Abstract
We tested the hypothesis that nitric oxide (NO) plays a role in CBF autoregulation in the brain stem during hypotension. In anesthetized rats, local CBF to the brain stem was determined with laser-Doppler flowmetry, and diameters of the basilar artery and its branches were measured through an open cranial window during stepwise hemorrhagic hypotension. During topical application of 10−5 mol/L and 10−4 mol/L Nω-nitro-L-arginine (L-NNA), a nonselective inhibitor of nitric oxide synthase (NOS), CBF started to decrease at higher steps of mean arterial blood pressure in proportion to the concentration of L-NNA in stepwise hypotension (45 to 60 mm Hg in the 10−5 mol/L and 60 to 75 mm Hg in the 10−4 mol/L L-NNA group versus 30 to 45 mm Hg in the control group). Dilator response of the basilar artery to severe hypotension was significantly attenuated by topical application of L-NNA (maximum dilatation at 30 mm Hg: 16 ± 8% in the 10−5 mol/L and 12 ± 5% in the 10−4 mol/L L-NNA group versus 34 ± 4% in the control group), but that of the branches was similar between the control and L-NNA groups. Topical application of 10−5 mol/L 7-nitro indazole, a selective inhibitor of neuronal NOS, did not affect changes in CBF or vessel diameter through the entire pressure range. Thus, endothelial but not neuronal NO seems to take part in the regulation of CBF to the the brain stem during hypotension around the lower limits of CBF autoregulation. The role of NO in mediating dilatation in response to hypotension appears to be greater in large arteries than in small ones.
Autoregulation of CBF is an intrinsic ability to maintain constant CBF despite blood pressure changes. Pial arterioles have been regarded as the primary sites of autoregulatory function by changing their calibers in response to increase or decrease in blood pressure (Kontos et al., 1978a; MacKenzie et al, 1979), whereas the role of the larger cerebral arteries in CBF autoregulation has been poorly understood. We have recently shown that large arteries, such as the basilar artery, also contribute to reductions in cerebrovascular resistance around the lower limits of CBF autoregulation, and thereby maintain CBF to the brain stem during severe hypotension (Toyoda et al., 1996). However, in the study, the mechanism for dilatation of the basilar artery and its branches during hypotension remained undetermined.
Nitric oxide (NO) or a closely related compound was initially identified as the endothelium-derived relaxing factor (Ignarro et al., 1987; Palmer et al., 1987), and now is also shown to be produced by neurons and glia (Bredt et al., 1990; Knowles et al., 1989). Several studies indicated that NO modulates both basal cerebral vessel tone and vascular responses under specific pathophysiological circumstances like hypercapnia (Faraci, 1990; Wang et al., 1992a; Buchanan and Phillis, 1993; Pelligrino et al., 1993). In contrast, several conflicting results have been reported regarding the role of NO in CBF autoregulation (Wang et al., 1992b; Buchanan and Phillis, 1993; Tanaka et al., 1993; Kelly et al., 1994; Kobari et al., 1994; Takahashi et al., 1995).
The first goal of this study was to examine the hypothesis that NO regulates CBF autoregulation in the the brain stem as well as autoregulatory responses of different-sized vessels during hypotension. For this purpose, we used an open cranial window to measure local CBF and vessel diameters of the brain stem simultaneously in the presence and absence of Nω-nitro-
Recently, 7-nitro indazole (7-NI) was found to be a relatively selective inhibitor of neuronal NOS (Babbedge et al., 1993; Moore et al., 1993). This compound produces potent inhibition of rat cerebellar NOS with an IC50 of 0.9 ± 0.1 μmol/L (mean ± SEM) (Babbedge et al., 1993). Using this compound and other nonselective NOS inhibitors, we can differentiate the origin of NO involved in the CBF autoregulation. Thus, the second goal of this study was to determine, by using 7-NI, whether endothelial NO or neuronal NO is primarily responsible for autoregulatory adjustments.
METHODS
Animal preparation
Experiments were performed on 43 male Sprague-Dawley rats, weighing 300 to 450 g. Rats were anesthetized with amobarbital sodium, an initial dose of 50 mg/kg intraperitoneally, and followed by additional doses of 15 to 20 mg·kg−1 h−1 intravenously. The depth of anesthesia was examined often by observing the effects of pressure to the paw on heart rate and blood pressure. The trachea was cannulated and each rat was mechanically ventilated with room air and supplemental oxygen after immobilization by 5 to 10 mg/kg d-tubocurarine chloride. Catheters were placed in both femoral arteries: one for the measurement of systemic arterial pressure, and the other for withdrawing blood to change systemic arterial pressure and for sampling arterial blood. A femoral vein was cannulated for the infusion of drugs. Rectal temperature was maintained at 37°C with a heating pad.
A craniotomy was prepared over the ventral brain stem as previously described in detail (Faraci et al., 1987; Fujii et al., 1991a; Toyoda et al., 1996). Portions of the dura and pia mater and the arachnoid membrane were resected. The open cranial window was suffused with artificial CSF (composition in mmol/L: 132 NaCl, 2.95 KCl, 1.71 CaCl2, 0.65 MgCl2, 24.6 NaHCO3, and 3.69 D-glucose), warmed to 37°C, and continuously bubbled with a gas mixture of 5% CO2 and 95% N2. Suffusion rate of CSF using inlet and outlet ports over the exposed brain stem was 3.0 mL/min. In CSF sampled from the craniotomies, P
Cerebral blood flow to the ventral brain stem was measured by laser-Doppler flowmetry (ALF 21D Model, Advance Co. Ltd., Tokyo, Japan) equipped with a 550-μm-diameter needle probe as described previously (Fujii et al., 1991b; Toyoda et al., 1996). Briefly, the probe was placed lateral to, but near the basilar artery in the cranial window and advanced into the CSF approximately 0.2 mm above the surface of the brain stem. We regarded laser-Doppler flowmetry outputs as arbitrary units and expressed changes in CBF as a percentage of the baseline CBF.
Experimental protocol
Four treatment groups and a control group of animals were studied. Topical application of 10−5 mol/L
Responses of the basilar artery and its branches to 10−5 mol/L acetylcholine and 10–7 mol/L sodium nitroprusside were examined at baseline and during topical application of drugs before hypotension as well as at the MABP of 30 mm Hg in each group.
Statistical analysis
Values are expressed as means ± SD. One-way repeated-measures analysis of variance (ANOVA) followed by Fisher's PLSD test for significance was used for the comparison of stepwise CBF and vasodilator responses with the baseline value within a group. One-way factorial ANOVA followed by Fisher's PLSD test was used for the comparison of the responses among groups. Two-way repeated-measures ANOVA followed by Fisher's PLSD test was used for the comparison of whole MABP-response curves of vasodilation among groups. P < 0.05 was accepted as statistically significant.
RESULTS
Baseline physiological variables were within normal limits and similar among groups (Table 1). Topical application of drugs had no effect on systemic arterial blood pressure. Arterial P
Physiological variables
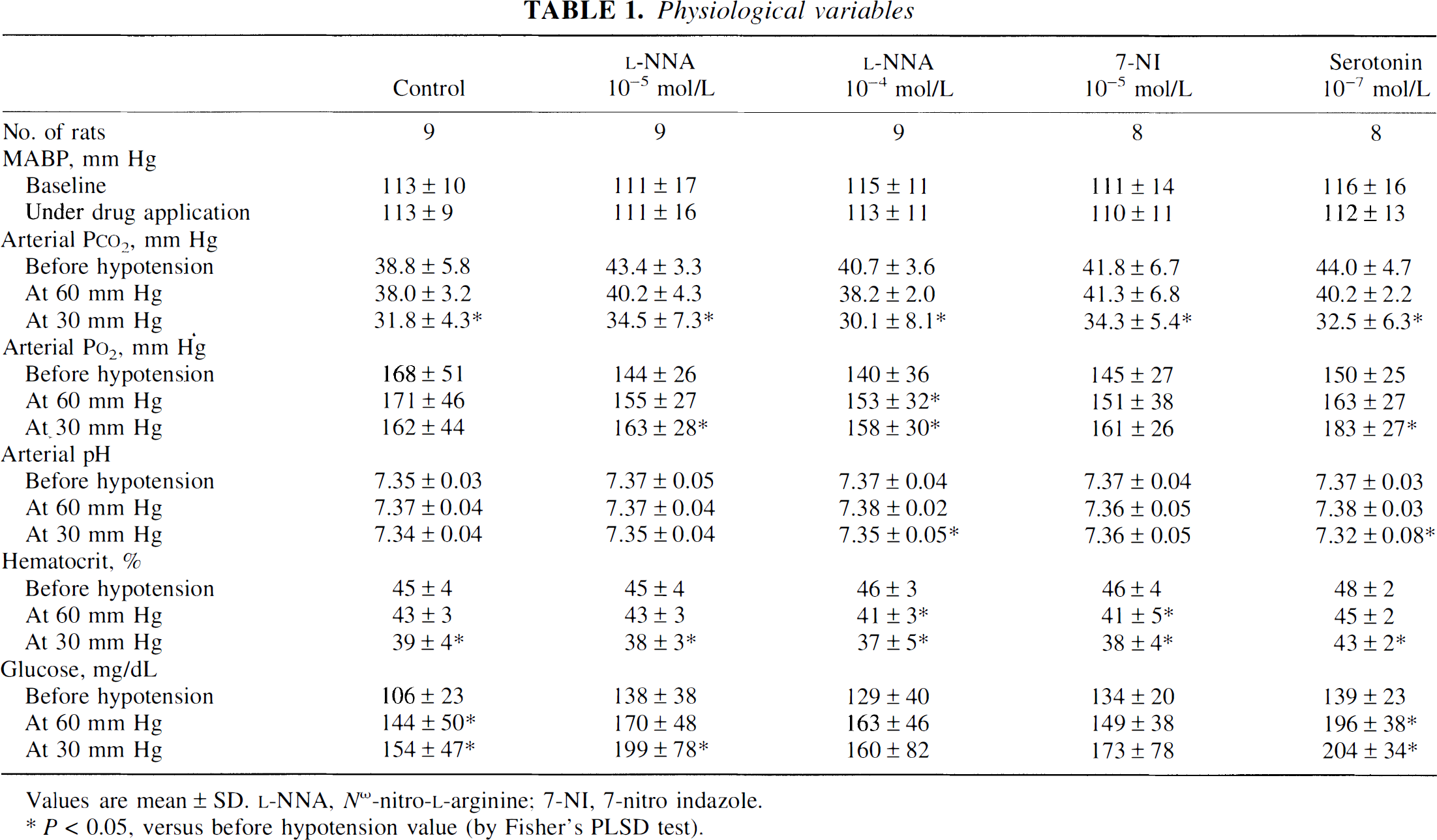
Values are mean ± SD.
P < 0.05, versus before hypotension value (by Fisher's PLSD test).
Effects of l-NNA and 7-NI on CBF and its changes during hypotension
Topical application of saline in the control group had no effect on CBF (0 ± 4% from the baseline value). Topical application of
In the control group, CBF was well maintained until MABP was reduced to 45 mm Hg and decreased significantly thereafter (Fig. 1). CBF started to decrease significantly at 45 mm Hg in the 10−5 mol/L
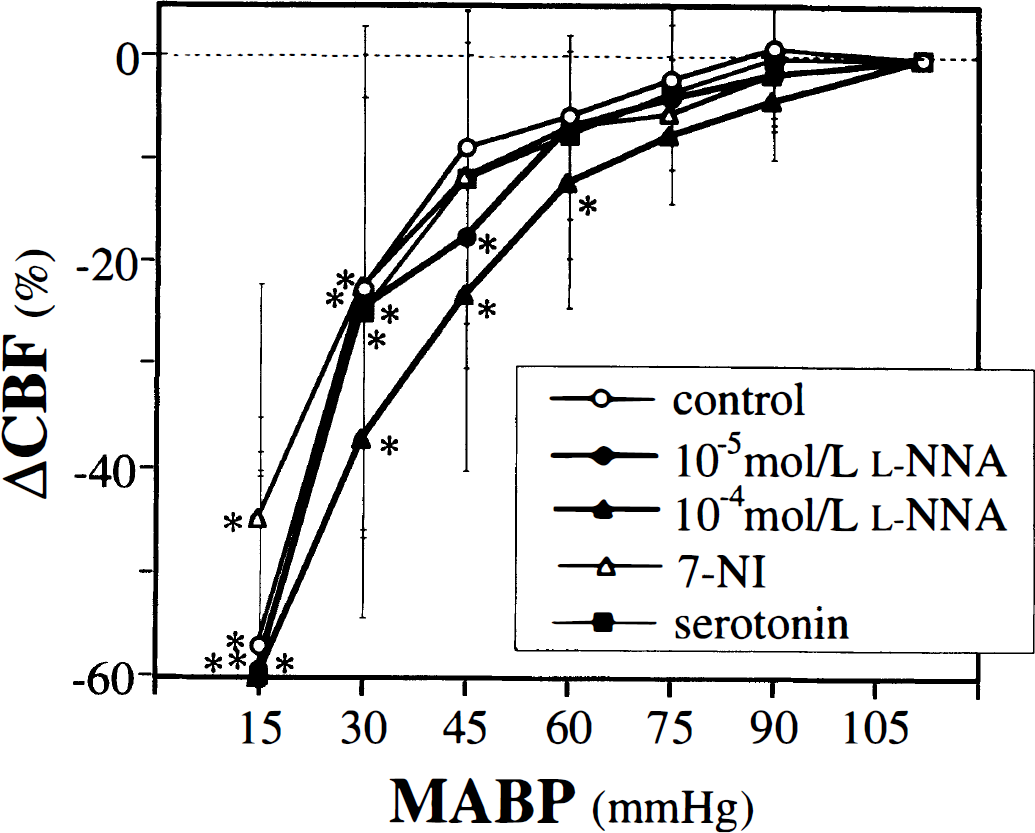
Changes in CBF in the five groups during stepwise hypotension. All values are means ± SD.
Effects of l-NNA and 7-NI on basal vessel diameter and on vascular responses to agonists
Baseline diameter of vessels did not differ among the five groups (Table 2). Topical application of
Vessel diameter and its dilator response to acetylcholine and nitroprusside
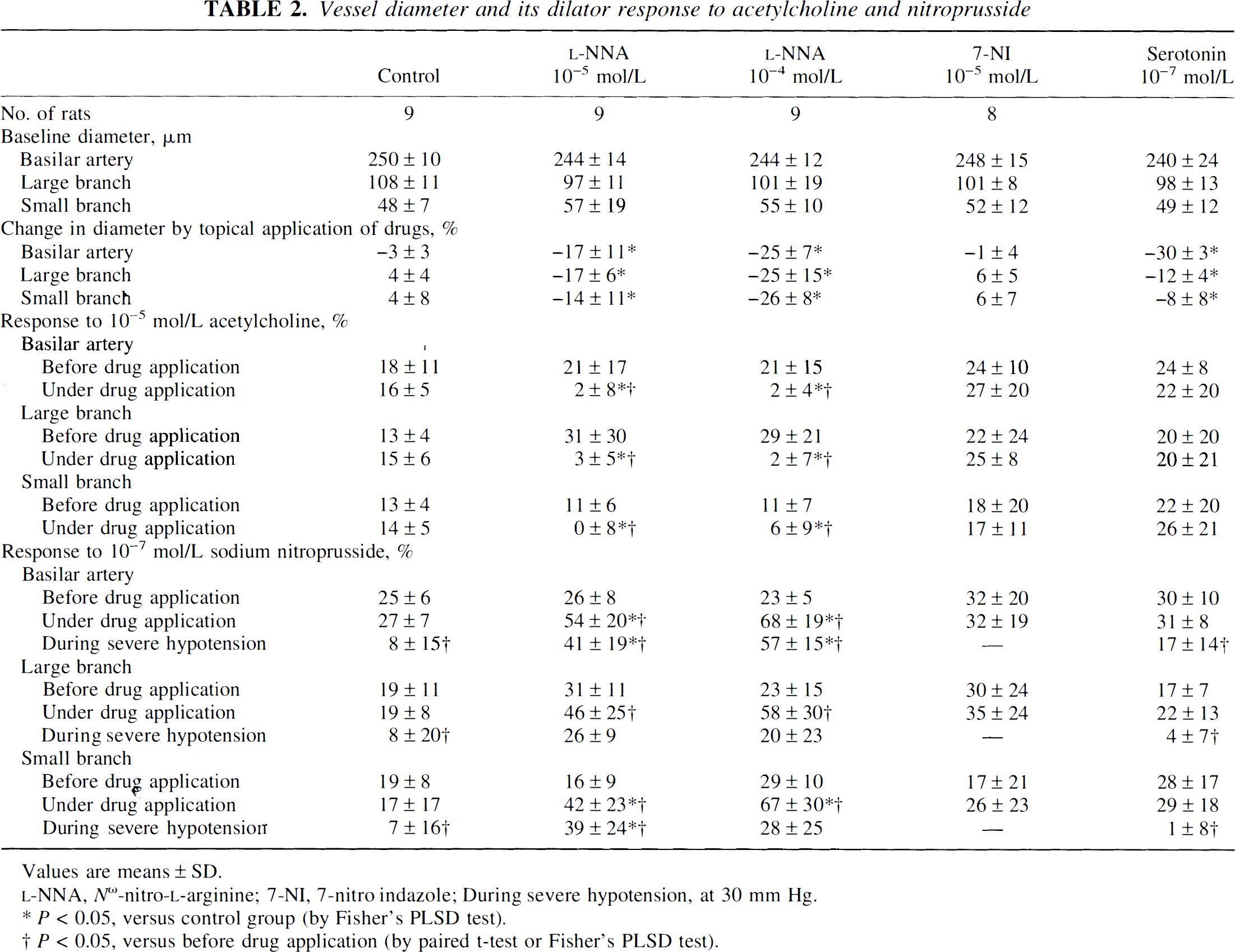
Values are means ± SD.
P < 0.05, versus control group (by Fisher's PLSD test).
P < 0.05, versus before drug application (by paired t-test or Fisher's PLSD test).
Effects of l-NNA and 7-NI on changes in vessel diameter during hypotension
In the control group, diameter of the basilar artery was essentially constant until MABP reached 60 mm Hg, and it dilated significantly at 45 mm Hg and below with maximum dilatation at 30 mm Hg (Fig. 2). In the
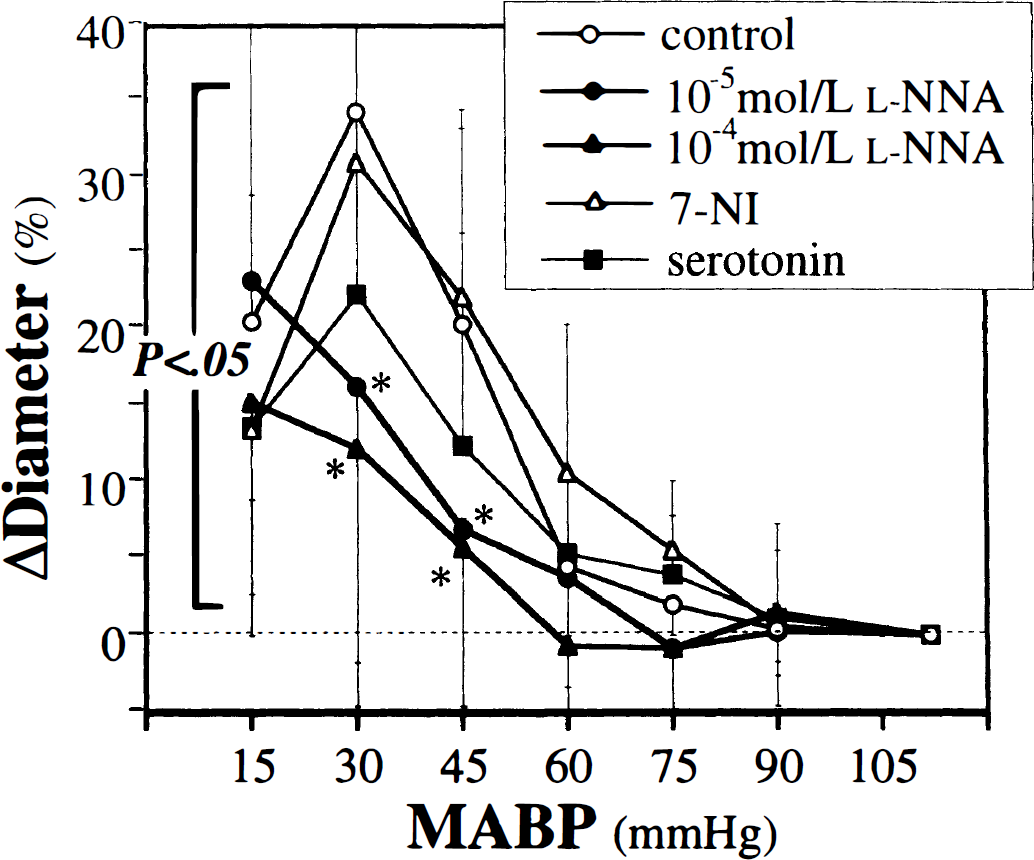
Changes in the diameter of the basilar artery in the five groups during stepwise hypotension. All values are means ± SD. MABP, mean arterial blood pressure;
The dilatation of the large branch reached the statistical significance at 45 mm Hg for the control and 10−4 mol/L
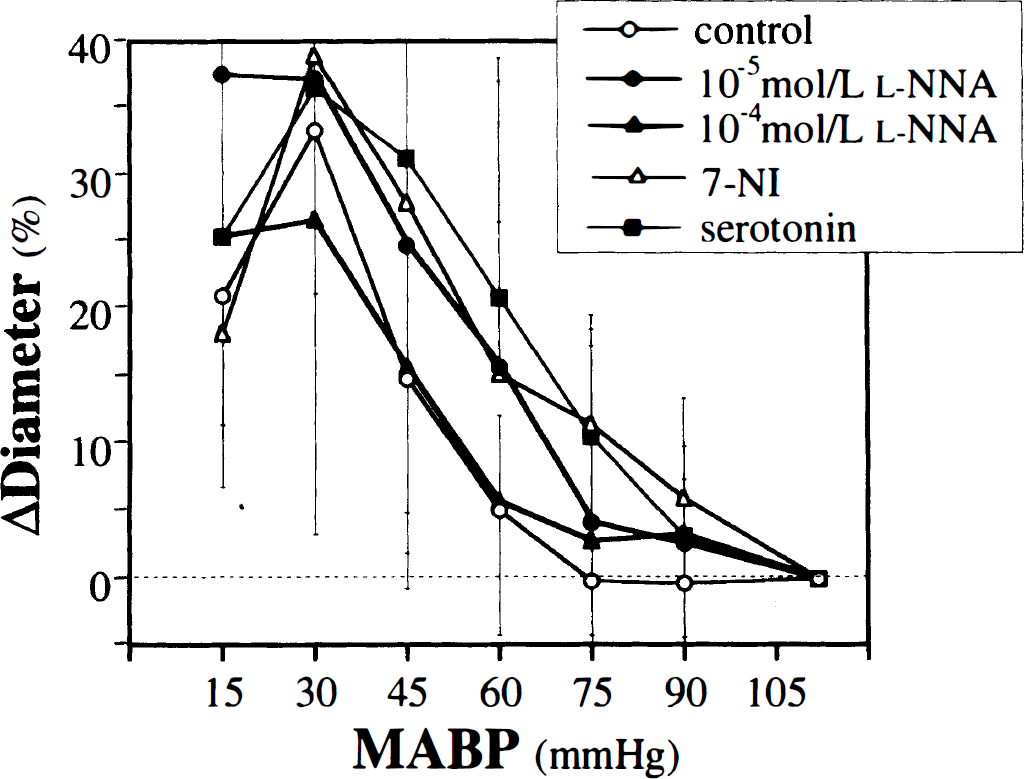
Changes in the diameter of the large branch of the basilar artery in the five groups during stepwise hypotension. All values are means ± SD.
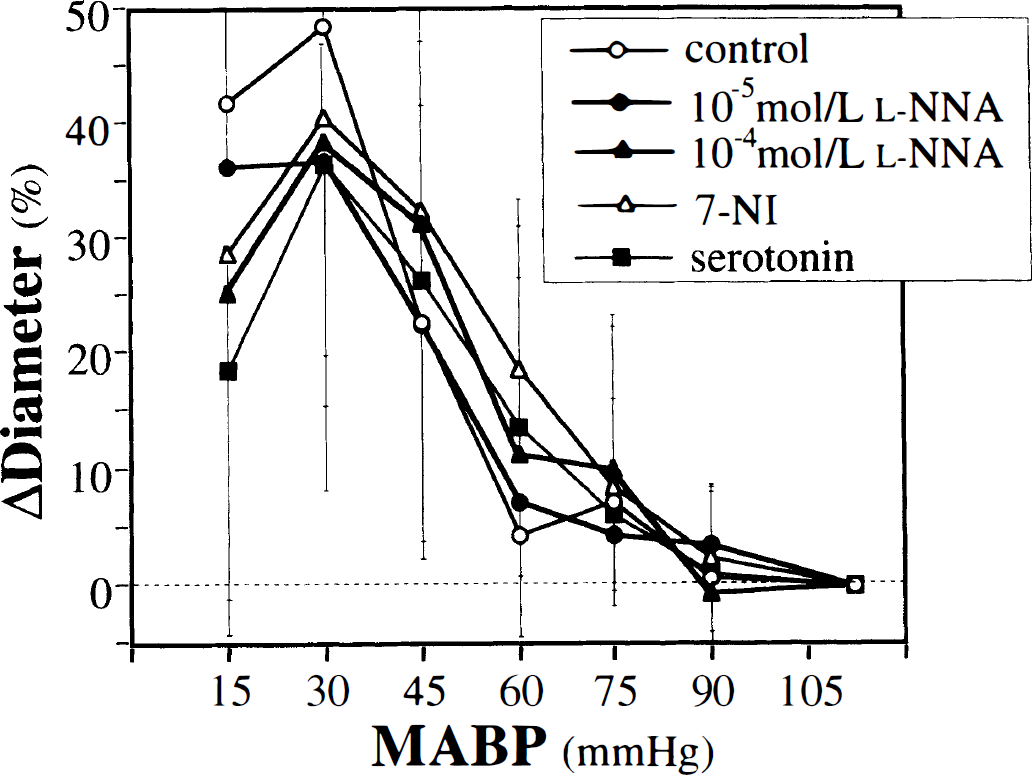
Changes in the diameter of the small branch of the basilar artery in the five groups during stepwise hypotension. All values are means ± SD.
DISCUSSION
There are three major findings in the present study. First, during inhibition of NOS by topical application of
Comparison with previous studies
The role of NO in CBF autoregulation is conflicting (Wang et al., 1992b; Buchanan and Phillis, 1993; Tanaka et al., 1993; Kelly et al., 1994; Kobari et al., 1994; Takahashi et al., 1995). Majority of previous studies showed that, although NOS inhibitors reduced the basal CBF, they did not affect CBF autoregulation during hypotension (Wang et al., 1992b; Buchanan and Phillis, 1993; Takahashi et al., 1995). In contrast, others showed that NO contributes to maintenance of CBF during mild hypotension of 30 to 40 mm Hg below the baseline blood pressure (Tanaka et al., 1993; Kobari et al., 1994). The present study agrees in part with the latter findings in that NOS inhibitors impair CBF autoregulation. However, our results are unique in that autoregulation was impaired principally during profound hypotension and not during mild hypotension. Recently, Huang et al. (1996) reported that endothelial NOS mutant mice showed significant decreases in parietal CBF measured by laser-Doppler flowmetry during severe hemorrhagic hypotension around 20 to 30 mm Hg compared with the wild-type strain, which strongly supports our findings that NO plays a major role in autoregulation under profound hypotensive condition. Discrepancies of the results between our studies and the previous ones may partly result from the differences of the sorts and routes of administration of NOS inhibitors, brain regions, and severity of hypotension. In some previous studies, forebrain CBF was usually measured under systemic application of N-monomethyl
We consider that the present methods have some advantages over previous ones. They are as follows: maintaining the resting systemic blood pressure by topical application of drugs; simultaneous observation of changes in CBF and diameters of different-sized vessels; and continuous observation of the changes throughout the pressure changes (Faraci et al., 1987; Fujii et al., 1991a; Toyoda et al., 1996).
Nitric oxide and the basilar artery
Nitric oxide contributes to the regulation of vascular tone of the large cerebral arteries under resting conditions and in response to diverse vasoactive stimuli (Faraci, 1990; Wang et al., 1992a; Buchanan and Phillis, 1993; Pelligrino et al., 1993). Regarding the basilar artery, topical application of NOS inhibitors produces concentration-dependent constriction under resting conditions and attenuates its dilator response to acetylcholine (Faraci, 1990). In the present study,
The present study showed a characteristic change in CBF;
Serotonin, another potent vasoconstrictor agent, tended to decrease the dilatation of the basilar artery during hypotension. However, this attenuation was smaller than that in
In this study, we did not clarify the mechanisms that activate NOS despite blood pressure changes. Hypoxia seems to be a dominant mechanism involved in hypotension-induced cerebral vasodilatation (Kontos et al., 1978b), and inhibition of NOS attenuates cerebral vasodilation (Pelligrino et al., 1995) and increases in CBF (Iwamoto et al., 1992) during severe hypoxia. Thus, local hypoxia may be an important trigger of activation of NOS under hypotensive conditions. It has been well known that increased shear stress and stretch forces exerted on endothelial cells by blood flow and pressure stimulate NO release (Cooke et al., 1990). Severe hemorrhagic hypotension decreases shear stress and stretch forces, and therefore may attenuate endothelial NO release in each group. If this were true, we would see the diminished effect of NOS inhibition during hypotension.
Nitric oxide and the small arteries
Responses to NOS inhibitors are dependent on the size of cerebral vessels, i.e. topical application of N-monomethyl
The present study showed that, while
Neuronal nitric oxide synthase for CBF autoregulation
Endothelial and neuronal NO may be involved in CBF autoregulation in a different manner. Huang et al. (1996) reported the impaired autoregulatory plateau of parietal CBF in endothelial NOS mutant mice, and suggested the contribution of endothelial NO to CBF autoregulation. In the contrast, some studies suggest that neuronal NO has a potential to regulate CBF (Kelly et al., 1995; Wang et al., 1995). Systemic injection of 7-NI caused significant reduction of both basal CBF and the response to hypercapnia, along with reduction of brain NOS activity and preservation of endothelial NOS-induced vasodilation.
In the present study, we topically applied a tenfold higher concentration of IC50 of 7-NI, which caused an approximate 80% reduction of rat cerebellar NOS activity in vitro (Babbedge et al., 1993). This inhibitor did not affect basal CBF, baseline diameter of any vessels, or vasodilator response to acetylcholine. During mild hypotension, autoregulatory plateau of CBF was preserved and dilator responses of each-sized vessels were somewhat augmented in 7-NI-treated rats. During severe hypotension around the lower limit of CBF autoregulation, 7-NI had no significant effects on reduction of CBF or dilatation of the vessels. These results do not support the positive contribution of neuronal NOS to the regulation of CBF during hypotension. Even the topical application of a higher concentration of 7-NI (10−4 mol/L) did not affect either basal CBF or CBF autoregulation during hypotension, accompanied by an increase in the basal vessel diameter (12% in the basilar artery) and exaggerated vasodilation during mild hypotension (14% at 60 mm Hg in the basilar artery) (unpublished observation). An in vitro study also demonstrated that 7-NI induced an unusual relaxation of the middle cerebral artery in dogs irrespective of presence of functional endothelium or pretreatment by nonselective NOS inhibitor, which suggested direct vasodilator actions of 7-NI (Medhurst et al., 1994). Thus, augmentation of the vasodilator responses during mild hypotension may partly support the effectiveness of 7-NI in this study.
The difference in the route of 7-NI administration may result in the disparate change in basal CBF between our study and previous ones. Our previous study using the same window technique showed that superfusion of vasopressin, having much higher molecular weight (F.W. 1084.2) than
Another mechanism for CBF autoregulation
We recently clarified using the same window technique that inhibition of opening of ATP-sensitive potassium channels by topical application of glibenclamide impaired the dilator response of rat brain stem arterioles with a baseline diameter <70 μm during hypotension and shifted the lower limit of CBF autoregulation upward (Toyoda et al., 1997). In contrast, glibenclamide did not attenuate the dilatation of the basilar artery during hypotension. Thus, different factors seem to contribute to the autoregulatory responses of brain stem vessels with different size, i.e., endothelial NO regulates the tone of large arteries and ATP-sensitive potassium channels regulate microcirculation. Kontos and Wei (1996) recently reported that
In conclusion, endothelial NO plays a significant role in the autoregulatory response of large arteries and thus maintaining CBF to the brain stem during severe hypotension around the lower limit of CBF autoregulation. However, NO does not seem to mediate the response during mild hypotension within the autoregulatory plateau of CBF.
