Abstract
The novel neuronal nitric oxide synthase inhibitors, 1-(2-trifluoromethylphenyl)imidazole (TRIM) and 7-nitro indazole (7-NI), were used to investigate the role of nitric oxide in a model of transient focal cerebral ischemia in vivo. In halothane-anesthetized rats, the middle cerebral artery (MCA) was occluded for 2 hours using an intravascular thread and then reperfused for 22 hours before histologic evaluation. TRIM (10, 20, or 50 mg/kg), 7-NI (60 mg/kg), TRIM (50 mg/kg) plus L-arginine (300 mg/kg), or L-arginine (300 mg/kg) alone was administered intraperitoneally, either at 5 or 90 minutes after MCA occlusion. Immediate administration (5 minutes after MCA occlusion) of TRIM produced a dose-related reduction in lesion size, which was reversed with L-arginine coadministration. Similarly, delayed administration of TRIM (90 minutes after MCA occlusion, 50 mg/kg) decreased total lesion volume by 48.4% ± 13.0% in comparison to a reduction of 39.3% ± 10.9% when TRIM (50 mg/kg) was administered immediately (5 minutes) after occlusion. 7-NI (60 mg/kg) reduced the total lesion volume by 38.5% ± 13.7% when administered immediately (5 minutes) after MCA occlusion, but had no effect when administration was delayed (90 minutes). Neither TRIM (50 mg/kg) nor 7-NI (60 mg/kg), administered 5 minutes after MCA occlusion, had any significant effect on mean arterial blood pressure throughout the ischemic period or for up to 10 minutes after reperfusion. These results indicate that immediate or delayed administration of the selective neuronal NOS inhibitor TRIM reduces the lesion volume after transient MCA occlusion. In contrast, only immediate administration of 7-NI reduces lesion volume.
Keywords
Nitric oxide plays multiple roles in the pathophysiology of focal cerebral ischemia (Dawson, 1994). Focal cerebral ischemia in rats causes an immediate increase in the production of nitric oxide in cerebral tissue (Malinski et al., 1993) and in the appearance of nitric oxide end products in plasma (Kumura et al., 1994). Experiments using conventional nitric oxide synthase (NOS) inhibitors or nitric oxide donors have provided evidence that nitric oxide can act both as a neuroprotective and neurotoxic agent in animal models of focal cerebral ischemia (for reviews see Iadecola et al., 1994; Dawson, 1994; Meldrum, 1996). For example, administration of nitric oxide donors (sodium nitroprusside, 3-morpholinosydnonimine) or
In contrast to these potential beneficial effects, nitric oxide may itself be a mediator of neuronal destruction. Several studies using nonselective NOS inhibitors have shown that NOS inhibition may protect against cerebral ischemic damage in vivo (Nowicki et al., 1991; Nagafuji et al., 1992; Buisson et al., 1993; Ashwal et al., 1993). In vitro, nitric oxide has been shown to exacerbate ischemic cell damage by generating peroxynitrite (Radi et al., 1991), inhibiting iron-containing enzymes (Stadler et al., 1991), and mediating glutamate neurotoxicity (Dawson et al., 1991).
However, there are conflicting reports showing that nonselective NOS inhibitors have a variety of effects on lesion size after cerebral ischemia. Nitric oxide synthase inhibitors have been shown to reduce, increase, or have no effect on focal ischemic lesions depending on (1) the animal model, (2) the dosing regimens (time, dose, and route), and (3) the vasopressor action of the specific nonselective NOS inhibitor used (Dawson, 1994; Iadecola et al., 1994; Dalkara and Moskowitz, 1994). The development of knockout mice deficient in the different isoforms of NOS (neuronal, endothelial, and inducible) has helped to clarify the role of nitric oxide in cerebral ischemia. Knockout mice with selective endothelial NOS deletion develop a larger infarct after MCA occlusion in comparison to wild-type controls (Huang et al., 1996). In contrast, neuronal NOS knockout mice demonstrate a reduction in the size of the infarct in comparison to wild-type controls after both permanent (Huang et al., 1994) and transient MCA occlusion (Hara et al., 1996). Therefore, neuronal NOS may have a harmful role to play in ischemia but endothelial NOS may be protective.
Novel nitric oxide synthase inhibitors (e.g., 7-nitro indazol [7-NI], 1-(2-trifluoromethylphenyl)imidazole [TRIM], and ARL 17477) that show a greater selectivity for certain NOS isoforms have recently been developed. 7-Nitro indazole is an inhibitor of the neuronal NOS in vitro and exhibits antinociceptive effects in vivo (Moore et al., 1993a,1993b). Although 7-NI also inhibits endothelial NOS in vitro, it is devoid of the vasopressor activity associated with existing nonselective NOS inhibitors such as L-NG-nitroarginine methyl ester (Moore et al., 1993a, 1993b; Babbedge et al., 1993; Rees et al., 1991). Administration of 7-NI produced a 25% reduction in lesion volume in permanent MCA occlusion in rats, which was reversed by
In this study we have used TRIM and 7-NI to further investigate the role of nitric oxide in a reperfusion model of MCA occlusion in the rat. These agents are administered after ischemia, which may be more relevant to the clinical situation. Our data indicate that TRIM and 7-NI are cerebroprotective when administered immediately after MCA occlusion; similarly, TRIM may also be effective when given 90 minutes after MCA occlusion.
MATERIALS AND METHODS
All experiments were carried out under U.K. Home Office regulations. Male Sprague-Dawley rats (300 to 350 g, Charles River, U.K.) were fasted overnight before surgery but allowed free access to water. The MCA was occluded for 2 hours using a method of intravascular occlusion (Koizumi et al., 1986; Zea Longa et al., 1989). For occlusion of the MCA, an occluder thread was prepared. The tip of a 3-0 nylon Ethilon suture (Ethicon Ltd., U.K.) was rounded with sandpaper (P1000) and coated with a silicone compound to a diameter of 0.3 mm and a length of 3 mm. Anesthesia was induced with 3% halothane in oxygen and maintained with 1.75% to 2% halothane in oxygen via a face mask in spontaneously breathing rats. The rectal body temperature was continuously monitored and maintained at 37.5°C throughout the surgery using a rectal probe and automated heat pad (ʻk-tempʼ unit, International Market supplies, U.K.). After a ventral midline incision in the neck, the right common carotid artery (CCA) was isolated and a cannula was inserted to collect blood samples for blood gas analysis immediately before occlusion (Table 1). The right CCA was temporarily clamped, using a microsurgical clip, proximal to the bifurcation of the internal carotid artery and external carotid artery and a small incision was made in the CCA. The occluder thread was inserted, secured and then advanced through the CCA into the internal carotid artery to a distance of 18 to 20 mm (depending on body weight) from the internal carotid artery and external carotid artery bifurcation, until the suture blocked the origin of the MCA. The incision was closed and the rat was allowed to recover. On average the surgery was performed within 25 minutes. After 2 hours of ischemia the intraluminal thread was removed under brief anesthesia (2% halothane in oxygen), to allow reperfusion of the ischemic tissue. A small increase in rectal temperature was observed at this time point but there was no significant difference between treatment groups (Table 1). Because the right CCA was ligated in this model, the ischemic tissue was reperfused via the circle of Willis through the contralateral carotid artery, basilar artery, and collateral circulation of the cerebral arteries.
Physiologic variables
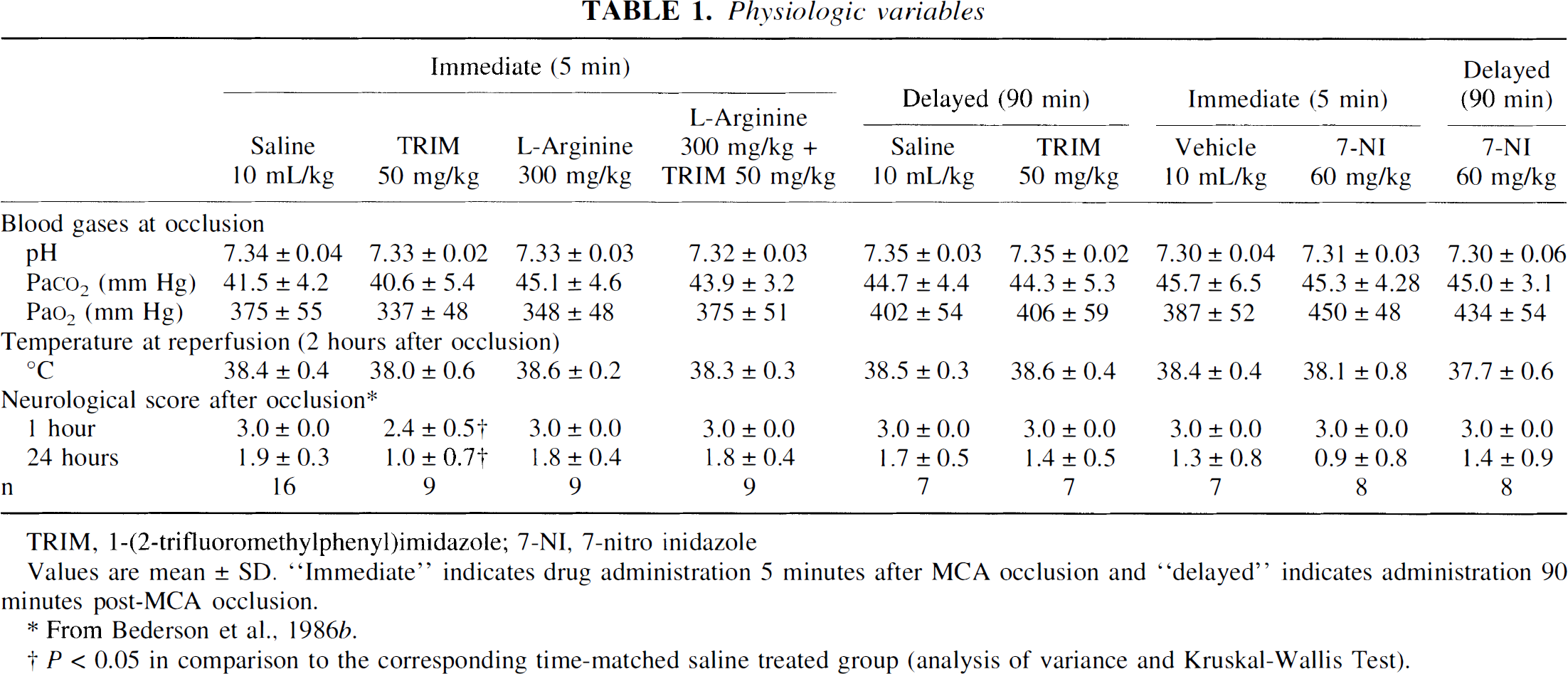
TRIM, 1-(2-trifluoromethylphenyl)imidazole; 7-NI, 7-nitro inidazole
Values are mean ± SD. “Immediate” indicates drug administration 5 minutes after MCA occlusion and “delayed” indicates administration 90 minutes post-MCA occlusion.
From Bederson et al., 1986b.
P < 0.05 in comparison to the corresponding time-matched saline treated group (analysis of variance and Kruskal-Wallis Test).
After MCA occlusion, neurologic examination was performed in the conscious rat at 1, 4, and 24 hours after MCA occlusion, using a 4-point scoring system (Bederson et al., 1986b). The neurologic deficit was assessed as follows: grade 1, forelimb flexion; grade 2, decreased resistance to lateral push; or grade 3, gait disturbances leading to circling or walking to the left; grade 4, no neurologic deficit (Bederson et al., 1986b). Successful occlusion was demonstrated by an animal with a grade 3 deficit. All vehicle- or saline-treated rats exhibited left circling 1 hour (grade 3) after MCA occlusion (Table 1).
Blood pressure measurements
In a separate group of animals the effect of saline, TRIM, or 7-NI on arterial blood pressure was assessed. Surgery was performed as described above for MCA occlusion, and blood pressure was measured via the right carotid artery using a MacLab/8s (ADInstruments Ltd., U.K.) and recorded by an Apple Macintosh computer (Powerbook 520c, Apple Ltd., U.K.). Blood pressure was monitored during ischemia and reperfusion in the anesthetized rat (maintenance, 1% to 1.5% halothane). This group of animals were not allowed to recover from the anesthesia at the end of this experiment.
Determination of lesion size
Neuropathologic evaluation was performed at 24 hours after the induction of ischemia. The animals were anesthetized using 3% halothane and decapitated. The brains were removed and examined to confirm the position of the intravascular thread and the absence of subarachnoid hemorrhage or arterial penetration with the thread. Five of 97 animals were excluded from the study because they demonstrated signs of subarachnoid hemorrhage after brain removal. The brains were sectioned coronally with a Jacobowitz rat brain slicer (Zivic-Miller Lab, Inc., Allison Park, PA U.S.A.) at 2-mm intervals and incubated for 30 minutes in 2% 2,3,5-triphenyltetrazolium chloride (TTC) at 37°C for vital staining (Bederson et al., 1986a). Then the sections were fixed by immersion in 10% formalin solution. The brain sections were scanned using a conventional, high-resolution flatbed scanner system (Agfa Ltd., U.K.) and lesion size was determined using an in-house semiautomated image analysis system (Dispim software). Areas not stained red with TTC were considered lesioned. The total and lesion size (mm2) was obtained for each coronal section. Lesion volume (mm3) for each section was obtained by multiplying the lesion size by the slice thickness (2 mm). Total brain lesion volume was calculated as the sum of each slice volume.
Drug treatment
Animals were randomly assigned to drug or vehicle-treated groups. In group 1, TRIM (10, 20, or 50 mg/kg),
Materials and solutions
1-(2-Trifluoromethylphenyl)imidazole was obtained from Lancaster Synthesis Ltd., U.K. and dissolved in warm (37°C) physiologic saline (0.9%) at a concentration of 5 mg/mL. 7-Nitro indazole (Lancaster Synthesis Ltd.) was suspended in 0.25% wt/vol methylcellulose (400 cp, Sigma Ltd., U.K.) in HPLC-grade distilled water at 37°C, using sonication, at a concentration of 5 mg/mL. 2,3,5-Triphenyltetrazolium chloride was obtained from Sigma.
Statistical analysis
Data are presented as mean ± SD and were evaluated by analysis of variance and Tukey's test. Neurologic data were analyzed using Kruskal-Wallis followed by Dunn's procedure. Statistical significance was accepted at the P < 0.05 level.
RESULTS
Immediate or delayed NOS inhibition on lesion size
Immediate administration of TRIM (5 minutes after MCA occlusion) produced a dose-related inhibition of ischemic lesion size when measured at 24 hours after MCA occlusion (Fig. 1). The highest dose of TRIM (50 mg/kg) significantly reduced total lesion volume by 39.3% ± 10.9% in comparison to saline-treated animals (Table 2). Similarly, delayed administration of TRIM (50 mg/kg, 90 minutes) significantly reduced the total lesion size by 48.4% ± 13.0%. There was no significant difference in the reduction in lesion size between immediate or delayed TRIM-treated (50 mg/kg) groups (Table 2). Also, immediate (5 minutes) administration of TRIM (50 mg/kg) produced a significant (P < 0.05) improvement in the neurologic score at 1 and 24 hours after MCA occlusion, in comparison to saline-treated animals (Table 1). The neurologic score (1 or 24 hours after MCA occlusion) after delayed administration of TRIM was not significantly (P > 0.05) different from the appropriate saline-treated group.
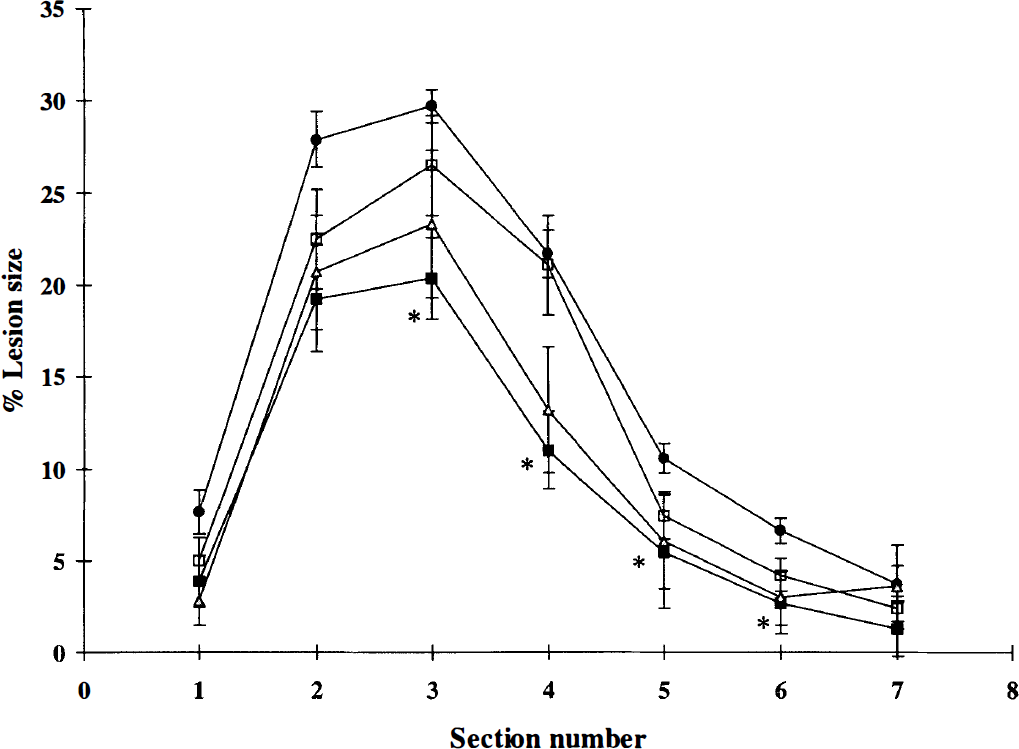
Dose-response curve for 1-(2-trifluoromethylphenyl)imidazole- (TRIM-) versus saline-treated rats. Graph shows the percent lesion size for each coronal section (2 mm) from anterior to posterior. Values are means and SD. Saline (•, 10 mL/kg, n = 14) or TRIM at 10 mg/kg (□, n = 5), 20 mg/kg (△, n = 7), or 50 mg/kg (▪, n = 9) was administered 5 minutes after MCA occlusion. Administration of TRIM (50 mg/kg) significantly reduced the lesion size (*P < 0.05) as compared with the saline-treated group (analysis of variance and Tukey's test).
Effect of NOS inhibitors on lesion volume (mm3) resulting from transient MCA occlusion
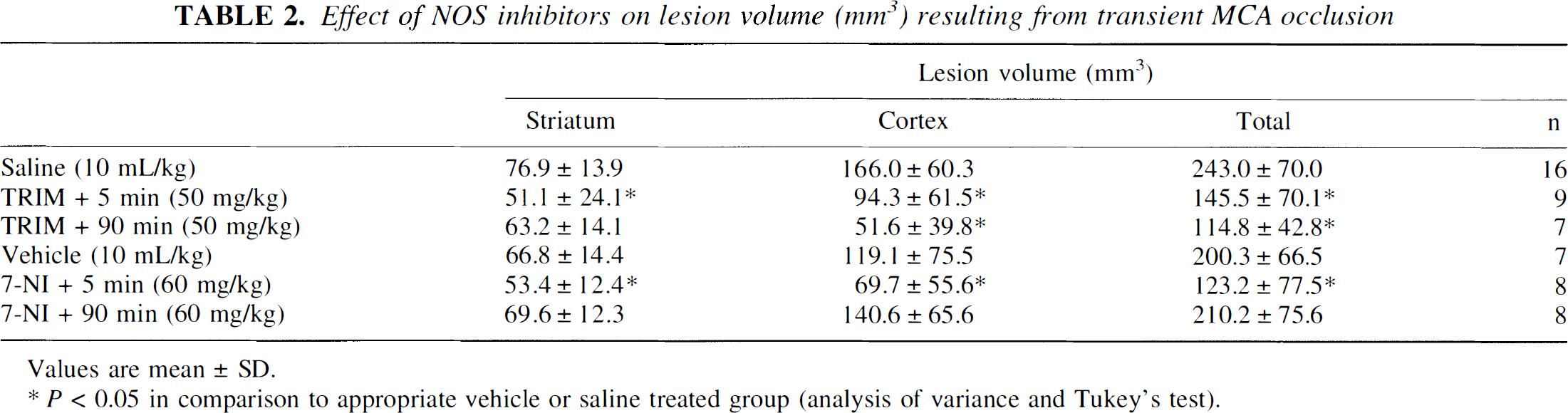
Values are mean ± SD.
P < 0.05 in comparison to appropriate vehicle or saline treated group (analysis of variance and Tukey's test).
Administration of 7-NI (60 mg/kg, intraperitoneally) produced a significant 38.5% ± 13.7% reduction in total lesion volume when given immediately (5 minutes) after MCA occlusion, but no significant (P > 0.05) cerebroprotection was observed on delayed administration (90 minutes) (Table 2 and Fig. 2). Neither immediate nor delayed administration of 7-NI had any significant (P > 0.05) effect on neurologic score in comparison to vehicle-treated group (Table 1).
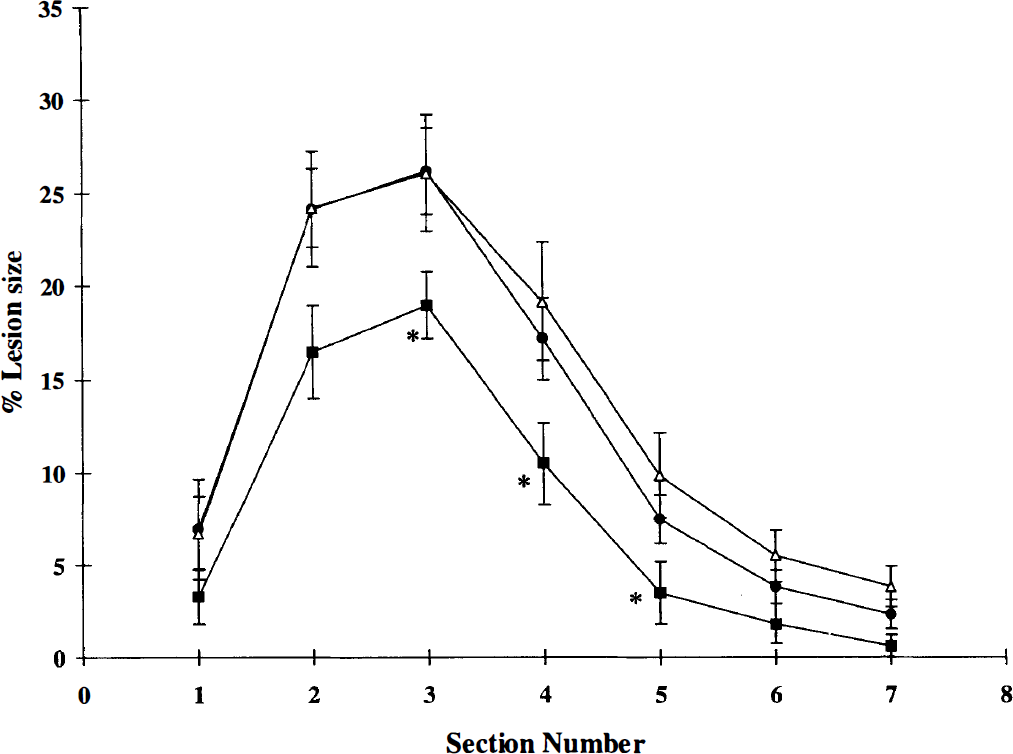
Effect of 7-nitro indazole (7-NI) on lesion size. Graph shows percent lesion size for each coronal section (2 mm) from anterior to posterior. Values are means and SD. Vehicle (•, 10 mL/kg, n = 7) or 7-NI (60 mg/kg) were administered at 5 (▪, n = 8) or 90 (△, n = 8) minutes after MCA occlusion. Immediate (5 minutes) administration of 7-NI significantly (*P < 0.05) reduced lesion size as compared with the vehicle-treated group (analysis of variance and Tukey's test).
Striatal and cortical lesion volumes were analyzed to investigate further the area of the ischemic lesion that was affected by NOS inhibition. Immediate (5 minutes) administration of either TRIM or 7-NI reduced the lesion volume in both the striatum and the cortex (Table 2). The reduction in the size of the cortical lesion was, however, more pronounced in comparison to the striatal lesion. Delayed administration of TRIM, but not 7-NI, also decreased the size of the striatal and cortical lesion (Table 2).
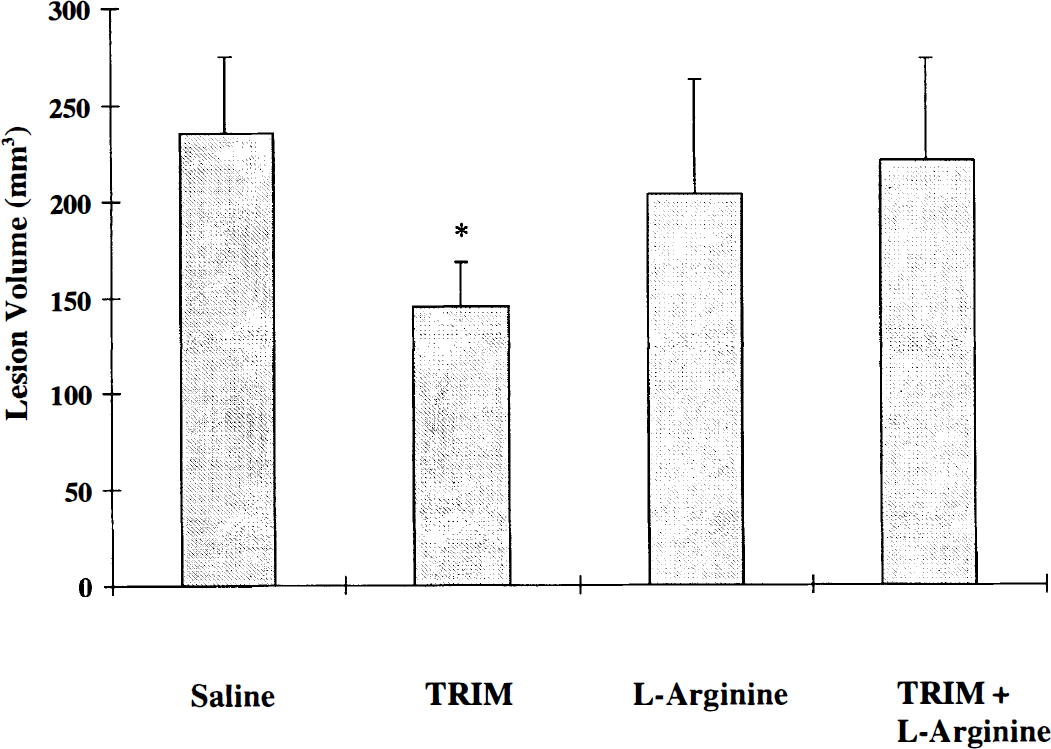
l-Arginine reversal of TRIM cerebroprotection
Immediate administration (5 minutes) of
Effect of NOS inhibition on blood pressure
Middle cerebral artery occlusion caused a transient reduction in mean arterial blood pressure, which returned to baseline levels before intraperitoneal injections at 5 minutes after MCA occlusion. Neither saline, TRIM, nor 7-NI had any significant effect on blood pressure during either the ischemic or reperfusion periods after MCA occlusion (Table 3). These results are in keeping with previous studies in normal animals, which have shown no significant (P > 0.05) change in blood pressure on administration of TRIM or 7-NI (Handy et al., 1995; Moore et al., 1993a).
Effect of saline, TRIM, or 7-NI on mean arterial blood pressure after transient MCA occlusion in the anesthetized rat
Values are mean ± SD; P > 0.05 (analysis of variance and Tukey's test). Arterial pressure values (mm Hg) are given as follows: MCA occlusion was performed at t = 0 minutes, test agents were injection at t = 5 minutes, and reperfusion was performed at t = 120 minutes.
DISCUSSION
This study demonstrates that inhibition of NOS with either TRIM or 7-NI reduces the size of the ischemic lesion measured 24 hours after transient focal cerebral ischemia. Immediate administration of TRIM (5 minutes after occlusion) produced a dose-related inhibition of the lesion size after reversible MCA occlusion. Delayed administration (90 minutes after occlusion, i.e., 30 minutes before reperfusion) of the highest dose of TRIM (50 mg/kg) also reduced the size of the ischemic lesion. The observation that TRIM is protective even after delayed treatment is similar to a previous study using the novel selective neuronal NOS inhibitor, ARL 17477, which decreased infarct volume after transient MCA occlusion in rats when administered at the time of reperfusion (120 minutes after occlusion; Zhang et al., 1996b). These results show that there is a modest therapeutic time window for NOS inhibition in reversible focal cerebral ischemia and confirm previous findings that suggest a detrimental role for neuronally synthesized nitric oxide in the acute stages of focal cerebral ischemia. Immediate administration of TRIM also partially reduces the deficits in motor function observed 1 and 24 hours after transient MCA occlusion, which is described as a decrease in the neurologic score in comparison to saline-treated groups. These results provide evidence that TRIM not only protects against the physical ischemic damage, indicated by TTC staining, but also reduces the deficit in neurologic function observed after transient focal cerebral ischemia. More recently, our group has confirmed the cerebroprotective effect of immediate administration of TRIM using magnetic resonance imaging (Haga et al., 1997).
Immediate administration of 7-NI (5 minutes after MCA occlusion) decreased the total lesion volume at a dose that does not alter blood pressure and that has been shown previously to reduce neuronal NOS activity by 50% in the cerebral cortex (Yoshida et al., 1994). The cerebroprotective effect of 7-NI is similar to a previous report in which 7-NI (50 mg/kg) was shown to inhibit lesion volume by 25% after permanent MCA occlusion, an effect that was reversed by
Interestingly,
It has been suggested that the mechanism of action of both TRIM and 7-NI is through the inhibition of NOS, preventing the conversion of
Slight hyperthermia, which could contribute to the development of the ischemic lesion, was observed at the time of reperfusion, and previously a reduction in body temperature has been shown to be neuroprotective (Chen et al., 1992). However, TRIM and 7-NI are unlikely to be cerebroprotective as a result of a reduction in body temperature because at the time of reperfusion there was no temperature difference between control and drug-treated groups. Similarly, 24 hours after MCA occlusion no difference in body temperature was observed (unpublished communication). Also, neither agent had any effect on systemic blood pressure on intraperitoneal administration. Therefore, we can conclude that there should not be any major change in body temperature because of any potential hemodynamic effects of TRIM or 7-NI.
Although both TRIM and 7-NI have been shown to be relatively selective inhibitors of neuronal NOS, a possible effect on inducible NOS should be considered (Moore et al., 1993a, 1993b; Babbedge et al., 1993; Handy et al., 1995). Expression of brain inducible NOS mRNA, however, begins at 12 hours and peaks 48 hours after MCA occlusion, and inducible NOS catalytic activity is observed only 48 hours after ischemia (Iadecola et al., 1995b). Therefore, it is unlikely that TRIM or 7-NI alters inducible NOS activity, because the protocol used in our study measured lesion size only 24 hours after occlusion. Also, both agents were administered in the acute stages (<2 hours) of ischemic cell damage, before expression of the inducible NOS protein.
In conclusion, inhibition of NOS with TRIM or 7-NI elicited protection against ischemic cell damage after transient focal cerebral ischemia in rats. These results confirm previous observations that neuronally derived nitric oxide is detrimental in the early stages of transient focal cerebral ischemia in vivo.
Footnotes
Abbreviations used
Acknowledgment
The authors thank Dave Plummer (UCH, London) for access to the suite of image display (Dispim) software.
