Abstract
Cerebral large arteries dilate actively around the lower limits of CBF autoregulation, mediated at least partly by nitric oxide, and maintain CBF during severe hypotension. We tested the hypothesis that this autoregulatory response of large arteries, as well as the response of arterioles, is altered in spontaneously hypertensive rats (SHR) and that the altered response reverts to normal during long-term antihypertensive treatment with cilazapril, an angiotensin-converting enzyme inhibitor. In anesthetized 6- to 7-month-old normotensive Wistar-Kyoto rats (WKY), 4- and 6- to 7-month-old SHR without antihypertensive treatment, and 6- to 7-month-old SHR treated with cilazapril for 10 weeks, local CBF to the brain stem was determined with laser—Doppler flowmetry and diameters of the basilar artery and its branches were measured through a cranial window during stepwise hemorrhagic hypotension. The lower limit of CBF autoregulation shifted upward in untreated SHR to 90 to 105 mm Hg from 30 to 45 mm Hg in WKY, and it reverted to 30 to 45 mm Hg in treated SHR. In response to severe hypotension, the basilar artery dilated by 21 ± 6% (mean ± SD) of the baseline internal diameter in WKY. The vasodilation was impaired in untreated SHR (10 ± 8% in 4-mo-old SHR and 4 ± 5% in 6- to 7-month-old SHR), and was restored to 22 ± 10% by treatment with cilazapril (P < 0.005). Dilator responses of branch arterioles to hypotension showed similar attenuation and recovery as that of the basilar artery. The data indicate that chronic hypertension impairs the autoregulatory dilation of the basilar artery as well as branch arterioles and that antihypertensive treatment with cilazapril restores the diminished dilation toward normal.
Autoregulation of CBF, an intrinsic ability to maintain constant cerebral perfusion in the face of blood pressure changes, is modified or disturbed by chronic hypertension in both forebrain and hindbrain circulations (Strandgaard et al., 1973, 1975; Jones et al., 1976; Fujishima and Omae, 1976; Barry et al., 1982; Sadoshima and Heistad, 1983). The disturbance presumably results mainly from the attenuation of the autoregulatory responses of cerebral arterioles because of their morphologic and functional alterations during chronic hypertension (Folkow et al., 1970; Hazama et al., 1978; Hart et al., 1980; Mayhan et al., 1987, 1988; Baumbach et al., 1988). We have recently demonstrated that large arteries, such as the basilar artery, also dilate actively and contribute to reductions in cerebrovascular resistance around the lower limits of CBF autoregulation, and thereby maintain CBF to the brain stem during severe hypotension (Toyoda et al., 1996). Chronic hypertension alters the structure and mechanics of cerebral large arteries as well as those of arterioles (Winquist and Bohr, 1983; Mayhan, 1990; Hajdu and Baumbach, 1994). Thus, it is anticipated that the autoregulatory response of large arteries is also attenuated during chronic hypertension. The first goal of this study was to determine the effect of chronic hypertension on autoregulatory responses of the basilar artery and its branch arterioles during acute hypotension in vivo.
Altered CBF autoregulation during chronic hypertension was restored to normal during antihypertensive treatment (Strandgaard, 1976; Hoffman et al., 1982; Vorstrup et al., 1984). Treatment also attenuated cerebral vascular hypertrophy (Folkow et al., 1971; Ibayashi et al., 1986; Clozel et al., 1989; Hajdu et al., 1991) and preserved various endothelium-dependent and -independent responses (Yang et al., 1993; Takaba et al., 1996). The second goal of this study was to examine whether long-term antihypertensive treatment with cilazapril, an angiotensin-converting enzyme (ACE) inhibitor, restores the diminished autoregulatory response of the basilar artery, as well as arterioles, to the normal level and recovers CBF autoregulation in the brain stem circulation during acute hypotension in vivo.
METHODS
Animal preparation
Experiments were performed in male Wistar-Kyoto rats (WKY, n = 6) and spontaneously hypertensive rats (SHR, n = 17) of the Izm strain (WKY/Izm, SHR/Izm), obtained from the Disease Model Cooperative Research Association (Kyoto, Japan) (Nabika et al., 1991). At the age of 4 months, we performed the experiment in five untreated SHR, and other SHR were randomly divided into two groups: a treated group that received cilazapril (approximately 9 mg·kg−1·day−1) in the drinking water for 10 weeks (n = 6) and an untreated group that received tap water (n = 6). The WKY rats were allowed free access to tap water. All the rats were fed stock chow diet (Clea Japan Inc., Tokyo, Japan), which contains 0.3% (by weight) sodium, during observation periods. Systolic blood pressure was measured with the tail-cuff method at 1-month intervals. Experiments were conducted when the rats were 6 to 7 months of age.
Rats were anesthetized intraperitoneally with 50 mg/kg amobarbital sodium, and were supplemented intravenously with this drug at a rate of approximately 15 to 20 mg · kg−1 · hour−1. The depth of anesthesia was examined often by observing the effects of pressure to the paw on heart rate and blood pressure. The trachea was cannulated and each rat was mechanically ventilated with room air and supplemental oxygen after immobilization with 5 to 10 mg/kg d-tubocurarine chloride. Catheters were placed in both femoral arteries, one for measuring systemic arterial pressure, and the other for withdrawing blood to change systemic arterial pressure and for sampling of arterial blood. A femoral vein was cannulated for the infusion of drugs. Rectal temperature was maintained at 37°C with a heating pad.
A craniotomy was prepared over the ventral brain stem as previously described in detail (Faraci et al., 1987; Fujii et al., 1991a; Toyoda et al., 1996, 1997a). Portions of the dura and pia mater and the arachnoid membrane were resected. The open cranial window was suffused with artificial CSF (NaCl, 132; KCl, 2.95; CaCl2, 1.71; MgCl2, 0.65; NaHCO3, 24.6;
Cerebral blood flow to the ventral brain stem was measured by laser—Doppler flowmetry (ALF 21D Model, Advance Co. Ltd., Tokyo, Japan) equipped with a 550-μm-diameter needle probe as described previously (Fujii et al., 1991b; Toyoda et al., 1996, 1997a). Briefly, the probe was placed lateral to, but near, the basilar artery in the cranial window and advanced into the CSF approximately 0.2 mm above the surface of the brain stem. We regarded laser—Doppler flowmetry outputs as arbitrary units and expressed changes in CBF as a percentage of the baseline CBF.
Experimental protocol
Responses of the basilar artery and branches to acetylcholine (10−6 and 10−5 mol/L) and sodium nitroprusside (10−8 and 10−7 mol/L) were examined in each group. Drugs were dissolved in artificial CSF and suffused over the craniotomy for 5 minutes. Vessel diameters were measured immediately before application of each agonist and during the last minute of application.
Stepwise hypotension, approximately 15 mm Hg per step, was then induced by controlled withdrawal of blood through the femoral arterial cannula. We maintained MABP at each step for approximately 4 minutes and measured vessel diameter and CBF simultaneously during the last minute, because these parameters were usually stable during the last 2 minutes at each step.
Statistical analysis
Values are expressed as mean ± SD. One-way factorial analysis of variance was used for comparison of variables among groups. One-way repeated-measures analysis of variance was used for comparison of CBF within a group to determine the lower limit of CBF autoregulation. Post hoc analysis for significance was made with Fisher's protected least significant difference test. A level of P < 0.05 was accepted as statistically significant.
RESULTS
Long-term treatment with cilazapril decreased systolic pressure in SHR to a level similar to that in WKY throughout the experimental period (Fig. 1). Values for arterial blood gases at rest and during hypotension were similar among the groups (Table 1). Acetylcholine (10−6 and 10−5 mol/L) -induced dilation of the basilar artery and large branch was significantly reduced in untreated SHR compared with WKY and treated SHR (P < 0.05), and that of the small branch tended to be reduced in untreated SHR (0.05 < P < 0.1), whereas the response to sodium nitroprusside (10−8 and 10−7 mol/L) did not differ among the groups (Table 2).
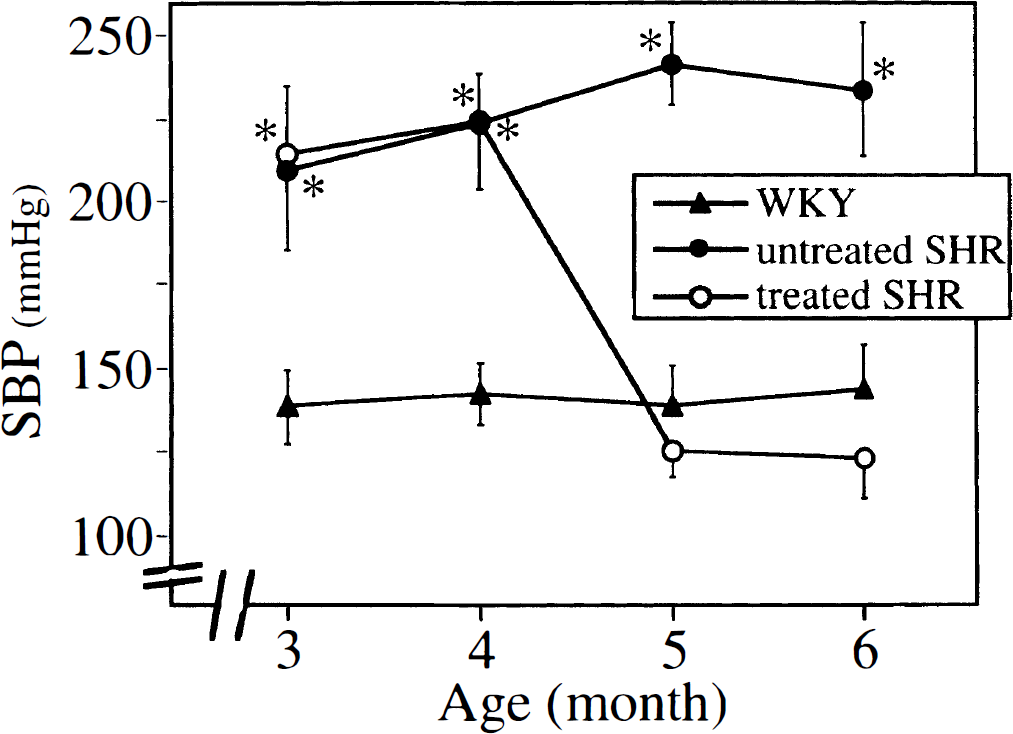
Changes in systolic blood pressure (SBP) of WKY (n = 6), untreated SHR (n = 6), and SHR treated with cilazapril (n = 6) during observation period. Values are means ± SD. * P < 0.0001 versus WKY. SHR, spontaneously hypertensive rats; WKY, Wistar-Kyoto rats.
Physiologic variables
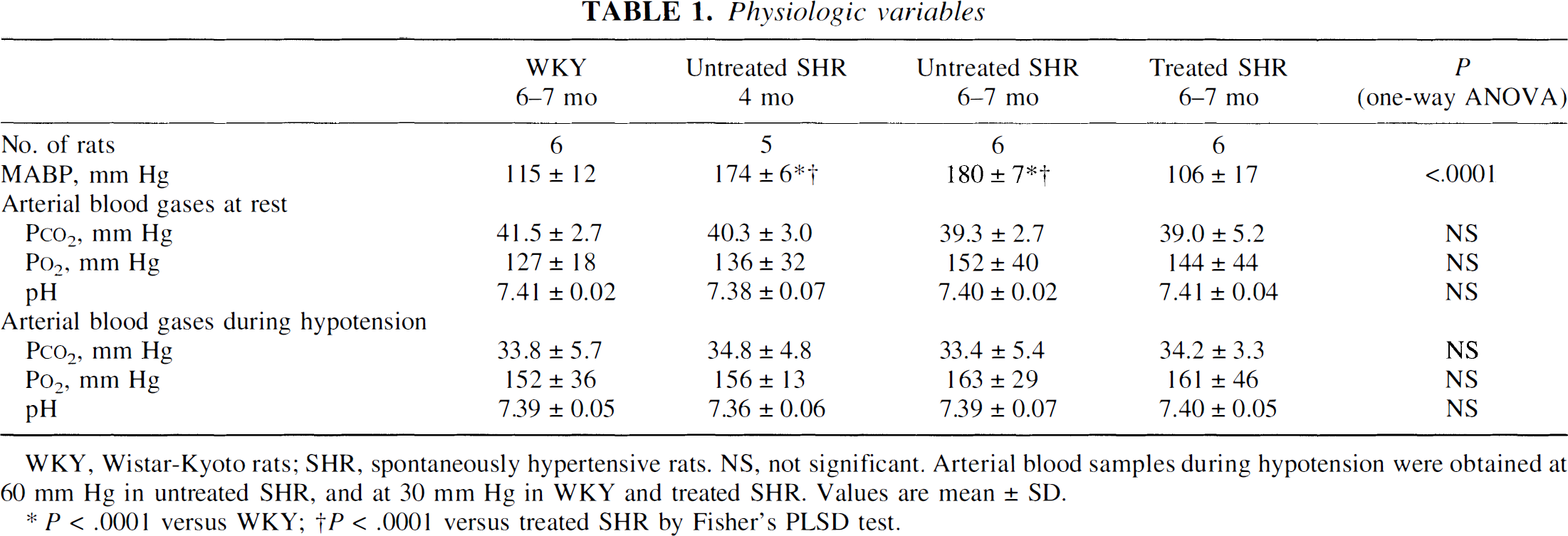
WKY, Wistar-Kyoto rats; SHR, spontaneously hypertensive rats. NS, not significant. Arterial blood samples during hypotension were obtained at 60 mm Hg in untreated SHR, and at 30 mm Hg in WKY and treated SHR. Values are mean ± SD.
P < .0001 versus WKY
P < .0001 versus treated SHR by Fisher's PLSD test.
Baseline vessel diameters and their changes by topical application of drugs
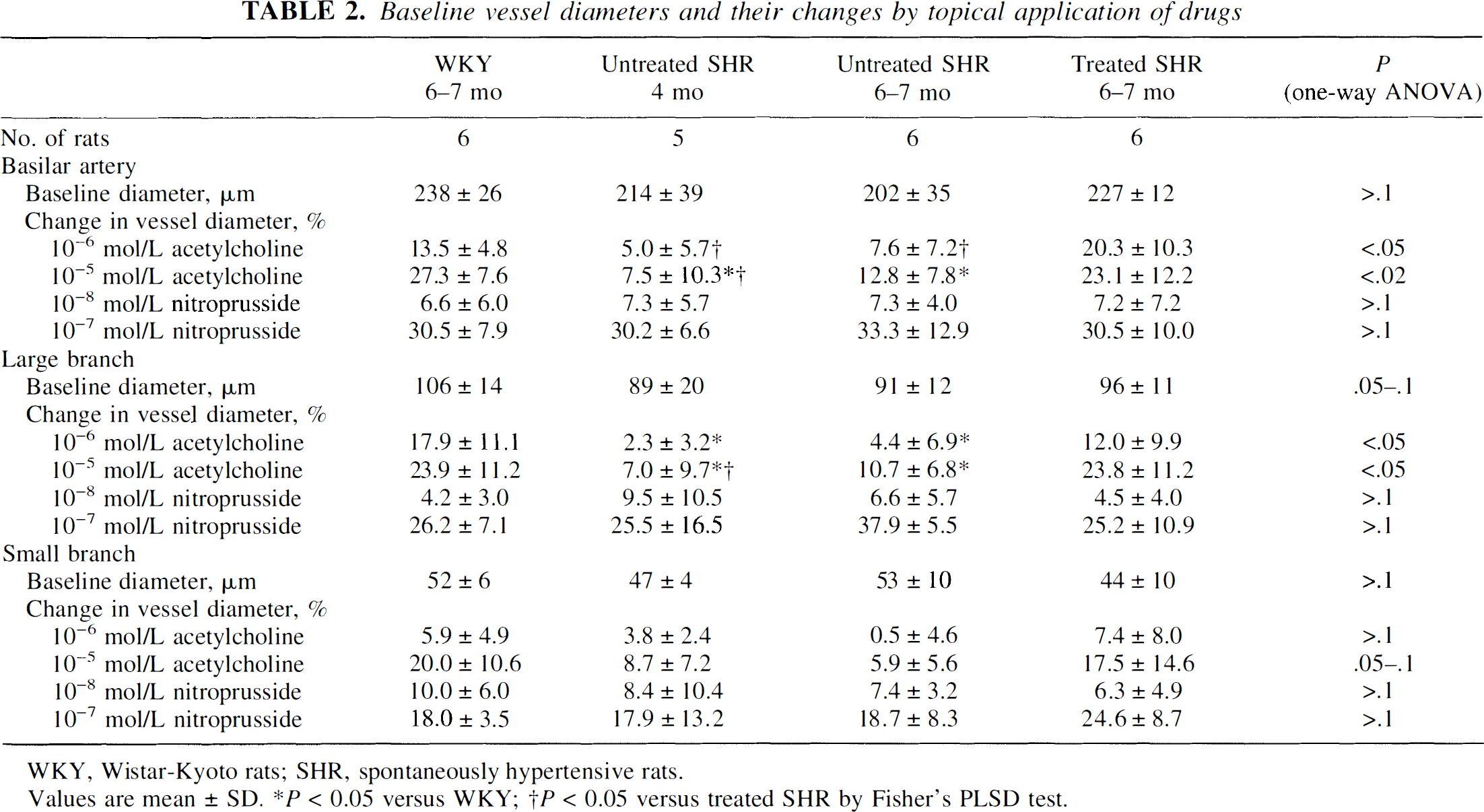
WKY, Wistar-Kyoto rats; SHR, spontaneously hypertensive rats.
Values are mean ± SD. *P < 0.05 versus WKY; †P < 0.05 versus treated SHR by Fisher's PLSD test.
In 4-month-old untreated SHR, CBF was preserved during reduction of MABP from 174 ± 6 to 90 mm Hg, decreased significantly at 75 mm Hg (P < 0.05), and fell steeply thereafter (Fig. 2). In 6- to 7-month-old untreated SHR, CBF was well maintained at MABP from 180 ± 7 to 105 mm Hg, and decreased significantly at 90 mm Hg (P < 0.05). In WKY and treated SHR, CBF was constant at MABP from 115 ± 12 mm Hg and 106 ± 17 mm Hg, respectively, to 45 mm Hg, and decreased significantly at 30 mm Hg (P < 0.05). Thus, the lower limit of CBF autoregulation was 75 to 90 mm Hg in 4-month-old untreated SHR, 90 to 105 mm Hg in 6- to 7-month-old untreated SHR, and 30 to 45 mm Hg in WKY and treated SHR.
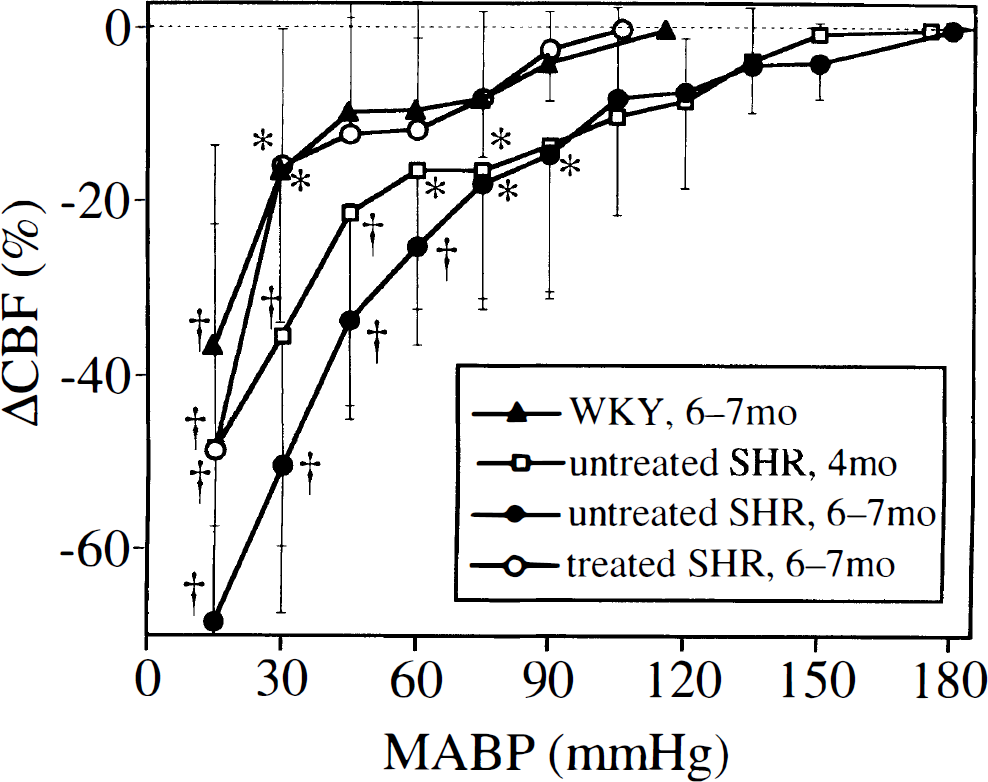
Changes in cerebral blood flow (CBF) to the brain stem during stepwise hypotension in WKY (6- to 7-month-old, n = 6), untreated SHR (4-month-old, n = 5; 6- to 7-month-old, n = 6), and SHR treated with cilazapril (6- to 7-month-old, n = 6). Values are means ± SD. *P < .05, †P < .01 versus baseline CBF before hypotension.
Internal diameter of the basilar artery increased by less than 10% during severe hypotension in 4-month-old untreated SHR, and was almost unchanged throughout hypotension in 6- to 7-month-old untreated SHR (Fig. 3). In WKY and treated SHR, it increased steeply and exceeded the baseline diameter by 20% at 30 mm Hg. Large and small branches dilated less than 20% of the baseline value during severe hypotension in 4-month-old untreated SHR, and less than 10% in 6- to 7-month-old untreated SHR, whereas they dilated more than 30% at 30 mm Hg in WKY and treated SHR. The maximum change in diameter of each vessel was significantly different between both untreated SHR (4- and 6- to 7-month-old) and the other two groups (P < 0.005 in the basilar artery, P < 0.05 in the large branch, and P < 0.01 in the small branch; Fig. 4).
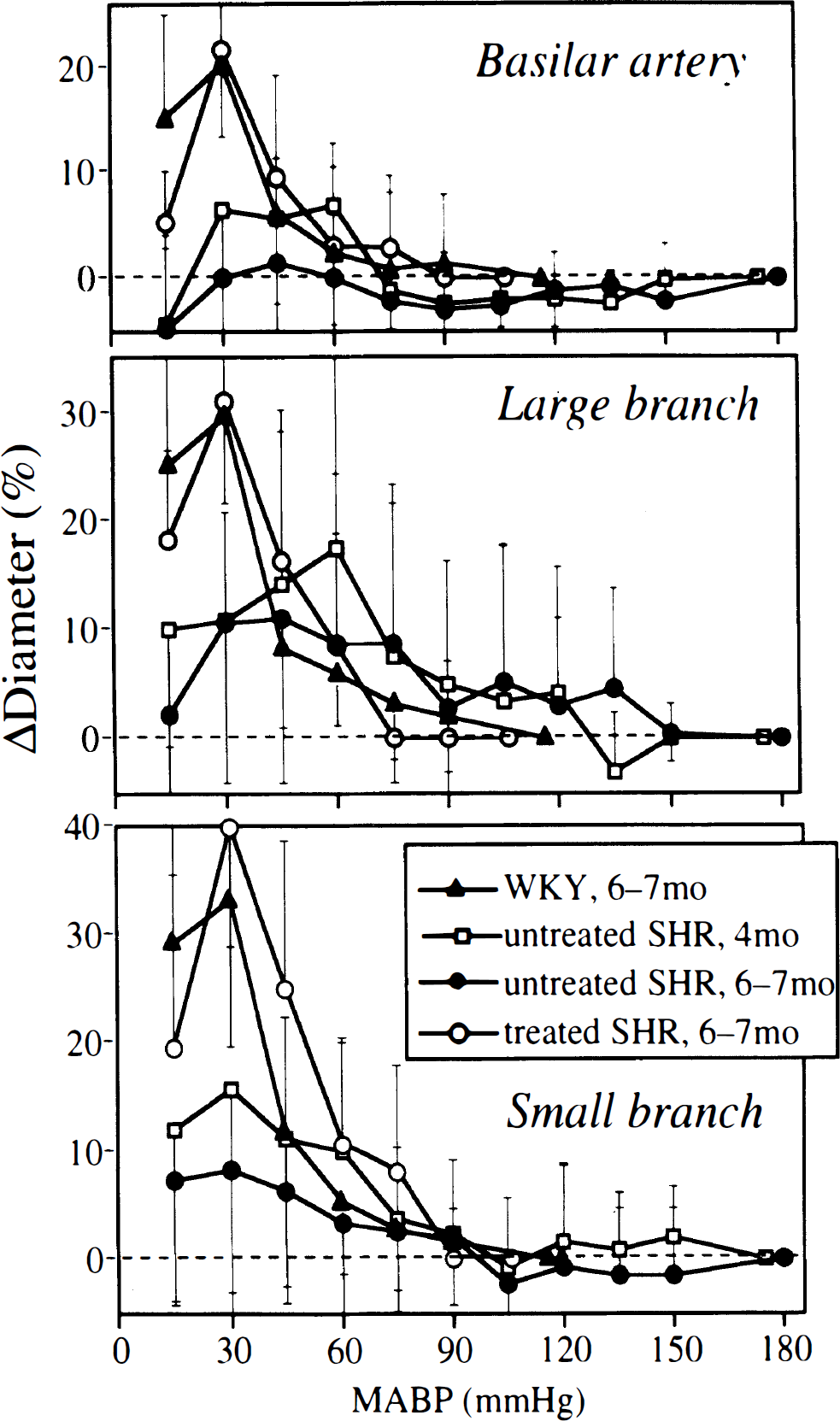
Changes in the internal diameter of the basilar artery, large branch with a baseline diameter ≥70 μm, and small branch with a baseline diameter <70 μm during stepwise hypotension in WKY (6 to 7-month-old, n = 6), untreated SHR (4-month-old, n = 5; 6- to 7-month-old, n = 6), and SHR treated with cilazapril (6- to 7-month-old, n = 6). Values are means ± SD.
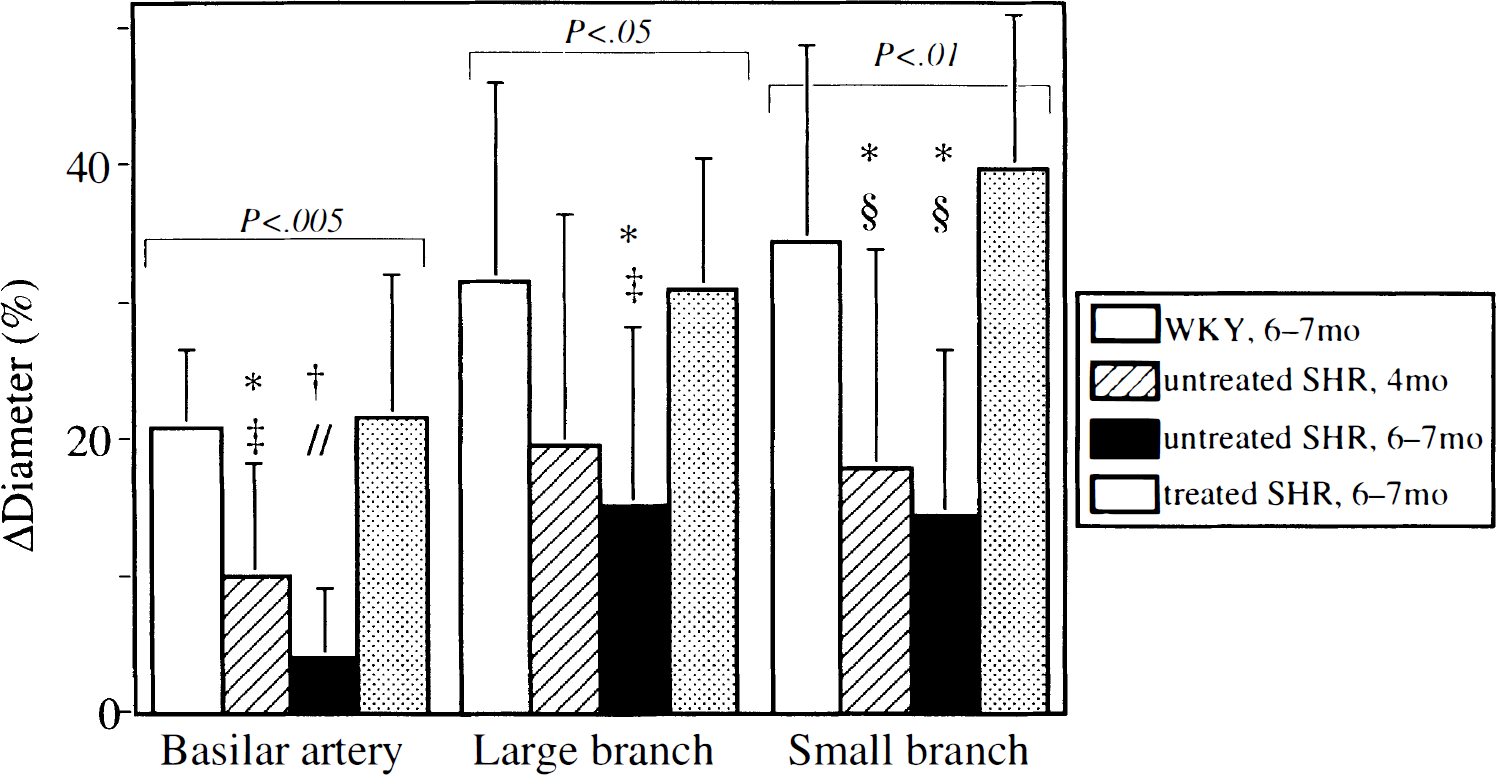
Maximum changes in the internal diameter of the basilar artery, large branch with a baseline diameter ≥70 μm, and small branch with a baseline diameter <70 μm during hypotension in WKY (6- to 7-month-old, n = 6), untreated SHR (4-month-old, n = 5, 6- to 7-month-old, n = 6), and SHR treated with cilazapril (6- to 7-month-old, n = 6). Values are means ± SD. P < 0.005 among the four groups in the basilar artery, P < 0.05 in the large branch, and P < 0.01 in the small branch by one-way factorial analysis of variance. *P < 0.05, †P < 0.001, versus WKY, ‡P < 0.05 §P < 0.005, ‖P < 0.001, versus treated SHR by Fisher's protected least significant difference test.
DISCUSSION
This study examined the effects of chronic hypertension and antihypertensive treatment on autoregulatory dilation of the basilar artery along with that of branch arterioles in response to acute hypotension. There are two major findings in the current study. First, the autoregulatory dilator response of the basilar artery was almost abolished and that of arterioles was severely attenuated in SHR compared with WKY. Second, the diminished response of the basilar artery and arterioles in SHR reverted to the level of WKY during long-term antihypertensive treatment with cilazapril. Impairment of both CBF autoregulation and dilator responses of different-sized vessels during chronic hypertension and their concurrent recovery during antihypertensive treatment suggest that the large arteries, in conjunction with arterioles, play a role in autoregulatory adjustments in the brain stem circulation, as we previously proposed (Toyoda et al., 1996).
Effects of hypertension on brain stem autoregulation
Cerebral blood flow autoregulation of the brain stem circulation, as well as the forebrain circulation, is altered by chronic hypertension (Sadoshima and Heistad, 1983). This study has also demonstrated that the dilator response to hypotension was impaired in both large arteries and arterioles of the brain stem. The impairment of the response, in conjunction with presumably altered function in intraparenchymal arterioles, appears to disturb CBF autoregulation of the brain stem in SHR. Moreover, impairment of the responses was not simply caused by genetic differences between SHR and WKY (Kurtz et al., 1989), because antihypertensive treatment restored the responses in SHR toward normal.
The altered vasodilator responses may result from the structural and functional alterations of arteries and arterioles of the brain stem during chronic hypertension; structural features include reduction in lumen size as a result of vascular hypertrophy and “remodeling,” with a reduction in external diameter (Baumbach et al., 1988), and the functional features are dysfunction of endothelium and preserved smooth muscle relaxation (Heistad et al., 1995). We recently demonstrated that topical application of an inhibitor of nitric oxide synthase attenuated dilator responses of the basilar artery but did not change those of the branches during severe hypotension in vivo, suggesting that nitric oxide may mediate the autoregulatory response of the basilar artery (Toyoda et al., 1997b). Thus, endothelial dysfunction of the basilar artery (confirmed by attenuated response to acetylcholine) may cause, at least in part, the impairment of the autoregulatory vasodilation in this study.
The attenuated autoregulatory response of branch arterioles does not seem to result from the inhibition of nitric oxide synthase by endothelial dysfunction (Toyoda et al., 1991b). Opening of ATP-sensitive potassium (KATP) channels has been demonstrated to contribute to CBF autoregulation; topical application of glibenclamide, a KATP channel blocker, disturbed the autoregulatory dilation of rat pial arterioles (Lee et al., 1993; Hong et al., 1994). We recently reported that glibenclamide also disturbed dilator responses of brain stem arterioles and raised the lower limit of CBF autoregulation in the brain stem (Toyoda et al., 1997c). Moreover, responses to opening of KATP channels are disturbed in cerebral vessels during chronic hypertension (Kitazono et al., 1993). Thus, the impairment of the autoregulatory dilation of branch arterioles in SHR may result from the altered response to this channel. The production of a cyclooxygenase constrictor substance is another likely mechanism for the impairment of cerebral vasodilator responses in SHR (Mayhan et al., 1988).
Effects of antihypertensive treatment on brain stem autoregulation
Cerebral blood flow autoregulation of SHR during hypotension was reported to be disturbed in animals as young as 3 to 4 months old (Hoffman et al., 1981; Barry et al., 1982). The present study has demonstrated that autoregulatory vasodilation of different-sized vessels is also diminished in 4-month-old SHR. Moreover, treatment with cilazapril does not merely prevent the aggravation of brain stem autoregulation during chronic hypertension but also restores the previously altered autoregulation toward normal.
Antihypertensive treatment with drugs other than ACE inhibitors also restored altered CBF autoregulation toward normal (Strandgaard, 1976; Hoffman et al., 1982; Vorstrup et al., 1984). Thus, we believe that recovery of dilator responses of brain stem vessels as well as brain stem blood flow autoregulation after treatment with cilazapril is produced, at least in part, by its effect on blood pressure. It is possible, however, that acute as well as chronic specific effects related to ACE inhibition may also explain our findings. Actually, acute intravenous administration of captopril, another ACE inhibitor, was reported to decrease to some extent the lower limit of CBF autoregulation in the forebrain circulation in SHR (Barry et al., 1984; Waldemar, 1990). Thus, the lower limit of brain stem autoregulation may also shift downward by acute administration of cilazapril. In addition, it is suggested that chronic treatment with cilazapril but not hydralazine prevents vascular remodeling in stroke-prone SHR (SHRSP) (Hajdu et al., 1991). Acetylcholine-induced relaxation of the isolated aorta is preserved in SHRSP treated with cilazapril but not with hydralazine (Clozel et al., 1990). We did not determine, however, to what extent the effect of cilazapril in this study was produced by its antihypertensive effect and by other specific effects related to ACE inhibition. In a future study, we plan to clarify mechanisms of changes in brain stem autoregulation by treatment with cilazapril.
In any event, recovery of the dilator responses of both the basilar artery and branch arterioles during treatment with cilazapril does not seem to conflict with our previous findings that nitric oxide and KATP channels are important modulators of autoregulatory responses of the brain stem vessels (Toyoda et al., 1997b, 1997c), because treatment with cilazapril prevented the endothelial dysfunction (Clozel et al., 1990; Yang et al., 1993) as well as impaired response to a KATP channel opener (Takaba et al., 1996).
In summary, chronic hypertension attenuated the autoregulatory responses of the brain stem artery and arterioles, and antihypertensive treatment with cilazapril restored them toward normal in vivo.
Footnotes
Abbreviations used
Acknowledgments
Cilazapril was kindly provided by Nippon Roche KK, Tokyo, Japan. The authors thank Drs. Jiro Kitayama and Hiroshi Nakamura for their technical supports.
