Abstract
To investigate the reliability of unilateral jugular venous monitoring and to determine the appropriate side, we performed bilateral jugular venous monitoring in 22 head-injured patients. Fiberoptic catheters were placed in both jugular bulbs. Arterial and bilateral jugular venous blood samples were obtained simultaneously for in vitro determination of jugular venous oxygen saturation (SJO2), arterial minus jugular venous lactate content difference (AJDL), and modified lactate-oxygen index (mLOI). Ischemia was assumed if one of the following pathologic values occurred at least unilaterally: SJO2 <54%, AJDL <−0.37 mmol/L, mLOI >0.08. The sensitivity of calculated unilateral monitoring in detecting ischemia was evaluated by comparing the incidence detected unilaterally with that disclosed bilaterally. The mean and maximum bilateral SJO2 differences varied between 1.4% and 21.0%, and 8.1% and 44.3%, respectively. The bias and limits of agreement (mean differences ± 2 SD) between paired samples were 0.4% ± 12.8%. There was no significant variation in bilateral SJO2 differences with time. Decreasing cerebral perfusion pressure (r = −0.559, P < 0.001) and arterial P
Monitoring of global cerebral hemodynamics and metabolism based on the Fick principle has provided significant information concerning the pathophysiology of severe head injuries, and has become an important tool for clinical management. According to this method, arterial and cerebral venous blood samples are analyzed for concentration differences of tracers or various metabolites such as oxygen, glucose, and lactate (Kety and Schmidt, 1945; Robertson et al., 1989, 1987). Moreover, the technique of continuous jugular venous oxygen saturation (SJO2) measurement, although artifact prone, yields important real-time data about the state of brain oxygenation in severely head-injured patients at risk for cerebral ischemia (Andrews et al., 1991; Sheinberg et al., 1992). Measurements of oxygen and glucose concentrations performed on samples drawn simultaneously from both internal jugular bulbs have demonstrated that only small differences exist in healthy adults (Gibbs et al., 1942) Consequently, cerebral venous blood samples for oxygen saturation monitoring, as well as for all other derived calculations, are usually obtained from unilateral jugular bulb catheterization. However, as recently demonstrated, marked differences can occur in patients with head injuries, especially in cases of unilateral lesions. Unfortunately, the more appropriate side for monitoring could not be identified (Stocchetti et al., 1994). In head-injured patients cerebral blood flow is often heterogeneous in different areas of the brain (Raichle et al., 1976; Bouma et al., 1991) In this setting, the major problem is that therapeutic procedures based on information obtained from unilateral jugular bulb catheterization may be dangerous to the contralateral hemisphere (Stocchetti et al., 1994). The aim of our study was to assess the incidence and the extent of such potentially dangerous discrepancies between both jugular bulbs in comatose head-injured patients. To investigate this we considered two aspects. First, we investigated the absolute discrepancies between jugular venous oxygen saturation (SJO2) and arterial minus jugular venous lactate content difference (AJDL) between both jugular bulbs. Second, we tested the reliability of calculated unilateral monitoring to detect pathologic values in SJO2, AJDL, and the modified lactate-oxygen index (mLOI) indicating cerebral ischemia.
MATERIALS AND METHODS
After approval by our local ethics committee, 22 patients with Glasgow Coma Scale scores <8 on admission to the emergency room were enrolled in the study from June 1995 to October 1996. Patient demographic data and clinical characteristics are presented in Table 1.
Patient demographic data and clinical characteristics
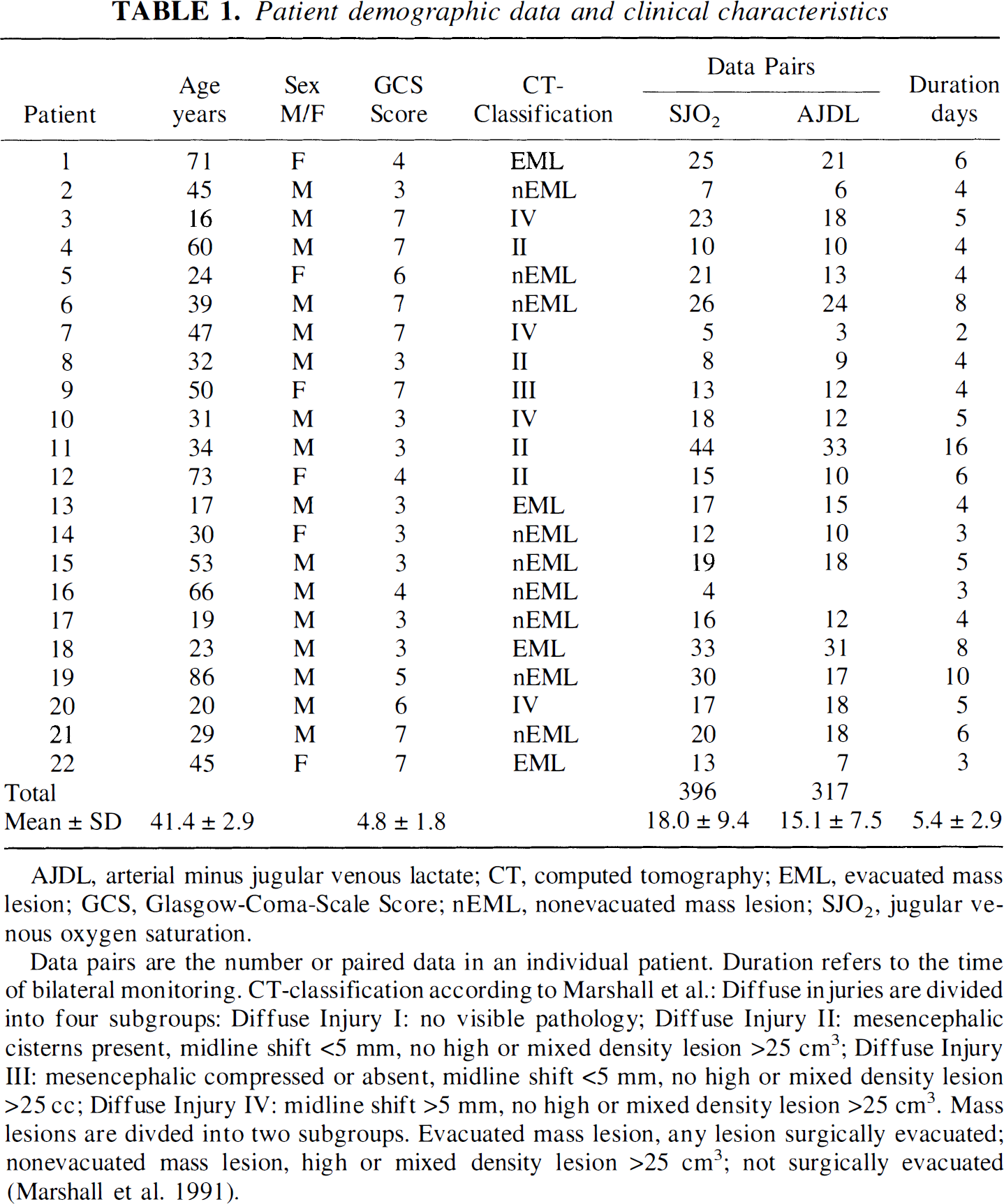
AJDL, arterial minus jugular venous lactate; CT, computed tomography; EML, evacuated mass lesion; GCS, Glasgow-Coma-Scale Score; nEML, nonevacuated mass lesion; SJO2, jugular venous oxygen saturation.
Data pairs are the number or paired data in an individual patient. Duration refers to the time of bilateral monitoring. CT-classification according to Marshall et al.: Diffuse injuries are divided into four subgroups: Diffuse Injury I: no visible pathology; Diffuse Injury II: mesencephalic cisterns present, midline shift <5 mm, no high or mixed density lesion >25 cm3; Diffuse Injury III: mesencephalic compressed or absent, midline shift <5 mm, no high or mixed density lesion >25 cc; Diffuse Injury IV: midline shift >5 mm, no high or mixed density lesion >25 cm3. Mass lesions are divded into two subgroups. Evacuated mass lesion, any lesion surgically evacuated; nonevacuated mass lesion, high or mixed density lesion >25 cm3; not surgically evacuated (Marshall et al. 1991).
Study protocol
All patients were managed according to our standard intensive care protocol, which is aimed at preventing cerebral ischemia (Metz et al., 1996). They were deeply sedated by continuous infusion of opioids and midazolam, and did not respond to pain. Hyperthermia (core temperature >37.5°C) was strictly avoided. Ventilator settings were adjusted to achieve sufficient arterial oxygenation (Pa
Monitoring
Invasive hemodynamic monitoring was performed according to the patient's critical illness. Intracranial pressure was continuously monitored by an intraparenchymal fiberoptic catheter with its tip in the white matter of the brain (OPX-100, Inner Space Medical Inc., Irvine, CA, U.S.A.). For bilateral jugular venous monitoring, patients were catheterized with the upper part of the body slightly elevated and without rotating the head. Introducer sheaths (4.0F, Arrow International, Reading, PA, U.S.A.) were placed upstream in both internal jugular veins. Through each introducer sheath a fiberoptic catheter (U 440, Oximetrix, Abbott Critical Care System, Abbott Lab., North Chicago, IL, U.S.A.) was inserted and advanced into the corresponding jugular bulb. To prevent jugular venous thrombosis both systems were flushed with 5 mL/h of 10 U/mL heparinized saline. Catheter positions were verified by roentgenography in anteroposterior and lateral projections or by computed tomographic (CT) scan. The lateral film was obtained from a position slightly less than perpendicular to the neck to allow for visualization of both catheters. We used fiberoptic catheters bilaterally to detect significant changes in oxygen saturation between both sides as early, and as completely, as possible. Because online SJO2 monitoring is artifact prone, whenever the SJO2 monitors appeared to show significant changes we drew blood simultaneously from both jugular bulbs and the arterial line to measure in vitro oxygen saturation and arterial minus jugular venous lactate content differences. In addition, blood samples were drawn in conjunction with in vitro calibration of fiberoptic catheters in 6-hour intervals. To avoid contamination of the samples with extracerebral blood, jugular venous blood was withdrawn slowly (approximately 2 mL/min).
Data analysis
All blood samples were analyzed in vitro for arterial (SaO2) and jugular venous (SJO2) oxygen saturation and AJDL. The blood gases were measured on a Radiometer 520 blood gas analyzer. (Fa. Radiometer, Copenhagen, Denmark). To improve the accuracy of lactate determinations, duplicate whole blood lactate concentrations in arterial and venous blood were determined in each case by an enzymatic method and average values were calculated (Beckman CX 7, Beckman Instruments, Inc., Fullerton, CA, U.S.A.). The data of the lactate measurements were excluded from analysis if the results from the same sample differed by more than 5%.
The lactate-oxygen index (LOI = negative arterial minus jugular venous lactate content difference divided through arterial minus jugular venous content difference = AJDL × AJDO2−1 represents the ratio of the amount of glucose metabolized anaerobically to the amount metabolized aerobically. When the LOI is 0.08 or greater, ischemia or infarction is present (Robertson et al., 1989). However, in the case of acute anemia the LOI may lead to false-positive results because the AJDO2 is abnormally low. To avoid this we used the modified lactate-oxygen index (mLOI, Cruz et al., 1994). This physiologic variable relies on cerebral extraction of oxygen instead of arteriojugular oxygen content difference. Thus, the undesired effects of acute anemia on assessment of cerebral ischemia are eliminated. The mLOI was calculated using the following formula: mLOI = –AJDL (mmol/L)/(SaO2 – SJO2) (%) × 10 (Cruz et al., 1994). Normally SJO2 varies between 54% and 75% (Dearden, 1991), AJDL amounts to 0.17 ± 0.1 mmol/L (Gibbs et al., 1942), and mLOI values are <0.08 (Cruz et al., 1994). Ischemia was assumed if at least one of the following criteria was fulfilled (Sheinberg et al., 1992; Cruz et al., 1994): AJDL <–0.37 mmol/L, or mLOI >0.08. In the case of SJO2 <54% without pathologic AJDL or mLOI values, compensated cerebral hypoperfusion was assumed. To determine the appropriate side for jugular venous monitoring, we investigated the sensitivity of unilateral measurements in detecting cerebral ischemia or global cerebral hypoperfusion. In accordance with various recommendations in the literature, the incidence of ischemia or hypoperfusion appearing on the actual suggested side was compared with the maximum possible incidence detectable using bilateral monitoring. The following recommendations were studied: Robertson et al. (1989) cannulated the side where there was the most severe injury, or the right side in case of diffuse lesions. Dearden (1991) suggested the side of predominant venous outflow, which was identified by the larger jugular foramen on a computerized tomography of the head. Furthermore, the reliability of exclusively monitoring the right side was tested (Cruz et al., 1994). Finally, the so-called CT approach was investigated, consisting of a synthesis of Robertson's and Dearden's recommendations. Based on CT scan, the side of predominant lesion or, in case of diffuse lesions, the side of the largest jugular foramen was selected for jugular venous monitoring.
Statistical analysis
For statistical analysis the Kolmogoroff-Smirnoff modification of the Lilliefor's test was used to test for normal distribution. Bilateral SJO2 and AJDL data were compared using the method described by Bland and Altman (1986). To determine if two different techniques are in agreement and therefore interchangeable, a plot of the difference between the paired results from the two methods of measurement against the average of the paired results was performed. The agreement between the two measurements can be summarized by calculating the bias, estimated by the mean difference and the standard deviation of the differences, which indicates the measure of precision. The limits of agreement (95% confidence interval) are defined by the bias ± 2 SD of the differences (Bland and Altman, 1986). To avoid the statistical error caused by repeated measurements, we used the patients' mean data of each jugular bulb for analysis. To investigate the variations of bilateral SJO2 differences with time, the patients' individual mean differences, maximum differences, and crossover events were divided into three subsequent time intervals up to 24, 48, and 72 hours after admission to the intensive care unit. For each individual patient the corresponding areas under the curve were calculated (Matthews et al., 1990). The resulting mean values concerning mean SJO2 differences, maximum SJO2 differences, and crossover events for each time interval were analyzed by the paired Student's t test for statistical significance. Next, bilateral SJO2 differences were plotted against the corresponding CPP, ICP, and Pa
RESULTS
In 22 patients the catheters were used for 5.4 ± 2.9 days (range, 2 to 16 days). During the study period 396 paired samples of right and left SJO2 were obtained. In 317 cases the corresponding AJDL and mLOI values were determined simultaneously (Table 2). Precision and accuracy of the in vitro blood gas measurements were high. The results were checked daily for repeatability and were found to be consistently below 1% difference in the same sample. With respect to lactate measurements, the cumulative variation coefficient was 3.1% and the SD was 0.10 mmol/L.
Mean values for oxygen saturation (SJO2) in both jugular bulbs and corresponding bilateral mean and maximum differences in SJO2, and AJDL in individual patients
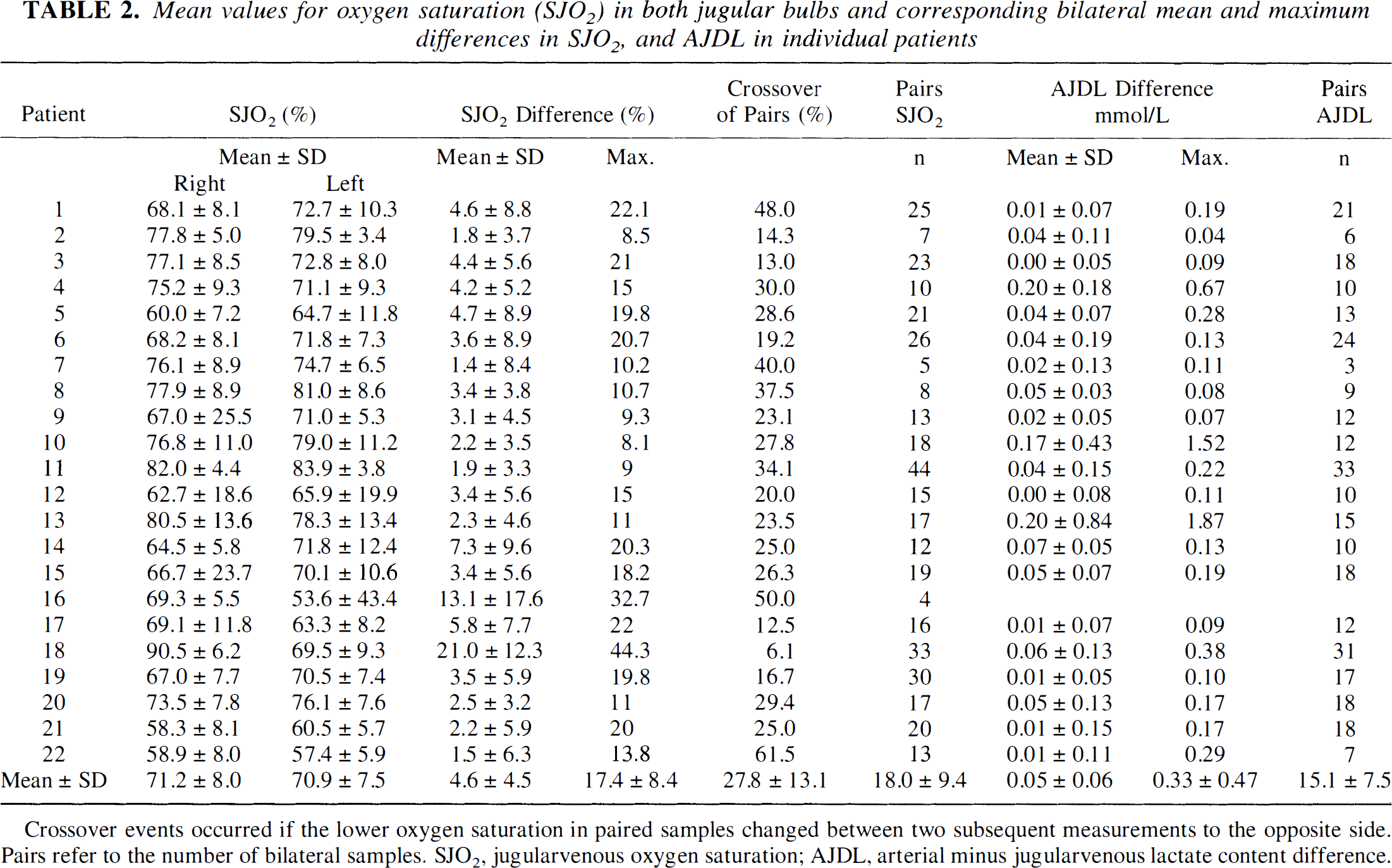
Crossover events occurred if the lower oxygen saturation in paired samples changed between two subsequent measurements to the opposite side. Pairs refer to the number of bilateral samples. SJO2, jugularvenous oxygen saturation; AJDL, arterial minus jugularvenous lactate content difference.
The overall mean values of the right and left SJO2 and AJDL data obtained from all patients were not significantly different (Table 2). In individual patients the mean bilateral SJO2 differences varied between 1.4% and 21.0%, and the maximum SJO2 differences were between 8.1% and 44.3%, respectively. Four patients had maximum bilateral differences between 5% and 10%; 11 patients between 10% and 20%, and 7 patients greater than 20%. Eighteen of 22 patients had an SJO2 difference above 10% at least once, and discrepancies greater than 20% were found in one third of the patients. A crossover in SJO2 is defined as the change of the lower oxygen saturation to the opposite side between two subsequent measurements. In individual patients the frequency of crossover in paired samples varied between 6.1% and 61.5%. On the average, in approximately every fourth data pair the lower SJO2 value changes in subsequent measurements (Table 2).
Figure 1 demonstrates the distribution of individual mean differences between the corresponding SJO2 values of the right and left side plotted against their mean values (Bland and Altman, 1986). The bias and precision (mean differences ± SD) between the corresponding mean right and left SJO2 was −0.4% ± 6.4%. The resulting wide limits of agreement were −13.2% and 12.4%. Next, exclusively paired samples with at least unilateral desaturation (i.e., SJO2 <54%) were selected for analysis. Again, Bland and Altman plots were used for analysis of agreement (Fig. 1). In comparison to mean SJO2 data the limits of agreement during jugular venous desaturation had improved to ± 7.4%.
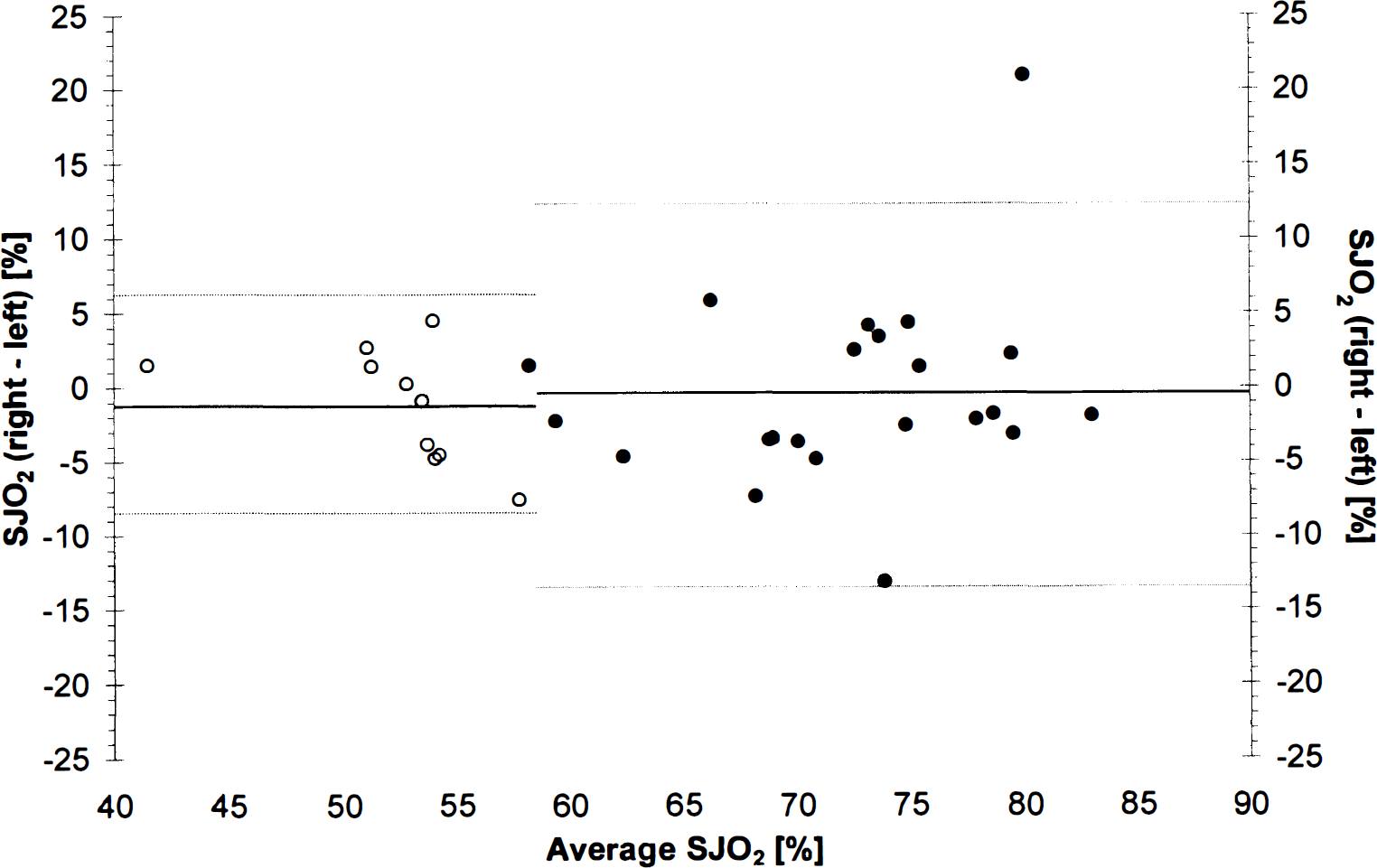
Distribution of individual differences between mean right and left jugular bulb oxygen saturation (SJO2) against their average values. Full dots represent overall mean values in individual patients. Open circles consist of mean right and left SJO2 and the resulting average values exclusively from paired samples with at least unilateral desaturation (SJO2 <54%). The corresponding solid lines represent the bias or the overall mean difference, dashed lines correspond to mean ± 2 SD of the differences (i.e., 95% confidence interval), respectively.
In 21 patients, with a total number of 317 paired samples processed, we found a mean AJDL of −0.16 ± 0.19 mmol/L. The mean differences (accomplished by subtracting the smaller number from the larger one) between both jugular bulbs was 0.05 mmol/L ± 0.06 mmol/L; the maximum differences varied greatly between 0.04 mmol/L and 1.52 mmol/L (Table 2). Using the Bland and Altman plot for AJDL data, we found a bias of −0.01 mmol/L and corresponding limits of agreement of −0.19 mmol/L and 0.17 mmol/L, respectively (Fig. 2). In contrast to SJO2, in the case of pathologic AJDL <–0.37 mmol/L, the limits of agreement between both sides decreased markedly as a function of increasing cerebral lactate production (–1.38 mmol/L and 1.42 mmol/L). This range by far exceeds the defined ischemic threshold of −0.37 mmol/L.
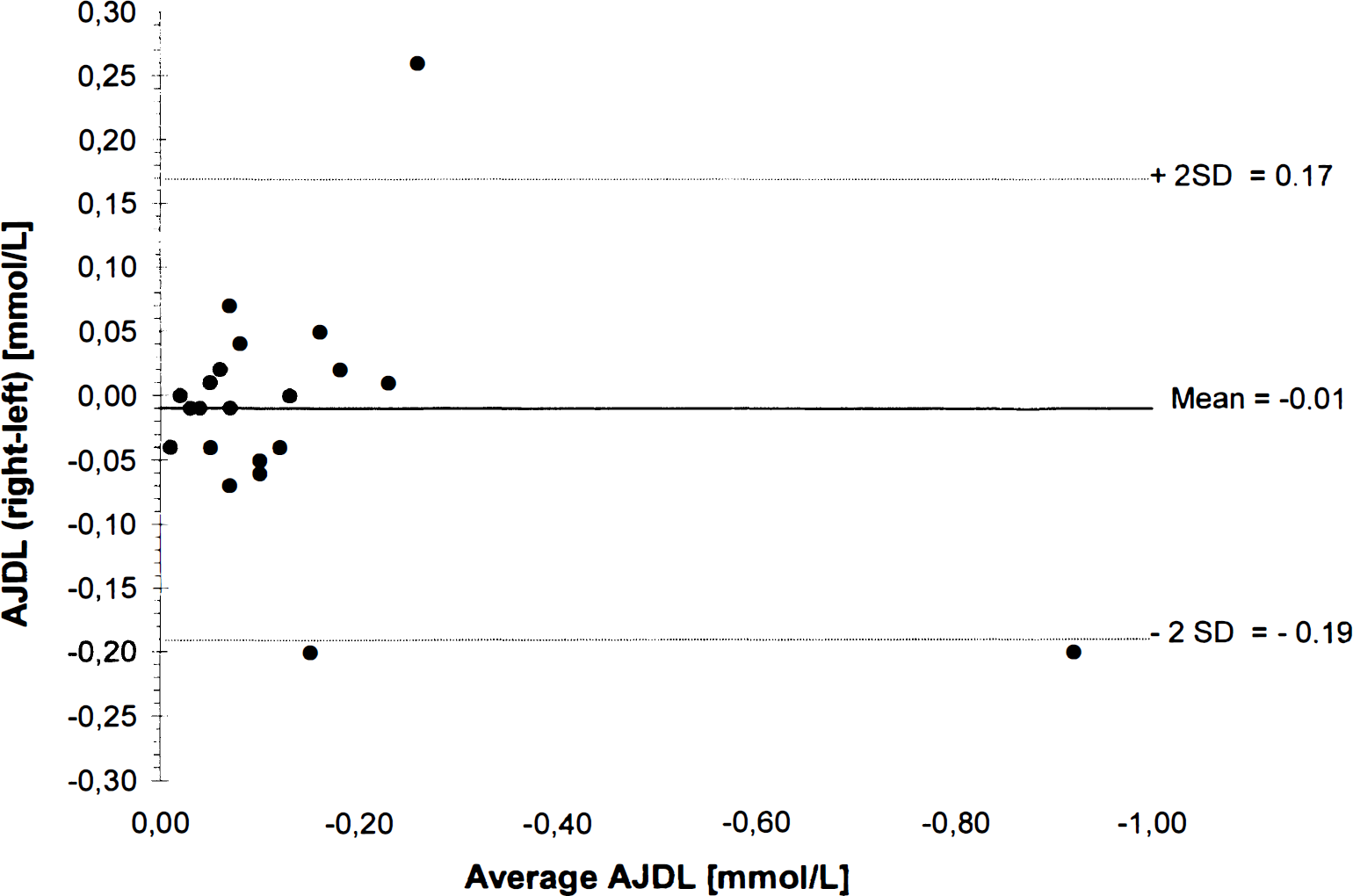
Distribution of individual differences between mean arterial minus jugular venous lactate content difference (AJDL) in the right and left jugular bulb against their average values. The solid line represents the bias or the overall mean difference, dashed lines correspond to mean ± 2 SD of the differences (i.e., 95% confidence interval).
Depending on the type of pathology examined, between 65% and 80% of pathologic values occurred exclusively unilaterally (Table 3). Episodes of jugular venous desaturation were present in 10 patients. The maximum differences in SJO2 between both jugular bulbs varied between 1.5% and 15.8%. With respect to AJDL and mLOI data, we observed large bilateral discrepancies in the majority of patients.
Incidence of exclusively unilateral pathological values in SJO2, AJDL, and mLOI in conjunction with corresponding maximum bilateral differences

SJO2, jugular venous oxygen saturation; AJDL, arterial minus jugular venous lactate content difference, mLOI, modified lactate oxygen index. Maximum bilateral differences are calculated by subtracting the smaller value from the larger one.
Distinguishing between unilateral and diffuse brain injuries, we found significantly lower bilateral mean and maximum SJO2 differences in patients with diffuse lesions (2.8 ± 1.0% versus 5.9 ± 5.4% for mean differences and 12.5 ± 4.0% versus 20.2 ± 9.0% for maximum differences). Regarding AJDL, maximum differences were also lower in diffuse injuries, but did not achieve statistical significance.
Neither the corresponding mean, nor the maximum bilateral SJO2 differences, nor the incidence of crossover events changed significantly during the first 72 hours after hospital admission. However, the frequency of jugular venous desaturations (SJO2 <54%) declined continuously from the first 24 hours up to 48 and 72 hours after admission (20.5% versus 11.2% versus 8.5% of data pairs, P < 0.05). Correlation analysis revealed significant associations between bilateral SJO2 differences and corresponding CPP, ICP, and arterial P
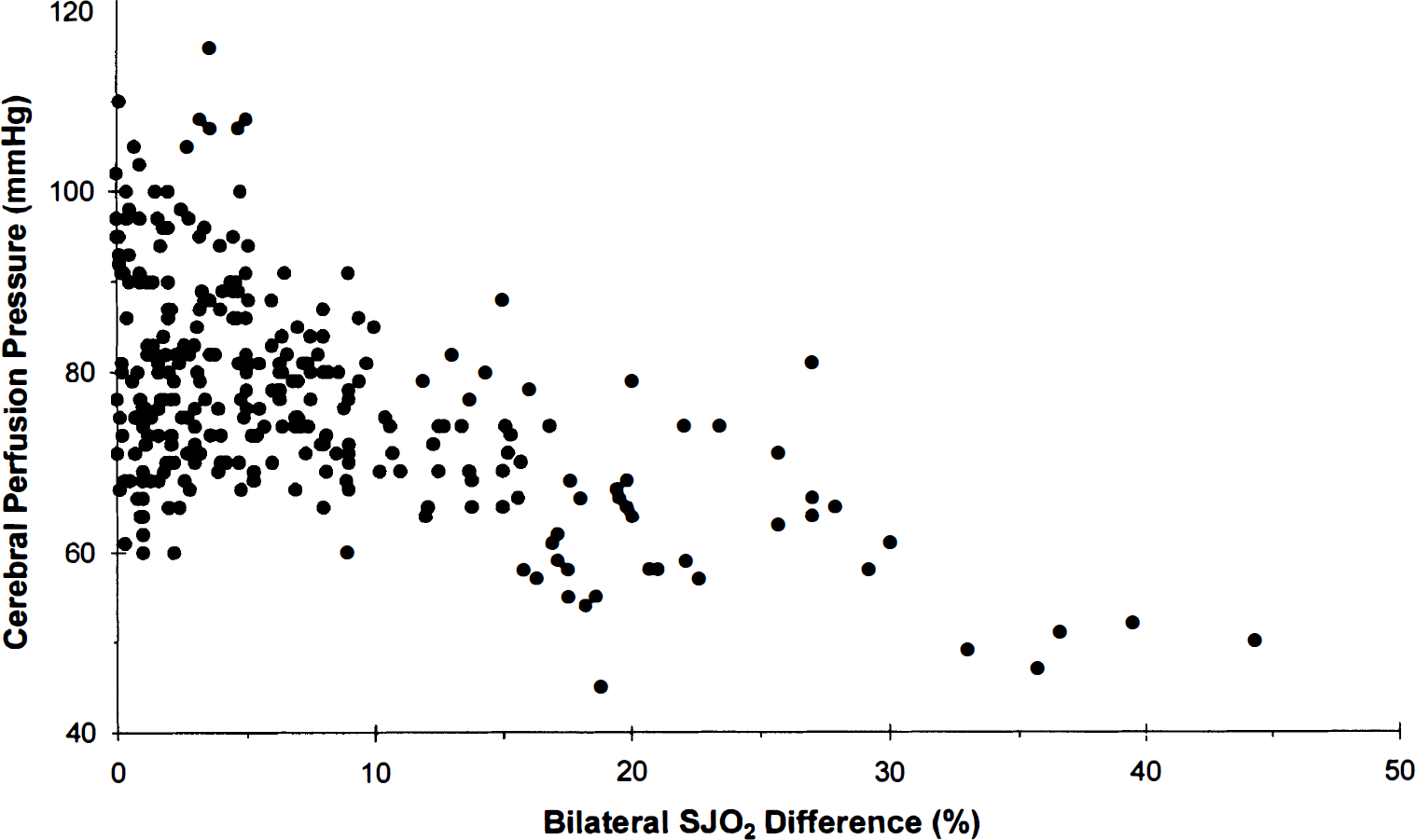
Correlations between cerebral perfusion pressure (CPP) and bilateral differences in jugular venous oxygen saturation (SJO2). Pearson's correlation coefficient r = −0.559, P < 0.001, n = 305 data pairs.
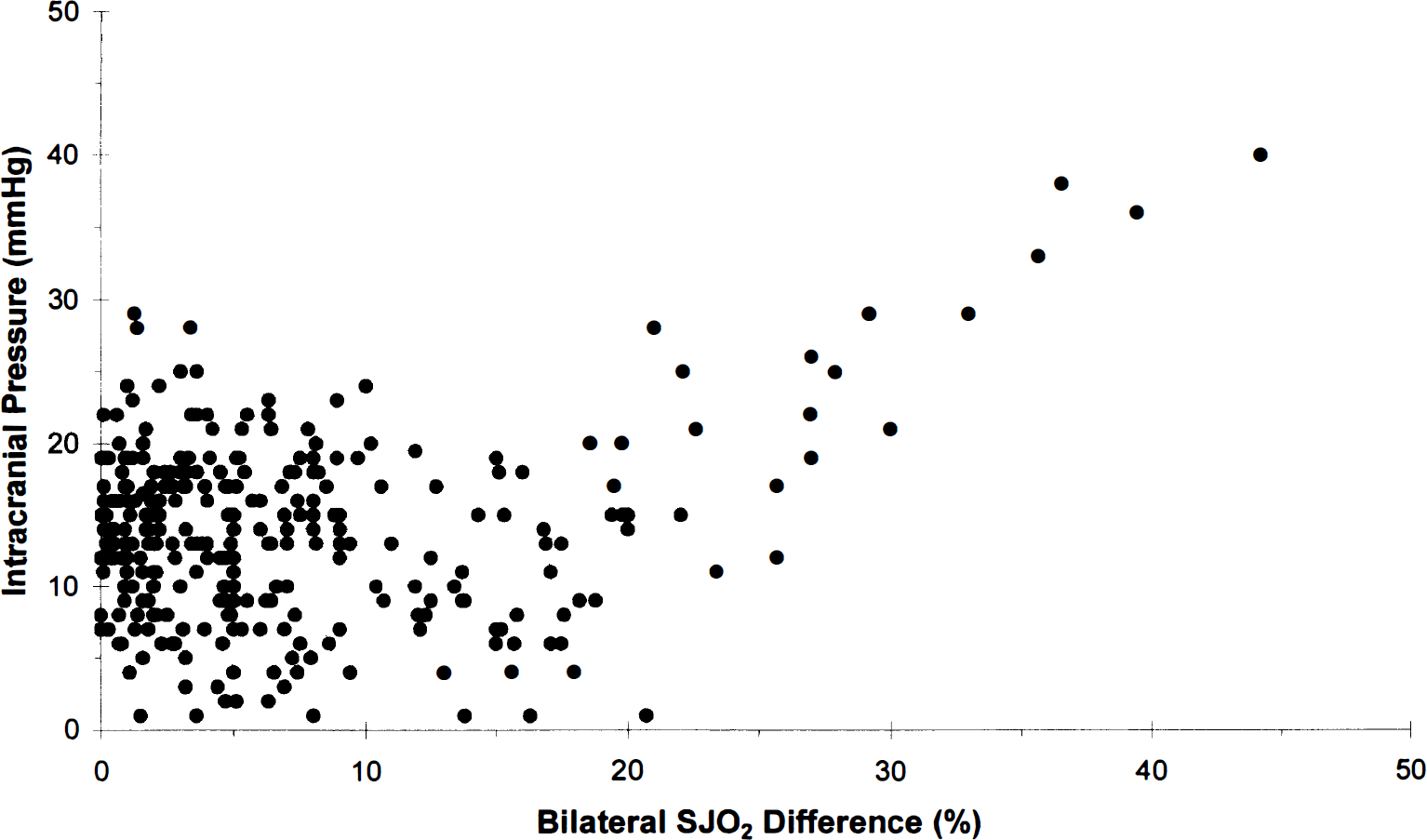
Correlations between intracranial pressure (ICP) and bilateral differences in jugular venous oxygen saturation (SJO2). Pearson's correlation coefficient r = 0.249, P < 0.001, n = 305 data pairs.
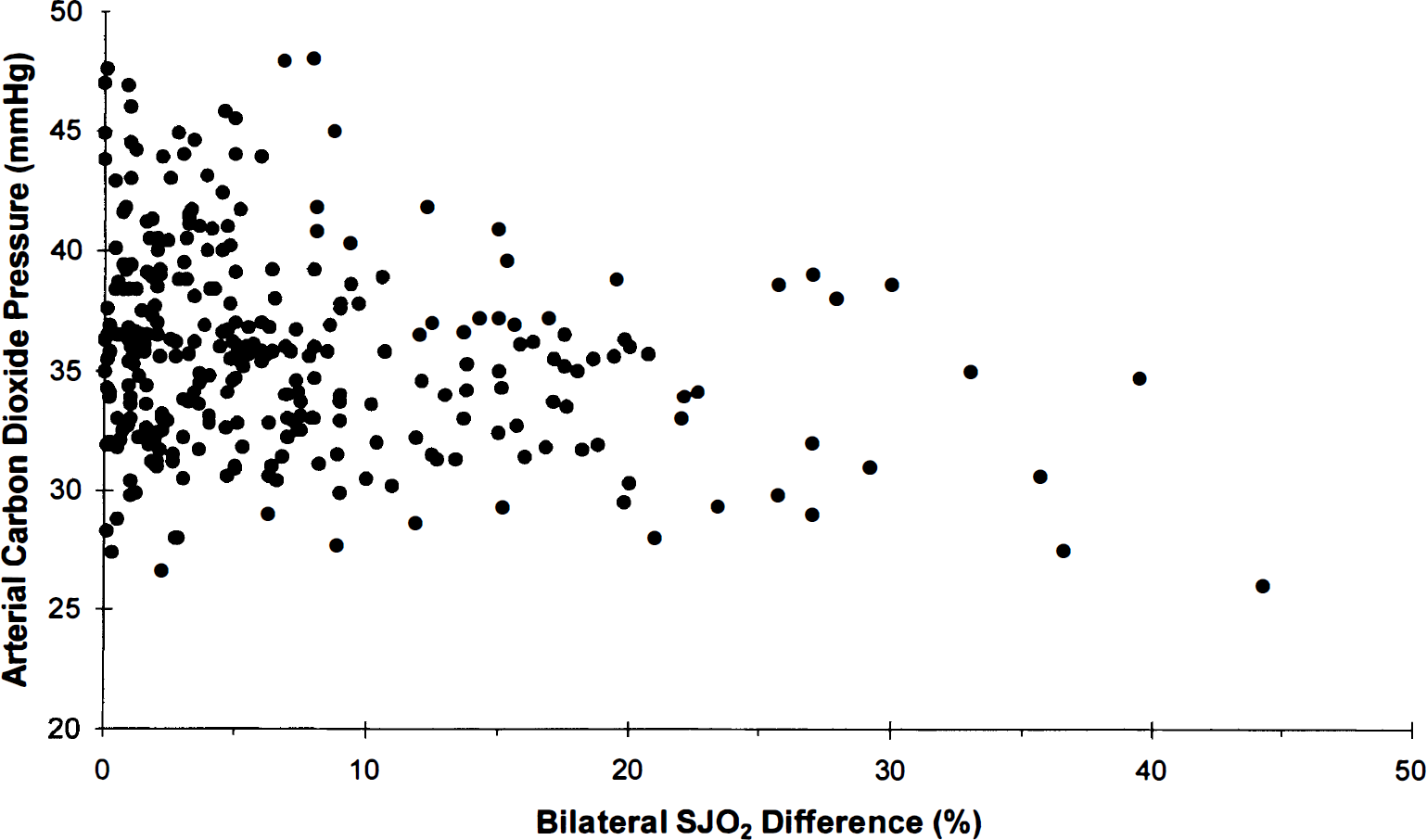
Correlations between arterial carbon dioxide pressure (Pa
Finally we investigated the reliability of unilateral measurements in identifying pathologic values indicative of cerebral hypoperfusion and ischemia (Table 4). Using bilateral jugular venous monitoring we found a total of 134 pathologic values, representing a sensitivity of 100%. Using Robertson's recommendation, only 105 pathologic values suggestive of ischemia could be identified, corresponding to a sensitivity of 79%. Dearden's method disclosed 110 pathologic values with a sensitivity of 82%. In our patient population a maximum of 117 events or 87% could be evaluated if the side of monitoring was chosen according to the so-called CT approach, which consists of a synthesis of Robertson's and Dearden's recommendations. Based on a routine CT scan the side of predominant lesion or, in case of diffuse lesions, the side of the largest jugular foramen was selected for jugular venous monitoring. Monitoring exclusively in the right jugular bulb resulted in the lowest sensitivity of 75%. With respect to the type of event, the highest sensitivity was obtained by AJDL measurements. Again the CT approach proved best and allowed 16 of 17 diagnoses (Table 4). In contrast, by monitoring on the right side only 11 pathologic AJDL values were detected (i.e., 65%). Overall, mLOI data had the highest variability. Once again, the best sensitivity of 86% was achieved by using the CT approach.
Sensitivity of various recommendations for unilateral jugularvenous monitoring in detecting compensated cerebral hypoperfusion and/or cerebral ischemia
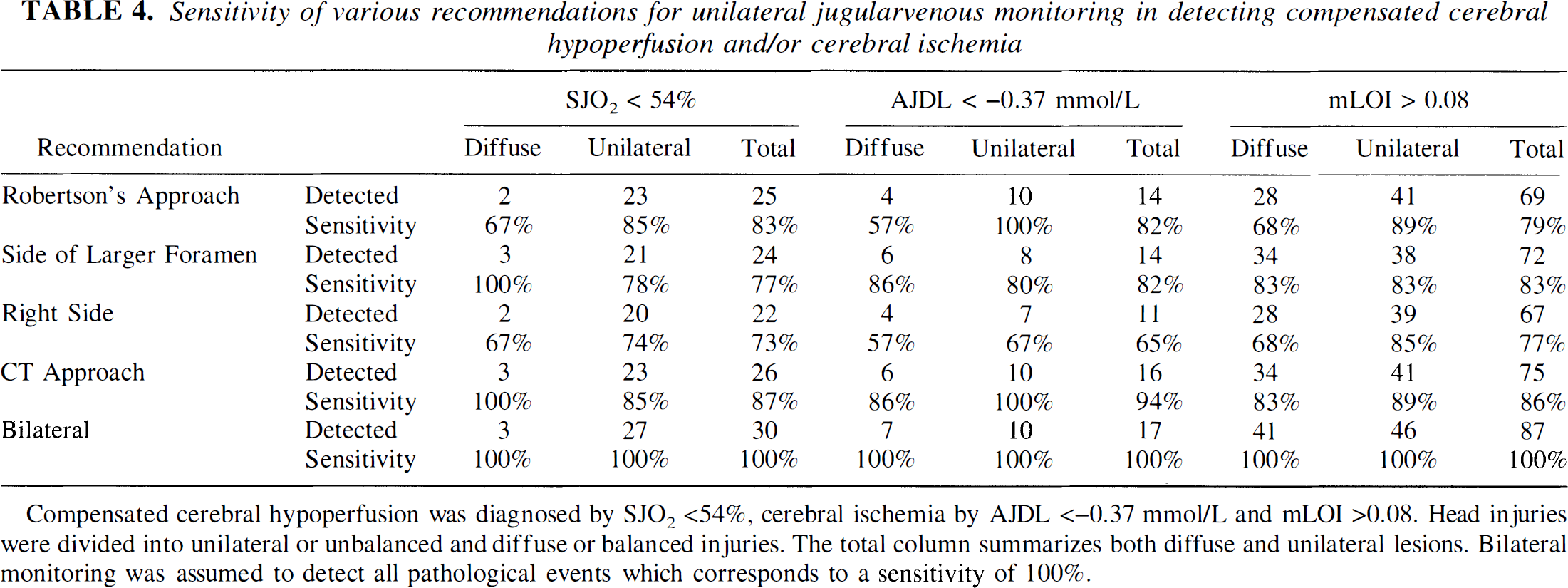
Compensated cerebral hypoperfusion was diagnosed by SJO2 <54%, cerebral ischemia by AJDL <–0.37 mmol/L and mLOI >0.08. Head injuries were divided into unilateral or unbalanced and diffuse or balanced injuries. The total column summarizes both diffuse and unilateral lesions. Bilateral monitoring was assumed to detect all pathological events which corresponds to a sensitivity of 100%.
Serious complications associated with bilateral jugular venous monitoring did not occur. During and after bilateral catheter insertion, clinically significant increases in intracranial pressure because of obstruction of venous outflow of the brain was not observed. Carotid puncture was noted once (2.3%) with no apparent adverse effect. Minor venous hematoma occurred unilaterally in two patients (9.1%). Clinically apparent venous thrombosis was not observed in our patients. Routine catheter cultures after removal showed totally negative results.
DISCUSSION
Studies comparing bilateral jugular bulb oxygen saturation in conjunction with indices of cerebral oxygen deficit (AJDL, mLOI) are still lacking. In this study, conducted on severely head-injured patients, we investigated the accuracy of unilateral jugular venous monitoring on parameters indicative of cerebral hypoperfusion and ischemia (SJO2, AJDL, mLOI) as compared with bilateral measurements, which yield the maximum possible information. Regarding jugular venous oxygen saturation in head-injured patients, a difference of 5% in bilateral SJO2 seemed acceptable; a difference of 10% seemed highly suspicious, and differences above 15% were unacceptable (Stocchetti et al., 1994). We found a mean difference of 4.8% in SJO2 values between the right and the left jugular bulb. This value corresponds to those reported previously in the literature (Stocchetti et al., 1994). Although mean bilateral differences would suggest minor discrepancies, Fig. 1 shows that the limits of agreement between both sides were poor. For example, if right SJO2 was 65%, which was well within the normal range, left SJO2 values might have been between 52% and 78%. The lowest SJO2 would suggest hypoperfusion, whereas the highest value indicates relative hyperemia. This lack of agreement could significantly influence patient management. Moreover, in our patients the side showing greater oxygen saturation was highly inconsistent and changed frequently with time (Table 2). In the case of at least unilateral jugular venous desaturation (SJO2 <54%) the corresponding limits of agreement improved to ±7.4%. However, an ipsilateral SJO2 of 50%, indicating compensated hypoperfusion, may be associated with a contralateral SJO2 between 43% and 57%. The lower value suggests marked hypoperfusion, the higher one falls within the normal range. Considering the wide limits of agreement, the high frequency of suspicious differences in individual data pairs, and finally the high frequency of crossover values between both sides, we conclude that arbitrarily selecting a particular side for SJO2 monitoring results in an increased probability of invalid results in a large number of patients. This finding is in agreement with Stocchetti and coworkers, who could not identify the more appropriate side for SJO2 monitoring in patients with bilateral or predominantly monolateral cortical or deep lesions (Stocchetti et al., 1994). As expected from studies dealing with early evaluation of cerebral blood flow and oxygen extraction in head-injured patients, the frequency of jugular venous desaturations decreased significantly during the first 3 days of monitoring (Bouma et al., 1991, 1992). However, the extent of mean and maximum bilateral SJO2 differences did not change significantly during this period. On average, absolute SJO2 values increased bilaterally in nearly the same way. Moreover, the frequency of changes in the lower value in SJO2 to the opposite side in two subsequent measurements (crossover) did not change significantly during the first 3 days after trauma. Therefore, the causal pathophysiology for these individual side-to-side differences and their dynamic changes seem to remain consistent during this time period.
Global cerebral hypoperfusion can be diagnosed by a jugular venous desaturation below 54%. However, SJO2 alone is insufficient to detect regional cerebral hypoperfusion or ischemia. In this case there is a heterogeneity of cerebral blood flow (CBF). Hypoperfused or ischemic areas with high oxygen extraction (i.e., low SJO2) overlap with areas of normal or even elevated CBF and corresponding normal or low oxygen extraction (i.e., normal or high SJO2). The resulting SJO2 in the jugular bulb represents mixing of blood drained from these heterogeneous areas and therefore could be in the normal range (Metz et al., 1996). If CBF fails to meet metabolic demand, anaerobic metabolism occurs, causing accumulation of lactate in ischemic brain tissue (Frerics et al., 1990; Inao et al., 1988; Unterberg et al., 1988). It has been further shown that lactate rapidly moves across the blood—brain barrier and appears in cerebral venous blood (Frerics et al., 1990). The production of lactate in brain tissue and blood increases in proportion to the severity of injury, and the point of most negative AJDL coincides with the point of maximum brain tissue lactate content (Inao et al., 1988). In head-injured patients, global as well as regional cerebral ischemia can be diagnosed by the AJDL, or by calculating the mLOI, which can be easily obtained in the intensive care unit (Cruz et al., 1994).
However, the blood—brain barrier transport mechanism for lactate is bidirectional and dependent on the relative concentrations in brain tissue and blood (Nemoto et al., 1974). The higher the difference in lactate content, the higher the clearance from the brain to jugular venous blood. Therefore, jugular venous lactate concentration may be influenced by elevated systemic lactate production. As long as arterial lactate concentration is normal, increases in cerebral lactate production should be reflected by a net efflux of lactate. In the presence of a systemic lactic acidosis, a minor increase in cerebral lactate production could be obscured. However, as demonstrated in comatose, head-injured patients an increased cerebral lactate production was consistently present in patients who developed a cerebral infarction, even when arterial lactate concentration was increased (Robertson et al., 1989).
Of particular interest are the findings of Inao et al. (1988), who assessed the dynamics of brain tissue lactate compared with cerebrospinal fluid lactate and the arteriovenous difference in lactate concentrations after fluid-percussion injury in cats. The observation that lactate levels in arterial blood and cerebrospinal fluid are maximum immediately after impact, while brain tissue lactate content gradually increases suggests that the major lactate contributor to serum and cerebrospinal fluid compartments may not be the brain. The authors found a much higher correlation between brain tissue lactate and the arteriovenous lactate difference, especially in the presence of systemic metabolic changes. The production of lactate in brain tissue and blood increases in proportion to the severity of injury, and the point of most negative arteriovenous lactate difference coincides with the point of maximum brain tissue lactate content. Studies undertaken more than 50 years ago showed a mean AJDL difference of 0.17 ± 0.1 mmol/L in healthy young men (Gibbs et al., 1942). The mean AJDL found in our patients was nearly equal to that value. However, in our patients the observed intra- and interindividual variability far exceeded the normal range. Although the mean differences between both jugular bulbs were usually low, their maximum bilateral discrepancies were marked and increased with increasing pathologic values (i.e., more negative AJDL, Table 2). Again, side-to-side differences in AJDL, especially in the pathologic range, rule out the interchangeability of right and left jugular venous monitoring.
Ischemia was far more often diagnosed by calculation of mLOI than by being detected by AJDL measurements. In head-injured patients oligemia in the contused areas is associated with a subsequent hyperemic rim around the contusion (Alexander et al., 1994). Provided that regional CBF in contused areas actually failed to meet metabolic demand, anaerobic metabolism and consequently regional lactate production in the affected areas occurs. Depending on the extent of ischemia and the size of the ischemic area, the resulting AJDL either exceeds the ischemic threshold (<–0.37 mmol/L) or not. Therefore, focal ischemia may be present even if AJDL is in the normal range. In this case, calculation of mLOI enables diagnosis of disproportionately elevated anaerobic glucose utilization and might be more sensitive for detecting focal ischemia than the AJDL alone. As was true for AJDL measurements, we also found unacceptable differences between both sides in paired mLOI samples. As demonstrated in Table 3, between 65% and 80% of ischemic events occurred exclusively unilaterally. In summary, even if only ischemic events are considered, our data suggest unsatisfactory agreement between both sides of jugular venous monitoring.
From the anatomic point of view, blood entering the brain via each internal carotid artery is distributed almost totally to the ipsilateral hemisphere. On the venous side the large dural sinuses in the midline drain both hemispheres, dividing their contents at the torcular with the blood being conveyed to the torcular via the lateral sinuses. Thus, almost complete mixing of the venous drainage from the two hemispheres should be expected. However, on average two thirds of the blood supplied to one hemisphere through an internal carotid artery is actually drained through the ipsilateral internal jugular vein (Shenkin et al., 1948). The anatomic basis for this observation is, first, the superficial cerebral veins passing directly to the lateral and petrosal sinuses, which join to form the jugular bulb, and second, the cross-section of any given vessel in the torcular is extremely variable, even relative to the cross-sectional area of the other vessels in the same torcular (Gibbs and Gibbs, 1934; Woodhall, 1936). These superficial cerebral veins are principally the inferior cerebral veins from the temporal lobe, the under surfaces of the occipital and frontal lobe, and the great vein of Labbé, which drains the lateral surface of the hemisphere (Shenkin et al., 1948). Considering the distribution of flow out of the superior sagittal sinus and straight sinus between the right and the left lateral sinus, the right lateral sinus was composed of, on average, 94.5% superior sagittal venous fluid and 5.5% straight sinus fluid, whereas the left lateral sinus contained on average 83.5% superior sagittal venous fluid and 16.5% straight sinus fluid. However, the interindividual variations in flow distribution were extremely high (Gibbs and Gibbs, 1934). In summary, the venous drainage of the brain is characterized by high interindividual variability. Therefore, venous outflow of specific areas in the brain cannot be clearly assigned to a particular jugular bulb. Furthermore, in persons without intracranial pathologic damage, it is suggested that the blood in either jugular bulb is representative of the drainage of all regions of the brain, and therefore the values obtained from such samples would be an approximate average of the whole (Lassen, 1959; Kety and Schmidt, 1948). Normally, based on coupling between cerebral blood flow and metabolism, local CBF increases or decreases depending on tissue metabolic requirements (Raichle et al., 1976). Because the ratio between cerebral metabolic rate for oxygen (CMRO2) and CBF does not change, according to the Fick equation (CMRO2 = AJDO2 × CBF) the arteriovenous oxygen content difference (AJDO2) remains constant. Thus, the oxygen saturation in venous blood drained from different areas of the brain should not be significantly different. Under these conditions, despite incomplete mixing of cerebral venous blood, the corresponding SJO2 values in both jugular bulbs are expected to agree within narrow bounds. However, in most severely head-injured patients coupling of CBF and CMRO2 is abnormal, resulting in regionally increased or decreased CBF independent of regional metabolic demand (Robertson et al., 1989). Under these conditions, incomplete mixing of cerebral venous blood results in different SJO2 and AJDL values in both jugular bulbs. Therefore, patient-to-patient variations in bilateral SJO2, AJDL, and mLOI differences, as found in our study, are reasonable. However, variations in bilateral differences with time in an individual patient are not adequately explained. From a theoretical point of view, the following circumstances might contribute to these findings. Cerebral blood flow is not inevitably homogeneous throughout the brain. Regional cerebral blood flow trends in head-injured patients have shown different dynamics in contused areas, the surrounding brain, and cortical blood flow (Alexander et al., 1994). If heterogeneous CBF is present, decreasing (global) CBF may cause different compensatory mechanisms depending on the actual relation of CBF to metabolism and the status of autoregulation. In areas with previously intact autoregulation and normal perfusion, vasodilation occurs causing relatively unchanged SJO2. In areas with prior hypoperfusion and impaired or exhausted autoregulation, decreasing CBF causes increasing oxygen extraction associated with decreasing SJO2. Consequently, bilateral SJO2 difference will rise. If this compensation is exhausted, ischemia follows manifested by a fall in CMRO2 and an increase in cerebral lactate production. As a result, oxygen extraction decreases and SJO2 will increase (Robertson et al., 1989). Again, bilateral SJO2 difference may change significantly as a function of time. Based on the sigmoid shape of the hemoglobin—oxygen dissociation curve comparable changes in bilateral P
Our data suggest an association between bilateral SJO2 differences and CPP. Although low cerebral perfusion pressure does not necessarily generate large bilateral SJO2 differences in an individual patient, greater variations occur overall if CPP declines. As mentioned, autoregulation of CBF may be heterogeneous throughout the brain. In areas with intact autoregulation, declining CPP does not change CBF. However, with respect to areas with impaired autoregulation, CBF declines causing an increase in oxygen extraction. Therefore, if intact autoregulation coincides with impaired autoregulation, greater SJO2 differences may occur. As expected from the association between SJO2 differences and CPP, intracranial hypertension may also increase SJO2 discrepancies. Impairment of microcirculation decreases regional CBF. In case of existing CBF heterogeneity, the same mechanism takes place as seen in declining CPP. In addition, CO2 vasoresponsivity may also be heterogeneous throughout the injured brain (Marion and Bouma, 1991). Hyperventilation reduces CBF in the corresponding brain areas in a different way, causing dynamic changes in bilateral SJO2 differences.
The knowledge about physiologic variations in venous outflow from the brain, the frequently occurring uncoupling of blood flow and metabolism, the effects of changes in CPP, ICP, and Pa
The final remaining question was whether we could prospectively identify the more appropriate side for diagnosing cerebral hypoperfusion or cerebral ischemia. We therefore tested the accuracy of unilateral jugular venous monitoring according to the various recommendations reported in the literature (Cruz et al., 1994; Dearden, 1991; Robertson et al., 1989). For each different pathologic measure investigated, the CT approach for selecting the appropriate side showed the highest sensitivity (Table 4). Combining diffuse and unbalanced lesions, the CT approach enables the detection of at least 84% and, at best, 94% of defined pathologic values. One might assume that these data support sufficient accuracy of unilateral monitoring, provided that the objective is detection of cerebral ischemia. However, it must be kept in mind that even a single ischemic episode may cause cerebral infarction associated with poor neurologic outcome (Robertson et al., 1987). On the basis of the bilateral differences in AJDL, SJO2, and mLOl found in our patients, one must argue that the incidence of cerebral hypoperfusion or ischemia in clinical investigations using unilateral jugular venous monitoring was probably too low. In an important study of diffuse head injuries, jugular venous monitoring was performed exclusively on the right side (Cruz et al., 1994). If we had applied this procedure to our data, 32% of pathologic mLOl values would have been missed. Using the methodology of another pioneering study of severely head-injured patients, in which jugular venous monitoring was performed on the right side if the injury was diffuse, would have also resulted in our missing 13 of 41 or 32% of the pathologic mLOl values (Robertson et al., 1989). In contrast, cannulation on the side of larger jugular foramen would reduce the rate of false negative mLOl data to 7 of 41, or 16%.
Some of the possible limitations of our results should also be addressed. First, technical errors such as inappropriate positioning of the catheter tip and rapid withdrawal of blood might occur. In our study, the correct positioning of both catheter tips high in the jugular bulbs just below the skull base was always confirmed by radiography. In this position the negligible proportion of about 3% of jugular venous blood arises from extracranial sources (Shenkin et al., 1948). To avoid additional extracerebral contamination by high sampling flow, venous sampling via the catheters was performed slowly (i.e., about 2 mL/min). Therefore, we can safely assume that representative cerebral venous blood was obtained for analysis.
The influence of an elevated arterial lactate content on AJDL affects the whole brain. In the present study, borderline AJDL values near the ischemic threshold possibly remained undetected, which would have reduced the incidence of ischemic episodes. However, the large increase in lactate production by the ischemic brain presumably overshadows the fluctuations in calculated AJDL as a result of changes in arterial lactate content. Bilateral AJDL differences would have been more likely reduced than pronounced by this influence. As long as samples are obtained during steady state conditions, these theoretical concerns appear to be of limited practical significance (Robertson et al., 1987). We did not measure cerebral blood flow and therefore evidence about absolute interhemispheric differences in cerebral oxygen consumption and cerebral lactate production, as well as the true mixed cerebral venous oxygen saturation, cannot be given. Nevertheless, even unilateral occurrence of pathologic values in SJO2, AJDL, and mLOl indicates at least regional disproportion of cerebral oxygen delivery and oxygen consumption and therefore requires therapeutic intervention. However, the significance of the differences in SJO2 falling in the normal range between 54% and 75% cannot be judged.
Potential complications can be described as those associated with insertion of the catheter, including carotid puncture, nerve injury, and pneumothorax, and those associated with the catheter remaining in the jugular vein, including increase in ICP, thrombosis, and infection. The most common complication during insertion is carotid puncture. The incidence found in our study (2.3%) corresponds to the results previously reported in the literature. In a study of 123 pediatric patients common carotid artery puncture occurred in 3% of patients (Goetting and Preston, 1990). Monitoring SJO2 bilaterally, puncture of the carotid artery was recorded in 2 of 45 (4%) attempts (Stocchetti et al., 1994). As found in our study, there were no sequelae in either of these series. Hemostasis was achieved without exception by applying local pressure for several minutes.
Particularly in cases of reduced intracranial compliance, ICP can be increased by procedures that obstruct venous outflow from the brain. Possible causes include endovascular obstruction by indwelling catheters or thrombosis, and external venous compression by hematoma or edema. Therefore, it is reasonable to be concerned that a catheter in the jugular vein might raise ICP. However, the 4F introducer sheath, as used in our study, is quite small relative to the lumen of the internal jugular vein. Even pediatric patients with 4F jugular bulb catheters showed no significant rise in ICP either during or after cannulation. The maximum rise in ICP was 2 mm Hg in a single patient, whereas six others showed a decrease in ICP. The authors revealed no evidence of jugular venous obstruction in the catheterized vessel and concluded that jugular bulb cannulation can be performed in patients without causing further increases in elevated ICP. (Goetting and Preston, 1991). Bilateral jugular bulb catheter insertion in adults was associated with only minor ICP elevations of no clinical significance (Stocchetti et al., 1994). Large perivasular hematoma may even cause complete obstruction of ipsilateral venous return. Possible, serious sequelae are marked increases in ICP and facilitation of venous thrombosis. Venous hematomas were noted in 2.3% of jugular bulb catheterizations, all occurring in patients with multiple punctures (Goetting and Preston, 1990). Therefore, multiple punctures should be avoided, especially in patients with coagulopathy. Clinically relevant venous thrombosis around the indwelling catheters is uncommon in adults without malignancies and, although it has not been reported with jugular bulb catheters, it could have serious consequences. Obstruction of venous return from the head increases cerebral blood volume and subsequently ICP. At worst, bilateral jugular venous thrombosis or cerebral venous sinus thrombosis occurs. Although uncommon, both are potentially fatal complications. Based on experience with other intravascular catheters, the larger the catheter relative to the size of the vessel and the lower the flow through the vessel, the greater the risk of thrombosis. To minimize this risk of thrombosis it seems reasonable to flush the system continuously with heparinized saline. Line sepsis is associated with all types of indwelling catheters and occurs in most studies at an overall rate of between 0% and 5% (Seneff and Rippe, 1985). As also reported by other investigators (Goetting and Preston, 1990), no cases of catheter-induced infection were observed in our patients.
Under the conditions of this study, which showed wide limits of agreement in addition to a high frequency of crossover values between both sides in individual patients, we conclude that arbitrarily selecting a particular side for SJO2 monitoring causes misleading results in a large number of patients. Furthermore, even when using CT-guided unilateral SJO2 monitoring, the most appropriate value for making therapeutic decisions cannot be definitively determined. For diagnosing ischemic events based on jugular venous desaturation and pathologic AJDL and mLOI values, the CT approach using the side of predominant injury for unbalanced lesions, or the side of the larger jugular foramen for diffuse or balanced lesions has the highest sensitivity. We conclude that in an individual patient presenting intracranial pathology, as is the case with severe head injury, even calculated unilateral jugular venous monitoring has an unpredictable risk for misleading or missing data.
