Abstract
The participation of nitric oxide and vasoactive intestinal peptide (VIP) in the neurogenic regulation of bovine cerebral arteries was investigated. Nitrergic nerve fibers and ganglion-like groups of neurons were revealed by NADPH-diaphorase staining in the adventitial layer of bovine cerebral arteries. NADPH diaphorase also was present in endothelial cells but not in the smooth muscle layer. Double immunolabeling for neuronal nitric oxide synthase and VIP indicated that both molecules co-localized in the same nerve fibers in these vessels. Transmural nerve stimulation (200 mA, 0.2 milliseconds, 1 to 8 Hz) of endothelium-denuded bovine cerebral artery rings precontracted with prostaglandin F2α, produced tetrodotoxin-sensitive relaxations that were completely suppressed by NG-nitro-
Keywords
Immunohistochemical and ultrastructural studies show that cerebral arteries are surrounded by a dense network of perivascular nerves, part of which contains vasodilator neuropeptides such as vasoactive intestinal peptide (VIP) (Larsson et al., 1976; Suzuki et al., 1988), substance P (Uddman et al., 1981; Suzuki et al., 1989), or calcitonin gene-related peptide (Suzuki et al., 1989). In addition, functional studies reveal that cerebral arteries relax when perivascular nerves are electrically stimulated. This effect occurs in the presence of adrenergic and cholinergic receptor blockade (Lee et al., 1978; Toda, 1981) and it is therefore considered to be a nonadrenergic noncholinergic (NANC) neurogenic relaxation. It has been recently reported that the neurogenic relaxation of cerebral arteries from several species is substantially reduced when nitric oxide (NO) synthesis is prevented by
Although NO currently is considered as a peripheral neurotransmitter involved in NANC relaxation in many organs (Ny et al., 1995; Kim et al., 1995; Takahashi et al., 1995; Takahashi and Owyang, 1995), a controversy exists as to whether neuropeptides, such as VIP or calcitonin gene-related peptide, also contribute to the neurogenic relaxation, based on the existence of a relaxation component resistant to NOS inactivation in some tissues (Kim et al., 1995; Ny et al., 1995; Takahashi et al., 1995; Takahashi and Owyang, 1995). In fact, VIP, which colocalizes with NOS in peripheral nerves from different territories (Ekblad et al., 1994; Hayashida et al., 1996; Takahashi et al., 1995; Klimaschewski et al., 1994; Barroso et al., 1996), has been shown to contribute to NANC relaxation in some tissues, such as the rabbit corpus cavernosum (Kim et al., 1995), the cat lung (Takahashi et al., 1995), or the rat stomach (Takahashi and Owyang, 1995), but not in others, such as the cat esophagus (Ny et al., 1995) or the human trachea (Ward et al., 1995). In cerebral blood vessels, VIP has been proposed as a functional dilator neurotransmitter (Lee et al., 1984; Bevan et al., 1986; Gaw et al., 1991), but conclusive experimental evidence has never been provided.
Nitric oxide and VIP exert their relaxant effect on smooth muscle cells through two separate signal transduction mechanisms. Activation of guanylyl cyclase by NO results in the accumulation of cyclic GMP (cGMP) (Schmidt et al., 1993), whereas the interaction of VIP with specific membrane receptors leads to increases in cyclic AMP (cAMP) concentration on activation of adenylyl cyclase (Huang and Rorstad, 1984; Suzuki et al., 1985).
In the current report, we analyze the participation of these two pathways in the relaxation produced by stimulation of perivascular nerves in bovine cerebral arteries and their possible interaction. We show that neuronal NO fully accounts for the neurogenic relaxation and that VIP, which is present in the same nerve terminals, acts as a neuromodulator by regulating NO synthesis in the nitrergic nerves.
METHODS
Bovine brains were obtained from a local slaughterhouse and transported to the laboratory in phosphate-buffered saline solution (PBS) at 4°C. A first-order branch of the anterior cerebral artery was isolated for functional experiments and for biochemical determinations. The endothelium was removed by gently introducing a cotton thread through the vascular lumen, taking care to preserve the adventitial layer.
Isometric tension recordings
Arterial rings, 4 mm in length, were suspended on two intraluminal parallel wires, introduced in an organ bath containing a physiologic solution (in mmol/L: 115 NaCl, 4.6 KCl, 1.25 CaCl2, 25 NaHCO3, 1.2 KH2PO4, 1.2 MgSO4, 0.01 EDTA, and 11 glucose), and connected to a Piodem strain gauge for isometric tension recording. All rings were equilibrated at a passive tension of 1 g for 90 minutes. Before starting the experiments, rings were contracted with prostaglandin F2α and acetylcholine (1 to 10 μmol/L) was added to confirm functionally the absence of endothelium. Only arteries that did not dilate in the presence of acetylcholine were used. Sodium nitroprusside (10 μmol/L) was added at the end of each experiment, and the relaxation obtained was considered to be 100%. In experiments in which guanylyl cyclase was inhibited, the maximal relaxation was obtained with papaverine (100 μmol/L) in both control and treated vessels.
Transmural nerve stimulation (TNS; 200 mA, 0.2 milliseconds, 1 to 8 Hz) was applied to precontracted arterial rings using two parallel platinum electrodes, one at each side of the vessel, connected to a CS-20 stimulator (Cibertec, Madrid, Spain). The neurogenic nature of the stimuli was demonstrated, since TNS responses were blocked by tetrodotoxin (1 to 10 μmol/L). The interval between consecutive stimuli was at least 8 to 10 minutes. TNS always was performed in the presence of 5 μmol/L indomethacin, 1 μmol/L phentolamine, 1 μmol/L propranolol, and 0.1 μmol/L atropine to prevent any effect mediated by prostaglandins, noradrenaline, or acetylcholine. Two or three reproducible responses were obtained before starting the experiments. NG-nitro-
The VIP concentration curves were performed in a cumulative manner in precontracted arteries. Only one curve was obtained for each vascular ring to avoid tachyphylaxis. Experiments in the presence and absence of
Cyclic GMP and cyclic AMP measurements
Arterial rings were mounted in the organ baths as for the functional experiments. The absence of relaxing responses to acetylcholine and the presence of TNS-induced relaxations were assessed in each segment. When
Histochemistry
The entire circle of Willis and its major branches were dissected out of the brain in the slaughterhouse, rinsed and perfused with PBS, and immediately introduced in fixative. The arteries used for NADPH-diaphorase staining were fixed for 2 hours in 4% paraformaldehyde in 0.1 mol/L phosphate buffer, pH 7.4, at 4°C, and then washed with phosphate buffer and crioprotected by incubation for 1 hour with increasing concentrations of sucrose (5% to 20%, weight/volume) and overnight with 30% (weight/volume) sucrose. Longitudinal sections (40 μm) were obtained with a freezing microtome, mounted on slides, and incubated for 70 minutes at 37°C in a mixture containing 1 mmol/L NADPH, 1 mmol/L nitro blue tetrazolium, and 0.3% Triton-X-100, in 0.1 mol/L phosphate buffer, pH 8.0. The sections were rinsed in phosphate buffer, dehydrated, and coverslips were fixed with Permount.
The vessels used for immunohistochemical study were fixed for 16 hours at 4°C in Zamboni's fixative (Steffanini et al., 1967) and washed in PBS containing 15% (weight/volume) sucrose and 0.01% (weight/volume) sodium azide. After pretreatment with 0.2% (volume/volume) Triton X-100 in PBS for 2 hours, and with Pontamine Sky blue (Cowen et al., 1985) for 30 minutes, the arterial segments were incubated overnight at room temperature with a rabbit antibody against NOS (Hammersmith Hospital/Wellcome, 1:400) and a guinea-pig antibody against VIP (Euro-Diagnostica, 1:300) diluted in PBS. Arteries then were washed in PBS and incubated with a mixture of tetramethylrhodamine isothiocyanate–conjugated mouse antiserum against rabbit IgG (Sigma, 1:100) and fluorescein isothiocyanate–conjugated mouse antiserum against guinea-pig IgG (Sigma, 1:10) for 1 hour at room temperature. The arteries were mounted and examined under a microscope equipped for epiillumination with filters selective for fluorescein and rhodamine fluorescence. In control experiments, no immunostaining was observed when primary antisera were omitted, replaced with nonimmune serum, or preadsorbed with their corresponding antigens (0.1 to 1 μmol/L) for 24 hours at 4°C. Labeled secondary antisera did not exhibit cross-reactivity with IgG from inappropriate species.
Statistical analysis
Data are expressed as arithmetic means plus/minus standard deviation, except for EC50 values, which are presented as geometric means with confidence limits. Statistical comparisons between groups were made using Student's t test for paired or unpaired samples, except for EC50 values, which were analyzed by the nonparametric Wilcoxon test. P < 0.05 was considered to be statistically significant.
Chemicals
The following compounds were used: prostaglandin F2α, acetylcholine chloride, sodium nitroprusside, tetrodotoxin, indomethacin, phentolamine hydrocloride,
RESULTS
Histochemical staining for NADPH-diaphorase activity in longitudinal sections of bovine pial arteries revealed the presence of large bundles of positive-staining fibers running longitudinally in the adventitial layer (Fig. 1A). These bundles branched off in different directions, leading to single varicose nerve fibers (Fig. 1B). In some areas, heavily stained neurons presenting clear nuclei and grouped in a ganglion-like manner were observed (Figs. 1C and 1D). NADPH-diaphorase staining also was present in endothelial cells but not in the smooth muscle layer (Fig. 1C). Double immunolabeling for neuronal NOS and VIP in whole-mounted arterial segments showed a co-localization of both markers in the same perivascular nerve fibers (Figs. 1E and 1F).
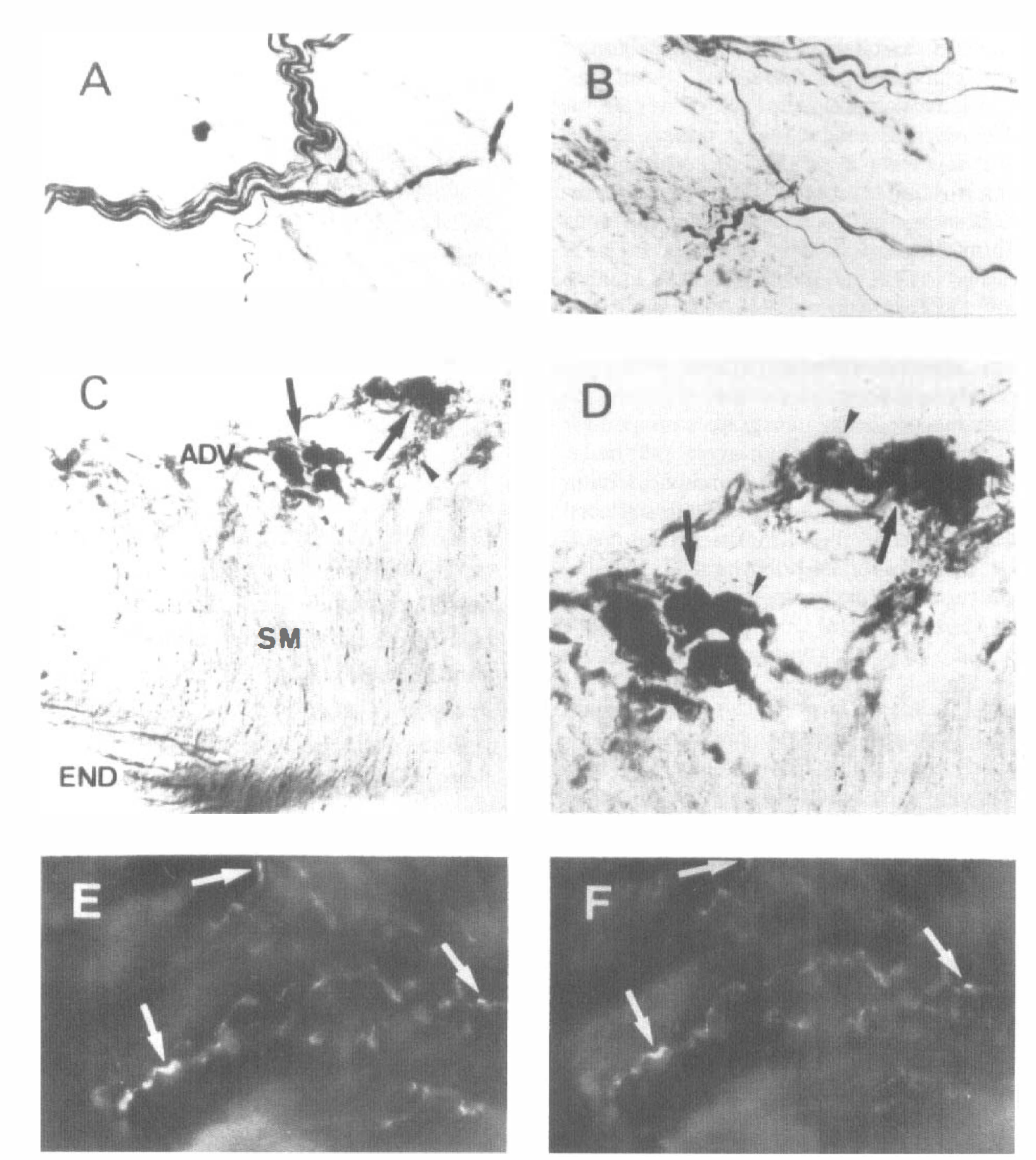
(
To determine the contribution of NO and VIP to the relaxation induced by depolarization of perivascular nerves, TNS was applied to endothelium-denuded precontracted arterial rings before and after blockade of NOS, guanylyl cyclase, or adenylyl cyclase. TNS induced a transient relaxation that was abolished by the NOS inhibitor
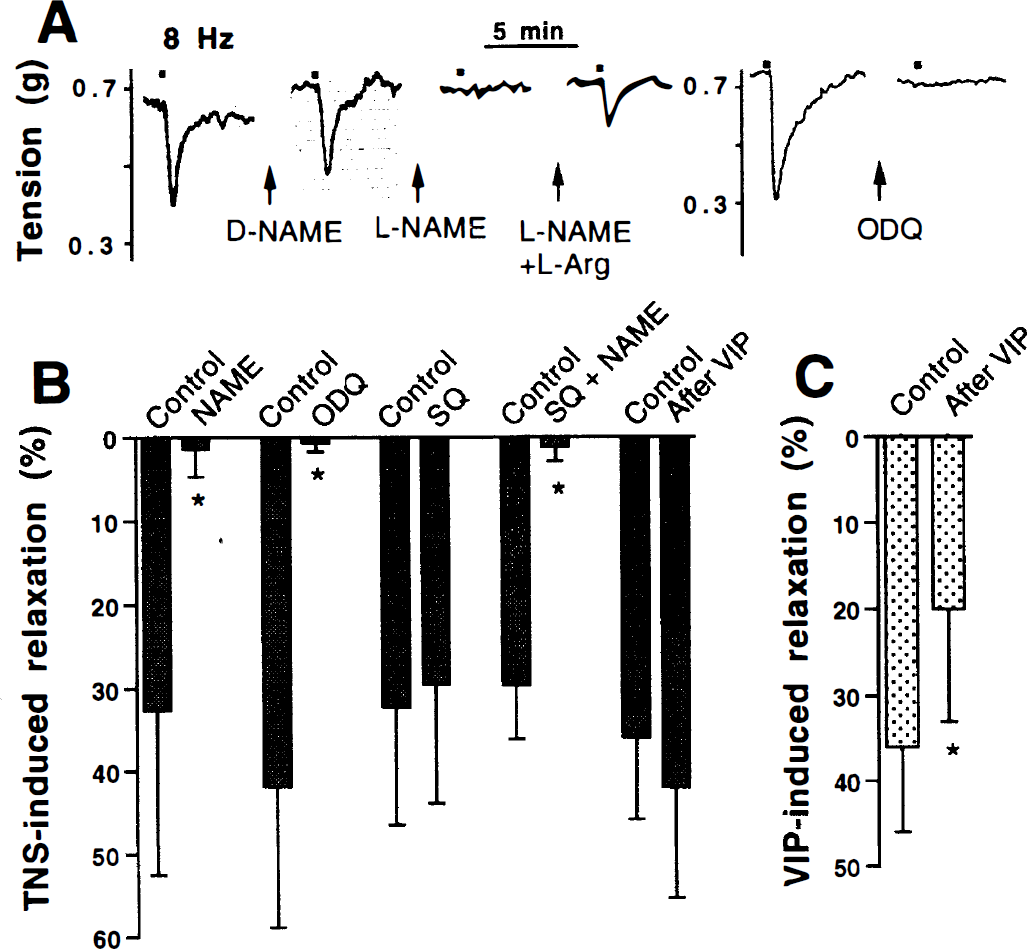
The intracellular concentrations of the cyclic nucleotides cGMP and cAMP were measured before and during TNS-induced relaxation. In agreement with the functional results described earlier, significant increases of cGMP concentration were elicited by TNS (Fig. 3A), whereas a concomitant small decrease in cAMP concentration was produced (Fig. 3B). The TNS-induced increment in cGMP concentration was caused by guanylyl cyclase activation by NO because it was abolished in the presence of
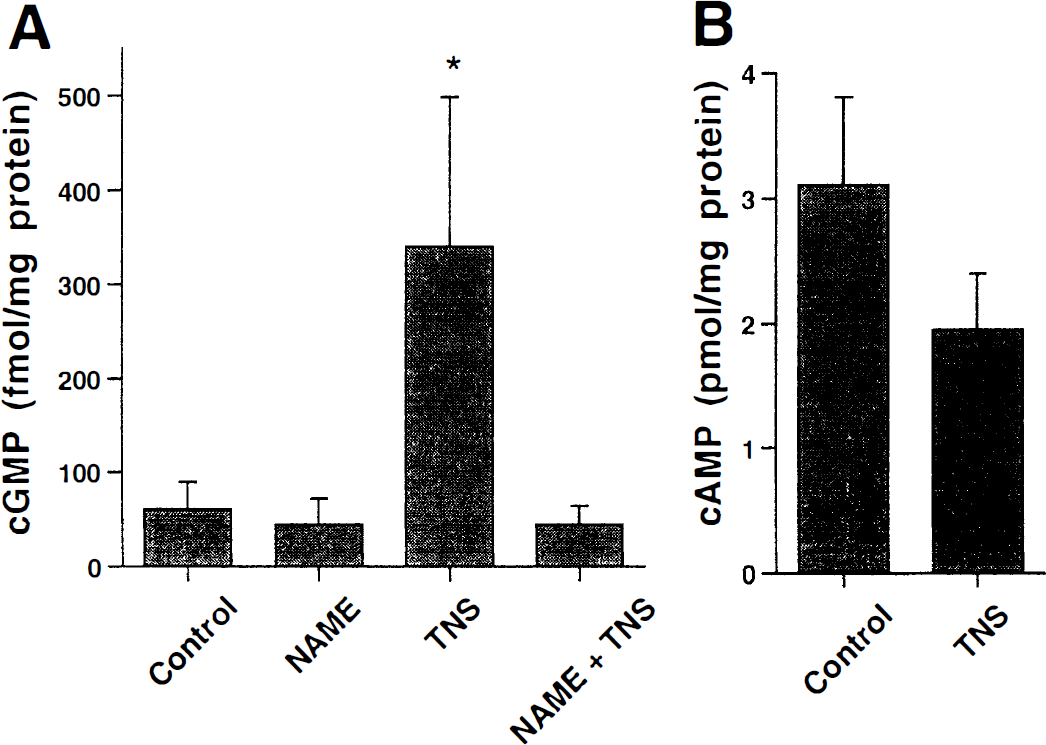
To analyze the signal transduction mechanisms of VIP action in bovine cerebral arteries, concentration–response curves were obtained when the NO/cGMP pathway was blocked by either
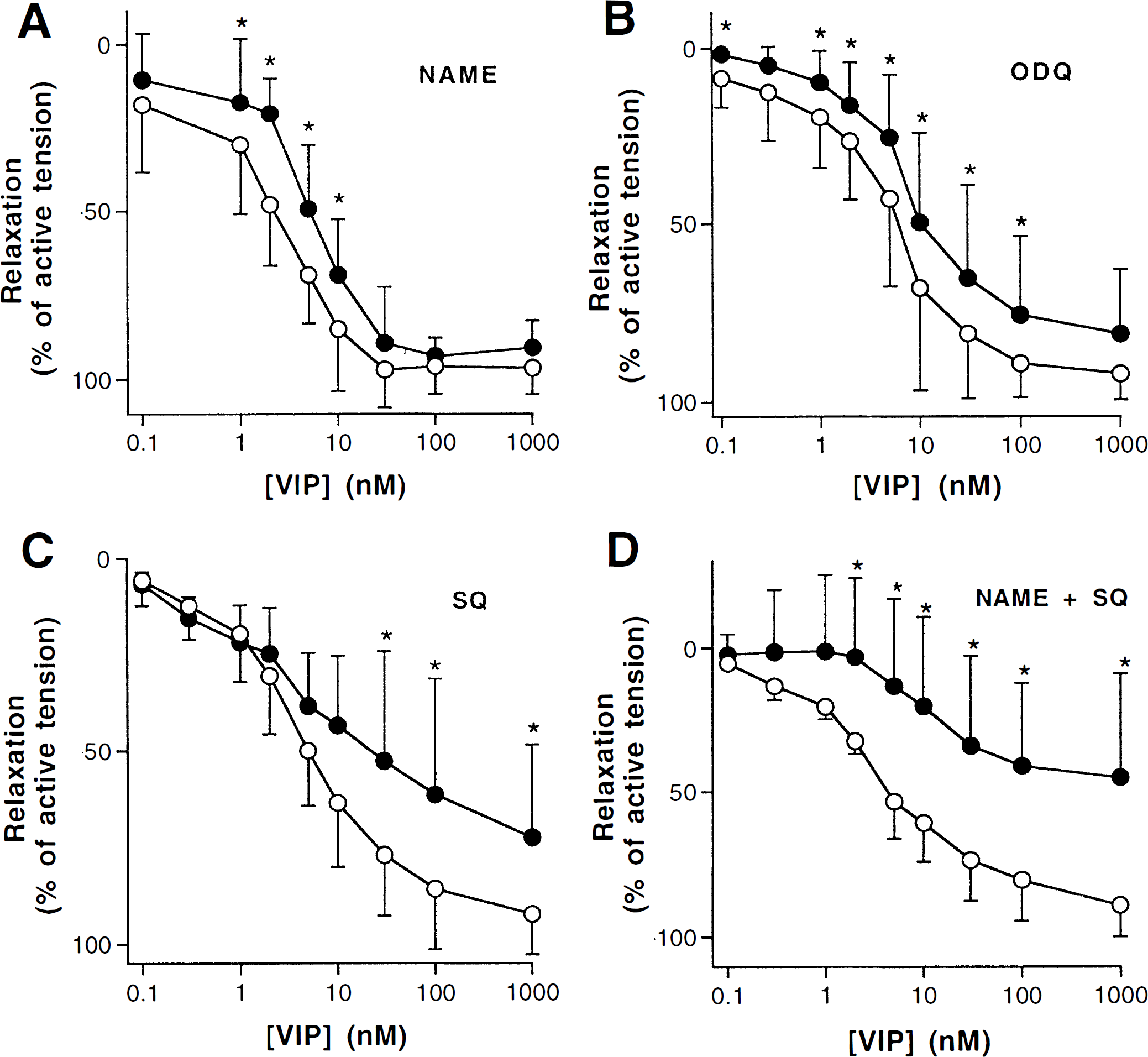
Relaxation induced by cumulative concentrations of VIP in bovine cerebral arteries without endothelium in the absence (open circles) and in the presence (filled circles) of 100 μmol/L
EC50 and Emax values for VIP-induced relaxation in bovine cerebral arteries in the absence and presence of L-NAME, SQ 22,536 or both
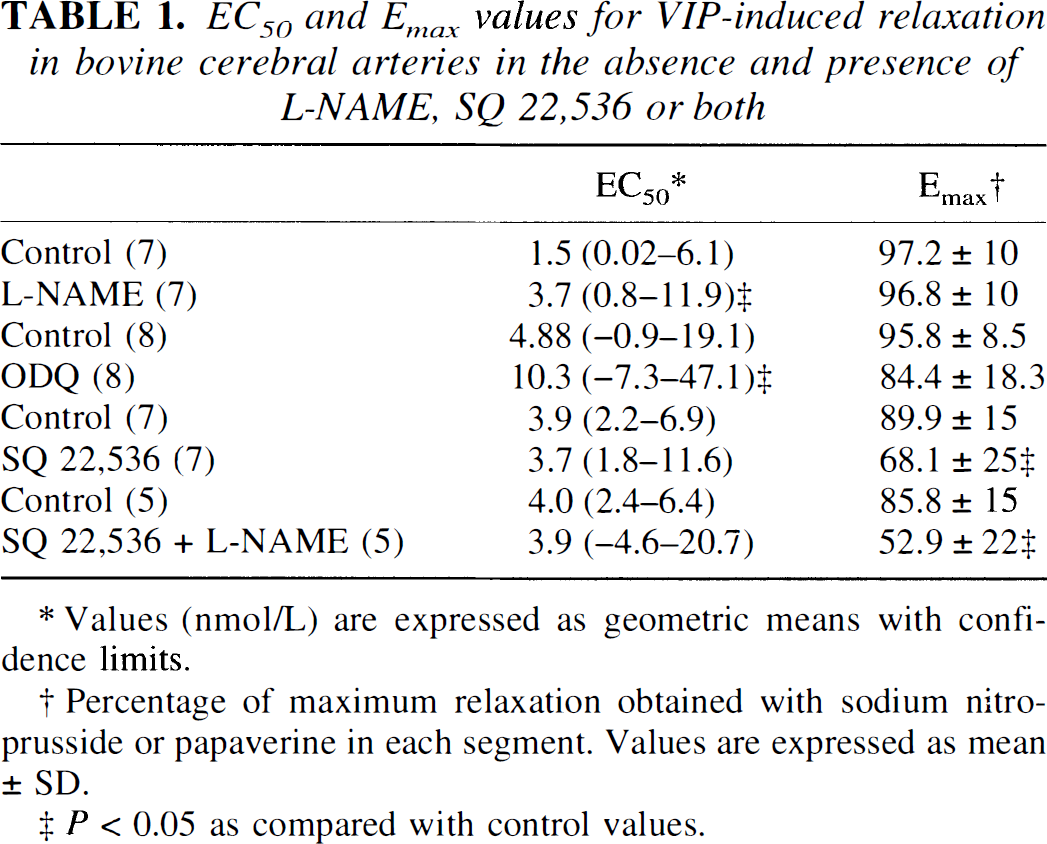
Values (nmol/L) are expressed as geometric means with confidence limits.
Percentage of maximum relaxation obtained with sodium nitroprusside or papaverine in each segment. Values are expressed as mean ± SD.
P < 0.05 as compared with control values.
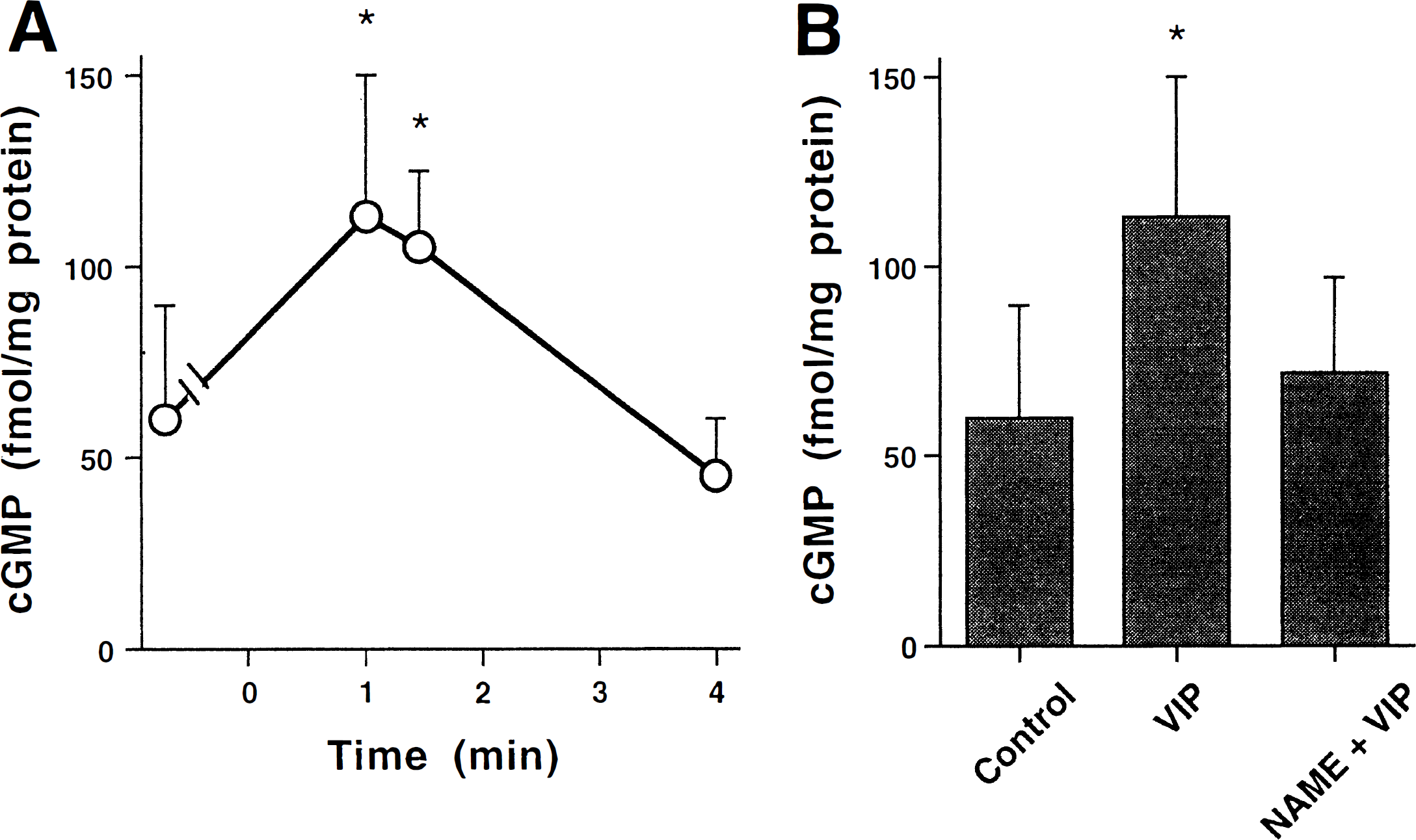
To assess further the participation of the NO–cGMP pathway in the relaxation induced by VIP in bovine cerebral arteries, cGMP levels were measured at different times after addition of the peptide. Cyclic GMP increased 1 minute after addition of VIP to the organ bath, remained elevated for 2 minutes, and returned to control levels 4 minutes after addition of the peptide (Fig. 5A). In the presence of
DISCUSSION
We investigated the involvement of NO and VIP in the regulation of bovine cerebral artery function by perivascular nerves. Our results suggest that there is an interplay between these two messengers, which are contained in the same nerve population, with NO being the peripheral neurotransmitter directly relaxing smooth muscle cells and VIP acting as an autocrine neuromodulator.
The presence of perivascular nitrergic nerves, shown here by NADPH-diaphorase staining, used as a marker for NOS (Hope et al., 1991; Dawson et al., 1991), and by NOS immunohistochemical study, is in agreement with previous results in the cerebral arteries of rats, cats, guinea-pigs, and humans (Barroso et al., 1996; Bredt et al., 1990, Estrada et al., 1993, Nozaki et al., 1993). However, it is interesting to notice the detection in bovine vessels of stained neuronal cell bodies grouped in ganglion-like structures, with morphologic features similar to those previously described in cats (Estrada et al., 1993; Lee, 1994). Although nitrergic neurons in the sphenopalatine ganglion have been shown to project to cerebral arteries in the rat (Nozaki et al., 1993; Minami et al., 1994; Suzuki et al., 1994), the presence of intrinsic NOS neurons in the cerebral vessel wall in cats and cows suggests that some of the nitrergic nerves have a local origin, at least in these species.
The coexistence of NOS and VIP demonstrated here for bovine cerebral arteries seems to be a common feature of NANC nerves in the peripheral nervous system. Partial or total co-localization of these two transmitters has been reported in different locations, including the gastrointestinal tract of several species (Ekblad et al., 1994), the canine corpus cavernosum (Hayashida et al., 1996), rat nasal mucosa (Takahashi et al., 1995), guinea-pig heart (Klimaschewski et al., 1994), and cerebral blood vessels (Nozaki et al., 1993; Barroso et al., 1996). This frequent co-localization of VIP and NOS in peripheral nerve terminals suggests a possible functional interaction between these two relaxing pathways.
Our results show that VIP-mediated activation of adenylyl cyclase did not occur during neurogenic stimulation of bovine cerebral blood vessels, because of the following: (1) there was no intracellular accumulation of cAMP, (2) the relaxation was not impaired by inhibition of adenylyl cyclase with SQ 22,536, and (3) VIP-induced tachyphylaxis did not modify the vascular relaxation to TNS. On the contrary, the guanylyl cyclase pathway must have been activated, since (1) significant increases in intracellular cGMP concentrations were detected during TNS, and (2) the relaxing response to TNS was completely blocked by the adenylyl cyclase inhibitor ODQ. The disappearance of any significant relaxation or cGMP accumulation in the presence of the NOS inhibitor
One interesting finding is that cGMP levels were low in endothelium-denuded arteries and that they were not further decreased by
Although VIP does not behave as a neurotransmitter in bovine cerebral vessels, the possibility exists that it acts as a neuromodulator by modifying the release of the coexisting neurotransmitter, as is the case with other neuropeptides. We show here that VIP causes relaxation of bovine cerebral arteries by two independent pathways. First, and in accordance with the well known interaction of VIP with adenylyl cyclase–coupled receptors (Huang and Rorstad, 1984; Suzuki et al., 1985), VIP relaxes the vessel by adenylyl cyclase activation, since the relaxation decreased in the presence of an inhibitor of this enzyme. Second, it activates the NO–cGMP pathway, as demonstrated by the increase in cGMP concentration and the partial inhibition of the VIP-induced relaxation by
Shortly before the identification of perivascular nitrergic plexi in the cerebral vasculature, Gaw and others (1991) described a decrease in the relaxation produced by VIP in sheep cerebral arteries on incubation with hemoglobin or the NOS inhibitor NG-monomethyl-
