functional connectivity better than either method applied in isolation.
These issues apply to all statistical techniques that use covariance matrices. We reported matrices derived from Pearson product moment correlations (which assume a simple linear relationship) for this demonstration because they are easily understandable. But the same issues apply to nonlinear data and matrices using rank-ordered data to avoid the assumption of linearity (e.g., Spearman's rho or Kendall's tau).† These issues also pertain to studies of effective connectivity, in which covariance matrices are examined in relation to specified models of how activity in different brain regions influence each other (Friston, 1994).
A number of methodologic difficulties complicate the widespread application of covariance analyses to neuroimaging data. Most of these topics are dealt with else-where (Ford, 1986; Horwitz et al., 1992b). However, the issue of sample size warrants special attention in applying multiple covariance analyses to PET data. The statistical power of covariance analysis depends heavily on sample size-a problem given the small number of subjects in typical PET studies (Ford, 1986). Exploratory analyses in our own laboratory suggest that relatively stable correlations between brain regions emerge with data sets of approximately ten subjects. We find that 20 or more scan pairs usually are necessary to gain sufficient statistical power to test hypotheses, although correlations of similar magnitude may emerge with smaller sample sizes. However, the risk of false-positive correlations (type I statistical error) increases rapidly as more ROIs are examined. This problem is compounded if both within-task and between-task matrices are examined because every pair of ROIs produces three correlations (two within-task and one between-task). Although application of Bonferroni corrections for multiple comparisons limits the risk of false-positive results, these corrections are overly conservative in small data sets. Given the magnitude of correlations typically observed in PET studies, most PET data sets simply are too small to provide sufficient statistical power to examine large numbers of ROIs. The issue of poor statistical power due to small sample sizes also is problematic if one wishes to formally test for changes in functional connectivity across conditions. Changes in functional connectivity between regions must be substantial to reach statistical significance. Therefore, many studies lack the statistical power to formally test for changes in functional connectivity across conditions.
The present analyses highlight the dynamic nature of functional connectivity in the human brain. The pattern of correlations between the OFC regions and the amygdalae change dramatically depending on the experimental condition, despite high correlations of CBF within the same ROI in different conditions. The variance of CBF in the control condition accounts for 50% to 80% of the variance of CBF in the stimulation condition. Nevertheless, the pattern of correlations between different ROIs varies substantially across conditions. Of note, significant correlations between two regions at rest do not imply significant correlations during activation conditions. For instance, the RAMG and LAMG, which have a significant correlation at rest, show no substantial association during olfactory tasks.
Several insights about the interactions between the orbitofrontal cortices and the amygdalae emerge from this application of covariance analysis. First, the LOFC and LAMG respond to exposure to aversive odorants relative to odor detection in a functionally similar manner. This does not indicate that all, or even a majority, of the processing in these two regions is functionally linked. In fact, the within-task data suggest that only a small portion of the variance in either the LOFC or LAMG is related to the activity level in the other region. However, CBF increases proportionally in the LOFC and LAMG between the aversive sulfide and odor detection conditions, when common sources of variance are removed. The cause of this association remains a matter of speculation. Both regions receive direct projections from the primary olfactory cortex (Carmichael et al., 1994; Price, 1991), which could allow parallel processing of olfactory information. The OFC and amygdala also possess strong bidirectional connections capable of supporting either unidirectional or bidirectional influences of one region on the other (Amaral et al., 1992; Carmichael and Price, 1995; Zald and Kim, 1996a). Either way, the commonality of the response to aversive stimuli in the LAMG and LOFC converges with previous electrophysiologic and lesion studies of nonhuman primates that show striking similarities in the manner in which the OFC and amygdala process hedonically valenced information (Thorpe et al., 1983; Gaffan and Murray, 1990; Gaffan et al., 1993; Zald and Kim, 1996b).
The ability to interpret correlations arising from between-task matrices depends heavily on how well the control condition matches the stimulation condition on all variables outside of the parameter of interest. In situations in which the stimulation and control conditions appropriately isolate a single parameter, correlations may be interpreted in specific relation to the parameter of interest with far greater confidence than in situations in which multiple parameters vary between conditions. Similar interpretational issues arise in all experimental designs that rely on subtraction analyses. For instance, the ability to interpret the factors leading to a peak ΔCBF arising in the LAMG during the aversive odor-minus-odor detection condition depends on the adequacy of the odor detection condition as a control for aspects of olfaction. The interpretation of the LAMG-LOFC correlation in the between-task ΔCBF matrix similarly depends on the ability of the odor detection condition to control for aspects of olfactory processing without introducing additional confounding variables. Because the odor detection condition differs from the aversive olfaction condition on a number of parameters (e.g., presence of an odorant, induction of a negative emotional experience, degree to which the subjects attempt to detect an odorant, etc.), the interpretation of the emergent specific functional connectivity becomes more difficult. We have not observed significant between-task correlations between the LOFC and LAMG in experiments comparing exposure to pleasant odorants with odor detection, (Zald and Pardo, unpublished observations). This suggests that olfactory perception per se cannot account for the specific functional connectivity between the LOFC and LAMG in the aversive olfaction-minus-odor detection comparison. However, additional studies using carefully matched control conditions will be necessary to isolate the cognitive or perceptual factors leading to the specific functional coupling of the LOFC and LAMG during aversive olfaction.
The LOFC and LAMG also showed a modest but significant resting correlation in the within-task matrix from the ECR condition. A similarly modest but significant association also emerged between the ROFC and RAMG. These correlations could reflect a modest functional coupling in the process of silent mentation or a byproduct of the robust bidirectional anatomic connections between these two regions (Amaral et al., 1992; Carmichael and Price, 1995; Zald and Kim, 1996a).
Potential methodologic contributions to the observed within-task correlations merit specific attention. Global confounds, such as variations in the timing of radioisotope delivery or the inadequate application of normalization for global activity, could lead to spuriously high correlations. However, two arguments dispute that the observed correlations reflect methodologic artifacts. First, ROIs within the same cerebral hemisphere that lack close anatomic connections generally fail to produce significant correlations in this normative data set (Zald and Pardo, unpublished observation). Second, in the olfactory conditions (which used identical imaging methods as the normative data set), we observe highly dynamic correlations, at times approaching zero. If shared methodologic artifacts determine the observed correlations, these artifacts should exert similar effects across conditions. Thus, the sheer dynamic nature of the observed correlations argues against the possibility that resting correlations reflect an artifact of data acquisition or image processing.
The use of the ECR condition as a baseline measure remains open to debate. Resting with eyes closed (ECR) represents an unconstrained condition in which subjects may partake in multiple types of silent mentation. Thus, functional connectivity in the ECR condition may reflect more than just anatomic characteristics. Nevertheless, ECR scans show a high degree of stability both within and between scanning sessions (Ball et al., 1988a; 1988b) and represent the most commonly used baseline task in human brain-mapping studies. As an alternative to the ECR condition, the average covariance between two ROIs during a series of tasks not believed to involve those ROIs might aid in estimating “baseline” covariance levels. However, the theoretical and practical limitations of such an approach remain substantial.
Despite potential concerns about utilizing ECR conditions, their inclusion significantly aids in the interpretation of correlations arising in other task conditions. For instance, examination of the ROFC-LOFC correlation without reference to the ECR condition might wrongly lead to the assumption that the two OFC areas become uniquely coupled during odor detection. However, the ECR data make clear that this high level of functional connectivity is not unique to odor detection. Unfortunately, studies examining functional and effective connectivity frequently omit information on resting correlations. In the absence of resting data, interpretation of reported levels of functional or effective connectivity often remains ambiguous.
Comparison to a baseline condition also appears extremely useful in revealing functional uncouplings. Functional uncoupling has received little attention in discussions of network interactions. However, the process probably is critical to network functioning. Functional uncoupling occurs when the activity within two components of a network becomes independent or functionally disconnected. If functional coupling reflects an active process that facilitates the interaction or communication between regions, functional uncoupling may optimize neurocognitive functioning by isolating the processing in different regions. Determining the specific situations in which normally coupled regions become functionally uncoupled provides a potentially rich, and so far unexplored, strategy for understanding the different contributions of regions within a distributed network.
The decline in the functional connectivity of the RAMG and LAMG during the odor detection task relative to the resting state provides a clear example of functional uncoupling. Such dramatic uncoupling suggests that processing in these regions becomes disengaged during odor detection. Interestingly, the RAMG shows substantially higher CBF levels than the LAMG during odor detection (Zald and Pardo, 1997), whereas no such asymmetry exists at rest (Zald and Pardo, unpublished observations). Thus, this independence of the two amygdalae during odor detection might reflect the asymmetric or isolated activation of the RAMG during odor detection. This is consistent with neuropsychological studies which indicate asymmetrical involvement of right hemisphere structures in certain aspects of olfactory processing (Zatorre and Jones-Gotman, 1991; Jones-Gotman and Zatorre, 1993).
A different pattern of functional coupling and uncoupling emerges between the ROFC and LOFC. Unlike the amygdalae, the OFC regions exhibit substantial correlations during both ECR and odor detection conditions. However, this association decreases substantially during exposure to aversive odorants. This breakdown in the normally high level of functional connectivity may reflect relatively independent processing in the right and left OFC during exposure to an aversive odorant. The reason this occurs remains unclear, but it provides strong evidence that the left and right OFC do not operate in unison when exposed to an unpleasant odorant.
The high level of resting correlations between the contralateral OFC regions and contralateral amygdalae converge with previous correlational analyses of resting brain activity. In exploratory analyses in our laboratory, we consistently observe the largest correlations between regions at similar coordinates in the opposite hemisphere. High correlations between identical brain regions in the opposite hemisphere similarly characterize regional glucose metabolism levels at rest (Horwitz et al., 1984). This pattern probably reflects a combination of static commonalties in the anatomy and vascular supply of the two hemispheres and commissural and/or callosal connections between homologous contralateral regions. Nevertheless, these functional couplings clearly are not present in all conditions. To the extent that anatomic factors cause covariance in these regions at rest, these influences break down with relative ease during stimulation conditions.
A potential confounding variable in this type of study arises from the placement of ROIs. Use of ROIs based on empirically determined foci from subtraction analyses affords confidence that the regions under study are relevant to the neurocognitive functions of interest. Because the interpretation of functional relationships is constrained by the anatomic connections between regions, the use of empirically defined regions finds greatest justification when the activated regions correspond closely to previously defined anatomic and functional circuits. In the present example, the OFC and amygdala already were known to possess a high degree of anatomic and functional inter-relations, and both structures were expected a priori to show an involvement in olfactory hedonics (Zald and Kim 1996a; 1996b; Zald and Pardo, 1997). In contrast, there exists far weaker justification for taking multiple extrema from a brain mapping study when these foci do not map to previously defined anatomic or functional networks.
Placement of ROIs based on peak foci from substraction analyses also may influence the emergent covariance matrices because of their coherence or lack of coherence to anatomic boundaries. For instance, in the present study, the coordinates for the LAMG peak are quite lateral relative to those for the center of the amygdala, whereas the RAMG peak is closer to the midline of the amygdala. Because of this placement, the ROIs cover slightly different portions of the medial temporal lobe. Thus, slight difference in anatomy could be reflected in different correlational patterns. However, the consistency in the level of ipsilateral OFC-amygdala connectivity in each hemisphere during the ECR condition suggests that these anatomic differences did not dramatically limit the ability to observe functional connectivity. These slight differences in anatomic placement also did not limit the ability to observe robust associations between the two amygdalae in the ECR condition.
Another issue in the placement of ROIs in PET studies relates to the collapsing of data from potentially heterogeneous areas. This may be particularly problematic in an area such as the amygdala, which is composed of a heterogeneous group of nuclei. In treating the amygdala as a unitary structure, the present techniques may obfuscate functional connectivity if it is limited to discrete nuclei. It seems likely that some of the amygdala nuclei possess greater functionally connectivity to the OFC than other nuclei, simply because of their different anatomic connections (Zald and Kim, 1996a). Nevertheless, it is no less appropriate to examine the functional connectivity within a heterogeneous portion of the amygdala than to identify the amygdala as a maxima in a subtraction analysis despite having blurred over these same heterogeneous sectors of the amygdala. Furthermore, the high degree of anatomic and functional interconnections between different amygdala nuclei make it likely that examination of the entire amygdala region often will be as or even more useful than examining discrete amygdala nuclei. This is particularly true in a study such as the present one, in which CBF increases extended over wide portions of both amygdalae (Zald and Pardo, 1997). The very robustness of the response to aversive odorants may in fact relate to the coactivation of multiple amygdala nuclei. If so, inclusion of the entire activated region may be more informative than limiting the study of functional connectivity to a discrete portion of the activated region. Most likely, the fullest understanding of functional connectivity will be gained by examining neural systems on both a finely detailed and a broader regional approach.
The present analyses were undertaken to empirically explore the functional connectivity of the OFC and amygdala in olfactory tasks. Given the empirically driven nature of this study, replication in another sample would improve confidence in the observed findings. Such empirically driven approaches may produce results that exceed our current understanding of how and why networks operate. Little physiologic or anatomic data suggested a priori that the amygdalae uncouple during odor detection. The cause and purpose of this uncoupling remains speculative. However, the very novelty of the finding highlights the unique information provided by covariance analyses of PET data. Demonstration of functional connectivity or lack of functional connectivity does not indicate the causal direction through which regions become coupled or uncoupled. Structural equation modeling procedures, which allow researchers to test the fit of models that incorporate causal links, may provide information on directionality, but evidence of functional connectivity alone cannot. However, in poorly understood networks, a simple empirical demonstration of functional connectivity may contribute greatly to our understanding of how brain regions interact during different tasks.
As the limitations of subtraction analysis of functional brain-mapping data become increasingly apparent (see Friston et al., 1996) and use of alternative approaches to data analysis increases, the strengths and weaknesses of alternative approaches must receive a thorough examination. The present results demonstrate some of the strengths and weaknesses of analyzing covariance matrices based on subtracted and nonsubtracted data. Covariance matrices based on these different types of data provide complementary information about regional interactions. The decision to analyze subtracted or nonsubtracted data dictates which features of functional connectivity can be elucidated and which features will remain unaddressed. In many cases, inclusion of both types of analyses can elucidate issues of functional connectivity better than either method applied in isolation. If applied judiciously, such techniques may dramatically increase our understanding of the dynamic functional interactions that characterize large-scale distributed neural networks.
Footnotes
Acknowledgements
The authors thank the technical staff of the PET Imaging Service; Satoshi Minoshima (University of Michigan) for providing analysis software; Joel Lee for computer assistance and advice; Patricia Pardo, Michael Kuskowski, and anonymous reviewers for helpful comments on a draft of this manuscript; and our volunteer subjects for their patience and generosity.
*
Structural equation modeling also might be included in this list, although technically it produces estimates of “effective connectivity” (which requires a model of the direction of the influence between two regions) rather than functional connectivity (which requires no such model; e.g., McIntosh and Gonzales-Lima, 1992; 1994; Friston, 1994).
†
In small data sets, Spearman's rho or Kendall's tau may be preferable to Pearson product moment correlations because of the sensitivity of Pearson product moment correlations to outliers. In the present data set, there was little evidence of outliers or nonlinear relations, and post hoc application of Spearman's rho produced negligible changes in the pattern of correlations.