Abstract
Hypoxia—ischemia is accompanied by abundant corticosterone secretion that could exacerbate brain damage after the insult. The authors demonstrate that the steroid synthesis inhibitor metyrapone (150 mg/kg subcutaneously) suppresses the hypoxia—ischemia-induced rise of plasma corticosterone levels (17.3 ± 3.6 μg/dL) when compared with corticosterone-treated animals (72.2 ± 4.8 μg/dL) immediately after hypoxia—ischemia. In parallel, metyrapone reduced brain damage (P < 0.05). Moreover, none of the metyrapone-treated animals displayed seizures, whereas seven of eight corticosterone-treated animals had seizures after hypoxia—ischemia. Although corticosterone administration in metyrapone-treated animals elevated plasma corticosterone levels (39.0 ± 5.3 μg/dL), this did not result in a subsequent increase in brain damage and seizures when compared with metyrapone-treated animals. The authors conclude that metyrapone reduces brain damage and the incidence of seizures after hypoxia—ischemia but that this effect might partially be independent from its effect on modulating plasma corticosterone levels.
Prolonged exposure to corticosterone causes hippocampal dendritic atrophy and may induce loss of hippocampal neurons (Sapolsky et al., 1985; Woolley et al., 1990). A shorter period of exposure to high levels of corticosterone exacerbates loss of rat hippocampal neurons in response to hypoxia—ischemia, neurotoxins, and seizures (e.g., Sapolsky and Pulsinelli, 1985). Such insults themselves are likely to be accompanied by elevated corticosterone levels that could worsen the outcome of the insult. In accordance with this, we demonstrated previously that reducing plasma corticosterone levels during hypoxia—ischemia by down-regulating the hypothalamo-pituitary-adrenal axis reduces neuronal damage after hypoxia—ischemia (Krugers et al., 1995). Moreover, we showed that this was accompanied with a reduction in seizures after hypoxia—ischemia.
Metyrapone inhibits the conversion of deoxycorticosterone in the rat adrenal cortex, thereby preventing the stress-induced elevation of plasma corticosterone levels (Goodman Gilman et al., 1985). Hence, we hypothesize that metyrapone could reduce brain damage after hypoxia—ischemia by suppressing the insult-induced rise in corticosterone levels (Kemper et al., 1994; Smith-Swintoski et al., 1996). The present study was initiated to study whether metyrapone exerts a neuroprotective action and alters the incidence of seizures after hypoxia—ischemia. Also, in a preliminary study, we found that metyrapone caused anesthesia-like effects (Kemper et al., 1994). To establish this, we studied the effect of metyrapone on general activity of the animals.
MATERIALS AND METHODS
Animals and experimental treatments
All experiments were approved by the committee on animal bioethics of the University of Groningen. Male Wistar rats (250–300 g) were housed individually with a light—dark regimen (lights on from 8:00
Surgical procedures and hypoxia—ischemia
The animals were exposed to hypoxia—ischemia as described previously (Knollema et al., 1995; Krugers et al., 1995). Briefly, animals were anesthetized with halothane (0.9%). Next, the left arteria femoralis was cannulated for continuous blood pressure monitoring during the normoxia and hypoxia/ischemia periods (Hewlett Packard Pressure Transducer, Boeblingen, Germany), and the right jugular vein was cannulated for determination of corticosterone and blood glucose levels. Subsequently, the animals were intubated and ventilated (Infant Ventilator MK2, Loosco, Amsterdam, The Netherlands) for 10 minutes with 30% oxygen (O2) and 70% nitric oxide (N2O) (first normoxia period), for 20 minutes with 10% O2 and 90% N2O (hypoxia), and for 10 minutes with 30% O2 and 70% N2O (second normoxia period). During the hypoxia period, the left carotid artery was occluded (ischemia). The rectal temperature was maintained between 36.5°C and 37.5°C.
Histology
Twenty-four hours after hypoxia—ischemia, animals were anesthetized deeply with pentobarbital (50 mg/kg intraperitoneally). Next, they were perfused transcardially with 0.9% saline for 1 minute, followed by 4% paraformaldehyde in 0.1 mol/L phosphate-buffered saline (pH 7.4) for 10 minutes. Brains were removed from the skull and postfixed in 4% paraformaldehyde. Before cutting, the brains were cryoprotected by overnight storage in 30% sucrose in 0.1 mol/L phosphate buffer (pH 7.4), cut to 30-μm thick coronal sections at −15°C using a cryostat microtome, and collected in paraformaldehyde. The modified Gallyas silver impregnation procedure of Nadler and Evenson (1983), which labels damaged cells and their processes, was employed on free-floating sections, with all steps being identical and run simultaneously for all animals. For details of the procedure, see Ter Horst et al. (1995).
Brain damage was determined in coronal sections of different levels. Levels were 4.2, 5.7, 6.7, 7.7, and 9.2 mm from the interaural line (Paxinos and Watson, 1982). The damage was determined using bright-and-dark field microscopy (Knollema et al., 1995; Krugers et al., 1995). Camera lucida drawings of the silver staining in the ipsilateral side to the clamped artery were made on millimeter paper and used for damaged-area size calculations. Damage was expressed as the ratio between the area silver stained and the total area. The total damage per animal was calculated throughout the brain using the damage at the five levels and subsequently averaged per group. Data are expressed as mean ± SEM. Histology data, behavioral data, and plasma corticosterone levels were evaluated statistically, as described previously (Knollema et al., 1995; Krugers et al., 1995) using the (two-tailed) Student's t test. Significance level was P < 0.05.
Quantification of plasma corticosterone levels
Blood samples were taken in the second normoxia period. Subsequently, the samples were transferred to centrifuge tubes containing 10 μL heparin solution (500 units/mL) and centrifuged for 20 minutes at 3500 g. The supernatant was stored at −20°C for the corticosterone assay. Corticosterone was extracted from 75 μL of plasma using a liquid extraction method. Plasma corticosterone levels were quantified with high-pressure liquid chromatography in combination with ultraviolet detection (Shimizu et al., 1983). The absolute detection threshold for corticosterone in plasma was 8 ng/mL.
Behavior
We measured the activity of the animals before hypoxia/ischemia to test whether the different treatments affected locomotor activity. We recorded the number of steps and rearings during a 3-minute period in the staircase test (Willig et al., 1991).
Seizures
To establish whether modulation of steroid levels during hypoxia—ischemia affected hypoxia—ischemia-induced seizures, we recorded the number of animals per group that displayed visible seizures. Seizures were recorded from the period immediately after hypoxia—ischemia until perfusion 24 hours later.
RESULTS
Temperature, blood pressure, blood glucose, and plasma corticosterone levels
Body temperature and blood glucose levels were not significantly different between the groups during the whole procedure (data not shown). During the normoxia periods, blood pressure was similar in all groups, whereas MET-treated animals had slightly elevated blood pressure during the hypoxia—ischemia period when compared with the other groups (Table 1).
Blood pressure, plasma corticosterone levels, the number of animals displaying visible seizures after hypoxia—ischemia as well as animals' activity (number of steps and rearings) before hypoxia—ischemia
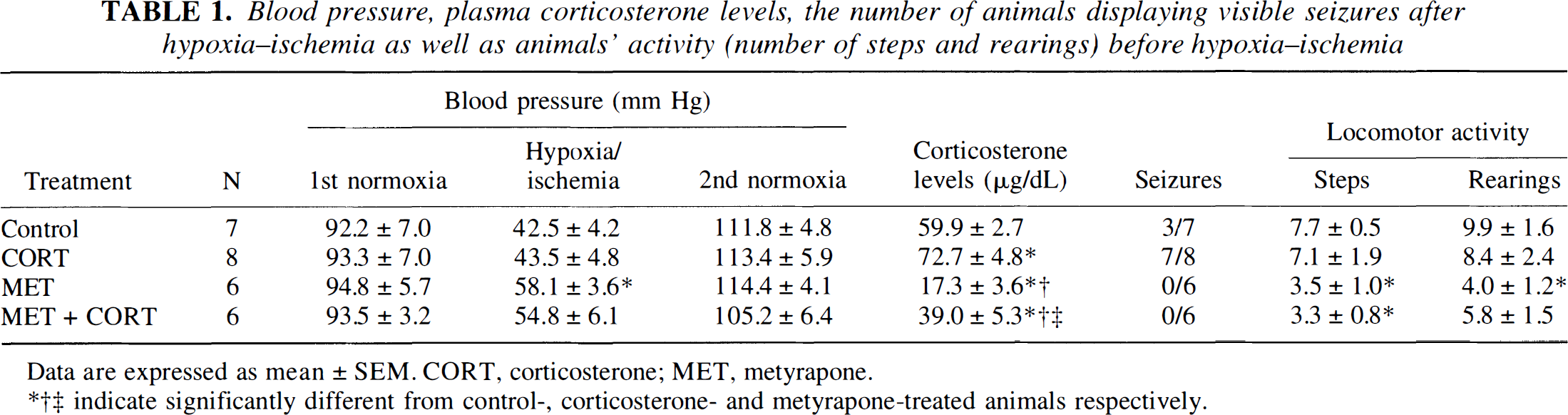
Data are expressed as mean ± SEM. CORT, corticosterone; MET, metyrapone.
indicate significantly different from control-, corticosterone- and metyrapone-treated animals respectively.
Control rats displayed very high plasma corticosterone levels shortly after 20 minutes of hypoxia—ischemia (59.9 ± 2.7 μg/dL, Table 1). Pretreatment with corticosterone significantly elevated the plasma steroid levels (72.7 ± 4.8 μg/dL), whereas pretreatment with metyrapone reduced plasma steroid levels (17.3 ± 3.6 μg/dL). Corticosterone administration in these animals (MET + CORT) resulted in a marked and significant increase in the plasma steroid levels over metyrapone-treated animals (39.0 ± 5.3 μg/dL).
Histology
Analysis of silver impregnation revealed striking effects of the treatments on brain damage, which appeared at least partially related to the plasma corticosteroid levels. In control animals, the hippocampus, the dorsolateral parts of the striatum, the thalamic areas, and cortex were damaged (Fig. 1). Total damage in control animals was 48.9 ± 9.1%. Brain damage was evident particularly in the animals displaying extremely high corticosteroid levels shortly after ischemia (CORT, total brain damage 65.8 ± 4.0%). Conversely, brain damage in animals pretreated with MET and MET + CORT (those with low plasma corticosteroid levels after hypoxia—ischemia) showed significantly less damage (P < 0.05) when compared with CORT-treated animals (total brain damage 26.3 ± 12.8% and 25.7 ± 13.4%, respectively). Corticosterone administration in MET animals did not influence brain damage when compared with MET animals.
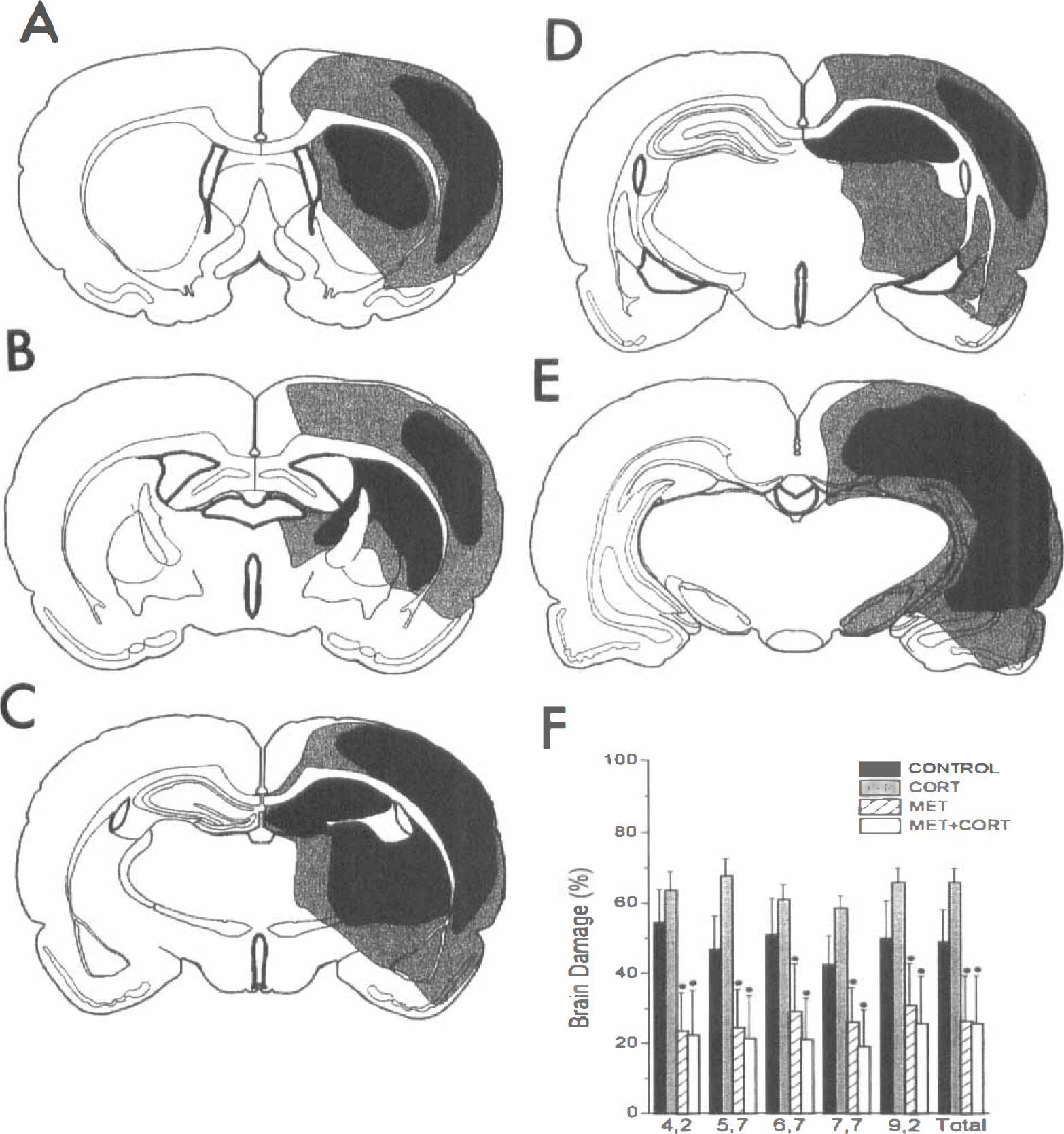
Camera lucida drawings of brain damage (
Seizures
Three of seven animals in the control group displayed visible seizures during the 24-hour period after hypoxia—ischemia (Table 1). These seizures were characterized by spinning of the animal around its body-axis, jerking movements, and/or clonic contractions of the paws. The seizures were intermittent and lasted less than 30 seconds. No between-group differences in the pattern of seizures was apparent. The number of animals displaying seizures showed a clear relationship with brain damage. In the CORT group, seven of eight animals displayed seizures whereas none of the metyrapone-treated animals displayed visible seizures. Corticosterone treatment of metyrapone-treated animals did not increase the number of visible seizures.
Behavior
Behavioral measurements before hypoxia—ischemia revealed that metyrapone affected activity of the animals. Metyrapone reduced both the number of steps and rearings in the staircase test, indicating a reduced general locomotor activity (Willig et al., 1991; Table 1).
DISCUSSION
Considerable evidence indicates that elevated plasma corticosterone levels exacerbate neuronal damage in response to excitotoxic events (Sapolsky and Pulsinelli, 1985; Stein and Sapolsky, 1988; Tombaugh and Sapolsky, 1992). It has been hypothesized that corticosteroids endanger neurons by disrupting glucose uptake and astrocyte functioning, resulting in elevated glutamate levels and an impaired capacity to maintain neuronal calcium homeostasis during and after hypoxia—ischemia or seizures (Virgin et al., 1991; Tombaugh and Sapolsky, 1992; Stein-Behrens et al., 1992; Elliott and Sapolsky, 1993; Elliot et al., 1993; Stein-Behrens et al., 1994). Because cerebral ischemia and seizures can induce considerable corticosterone secretion, corticosteroids could worsen the outcome of these insults. In the present study, we have shown that the steroid synthesis inhibitor metyrapone reduces plasma corticosterone levels during and shortly after hypoxia—ischemia. As predicted, this is accompanied by increased cell survival and a reduced incidence of seizures after hypoxia—ischemia when compared with animals exhibiting very high plasma corticosterone levels. These findings confirm our previous data (Kemper et al., 1994) and the observations by Smith-Swintoski et al. (1996) that metyrapone reduces brain damage after middle cerebral artery occlusion and kainic acid-induced seizures. Essentially, this may indicate that the production of corticosteroids after injury contributes to the severity of hypoxia—ischemia-induced damage. Accordingly, we demonstrated that down-regulation of the hypothalamo-pituitary-adrenal axis prevents the release of glucocorticoids during hypoxia—ischemia and increased neuronal survival (Krugers et al., 1995). Moreover, our data show that metyrapone reduces the incidence of seizures after hypoxia—ischemia. If—as our data at least partly demonstrate—corticosterone exacerbates brain damage after ischemia and seizures, this might have clinical implications. Accordingly, elevated urinary and plasma cortisol levels have proven to be predictive indicators of prognosis after stroke and status epilepticus (Olsson et al., 1992; Gallagher et al., 1984).
The glucocorticoid receptor, rather than the mineralocorticoid receptor, appears to be involved in the corticosteroid endangerment of neurons (Packan and Sapolsky, 1990). Our results in the present are in agreement with such a hypothesis because cells throughout the brain were protected by metyrapone, which coincides with the distribution of the glucocorticoid receptor (whereas the mineralocorticoid receptor is localized mainly in the hippocampus, De Kloet, 1991).
We found that corticosterone treatment did increase plasma corticosterone levels over that of metyrapone-treated animals, but this did not result in a subsequent increase in damage or seizures. Therefore, one might argue against a neuroprotective effect of metyrapone by suppressing the insult-induced rise in corticosterone. Metyrapone reduces corticosteroid production by inhibition of the 11-β-hydroxylation reaction, resulting in an increased production of 11-deoxycorticosterone (DOC, Goodman Gilman et al., 1985). Metabolites of DOC (such as 3α 21-dihydroxy-5α pregnan-20-one, 5α-THDOC) are known for their anesthetic properties, presumably via gamma-aminobutyric acidA-receptors (Lambert et al., 1995). Although we did not measure levels of these neuroactive steroids, we do have some indirect evidence for an anesthesia-like effect of metyrapone. Metyrapone-treated animals were tested for their locomotor activity in a staircase test: metyrapone reduced the activity of the rats in a staircase test. Because gamma-aminobutyric acid-ergic actions of neuroactive steroid actions may prevent excitotoxic brain damage, this could be an additional way to explain the neuroprotective effect of metyrapone (Lambert et al., 1995).
We conclude that the steroid synthesis inhibitor metyrapone reduces neuronal damage and seizures after hypoxia—ischemia but that the nature of its neuroprotective action might be partially independent from suppressing the increase in plasma corticosteroid levels.
