Abstract
Two populations of scattered neurons containing nitric oxide synthase activity were detected in the wall of the third and lateral cerebral ventricles of rat brain, using histochemistry for NADPH-diaphorase activity. One type was multipolar and lay supraependymally, with dendrites oriented in the plane of the ependymal layer. The second type was bipolar and was situated subependymally, with dendrites extending in opposite directions, either into the surrounding brain tissue or to the ventricular surface. Moreover, multipolar neurons, situated in the corpus callosum and in the subcortical white matter, had long varicose dendrites extending toward the roof of the lateral ventricles. As a result, numerous NADPH-diaphorase neurites spread out on the free surface of the ependymal layer in contact with the CSF. These observations raise the possibility that periventricular nitrergic neurons play an essential role in registering the composition of the CSF and in modulating subcortical cerebral blood flow. A further possibility is that supraependymal nitrergic neuronal processes are effectors regulating activity of ependymal cells.
Keywords
Cerebrospinal fluid-contacting neurons extend their processes on the wall of the ventricles in the brain (Vigh and Vigh-Teichmann, 1971, 1973). These neurons appear in large numbers in the wall of the third ventricle, arising from the periventricular hypothalamic nuclei, and may be responsible for a presumptive sensory function in this site (Vigh and Vigh-Teichmann, 1973). Moreover, intraventricular nerve endings have been observed in all cerebral ventricles and in the area around the central canal of the spinal cord and medulla oblongata (reviewed by Westergaard, 1972). The origin of these nerve fibers, the types of transmitter(s) employed, and their functions are unclear (Richards et al., 1973). Location at the interface between CSF and the brain makes such nerve endings either potential effectors regulating ependymal cell activity or sensory targets for signaling the CSF composition (Westergaard, 1972).
By the NADPH-diaphorase histochemical technique (Thomas and Pearse, 1964; Scherer-Singler et al., 1983), which gives a Golgi-like staining of specific neuronal bodies, dendritic tree, and nerve fibers, we serendipitously found the presence of a network of NADPH-diaphorase nerve endings on the ependymal surface of the third and lateral ventricles, as well as of sparse NADPH-diaphorase-positive neurons situated supraependymally.
NADPH-diaphorase activity colocalizes with nitric oxide synthase activity (NOS) in a class of so-called nitrergic neurons that uses nitric oxide (NO) as neurotransmitter (Bredt et al., 1991; Dawson et al., 1991; Hope et al., 1991). Several previous studies have reported the presence of NADPH-diaphorase-positive neurons in particular regions of the CNS of various species (Leigh et al., 1990; Vincent and Kimura, 1992). Similar cells have been noted in the subcortical white matter (interstitial neurons; Sandell, 1986; Mizukawa et al., 1988), in the corpus callosum (Thomas and Pearse, 1964) or lying just beneath the ependymal layer in the diencephalon (Vincent and Kimura, 1992). Interstitial neurons, scattered in the adult subcortical white matter and corpus callosum, are considered remnants of the large number of neurons present during fetal and early postnatal life within the subplate (future white matter; Chun and Shatz, 1989). These neurons seem to have no obvious relevance to the adult cerebral cortical circuitry and function (Chun and Shatz, 1989).
The presence of nitrergic neurons in the third and lateral ventricles, at the inner surface of the brain in contact with the CSF, may be related to specialized functions at this site. We examined the distribution, morphologic features, and possible functional implications of NADPH-diaphorase neurons extending their cell body or cell processes or both to the wall of the cerebral ventricles in the rat brain.
ANIMALS AND METHODS
For this study, 12 adult Wistar rats of either sex weighing 200–250 g were maintained on a 12 h light/12 h dark cycle and with free access to food and water. All chemicals were obtained from Sigma (Milan, Italy).
The animals were deeply anesthetized with thiopental sodium and transcardially perfusion-fixed with 4% paraformaldehyde in 0.1 M phosphate buffer (PB; pH 7.4), following a brief rinse with physiological saline and heparin (1%) at room temperature. The brains were removed from the skull and postfixed in the same fixative for 2 h at 4°C and then stored in PB overnight.
The brains were cut with a vibratome into coronal slices 40 μm thick, including the forebrain and brainstem, and collected in PB containing 0.02% sodium azide. Semiserial sections (one of five) were used to study qualitatively the distribution of NADPH-diaphorase neurons and nerve fibers in the brain ventricles. In addition, serial sections 40 μm thick were collected from the brain, including the lateral ventricles from the frontal to the tem-porooccipital pole of one animal, to count the number and evaluate morphometric characteristics of NADPH-diaphorase neurons contacting the CSF.
Free-floating brain slices were incubated in a solution containing 0.1 MTris-HCl (pH 8.0), 1 mM β-NADPH, 0.2 mM nitro blue tetrazolium (NBT), 0.2% Triton X-100, for 60–90 min at 37°C. For a negative control test, NADPH was omitted from the incubation medium. After the histochemical reaction, the sections were rinsed and counterstained with cresyl violet to visualize the ependymal cell layer and to evaluate better the topographic relation of NADPH-diaphorase neuronal bodies and nerve endings to the ventricular surface. The sections were mounted on gelatine-coated slides, dehydrated, and coverslipped with permount for light microscopic examination.
NADPH-diaphorase neurons were considered contacting the CSF when the cell body or its processes or both were lying on the ependymal surface facing the CSF. To evaluate the morphometric characteristics of NADPH-diaphorase neurons contacting the CSF, these neurons were counted in the brain of one animal, on 135 coronal serial sections (40 μm thick; levels from bregma = + 1.70 mm to −4.30 mm, according to the Paxinos and Watson atlas, 1986), using a microscopic magnification of ×200. Morphometric evaluation of such neurons was performed by means of a quantitative image analyzer, Quantimet 500-Leica. The data were expressed as the cumulative number of all positive neurons counted in both lateral ventricles along their frontotemporal parts or in the third ventricle.
RESULTS
NADPH-diaphorase-positive neurons of strong staining intensity were present throughout various regions of the brain, confirming previous descriptions (Thomas and Pearse, 1964; Leigh et al., 1990; Vincent and Kimura, 1992). Moreover, a number of medium-sized NADPH-diaphorase neurons (Table 1) spread on the ventricular surface (supraependymal neurons) or lay just beneath the ependymal layer (subependymal neurons) and extended their processes supraependymally (Fig. 1A and B). Supra- and subependymal (juxtaependymal) neurons could be observed to better advantage in 40-μm thick coronal slices in which the ventricular surface appeared as a strip inclined toward the plane of the slide (Fig. 1A and B). In addition, intensely stained NADPH-diaphorase-positive neurons were seen in the corpus callosum or in the subcortical white matter (Fig. 1C and D). These so-called interstitial neurons showed linear long neurites extended for several hundred micrometers either parallel to the callosal fibers (Fig. ID) or upward to the cerebral cortex and downward to the roof of the nearer lateral ventricle (Fig. 1C). As a result, a sparse delicate meshwork extended under and on the ependymal layer, consisting of thin and thick varicose processes (Fig. 2A), which were at times in apparent continuity with nearby NADPH-diaphorase neuronal bodies (Fig. 1).
Distribution and morphometric characteristics of NADPH-diaphorase neurons contacting the CSF in the rat cerebral ventricles
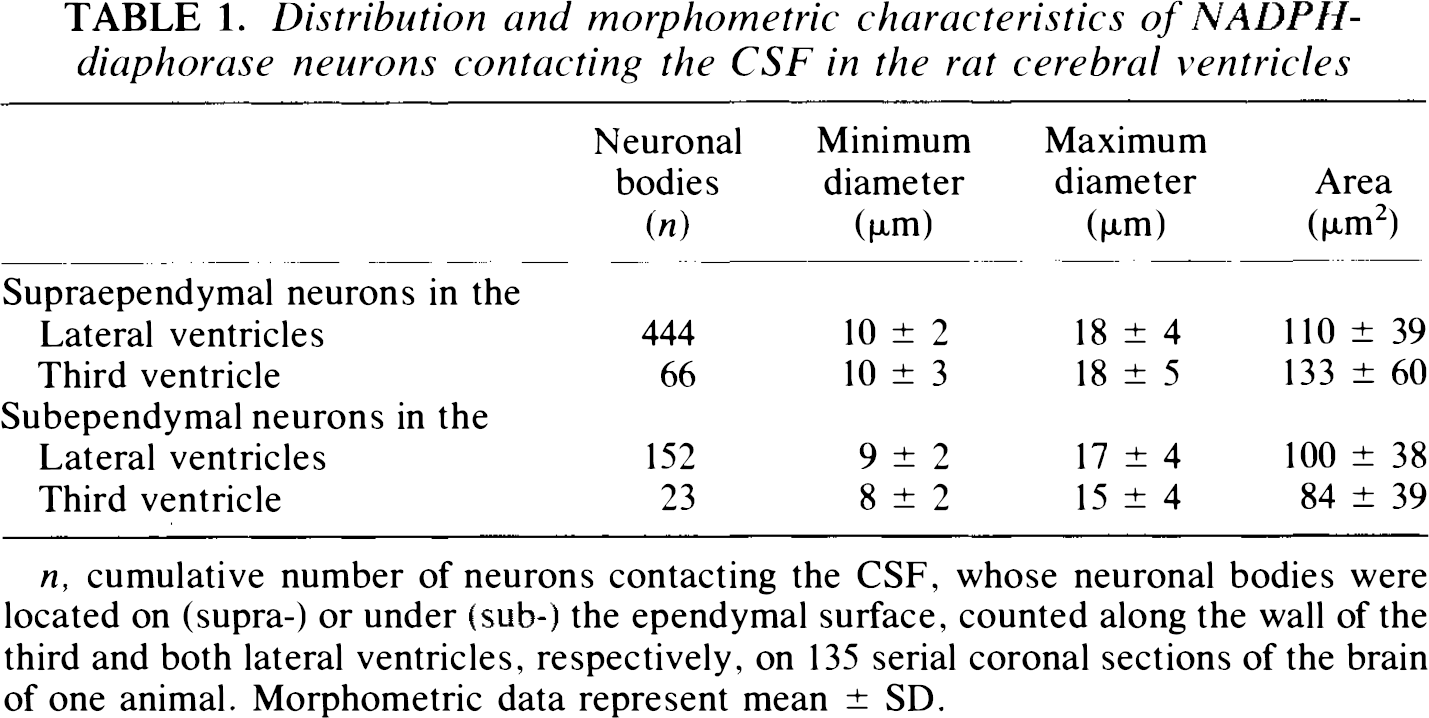
n, cumulative number of neurons contacting the CSF, whose neuronal bodies were located on (supra-) or under (sub-) the ependymal surface, counted along the wall of the third and both lateral ventricles, respectively, on 135 serial coronal sections of the brain of one animal. Morphometric data represent mean ± SD.
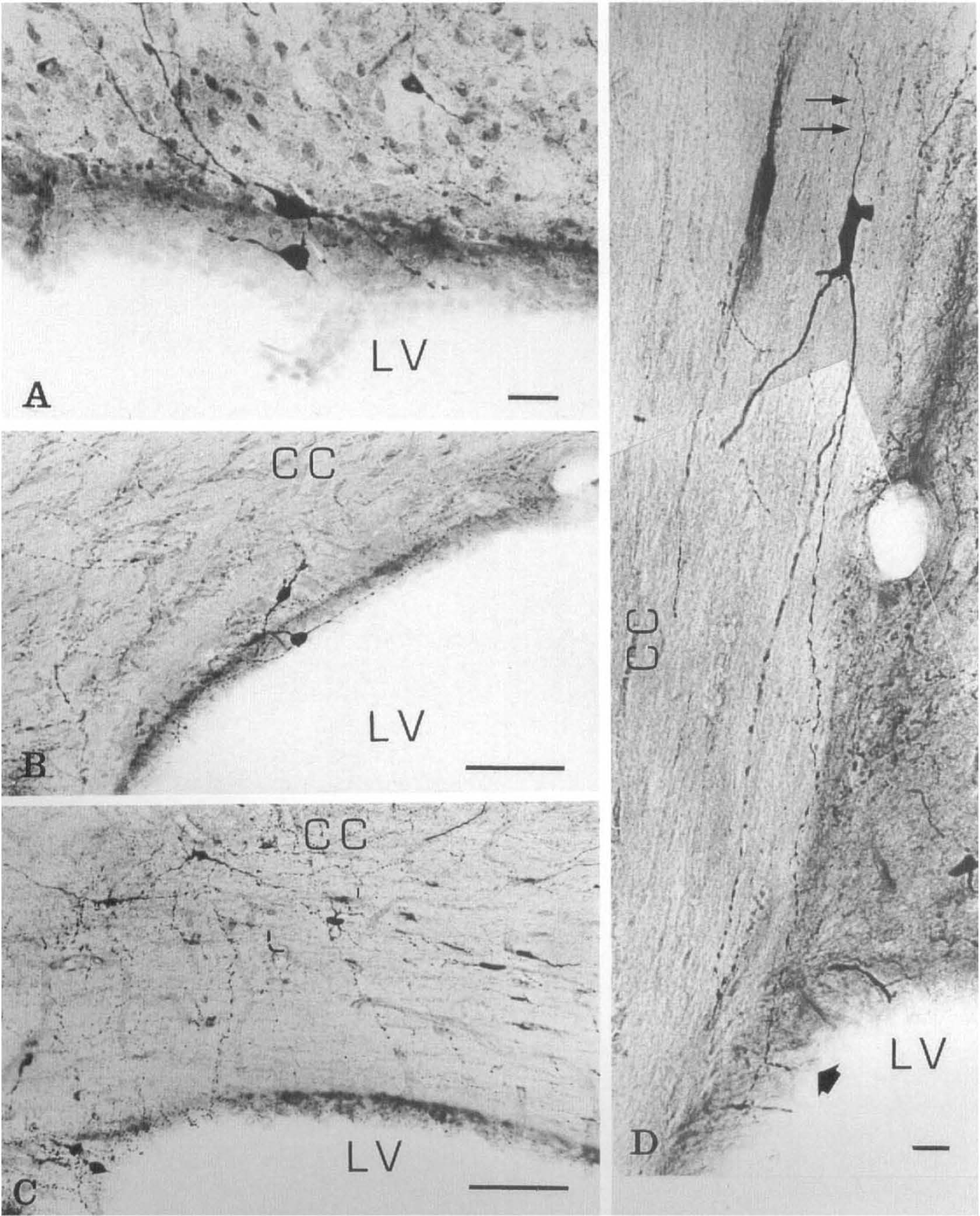
Sections through the frontal horn of the lateral ventricle of rat brain. NADPH-diaphorase staining followed by counter-stain with cresyl violet.
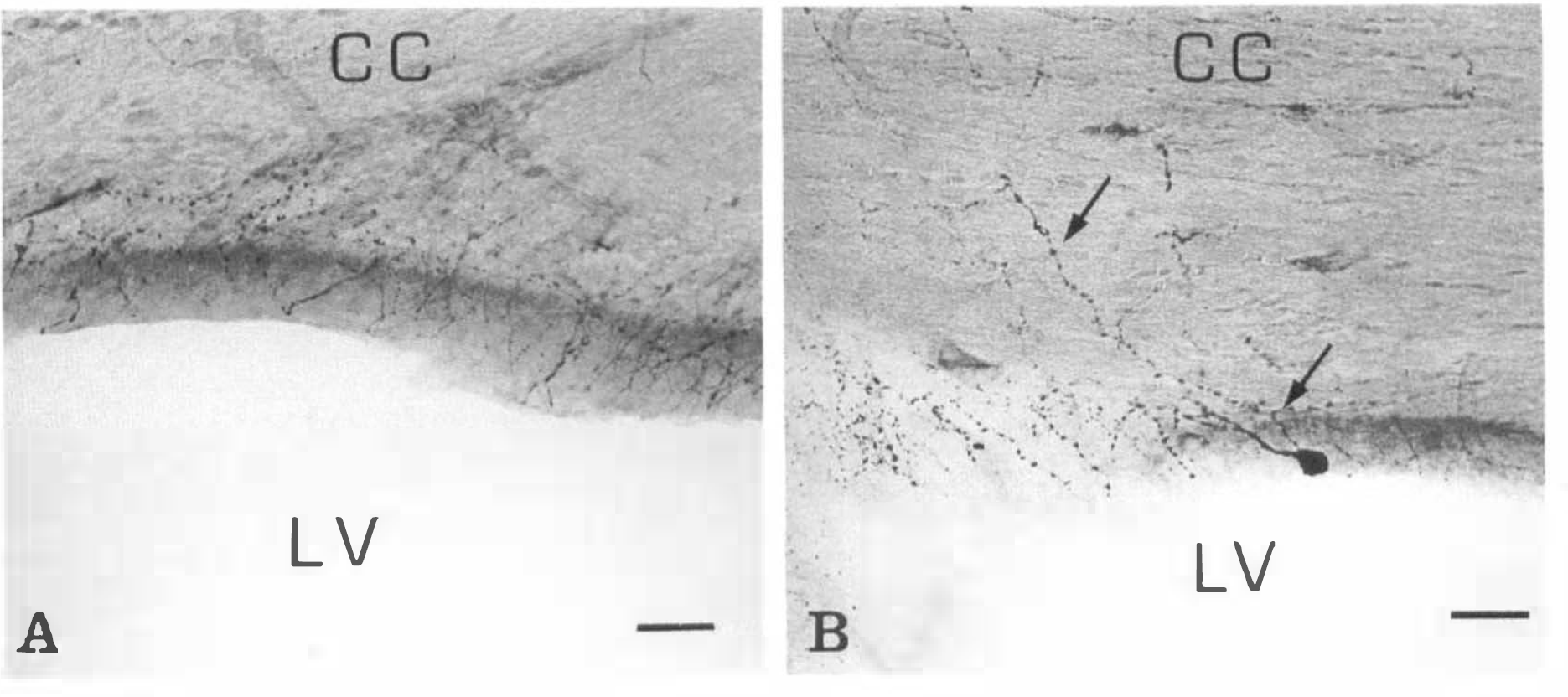
Sections through the frontal horn of the lateral ventricle of rat brain. NADPH-diaphorase staining followed by counter-stain with cresyl violet.
Generally, supraependymal neurons were of the multipolar type, and the dendrites were oriented in the plane of the ependymal layer. Subependymal and interstitial neurons were of the bipolar or multipolar type, having neurites oppositely oriented either toward the ventricular surface or into the surrounding brain tissue. A characteristic feature of these cells was thin, relatively straight dendrites, displaying regular ovoid distention along their course (Fig. 1B and D). This feature was critical to distinguishing between dendrites and axons. Occasionally, a thin neurite took origin from the cell body and was uniform in caliber along its observable course. Such axon-like processes, derived from juxtaependymal and interstitial neuronal bodies, were directed toward the surrounding brain tissue (Figs. 1D and 2B).
Between one and five NADPH-diaphorase neurons contacting the CSF were counted on each 40-μm thick coronal section. They appeared solitary or in couples and were scattered around the ventricular wall at all levels along the frontotemporal parts. Not all ventricular linings contained such neurons. Juxtaependymal neurons were located on the ventricular surface covering the corpus callosum, the caudate-putamen, and the lateral septal nucleus, whereas they were absent on the ventricular surface covering the septofimbrial nucleus and the fimbria hippocampi. Interstitial neurons contacting the CSF were evident in the white matter of the corpus callosum and of the external capsule surrounding the frontal and temporal parts of the lateral ventricles.
In the third ventricle, there were two distinctive types of NADPH-diaphorase neurons contacting the CSF: either these appeared medium-sized (Table 1) and scattered around the ventricular wall, or they were large neurons grouped in clusters beneath the ependyma. The former have morphologic characteristics (Table 1) and distribution patterns like those in the lateral ventricles, so that it is conceivable that such neurons play a unitary role in the different ventricles. Even though some of the NADPH-diaphorase neurons grouped in clusters in the third ventricle appeared to be in contact with the CSF, these were not further objects of our study, because they morphologically pertain to the periventricular hypothalamic nuclei, and their role may be limited to the maintenance of specific hypothalamic functions.
NADPH-diaphorase neurons contacting the CSF were not observed in the cerebral aqueduct or in the fourth ventricle.
Morphometry data are summarized in Table 1. NADPH-diaphorase neurons contacting the CSF displayed similar characteristics in the third and in the lateral ventricles. Supraependymal neurons were just slightly larger than subependymal neurons. The interstitial NADPH-diaphorase neurons were not counted, as it was not easy in each case to outline the continuity between neuronal body located in the white matter and neurites reaching the ventricular wall.
DISCUSSION
We have demonstrated that a particular population of nitrergic neurons, detected by NADPH-diaphorase staining, is localized in or extends its processes to the wall of the third and lateral ventricles of rat brain. This observation raises the possibility that juxtaependymal and interstitial nitrergic neurons may have common specialized functions, depending on their contact with ependymal cells and exposure to the CSF bathing.
Ependymal cells may be under the influence of NO from intraventricular nerve endings. A mesh-work of nitrergic/NADPH-diaphorase neurites runs parallel to and comes in contact with the free surface of the ependymal cells. The fact that similar nitrergic neuronal processes have not been detected in the cerebral aqueduct and in the fourth ventricle may indicate regional specialized functions of nitrergic innervation, as well as of the ependymal layer (Balercia et al., 1992). We cannot determine the structural characteristics and the types of contacts between nitrergic neuronal processes and ependymal cells. Previous electron microscopic investigations revealed that intraventricular nerve endings come close to the ependymal cell plasma membrane and sometimes form junctions like zonulae adherens (Westergaard, 1972). Thus it is conceivable that ependymal cells are under the diffusion-limited domain of NO derived from adjacent nitrergic neurites. It has been found that the secretory functions of epithelial cells and epithelial ciliary motility appear to be prominent NO targets in the lung and gastrointestinal mucosa (reviewed by Schmidt and Walter, 1994). By analogy, we could hypothesize that NO regulates ependymal cell functions, such as movements of ependymal cilia, permeability to transependymal passage of CSF, and ependymal secretion of substances in the CSF (Bruni et al., 1985; Peters et al., 1991).
Nitrergic neurons contacting the CSF may have a sensory function. On the basis of a number of morphologic and pharmacologic studies, it has been suggested that intraventricular nerve endings register both physical and chemical changes in the CSF (Westergaard, 1972; Richards et al., 1973). We could speculate on functions of nitrergic neurons contacting the CSF on the basis of their morphologic characteristics and their topographic distribution. As a general rule, orientation of neurons with respect to a given structure relates to their function (Peters et al., 1991). It is worthy of note that supraependymal nitrergic neurons are parallel to the ventricular wall and that the course of the dendritic tree of subependymal and interstitial neurons is pointed, at least in part, toward the free surface of the ependymal cells. Although axons of the juxtaependymal nitrergic neurons are seldom identifiable with certainty in the light microscope, they are invariably seen oriented toward surrounding brain tissue. This morphologic polarization suggests that nitrergic neurons contacting the CSF are primarily concerned with a receptive function. Such a role would be in agreement with the fact that NO has been implicated in the primary transduction processes of several sensory modalities, including adaptive regulation of visual and olfactory signaling and possibly pain (reviewed by Schmidt and Walter, 1994).
If the nitrergic neurons contacting the CSF are involved in a receptive function, important questions to settle will be which CSF specific changes they respond to and which homeostatic responses in the brain are due to such a stimulation. The CSF, secreted mainly by the choroid plexuses, contributes to the homeostasis of brain parenchyma and is in equilibrium with the extracellular fluid. Owing to the brain metabolism, the Pco2 is higher (>7–9 mm Hg) and consequently the pH is lower (<0.1 unit) in the CSF than in venous blood (Schuller, 1993). Recent studies indicate that neuronal activity gives rise to significant changes in intracellular and extracellular pH (Chesler and Kaila, 1992), and that neural activity is one of the main factors regulating regional cerebral blood flow (Ginsberg et al., 1987; Sokoloff, 1989). Furthermore, there is evidence that little changes in the pH level or in electrolytic composition of the CSF influence the cerebral blood flow (reviewed by Heuser, 1978; Rabary et al., 1994). More recently, a current hypothesis assumes that neural NO, a potent vasodilator produced by specific neurons, participates in the increases in cerebral blood flow elicited by neural activation (Iadecola, 1993). With these considerations in mind, we speculate that a physiological or pathological rise in H+ and CO2 concentration of the CSF could stimulate nitrergic neurons contacting the CSF and that such neurons could be involved in regulation of blood flow in periventricular cerebral structures.
Anatomic evidence supports this suggestion. Nitrergic neurons contacting the CSF on one side extend their processes also toward the contiguous periventricular structures such as the striatum, the septal nucleus, the corpus callosum, and the thalamus. Moreover, it is known that NADPH-diaphorase neurons contained in the white matter (interstitial neurons) give rise to association fibers that project mostly into the cortical gray matter (Meyer et al., 1991). Thus it is conceivable that nitrergic neurons may play a role in the maintenance of local circuitry and function in the wall of the third and lateral ventricles. Alternatively, nitrergic neurons may form a periventricular network contacting the CSF on one side and having connection with the periventricular nuclei and the subcortical white matter on the other. Although such neurons are few and scattered, they could have extensive connections.
In conclusion, we have demonstrated the presence of nitrergic neuronal processes on the ependymal surface of the third and lateral ventricles. It should be clarified whether nitrergic neurons have relevance to functions of ependymal cells and for signaling of the CSF composition.
Footnotes
Acknowledgment:
We thank Dr. Michelangelo Iannone (University of Catanzaro) for helpful discussion. This work was supported by Consiglio Nazionale delle Ricerche, grant n. 9202783CT04, and by IRCCS-Clinica S. Lucia.
