Abstract
The authors used functional magnetic resonance imaging (fMRI) to determine whether acute intravenous (IV) cocaine use would change global cerebral blood flow (CBF) or visual stimulation-induced functional activation. They used flow-sensitive alternating inversion recovery (FAIR) scan sequences to measure CBF and blood oxygen level-dependent (BOLD) sensitive T2* scan sequences during visual stimulation to measure neuronal activation before and after cocaine and saline infusions. Cocaine (0.6 mg/kg IV over 30 seconds) increased heart rate and mean blood pressure and decreased end tidal carbon dioxide (CO2). All measures returned to baseline by 2 hours, the interinfusion interval, and were unchanged by saline. Flow-sensitive alternating inversion recovery imaging demonstrated that cortical gray matter CBF was unchanged after saline infusion (–2.4 ± 6.5%) but decreased (–14.1 ± 8.5%) after cocaine infusion (n = 8, P < 0.01). No decreases were detected in white matter, nor were changes found comparing BOLD signal intensity in cortical gray matter immediately before cocaine infusion with that measured 10 minutes after infusion. Visual stimulation resulted in comparable BOLD signal increases in visual cortex in all conditions (before and after cocaine and saline infusion). Despite a small (14%) but significant decrease in global cortical gray matter CBF after acute cocaine infusion, specific regional increases in BOLD imaging, mediated by neurons, can be measured reliably.
Keywords
Cocaine addiction is a serious social problem with few effective treatment strategies. There is limited knowledge of the neurobiology of cocaine addiction, and such knowledge might help develop more specific treatments. We wished to apply the newly developed technology of functional magnetic resonance imaging (fMRI) to study the mediating neuroanatomy of the cognitive and rewarding aspects of cocaine abuse in cocaine-dependent subjects. However, we were concerned that the effects of cocaine on systemic and cerebral hemodynamics might interfere with the acquisition of meaningful local neuronal activation maps because fMRI signals depend on intact coupling between metabolic demand and cerebral blood flow (CBF).
The most widely used fMRI method, blood oxygenation level-dependent (BOLD) scanning, uses changes in venous oxygenation of brain tissue as an indirect marker of detecting neuronal activation (Kwong et al., 1992; Bandettini et al., 1992; Blamire et al., 1992; Ogawa et al., 1992; Ogawa et al., 1990; Turner et al., 1991). Positron emission tomography studies have demonstrated that regional neuronal activation due to sensory stimulation results in an increase in CBF that exceeds the increase in oxygen metabolic demand of that region of brain tissue (Fox and Raichle, 1986; Fox et al., 1988). Enhanced CBF leads to a decrease in local venous deoxyhemoglobin concentration. Because deoxygenated hemoglobin is a paramagnetic endogenous contrast agent that decreases local signal intensity (Ogawa et al., 1992), enhanced CBF leads to increased BOLD signal.
Cocaine has several direct and indirect effects on the cerebral vascular bed that may influence the neuronal—vascular coupling. Most importantly, acute cocaine administration increases heart rate and may alter systemic vascular resistance which, despite central nervous system autoregulatory mechanisms, may influence CBF. Cocaine-induced hyperventilation (Richard et al., 1993) may reduce the partial pressure of carbon dioxide (Pa
Recent human neuroimaging studies investigating the effects of acute cocaine infusion suggest that this drug may cause fairly widespread changes in glucose metabolism and CBF. A 14% decrease in global glucose use after acute cocaine infusion has been demonstrated using fluorodeoxyglucose positron emission tomography (London et al., 1990). In this study, 26 of 29 brain regions (including all neocortical areas, basal ganglia, portions of the hippocampal formation, thalamus, and midbrain) that were investigated showed a significant decrement in regional cerebral metabolic rate for glucose. Decreased relative CBF in multiple brain regions, including the basal ganglia, inferior cingulate, and inferior frontal cortex, using single photon emission computed tomography (SPECT) imaging also has been reported (Pearlson et al., 1993). More recently, an approximate 30% global decrease in absolute CBF has been reported using SPECT and a microsphere model in a small cohort of cocaine-dependent subjects (Wallace et al., 1996). However, a conflicting recent study reports increased global CBF when assessed using the xenon 133 inhalation technique with SPECT imaging after acute cocaine infusion (Mathew et al., 1996).
These observations suggest the possibility that cocaine effects on CBF may decrease the sensitivity of fMRI signals associated with BOLD imaging during and immediately after acute cocaine infusion. We used a recently developed fMRI pulse sequence, flow-sensitive alternating inversion recovery (FAIR) (Kwong et al., 1993; Kwong et al., 1994; Kwong et al., 1995; Kim, 1995), to measure global effects of cocaine on CBF. The FAIR technique uses endogenous water protons as a tracer to detect relative CBF changes. Pairs of inversion recovery images are acquired by interleaving slice-selective inversion and nonselective inversion. The signal enhancement (FAIR image) measured by the difference between the paired images is directly proportional to blood flow. Relative changes in images collected during two different conditions estimate CBF changes (Kwong et al., 1995; Kim, 1995). This method has been used previously to map functional activation on the basis of changes in CBF after visual stimulation (Kwong et al., 1993) and finger movements (Kim, 1995).
In addition to directly assessing the effect of cocaine on CBF using FAIR imaging, we also were interested in determining the stability of the BOLD signal in the setting of the many nonspecific effects of the drug on the cerebral vascular system. We used a high-contrast, moving visual stimulus that reliably produces near maximal fMRI activation in primary sensory occipital cortex (Kwong et al., 1992, Tootell et al, 1995) to determine the effect of cocaine on BOLD activation.
The goals of this study were 1) to record the detailed physiologic response to a cocaine infusion in the fMRI environment; 2) to determine the effect of the cocaine-induced physiologic changes on global CBF as determined by the FAIR technique and compare this finding with the effects of cocaine on the global BOLD signal; and 3) to quantify the effects of cocaine on functional activation in occipital cortex using a BOLD scan sequence in a robust, well-characterized visual stimulation paradigm. The results of this study support the specificity of the findings we have reported on the regional brain activation due to acute cocaine administration (Breiter, et al., 1997).
MATERIALS AND METHODS
Subject characterization
Seventeen actively abusing cocaine-dependent subjects between the ages of 27 and 46 years (34.5 ± 4.6 years, mean ± standard deviation [SD]; 13 men, 4 women; 10 white, 7 black) were recruited by advertising. Substance abuse and other psychiatric symptoms were evaluated using standard instruments (Addiction Severity Index [McLellan et al., 1980], Mini-Structured Clinical Interview for DSM-III/R [American Psychiatric Association, 1987], Hamilton Anxiety Scale and the Hamilton Depression Scale [Hamilton, 1959; 1960]). Evidence or history of physical disease, history of head trauma with loss of consciousness, family history of sudden cardiac death or cardiac disease, or fulfillment of criteria for any axis I psychiatric diagnosis other than nicotine and/or cocaine dependence, alcohol or marijuana abuse, or abnormal response to a test dose (0.2 mg/kg intravenously over 30 seconds) of cocaine were exclusionary criteria. Subjects reported heavy, long-term cocaine use (median 7.8 ± 6.0 years; range, 2–25 years) with current use being 16 ± 8.2 days/month (range, 6–30 days). Smoking was the current primary route of self-administration for most subjects; many had abused cocaine intravenously in the past. None were receiving or seeking treatment for substance abuse. All subjects had a urinalysis confirming recent cocaine use but were abstinent from cocaine and alcohol for at least 19 to 28 hours before each imaging session. All were right handed, as assessed by self-report. Subjects gave informed consent and the experimental protocol was approved by the Subcommittee on Human Studies at the Massachusetts General Hospital (MGH).
Experimental design
Each subject was admitted to the MGH General Clinical Research Center (GCRC) for the screening procedures; those meeting all criteria were boarded overnight on the unit in preparation for imaging the following day. In the morning, each subject had bilateral intravenous catheters placed (right forearm for cocaine infusion, left forearm for serial venous blood sampling for quantitative cocaine levels). Scanning was performed between 11
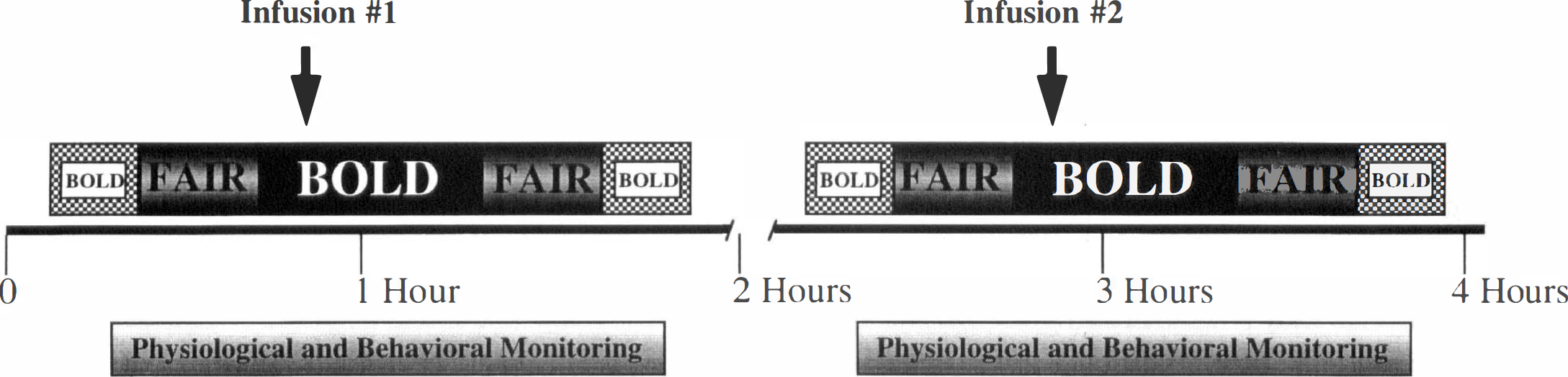
Schematic diagram of experimental paradigm. BOLD, blood oxygen level-dependent; FAIR, flow-sensitive alternating inversion recovery.
Sequential 3-mL venous blood samples were collected immediately before and at 1, 3, 5, 10, 15, 30, 60, 90, and 120 minutes after each infusion. The 120-minute sample for the first infusion also was the preinfusion sample for the second infusion. Cocaine quantitative assays were performed by the MGH Clinical Chemistry Laboratory (Puopolo et al., 1992).
Physiologic monitoring
Physiologic monitoring was conducted using an InVivo Omni-Trak 3100 patient monitoring system (Invivo Research, Orlando, FL, U.S.A.) modified to permit on-line computer acquisition of physiologic measurements. Electrocardiogram, heart rate (HR), end-expiratory carbon dioxide (ETCO2), and noninvasive systemic mean blood pressure (MBP) were measured continuously. The temporal resolution of the system for sampling blood pressure was once every 2 minutes. Values for each of the other parameters were sampled once per second, except for the electrocardiogram trace, which was digitized at a rate of 100 Hz.
The measured physiologic parameters were ported to a Macintosh Power PC 7100 (Cupertino, CA, U.S.A.) running a custom National Instruments LabView data acquisition program (National Instruments, Austin, TX, U.S.A.). This program allowed simultaneous acquisition of 1) digitized analog electrocardiogram trace, 2) the scanner trigger pulse, which indicated when the gradient coils of the magnet were firing, and 3) physiologic measures from the InVivo system.
Precautions taken to ensure safe conduct of the study included use of advanced cardiac life support (ACLS) trained personnel, frequent running of mock codes, and the presence of a cardiologist at the time of all infusions, whose sole responsibility was to monitor subject safety. Because of magnetohydrodynamic effects on the electrocardiogram tracing, a baseline rhythm strip was obtained before each drug infusion, to which all subsequent tracings were compared.
Imaging parameters
Images were collected on a GE 1.5T Signa imager, retrofitted for echo planar imaging by Advanced NMR, Inc. (Wilmington, MA, U.S.A.), using a quadrature head volume coil for signal reception. A dental impression bite bar was used to help stabilize head position and minimize movement between scans. A sagittal localizer scan was performed for placement of the experimental slices, followed by an automated shim procedure to improve Bo magnetic field homogeneity (Reese et al., 1995). An echo planar imaging multislice T1-weighted spin echo high-resolution scan for anatomic referencing of functional data was performed.
Functional scans were obtained to examine task-related differences in cerebral activity during visual stimulation using a T2*-weighted asymmetric spin echo sequence (the BOLD scans) (Δ τ [offset time] = −25 ms, TR [repetition time] = 3000 ms, TE [echo time] = 70 ms, Flip = 90°, in plane resolution = 3.1 × 3.1 mm, through-plane resolution = 8 mm). Fifteen contiguous axial slices were chosen to extend from the superior surface of the cortex through the temporal lobes for the BOLD scans. Sixty images per slice were collected for a total scan duration of 3 minutes.
Flow-sensitive alternating inversion recovery scans were collected for a single slice, using the center slice of the 15 slices used for the BOLD scans so that all imaging could be performed without movement of the subject within the bore of the magnet. This slice plane sampled frontal, parietal, and cingulate cortical regions. The FAIR imaging consisted of an inversion recovery sequence (TR time = 3500 ms, TE = 40 ms, TI [inversion time] = 1300 ms, in plane resolution = 3.1 × 3.1 mm, through-plane resolution = 8 mm). One hundred twenty-eight images (64 pairs) of the single 8-mm slice were collected during each 7:38 minute scan.
The BOLD imaging parameters during the infusion scan were identical to those reported previously for the visual stimulation BOLD scans, with the exception of a longer TR (8000 ms) and longer scanning duration (136 images per slice, total scan time 18:24 minutes) (Breiter et al., 1997).
Visual stimulation paradigm
Subjects lay supine in the magnet looking up into a mirror aimed at a rear-projection screen situated at chin level. Images were projected from outside the scanner room, through a collimating lens, onto the rear-projection screen. S-VHS video signals were generated from a Macintosh computer equipped with a Radius VideoVision interface.
The visual stimulus used was a circular black-and-white checkerboard pattern counterphase flickering at 4 Hz. This stimulus optimally evokes fMRI measurable cortical gray matter activity in both primary and secondary visual areas, including the motion sensitive area mediotemporal (Tootell et al., 1995; Sereno et al., 1995). The experimental paradigm consisted of alternating 45-second blocks of rest (darkness) and visual stimulation. The subject was instructed to remain still with eyes open and fixed on the center of the screen throughout the 3-minute scan.
DATA ANALYSIS
Physiology
Continuously collected physiologic measures were matched to concurrently collected scanner trigger pulses to determine values at relevant time points in the study. Data were analyzed first by an analysis of variance (ANOVA) with time as the factor. When significant F values were obtained for one of the physiologic measures, the individual time points were compared by post hoc Student's t tests to determine at what times the change from baseline, defined as the value just before the infusion, was significant. The Bonferroni correction for multiple comparisons was used; the criteria for significance at the 0.05 level was P < 0.007.
Motion correction
All data sets were motion-corrected using a software registration algorithm based on the work of Woods and colleagues (Jiang et al., 1995; Woods et al., 1992). As discussed previously (Breiter et al., 1996), the motion correction process often causes artifact in the first and last slices in a stack of slices. In the case of a single slice (as in the FAIR data sets), the algorithm minimizes within plane motion only and does not corrupt the slice data. Because of the complexity of MRI signal dependence on bulk movement, this motion correction algorithm remains an approximation. As a result, any data set evidencing excessive motion (movement > 1.5 mm for the FAIR scans, and > 3.5 mm for the BOLD scans) was excluded from further analysis.
Flow-sensitive alternating inversion recovery data
Flow-weighted images were obtained by subtracting the flow-insensitive images from the flow-sensitive images. The resulting subtraction images were used as an index of steady-state flow values (Kim, 1995; Kwong et al., 1995). The percent change at each pixel between steady state flow measured during the preinfusion scan and the postinfusion scan was used to estimate the percent CBF change due to the infusion (Kwong et al., 1995; Kim, 1995). Regions of interest (ROIs) (whole slice, cortical gray matter and white matter) were drawn on the first FAIR image collected for each subject. The average signal intensity value for the averaged pixels within a given ROI was calculated for each of the four scans from each subject. To determine the effect of cocaine on CBF, we calculated the percent change between the preinfusion and postinfusion FAIR scans in the average signal intensity for each of these ROIs. The CBF measurements made by this FAIR technique are relative values; however, the percent change in this measurement after cocaine infusion provides a reasonable estimate of the absolute change in CBF.
The same ROIs generated for the FAIR data analysis were used to determine the average signal intensity of the first 38 images (5 minutes of preinfusion resting state) and the last 38 images (last 5 minutes of BOLD scan). The percent difference between these values for each subject was used as the measure of global BOLD signal change due to the infusion. Statistical significance was determined using Student's t tests to compare the change in CBF after saline (pre minus post) and cocaine (pre minus post) infusion with degrees of freedom (n − 1) determined by the number of subjects (n) in the comparison.
Of the 17 subjects studied, 8 subjects had interpretable FAIR data sets for both the saline and cocaine infusions (2 did not have saline infusions, 6 were excluded because of excessive movement, and 1 was excluded because of technical problems with the scanner). One of the studies included in the final analysis was collected during a replicate imaging session; data from the original imaging session had to be discarded; thus, the subject is represented only once in the final cohort.
Visual stimulation data
Motion-corrected images were transformed into statistical maps using parametric unpaired Student's t tests to compare, at each pixel, the mean of the time points in the stimulated versus rest conditions. The image data were smoothed, using a two-dimensional, center-weighted kernel analogous to a Hanning filter, so that each pixel was correlated with its neighboring pixels. This process decreased the effective spatial resolution to 6 × 6 × 8 mm3. The statistical map was transformed to a P value map and displayed in pseudocolor (as a useful surrogate for a map of neural activity) and superimposed on a gray-scale high resolution T1-weighted echo planar image. The resultant maps were examined to identify regions that showed significant visual stimulation-related signal change (activation) (Breiter et al., 1996; Breiter et al., 1997).
The values used to make quantitative preversus postsaline and cocaine infusion comparisons of fMRI signal change were obtained using an ROI that represented equally weighted activation in each of the four scans. Thus, for each subject, the four visual stimulation BOLD scans were averaged together, and the averaged data were used to generate an averaged Student's t test statistical map. This averaged t map was adjusted to display all voxels with a threshold above to the conservative P < 10−6 level, which would represent the Bonferroni correction for every voxel we acquired in the brain (Breiter, et al., 1997), and was used to draw an ROI for each slice containing significantly activated pixels in the primary visual cortical region. These ROIs then were used to calculate the percent signal change in each of the four visual stimulation scans. A weighted average percent signal change (% D) from the ROIs was calculated for each of the four visual stimulation BOLD scans. Two-way ANOVA was used to determine significant change due to drug and condition.
Eight of the 17 subjects had interpretable visual stimulation BOLD scans for both the saline and cocaine infusions (2 did not have saline infusions, 3 were excluded because of technical problems with the scanner, and 4 were excluded because of flawed timing of the visual stimulus presentation). One of the studies included in the final analysis was collected during a replicate imaging session because their data from the original imaging session had to be discarded.
RESULTS
Cocaine levels
Plasma samples taken before the first infusion demonstrated absence of residual cocaine in all of the subjects studied. Peak plasma cocaine leve (Cmax) after the cocaine infusion ranged from 197 to 893 μg/L with a mean of 389 ± 233 (n = 7 subjects). The time to peak varied from 3 to 15 minutes in the group.
Physiology
Physiologic monitoring during the fMRI study showed the expected effects of cocaine on the cardiorespiratory system (Figs. 2 and 3). In all subjects, the HR began to increase within the first minute after cocaine infusion; MBP increased more slowly and less dramatically, and the ETCO2 dropped slightly after a delay. The changes in HR, MBP, and Δ ETCO2 after cocaine infusion were statistically significant according to the ANOVA criteria (P < 0.0001, P < 0.002 and P < 0.0001, respectively). Figure 2 shows the averaged physiologic data from all subjects at selected time points most relevant to the interpretation of the fMRI data. Figure 3 shows the continuous physiologic data (HR, MBP, and Δ ETCO2) recorded from a representative subject during each of the two scanning epochs (saline and cocaine infusions). In both the averaged and the individual data, note the stability of the measurements over the duration of the saline infusion scanning epoch as well as during the preinfusion phase of the cocaine infusion scanning epoch.
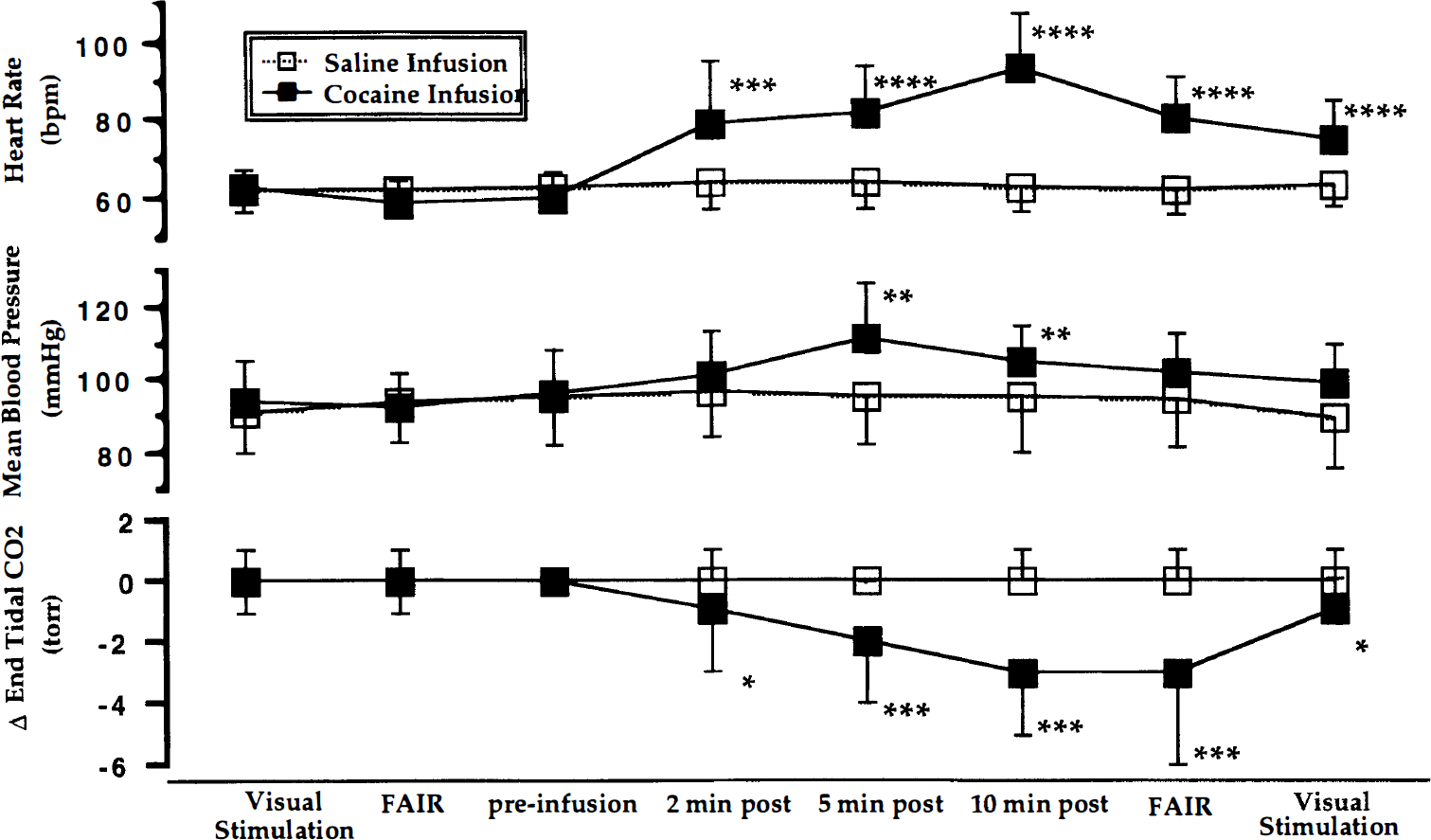
Averaged physiologic responses at specific time points relative to the cocaine (▪) and saline (□) infusions are shown; heart rate (HR) in the top portion, mean blood pressure (MBP) in the center, and change in end-expiratory carbon dioxide (ETCO2) at the bottom. The measures shown are at discrete time points; the connecting lines do not represent continuous data but rather serve to facilitate comparisons between data points. Each graph includes measurements taken during the preinfusion visual stimulation blood oxygen level-dependent (BOLD) scan, during the preinfusion flow-sensitive alternating inversion recovery (FAIR) scan, during the infusion BOLD scan (preinfusion and at 2, 5, and 10 minutes postinfusion), and during the postinfusion FAIR and visual stimulation scans. Note how stable the physiologic measures are throughout the saline infusion and during the preinfusion interval before cocaine. Data were analyzed first by analysis of variance, with time of measurement as the factor. When significant F values were obtained for one of the physiologic measures, individual time points were compared by post hoc Student's t tests to determine at what times the change from baseline was significant. The Bonferroni correction for multiple comparisons was used; the criteria for significance at the 0.05 level was P < 0.007. The mean ± standard deviation is presented for each measure for the entire group (n = 17 for cocaine, n = 14 for saline) studied. There were no significant differences between the values shown here and those of the subgroups used for the final quantitative visual stimulation BOLD or FAIR studies. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 10−7.
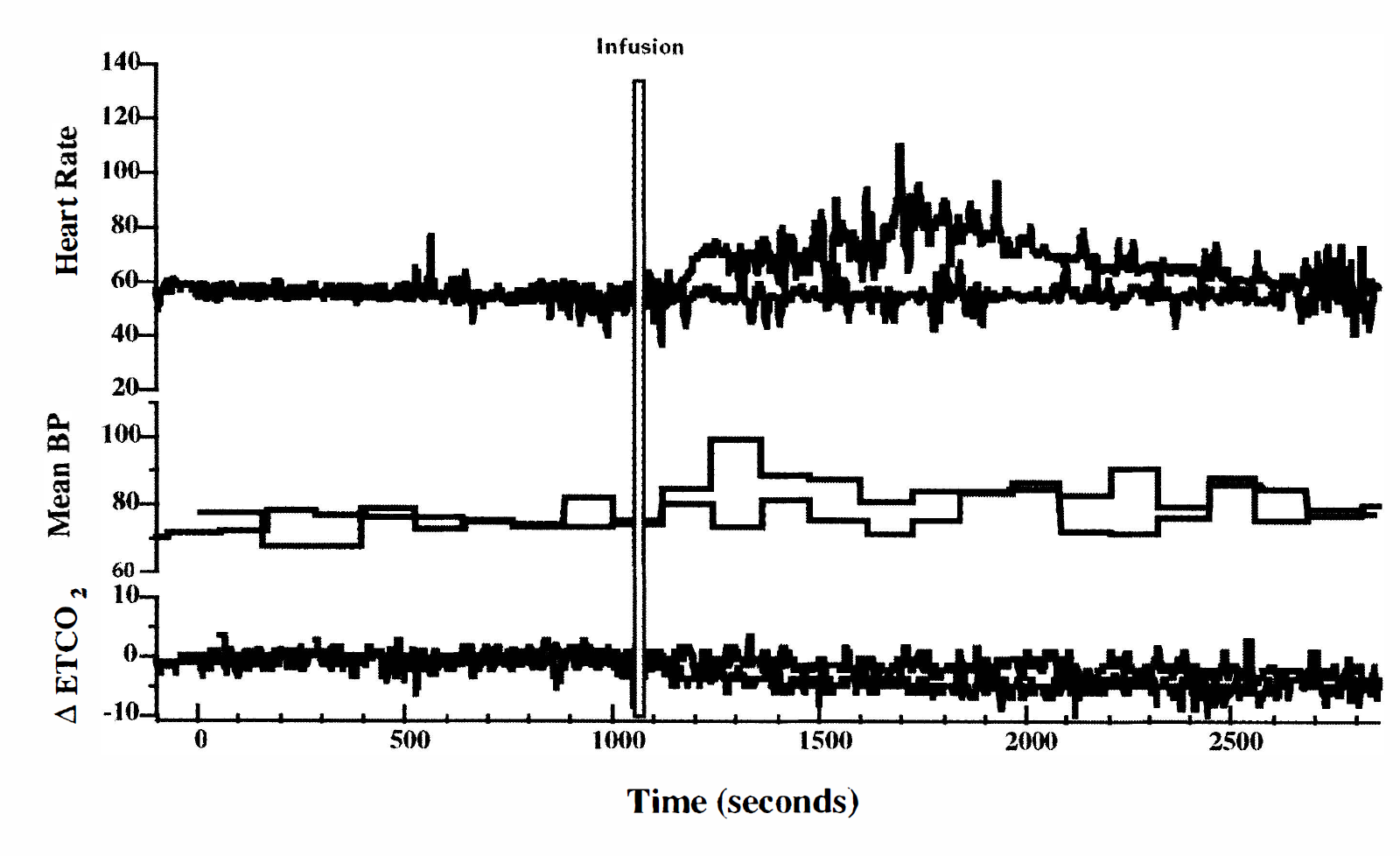
Continuously recorded physiologic measures from a representative subject collected during the saline infusion epoch (dotted lines) and cocaine infusion epoch (solid lines). Heart rate (HR) is shown in the top portion, mean blood pressure (MBP) in the center, and change in end-expiratory carbon dioxide (ETCO2) at the bottom. The 30-second infusion is indicated by the thin gray bar. The HR begins to increase rapidly within the first minute after the cocaine infusion; the increase in MBP is slower and less dramatic, as is the delayed decrease in ETCO2. Note the greater variability of the measures in the interval immediately following the cocaine infusion.
Cocaine (n = 17) caused the HR to rapidly increase from a preinfusion value of 60 ± 7 bpm to 79 ± 16 bpm at 2 minutes postinfusion (P < 0.0001), to 82 ± 12 bpm at 5 minutes postinfusion (P < 10−6), to 93 ± 14 bpm at 10 minutes postinfusion (P < 10−8). By the time the postcocaine infusion FAIR scan was collected, the HR (81 ± 11 bpm) had begun to return to baseline; however, it still was elevated significantly over the HR (59 ± 6 bpm) at the time of the preinfusion FAIR scan (P < 0.0001). Similarly, the HR continued to return toward baseline as the postcocaine infusion visual stimulation BOLD scan was collected; however, the HR still was elevated significantly over the preinfusion visual stimulation time point (62 ± 4 bpm compared with 75 ± 10 bpm, P < 0.01). Normal sinus rhythm was observed in all subjects throughout the study.
Mean blood pressure increased slightly from 96 ± 12 torr before the cocaine infusion to 101 ± 12 torr at 2 minutes postinfusion (P < 0.11, not significant [NS]), then up to 111 ± 15 torr at 5 minutes (P < 0.002) before starting to slowly decrease. The MBP still was slightly but not significantly elevated by the time the postcocaine infusion FAIR and postcocaine visual stimulation scans were collected.
ETCO2 measures do not reflect absolute measures of PaCO2; however, ETCO2 provides a reliable estimate of true changes in PaCO2. Because of this, we chose to analyze and display the data in the figures in terms of the change in ETCO2 (Δ ETCO2) from the baseline measure for each individual. The absolute values of ETCO2 decreased slowly from a preinfusion value of 39 ± 4 mm Hg to 36 ± 4 mm Hg by 10 minutes and remained at this level during the time the postinfusion FAIR scan was collected. The Δ ETCO2 was significantly different between the pre- and postcocaine infusion FAIR (P < 0.001) and visual stimulation BOLD (P < 0.05) scans.
There was no change in any of the measured physiologic parameters during the scanning epoch in which saline was infused. Note the greater variability in the group data points after the cocaine infusion in all three measures. This indicates the interindividual variability not only in magnitude of change but also in the duration of the response. In all subjects, all measures had returned to baseline by 2 hours after the cocaine infusion. Statistical analysis of these physiologic measures for the subgroups used for the FAIR data analysis and the visual stimulation BOLD data analysis did not differ from the whole group.
Flow-sensitive alternating inversion recovery imaging data
Eight subjects had interpretable data from matched pre- and postcocaine and pre- and postsaline FAIR scans. The three ROIs generated for each subject to analyze the FAIR data included whole slice (4311 ± 363 mm2), cortical gray matter (2022 ± 321 mm2) and white matter (354 mm2). These are shown for a typical subject in Fig. 4. The slice plane included regions of frontal, cingulate, and parietal cortex.
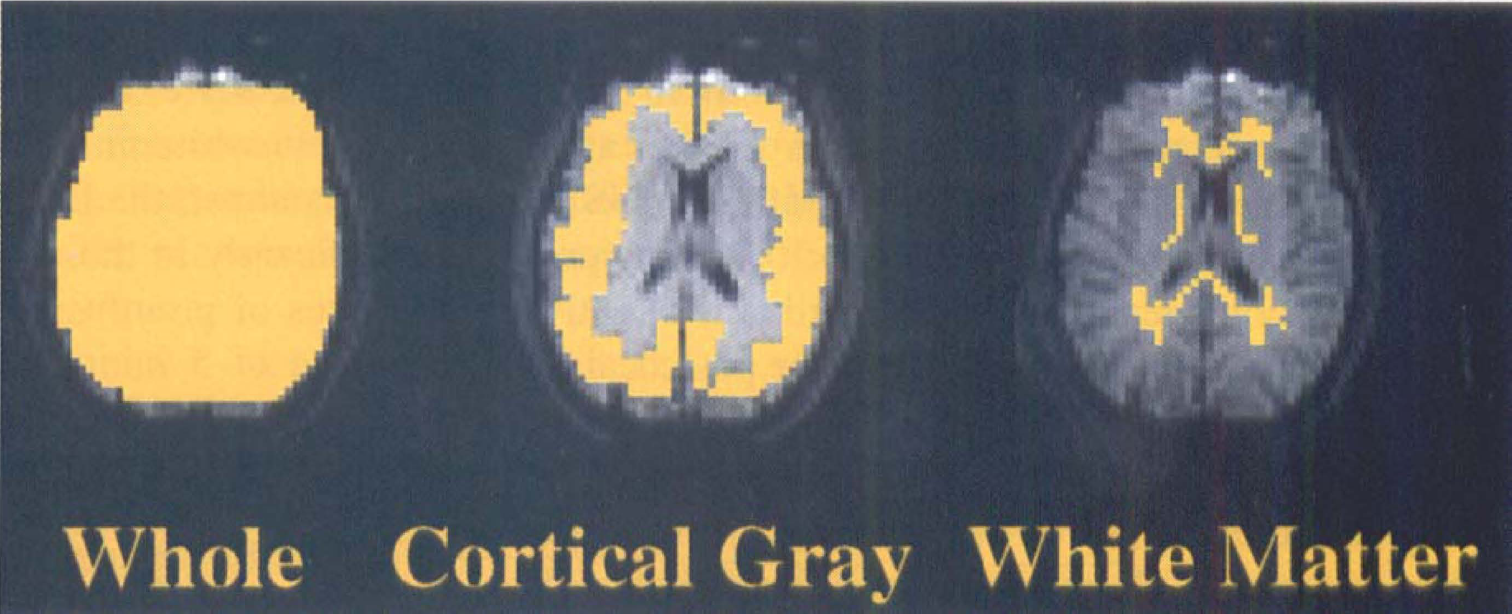
The regions of interest (ROIs) used in the quantitative flow-sensitive alternating inversion recovery (FAIR) data analysis for one representative subject are shown in yellow superimposed on first FAIR image. In this subject, the ROI for the whole slice was 4311 ± 363 mm2, cortical gray matter was 2022 ± 321 mm2, and white matter was 354 mm2. The ROIs were used to calculate the percent change pre- versus postinfusion for both the FAIR and blood oxygen level-dependent (BOLD) scans.
By FAIR imaging, both whole-slice and cortical gray matter CBF were unchanged (–3.7 ± 5.3% and −2.4 ± 6.5%, respectively) after saline infusion, but decreased (–13.3 ± 6.8% and −14.1 ± 8.5%, respectively) after cocaine infusion (whole slice P < 0.01, cortical gray matter P < 0.01; Table 1). This effect was not seen when the ROI included only areas of white matter (Table 1). Increasing the cohort to include subjects with usable data from only one of the two infusions did not alter the results; for n = 9 saline infusions and n = 14 cocaine infusions, there was a significant decrease in FAIR measured CBF after cocaine but not saline in the whole slice (–12.7 ± 7.1%, P < 0.001) and in cortical gray matter (–14.0 ± 8.1%, P < 0.0006). This decrease was not detected in white matter (—5.8 ± 17.6%).
FAIR percent change preinfusion versus postinfusion
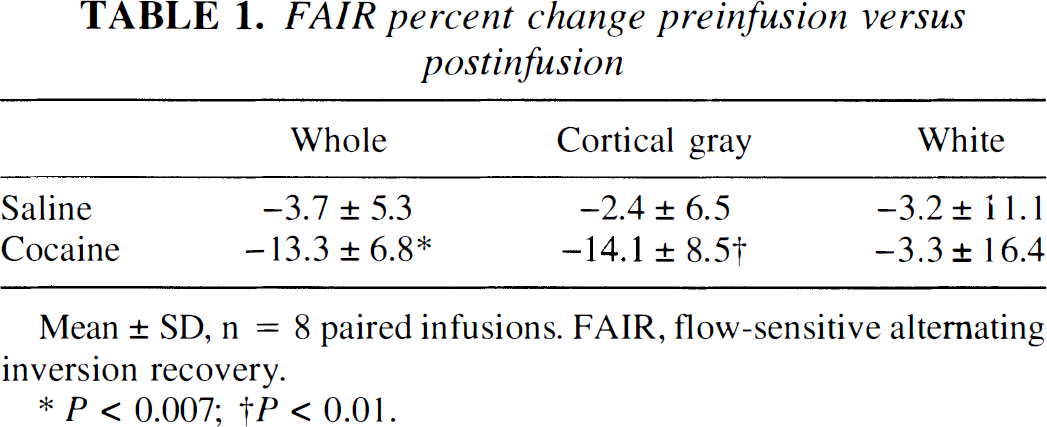
Mean ± SD, n = 8 paired infusions. FAIR, flow-sensitive alternating inversion recovery.
P < 0.007
P < 0.01.
In contrast, the BOLD signal intensity did not change significantly in these same ROIs when the average of 5 minutes of preinfusion images were compared with the average of 5 minutes of postinfusion images after cocaine or saline infusions (n = 8, Table 2). The percent signal change was less than 0.5%.
BOLD percent change preinfusion versus postinfusion
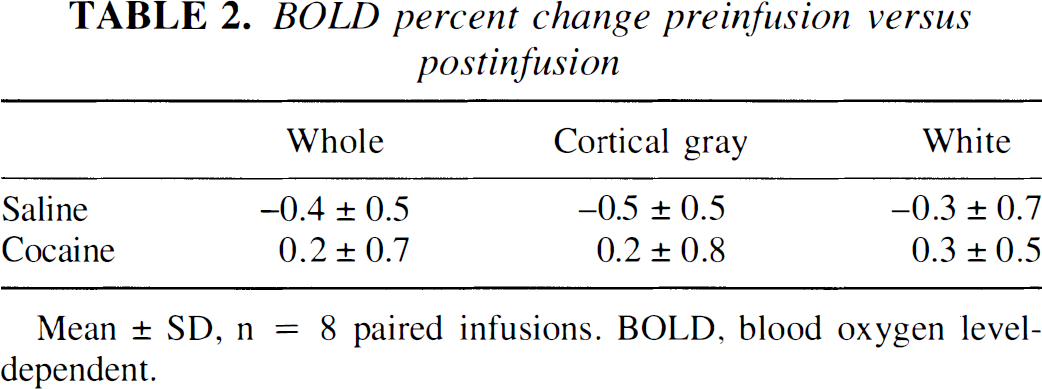
Mean ± SD, n = 8 paired infusions. BOLD, blood oxygen level-dependent.
Visual stimulation data
All subjects showed specific fMRI signal increases in the primary visual cortical area that temporally correlated with the stimulus presentations. Quantitative analysis showed no significant difference in the visual stimulation-induced fMRI signal increase in primary visual cortex after cocaine or saline infusion (Table 3, Fig. 5). The marked stability of this activation across multiple scans for each subject is apparent in the group data presented in Table 3. The range of activation that was observed (0.77–2.97%), including all four scans from each of the eight subjects analyzed, was well within that reported by others (Kwong et al., 1992, Tootell et al., 1995) using this type of paradigm. Figure 5 presents the data from a representative subject; the Student's t test statistical maps of a single slice, which include a portion of the primary visual cortex from each of the four visual stimulation scans, thresholded to the conservative P < 10−6 level, are shown superimposed on the high-resolution T1-weighted scan. The variance in the fMRI signal itself did not change after cocaine infusion for either the baseline (dark) or activation (flashing lights) condition (n = 7).
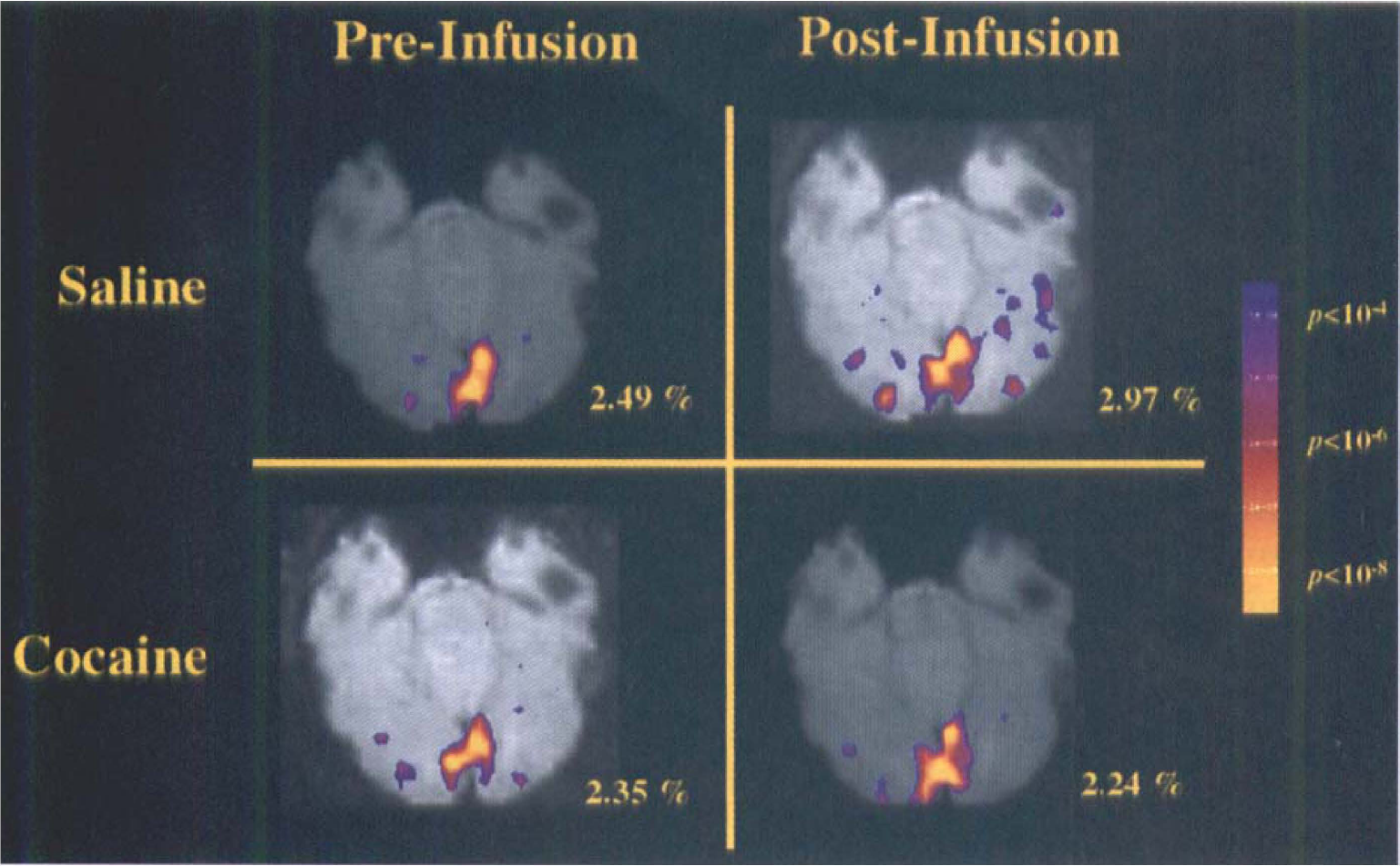
Demonstration that visual stimulation produces equivalent functional magnetic resonance imaging (fMRI) blood oxygen level-dependent (BOLD) activation in primary visual cortex in the presence or absence of cocaine. Quantitative and qualitative assessment of a single subject is displayed in these Student's t test maps from each of the four visual stimulation scans (before and after the saline and cocaine infusions), thresholded to the conservative P < 10−6 level and displayed superimposed on the first image of the BOLD scan from that slice, which includes a portion of the primary visual cortex. The percent signal change is indicated adjacent to each map.
Percent change in BOLD signal intensity in primary visual cortex during visual stimulation

Mean ± SD, n = 8 paired infusions. BOLD, blood oxygen level-dependent.
DISCUSSION
The physiologic responses to the 0.6-mg/kg intravenous cocaine infusion reported in the present study are in close accord with previously published studies in experienced cocaine abusers (Fischman and Schuster, 1982; Fischman et al., 1985; Foltin and Fischman, 1991; Mathew et al., 1996). All subjects responded to the drug with a rapid increase in HR that slowly returned to resting rate by 45 to 90 minutes, a moderately fast but more modest rise in MBP that also returned to baseline levels by 45 to 90 minutes, and a slow-onset, small decrease in ETCO2. Cocaine has been shown to increase systolic BP with less effect on diastolic BP (Foltin et al., 1995). Because our noninvasive pressure transducer only measures MBP, the apparent cocaine effect on BP may appear smaller than reported in other studies (Mathew et al., 1996). Although we did not measure respiratory rate systematically, we did note a rise in respiratory rate in several of the subjects similar to that reported in a recent study (Mathew et al., 1996).
Cocaine (0.6 mg/kg) reduced the relative CBF in cortical gray matter by 14%, as assessed by FAIR imaging. The cocaine effect was specific because the decrease was found in cortical gray matter but could not be detected in white matter and was not present after saline infusions. Global BOLD fMRI signal intensity in cerebral gray matter was unchanged at a comparable time point. At a time point very close to the FAIR-measured decrease in CBF, stimulus-induced BOLD activation in primary visual cortex was unchanged quantitatively by cocaine infusion. Thus, despite a measurable global cortical gray matter decrease in CBF after cocaine infusion, neuronally mediated specific regional increases in BOLD imaging still could be measured reliably.
The observation that acute cocaine infusion decreased global cortical gray matter CBF but did not change visual stimulation-induced increases in CBF, as measured by the BOLD imaging, is noteworthy. This result suggests that fMRI imaging with BOLD contrast is a viable method for future neuroimaging studies that use pharmacologic challenges with drugs that have effects on the cardiac and/or respiratory system.
A limitation of this study is that the quantitative assessment of cocaine effects on the visual stimulation BOLD activation was not performed at the time of maximal drug effect. Although not maximal, acute cocaine effects still were evident during the postinfusion visual stimulation BOLD scan, as evidenced by subjective ratings (data not shown), persistently elevated HR and MBP, and persistently decreased ETCO2.
Our results, a 14% decrease in CBF in frontal, parietal, and cingulate cortical areas 15 to 30 minutes after cocaine infusion, are consistent with several prior neuroimaging studies demonstrating widespread psychostimulant-induced decrease in CBF. Acute cocaine infusion decreased relative CBF, as assessed by SPECT imaging using the tracer technetium Tc 99m hexamethyl-propyleneamine-oxime (HM-PAO; Pearlson et al., 1993). Although these investigators measured CBF 1 and 5 minutes after drug infusion, the relative decrease in CBF was evident in multiple cortical regions. More recently, Wallace and colleagues (1996) reported a 30% decrease in absolute CBF at the time of peak cocaine subjective effects (within the first few minutes after intravenous infusion), using technetium Tc 99m HM-PAO SPECT with a modified microsphere model, in all brain regions assessed (including right and left sides of the caudate, putamen, globus pallidus, thalamus, anterior cingulate, prefrontal, precentral, and occipital cortex, as well as cerebellum). This seems consistent with our 14% decrease measured 15 to 30 minutes after infusion of a comparable dose of cocaine. Acute methylphenidate infusion decreased CBF globally, as assessed by positron emission tomography 15O-water studies (Wang et al., 1994). The CBF decrease was present by 5 to 10 minutes after methylphenidate infusion, persisted at 30 minutes postinfusion, and was seen in all cortical regions investigated. Our results conflict with those presented by Mathew and colleagues (1996), who used the xenon 133 inhalation technique with SPECT imaging to measure an increase in CBF after acute 0.3-mg/kg cocaine infusion. This increase still was evident at the 30-minute postinfusion time point, which corresponds to the time at which our FAIR data were collected. A possible explanation for the difference between studies is dose dependence because Mathew et al. (1996) studied a dose roughly half the amount that was used in the present study. Also, they did not determine cocaine use by toxicology screens before or by quantitative levels at the time of the study, so prior exposure or current intoxication are possible confounding factors.
The cocaine-induced decrease in CBF in cortical gray matter documented by the FAIR scans could have resulted, at least in part, from an increase in respiratory rate, which drove down the partial pressure of CO2 (PaCO2), signaling vasoconstriction in brain tissue (Guyton and Hall, 1996; Edvinsson et al., 1993). Coupling of CBF and neuronal oxygen metabolism is maintained in nonpathologic and normocapnic humans. However, this coupling is perturbed by changes in PaCO2 over the range of 20 to 80 mm Hg; across this range, CBF increases fourfold without a change in the cerebral metabolic rate of oxygen (CMRO2). Current literature suggests that in the human brain, a 1-mm Hg change in partial pressure of CO2 in the blood signals a 3% to 5% change in CBF (Maximilian et al., 1980; Kety and Schmidt, 1948). The 2-mm Hg decrease in mean ETCO2, measured at the time the postcocaine infusion FAIR scan was acquired, would predict approximately a 6% to 10% CBF decrease; such a decrease is consistent with our results. However, for each individual, the correlation between change in ETCO2 and percent change in FAIR after cocaine infusion did not reach statistical significance (r = 0.15, df 12, P < 0.6, NS). When the data from both saline and cocaine infusions were included in the analysis, the trend was concordant with the predicted relationship between PaCO2 and CBF, yet the correlation between the measures still did not reach statistical significance (r = 0.36, df 23, P < 0.09, NS) (Fig. 6). The calculated regression slope for the correlation between change in ETCO2 and percent change in FAIR is a 2.1% change per mm Hg (Fig. 6). We conclude that although it is possible that a respiratory-driven decrease in PaCO2 may explain partially the drop in CBF after cocaine infusion, this explanation still is questionable.
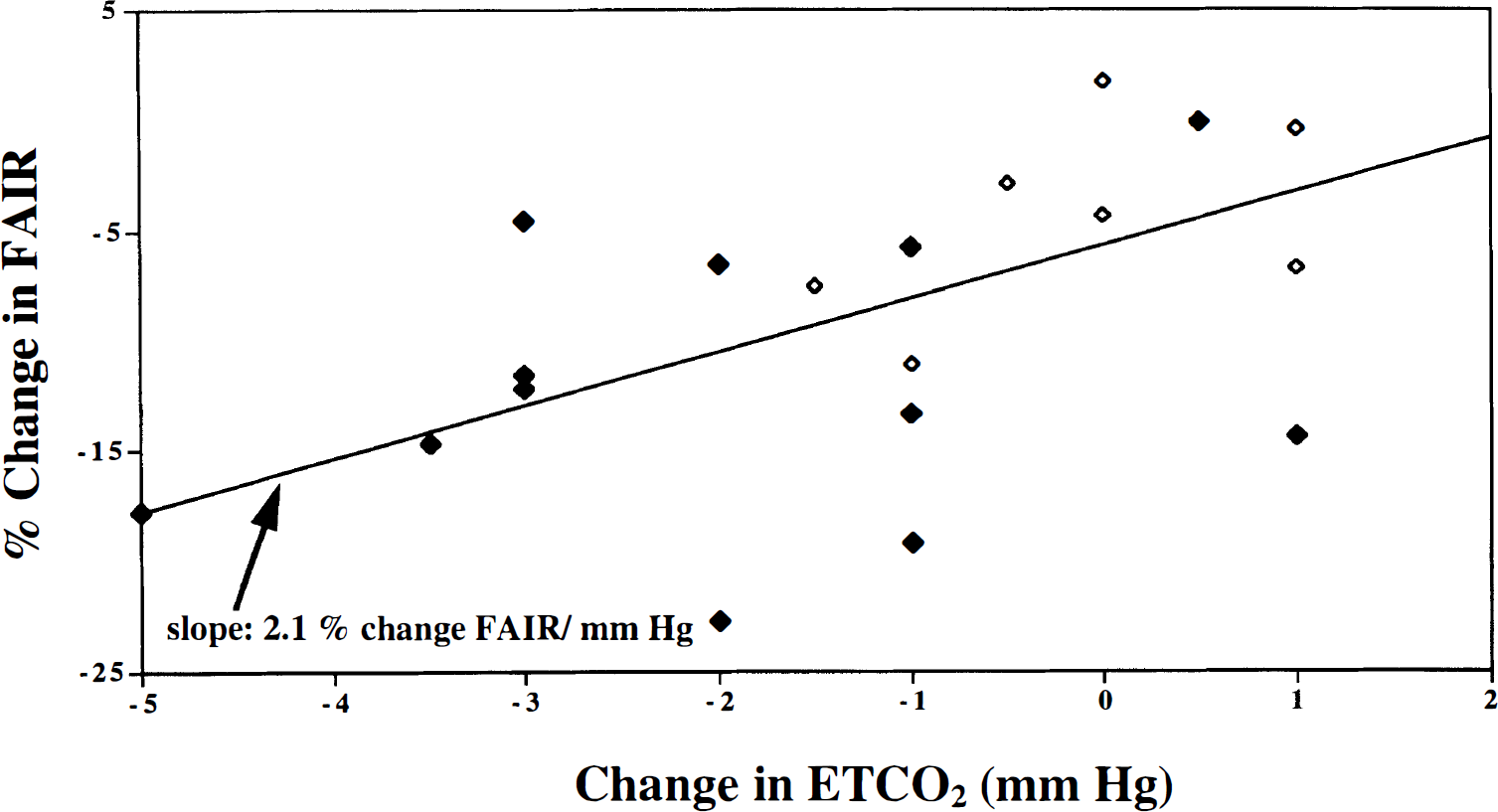
Correlation between change in end-expiratory carbon dioxide (ETCO2) and percent change in flow-sensitive alternating inversion recovery (FAIR) after cocaine (♦) and saline (⋄) infusions. Data from 14 individuals are presented, eight sets of which are represented twice, once for each infusion. Calculated slope of the regression line is 2.1% change in FAIR per millimeters of mercury change in ETCO2.
The absence of a concomitant BOLD decrease at a time point close to the occurrence of a FAIR decrease has several potential interpretations. The first is that the decrease in CBF was so small that it is detectable with fMRI only when using a flow-sensitive technique such as FAIR. Cerebral blood flow changes contribute less to changes in BOLD signal intensity than do changes in oxygenation level. However, we do not think that the absence of BOLD signal change is due to inadequate sensitivity. Monte Carlo simulations predict that each 1% change in T2*-weighted fMRI signal intensity corresponds to a CBF change of approximately 13% (Boxerman et al., 1995). A change of this magnitude in the T2* fMRI signal intensity measured 13 to 18 minutes after cocaine infusion should have been large enough to detect in our cohort, given the low noise in the data. We believe that a concomitant decrease in glucose metabolism after acute cocaine infusion (London et al., 1990) offset the decrease in BOLD signal intensity caused by decreased CBF, with the result that matching changes in blood flow and oxygen consumption left the hemoglobin oxygen saturation nearly unchanged. A similar decrease in glucose metabolism also has been demonstrated using fluorodeoxyglucose positron emission tomography studies of healthy human subjects after acute administration of another psychostimulant, D-amphetamine (Wolkin et al., 1987).
A possible alternative explanation is that the decrease in FAIR is an artifact caused by cocaine-induced vasospasm. Flow-sensitive alternating inversion recovery measurements of CBF can be affected by the transit time of blood through large vessels (Buxton et al., 1996). Cocaine is a known stimulant of the sympathetic nervous system and thus would activate the superior cervical ganglion that innervates cerebral vasculature. Stimulation of this pathway signals vasoconstriction; if autoregulatory mechanisms maintain constant CBF, the increased velocity of flow through the large vessels may create errors in the quantitation of perfusion. Although the FAIR technique is less sensitive to these artifacts than other inversion-preparation schemes, we cannot rule out this potential artifact.
CONCLUSION
These infusion experiments demonstrate that the effects of intravenous cocaine infusion do not obscure neuronally mediated regional changes in fMRI BOLD signal. Although a global CBF decrease was measured, visual stimulation produced regionally specific changes in primary visual cortex by fMRI BOLD imaging that were indistinguishable from baseline. We conclude that in the presence of cocaine, the fundamental coupling of neuronal activity to CBF remains intact. Our results strongly suggest that despite the cardiovascular and respiratory effects of cocaine in humans, fMRI scanning using the BOLD technique will allow visualization of neuronally mediated changes in BOLD signal corresponding to the subjective experience of the cocaine.
Footnotes
Abbreviations used
Acknowledgments
The authors thank Ken Kwong for consultation on the FAIR studies, John Baker for assistance in development of the physiologic data acquisition system, and the staff of the Mallinckrodt GCRC at Massachusetts General Hospital for excellent care of the research subjects.
